 Open Access Article
Open Access ArticleBroadband cross polarization for ultra-wideline magic-angle spinning NMR†
James J.
Kimball
 ab,
Sophia
Keil
ab,
Sophia
Keil
 c,
Max
Bußkamp
c,
Adam R.
Altenhof
c,
Max
Bußkamp
c,
Adam R.
Altenhof
 ab,
Michael Ryan
Hansen
ab,
Michael Ryan
Hansen
 *cd and
Robert W.
Schurko
*cd and
Robert W.
Schurko
 *ab
*ab
aDepartment of Chemistry & Biochemistry, Florida State University, Tallahassee, FL 32306, USA. E-mail: rschurko@fsu.edu
bNational High Magnetic Field Laboratory, Tallahassee, FL 32310, USA
cInstitute for Physical Chemistry, University of Münster, Münster, DE-48149, Germany. E-mail: mhansen@uni-muenster.de
dCenter for Multiscale Theory and Computation (CMTC), University of Münster, Münster, DE-48149, Germany
First published on 11th March 2026
Abstract
Over the past decade, there has been a sustained interest in using frequency-swept (FS) pulses for the efficient acquisition of wideline and ultra-wideline (UW) NMR powder patterns. Such experiments are typically conducted under static conditions, employing both direct- and indirect-excitation methods (i.e., WCPMG and BRAIN-CP/WCPMG, respectively). Recently, Koppe et al. demonstrated that the WCPMG pulse sequence can be used to efficiently acquire wideline and UW NMR spectra with spinning sideband (SSB) manifolds under magic-angle spinning (MAS) conditions, capitalizing on the increased signal-to-noise ratios (SNR) afforded by MAS. To date, there have been only a few instances of broadband cross-polarization (CP) experiments using FS pulses under MAS conditions and no applications to systems exhibiting wideline and/or ultra-wideline powder patterns, despite the clear advantages these experiments could offer. Herein, we demonstrate that FS pulses selectively applied to a single sideband of the S spin can be used for efficient 1H-S polarization transfer to S = 1/2 nuclides with large anisotropic chemical shift interactions at slow to moderate MAS rates. The Hartmann–Hahn matching conditions in BRAIN-CP/WCPMG-MAS experiments bear similarity to those of standard CP sequences, yet operate over UW frequency ranges and only require low-amplitude RF pulses on the S channel. Crucial to the success of the BRAIN-CP/WCPMG-MAS experiment is careful calibration of the RF amplitude, transmitter offset, and effective frequency sweep of the FS pulse applied to the S spins at a given MAS rate. Thus, by means of numerical simulations and experimental testing, we provide recommendations for the parameterization and setup of BRAIN-CP/WCPMG-MAS experiments for their most efficient use. Results showcasing the capability of the BRAIN-CP/WCPMG-MAS pulse sequence are presented, including applications to 119Sn, 195Pt, and 103Rh NMR.
1. Introduction
In recent years, the increasing availability of high magnetic fields has posed a significant challenge for solid-state NMR (SSNMR) of nuclides affected by strong chemical shift anisotropy (CSA), as their powder pattern breadths scale proportionally with the applied magnetic field strength, B0. In instances of SSNMR spectra with ultra-wideline (UW) powder patterns (i.e., those with breadths surpassing 250 kHz), the effectiveness of traditional pulsed NMR methods is compromised due to the inability to manipulate spin polarization across large frequency bandwidths using rectangular pulses and conventional probes.1 Nevertheless, acquisition of such SSNMR spectra is not insurmountable, as numerous pulse sequences have been designed for this very purpose.2–5At the heart of these methods is the use of frequency-swept (FS) pulses, which feature time-dependent phases with the optional addition of modulated amplitudes.6,7 These pulses have been used extensively for a wide variety of purposes, including but not limited to excitation, refocusing, and polarization transfer – all of which are crucial building blocks for many commonly used techniques in NMR.8–10 Of the wide variety of FS pulses available, wideband, uniform rate, smooth truncation (WURST) pulses featuring linear frequency sweeps have found the most success in UW NMR experiments.11,12 The most prevalent example is the WURST-CPMG (WCPMG) sequence, wherein WURST pulses are used for direct excitation (DE) in CPMG-style experiments under both static and MAS conditions.13–15 For indirect excitation (i.e., 1H → S cross polarization, CP) experiments, WURST pulses are used in the framework of the broadband adiabatic inversion cross-polarization (BRAIN-CP) sequence, enabling efficient CP across bandwidths well into the UW regime.16 BRAIN-CP has been thoroughly explored in applications to static samples;17,18 however, the use of FS pulses for broadband CP under MAS conditions has been limited to date,19–21 and there exist no examples of its application for the acquisition of UW NMR spectra under MAS conditions without the assistance of techniques such as acquisition of a variable offset cumulative spectrum (VOCS), wherein the transmitter is “frequency stepped” across the pattern breadth.22,23
