Open Access Article
Open Access ArticleThe superior catalytic effect of N vs. Ni for improving hydrogen storage kinetics of LiBH4@X-doped-C-MSU-H (X = N or Ni) nanoporous carbon composites
Petru
Palade
 *,
Catalin
Negrila
,
Anca G.
Mirea
,
Cristian
Radu
and
Cezar
Comanescu
*,
Catalin
Negrila
,
Anca G.
Mirea
,
Cristian
Radu
and
Cezar
Comanescu

National Institute of Materials Physics, Atomistilor Str. 405A, 077125, Magurele, Romania. E-mail: petru.palade@infim.ro
First published on 23rd February 2026
Abstract
Lithium borohydride (LiBH4) is a promising hydrogen storage material releasing 13.8 wt% H2 upon decomposition in lithium hydride and boron, significantly surpassing other complex hydrides. However, sluggish dehydrogenation kinetics still hinder the use for practical applications. The infiltration of LiBH4 into carbon nanoscaffolds has proved to be effective in improving the hydrogen absorption/desorption (a/d) kinetics. Further improvement of storage kinetics can be achieved by modification of the nanocarbon with dopant elements. The present work compares nanoporous carbon (C-MSU-H) and C-MSU-H doped either with 1 at% N or 1 at% Ni as the matrix for infiltration of LiBH4. The catalytic effect of nitrogen proved to be superior to that of nickel (keeping the same doping level) for improving the hydrogen a/d kinetics of LiBH4 infiltrated in doped C-MSU-H. X-ray photoelectron spectroscopy was used to detect the amount and chemical proximity of nitrogen in nanoporous carbon following the thermal treatment in ammonia flow. The morphology and porosity of doped C-MSU-H were investigated by X-ray diffraction, FTIR, TEM, and BET. Hydrogen a/d kinetics of LiBH4@C-MSU-H nanocomposites was investigated by a volumetric method. The desorption peak temperatures (measured at 2 °C min−1 rate) are 339 °C for the undoped LiBH4@C-MSU-H, 328 °C for the LiBH4@C-MSU-H doped with 1 at% Ni and 318 °C for the LiBH4@C-MSU-H doped with 1 at% N nanocomposites. The activation energies of hydrogen desorption for the investigated nanocomposites were obtained from Kissinger plots: 142.7 kJ mol−1 for undoped LiBH4@C-MSU-H, 123.8 kJ mol−1 for LiBH4@C-MSU-H 1 at% Ni and 119.5 kJ mol−1 for LiBH4@C-MSU-H 1 at% N nanocomposites. The catalytic effect on LiBH4 dehydrogenation due to N-doping of nanocarbons is discussed.
1. Introduction
Due to the greenhouse effect caused by the burning of fossil fuels, a top priority for today's society is the development of a non-polluting and sustainable economy. Moreover, fossil fuels could meet society's needs for a limited period of time. Hydrogen, as an energy carrier, represents a non-polluting energy alternative. Storing hydrogen in hydrides is a safe and inexpensive method. Metal hydrides have a hydrogen mass content below 2 wt% H2. Lithium borohydride, LiBH4, upon decomposition into LiH and B provides an amount of hydrogen equal to 13.8 wt% H2, well above the 7 wt% H2 obtained for magnesium hydride or sodium alanate NaAlH4.1 Unfortunately, for the rehydrogenation of LiBH4 after hydrogen desorption, very harsh conditions are required, namely 350 atm H2 at 600 °C.2The difficult absorption/desorption of hydrogen is caused by sluggish kinetics and thermodynamic barriers that negatively influence the dehydrogenation temperature. Significant efforts have been made to improve the hydrogenation/dehydrogenation kinetics of LiBH4 through the use of catalysts such as those described in very recent reviews.3,4 Another strategy for improving kinetics is nanoconfinement, by embedding borohydrides in various matrices that prevent the growth of hydride grains during hydrogen absorption/desorption cycles, adversely affecting hydrogen diffusion in the hydride bed. For this purpose, various matrices have been used: porous hollow carbon nanospheres,5 double-layered carbon nanobowls,6 silica and carbon scaffolds,7 nanoporous carbon,8,9 CoNi doped hollow carbon networks,10 carbon wrapped Fe3O4 nanospheres,11 TiO2 decorated porous carbonaceous networks,12 N-doped carbon nanosheets embedded with Co nanoparticles,13 and Ni nanoparticles coated with porous hollow carbon micropheres.14 Also mixing with carbon nanotubes15 or 2D structures like graphene doped with FeF2/FeOx16 or high entropy oxide nanoplates17 have proved to be useful in avoiding the growth of borohydride grains and improving hydrogenation/dehydrogenation kinetics. An interesting approach was to synthesize nanoparticles of LiBH4 supported on Ni doped graphene.18 Thermodynamic barriers can be reduced through thermodynamic destabilization, which means lowering the decomposition enthalpy by creating new compounds that stabilize the dehydrogenated state as an alternative to the decomposition reaction of LiBH4 into LiH and B.19 For LiBH4 containing composites, thermodynamic destabilization has been achieved through the systems: LiBH4/MgH2,19 LiBH4/LiAlH4,20 LiBH4/YH3,21 LiBH4/Mg2NiH4.22
The interaction between LiBH4 and the carbon matrix or between multi-component systems based on lithium borohydride was investigated using modern methods such as in situ X-ray Raman spectroscopy23 and QENS (quasielastic neutron scattering and neutron vibrational spectroscopy),24 whereas the identification of poorly crystallized phases and reaction mechanisms can be explored by 11B magic angle spinning (MAS) nuclear magnetic resonance.25
Both Ni catalysis and LiBH4 nanoconfinement have a synergistic effect on the reversible absorption/desorption of hydrogen.26 Nitrogen-doped nanoporous carbon has proven to be a very effective support for catalytic reactions or for hydrogen storage27,28 and has had a significant effect on improving the absorption/desorption kinetics of hydrogen for MgH2 confined in such matrices.29 Besides the physicochemical catalytic mechanism, carbon facilitates thermal conduction in the hydride mass (usually having low thermal conductivity), which enhances the hydrogen desorption kinetics (endothermic reaction).
Very recently, solar-driven reversible hydrogen storage was proposed as an efficient technique to generate H2 reversibly, without using any external heat source, except for solar radiation. MgH2 was catalyzed with Cu nanoparticles distributed on MXene nanosheets, having a dual effect: (i) photothermal effect under solar radiation and (ii) catalytic effect on hydrogenation/dehydrogenation of MgH2. Such a system can reversibly store 5.9 wt% H2 using only solar radiation as the heat source.30 A similar system using MgH2 catalyzed with flower-like microspheres made from N-doped TiO2 nanosheets coated with TiN nanoparticles has a hydrogen storage capacity of 6.1 wt% H2.31 Mg2Ni(Cu) and its hydrogenated compound exhibit intra/inter-band transitions generating 85% light absorption across the entire spectrum range raising the surface temperature of MgH2 to 261.8 °C under a light intensity of 2.6 W cm−2. Mg2Ni(Cu)/Mg2Ni(Cu)H4 acts as a light-enhanced “hydrogen pump” for MgH2 having both photothermal and catalytic effects, leading to a reversible hydrogen storage amount of 6.1 wt% H2 with 95% retention under 3.5 W cm−1.32 MgH2 catalyzed using a TiO2/Fe2O3 heterojunction photocatalyst releases 3.1 wt% H2 within 45 min under a light intensity of 1.78 W cm−2, exhibiting a dehydrogenation activation energy of 77.3 kJ mol−1.33 Among other hydrides, LiBH4 catalyzed using g-C3N4/Fe2O3 heterojunctions can desorb 2.70 wt% H2 very quickly within 1 min and 3.87 wt% in 30 min under a light intensity of 1.78 W cm−2.34 A Ni/ZrO2 catalyst derived from metal–organic frameworks has been proven useful both for solar-driven hydrogen storage and photothermal conversion of CO2 to CH4.35 Reversible solar-driven hydrogen absorption/desorption of 4.9 wt% H2 was achieved for the sodium cyclohexanolate/phenoxide pair with 99.9% conversion and selectivity. The initial dehydrogenation rate was 23.4 mmol H2 g−1 h−1 (i.e. 2 orders of magnitude higher than that obtained via thermocatalysis).36
The present work investigates the catalytic efficiency of nitrogen and respectively nickel as doping elements for ordered nanoporous carbon on the hydrogenation/dehydrogenation reaction of LiBH4@doped nanoporous carbon nanocomposites. Via thermal treatment of the ordered nanoporous carbon matrix in gaseous ammonia, nitrogen doping of carbon exhibiting a strong catalytic effect in improving the kinetics of the hydrogen absorption/desorption reactions is achieved. The amount of nitrogen and its chemical proximity in the structure of the nanoporous carbon were carefully investigated through X-ray photoelectron spectroscopy. The Ni-doped ordered nanoporous carbon was obtained by reduction of Ni-salts deposited within the nanocarbon structure. The modification of the nanoporous carbon structure via thermal treatment in ammonia was analyzed through morphological and porosity investigations. The present work presents a comparison between the catalytic efficiency of C–MSU-H doped with the same amount of either N or Ni for improving the hydrogenation/dehydrogenation kinetics of LiBH4 demonstrating the superiority of N over Ni. This will be very useful for replacing the critical or toxic metal in the hydrogenation/dehydrogenation catalyst. We have comparatively discussed the origin of the catalytic effect for Ni and N doping. To our knowledge, this is the first time such a comparison (keeping the same amount of N and Ni dopants) has been carried out.
