 Open Access Article
Open Access ArticleUltraviolet photoeffects on oxygen–hydrogen interstitial clusters in rutile TiO2
Heonjae Jeong†
 b,
Ian I. Sunia,
Raylin Chen
b,
Ian I. Sunia,
Raylin Chen a,
Grace McKnightb,
Elif Ertekin
a,
Grace McKnightb,
Elif Ertekin b,
Xiao Su
b,
Xiao Su a and
Edmund G. Seebauer
a and
Edmund G. Seebauer *a
*a
aDepartment of Chemical and Biomolecular Engineering, University of Illinois at Urbana-Champaign, Urbana, Illinois 61801, USA. E-mail: eseebaue@illinois.edu
bDepartment of Mechanical Science and Engineering, University of Illinois at Urbana-Champaign, Urbana, Illinois 61801, USA
First published on 9th March 2026
Abstract
Super-bandgap illumination of semiconductors affects the diffusion and reaction rates of interstitial atoms, but photoeffects on clusters of interstitials remain virtually unexplored. In prototypical metal oxides such as TiO2 and ZnO, oxygen interstitials (Oi) appear to form stable clusters below about 300 °C. Such cluster formation becomes very important when chemically prepared surfaces of binary oxides submerged in liquid water inject Oi into the solid. New kinetically dominated phenomena occur, such as strong isotopic fractionation, that are influenced by interstitial trapping in clusters. Judicious coordination of temperature and illumination allows optimization of competing kinetic effects, but little is known about the composition of Oi-containing clusters or their photoresponse. This study begins to fill that gap by combining simulations of Oi–Hi clusters by density functional theory (DFT) with self-diffusion measurements of 18O in submerged single-crystal rutile TiO2(110) under ultraviolet (UV) illumination. The simulations show that Oi–(Hi)x exists in several isomers, each with multiple charge states that can change upon illumination. The diffusion measurements suggest that UV changes the populations of Oi-containing cluster isomers deep in the solid, and that the details of these changes depend upon the application of potential bias. The results indicate that illumination alters the rate constants for formation or dissociation of (Oi)y–(Hi)x or alters the concentration of the reactant Hi.
1. Introduction
Super-bandgap illumination of semiconductors has long attracted interest for altering equilibria among point defects1–3 and affecting their rates of diffusion4,5 and reaction.6,7 Photogenerated minority carriers change the defects’ charge state or recombine at defects to convert electronic to lattice vibrational energy.8 Lower temperatures accentuate such effects by decreasing the rate of thermal carrier generation. Illumination and temperature can then work together to affect semiconductor properties,9,10 or induce “thermo-photocatalysis.”11In prototypical metal oxides such as TiO2 and ZnO, recent experimental evidence suggests that oxygen interstitials (Oi) can react to form clusters that are stable below about 300 °C.12 Such clustering can neutralize unwanted donors, especially adventitious hydrogen13 that is common in oxides,14–18 but also restricts movement of Oi and thereby inhibits removal of potentially unwanted O vacancies (VO).19 These effects take on increased importance because of the recent discovery that chemically prepared surfaces of binary oxides submerged in liquid water inject Oi efficiently.13 This injection provides an effectual pathway for Oi to dominate low-temperature point defect populations.13,20,21 Submersion temperatures below 100 °C facilitate kinetically dominated phenomena like strong isotopic fractionation.22,23 Under such conditions, judicious coordination of temperature and illumination permits optimization among competing kinetic effects. However, apart from the special case of UO2,24,25 little is known about the composition of Oi-containing clusters in metal oxides or their response to photostimulation. In TiO2 and ZnO, the preponderance of such clustering appears to involve interstitial hydrogen (Hi).12
This work begins to fill these gaps by combining simulations of Oi–Hi clusters using density functional theory (DFT) with self-diffusion measurements of 18O in submerged single-crystal rutile TiO2(110) illuminated by ultraviolet (UV) light. The DFT simulations show that Oi–(Hi)x dimers and trimers exist in several isomers, and each has multiple charge states that can change upon illumination. The diffusion measurements indicate that UV changes the populations of Oi-containing cluster isomers deep in the solid, and that the details of these changes depend upon the application of potential bias. The results suggest that illumination alters the rate constants for formation or dissociation of (Oi)y–(Hi)x or changes the concentration of the reactant Hi.
2. Methods
2.1 Experiments
Experimental literature concerning illumination-stimulated behavior of defects has typically employed measurements of electrical conductivity,26 dopant compensation,27–29 solar cell lifetime,9,30 or oxygen incorporation rate.10,31,32 The present study measures depth profiles of 18O injected by TiO2(110) exposed to isotopically enriched water. Isotopic fractionation occurs in this system, resulting in profile metrics that respond to Oi trapping effects in distinct ways. Such differences enable disaggregation of photostimulation effects on trapping vs. other physical processes such as injection or site hopping.Protocols employed procedures for specimen preparation and diffusion under illumination described elsewhere.13,23,33 Single-crystal specimens of TiO2(110) (5 × 5 × 0.5 mm, MTI Corp.) were submerged in 18O-labeled water (10 atomic % 18O, Sigma-Aldrich) at constant temperatures (T) ranging from 30–80 °C. The diffusion time was 60 min except as noted below. Crystals were de-greased by 10 min of ultrasonic agitation in acetone, isopropanol, ethanol, and methanol, followed by wet etching (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 30% NH4OH
2, 30% NH4OH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O) for 40 min at room temperature to remove elemental poisons that inhibit Oi injection.
H2O) for 40 min at room temperature to remove elemental poisons that inhibit Oi injection.
Diffusion under submersion was studied with UV illumination using a protocol identical to that described previously.33 Light originated from an inspection lamp (Black-Ray™ B-100AP/R, UVP) operating with a central wavelength of 365 nm. At 25 cm, the lamp produced an intensity on the order of 5 mW cm−2 at the surface. The intensity was kept low to minimize possible heating by light absorption. Evidence that such heating was indeed negligible has been presented elsewhere.33
Application of potential bias in a conventional three-electrode cell configuration aids the measurement of self-diffusion metrics by increasing their magnitudes.23 Accordingly, some TiO2 specimens were immersed for 60 min at constant T with a potential bias applied using a Biologic SP200 potentiostat. The electrical connection to the TiO2 specimens was a Cu wire attached with double-stick carbon tape covered with Kapton. An Ag/AgCl reference electrode and a Pt counter electrode were employed. Before immersion, 10 min of air bubbling through the aqueous solution with simultaneous air flow through the headspace established liquid–gas equilibrium.
The undoped TiO2 was electrically insulating and no electrolyte was employed, so no faradaic electrochemistry occurred. Measured currents were small (10–100 nA) and did not correlate with any profile metric. Measurements of pH before and after each experiment hovered near 7 and changed 0.25 pH unit or less. Diffusion metrics did not change with the sign or magnitude of the applied potential. Unfortunately, the mechanism by which applied bias increases the injection flux remains unknown at this point. Applying a bias may unintentionally remove a contaminant that poisons injection sites. Other possible hypotheses include that enhanced electric fields or some form of electrostatic electrochemistry35,36 could play a role. Investigation of these effects will be the subject of future work.
Surface analysis by X-ray photoelectron spectroscopy (XPS) of specimens treated before diffusion by vacuum annealing, NH4OH etching or degreasing have been published previously.34 The spectra showed primarily Ti, O, and C. Surface elemental composition and chemical state varied but did not correlate with experimental conditions. Here we supplemented those studies by measuring XPS spectra on select specimens before and after diffusion with applied bias. Spectra were measured with a Kratos Axis ULTRA instrument using a monochromatic Al Kα source (1486.6 eV). Energies were calibrated based on the C1s peak (284.6 eV) of adventitious carbon. Elemental compositions and chemical state were determined using the CasaXPS software library. The results (Fig. S1 and S2 of the SI) for elemental composition and chemical state of Ti, O and C again show variability but no discernable correlation with experimental conditions.
The surface morphology and roughness were measured for some specimens immediately after surface preparation but before submerged diffusion by atomic force microscopy (AFM) with an MFP-3D instrument in tapping mode. A smaller body of such characterization focused on rms roughness has been reported previously,34 but the current study examines more specimens and reports additional surface parameters such as skew and kertosis. Specimens were dried in flowing N2 gas to remove droplets removal after removal from the liquid. For accurate comparison between specimens, the Al-coated silicon probes (Ted Pella Inc.) were changed for every measurement. Each measurement generated 512 × 512 data points. Table T1 in the SI shows that etching with NH4OH yielded a broader range of rms roughness than simple degreasing, with the average being slightly smoother than simple degreasing. Skew and kurtosis varied considerably, although exceptionally high values were distorted by single large protrusions that could have been foreign particulates or dust.