The complex behavior of FS pulses under MAS has likely hindered their widespread application; however, this is not unexplored territory. Pell and coworkers demonstrated the use of WURST pulses for broadband inversion of spin polarization under MAS.24–28 In such experiments, the phase modulation of the pulse is set such that the effective frequency sweeps across just one spinning sideband (SSB), yet inversion of the entire sideband manifold is observed. WURST applied in this way are referred to as single-sideband selective adiabatic pulses (S3APs) and their performance in terms of spin locking and adiabatic inversion hinges upon satisfaction of the following experimental protocols: (i) the sweep width (Δ, as determined by the phase modulation) is matched to the MAS rate (ωrot = 2πνrot, i.e., Δ = νrot)25 and (ii) the offset frequency (ΩS = ω0,S − ωTx,S, where ω0,S is the Larmor frequency and ωTx,S is the time-independent transmitter frequency) is set such that the frequency sweep centered at a transmitter frequency of ωTx,S passes through the resonance frequency of a single SSB.27 Also of importance is the pulse amplitude (ω1,S = 2πν1,S), which has direct implications on the adiabaticity of the S3AP. Ideally, the condition |ν1,S| ≪ |νrot| is in place;26 yet, efficient inversion across UW frequency ranges has been also reported in the more intermediate regime of |ν1,S| ≈ |νrot|.28
In this work, we demonstrate the implementation of S3APs for CP in a manner not unlike that of the BRAIN-CP sequence in combination with WCPMG incremented echo acquisition14 (hence the name BRAIN-CP/WCPMG-MAS). The S3AP is used for fulfillment of the HH matching conditions and subsequent spin locking of the magnetization produced from broadband CP, and culminates with the storage of magnetization along the z-axis. The principles outlined by Pell and coauthors are used in the theoretical description of the BRAIN-CP/WCPMG-MAS experiment. Hartmann–Hahn (HH) matching conditions are derived using a combination of analytical equations, simulated experiments, and experimental measurements. Parameters crucial to the success of the BRAIN-CP/WCPMG-MAS sequence, which include the transmitter offset, spin locking amplitude, and phase modulation of the S3AP, are addressed in detail. Uniform polarization transfer across bandwidths surpassing 1 MHz is demonstrated, showcasing the capability of a properly optimized BRAIN-CP/WCPMG-MAS sequence. A variety of experimental results are used to illustrate the potential of BRAIN-CP/WCPMG-MAS for significant reduction in the experimental times required to obtain spectra of both wideline (ca. 50–250 kHz in breadth) and UW SSB manifolds of spin-1/2 nuclides, compared to analogous static and/or direct-excitation experiments.
2. Experimental methods
2.1 Samples
Dibutyltin oxide [DBTO, Sigma Aldrich], cisplatin [Pt(NH3)2Cl2, Thermo Fisher], and chloro(1,5-cylcooctadiene)rhodium(I) dimer [Rh2Cl2(COD)2, Strem] were purchased from the listed sources and used in all subsequent NMR experiments without further purification. The identities and purities of the samples were verified through comparisons with previously reported SSNMR spectra and PXRD patterns. The novel [Sn(HPDABA)]Cl2 (HPDABA = 2-hydroxy-4-(propan-2-ylideneamino)benzoic acid) cocrystal was prepared mechanochemically via ball milling (see SI for the synthetic procedure). All samples were ground into fine powders on the benchtop under ambient conditions and were packed into 3.2 mm (for νrot ≤ 15 kHz) and 2.5 mm (for νrot > 15 kHz) zirconium NMR rotors.2.2 SSNMR spectroscopy
Bruker Avance NEO consoles were used for all experiments. 1H–119Sn NMR spectra were acquired using a 14.1 T Magnex/Bruker [ν0 (1H) = 600 MHz] wide bore magnet with a resonance frequency of ν0 (119Sn) = 223.744 MHz. 1H–195Pt NMR experiments were conducted using a 11.7 T Bruker [ν0 (1H) = 500 MHz] wide bore magnet with ν0 (195Pt) = 106.700 MHz. 1H–103Rh NMR experiments were conducted using a 21.1 T [ν0 (1H) = 900 MHz] home-built ultra-wide bore magnet with ν0 (103Rh) = 28.678 MHz. Home-built 3.2 mm triple-resonance (HXY) and 3.2 mm double-resonance (HX) probes were used for experiments at 14.1 T and 21.1 T, respectively. A 2.5 mm triple resonance (HXY) probe was used for experiments at 11.7 T. A Bruker MAS3 unit was used for all MAS NMR experiments with νrot set between 10 and 25 kHz. The magic angle of the NMR probes was calibrated to 54.74° by maximizing the number of rotational echoes observed in the 79Br FID of KBr29 or by minimizing the linewidth of the SSBs in the 23Na MAS NMR spectrum of NaNO3.30Pulse width calibrations for all S nuclides (excluding those for 195Pt which were performed with a Bloch decay) were performed under static conditions by arraying the width of an excitation pulse (τcal) inserted between the contact pulse and the first refocusing pulse in a CP/CPMG experiment (see SI for the pulse sequence, Scheme S1). In each case, the transmitter frequency was tuned to a discontinuity of high signal intensity in the powder pattern and care was taken to interpret the resulting nutation curve according to the on-resonance signal only. 119Sn chemical shifts were referenced to neat Sn(CH3)4 with δiso = 0 ppm using SnO(s) as a secondary reference (δ22 = δ33 = 121.3 ppm and δ11 = 866.7 ppm, where δ11 and δ22 represent the largest principal components (lowest shielding) of the chemical shielding tensor using the standard convention).31195Pt chemical shifts were referenced to 1.0 M K2PtCl6(aq) with δiso = 0 ppm. 103Rh chemical shifts were referenced such that δ(103Rh) = 0 ppm using a frequency ratio of Ξ = 3.16% relative to the 1H resonance frequency of TMS(l).32