2. Experimental
2.1 Synthesis of materials
In order to obtain functionalized nanoporous carbon–hydride nanocomposites, the nanoporous carbon was prepared as a replica of nanoporous silica with an ordered structure. Then it was functionalized by treating in gaseous ammonia or by depositing metal ions, followed by chemical reduction. Nanoporous silica with ordered hexagonal pores was obtained starting from cheap precursors (sodium silicate). To obtain MSU-H,37–39 sodium silicate solution (27% SiO2, 14% NaOH) (Sigma Aldrich, extra pure) and pluronic surfactant P123 (Sigma Aldrich, average Mn ∼ 5800 block copolymers PEG–PPG–PEG) were used in the stoichiometric molar ratio 1 SiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.017 P123
0.017 P123![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.83 CH3COOH
0.83 CH3COOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.78 NaOH
0.78 NaOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 230 H2O. Thus, 1.2 g of P123 were mixed with 10 mL of 1.0 M acetic acid solution and 10 mL of water. To this microemulsion were added 2.7 g of sodium silicate solution (27 wt% SiO2 and 14 wt% NaOH) and 30 mL H2O. The resulting product was stirred for 24 hours at ambient temperature. Subsequently, it was treated at 100 °C for 24 h. The product was filtered and dried and finally the remaining surfactant was removed by calcination in air at 550 °C for 5 h. The carbon replica of MSU-H, named in the following C-MSU-H,40 was made by impregnating sucrose solution into the MSU-H matrix in two reaction steps. In the first step, 4 g of nanoporous silica MSU-H were dispersed in a solution consisting of 20 g H2O and 0.64 g H2SO4 (the acid is the catalyst). Then, 5 g of sucrose (carbon source) were added to the formed mixture so that the solution filled the pores of MSU-H. The slurry was heated in an oven for 6 h at 100 °C followed by another 6 h at 160 °C so that the sucrose became polymerized. After that, in the second impregnation step 3.2 g of sucrose and 0.36 g of H2SO4 dissolved in 20 g of water were added. The heating in the oven was repeated for 6 h at 100 °C followed by another 6 h at 160 °C so that the sucrose became polymerized and completely filled the MSU-H pores. The product obtained was washed of impurities, filtered and subsequently calcined for 3 h at 400 °C and 4 h at 900 °C in a nitrogen flow (N2, purity 99.999%) to transform sucrose into carbon. Thereby, a carbon–SiO2 composite was obtained. The removal of SiO2 was done via treatment with an aqueous solution of 10% hydrofluoric acid and washing with double distilled water until neutral pH. A treatment in gaseous ammonia flow (NH3, purity 99.999%) with a rate of 100 ml min−1 for 4 h was performed on the nanoporous carbon C-MSU-H in order to achieve the doping with N atoms. Special precautions were taken the ammonia being neutralized at the outlet of the heating system. The treatment temperatures were 400 °C, 450 °C and 500 °C.
230 H2O. Thus, 1.2 g of P123 were mixed with 10 mL of 1.0 M acetic acid solution and 10 mL of water. To this microemulsion were added 2.7 g of sodium silicate solution (27 wt% SiO2 and 14 wt% NaOH) and 30 mL H2O. The resulting product was stirred for 24 hours at ambient temperature. Subsequently, it was treated at 100 °C for 24 h. The product was filtered and dried and finally the remaining surfactant was removed by calcination in air at 550 °C for 5 h. The carbon replica of MSU-H, named in the following C-MSU-H,40 was made by impregnating sucrose solution into the MSU-H matrix in two reaction steps. In the first step, 4 g of nanoporous silica MSU-H were dispersed in a solution consisting of 20 g H2O and 0.64 g H2SO4 (the acid is the catalyst). Then, 5 g of sucrose (carbon source) were added to the formed mixture so that the solution filled the pores of MSU-H. The slurry was heated in an oven for 6 h at 100 °C followed by another 6 h at 160 °C so that the sucrose became polymerized. After that, in the second impregnation step 3.2 g of sucrose and 0.36 g of H2SO4 dissolved in 20 g of water were added. The heating in the oven was repeated for 6 h at 100 °C followed by another 6 h at 160 °C so that the sucrose became polymerized and completely filled the MSU-H pores. The product obtained was washed of impurities, filtered and subsequently calcined for 3 h at 400 °C and 4 h at 900 °C in a nitrogen flow (N2, purity 99.999%) to transform sucrose into carbon. Thereby, a carbon–SiO2 composite was obtained. The removal of SiO2 was done via treatment with an aqueous solution of 10% hydrofluoric acid and washing with double distilled water until neutral pH. A treatment in gaseous ammonia flow (NH3, purity 99.999%) with a rate of 100 ml min−1 for 4 h was performed on the nanoporous carbon C-MSU-H in order to achieve the doping with N atoms. Special precautions were taken the ammonia being neutralized at the outlet of the heating system. The treatment temperatures were 400 °C, 450 °C and 500 °C.
Besides nitrogen doping, another functionalization procedure of C-MSU-H involved doping with nickel ions. A level of 1 at% Ni doping of nanoporous carbon was envisaged, which meant 4.65 wt% Ni in carbon in order to keep the same doping level previously obtained for N doped C-MSU-H. For this purpose, 0.233 g of nickel nitrate hexahydrate Ni(NO3)2·6H2O (Sigma Aldrich > 99.9% trace metal basis) (corresponding to 0.047 g of Ni) dissolved in 10 ml of absolute ethanol was used to impregnate 1 g nanoporous carbon by the incipient wetness method (6 steps). Using this method we obtained a loading of 4.65 wt% Ni/nanoporous carbon (respectively 1 at% Ni/nanoporous carbon). After drying at 70 °C for 10 hours, the nanoporous carbon impregnated with an ethanol solution of Ni(NO3)2·6H2O was treated in a 5% H2/Ar (purity 99.999%) flow (100 ml min−1) for 4 h at 430 °C. Subsequently, nanoporous carbon doped either with N or Ni was mixed with LiBH4 in a proportion of 50/50 wt% and the nanocomposites were synthesized by the melt infiltration under hydrogen pressure of 100 atm at 300 °C, so that LiBH4 (melting temperature 278 °C) infiltrates into the ordered pores of doped C-MSU-H. This method produces LiBH4@(Ni or N) doped C-MSU-H nanocomposites, avoiding the growth of LiBH4 grains during hydrogen absorption/desorption cycles, which would worsen the hydrogenation/dehydrogenation kinetics. Additionally, the heat transfer in the hydride mass was improved. The processing of hydrides was done in a Labstar Mbraun glove box (Garching, Germany) in a controlled atmosphere (<1 ppm of oxygen and moisture) in order to avoid sample contamination.
2.2 Characterization methods
X-ray diffraction measurements were performed using an Anton Parr diffractometer with Cu K-alpha radiation starting from very small angles for pristine ordered nanoporous carbon and respectively nitrogen doped samples with a step of 0.02° in the 2θ range of 0.1–60°. XRD measurements were performed in the 2θ range of 15–80° for LiBH4@ordered nanoporous carbon nanocomposites.FTIR measurements were performed on pressed pellets containing 120 mg KBr and the IR-active material. To avoid contamination, the powders were ground in a MBraun Labstar glove box. Measurements were performed in the wavenumber range of 300–4000 cm−1 using a JASCO-6600 FTIR spectrometer with a measuring step of 4 cm−1.
Nitrogen physisorption analysis was performed at −196 °C using a Micromeritics 3FLEX apparatus. Before determining the surface area, all powders were degassed at 200 °C for 4 h. The Brunauer–Emmett–Teller (BET) formalism was used to calculate the specific surface area from the data acquired at P/P0 between 0.025 and 0.3. The pore size distribution of the samples was determined from the desorption branch of the N2 isotherm. The pore size and volume analysis were calculated using the Barrett–Joyner–Halenda (BJH) formalism.
XPS (X-ray photoelectron spectroscopy) measurements were performed using a SPECS spectrometer based on a PHOIBOS 150 analyzer with a monochromatic X-ray source of 300 W with Al Kα radiation—1486.61 eV. Charge compensation was done using a Specs FG15/40 flood gun. The XPS core level lines were recorded using a pass energy of 20 eV while the survey spectra were recorded with a pass energy of 50 eV. The fittings were performed using Spectral Data Processor software using Voigt functions and usual relative sensitivity factors.
TEM images were obtained using a JEM-2100 analytical transmission electron microscope (Jeol, Tokyo, Japan) working at 200 kV endowed with a dispersive X-ray spectrometer. Prior to TEM investigation the samples were dispersed in hexane using an ultrasonic device with high power (VCX 750 Sonics, Newton, CT, USA), followed by drying onto Lacey carbon-coated Cu TEM grids.
Hydrogen storage kinetics measurements were performed using a commercially available Sievert (volumetric) apparatus built by Advanced Material Corporation, Pittsburgh, USA.
3. Results and discussion
3.1 X-ray diffraction
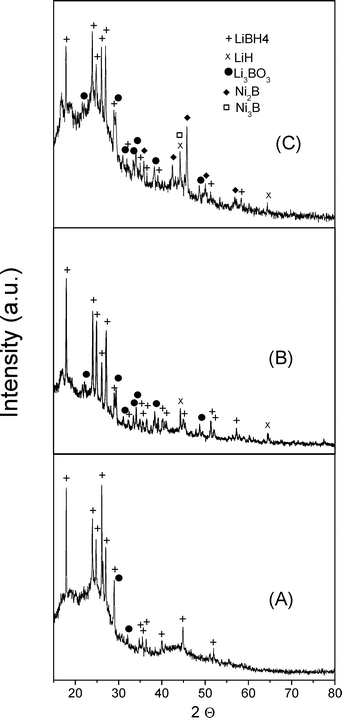
For the pristine ordered nanoporous carbon C-MSU-H and nanoporous carbon samples thermally treated in ammonia flow at 400 °C and 500 °C a hexagonal structure is observed with peaks at very low angles in the X-ray diffraction pattern provided in Fig. 1. The reflections at (1 0 0), (1 1 0) and (2 0 0) indicate a hexagonal lattice constant a = 10.9 nm. The peaks (0 0 2) and (1 0 0) from 2θ of about 23° and 43°, respectively, correspond to a graphitic phase in a very small amount with a lattice constant close to that of pure graphite. This phase appears in a significantly higher amount for the sample treated at 500 °C. The initially ordered nanoporous carbon C-MSU-H was functionalized either by treatment in a flow of gaseous ammonia or by doping with Ni ions. The XRD pattern of C-MSU-H doped with 1 at% Ni (Fig. 1D) exhibits differences from the undoped one due to the presence of very broad peaks corresponding to Ni and NiO which show very small crystallite size of a few nanometers. | ||
| Fig. 1 X-ray diffraction spectra of ordered nanoporous carbon C-MSU-H: (A) as obtained, (B) treated in ammonia gas at 400 °C, (C) treated in ammonia gas at 500 °C and (D) C-MSU-H doped with 1 at% Ni. | ||
The X-ray diffraction spectra for the re-hydrogenated (after cycles of hydrogen absorption/desorption) nanocomposites – 50 wt% LiBH4 – 50 wt% C-MSU-H (named LiBH4@C-MSU-H) (Fig. 2A) and 50 wt% LiBH4 – 50 wt% C-MSU-H doped with 1 at% N (named LiBH4@C-MSU-H 1 at% N) (nitrogen doping level was measured from XPS) (Fig. 2B) are very similar, and show mainly LiBH4 (ICDD file 01-084-8599) and a small amount of LiH (ICDD file 04-013-9487) according to the hydrogen generation reaction:
| LiBH4 ↔ LiH + B + 3/2H2 | (1) |
Boron cannot be detected in the XRD pattern due to amorphous state.