18O depth profiles were measured ex situ by secondary ion mass spectrometry (SIMS) using a PHI-TRIFT III instrument. Depth profiling was accomplished with a 3 keV Cs ion beam having a spot diameter near 0.5 mm. Analysis after each cycle of crater deepening employed a beam of Au at 22 keV. 18O concentrations were calibrated to the known natural abundance of 18O (0.2%) within as-received TiO2 specimens used as standards. 18O concentrations in deep regions of diffused profiles often departed only a few percent from the natural abundance baseline. Thus, special care was taken to calibrate the ion currents corresponding to 16O and 18O – typically measured daily on standard specimens. After diffusion (or initial etching in liquid), specimens were simply removed from the liquid, which did not wet the surface. Occasional residual drops that adhered were blown off with a gentle stream of air or nitrogen. Diffusion profiles were measured several days to weeks after diffusion at 2–5 different locations on the surface of each specimen.
After diffusion in submersion, some specimens were subjected to stages of annealing lasting 50 min at progressively higher temperatures between 100 and 300 °C in air using procedures akin to those described elsewhere.12 SIMS was performed after each stage to ascertain whether spreading of the 18O profile had occurred due to release of Oi due to dissociation of interstitial clusters containing this species.
2.2 DFT calculations
Clusters containing Oi and Hi were relaxed using first-principles DFT simulations.37,38 The projector augmented wave (PAW) method39,40 as implemented in the Vienna ab initio Simulation Package (VASP)41,42 was utilized, in conjunction with the Perdew–Burke–Ernzerhof (PBE) approximation to the exchange–correlation functional.43 Calculations employed a plane-wave basis set with a cutoff energy of 520 eV. The ground state lattice parameters of the conventional unit cell for rutile TiO2 were determined to be a = 4.648 Å and c = 2.969 Å for a 3 × 3 × 4 supercell, consistent with other computational44–46 values (experimental values47–50 are a = 4.59 Å and c = 2.96 Å). Defect structures were modeled in 216-atom supercells using the Monkhorst–Pack scheme51 with a 2 × 2 × 2 k-point mesh. Geometry optimization of defect structures was performed with a convergence criterion of 1 × 10−6 eV for the total energy and 2 × 10−2 eV Å−1 for atomic forces. The bandgap computed from PBE was 1.79 eV, consistent with other reported PBE bandgaps46,52–54 and showing the expected DFT-PBE underestimate compared to the experimental value of 3.03 eV.55 To obtain more realistic descriptions of charge transition levels lying outside the PBE bandgap, the PBE band edges were shifted via alignment to the band edges of the Heyd–Scuseria–Ernzerhof (HSE06) screened hybrid functional approximation, opening the predicted gap to 3.05 eV.45,56The defect formation energy Eform[Xqi] of a point or cluster defect in charge state q was obtained using the supercell approach:57–59
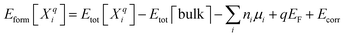 | (1) |
The situation is more complex for H. For TiO2(110), the surface normal is perpendicular to the c-axis. Interstitial hydrogen diffusion in this direction has a substantial barrier of 1.28 eV,17,60 which renders Hi incapable of diffusing at 70 °C and eliminates contact between the crystal and an external reservoir of H. Thus, the total amount of hydrogen is not expected to equilibrate with the liquid water environment during the experiment but instead remains constant at the initial adventitious value. However, as discussed below, assessment of which clusters are most likely to form considers reaction energies (Erxn) that do not depend upon the choice of reference states for O and H. Consequently, the chemical potential for H in eqn (1) was set to conditions associated with H in air, given by 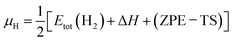 . Here, Etot(H2) denotes the DFT total energy of the H2 molecule, ΔH is the enthalpic correction to standard conditions, and ZPE–TS accounts for zero-point energy and thermal entropy contributions.
. Here, Etot(H2) denotes the DFT total energy of the H2 molecule, ΔH is the enthalpic correction to standard conditions, and ZPE–TS accounts for zero-point energy and thermal entropy contributions.
The Fermi level EF in eqn (1) accounts for the exchange of electrons from the reference electron reservoir. The energy correction term Ecorr accounts for the finite-size effects resulting from electrostatic interactions between charged defects in adjacent supercells. The method proposed by Lany and Zunger58 was employed to calculate the finite size effect corrections for potential alignment ΔEpa(D, q) and image charge ΔEi.
The experiments were carried out under kinetically controlled conditions far from equilibrium where it becomes more useful to examine reaction energies instead of formation energies. Erxn compares the formation energy of a cluster (Oi)y–(Hi)x with the summation of formation energies of its isolated point-defect constituents, as given by:
| Erxn = Eform[(Oi)y–(Hi)x] − y·Eform[Oi] − x·Eform[Hi]. | (2) |
Here, Eform[(Oi)y–(Hi)x] denotes the formation energy of a cluster comprising y interstitial O atoms and x H atoms, and Eform[Oi] and Eform[Hi] respectively represent the formation energies of isolated Oi and Hi. A negative value of Erxn signifies exothermicity at 0 K. The calculation does not account for entropy and therefore offers only an approximate guide to the free energy change at the nonzero temperature of the experiment.
3. Results
Fig. 1 illustrates a typical 18O SIMS profile following diffusion. The general shape of this profile matches those reported previously.22,23 The most dramatic feature is the isotopic fractionation represented by the near-surface “valley” where the 18O concentration drops well below the natural abundance (Cnat = 1.29 × 1020 cm−3) of 18O in TiO2. Isotopic fractionation is transient and represents an especially pronounced form of uphill diffusion.61The profiles may be quantified by several metrics,23 but the three of interest here are:
• F18: net 18Oi injection flux. The relationship between F18 and the total net flux Ftot of Oi (both isotopes) depends on the isotopic compositions of H2O and dissolved O2 as well as the chemical concentration of dissolved O2. In the present case of dissolved air, F18 should be multiplied by a factor of 10.6 to obtain Ftot.23
• λ1: characteristic length describing exponential decay at depths closely associated with uphill diffusion.
• λ2: characteristic length describing exponential decay at depths well beyond those associated with uphill diffusion.
Ref. 23 details the fitting procedures used to obtain these metrics, with the primary features described in the SI. All features are stable during long-term storage. However, details of the profile shapes vary with position on a given specimen, and between specimens. Such variations have been discussed at length elsewhere33,34 and arise mostly from differences in the level of surface contamination, especially adventitious carbon, which can poison the surface sites responsible for injection.
3.1 Effects of applied bias in the dark
As a reference point for UV studies, the empirical dependence of F18, λ1 and λ2 on T and diffusion time t was measured with applied bias in the dark. Under such conditions, the governing physical processes entail breaking and forming of chemical bonds, both in fluid species (O2, H2O) and in adsorbed OH, and presumably have a corresponding Arrhenius-type temperature dependence. Accordingly, Fig. 2 shows Arrhenius plots of F18, λ1 and λ2 from 30 ≤ T ≤ 70 °C. For comparison, Fig. 2 also shows data for these metrics without bias using a combination of previously published measurements,23 new measurements obtained by the methods reported there, and the analysis procedure employed in the SI. The plots make clear that use of applied bias considerably increases the average values of all three metrics. Table T2 in the SI provides the effective activation energies of F18, λ1 and λ2.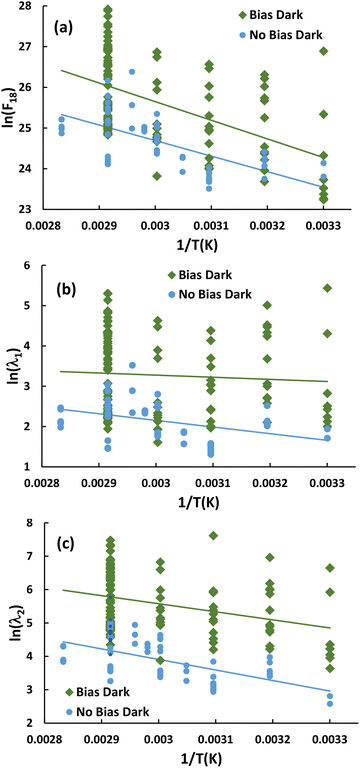 | ||
| Fig. 2 Arrhenius plots for profile metrics under dark conditions with and without applied bias: (a) F18, (b) λ1 and (c) λ2. Lines represent linear least squares fits. | ||
F18 depends upon the surface chemical processes of injection and annihilation. The wide variation in F18 at fixed temperature, often across the surface of a single specimen, results from variations in tiny concentrations of surface sites at which Oi injection is especially rapid.34 Variability in semiconductor behavior mediated by small concentrations of surface sites has ample precedent. Procedures to minimize such variability in Si device fabrication took several decades to develop fully.62 On TiO2 single crystals, small concentrations of defects catalytically mediate much of the surface chemistry.63 A general principle of heterogeneous catalysis is that the most reactive surface sites are often most vulnerable to deactivation by poisoning.64 Injection entails a form of catalysis wherein one reaction produce is Oi. Susceptibility to poisoning and the variability of injection rate that ensues34 is therefore not surprising.