Calibrated spin-locking fields applied to the S and I spins, ν1,S and ν1,I, were set as ν1,S = 5–25 kHz and ν1,I = 5–50 kHz for all experiments in order to fulfill the BRAIN-CP/WCPMG-MAS HH matching conditions (see Section 3.3). These were determined as the ratio of RF powers on the S and I channels yielding spectra with patterns of the highest signal intensity and overall pattern uniformity. In all experiments, the condition Δ = ν1,S = νrot was fulfilled in order to achieve broadband adiabatic spin locking, where Δ is the sweep width of the contact pulse as determined by the phase modulation, and νrot is the MAS rate. The contact pulse lengths, τCT, are on the order of milliseconds, with the exact experimental values determined from the relative magnitudes of the approximate heteronuclear dipolar couplings and SSB manifold breadths, as is conventionally done in the optimization of CP-type experiments.18,33
Rotor synchronization of excitation and refocusing WURST pulses during the CPMG train (Scheme 1) was achieved by ensuring that the condition 2Mτrot = 2τ2 + τref + τSE was satisfied, where 2M, τrot, τ2, τref, and τSE are the number of rotor periods per CPMG cycle, the rotor period, the dead time, the refocusing pulse width, and the length of the spin echo, respectively and M is an integer.34 In order to maximize signal intensity, the amplitudes of the excitation and refocusing WURST pulses were adjusted to satisfy the condition  where c is an experimentally determined constant, Rexc/ref = Δexc/ref/τexc/ref is the sweep rate of the WURST excitation and refocusing pulses, Δexc/ref is the sweep width, and τexc/ref is the pulse length.35,36Rexc/ref was set to satisfy the condition x ≥ 3, where x = Rexc/ref/(ΩSSB·νrot) and ΩSSB is the breadth (expressed in Hz) of the SSB manifold that is traversed by the effective frequency sweep over the course of one rotor period.14 As described by Koppe and coworkers,14 conditions of x ≥ 10 result in more efficient excitation and refocusing; however, for SSB manifolds in UW NMR spectra where ΩSSB is much larger, higher R values are required to meet these conditions, thereby necessitating higher amplitudes of νexc/ref. In some cases, this is unachievable due to probe power limitations (e.g., in 1H–103Rh experiments). All BRAIN-CP/WCPMG-MAS experiments required optimization of the transmitter offset of the pulse applied to the S spins (ΩS). This was accomplished by arraying ΩS and choosing the value resulting in the signal of highest intensity. The need for initial optimization of ΩS is less essential in cases where δiso is known, as a value of ΩS around δiso ± ωrot/2 (such that the transmitter is tuned adjacent to the isotropic shift) can be used as a starting point (see Section 3.3). It is nevertheless recommended to optimize this parameter after adjusting the HH matching conditions.
where c is an experimentally determined constant, Rexc/ref = Δexc/ref/τexc/ref is the sweep rate of the WURST excitation and refocusing pulses, Δexc/ref is the sweep width, and τexc/ref is the pulse length.35,36Rexc/ref was set to satisfy the condition x ≥ 3, where x = Rexc/ref/(ΩSSB·νrot) and ΩSSB is the breadth (expressed in Hz) of the SSB manifold that is traversed by the effective frequency sweep over the course of one rotor period.14 As described by Koppe and coworkers,14 conditions of x ≥ 10 result in more efficient excitation and refocusing; however, for SSB manifolds in UW NMR spectra where ΩSSB is much larger, higher R values are required to meet these conditions, thereby necessitating higher amplitudes of νexc/ref. In some cases, this is unachievable due to probe power limitations (e.g., in 1H–103Rh experiments). All BRAIN-CP/WCPMG-MAS experiments required optimization of the transmitter offset of the pulse applied to the S spins (ΩS). This was accomplished by arraying ΩS and choosing the value resulting in the signal of highest intensity. The need for initial optimization of ΩS is less essential in cases where δiso is known, as a value of ΩS around δiso ± ωrot/2 (such that the transmitter is tuned adjacent to the isotropic shift) can be used as a starting point (see Section 3.3). It is nevertheless recommended to optimize this parameter after adjusting the HH matching conditions.