A relatively small amount of Li3BO3 (ICDD file 00-018-0718) can be seen in the XRD pattern, probably appeared during the diffraction measurements, although we mounted a protective foil over the sample. The most important amorphous contribution is due to LiBH4 at 2θ ≈ 25°, but some amount of LiBH4 crystallizes outside of the ordered nanoporous carbon matrix during the hydrogen absorption/desorption cycles. The X-ray diffraction spectrum for the rehydrogenated composites 50 wt% LiBH4 – 50 wt% C-MSU-H doped with 1 at% Ni (named LiBH4@C-MSU-H 1 at% Ni) (Fig. 2C) presents novel elements compared to the previous ones. Thus, the presence of nickel borides with different percentages of B is observed, namely Ni3B (ICDD file 00-048-1223) and Ni2B (ICDD file 00-048-1222) in addition to the LiBH4, LiH and Li3BO3 phases. There is no evidence of metallic Ni or NiO in the XRD pattern, probably due to the reduction effect of LiBH4 and the affinity of Ni to the reaction with boron provided from LiBH4.
3.2 FTIR spectroscopy
The measurements were performed in the wavenumber range of 300–4000 cm−1 using a JASCO-6600 FTIR spectrometer with a measuring step of 4 cm−1 for both the initial C-MSU-H and LiBH4 and for the LiBH4@C-MSU-H and LiBH4@C-MSU-H 1 at% N rehydrogenated nanocomposites as shown in Fig. 3.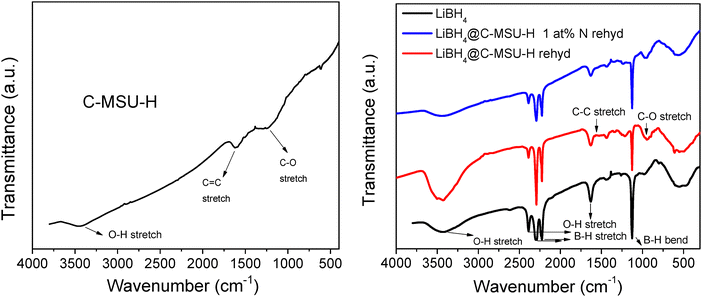 | ||
| Fig. 3 FTIR spectra of C-MSU-H and the original LiBH4 and LiBH4@C-MSU-H and LiBH4@C-MSU-H 1 at% N rehydrogenated nanocomposites. | ||
The band at 1568 cm−1 corresponds to the stretching mode of the carbon atoms in the aromatic ring (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) that exists in C-MSU-H. The band at 1036 cm−1 in C-MSU-H corresponds to the stretching mode for the C–O interaction, and the band at 3445 cm−1 can be assigned to the stretching mode for the O–H bond that appears due to hygroscopicity of KBr.
C) that exists in C-MSU-H. The band at 1036 cm−1 in C-MSU-H corresponds to the stretching mode for the C–O interaction, and the band at 3445 cm−1 can be assigned to the stretching mode for the O–H bond that appears due to hygroscopicity of KBr.
The vibrational bands at 2390, 2294 and 2226 cm−1 present in LiBH4@C-MSU-H correspond to the stretching mode for the B–H bond in LiBH4. The band at 1126 cm−1 can be assigned to the bending B–H vibration in the original LiBH4 and LiBH4@C-MSU-H re-hydrogenated nanocomposites. In LiBH4@C-MSU-H nanocomposites, bands corresponding to the O–H (3440 cm−1), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (1572 cm−1) and C–O (1004 cm−1) stretching vibrations also appear, originating from the initial C-MSU-H. Similar to X ray-diffraction, the FTIR spectra proved that after re-hydrogenation the LiBH4 phase is recovered. In the FTIR spectra of the rehydrogenated nanocomposites (Fig. 3) there is no evidence of Li2B12H12 whose distinctive feature would be the band at 2485 cm−1. Borane is not released below 450 °C for LiBH4 infiltrated in nanoporous carbon.41 We consider that the incomplete hydrogenation is caused by a kinetic barrier. The FTIR spectrum of LiBH4@C-MSU-H is very similar to that of LiBH4@C-MSU-H 1 at% N reydrogenated nanocomposites. There is no obvious change in the FTIR spectra due to the presence of N. This can be understood as being due to the very low nitrogen content in the sample. As expected, the XRD pattern of LiBH4@C-MSU-H 1 at% N is very similar to that of undoped LiBH4@C-MSU-H (Fig. 2). The reason why the presence of nickel borides can be detected in the XRD pattern of LiBH4@C-MSU-H 1 at% Ni rehydrogenated nanocomposites is the much higher atomic scattering factor of Ni compared to that of N, C, B and Li. However, we performed N 1s HRXPS (with a noisy spectrum due to very low N content) on LiBH4@C-MSU-H 1 at% N rehydrogenated sample showing the presence of Li3N/Li3BN2.
C (1572 cm−1) and C–O (1004 cm−1) stretching vibrations also appear, originating from the initial C-MSU-H. Similar to X ray-diffraction, the FTIR spectra proved that after re-hydrogenation the LiBH4 phase is recovered. In the FTIR spectra of the rehydrogenated nanocomposites (Fig. 3) there is no evidence of Li2B12H12 whose distinctive feature would be the band at 2485 cm−1. Borane is not released below 450 °C for LiBH4 infiltrated in nanoporous carbon.41 We consider that the incomplete hydrogenation is caused by a kinetic barrier. The FTIR spectrum of LiBH4@C-MSU-H is very similar to that of LiBH4@C-MSU-H 1 at% N reydrogenated nanocomposites. There is no obvious change in the FTIR spectra due to the presence of N. This can be understood as being due to the very low nitrogen content in the sample. As expected, the XRD pattern of LiBH4@C-MSU-H 1 at% N is very similar to that of undoped LiBH4@C-MSU-H (Fig. 2). The reason why the presence of nickel borides can be detected in the XRD pattern of LiBH4@C-MSU-H 1 at% Ni rehydrogenated nanocomposites is the much higher atomic scattering factor of Ni compared to that of N, C, B and Li. However, we performed N 1s HRXPS (with a noisy spectrum due to very low N content) on LiBH4@C-MSU-H 1 at% N rehydrogenated sample showing the presence of Li3N/Li3BN2.