Fig. 2 also shows considerable variability for λ1 and λ2. These metrics are measured well within the bulk and depend directly upon bulk processes such as hopping diffusion, atom exchange with the lattice, and interstitial cluster formation.23 However, progressive spatial and temporal evolution of clusters incorporating Oi provides a mechanism to couple bulk metrics to F18.34 Higher injection fluxes enable quicker evolution of the cluster isomer populations. Thus, much of the variability in λ1 and λ2 arises indirectly from the variations in F18.
To quantify this coupling, Fig. 3 shows plots of λ1 and λ2 vs. F18 in ln–ln form. All metrics with applied bias (green points) were measured at 70 °C. The plots of both λ1 and λ2 vs. F18 show good linearity, signifying an empirical power law dependence. Table 1 lists the corresponding slopes m, which provide the exponents m for normalization of bulk metrics by Fm18.
| Metric | Applied bias dark | Without bias dark | Without bias UV |
|---|---|---|---|
| λ1 | 1.05 ± 0.01 | 0.89 ± 0.04 | 0.72 ± 0.05 |
| λ2 | 0.83 ± 0.02 | 0.72 ± 0.10 | 0.27 ± 0.10 |
Fig. 3 also shows corresponding data for λ1 and λ2 (blue points) measured without applied bias using previously published results,33 new measurements obtained by the methods reported there, and the analysis procedure employed in the SI. However, fewer data were available at 70 °C than for measurements with applied bias. To compensate for the smaller data set, a scaling procedure was employed to remove the effects of differing T. The regression lines from the Arrhenius plots in Fig. 2 were used to estimate the value of each data point scaled to 70 °C. In a chosen Arrhenius plot this procedure in effect translates points at T ≠ 70 °C parallel to the regression line to T = 70 °C – quantitatively preserving the standard error around the regression line. We have employed this procedure elsewhere,33 but without a direct test of its validity. Here, such a test becomes possible using the data with applied bias because of the large number of points across the entire temperature range (about 125). When the entire data sets for λ1 and λ2 were scaled to 70 °C, the resulting lines changed slope by only a few percent. The scatter about the corresponding regression lines worsened, but only slightly. These results indicate that the scaling procedure does not introduce significant distortions or biases into the analysis.
Even after variability due to F18 is accounted for, Fig. 3's data for λ1 and λ2 without bias (both dark and UV) exhibit more scatter than the corresponding results with bias. The origin of this added scatter remains unclear at present, but procedural or metrological differences cannot explain it because the same equipment and methodology were employed for all data sets. However, an empirical linear correlation between ln(λ1) and ln(F18) works very well for data with bias. We therefore employed similar empirical linear fitting for data without bias (both dark and UV) to create a basis for comparing all the data sets quantitatively. The slopes of λ1 and λ2 without bias in the dark (Table 1) fall somewhat below those with applied bias. However, the slopes for λ2 fall within each other's error bars.
Experiments were performed to examine the time dependence of the three metrics with applied bias. Fig. 4a shows ln(F18) at 70 °C vs. ln(t) in the range 6–60 min. In this range, ln(F18) decreases with a slope of −0.90 ± 0.20. The decrease presumably originates from increasing back-diffusion and annihilation of Oi to the surface as the concentration of Oi in the bulk steadily increases. Surface annihilation is required by the principle of microscopic reversibility, as has been previously reported and discussed for Oi injection into metal oxides.23
Corresponding plots for λ1 and λ2 with bias at 70 °C appear in Fig. S3 of the SI. These metrics by themselves exhibit little time dependence. However, they are heavily convolved with the time dependence of F18 as discussed above. To examine the effects of time on profile evolution in these regions independent of F18, Fig. 4 also shows ln–ln plots of λ1/F1.0518 and λ2/F0.8318 vs. t. Both normalized metrics increase with time. The slopes in Fig. 4 are 1.05 ± 0.04 for λ1/F1.0518 and 0.78 ± 0.05 for λ2/F0.8318.
3.2 Effects of ultraviolet illumination
Fig. 5 shows Arrhenius plots of F18, λ1 and λ2 without applied bias, both in the dark and with UV illumination. Table T2 in the SI provides the corresponding effective activation energies. The data shown in blue (measured in the dark) replicate the corresponding results in Fig. 2, with expanded vertical scales in Fig. 5 to enable clearer comparison with UV results. The effects of UV illumination on F18 and λ1 broadly accord with those reported in a previous study33 (wherein λ1 was termed “λ”), with differences in detail arising from the enlarged data set and significant differences in the procedure for computing profile metrics. For example, existence of the λ2 region was not known at the time of the previous study. | ||
| Fig. 5 Arrhenius plots for profile metrics under dark and UV conditions without applied bias: (a) F18, (b) λ1 and (c) λ2. Lines represent linear least squares fits. | ||
Thus, the UV data in Fig. 5 were scaled to 70 °C as described above for dark data and were plotted in Fig. 3 as red points with empirical linear fits. Table 1 shows the corresponding slopes. For λ1, UV illumination exerts little effect on the general magnitude, but the slope m decreases noticeably. The ranges encompassed by the error bars with and without UV do not overlap. For λ2, UV illumination dramatically decreases m, and the ranges encompassed by their respective standard deviations come nowhere near overlapping. Visually, the points at the high end of the flux range lie noticeably lower than the corresponding points measured in the dark.
The variability of λ2, together with higher injection fluxes made possible by applied potential bias (increasing the signal-to-noise ratio for measuring λ2), prompted measurement of additional data under UV illumination with bias. An elevated temperature of 70 °C was chosen because Fig. 2(c) shows that λ2 increases with T. Although measurements are possible at 80 °C or higher, loss of labeled water to evaporation becomes a significant problem on the time scale of these experiments, even with refluxing.
Fig. 3 shows the results with points that are purple and orange. UV prompts no discernable change in λ1. Surprisingly, UV appears to increase λ2, although the effect is too small to assess its reliability with visual inspection alone. Statistical analysis is necessary.
Such analysis directly on F18, λ1 and λ2 can be misleading, as all these metrics vary over ranges nearly two orders of magnitude wide. Histograms of the distributions23 reveal significant deviations from normality in the form of strong positive skew. In addition, all three metrics are heteroscedastic (i.e., the standard deviation varies with the value of the metric itself), which is common for data sets encompassing such wide ranges. Various methods have been proposed for reliable statistical analysis of heteroscedastic, non-normal data.
Logarithmic transformation is a common approach employed across a wide range of disciplines including biology,65 chemistry,66 epidemiology,67 environmental science,68,69 social science,70 econometrics71 and others. The transformation often removes heteroscedasticity and sometimes leads to normal distributions. Arrhenius plotting as in Fig. 2 requires logarithmic transformation. The distributions for all three metrics become homoscedastic after transformation.
However, the distributions continue to deviate from normality. Classical parametric statistical tests (e.g., t-tests) exhibit considerable robustness to deviations from a normal distribution,72,73 but reformulation of the variables based on independent knowledge of the physical system is preferable whenever possible.65 Such a reformulation here is indeed possible as described above: determination of empirical exponents m for normalization of bulk metrics by Fm18. For a given bulk metric, m represents an empirical quantity that depends upon t and several other processes occurring in the bulk. Nevertheless, for profiles measured with applied bias, histograms of the normalized metrics λ1/F1.0518 and λ2/F0.8318 follow normal distributions, indicating that the non-normality previously reported23 for λ1 and λ2 was due to the influence of F18.