A full list of all BRAIN-CP/WCPMG-MAS and WCPMG-MAS parameters is given in Tables S1 and S2. All spectra were acquired using TPPM 1H decoupling with RF fields between 30–50 kHz, excluding the 195Pt spectra of cisplatin which were recorded using swept-frequency TPPM decoupling with an RF amplitude of 83 kHz. Direct-excitation (DE) schemes employ the eight-step phase cycling scheme used by Bhattacharyya and Frydman37 and CP schemes use the sixteen-step phase cycling scheme used by Larsen and co-workers.38 All pulse sequences described herein are available upon request or at https://github.com/rschurko.
2.3 Spectral processing and simulations
All data sets were processed in MATLAB via multiplication of each echo with a multi-peak Gaussian function, coaddition of echoes, fast-Fourier transform, and magnitude calculation (i.e., spectra are not phased) using a custom-written code (available on request). Numerical simulations, performed with both the SIMPSON 4.1.139 open source software package and a custom written simulation package in MATLAB R2023b, were used to monitor spin dynamics. An IS spin system (I = 1H, S = ½) was used, with an axially symmetrical CS tensor for the S spin where δiso = 0 ppm, ΩCS = either 675 or 1350 ppm (indicated in each case), and κ = −1, using the Herzfeld–Berger convention (or δiso = 0 ppm, Δδ = either 450 or 900 ppm (indicated in each case), and η = 0, using the Haeberlen convention).40 The heteronuclear dipolar coupling constant is given by bISD = γIγSrIS−3(ħ2π)(μ0/4π) and the corresponding dipolar frequency is given by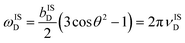 where θ describes the angle between the principle axis system (PAS) of the heteronuclear dipolar tensor and the rotor axis. In all cases, bISD/2π and θ were set to 4 kHz and 45°, respectively. All simulations were conducted using a magnetic field strength of 14.1 T.
where θ describes the angle between the principle axis system (PAS) of the heteronuclear dipolar tensor and the rotor axis. In all cases, bISD/2π and θ were set to 4 kHz and 45°, respectively. All simulations were conducted using a magnetic field strength of 14.1 T.
Throughout this work, we use the term isochromats to refer to groups of nuclear spins that precess at identical rates. Isochromats are distinguished by their orientations, defined by the angles α and β, where α is the azimuthal angle and β describes the relative orientation of the PAS for the chemical shift tensor and the unique axis of the MAS frame. In cases of axially symmetric CS tensors, the former angle is irrelevant and thus isochromats can be described completely by the angle β. Assuming a homogeneous B0, different isochromats thus represent unique resonance frequencies which collectively comprise a SSB manifold under MAS. For simplicity, axially symmetric CS tensors are used in all ensuing simulations unless otherwise denoted. Powder averaging is accomplished using the REPULSION scheme,41 with the number of orientations indicated in each case. Calculations were performed on a MacBook Pro operating on macOS 14.1.1 with an Apple M1 Pro chip and on NMRBox virtual machines.42
3. Protocols
3.1 A first example: wideline 1H–119Sn BRAIN-CP MAS NMR
The novel co-crystal [Sn(HPDABA)]Cl2 was chosen as a test case due to its favorable NMR properties for spectral acquisition using BRAIN-CP/WCPMG-MAS. Its large 119Sn CSA (ΩCS = 760 ppm, ca. 200 kHz in breadth at 14.1 T) and long T1(119Sn) time constant (estimated 40 s from an optimized recycle delay of 200 s) render DE methods impractical for the acquisition of 119Sn SSNMR spectra. However, the much shorter T1(1H) time constant (estimated from an optimized recycle delay of 5 s), in addition to 1H–119Sn heteronuclear dipolar coupling constants of moderate magnitude, set the stage for efficient CP experiments.The 1H–119Sn BRAIN-CP/WCPMG-MAS spectrum of [Sn(HPDABA)]Cl2 was acquired in just 8 minutes at 14.1 T and MAS rate of νrot = 15 kHz (Fig. 1c), revealing a SSB manifold closely resembling the simulated ideal spectrum (Fig. 1a). Spectral acquisition via BRAIN-CP/WCPMG-MAS (Fig. 1c) is more efficient than with WCPMG-MAS (i.e., by more than an order of magnitude, 8 minutes vs. 3.5 hours; Fig. 1b, see Scheme S2 for the pulse sequence) for two reasons: (i) the 1H–119Sn BRAIN-CP/WCPMG-MAS is efficient, resulting in enhanced signal intensity; and (ii) T1(1H) is much shorter than T1(119Sn), allowing for shorter recycle delays. The spectral distortions manifesting as low intensity peaks between the SSBs are from the use of short τSE values (Fig. S1, see Section 3.4) in an effort to maximize SNR.