3.3 X-ray photoelectron spectroscopy
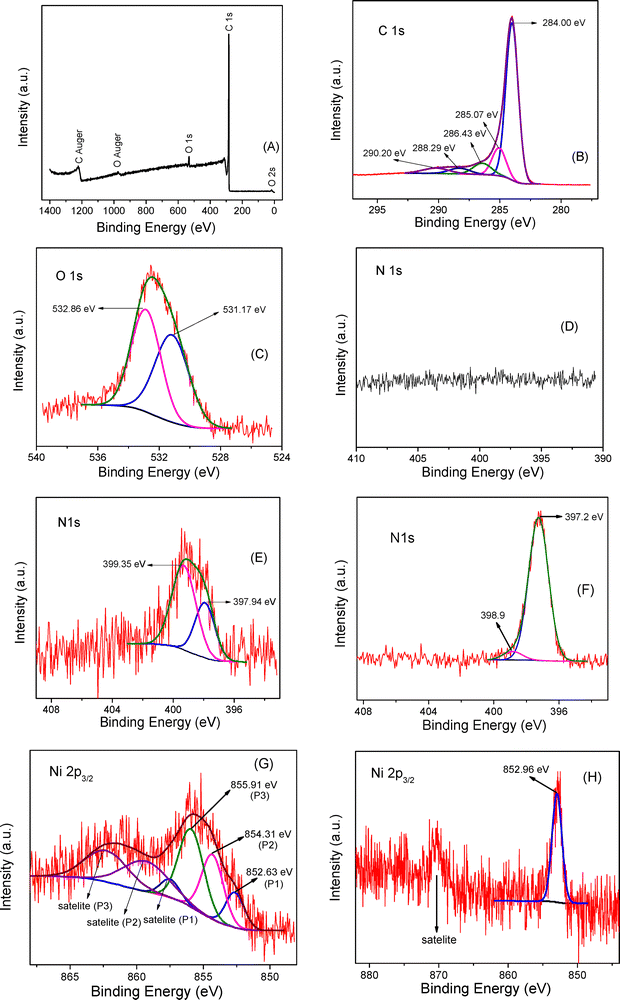
We performed X-ray photoelectron spectroscopy on C-MSU-H undoped and doped with nitrogen by thermal treatment under ammonia flow in order to find the amount and chemical state of N embedded in the carbon nanostructure (Table 1). We also performed XPS on Ni doped samples.| Sample | C (at%) | O (at%) | N (at%) |
|---|---|---|---|
| C-MSU-H pristine | 96.5 | 3.5 | — |
| C-MSU-H treated 4 h NH3 at 400 °C | 96.8 | 2.6 | 0.6 |
| C-MSU-H treated 4 h NH3 at 450 °C | 96.9 | 2.4 | 0.7 |
| C-MSU-H treated 4 h NH3 at 500 °C | 96.5 | 2.6 | 0.9 |
The reference energy was taken as the C 1s line C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds, which we set at 284 eV. The spectra from each sample were calibrated according to this reference. The samples have very few contaminants. Other elements, apart from C, O and N, were not observed in the samples. Monotonic increase in the amount of nitrogen is observed with increasing treatment temperature in NH3 (about 1 at% N doping for 4 h NH3 at 500 °C). A decrease in the O content is observed following treatment in NH3. In Fig. 4A–D are depicted the extended XPS spectrum and HRXPS C 1s, O 1s and N 1s spectra for nanoporous carbon sample without N doping. In C 1s HRXPS spectrum (Fig. 4B and Table 2) the binding energy of 284.00 eV corresponds to C-sp2 from the hexagonal carbon ring from nanoporous ordered structure, binding energy of 285.07 eV can be assigned to C–C, C–H, single bonds, binding energy of 286.43 eV indicates very likely C–O–C bonds, binding energy of 288.29 eV indicates C
C bonds, which we set at 284 eV. The spectra from each sample were calibrated according to this reference. The samples have very few contaminants. Other elements, apart from C, O and N, were not observed in the samples. Monotonic increase in the amount of nitrogen is observed with increasing treatment temperature in NH3 (about 1 at% N doping for 4 h NH3 at 500 °C). A decrease in the O content is observed following treatment in NH3. In Fig. 4A–D are depicted the extended XPS spectrum and HRXPS C 1s, O 1s and N 1s spectra for nanoporous carbon sample without N doping. In C 1s HRXPS spectrum (Fig. 4B and Table 2) the binding energy of 284.00 eV corresponds to C-sp2 from the hexagonal carbon ring from nanoporous ordered structure, binding energy of 285.07 eV can be assigned to C–C, C–H, single bonds, binding energy of 286.43 eV indicates very likely C–O–C bonds, binding energy of 288.29 eV indicates C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds, whereas binding energy of 290.20 eV can be assigned to specific shake-up C sp2-π–π* transition.42–44 In HRXPS O 1s spectrum (Fig. 4C and Table 3) the binding energy of 531.17 eV indicates molecular O2 adsorbed or C
O double bonds, whereas binding energy of 290.20 eV can be assigned to specific shake-up C sp2-π–π* transition.42–44 In HRXPS O 1s spectrum (Fig. 4C and Table 3) the binding energy of 531.17 eV indicates molecular O2 adsorbed or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds, whereas the binding energy of 532.86 eV can be assigned to C–O single bonds.44 In HRXPS N 1s spectrum for C-MSU-H – NH3 (4 h, 500 °C) (sample doped with ≈ 1 at% N) (Fig. 4E and Table 4) the binding energy of 397.94 eV indicates N in the pyridinic position whereas the binding energy of 399.35 can be assigned to N in the pyrrolic position.42,45
O double bonds, whereas the binding energy of 532.86 eV can be assigned to C–O single bonds.44 In HRXPS N 1s spectrum for C-MSU-H – NH3 (4 h, 500 °C) (sample doped with ≈ 1 at% N) (Fig. 4E and Table 4) the binding energy of 397.94 eV indicates N in the pyridinic position whereas the binding energy of 399.35 can be assigned to N in the pyrrolic position.42,45
| Sample | C-sp2 | C–C, C–H, C–N | C–O–C | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
C sp2-π–π* shake up | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| BE | Conc. | BE | Conc. | BE | Conc. | BE | Conc. | BE | Conc. | |
| (eV) | (at%) | (eV) | (at%) | (eV) | (at%) | (eV) | (at%) | (eV) | (at%) | |
| C-MSU-H | 284.0 | 69.70 | 285.07 | 13.70 | 286.43 | 7.40 | 288.29 | 4.20 | 290.2 | 5.00 |
| C-MSU-H | 283.9 | 69.60 | 285.15 | 13.00 | 286.48 | 7.40 | 288.42 | 5.00 | 290.4 | 5.10 |
| 4 h/400 °C | ||||||||||
| C-MSU-H | 284.0 | 73.80 | 285.00 | 13.90 | 286.16 | 5.00 | 287.48 | 3.00 | 289.6 | 4.30 |
| 4 h/450 °C | ||||||||||
| C-MSU-H | 284.0 | 76.30 | 285.18 | 12.80 | 286.60 | 5.50 | 288.48 | 2.90 | 290.1 | 2.40 |
| 4 h/500 °C | ||||||||||
| Sample | O ads, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
C–O | ||
|---|---|---|---|---|
| BE | Conc. | BE | Conc. | |
| (eV) | (at%) | (eV) | (at%) | |
| C-MSU-H | 531.17 | 49.60 | 532.86 | 50.40 |
| C-MSU-H 4 h/400 °C | 530.88 | 41.00 | 532.82 | 59.00 |
| C-MSU-H 4 h/450 °C | 531.11 | 25.40 | 532.61 | 74.60 |
| C-MSU-H 4 h/500 °C | 531.06 | 36.40 | 532.65 | 63.60 |
| Sample | N-pyridinic | N-pyrrolic | ||
|---|---|---|---|---|
| BE | Conc. | BE | Conc. | |
| (eV) | (at%) | (eV) | (at%) | |
| C-MSU-H | — | — | — | — |
| C-MSU-H 4 h/400 °C | 398.00 | 17.10 | 399.15 | 82.90 |
| C-MSU-H 4 h/450 °C | 397.92 | 34.90 | 399.30 | 65.10 |
| C-MSU-H 4 h/500 °C | 397.94 | 39.60 | 399.35 | 60.40 |
An increase in the C-sp2 content is observed with increasing treatment temperature in ammonia flow for C-MSU-H (Table 2). A decrease in the content of adsorbed oxygen and/or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds and an increase in the content of C–O bonds is observed following the thermal treatment in NH3 (Table 3). An increase in the N-pyridinic content and a decrease in the N-pyrrolic content are observed as a result of increasing the treatment temperature in gaseous ammonia (Table 4). However, C-MSU-H thermally treated in NH3 samples have a majority of nitrogen atoms into pyrrolic configurations. The nitrogen atoms incorporated into graphitic, pyridinic, or pyrrolic configurations modify the local electronic density and create electron-rich regions that can interact strongly with the metal cations of the hydride (e.g., Li+, Na+, Mg2+). These interactions lead to polarization of the Li–H bonds within the hydride, weakening them and lowering the activation energy for hydrogen desorption.46 Consequently, N-doped carbons not only provide physical confinement but also exert a catalytic effect on hydrogen release. The degree of doping and the type of nitrogen species play a decisive role in tuning these effects: graphitic N enhances conductivity and stabilizes the framework, while pyridinic and pyrrolic N sites provide strong chemical anchoring and catalytic reactivity. The theoretical simulations further demonstrate that N-doped sp2/sp3 hybrid carbon can migrate the Fermi level to the conduction band, leading to an n-type conductivity due to the additional electrons attributed to the N dopant.47 Particularly, pyridinic nitrogen from C-MSU-H thermally treated in ammonia has a strong catalytic effect. In this state, the N atom is sp2 hybridized with carbon atoms from the 2D hexagonal aromatic ring, participating in one π and two σ bonds with the nearest neighboring carbon atoms, providing a lone pair of electrons available for additional bonding. Pyridinic nitrogen embedded in the nanoporous carbon scaffold could act as a Lewis base and donate significant electron density to the confined hydride layered onto the scaffold's surface with an effect on hydride thermodynamic destabilization. XPS data confirm the pyridinic and pyrrolic nitrogen in our prepared N-doped C-MSU-H nanocarbons and the catalytic activity of such supports is expected to be higher than that of neat C-MSU-H as we will further notice from hydrogen storage behavior. The HRXPS N 1s spectrum of 50 wt% LiBH4–50 wt% 1 at% N-doped C-MSU-H rehydrogenated nanocomposites (noted as LiBH4@C-MSU-H 1 at% N) is depicted in Fig. 4F. The majority peak corresponding to binding energy of 397.2 eV can be attributed to the formation of Li3N or Li3BN2, both having binding energies close to 397 eV.48–50 The binding energy given in the literature for Li2NH is 398.2 eV,51 which is significantly higher than that of our experimental value. In the Ni 2p3/2 HRXPS spectrum for 1 at% Ni-doped C-MSU-H (Fig. 4G) the binding energy of 852.63 eV corresponds to Ni metal, the binding energy of 854.31 eV can be assigned to the Ni2+ oxidation state which belongs to NiO, and the binding energy of 855.91 eV corresponds to a higher oxidation state of Ni4+ which can be assigned to NiO2. In Ni 2p3/2 HRXPS for LiBH4@C-MSU-H 1 at% Ni re-hydrogenated nanocomposites (Fig. 4H) the binding energy of 852.96 eV can be assigned to Ni–B bonds.52,53