Table 2 shows the means and standard deviations of ln(F18), λ1/F1.0518 and λ2/F0.8318. All these variables are homoscedastic. The latter two obey normal distributions, but ln(F18) does not. For completeness, Table T3 in the SI shows the means and standard deviations of the original metrics (F18, λ1 and λ2) under both dark and UV conditions with applied bias at 70 °C. Because these variables are heteroscedastic and encompass ranges up to two orders of magnitude, their standard deviations look unnaturally large.
| Mean (dark) | Mean (UV) | Deg of freedom | pt | pMW | |
|---|---|---|---|---|---|
| a Two-tailed, equal variances (pooled) except for ln(F18), which assumes unequal variances. | |||||
| ln(F18) | 26.0 ± 0.9 | 26.5 ± 0.5 | 8 | 0.053 | 0.20 |
| λ1/F1.0518 | (4.03 ± 0.29) × 10−11 | (4.14 ± 0.32) × 10−11 | 76 | 0.40 | — |
| λ2/F0.8318 | (1.19 ± 0.24) × 10−7 | (1.38 ± 0.30) × 10−7 | 76 | 0.069 | — |
Table 2 also shows the results of t-tests comparing the values of ln(F18), λ1/F18 and λ2/F0.818 with and without UV illumination. For both ln(F18) and λ2/F0.8318, the likelihoods pt are 0.07 or below that illumination has no statistically significant effect. In contrast, pt is much larger at 0.4 for λ1/F1.0518. In other words, UV probably increases λ2 directly but exerts no direct effect on λ1.
The t-test for ln(F18) is suspect because of the non-normal, positively skewed distribution. Distributions with strong positive skew tends to overestimate statistical significance in t-tests.74 Thus, a non-parametric Mann–Whitney U-test was also applied to ln(F18) that does not presuppose normality. The non-parametric test effectively compares medians75 rather than means. Compared to a t-test, a Mann–Whitney test is less vulnerable to type I errors that reject the null hypothesis when it is true. The resulting probability pMW is 0.20. However, a Mann–Whitney test is more vulnerable than a t-test to type II errors that fail to reject the null hypothesis when it is false.
The statistical tests for ln(F18) thus provide a mixed assessment. The t-test implies strong statistical significance, while the Mann–Whitney test implies much weaker significance. However, both tests suffer drawbacks. As suggested by Fig. 5a without bias, the effect of UV varies with T and may be too small at 70 °C to reliably detect given the variability in the data sets.
A few crystals that were diffused with applied bias were subjected to progressive annealing steps between 100 and 300 °C. In contrast to previous work without applied bias,12 wherein such heating induced appreciable profile spreading, the profiles generated with bias exhibited no measurable spreading up to and including 300 °C.
3.3 DFT results for clusters of Oi and Hi
Fig. S4 in the SI shows the defect formation energies computed via eqn (1) for the simple point defects Oi, VO, Hi, and HO, where HO represents an H atom that substitutes for O in the TiO2 lattice. For the O-related defects under O-rich conditions, Oi is more stable than VO for n-type material (nearly universal for TiO2 single crystals). The ordering inverts for p-type material. In agreement with published studies,21,52,76 Oi exists in the 2− charge state over much of the EF range, and VO in the 2+ charge state.77,78 Hi exists primarily as a 1+ donor, in agreement with previous work.13,17,79–82 HO exists primarily as a 1+ donor, changing to a 1− acceptor at EF close to the conduction band minimum (CBM). The donor behavior largely agrees with a previous study80 that reported HO to be exclusively a donor over the entire range of EF A different study reported only acceptor behavior,79 but that work placed EF in the conduction band, and a full analysis of charge state vs. EF was not performed. HO is known to be amphoteric in other oxides such as ZnO,83–85 and to a modest extent that behavior appears in Fig. S4.For dimer clusters having stoichiometry Oi–Hi, the DFT calculations identified several isomers. This study did not attempt an exhaustive search of configuration space, but a random sampling method was employed to generate Oi–Hi defects. An O atom with randomly sampled spatial coordinates was placed within a single unit cell of the 3 × 3 × 4 TiO2 supercell. An H atom was then placed within an annular region centered on the O defect, with the O–H distance drawn from a Gaussian distribution and the orientation determined by a randomly selected angle. The defective supercells then underwent geometry relaxation to determine the corresponding energies.
Fig. 6 shows the four most stable dimer cluster configurations, and Fig. S5a shows the corresponding formation energies. Dimer a exhibits exclusively donor behavior until it nears the conduction band edge, maintaining a constant charge state of 1+ for EF < 2.8 eV, whereafter it exhibits acceptor behavior with a charge state of 2−. Dimers b–d adopt various forms of a split geometry, which means that the two O atoms associated with a single lattice site form no chemical bond between each other. These isomers are 1− and 3− acceptors for EF > 0.8 eV but become neutral for EF lower in the bandgap. From the thermodynamic perspective of formation energies, dimer a is most stable for 0 < EF < 1.7 eV, and dimer b dominates for EF > 1.7 eV. Dimers c and d are never the most stable species; c and d formation energies are 0.5–0.6 eV larger than that of b throughout the entire band gap. From the kinetic perspective, the situation could differ as detailed in the Discussion. Notably, the H atoms in dimers b and c bond directly to one of the O atoms comprising the defect's Oi “core.” In dimers a and b, the H atom forms the strongest bond to O atoms neighboring the defect core.
 | ||
| Fig. 6 Geometries of the four most stable dimers Oi–Hi. Blue, white and red spheres respectively represent Ti, lattice O and H. Green signifies the “extra” O in the core O interstitial. | ||
For trimer clusters of interstitials having stoichiometry Oi–(Hi)2, the DFT calculations again identified several isomers. Fig. 7 shows the five most stable configurations, and Fig. S5b shows the corresponding formation energies. Trimers a, d and e are donors, with a and d maintaining 2+ charge for all EF and e maintaining 1+. Trimer c is neutral for all EF up to 2.8 eV, whereafter it transitions through charge states 2−, 3−, 4−, and 5− for EF > 2.8 eV. Trimer b is neutral for EF < 1.0 eV, a 2− acceptor for 1.0 < EF < 2.8 eV and transitions through charge states 1−, 2−, 3−, 4−, and 5− for EF > 2.8 eV. As with dimers, the geometries of these trimers remain largely unchanged regardless of their charge state. From a thermodynamic perspective of formation energies, dimer a is most stable for 0 < EF < 2.0 eV. Trimer c dominates for a narrow range 2.0 < EF < 2.5 eV and trimer b dominates for EF > 2.5 eV, Trimers d and e are never the most stable species. The connection between stability and bonding of H differs from that of dimers. For trimers, only a has no H atoms bonded directly to the O atoms in the core Oi defect. In d and e, one H atom bonds to the core defect, while in b and c, both H atoms bond to core O atoms.
 | ||
| Fig. 7 Geometries of the five most stable trimers Oi–(Hi)2. Blue, white and red spheres respectively represent Ti, lattice O and H. Green signifies the “extra” O in the core O interstitial. | ||
As noted in the Methods section, H and to a lesser extent O were not fully in contact with an external reservoir of the corresponding elements. Under such kinetically limited conditions, it is more appropriate to gauge the propensity to form dimer and trimer species via their reaction energies (ΔErxn) with respect to the reactants Oi and Hi. Exothermic reactions with ΔErxn < 0 are more likely to occur than endothermic reactions.
Accordingly, Fig. 8 shows ΔErxn vs. EF for the dimers and trimers described above exhibiting the most negative values of ΔErxn. The details of the plots are complicated, but the high-level conclusion is that, over the entire range of EF, at least one dimer and one trimer exist for which formation from Oi and Hi is exothermic. For completeness, Fig. S6a and b depict ΔErxn vs. EF for all dimers and trimers, respectively.
4. Discussion
Most literature describing the effects of photostimulation on defect-mediated diffusion focuses on the formation energies of mobile charged defects.27,86,87 Photogenerated minority carriers affect these energies, which propagate into mobile defect concentrations and the corresponding effective diffusivities. The effects on hopping are usually not considered unless the dominant charge state changes (and thereby the relevant hopping barrier)88,89 or direct conversion of electronic to vibrational energy at the defect induces hopping.8 However, thermodynamic formation energies have little relevance in the present kinetically limited system. Effects of interstitial hopping and clustering are more important.The lack of knowledge about interstitial cluster formation in oxides near room temperature probably reflects their substantial mobility,13,90 which enables their rapid capture by bulk traps or surfaces. Interstitial clustering behavior near room temperature has started to attract attention only recently, and mainly in Si-based systems9 where illumination exerts noticeable effects.
An important experimental complication compounds the lack of knowledge; super-bandgap illumination is absorbed and ultimately converted into heat. Thus, illumination may simultaneously induce both thermal and nonthermal effects that are difficult to disaggregate.89 Keeping the illumination intensity low minimizes such effects, and an important aspect of the present data rules out this complication here. The effective activation energy of F18 is higher than that of λ2, implying that F18 has a stronger temperature dependence. Yet λ2 in Table 1 shows a greater fractional increase in response to UV than F18. Heating alone would result in F18 exhibiting the larger fractional increase.