In the sections that follow, we demonstrate that 1H-S BRAIN-CP/WCPMG-MAS spectra of similar quality can be acquired after optimization of several experimental parameters: the transmitter offset (ΩS = ω0,S − ωTx,S, Section 3.2), the RF amplitudes on the 1H and S channels (ω1,I = 2π·ν1,I and ω1,S = 2π·ν1,S, respectively, Section 3.3), and matching of the sweep width (Δ) to the MAS rate (ωrot = 2π·νrot, Section 3.4). As detailed below, BRAIN-CP/WCPMG-MAS offers further advantages with respect to conventional CP, such as reduced power requirements, increased bandwidths for polarization transfer, and high efficiency at slow to moderate MAS rates (10 to 25 kHz).
3.2 FS pulses for spin-locking under MAS
Crucial to the success of any direct-detection CP experiment is the ability to efficiently spin lock the S magnetization produced from the I source magnetization. Pell and coworkers have demonstrated the ability to efficiently spin lock S magnetization across UW frequency ranges using S3APs in inversion experiments.28 Importantly, these experiments were limited to inversion of spin polarization and did not involve cross-polarization. Early stages of the work with S3APs were limited to the “low-power” regime, delineated by the condition |ω1,S| ≪ |ωrot|,25 wherein the pulse irradiates only one SSB and thus avoids interference effects from its neighboring sidebands. However, later findings suggest this low power condition is not a strict requirement, especially in applications to UW SSB manifolds where values of |ω1,S| ≈ |ωrot| also result in efficient inversion.28Previous experiments involving the use of S3APs for inversion ensured that ΩS was set such that the frequency sweep traversed the resonance frequency of the SSB of highest intensity.25 In the low-power limit, this ensures inversion of the highest efficiency due to the fact that the most intense SSB represents a frequency at which resonance between most isochromats and the pulse is achieved at some point throughout the rotor period. Yet, efficient inversion has been reported with ΩS set to irradiate other SSBs of lower signal intensities, suggesting this condition need not be strictly met.27 Furthermore, complete inversion of all isochromats is not a rigid requirement for acquisition of a SSB manifold that allows for extraction of the corresponding CS tensor.
The ability to achieve efficient spin locking at multiple combinations of ω1,S and ΩS provides experimental flexibility, yet also raises questions concerning which (if any) of these combinations are superior. Numerical simulations (Fig. S2) demonstrate that the choice of the transmitter offset (ΩS/2π) can be correlated with the inversion efficiency, especially with the use of spin locking powers exceeding the MAS rate.
The correlation between the optimal value of ΩS/2π and inversion efficiency can be rationalized through an analysis of the Hamiltonian describing the S3AP in the jolting frame (J-frame). First introduced by Caravatti and coworkers,43 the J-frame is one that rotates synchronously with the time dependent chemical shift frequency for a single isochromat and is necessarily orientation dependent. Generally, FS pulses are described in the frequency modulated (FM) frame,44 which is one wherein the time dependence of the phase arises in the form of a linear frequency sweep. Accordingly, a description of the transformations from the FM frame to the J-frame to the MAS frame is presented in S2. The final frame represents one wherein the time dependence of the CS has been transferred to the RF pulse amplitude term with the effective frequency given by:
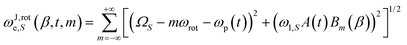 | (1) |
 are the Fourier coefficients of the mth SSB in the J-frame wherein ωJ,rote,S(β, t, m) is simultaneously swept across each mth SSB with an amplitude ω1,S scaled by the factor Bm(β) (see eqn (S12) for definition). Each component of ωJ,rote,S(β, t, m) can be evaluated in the J-frame in terms of its effective offset, ΩeS(t, m) = ΩS − mωrot − ωp(t), and effective amplitude, ωe1,S(t, m) = ω1,SA(t)Bm. In the event that ΩeS(t, m) ≫ ωe1,S(t, m) at all times throughout the duration of the pulse, the ωe1,S(t, m) terms can be ignored, allowing for an approximation of eqn (1) as:
are the Fourier coefficients of the mth SSB in the J-frame wherein ωJ,rote,S(β, t, m) is simultaneously swept across each mth SSB with an amplitude ω1,S scaled by the factor Bm(β) (see eqn (S12) for definition). Each component of ωJ,rote,S(β, t, m) can be evaluated in the J-frame in terms of its effective offset, ΩeS(t, m) = ΩS − mωrot − ωp(t), and effective amplitude, ωe1,S(t, m) = ω1,SA(t)Bm. In the event that ΩeS(t, m) ≫ ωe1,S(t, m) at all times throughout the duration of the pulse, the ωe1,S(t, m) terms can be ignored, allowing for an approximation of eqn (1) as: | (2) |
The theoretical insight gained from investigation into pulse parameters enabling efficient spin-locking with S3APs is used to guide choices for optimizing the BRAIN-CP/WCPMG-MAS experiment. In all cases, the condition
| 2πΔ = ω1,S = ωrot | (3) |
3.3 FS pulses for broadband CP under MAS
Optimal fulfillment of the HH matching conditions ensures maximum polarization transfer efficiency in any CP experiment. The HH matching conditions for CP/MAS experiments employing standard rectangular pulses (i.e., no amplitude or phase modulations) are well understood.45–47 Due to sample rotation, the dipolar frequency takes on a time dependence and can be expressed as , where bm are the Fourier coefficients. Provided ωrot > |bm|, a time dependence is imparted upon the HH matching conditions, which can be expressed in a frame rotating synchronously with the rotor as45,48
, where bm are the Fourier coefficients. Provided ωrot > |bm|, a time dependence is imparted upon the HH matching conditions, which can be expressed in a frame rotating synchronously with the rotor as45,48| ωe,I ± ωe,S = ±n·ωrot | (4) |
Use of a S3AP introduces a time dependence to eqn (4):
| ω1,I ± ωe,S(t) = ±n·ωrot | (5) |
| ωe,S(t) = [(ΩS − ωp(t))2 + (ω1,SA(t))2]1/2. | (6) |
A wide range of matching conditions, as determined by various combinations of offsets and amplitudes, are available in BRAIN-CP/MAS experiments and the selected conditions influence the resulting sign of 〈Sz〉 upon conclusion of the contact pulse (NB: distinction is made between BRAIN-CP/MAS, referring only to the CP period, and BRAIN-CP/WCPMG-MAS, referring to the experimental combination of BRAIN-CP with WCPMG-MAS). The effective offset, ΩS,e(t) = ΩS − ωp(t), is defined from eqn (6) and its instantaneous sign dictates that of 〈Sz〉. The impact of the sign of ΩS,e(t)/2π is evaluated through inspection of the time trajectories of both 〈Ix〉 and 〈Sz〉 using different values of ΩS/2π (Fig. 3). In all simulations, the direction of the frequency sweep is from low to high frequency and ν1,I = 10 kHz. The time trajectories of ΩS,e(t)/2π for two different values of ΩS/2π are shown relative to the resonance frequency of the S spin (Fig. 3a). The two cases for consideration are ΩS/2π = +5 kHz (Fig. 3b and c) and +10 kHz (Fig. 3d and e). Solutions to eqn (5) (Fig. 3b and d) predict the fulfillment of DQ matching conditions at times close to the start of the FS for both cases. Indeed, the simultaneous buildups of 〈Sz〉 and decreases in 〈Ix〉 agree with this prediction (Fig. 3c and e). The opposite signs of 〈Sz〉 towards the start of the contact pulse in Fig. 3c and e are attributed to the signs of ΩS,e(t)/2π relative to the resonance frequency of the target spin isochromat during the time at which the matching conditions are met: an offset of ΩS/2π = +5 kHz makes ΩS,e(t)/2π negative at this point, whereas for ΩS/2π = +10 kHz, ΩS,e(t)/2π is positive at all times (Fig. 3a). In the case of ΩS/2π = +5 kHz (Fig. 3c), the resonance condition is traversed shortly after the matching condition is met, which results in the inversion of Sz (i.e., 〈Sz〉 < 0). Inversion is not observed in the case of ΩS/2π = +10 kHz (Fig. 3d and e) as the resonance condition is avoided. The fulfillment of additional matching conditions in the latter case explains the observed oscillatory transfer between the I and S spin polarization. Changing the sign of ΩS to a negative value results in 〈Sz〉 with signs opposite to those discussed above, as does reversing the direction of the frequency sweep from high to low frequency (Scheme S3). Thus, it is a combination of factors that affect the sign of 〈Sz〉 produced in the BRAIN-CP/MAS experiment: the sign of ΩS, the sign of ΩS,e(t) at the point in time in which the match is fulfilled, the direction of the frequency sweep, and whether or not the sweep traverses the resonance condition. Additionally, the nature of the matching condition (i.e., either ZQ or DQ) influences the sign of 〈Sz〉 produced in BRAIN-CP/MAS experiments (Fig. S5).