O bonds and an increase in the content of C–O bonds is observed following the thermal treatment in NH3 (Table 3). An increase in the N-pyridinic content and a decrease in the N-pyrrolic content are observed as a result of increasing the treatment temperature in gaseous ammonia (Table 4). However, C-MSU-H thermally treated in NH3 samples have a majority of nitrogen atoms into pyrrolic configurations. The nitrogen atoms incorporated into graphitic, pyridinic, or pyrrolic configurations modify the local electronic density and create electron-rich regions that can interact strongly with the metal cations of the hydride (e.g., Li+, Na+, Mg2+). These interactions lead to polarization of the Li–H bonds within the hydride, weakening them and lowering the activation energy for hydrogen desorption.46 Consequently, N-doped carbons not only provide physical confinement but also exert a catalytic effect on hydrogen release. The degree of doping and the type of nitrogen species play a decisive role in tuning these effects: graphitic N enhances conductivity and stabilizes the framework, while pyridinic and pyrrolic N sites provide strong chemical anchoring and catalytic reactivity. The theoretical simulations further demonstrate that N-doped sp2/sp3 hybrid carbon can migrate the Fermi level to the conduction band, leading to an n-type conductivity due to the additional electrons attributed to the N dopant.47 Particularly, pyridinic nitrogen from C-MSU-H thermally treated in ammonia has a strong catalytic effect. In this state, the N atom is sp2 hybridized with carbon atoms from the 2D hexagonal aromatic ring, participating in one π and two σ bonds with the nearest neighboring carbon atoms, providing a lone pair of electrons available for additional bonding. Pyridinic nitrogen embedded in the nanoporous carbon scaffold could act as a Lewis base and donate significant electron density to the confined hydride layered onto the scaffold's surface with an effect on hydride thermodynamic destabilization. XPS data confirm the pyridinic and pyrrolic nitrogen in our prepared N-doped C-MSU-H nanocarbons and the catalytic activity of such supports is expected to be higher than that of neat C-MSU-H as we will further notice from hydrogen storage behavior. The HRXPS N 1s spectrum of 50 wt% LiBH4–50 wt% 1 at% N-doped C-MSU-H rehydrogenated nanocomposites (noted as LiBH4@C-MSU-H 1 at% N) is depicted in Fig. 4F. The majority peak corresponding to binding energy of 397.2 eV can be attributed to the formation of Li3N or Li3BN2, both having binding energies close to 397 eV.48–50 The binding energy given in the literature for Li2NH is 398.2 eV,51 which is significantly higher than that of our experimental value. In the Ni 2p3/2 HRXPS spectrum for 1 at% Ni-doped C-MSU-H (Fig. 4G) the binding energy of 852.63 eV corresponds to Ni metal, the binding energy of 854.31 eV can be assigned to the Ni2+ oxidation state which belongs to NiO, and the binding energy of 855.91 eV corresponds to a higher oxidation state of Ni4+ which can be assigned to NiO2. In Ni 2p3/2 HRXPS for LiBH4@C-MSU-H 1 at% Ni re-hydrogenated nanocomposites (Fig. 4H) the binding energy of 852.96 eV can be assigned to Ni–B bonds.52,53
3.4 TEM investigation
The transmission electron microscopy image for as prepared C-MSU-H sample is presented in Fig. 5A. The size of the hexagonal nanopores of the ordered structure is approximately 4 nm. It can also be observed that in the case of the C-MSU-H thermally treated in NH3 flow at 500 °C (Fig. 5B), the ordered structure remains similar to the untreated C-MSU-H sample, with hexagonal nanopores of almost the same size. In Fig. 5C are shown TEM images at various magnifications for C-MSU-H decorated with Ni nanoparticles. The samples indicate an ordered nanoporous carbon structure with uniformly distributed Ni/NiO clusters, without agglomeration. In the EDAX spectrum (Fig. 5D) taken over a large area where nanoparticles are present, a maximum corresponding to nickel is observed and the composition of the nanoparticles is confirmed. The nanoparticles are uniformly distributed in the structure, as can be seen from Fig. 5C. The histogram corresponding to their size distribution and the lognormal fit is presented in Fig. 5E, indicating a mean diameter of 5.3 nm.The rehydrogenated sample LiBH4@C-MSU-H 1 at% Ni consists of large amorphous particles containing well-dispersed nanoparticles (Fig. 5F). EDX chemical mapping of the amorphous particles reveals a uniform distribution of B and C, while the nanoparticles are rich in Ni. The nickel distribution appears to be concentrated in the nanoparticles, and the speckles appearing on maps outside of them can be attributed to noise. The rehydrogenated LiBH4@C-MSU-H 1 at% N sample consists of large amorphous particles with irregular shapes that do not show any special features (Fig. 5G). EDX chemical mapping reveals a uniform distribution of B, N, and C throughout the surface of the investigated particles, although the concentration of N is low, close to the detection quantification limit.
3.5 Porosity measurements
The porosity parameters of the samples are summarized in Table 5. The surface area calculated using the BET formalism for the samples C-MSU-H and C–MSU-NH3 (4 h, 500 °C) is 1264 m2 g−1 and, respectively, 1198 m2 g−1 indicating that the ordered nanoporous structure is maintained after the treatment at 500 °C in ammonia gas.| Sample | Surf. area | Surf. area | Surf. area | Pore vol | Pore vol | Pore diam. | Pore diam. |
|---|---|---|---|---|---|---|---|
| BET | BJH ads | BJH des | BJH ads | BJH des | BJH ads | BJH des | |
| (m2 g−1) | (m2 g−1) | (m2 g−1) | (cm3 g−1) | (cm3 g−1) | (nm) | (nm) | |
| C-MSU-H | 1264 | 1126 | 1197 | 1.158 | 1.288 | 4.113 | 4.1057 |
| C-MSU-H | 1198 | 1073 | 1131 | 1.051 | 1.105 | 3.915 | 3.9078 |
| NH3, 4 h, 500 °C |
The isothermal N2 adsorption–desorption plots, shown in Fig. 6(A and C), have isotherms of type IV shape. The pore volume and average pore diameter for the pristine C-MSU-H and C–MSU-NH3 (4 h, 500 °C) are presented in Fig. 6(B and D) and Table 5 and the values obtained indicate once again the preservation of the nanostructure of the sample after the treatment at 500 °C in NH3. The combination of nanoporosity and N-doping offers synergistic benefits. The nanoporous network ensures that hydrides are spatially confined, maximizing surface contact with the doped carbon walls, while the doped sites locally destabilize the hydride, effectively lowering both the desorption enthalpy and activation energy. This results in improved hydrogen release at significantly lower temperatures compared to bulk hydrides. Additionally, the carbon host enhances thermal conductivity with positive effect on hydrogen desorption which requires external heating because it is an endothermic reaction and mitigates phase segregation or irreversible decomposition during cycling.
 | ||
| Fig. 6 N2 adsorption/desorption isotherms and pore size distributions for as-prepared C-MSU-H (A) and (B) and C-MSU-H thermally treated in ammonia flow at 500 °C (C) and (D). | ||
3.6 Hydrogen storage behavior
Hydrogen absorption–desorption measurements were taken using an automated volumetric apparatus (Sievert). We performed isothermal kinetic hydrogen absorption and desorption and thermal programmed desorption measurements. In Fig. 7A, it can be observed that a large amount of hydrogen is released after the first desorption. The theoretical amount of hydrogen reversibly stored in the reaction LiBH4 ↔ LiH + B + 3/2H2 is 13.8 wt% H2. The weight ratio between LiBH4 and C-MSU-H was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
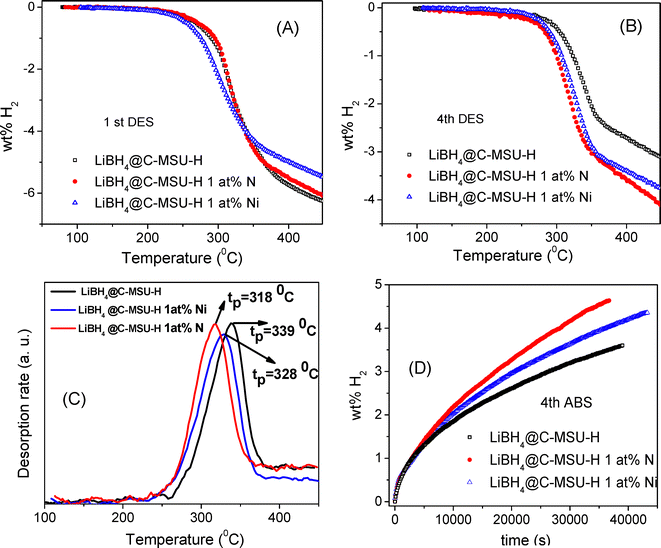
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. That means that the theoretical amount of hydrogen reversibly stored by LiBH4@C-MSU-H should be 6.9 wt% H2. However, the theoretical reversible amount should be approximately 6.5–6.7 wt% H2 (less than 6.9 wt% H2) because nickel borides or Li3N/Li3BN2 are also formed in the rehydrogenated nanocomposites containing either Ni or N. By making nanocomposites of LiBH4 infiltrate under H2 pressure in ordered nanoporous carbon, the aim is to avoid the growth of LiBH4 grains following the hydrogen absorption–desorption cycles, which would lead to a drastic deterioration of the hydrogen absorption–desorption kinetics. LiBH4@C-MSU-H desorbs 6.2 wt% H2 up to 450 °C for the first desorption with a temperature ramp of 2 °C min−1 (Fig. 7A). For complete desorption a much longer desorption time at 450 °C is required. Due to the presence of 1 at% Ni or N as doping elements, the LiBH4@doped C-MSU-H nanocomposites should desorb less than the theoretical amount of 6.9 wt% H2. LiBH4@C-MSU-H 1 at% Ni desorbs up to 450 °C, the least among all the samples, approximately 5.3 wt% H2 for the first desorption. After four hydrogen absorption–desorption cycles LiBH4@C-MSU-H releases only 3.1 wt% H2 up to 450 °C (Fig. 7B). LiBH4–C-MSU-H 1 at% N and LiBH4–C-MSU-H 1 at% Ni samples desorb 4.1 and 3.8 wt% H2 up to 450 °C, respectively, using the same temperature ramp of 2 °C min−1 (Fig. 7B). For complete desorption, a much longer desorption time at 450 °C is required. Faster desorption kinetics is observed for the N-doped sample compared to Ni-doped sample, but both doped samples have much faster desorption kinetics than the sample containing undoped C-MSU-H.