4.1 Response of the profile metrics to UV
Fig. 3 and 4 show that F18, λ1 and λ2 all change in different ways in response to applied bias, t and UV illumination. These trends all point to differences in the primary governing mechanism for each metric.Little theory exists for λ1, which contains 18O that has diffused from the “valley” region of isotopic fractionation.22 However, the λ1 region is much closer to the surface than the λ2 region. Therefore, the λ1 develops sooner than the λ2 region, and the concentration of Oi at any given time is higher. For these reasons, the λ1 region responds less to trapping effects than the λ2 region. If trapping effects are weak, the normalized metric λ1/Fm18 depends primarily upon the diffusivity D. As mentioned above, there is precedent for super-bandgap illumination to affect the interstitial hopping barrier88,89 due to a change in the majority charge state. However, this effect seems to be either small or nonexistent here, both with and without applied bias.
In contrast, λ2 samples a region far deeper in the solid, which is exposed to lower concentrations of Oi for shorter times. A metric measured in this region is more sensitive to the effects of small concentrations of saturable Oi traps. D probably affects λ2, but photostimulated effects on D appear to be negligible. With possible effects of UV on F18 removed from the normalized variable, the evidence points to significant changes in λ2 due to UV, corresponding to modified Oi capture by traps.
F18 contains primarily surface rate parameters as well as the coverage of injectable O, which is presumably set by liquid–surface interactions. As mentioned in the Methods, the application of bias may remove dissolved impurities that poison injection sites with high activity. Also, the electrodes and associated wiring may unintentionally introduce to the liquid small amounts of impurities such as Na. Ions of Na have recently been shown91 to shed their hydration shell and bond directly to TiO2(110), thereby setting up very strong electric fields (108 V cm−1) that could influence the injection of charged species such as Oi.
Although surface kinetic parameters dominate F18, it also contains D via the surface annihilation rate. Thus, UV can affect F18 directly via the surface rate parameters and indirectly via D. Here, however, UV exerts little if any influence on D.
Without applied bias, UV increases F18 (Fig. 2a), at the lower end of the temperature range. UV exerts little effect at 70 °C, a finding consistent with the mixed statistical results (Mann–Whitney and t-tests) discussed above with applied bias. This study cannot determine why UV increases F18 at lower temperatures, although removal of carbonaceous impurities by photo-oxidation has been proposed to cause UV-induced increases in hydrophilicity on TiO2(110).92 A similar mechanism may clear Oi injection sites of carbonaceous poisons.
4.2 Properties of interstitial clusters
Oi and Hi are known to form clusters when both species are extrinsic to the semiconductor, as in Si.93 In metal oxides, progressive annealing measurements12 of 18O profiles after aqueous submersion of TiO2 and ZnO single crystals have suggested that families of (Oi)y–(Hi)x interstitial clusters immobilize Oi, but supporting computational evidence was lacking. The DFT calculations detailed in this work provide such evidence.Interstitial families of clusters exist for dopants in silicon, such as boron.94 In UO2, wherein the U cation and O anion have comparable sizes, “Willis clusters” containing multiple Oi atoms exist95 along with mixed interstitial clusters containing Oi and Hi.96 Thus, the identification of dimers and trimers containing Oi and Hi by the present calculations is not surprising. However, given the rarity of Oi in oxides not synthesized specifically to be hyperstoichiometric,97,98 the behavior of Oi-containing clusters in oxides remains little investigated.
Certain aspects of the behavior observed here are straightforward to rationalize. The dominant charge states of the most thermodynamically stable dimers often represent the sums of the dominant charge states of the constituent Oi and Hi. The geometry of the dimer Oi core (dumbbell or split) mimics that of the parent Oi reactant. For trimers, the same patterns also hold for the most thermodynamically stable species except in highly n-type material, for which a split acceptor trimer exists. Typically, the most thermodynamically stable dimers and trimers isomerize to different geometries when changing ionization states – much like Oi does.
The dimer and trimer isomers differ considerably among themselves in geometry. The interconversion barriers were not examined but could be considerable given the rearrangements involved. Under kinetically controlled conditions near room temperature, thermodynamically unstable clusters may survive intact. The exothermicity of certain species in Fig. 8 are not helpful for predicting such isomerization; some of the depicted reactions are very exothermic in p-type material because the Oi reactant is very unstable thermodynamically under those conditions.
4.3 Effects of interstitial clustering
Traps for Oi in as-received rutile TiO2 take several forms at submersion temperatures, including VO, extended defects, and families12,99,100 of small interstitial clusters. Hydrogen is ubiquitous in metal oxides101,102 as an adventitious impurity and forms complexes with various defects,102,103 including electron acceptors101 such as Oi. The wide range of activation barriers that characterizes dissociation of Oi traps in TiO2 suggests that the (Oi)x–(Hi)y family of clusters may include many members. One isomer of the dimer has been reported previously13 but the DFT results presented here show that several isomers of the dimer Oi–Hi and trimer Oi–(Hi)2 may also form exothermically from Oi and Hi.Hi may be produced if Oi replaces HO in the lattice via a standard lattice kickout reaction, which seems likely. Oi may also react with molecular H2 trapped in the lattice. H2 has not been positively identified in rutile, although H2 has been reported likely to exist in anatase.104 Assuming H2 exists in rutile, reaction with Oi would presumably lead to either Oi–(Hi)2 or Oi–Hi plus free Hi. Clusters involving multiple Oi species may exist, but that possibility was not investigated here.
Some of the available traps for Oi, such as VO and HO substituting for O in the lattice,79,83,105 can be saturated if exposed to enough Oi. The ability of clusters with the general formula (Oi)x–(Hi)y to saturate remains unclear because many family members have not yet been identified. Saturable traps for Oi affect the λ2 region most strongly where isotopic label concentrations remain close to natural abundance near the leading edge of the tail.
Smaller values of λ2 imply more trapping. The correspondence suggests that in the absence of applied bias, UV increases the concentration of one or more small clusters containing O that absorb Oi. The effect is larger in the higher ranges of injection fluxes characteristic observed without bias. Candidate clusters include the Oi–Hi− cluster as well as larger clusters of formula (Oi)y–(Hi)x for which Oi–Hi− serves as a kinetic precursor.
The primary cluster isomers identified in the DFT calculations exist in multiple charge states. This is probably also true of other isomers not yet identified. In the dark, the relative populations of the isomers and their charge states depend upon EF. Under illumination, both the isomeric makeup and the charge states change depend on the isomers’ and point defects’ interactions with excess minority carriers via cross sections for carrier capture, excited state lifetimes, and other factors. The isomeric makeup can change by several pathways. One or more isomers’ stability may increase or decrease due to altered charge state. The charge state of Oi or Hi may change, affecting the exothermicity of the formation reaction of one or more isomers. The concentration of Hi may change because its rate of formation from HO or H2 may depend upon the charge state of Oi or HO. In other words, illumination alters the rate constants for formation or dissociation of (Oi)y–(Hi)x or changes the concentration of the reactant Hi. Fig. 9a and b depict these ideas schematically.
With applied bias, F18 is larger than for any of the experiments without applied bias. The (Oi)y–(Hi)x clusters are therefore more evolved and have a different balance of stoichiometries. This evolution is clear from the lack of spreading in progressive annealing experiments up to 300 °C, for which a great deal of spreading occurs when diffusion occurs without applied bias. The lack of measurable spreading implies that few clusters have dissociated to release the Oi required for spreading. Fig. 9c illustrates this evolution schematically. The concentration of the preponderant cluster in this different population evidently decreases in response to UV illumination as depicted in Fig. 9d, leading to behavior of λ2 that is opposite to illumination without bias wherein the preponderant cluster concentration increases in response to UV.
5. Conclusion
This study combines simulations of Oi–Hi clusters by density functional theory (DFT) with self-diffusion measurements of 18O in submerged single-crystal rutile TiO2(110) illuminated by ultraviolet (UV) light. The simulations show that Oi–(Hi)x dimers and trimers exist in several stable isomers, and each has multiple charge states that can change upon illumination. The diffusion measurements indicate that UV affects the concentration of interstitial clusters containing Oi, which changes the characteristic decay length for penetration of 18O into the solid. Together, the simulations and experiments suggest that illumination alters the rate constants for formation or dissociation of (Oi)y–(Hi)x or changes the concentration of the reactant Hi.At a high level, this work introduces methods to facilitate kinetically dominated phenomena like strong isotopic fractionation by using submersion-created interstitials from oxide surfaces at temperatures below 100 °C. An important step has been taken to solidify the idea that families of interstitial clusters such as (Oi)y–(Hi)x exist that can immobilize interstitials, and that coordination of temperature and illumination should enable their optimization for defect control.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data that support the findings of this study are included within the article and its supplementary information (SI). Supplementary information: summary method for determination of profile metrics; supplementary figures and tables. See DOI: https://doi.org/10.1039/d5cp04013b.Acknowledgements
This work was supported by the U.S. National Science Foundation under grant DMR 17-09327 and DMR 23-22121, and the Air Force Office of Scientific Research under grant FA9550-21-0078. SIMS and XPS measurements were performed in the Materials Research Laboratory Central Research Facilities, University of Illinois at Urbana-Champaign. This work used PSC Bridges-2 at the Pittsburgh Supercomputing Center through allocation MAT220011 from the Advanced Cyberinfrastructure Coordination Ecosystem: Services and Support (ACCESS) program, which is supported by the National Science Foundation Grants No. 2138259, No. 2138286, No. 2138307, No. 2137603, and No. 2138296. The authors thank Jaeyoung Hong, Hexi Huang and Andros Gao for extensive help with performing the progressive annealing experiments.References
- H. Roth, W. Bernard, P. Zeldes and A. P. Schmid, Voltage-Annealing of Radiation Damage in Tunnel Diodes, J. Appl. Phys., 1963, 34, 669–671 CrossRef.