 | ||
| Fig. 3 (a) Ideal simulation for the isochromat oriented at β = 0° with respect to the rotor axis. Purple and green arrows indicate the frequency ranges traversed by S3AP's centered at ΩS/2π = 5 kHz and 10 kHz, respectively. Analytical (b) and (d) and numerical (c) and (e) simulations for BRAIN-CP/MAS I → S polarization transfer during a S3AP under MAS conditions of νrot = 15 kHz using ΩS/2π = 5 kHz (b) and (c) and 10 kHz (d) and (e). Analytical plots show DQ solutions to eqn (5) of the main text. Numerical simulations make use of a two-spin system (I = 1H, S = 13C) with the 13C chemical shift tensor parameters described by: δiso = 0 ppm, ΩCS = 1350 ppm, and κ = −1. In all cases, ν1,I = 10 kHz and Δ = ν1,S = νrot = 15 kHz. The contact time τCT is 10 ms and the heteronuclear dipolar coupling is given by bISD/2π = 4 kHz with the dipolar vector oriented at θ = 45° relative to the rotor. | ||
Similarities between conventional CP/MAS and BRAIN-CP/MAS are observed, despite the time dependence of the HH matching conditions in the latter which can convolute such comparisons. The sign of the magnetization produced in both experiments depends on both the sign of the effective frequencies on both channels fulfilling the matching condition and the nature of the matching condition.49 In principle, a plot demonstrating the HH matching conditions for CP/MAS experiments in a manner similar to that of BRAIN-CP/MAS experiments (Fig. 2) could be envisioned. However, there exist no previous studies showcasing such results. This is largely due to the fact that CP/MAS experiments targeting spin-1/2 nuclides typically employ RF pulses with amplitudes on the order of 50 kHz to ensure efficient spin-locking. The potential for fulfilling as many matching conditions as are depicted in Fig. 2 only arise with the unique combination of low power RF pulses and slow MAS rates. Such experimental protocols have the distinct effect of clustering the matching conditions together in small frequency ranges relative to those at faster MAS rates and/or those with the use of higher powers. Thus, while the spin physics governing the polarization transfers in both CP/MAS and BRAIN-CP/MAS experiments are similar, it is the ability to efficiently spin-lock S magnetization at low RF powers using S3APs that differentiates the two experiments and offers the potential for a wide range of accessible matching conditions in the BRAIN-CP/MAS experiment.
| ω1,I = ωJ,rote,S(β, t, m) ± n·ωrot | (7) |
Differing only by the orientation of the S spin isochromat as determined by the angle β, a spin system otherwise identical to the one described above for I–S BRAIN-CP/MAS transfer (Fig. 2 and Fig. S5) is used to demonstrate the influence of a time-dependent chemical shift on the HH matching conditions. The isochromat described by β = 35° is chosen and simulated values of 〈Sz〉 are used to create a contour plot (Fig. 4) that enables visualization of the HH matching conditions as a function of ν1,I and ΩS/2π, as was done similarly in Fig. 2. This time, the observed matching conditions follow a strikingly different pattern. Narrow bands of light yellow and dark red, again indicating efficient polarization to produce high values of 〈Sz〉 in the positive and negative directions, respectively, are centered around each SSB.
A J-frame analysis can be used to rationalize the HH matching conditions for the case of the time-dependent chemical shift, as verified by a comparison of the analytical and numerical simulations (Fig. S8). As in the case of the isotropic chemical shift, the sign of 〈Sz〉 at t = τCT hinges upon several factors, including the direction of the FS, the nature of the matching condition, and the relative time occurrences of the matching condition and the traversal of the resonance condition. Interestingly, the J-frame analysis results in a description of several matching conditions met simultaneously as a result of the consideration of each individual component of ωJ,rote,S(β, t, m)/2π (Fig. S8).
Qualitative agreement between experimental and theoretical data is observed, yet several discrepancies exist. The most apparent difference is the dependence of the observed matching conditions on the parameters ΩS/2π and ν1,I (Fig. S10). While simulations predict narrow-banded matching conditions (Fig. 5), experimental data suggests a broadening of these conditions (Fig. S11), with maximal signal obtained when ΩS/2π is between two consecutive SSBs and ν1,I takes on a value between half-integer multiples of νrot. Several factors could contribute to such behavior, including the inability to perfectly model spin dynamics using a two-spin system (see Fig. S12), broadening of the matching conditions due to the range of homonuclear dipolar couplings present in solids, and/or RF inhomogeneity. However, both theory and experiment similarly predict null signal obtained using ν1,I of half-integer multiples of νrot. In contrast to simulations which predict maximal CP efficiency using values of ν1,I closer to νrot, experimentally it is maximized at higher values of ν1,I. This is attributed to inefficient spin locking on the I channel using low RF pulse powers (i.e., short T1ρ(1H), see Fig. S13). Drawing upon the results presented in this section, a recommended starting value of ν1,I is one halfway between the half-integer multiples of νrot. At slow MAS rates, higher powers are recommended to ensure efficient spin-locking.