1. That means that the theoretical amount of hydrogen reversibly stored by LiBH4@C-MSU-H should be 6.9 wt% H2. However, the theoretical reversible amount should be approximately 6.5–6.7 wt% H2 (less than 6.9 wt% H2) because nickel borides or Li3N/Li3BN2 are also formed in the rehydrogenated nanocomposites containing either Ni or N. By making nanocomposites of LiBH4 infiltrate under H2 pressure in ordered nanoporous carbon, the aim is to avoid the growth of LiBH4 grains following the hydrogen absorption–desorption cycles, which would lead to a drastic deterioration of the hydrogen absorption–desorption kinetics. LiBH4@C-MSU-H desorbs 6.2 wt% H2 up to 450 °C for the first desorption with a temperature ramp of 2 °C min−1 (Fig. 7A). For complete desorption a much longer desorption time at 450 °C is required. Due to the presence of 1 at% Ni or N as doping elements, the LiBH4@doped C-MSU-H nanocomposites should desorb less than the theoretical amount of 6.9 wt% H2. LiBH4@C-MSU-H 1 at% Ni desorbs up to 450 °C, the least among all the samples, approximately 5.3 wt% H2 for the first desorption. After four hydrogen absorption–desorption cycles LiBH4@C-MSU-H releases only 3.1 wt% H2 up to 450 °C (Fig. 7B). LiBH4–C-MSU-H 1 at% N and LiBH4–C-MSU-H 1 at% Ni samples desorb 4.1 and 3.8 wt% H2 up to 450 °C, respectively, using the same temperature ramp of 2 °C min−1 (Fig. 7B). For complete desorption, a much longer desorption time at 450 °C is required. Faster desorption kinetics is observed for the N-doped sample compared to Ni-doped sample, but both doped samples have much faster desorption kinetics than the sample containing undoped C-MSU-H.
The experimental maximum reversible capacity was determined at 450 °C by allowing hydrogen uptake for at least 20 h under 100 atm and prolonged hydrogen desorption at the same temperature (at least 8 h). Fig. S1 (SI) describes thermal programmed desorption up to 450 °C and prolonged desorption at this final temperature. We obtained a reversible capacity of 6.14 wt% H2 for LiBH4@C-MSU-H 1 at% N and 6.05 wt% H2 for LiBH4@C-MSU-H 1 at % Ni rehydrogenated nanocomposites. The amount of hydrogen released after several absorption–desorption cycles is lower than that in the first desorption due to the impossibility of complete rehydrogenation of the dehydrogenated nanocomposites due to a kinetic barrier. Fig. 7C shows the desorption peak temperatures after four absorption/desorption cycles for undoped LiBH4@C-MSU-H nanocomposites and the ones doped with N or Ni. A desorption peak temperature of 339 °C is observed for LiBH4@C-MSU-H, 328 °C for LiBH4@C-MSU-H 1 at% Ni and 318 °C for LiBH4@C-MSU-H 1 at% N, thus a clear advantage for the N-doped nanocomposites compared with the other two samples. Fig. 7D shows the absorption kinetics after 4 a/d cycles for LiBH4 infiltrated in nanoporous carbon containing undoped, N doped and Ni doped C-MSU-H performed at 450 °C and 100 atm H2. In 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s (approximately 11 h) the LiBH4@C-MSU-H sample absorbs 3.6 wt% H2, LiBH4@C–MSU-H 1 at% Ni absorbs 4.3 wt% H2 and LiBH4@C–MSU-H 1 at% N almost 4.6 wt% H2. Once again, also regarding the absorption kinetics, N-doped nanocomposites show the best behavior, while undoped nanocomposites behave the worst.
000 s (approximately 11 h) the LiBH4@C-MSU-H sample absorbs 3.6 wt% H2, LiBH4@C–MSU-H 1 at% Ni absorbs 4.3 wt% H2 and LiBH4@C–MSU-H 1 at% N almost 4.6 wt% H2. Once again, also regarding the absorption kinetics, N-doped nanocomposites show the best behavior, while undoped nanocomposites behave the worst.
The Kissinger plot54 allows obtaining the activation energy of hydrogen desorption from the slope of the ln(β/Tp2) vs. 1/Tp graph according to the relation
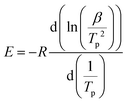 | (2) |
 | ||
| Fig. 8 Desorption peak temperature measured at various heating rates and corresponding Kissinger plots for (A) LiBH4@C-MSU-H, (B) LiBH4@C-MSU-H 1 at% Ni and (C) LiBH4@C-MSU-H 1 at% N. | ||
The incorporation of Li3N, LiNH2, or Li2NH into LiBH4@N-doped mesoporous carbons significantly enhances both hydrogen desorption kinetics and reversibility, primarily via the formation of stable Li–B–N–H intermediates and the reduction of activation energy barriers. Within N-doped nanoporous carbons, the dispersion of Li3N or LiNH2 nanoparticles ensures intimate contact with LiBH4 and maximizes interface area. Nitrogen functionalities on the carbon surface (pyridinic or pyrrolic N) act as Lewis base sites that coordinate to Li+ ions from LiBH4 or Li3N, inducing polarization of B–H bonds and facilitating H− transfer. In confined geometries, this reaction is spatially controlled, producing defect-rich interphases that sustain reversibility by minimizing B segregation and LiH aggregation. During cycling, the LiNH2 ↔ Li2NH ↔ Li3N coupled equilibrium acts as a hydrogen reservoir, allowing dynamic hydrogen exchange.55 Additionally, these equilibria maintain reactive species within the composite, stabilizing the system during multiple hydrogen absorption–desorption cycles.
| Li3N + 2H2 ↔ Li2NH + LiH + H2 ↔ LiNH2 + 2LiH | (3) |
The underlying chemistry encompasses formation of Li4BN3H1056 and its dehydrogenation to another catalytic species, Li3BN2.50 When LiBH4 is mixed or confined with Li3N, or equivalently with LiNH2 (derived in situ from Li3N + H2), a solid–solid reaction occurs upon heating, yielding the complex hydride Li4BN3H10:
| 3LiNH2 + LiBH4 → Li4BN3H10 | (4) |
This reaction represents a chemical destabilization of LiBH4 through amide/nitride incorporation, resulting in a hydrogen-rich compound (∼11.4 wt% H) that can dehydrogenate at lower temperatures (∼250 °C). Li4BN3H10 is the most thermodynamically favorable intermediate in the Li–B–N–H system.57 Upon heating, it may undergo several reactions leading to Li3BN2 as the more probable reaction product according to the reaction58
| Li4BN3H10 → Li3BN2 + LiNH2 + 4H2 | (5) |
Our HRXPS N 1s spectrum is consistent with the formation of Li3BN2 as an active catalytic species in LiBH4 dehydrogenation/rehydrogenation. The catalytic enhancement of LiBH4 hydrogen sorption by Li3BN2, can be attributed to the formation and decomposition of the Li4BN3H10 intermediate, and the interfacial electronic effects induced by N-doped carbon supports. The synergy between LiBH4 and N-doped carbon nanostructures—which provides conductive, confining, and catalytically active environments—yields a composite system with enhanced kinetics, reduced activation energy, and improved reversibility for hydrogen storage applications. Ni as a dopant for C-MSU-H forms catalysts such as Ni2B and Ni3B after interaction with LiBH4 which are less efficient than Li3BN2. Moreover, Li3BN2 catalytic centers formed around nitrogen inside the C-MSU-H matrix are much more dispersed compared to Ni2B and Ni3B clusters (provided from Ni/NiO initial clusters of 5.3 nm dispersed onto the C-MSU-H surface), leading to higher catalytic efficiency. Nickel borides actively participate in H2 dissociation/recombination. Using first principles calculations Liu et al.14 demonstrated that the dissociation energy for removing one H from LiBH4 decreased down to 1.00 eV for LiBH4 on Ni2B, much lower than the value of 4.22 eV for bulk LiBH4 and even lower than that of LiBH4 on Ni (1.27 eV) showing a superior catalytic effect of nickel boride compared to pure nickel. Additionally, using EXAFS spectroscopy at the Ni K-edge for 5 wt% Ni-catalyzed LiBH4, Ngene et al.59 identified NixB phases with different Ni–B and Ni–Ni coordinations for the dehydrogenated and rehydrogenated samples, which behave reversibly. Specifically, for the dehydrogenated samples Ni–Ni coordination decreased and Ni–B coordination increased whereas for the rehydrogenated samples Ni–Ni coordination increased and Ni–B coordination decreased.
The purpose of the work was to experimentally demonstrate the superiority of N over Ni as a doping element for the C-MSU-H-based catalyst for improving the dehydrogenation kinetics of LiBH4 and to explain this experimental finding. Table S1 (SI) shows the comparison of the hydrogen storage performances of the LiBH4@C-MSU-H 1 at% N studied in the present work with those presented in state-of-the-art literature for LiBH4-based systems. Our material was obtained through a simple procedure (melt infiltration), which does not consume much time and resources such as expensive precursors or toxic and critical elements. Further optimization of the catalyst is required to achieve better performances.