- D. Macdonald, F. Rougieux, A. Cuevas, B. Lim, J. Schmidt, M. Di Sabatino and L. J. Geerligs, Light-induced boron-oxygen defect generation in compensated p-type Czochralski silicon, J. Appl. Phys., 2009, 105, 093704 CrossRef.
- F. Fertig, R. Lantzsch, F. Frühauf, F. Kersten, M. Schütze, C. Taubitz, J. Lindroos and J. W. Müller, Excessive light-induced degradation in boron-doped Cz silicon PERC triggered by dark annealing, Sol. Energy Mater. Sol. Cells, 2019, 200, 109968 CrossRef CAS.
- Y.-C. Zhao, W.-K. Zhou, X. Zhou, K.-H. Liu, D.-P. Yu and Q. Zhao, Quantification of light-enhanced ionic transport in lead iodide perovskite thin films and its solar cell applications, Light: Sci. Appl., 2017, 6, e16243 CrossRef CAS PubMed.
- G. Y. Kim, A. Senocrate, T.-Y. Yang, G. Gregori, M. Grätzel and J. Maier, Large tunable photoeffect on ion conduction in halide perovskites and implications for photodecomposition, Nat. Mater., 2018, 17, 445–449 CrossRef CAS PubMed.
- D. V. Lang and L. C. Kimerling, Observation of Recombination-Enhanced Defect Reactions in Semiconductors, Phys. Rev. Lett., 1974, 33, 489–492 CrossRef CAS.
- E. Mosconi, D. Meggiolaro, H. J. Snaith, S. D. Stranks and F. De Angelis, Light-induced annihilation of Frenkel defects in organo-lead halide perovskites, Energy Environ. Sci., 2016, 9, 3180–3187 RSC.
- J. D. Weeks, J. C. Tully and L. C. Kimerling, Theory of recombination-enhanced defect reactions in semiconductors, Phys. Rev. B, 1975, 12, 3286–3292 CrossRef CAS.
- J. Coutinho, D. Gomes, V. J. B. Torres, T. O. Abdul Fattah, V. P. Markevich and A. R. Peaker, Theory of reactions between hydrogen and group-III acceptors in silicon, Phys. Rev. B, 2023, 108, 14111 CrossRef CAS.
- M. Siebenhofer, A. Viernstein, M. Morgenbesser, J. Fleig and M. Kubicek, Photoinduced electronic and ionic effects in strontium titanate, Mater. Adv., 2021, 2, 7583–7619 RSC.
- M. Lyulyukin, N. Kovalevskiy, I. Prosvirin, D. Selishchev and D. Kozlov, Thermo-photoactivity of pristine and modified titania photocatalysts under UV and blue light, J. Photochem. Photobiol., A, 2022, 425, 113675 CrossRef CAS.
- H. Jeong and E. G. Seebauer, Influence of interstitial cluster families on post-synthesis defect manipulation and purification of oxides using submerged surfaces, J. Chem. Phys., 2024, 161, 121103 CrossRef CAS PubMed.
- H. Jeong, E. Ertekin and E. G. Seebauer, Surface-Based Post-synthesis Manipulation of Point Defects in Metal Oxides Using Liquid Water, ACS Appl. Mater. Interfaces, 2022, 14, 34059–34068 CrossRef CAS PubMed.
- A. Janotti and C. G. Van de Walle, Hydrogen multicentre bonds, Nat. Mater., 2007, 6, 44–47 CrossRef CAS PubMed.
- H. Li and J. Robertson, Behaviour of hydrogen in wide band gap oxides, J. Appl. Phys., 2014, 115, 203708 CrossRef.
- A. Venzie, A. Portoff, E. C. P. Valenzuela, M. Stavola, W. B. Fowler, S. J. Pearton and E. R. Glaser, Impurity-hydrogen complexes in β-Ga2O3: Hydrogenation of shallow donors vs. deep acceptors, J. Appl. Phys., 2022, 131, 035706 CrossRef CAS.
- A. J. Hupfer, E. V. Monakhov, B. G. Svensson, I. Chaplygin and E. V. Lavrov, Hydrogen motion in rutile TiO2, Sci. Rep., 2017, 7, 17065 CrossRef CAS PubMed.
- M. D. McCluskey, M. C. Tarun and S. T. Teklemichael, Hydrogen in oxide semiconductors, J. Mater. Res., 2012, 27, 2190–2198 CrossRef CAS.
- Y. Knausgård Hommedal, M. Etzelmüller Bathen, V. Mari Reinertsen, K. Magnus Johansen, L. Vines and Y. Kalmann Frodason, Theoretical modeling of defect diffusion in wide bandgap semiconductors, J. Appl. Phys., 2024, 135, 170902 CrossRef.
- H. Jeong, M. Li, J. Kuang, E. Ertekin and E. G. Seebauer, Mechanism of Creation and Destruction of Oxygen Interstitial Atoms by Nonpolar Zinc Oxide(10-10) Surfaces, Phys. Chem. Chem. Phys., 2021, 23, 16423–16435 RSC.
- H. Jeong, E. Ertekin and E. G. Seebauer, Kinetic Control of Oxygen Interstitial Interaction with TiO2(110) via the Surface Fermi Energy, Langmuir, 2020, 36, 12632–12648 CrossRef CAS PubMed.
- H. Jeong and E. G. Seebauer, Strong Isotopic Fractionation of Oxygen in TiO2 Obtained by Surface-Enhanced Solid-State Diffusion, J. Phys. Chem. Lett., 2022, 13, 9841–9847 CrossRef CAS PubMed.
- H. Jeong, I. I. Suni, R. Chen, M. Miletic, X. Su and E. G. Seebauer, Reactions of fluid and lattice oxygen mediated by interstitial atoms at the TiO2(110)–water interface, Phys. Chem. Chem. Phys., 2025, 27, 9522–9536 RSC.
- J. Wang, R. C. Ewing and U. Becker, Average structure and local configuration of excess oxygen in UO2 + x, Sci. Rep., 2014, 4, 4216 Search PubMed.
- D. A. Andersson, T. Watanabe, C. Deo and B. P. Uberuaga, Role of di-interstitial clusters in oxygen transport in UO2 + x from first principles, Phys. Rev. B:Condens. Matter Mater. Phys., 2009, 80, 60101 Search PubMed.
- Z. Zhang and A. Janotti, Cause of Extremely Long-Lasting Room-Temperature Persistent Photoconductivity in SrTiO3 and Related Materials, Phys. Rev. Lett., 2020, 125, 126404 CrossRef CAS PubMed.
- K. Alberi and M. A. Scarpulla, Suppression of compensating native defect formation during semiconductor processing via excess carriers, Sci. Rep., 2016, 6, 27954 CrossRef CAS PubMed.
- X. Xie, B. Li, Z. Zhang, S. Wang and D. Shen, Controlled compensation via non-equilibrium electrons in ZnO, Sci. Rep., 2018, 8, 17020 CrossRef PubMed.
- A. Klump, M. P. Hoffmann, F. Kaess, J. Tweedie, P. Reddy, R. Kirste, Z. Sitar and R. Collazo, Control of passivation and compensation in Mg-doped GaN by defect quasi Fermi level control, J. Appl. Phys., 2020, 127, 045702 CrossRef CAS.
- B. Hammann, P. Vieira Rodrigues, N. Aßmann, W. Kwapil, F. Schindler, M. C. Schubert and S. W. Glunz, Deciphering the Role of Hydrogen in the Degradation of Silicon Solar Cells under Light and Elevated Temperature, Sol. RRL, 2024, 8, 2400457 CrossRef CAS.
- D. Rana, S. Agarwal, M. Islam, A. Banerjee, B. P. Uberuaga, P. Saadatkia, P. Dulal, N. Adhikari, M. Butterling, M. O. Liedke, A. Wagner and F. A. Selim, Light-driven permanent transition from insulator to conductor, Phys. Rev. B, 2021, 104, 245208 CrossRef CAS.
- A. Viernstein, M. Kubicek, M. Morgenbesser, T. M. Huber, E. Ellmeyer, M. Siebenhofer, C. A. F. Vaz and J. Fleig, Mechanism of photo-ionic stoichiometry changes in SrTiO3, Solid State Ionics, 2022, 383, 115992 CrossRef CAS.
- H. Jeong and E. G. Seebauer, Effects of Ultraviolet Illumination on Oxygen Interstitial Injection from TiO2 under Liquid Water, J. Phys. Chem. C, 2022, 126, 20800–20806 CrossRef CAS.
- H. Jeong and E. G. Seebauer, Effects of adventitious impurity adsorption on oxygen interstitial injection rates from submerged TiO 2 (110) and ZnO(0001) surfaces, J. Vac. Sci. Technol., A, 2023, 41, 033203 CrossRef CAS.
- J. Zhang, S. Ferrie, S. Zhang, Y. B. Vogel, C. R. Peiris, N. Darwish and S. Ciampi, Single-Electrode Electrochemistry: Chemically Engineering Surface Adhesion and Hardness To Maximize Redox Work Extracted from Tribocharged Silicon, ACS Appl. Nano Mater., 2019, 2, 7230–7236 CrossRef CAS.
- C. Liu and A. J. Bard, Electrons on dielectrics and contact electrification, Chem. Phys. Lett., 2009, 480, 145–156 CrossRef CAS.
- P. Hohenberg and W. Kohn, Inhomogeneous Electron Gas, Phys. Rev., 1964, 136, B864–B871 CrossRef.
- W. Kohn and L. J. Sham, Self-Consistent Equations Including Exchange and Correlation Effects, Phys. Rev., 1965, 140, A1133–A1138 CrossRef.
- G. Kresse and D. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- P. E. Blöchl, Projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- G. Kresse and J. Furthmüller, Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- G. C. Vásquez, S. Zh Karazhanov, D. Maestre, A. Cremades, J. Piqueras and S. E. Foss, Oxygen vacancy related distortions in rutile TiO2 nanoparticles: A combined experimental and theoretical study, Phys. Rev. B, 2016, 94, 235209 CrossRef.
- A. Janotti, J. B. Varley, P. Rinke, N. Umezawa, G. Kresse and C. G. Van de Walle, Hybrid functional studies of the oxygen vacancy in TiO2, Phys. Rev. B:Condens. Matter Mater. Phys., 2010, 81, 85212 CrossRef.
- Y.-S. Kim, Y.-C. Chung and K. S. Lee, The electronic structure of Ni doped rutile TiO2, J. Electroceram., 2006, 17, 951–953 CrossRef CAS.
- K. V. K. Rao, S. V. N. Naidu and L. Iyengar, Thermal Expansion of Rutile and Anatase, J. Am. Ceram. Soc., 1970, 53, 124–126 CrossRef CAS.
- M. E. Straumanis, T. Ejima and W. J. James, The TiO2 phase explored by the lattice constant and density method, Acta Crystallogr., 1961, 14, 493–497 CrossRef CAS.
- D. T. Cromer and K. Herrington, The Structures of Anatase and Rutile, J. Am. Chem. Soc., 1955, 77, 4708–4709 CrossRef CAS.
- J. K. Burdett, T. Hughbanks, G. J. Miller, J. W. Richardson and J. V. Smith, Structural-electronic relationships in inorganic solids: powder neutron diffraction studies of the rutile and anatase polymorphs of titanium dioxide at 15 and 295 K, J. Am. Chem. Soc., 1987, 109, 3639–3646 CrossRef CAS.
- H. J. Monkhorst and J. D. Pack, Special points for Brillouin-zone integrations, Phys. Rev. B, 1976, 13, 5188 CrossRef.
- T. S. Bjørheim, A. Kuwabara and T. Norby, Defect Chemistry of Rutile TiO2 from First Principles Calculations, J. Phys. Chem. C, 2013, 117, 5919–5930 CrossRef.
- X. Cai, P. Zhang and S.-H. Wei, Revisit of the band gaps of rutile SnO2 and TiO2: a first-principles study, J. Semicond., 2019, 40, 092101 CrossRef CAS.
- S.-D. Mo and W. Y. Ching, Electronic and optical properties of three phases of titanium dioxide: Rutile, anatase, and brookite, Phys. Rev. B:Condens. Matter Mater. Phys., 1995, 51, 13023–13032 Search PubMed.
- J. Pascual, J. Camassel and H. Mathieu, Resolved Quadrupolar Transition in TiO2, Phys. Rev. Lett., 1977, 39, 1490–1493 CrossRef CAS.
- J. Heyd, G. E. Scuseria and M. Ernzerhof, Hybrid functionals based on a screened Coulomb potential, J. Chem. Phys., 2003, 118, 8207–8215 CrossRef CAS.
- C. Freysoldt, B. Grabowski, T. Hickel, J. Neugebauer, G. Kresse, A. Janotti and C. G. Van de Walle, First-principles calculations for point defects in solids, Rev. Mod. Phys., 2014, 86, 253–305 CrossRef.
- S. Lany and A. Zunger, Accurate prediction of defect properties in density functional supercell calculations, Modell. Simul. Mater. Sci. Eng., 2009, 17, 084002 Search PubMed.
- J. M. Adamczyk, L. C. Gomes, J. Qu, G. A. Rome, S. M. Baumann, E. Ertekin and E. S. Toberer, Native Defect Engineering in CuInTe2, Chem. Mater., 2021, 33, 359–369 CrossRef CAS.
- O. W. Johnson, S.-H. Paek and J. W. DeFord, Diffusion of H and D in TiO2: Suppression of internal fields by isotope exchange, J. Appl. Phys., 1975, 46, 1026–1033 Search PubMed.
- R. Krishna, Uphill diffusion in multicomponent mixtures, Chem. Soc. Rev., 2015, 44, 2812–2836 RSC.
- W. Kern, in Handbook of Silicon Wafer Cleaning Technology, ed. K. A. Reinhardt and W. Kern, William Andrew Publishing, 3rd edn, 2018, pp. 3–85 Search PubMed.
- O. Berger, Understanding the fundamentals of TiO2 surfacesPart II. Reactivity and surface chemistry of TiO2 single crystals, Surf. Eng., 2022, 38, 846–906 Search PubMed.
- M. Králik, Adsorption, chemisorption, and catalysis, Chem. Pap., 2014, 68, 1625–1638 Search PubMed.
- A. P. St-Pierre, V. Shikon and D. C. Schneider, Count data in biology—Data transformation or model reformation?, Ecol. Evol., 2018, 8, 3077–3085 CrossRef PubMed.
- M. Bylesjö, O. Cloarec and M. Rantalainen, in Comprehensive Chemometrics, ed. S. Brown, R. Tauler and B. Walczak, Elsevier, Oxford, 2nd edn, 2009, pp. 101–114 Search PubMed.
- E. Vittinghoff, C. E. McCulloch, D. V. Glidden and S. C. Shiboski, in Handbook of Statistics, ed. C. R. Rao, J. P. Miller and D. C. Rao, Elsevier, 2007, vol. 27, pp. 148–186 Search PubMed.
- Q. Zhang, A. M. Schmidt and Y. P. Chaubey, Modeling left-censored skewed spatial processes: The case of arsenic drinking water contamination, Spat. Stat., 2024, 59, 100816 CrossRef.
- J. Li and A. D. Heap, Spatial interpolation methods applied in the environmental sciences: A review, Environ. Modell. Software, 2014, 53, 173–189 CrossRef.
- D. Ruppert, International Encyclopedia of the Social & Behavioral Sciences, Elsevier, 2001, pp. 15007–15014 Search PubMed.
- V. Corradi and N. R. Swanson, The effect of data transformation on common cycle, cointegration, and unit root tests: Monte Carlo results and a simple test, J. Econom., 2006, 132, 195–229 Search PubMed.
- M. J. Blanca, R. Alarcón, J. Arnau, R. Bono and R. Bendayan, Datos no normales: ¿es el ANOVA una opción válida?, Psicothema, 2017, 29, 552–557 Search PubMed.
- M. M. Ali and S. C. Sharma, Robustness to nonnormality of regression F-tests, J. Econom., 1996, 71, 175–205 Search PubMed.