4. Conclusions
Ordered nanoporous carbon with nitrogen substitution was prepared using the nanotemplating method starting from nanoporous silica by sucrose impregnation and pyrolysis followed by etching in HF. Finally, the treatment in a gaseous ammonia flow at temperatures between 400 °C and 500 °C was used for nitrogen doping. For the doping of the ordered nanoporous carbon with 1 at% Ni, nickel nitrate hexahydrate dissolved in absolute ethanol was used. The final thermal treatment was performed for 4 h at 430 °C in a 5% H2/Ar flow. The ordered carbon nanoporous structure is maintained even after nitrogen doping according to diffraction spectra and TEM images. Doping levels of up to 1 at% are obtained by treatments in a gaseous ammonia flow at 500 °C. For the ordered nanoporous carbon C–MSU-H doped with N, an increase in the N-pyridinic content and a decrease in the N-pyrrolic content are observed as a result of increasing the treatment temperature in gaseous ammonia flow. However, the samples have mostly N-pyrrolic. After nickel doping, homogeneously distributed clusters with average size of 5.3 nm containing Ni and NiO are observed in TEM decorating the ordered nanoporous carbon structure C–MSU-H. Nanocomposites 50 wt% LiBH4–50 wt% undoped or doped with N or Ni ordered nanoporous carbon (LiBH4@C-MSU-H, LiBH4@C-MSU-H 1 at% N, LiBH4@C-MSU-H 1 at% Ni) were prepared using the method of infiltration under hydrogen pressure above the melting temperature of LiBH4. According to XRD, for the re-hydrogenated LiBH4@C-MSU-H 1 at% Ni nickel borides are also observed. The fastest hydrogen absorption and desorption kinetics were obtained for the LiBH4@C-MSU-H 1 at% N, followed by LiBH4@C-MSU-H 1 at% Ni and much slower desorption kinetics for the undoped nanocomposites. The desorption peak temperatures (2 °C min−1 ramp) are 339 °C for LiBH4@C-MSU-H, 328 °C for LiBH4@C-MSU-H 1 at% Ni and 318 °C for the LiBH4@C-MSU-H 1 at% N. From the Kissinger plots the activation energies of hydrogen desorption for the nanocomposites were obtained: 142.7 kJ mol−1 for LiBH4@C-MSU-H, 123.8 kJ mol−1 for LiBH4@C-MSU-H doped with 1 at% Ni and 119.5 kJ mol−1 for LiBH4 infiltrated in 1 at% N-doped C-MSU-H.Author contributions
Conceptualization: P. P. and C. C.; investigation: P. P., C. N., A. G. M., and C. R., formal analysis: P. P., C. C., C. N. A. G. M., and C. R.; software: C. C., P. P., C. R., and C. N.; data curation: P. P. and C. C.; writing original draft: P. P., C. C., C. N., and A. G. M.; writing – revised editing: all authors. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data that support the findings of this study are available from the authors upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5cp03997e.
Acknowledgements
This work was funded by the Core Program of the National Institute of Materials Physics, granted by the Romanian Ministry of Research, Innovation and Digitization through the Project PC3-PN23080303.References
- L. Schlapbach and A. Züttel, Hydrogen-storage materials for mobile applications, Nature, 2001, 414, 353–358 CrossRef CAS PubMed.
- S. Orimo, Y. Nakamori, G. Kitahara, K. Miwa, N. Ohba, S. Towata and A. Züttel, Dehydriding and rehydriding reactions of LiBH4, J. Alloys Compd., 2005, 404–406, 427–430 CrossRef CAS.
- Y. Xu, Y. Zhou, Y. Li, M. Ashuri and Z. Ding, Engineering LiBH4-based materials for advanced hydrogen storage: A critical review of catalysis, nanoconfinement, and composite design, Molecules, 2024, 29, 5774 CrossRef CAS PubMed.
- D. Yin, X. Wang, F. Wu, D. Shang, H. Li, P. Huang and L. Zhang, A mini review on the state-of-the-art progress of LiBH4 for hydrogen storage: Challenges and perspectives, J. Alloys Compd., 2025, 1041, 183883 Search PubMed.
- J. Zheng, Z. Yao, X. Xiao, X. Wang, J. He, M. Chen, H. Cheng, L. Zhang and L. Chen, Enhanced hydrogen storage properties of high-loading nanoconfined LiBH4-Mg(BH4)2 composites with porous hollow carbon nanospheres, Int. J. Hydrogen Energy, 2021, 46, 852–864 Search PubMed.
- R. Wu, X. Zhang, Y. Liu, L. Zhang, J. Lu, M. Gao and H. Pan, A unique double-layered carbon nanobowl-confined lithium borohydride for highly reversible hydrogen storage, Small, 2020, 16, 2001963 Search PubMed.
- Suwarno, P. Ngene, A. Nale, T. M. Eggenhuisen, M. Oschatz, J. P. Embs, A. Remhof and P. E. de Jongh, Confinement effects for lithium borohydride: comparing silica and carbon scaffolds, J. Phys. Chem. C, 2017, 121, 4197–4205 CrossRef CAS PubMed.
- S. D. House, X. Liu, A. A. Rockett, E. H. Majzoub and I. M. Robertson, Characterization of the dehydrogenation process of LiBH4 confined in nanoporous carbon, J. Phys. Chem. C, 2014, 118, 8843–8851 CrossRef CAS.
- A. Gasnier and F. C. Gennari, Graphene entanglement in a mesoporous resorcinol-formaldehyde matrix applied to the nanoconfinement of LiBH4 for hydrogen storage, RSC Adv., 2017, 7, 27905–27912 Search PubMed.
- Y. Ding, C. Li, X. Zhang, W. Chen, X. Yu and G. Xia, Spatial confinement of lithium borohydride in bimetallic CoNi-doped hollow carbon frameworks for stable hydrogen storage, ACS Appl. Mater. Interfaces, 2024, 16, 50717–50725 CrossRef CAS PubMed.
- S. Wang, M. Gao, Z. Yao, Y. Liu, M. Wu, Z. Li, Y. Liu, W. Sun and H. Pan, A nanoconfined-LiBH4 system using a unique multifunctional porous scaffold of carbon wrapped ultrafine Fe3O4 skeleton for reversible hydrogen storage with high capacity, Chem. Eng. J., 2022, 428, 131056 CrossRef CAS.
- K. Xian, B. Nie, Z. Li, M. Gao, Z. Li, C. Shang, Y. Liu, Z. Guo and H. Pan, TiO2 decorated porous carbonaceous network structures offer confinement, catalysis and thermal conductivity for effective hydrogen storage of LiBH4, Chem. Eng. J., 2021, 407, 127156 CrossRef CAS.
- W. Chen, Y. Liu, C. Li, Y. Pang, X. Yu and G. Xia, Enhancing reversible hydrogen storage performance of LiBH4 catalyzed by N-doped carbon nanosheet networks embedded with Co nanoparticles, ACS Appl. Mater. Interfaces, 2025, 17, 50603–50611 CrossRef CAS PubMed.
- Y. Liu, W. Chen, S. Ju, X. Yu and G. Xia, Stable hydrogen storage of lithium borohydrides via the catalytic effect of Ni2B induced by thermodynamic destabilization reaction, J. Mater. Sci. Technol., 2024, 202, 192–200 Search PubMed.
- Z. Z. Fang, X. D. Kang, P. Wang and H.-M. Cheng, Improved reversible dehydrogenation of LiBH4 by milling with as-prepared single-walled nanotubes, J. Phys. Chem. C, 2008, 112, 17023–17029 CrossRef CAS.
- G. Na, W. G. Cui, H. Shi, Z. Li, F. Gao, X. Wang, K. Wang, Y. Gao, Y. Yang, Z. Shen, Y. Liu, J. Miao and H. Pan, In situ generation and stabilization of multiple catalysts by introducing a graphene-supported FeF2/FeOx additive for enhancing the hydrogen storage of LiBH4, ACS Appl. Energy Mater., 2025, 8, 3802–3811 CrossRef CAS.
- Y. Pang, X. Hu, X. Zhang, X. Yu and G. Xia, Two-dimensional transition metal-based high-entropy oxide nanoplates for enhanced hydrogen storage of LiBH4, J. Alloys Compd., 2025, 1040, 183613 CrossRef CAS.
- X. Zhang, L. Zhang, W. Zhang, Z. Ren, Z. Huang, J. Hu, M. Gao, H. Pan and Y. Liu, Nano-synergy enables highly reversible storage of 9.2 wt% at mild conditions with lithium borohydride, Nano Energy, 2021, 83, 105839 CrossRef CAS.
- J. J. Vajo, S. L. Skeith and F. Mertens, Reversible storage of hydrogen in destabilized LiBH4, J. Phys. Chem. B, 2005, 109, 3719–3722 CrossRef CAS PubMed.
- Q. He, D. Zhu, X. Wu, D. Dong, M. Xu and Z. Tong, Hydrogen desorption properties of LiBH4/xLiAlH4 (x = 0.5, 1, 2) composites, Molecules, 2019, 24, 1861 CrossRef CAS PubMed.
- J. H. Shim, Y. S. Lee, J. Y. Suh, W. Cho, S. S. Han and Y. W. Cho, Thermodynamics of the dehydrogenation of the LiBH4-YH3 composite: Experimental and theoretical studies, J. Alloys Compd., 2012, 510, L9–L12 CrossRef CAS.
- N. Bergemann, C. Pistidda, M. Uptmoor, C. Milanese, A. Santoru, T. Emmler, J. Puszkiel, M. Dornheim and T. Klassen, A new mutually destabilized reactive hydride system: LiBH4-Mg2NiH4, J. Energy Chem., 2019, 34, 240–254 CrossRef.
- P. S. Miedema, P. Ngene, A. M. J. van der Eerden, D. Sokaras, T. C. Weng, D. Nordlund, Y. S. Au and F. M. F. de Groot, In-situ X-ray Raman spectroscopy study of hydrogen sorption properties of lithium borohydride composites, Phys. Chem. Chem. Phys., 2014, 16, 22651–22658 RSC.
- X. Liu, E. H. Majzoub, V. Stavila, R. K. Bhakta, M. D. Allendorf, D. T. Shane, M. S. Conradi, N. Verdal, T. J. Udovic and S. J. Hwang, Probing the unusual anion mobility of LiBH4 confined in highly ordered nanoporous carbon frameworks via solid state NMR and quasielastic neutron scattering, J. Mater. Chem. A, 2013, 1, 9935–9941 Search PubMed.
- Y. Yan, H. Wang, M. Zhu, W. Cai, D. Rentsch and A. Remhof, Direct rehydrogenation of LiBH4 from H-deficient Li2B2H12−x, Crystals, 2018, 8, 131 CrossRef.
- P. Ngene, M. R. van Zwienen and P. E. de Jongh, Reversibility of the hydrogen desorption from LiBH4: A synergetic effect of nanoconfinement and Ni addition, Chem. Commun., 2010, 46, 8201–8203 RSC.
- F. Wang, J. Xu, X. Shao, X. Su, Y. Huang and T. Zhang, Palladium on nitrogen-doped mesoporous carbon: A bifunctional catalyst for formate-based, carbon-neutral hydrogen storage, ChemSusChem, 2016, 9, 246–251 CrossRef CAS PubMed.
- K. Koh, M. Jeon, D. M. Chevrier, P. Zhang, C. W. Yoon and T. Asefa, Novel nanoporous N-doped carbon-supported ultrasmall Pd nanoparticles: Efficient catalyst for hydrogen storage and release, Appl. Catal., B, 2017, 203, 820–828 CrossRef CAS.
- I. Muhammad, J. Saddique, C. Wu, M. ur Rahman, Z. U. Khan, W. Ali and R. Zhang, Nitrogen-doped -graphene-supported nickel nanoparticles reveal low dehydrogenation temperature and long cyclic life of magnesium hydrides, ACS Omega, 2024, 9, 19261–19271 CrossRef CAS PubMed.
- X. Zhang, Y. Sun, S. Ju, J. Ye, X. Hu, W. Chen, L. Yao, G. Xia, F. Fang, D. Sun and X. Yu, Solar-driven reversible hydrogen storage, Adv. Mater., 2023, 35, 2206946 CrossRef CAS PubMed.
- X. Hu, X. Chen, X. Zhang, Y. Meng, G. Xia, X. Yu, D. Sun and F. Fang, In situ construction of interface with photothermal and mutual catalytic effect for efficient solar-driven reversible hydrogen storage of MgH2, Adv. Sci., 2024, 11, 2400274 CrossRef CAS PubMed.
- X. Zhang, S. Ju, C. Li, J. Hao, Y. Sun, X. Hu, W. Chen, J. Chen, L. He, G. Xia, F. Fang, D. Sun and X. Yu, Atomic reconstruction for realizing stable solar-driven reversible hydrogen storage of magnesium hydride, Nat. Commun., 2024, 15, 2815 CrossRef CAS PubMed.
- Y. Jiang, Y. Sun, Y. Liu, M. Yue, Y. Cao, Q. Yuan and Y. Wang, Solar-driven reversible hydrogen storage in metal oxides-catalyzed MgH2, Int. J. Hydrogen Energy, 2025, 149, 150101 CrossRef CAS.
- Y. Sun, Y. Jiang, Y. Liu, L. Feng, M. Yue, Y. Cao and Y. Wang, Solar-driven rapid dehydrogenation of LiBH4 catalyzed by g-C3N4/α-Fe2O3 heterojunction, Int. J. Hydrogen Energy, 2026, 200, 152958 CrossRef CAS.
- H. Wang, Q. Li, J. Chen, J. Chen and H. Jia, Efficient solar-driven CO2 methanation and hydrogen storage over nickel catalyst derived from metal-organic frameworks with rich oxygen vacancies, Adv. Sci., 2023, 10, 2304406 CrossRef CAS PubMed.
- K. Tan, Q. Pei, Y. Yu, L. Liu, J. Li, L. Han, Y. Yu, Z. Li, A. Munyentwali, J. Guo, Y. Wang, L. Rao, T. He and P. Chen, Solar-driven reversible hydrogen storage of sodium cyclohexanolate/phenoxide pair, Angew. Chem., Int. Ed., 2025, 64, e202506275 CrossRef CAS PubMed.
- S. S. Kim, A. Karkamkar, T. J. Pinnavaia, M. Kruk and M. Jaroniek, Synthesis and characterization of ordered, very large pore MSU-H silicas assembled from water-soluble silicates, J. Phys. Chem. B, 2001, 105, 7663–7670 CrossRef CAS.
- S. S. Kim, T. R. Pauly and T. J. Pinnavaia, Non-ionic surfactant assembly of ordered, very large pore molecular sieve silicas from water soluble silicates, ChemComm, 2000, 17, 1661–1662 RSC.
- S. H. Joo, R. Ryoo, M. Kruk and M. Jaroniec, Evidence for general nature of pore interconnectivity in 2-dimensional hexagonal mesoporous silicas prepared using block copolymer templates, J. Phys. Chem. B, 2002, 106, 4640–4646 CrossRef CAS.
- S. S. Kim and T. J. Pinnavaia, A low cost route to hexagonal mesostructured carbon molecular sieves, ChemComm, 2001, 23, 2418–2419 RSC.
- X. Liu, D. Peaslee, C. Z. Jost and E. H. Majzoub, Controlling the decomposition pathway of LiBH4 via confinement in highly ordered nanoporous carbon, J. Phys. Chem. C, 2010, 114, 14036–14041 CrossRef CAS.
- M. Oschatz, J. P. Hofmann, T. W. van Deelen, W. S. Lamme, N. A. Krans, E. J. M. Hensen and K. P. de Jong, Effects of the functionalization of the ordered mesoporous carbon support surface on iron catalysts for the Fischer-Tropsch synthesis of lower olefins, ChemCatChem, 2017, 9, 620–628 CrossRef CAS PubMed.
- B. Lesiak, L. Kövér, J. Tóth, J. Zemek, P. Jiricek, A. Kromka and N. Rangam, C sp2/sp3 hybridisations in carbon nanomaterials – XPS and (X)AES study, Appl. Surf. Sci., 2018, 452, 223–231 CrossRef CAS.
- H. Darmstadt, C. Roy, S. Kaliaguine, S. J. Choi and R. Ryoo, Surface chemistry of ordered mesoporous carbons, Carbon, 2002, 40, 2673–2683 CrossRef CAS.
- L. M. Chew, W. Xia, H. Düdder, P. Weide, H. Ruland and M. Muhler, On the role of the stability of functional groups in multi-walled carbon nanotubes applied as support in iron-based high-temperature Fischer–Tropsch synthesis, Catal. Today, 2016, 270, 85–92 CrossRef CAS.
- Y. J. Cho, S. Y. Kang, B. C. Wood and E. S. Cho, Heteroatom-doped graphenes as actively interacting 2D encapsulation media for Mg based hydrogen storage, ACS Appl. Mater. Interfaces, 2022, 14, 20823–20834 CrossRef CAS PubMed.
- Q. Wang, J. Su, H. Chen, D. Wang, X. Tian, Y. Zhang, X. Feng, S. Wang, J. Li and H. Jin, Highly conductive nitrogen-doped sp2/sp3 hybrid carbon as a conductor-free charge storage host, Adv. Funct. Mater., 2022, 32, 2209201 CrossRef CAS.
- S. D. S. Fitch, G. E. Moehl, N. Meddings, S. Fop, S. Solue, T. L. Lee, M. Kazemian, N. G. Araez and A. L. Hector, Combined electrochemical, XPS, and STXM study of lithium nitride as a protective coating for lithium metal and lithium-sulfur batteries, ACS Appl. Mater. Interfaces, 2023, 15, 39198–39210 CrossRef CAS PubMed.
- W. Hou, Y. Li, S. Li, Z. Liu, P. G. Ryan, M. Xu, J. K. Kim, B. Yuan, R. Hu and Z. Luo, Lithium dendrite suppression with Li3N-rich protection layer formation on 3D anode via ultra-low temperature nitriding, Chem. Eng. J., 2022, 441, 136067 CrossRef CAS.
- S. Emani, C. Liu, M. Ashuri, K. Sahni, J. Wu, W. Yang, K. Nemeth and L. L. Shaw, Li3BN2 as a transition metal free, high capacity cathode for Li-ion batteries, ChemElectroChem, 2019, 6, 320–325 CrossRef CAS.
- Y. Pang, C. Wei, X. Ye, X. Li, H. Sun, S. Luo, T. Chen, S. Xia, T. Yuan and S. Zheng, Hδ−–Hδ+ comproportionation enables stable Li-N-H-F solid electrolyte, Angew. Chem., Int. Ed., 2025, 64, e202511344 CrossRef CAS PubMed.
- J. Legrand, A. Taleb, S. Gota, M. J. Guittet and C. Petit, Synthesis and XPS characterization of nickel boride nanoparticles, Langmuir, 2002, 18, 4131–4137 CrossRef CAS.
- J. Hong, M. Miola, D. Gerlach, M. C. A. Stuart, P. Rudolf, D. M. Morales, L. Protesescu and P. P. Pescarmona, An exploration of the electrocatalytic activity of nickel boride nanocrystals in the oxidation of 5-HMF, Catal. Sci. Technol., 2025, 15, 457–475 RSC.
- H. E. Kissinger, Variation of peak temperature with heating rate in differential thermal analysis, J. Res. Natl. Bur. Stand., 1956, 57, 217–221 CrossRef CAS.
- J. L. White, A. A. Baker, M. A. Marcus, J. L. Snider, T. C. Wang, J. R. I. Lee, D. A. L. Kilcoyne, M. D. Allendorf, V. Stavila and F. El Gabaly, The inside-outs of metal hydride dehydrogenation: imaging the phase evolution of the Li-N-H hydrogen storage system, Adv. Mater. Interfaces, 2020, 7, 1901905 CrossRef CAS.
- Y. E. Filinchuk, K. Yvon, G. P. Meisner, F. E. Pinkerton and M. P. Balogh, On the composition and crystal structure of the new quaternary hydride phase Li4BN3H10, Inorg. Chem., 2006, 45, 1433–1435 CrossRef CAS PubMed.
- D. J. Siegel, C. Wolverton and V. Ozoliņs, Reaction energetics and crystal structure of Li4BN3H10 from first principles, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 75, 014101 CrossRef.
- J. F. Herbst and L. G. Hector, Jr, Electronic structure and energetics of the quaternary hydride Li4BN3H10, Appl. Phys. Lett., 2006, 88, 231904 CrossRef.
- P. Ngene, M. H. W. Verkuijlen, Q. Zheng, J. Kragten, P. J. M. van Bentum, J. H. Bitter and P. E. de Jongh, The role of Ni in increasing the reversibility of the hydrogen release from nanoconfined LiBH4, Faraday Discuss., 2011, 151, 47–58 RSC.
| This journal is © the Owner Societies 2026 |