- T. Holsclaw, K. A. Hallgren, M. Steyvers, P. Smyth and D. C. Atkins, Measurement error and outcome distributions: Methodological issues in regression analyses of behavioral coding data, Psychol. Addict. Behav., 2015, 29, 1031–1040 Search PubMed.
- M. P. Perme and D. Manevski, Confidence intervals for the Mann–Whitney test, Stat. Methods Med. Res., 2018, 28, 3755–3768 CrossRef PubMed.
- H. Jeong, E. G. Seebauer and E. Ertekin, First-principles description of oxygen self-diffusion in rutile TiO2: Assessment of uncertainties due to enthalpy and entropy contributions, Phys. Chem. Chem. Phys., 2018, 20, 17448–17457 RSC.
- H. Peng, First-principles study of native defects in rutile TiO2, Phys. Lett. A, 2008, 372, 1527–1530 CrossRef CAS.
- H.-Y. Lee, S. J. Clark and J. Robertson, Calculation of point defects in rutile TiO2 by the screened-exchange hybrid functional, Phys. Rev. B:Condens. Matter Mater. Phys., 2012, 86, 75209 CrossRef.
- L.-B. Mo, Y. Wang, Y. Bai, Q.-Y. Xiang, Q. Li, W.-Q. Yao, J.-O. Wang, K. Ibrahim, H.-H. Wang, C.-H. Wan and J.-L. Cao, Hydrogen Impurity Defects in Rutile TiO2, Sci. Rep., 2015, 5, 17634 CrossRef CAS PubMed.
- T. S. Bjørheim, S. Stølen and T. Norby, Ab initio studies of hydrogen and acceptor defects in rutile TiO2, Phys. Chem. Chem. Phys., 2010, 12, 6817–6825 RSC.
- P. W. Peacock and J. Robertson, Behavior of hydrogen in high dielectric constant oxide gate insulators, Appl. Phys. Lett., 2003, 83, 2025–2027 Search PubMed.
- E. V. Lavrov, T. Mchedlidze and F. Herklotz, Photoconductive detection of hydrogen in ZnO and rutile TiO2, J. Appl. Phys., 2016, 120, 055703 CrossRef.
- J. Weber, E. V. Lavrov and F. Herklotz, Hydrogen shallow donors in ZnO and rutile TiO2, Phys. B, 2012, 407, 1456–1461 CrossRef CAS.
- A. Janotti and C. G. Van de Walle, Native point defects in ZnO, Phys. Rev. B:Condens. Matter Mater. Phys., 2007, 76, 165202 CrossRef.
- J. L. Lyons, J. B. Varley, D. Steiauf, A. Janotti and C. G. Van de Walle, First-principles characterization of native-defect-related optical transitions in ZnO, J. Appl. Phys., 2017, 122, 035704 CrossRef.
- E. J. Skiba, H. B. Buckner, C. Lee, G. McKnight, R. F. Wallick, R. van der Veen, E. Ertekin and N. H. Perry, UV-Driven Oxygen Surface Exchange and Stoichiometry Changes in a Thin-Film, Nondilute Mixed Ionic Electronic Conductor, Sr(Ti,Fe)O3-d, J. Am. Chem. Soc., 2024, 146, 23265–23277 CrossRef CAS PubMed.
- K. Alberi and M. A. Scarpulla, Effects of excess carriers on charged defect concentrations in wide bandgap semiconductors, J. Appl. Phys., 2018, 123, 185702 CrossRef.
- R. Vaidyanathan, S. Felch, H. Graoui, M. A. Foad, Y. Kondratenko and E. G. Seebauer, Nonthermal illumination effects on ultra-shallow junction formation, Appl. Phys. Lett., 2011, 98, 194104 Search PubMed.
- E. G. Seebauer, M. Y. L. Jung, C. T. M. Kwok, R. Vaidyanathan and Y. V. Kondratenko, Measurement of photostimulated self-diffusion in silicon, J. Appl. Phys., 2011, 109, 103708 Search PubMed.
- J. Meng, M. S. Sheikh, R. Jacobs, J. Liu, W. O. Nachlas, X. Li and D. Morgan, Computational discovery of fast interstitial oxygen conductors, Nat. Mater., 2024, 23, 1252–1258 Search PubMed.
- I. M. Nadeem, C. Penschke, J. Chen, X. Torrelles, A. Wilson, H. Hussain, G. Cabailh, O. Bikondoa, J. Imran, C. Nicklin, R. Lindsay, J. Zegenhagen, M. O. Blunt, A. Michaelides and G. Thornton, Ultracompact Electrical Double Layers at TiO2(110) Electrified Interfaces, J. Am. Chem. Soc., 2024, 146, 33443–33451 CrossRef CAS PubMed.
- A. Selloni, Aqueous Titania Interfaces, Annu. Rev. Phys. Chem., 2024, 75, 47–65 Search PubMed.
- B. B. Nielsen, K. Tanderup, M. Budde, K. B. Nielsen, J. L. Lindström, R. Jones, S. Öberg, B. Hourahine and P. R. Briddon, Local Vibrational Modes of Weakly Bound O–H Complexes in SI, Mater. Sci. Forum, 1997, 258–263, 391–398 Search PubMed.
- W. Luo, P. B. Rasband, P. Clancy and B. W. Roberts, Tight-binding studies of the tendency for boron to cluster in c-Si. II. Interaction of dopants and defects in boron-doped Si, J. Appl. Phys., 1998, 84, 2476–2486 CrossRef CAS.
- J. Yu, X.-M. Bai, A. El-Azab and T. R. Allen, Oxygen transport in off-stoichiometric uranium dioxide mediated by defect clustering dynamics, J. Chem. Phys., 2015, 142, 94705 Search PubMed.
- J. M. Flitcroft, M. Molinari, N. A. Brincat, N. R. Williams, M. T. Storr, G. C. Allen and S. C. Parker, The critical role of hydrogen on the stability of oxy-hydroxyl defect clusters in uranium oxide, J. Mater. Chem. A, 2018, 6, 11362–11369 Search PubMed.
- G. Singh, M. Kaur, V. Kumar Garg and A. C. Oliveira, Oxygen hyper stoichiometric trimetallic titanium doped magnesium ferrite: Structural and photocatalytic studies, Ceram. Int., 2022, 48, 24476–24484 Search PubMed.
- J. K. Grewal, M. Kaur, R. K. Sharma, A. C. Oliveira, V. K. Garg and V. K. Sharma, Structural and Photocatalytic Studies on Oxygen Hyperstoichiometric Titanium-Substituted Strontium Ferrite Nanoparticles, Magnetochemistry, 2022, 8, 120 CrossRef CAS.
- J. K. Cooper, S. B. Scott, Y. Ling, J. Yang, S. Hao, Y. Li, F. M. Toma, M. Stutzmann, K. V. Lakshmi and I. D. Sharp, Role of hydrogen in defining the n-type character of BiVO4 photoanodes, Chem. Mater., 2016, 28, 5761–5771 CrossRef CAS.
- A. Lushchik, V. N. Kuzovkov, E. A. Kotomin, G. Prieditis, V. Seeman, E. Shablonin, E. Vasil’chenko and A. I. Popov, Evidence for the formation of two types of oxygen interstitials in neutron-irradiated α-Al2O3 single crystals, Sci. Rep., 2021, 11, 20909 CrossRef CAS PubMed.
- M. D. McCluskey, M. C. Tarun and S. T. Teklemichael, Hydrogen in oxide semiconductors, J. Mater. Res., 2012, 27, 2190–2198 CrossRef CAS.
- H. Li and J. Robertson, Behaviour of hydrogen in wide band gap oxides, J. Appl. Phys., 2014, 115, 203708 CrossRef.
- M. E. Ingebrigtsen, A. Y. Kuznetsov, B. G. Svensson, G. Alfieri, A. Mihaila, U. Badstübner, A. Perron, L. Vines and J. B. Varley, Impact of proton irradiation on conductivity and deep level defects in β-Ga2O3, APL Mater., 2019, 7, 22510 CrossRef.
- E. V. Lavrov, I. Chaplygin, F. Herklotz and V. V. Melnikov, Hydrogen Donors in Anatase TiO2, Phys. Status Solidi B, 2021, 258, 2100171 CrossRef CAS.
- E. V. Lavrov, I. Chaplygin, F. Herklotz, V. V. Melnikov and Y. Kutin, Hydrogen in single-crystalline anatase TiO2, J. Appl. Phys., 2022, 131, 030902 CrossRef CAS.
Footnote |
| † Current address: Department of Mechanical Engineering, Sogang University, Seoul 04107, South Korea. |
| This journal is © the Owner Societies 2026 |