3.4 On the choice of MAS rate and contact time
The choice of MAS rate for BRAIN-CP/WCPMG-MAS experiments is somewhat arbitrary and ultimately left to the experimentalist. In general, νrot > 10 kHz seems to be advantageous in terms of maximizing SNR, but, as with most choices in NMR, there are pros and cons associated with the specific choice of νrot. The optimal value for νrot ultimately depends on the attainable spin locking amplitudes (due to the requirement that ν1,S = νrot) and width of the SSB manifold (due to the direct relationship between νexc/ref and νrot·ΩCS, see Section 3.2). The use of very slow MAS rates is likely not optimal as spin-locking efficiency is expected to suffer. Simulations and experiments similar to those presented in Fig. 5 and 6 were carried out at a νrot = 10 kHz (Fig. S14 and S15), with results similar to those discussed above. While uniform experimental SSB manifolds are produced at different values of νrot, faster MAS rates generally result in higher SNRs. This can be attributed to two factors: (i) higher MAS rates tend to give increased SNRs due to localization of signal in fewer SSBs; and (ii) the dependence of the HH match on νrot is such that faster MAS rates demand higher values for ν1,I, which in turn results in better 1H spin locking. This latter point comes with several caveats: (i) high-powered pulses on both channels place more demand on the NMR probe, especially for experiments on low-γ nuclides; (ii) larger values of νrot decrease the number of SSBs, which can cause an increased dependence of the BRAIN-CP/MAS experiment on ΩS/2π; (iii) faster MAS rates also require increased rates Rexc/ref for the WCPMG excitation and refocusing pulses, in order to obtain high values of x,14 which in turn require higher amplitudes;51 and (iv) very fast MAS rates are known to reduce CP efficiency and would thus introduce further complications to the experiment.52,53Directly dependent on the MAS rate is the spacing of rotary echoes in each CPMG loop. As mentioned in Section 3.1, the echo length, τSE, and thus, the number of rotary echoes acquired, influence the appearance of the NMR spectra, with shorter echo lengths resulting in spectral distortions present in many of the spectra shown herein. However, shorter values of τSE allow for the acquisition of more CPMG echoes and thus result in spectra with higher SNRs, albeit with decreased resolution (this poses no issues if only one site is expected). We note that these distortions do not hinder the extraction of CS tensor parameters and thus, the choice of τSE is left to the experimentalist.
A final comment is directed towards the choice of the contact time, τCT. As with conventional CP (both static and MAS) and static BRAIN-CP experiments, intuition concerning the relative magnitudes of the heteronuclear couplings can help determine initial values for τCT. However, the optimal value of τCT is generally deciphered through experimental optimizations (Fig. S16). Values of τCT below several milliseconds are not advised, as these are unlikely to allow for sufficient polarization transfer and run the additional risk of decreasing the spin locking efficiency.
4. Experimental examples
Having established the theoretical principles governing the spin dynamics in the BRAIN-CP/WCPMG-MAS, several test cases are presented that demonstrate its capability, versatility, and robustness. First, a case of a non-axially symmetric 119Sn CS tensor is investigated. Then, a 195Pt example is chosen to showcase the ability to CP across bandwidths approaching 1 MHz. Finally, an example of 103Rh SSNMR is shown as a demonstration of the acquisition of wideline SSNMR SSB manifolds of low-γ nuclides.4.1 Wideline 1H–119Sn BRAIN-CP/MAS NMR: a case of a non-axially symmetric CS tensor
Thus far, all theoretical and experimental examples have focused on S nuclides with axially symmetric CS tensors; hence, the case of DBTO is presented to demonstrate that the principles outlined above also apply to systems in which the CS tensor is not axially symmetric. Excellent agreement is observed between the ideal and experimental 119Sn SSB manifolds of DBTO acquired at 14.1 T and with νrot = 12 kHz (Fig. 7). As in the case of axially symmetric CS tensors, values of ΩS/2π that position the transmitter near the edge of the SSB manifold provide more efficient spin locking and thus result in more efficient CP (see again Fig. S4). The experimentally optimized matching conditions are ν1,S = 12 kHz and ν1,I = 40 kHz. Predictions for the optimal HH matching conditions for ν1,I falling between both odd and even half-integer multiples of νrot include ν1,I ≈ 15, 21, 27, or 43 kHz, for which the optimized value of ν1,I = 40 kHz is close.Similar to the case of [Sn(HPDABA)]Cl2, the T1(119Sn) is several orders of magnitude larger than the T1(1H) and thus, 1H–119Sn BRAIN-CP/WCPMG-MAS experiments represent a much more efficient method of acquisition relative to DE methods.54 While spectral acquisition via BRAIN-CP/WCPMG-MAS was accomplished in just 32 seconds (Fig. 7), WCPMG-MAS acquisition with the same number of scans is predicted to take ca. 7 hours and was thus not attempted.
4.2 Ultra-wideline 1H–195Pt BRAIN-CP/MAS experiments: pushing the CP bandwidth capability
195Pt is oftentimes selected as a target for methods aimed at the acquisition of wideline and UW NMR powder patterns.2,55–58 The 195Pt NMR SSB manifold of cisplatin spans 965 kHz (8975 ppm)59 in breadth at 11.7 T (Fig. 8). Here, the WCPMG-MAS (Fig. 8b) and BRAIN-CP/WCPMG-MAS (Fig. 8c) NMR experiments at MAS rates of νrot = 25 kHz both enable acquisition of 195Pt NMR spectra displaying the entire SSB manifold at a single transmitter frequency; however, the experimental time required for acquisition of the BRAIN-CP/WCPMG-MAS spectrum (Fig. 8c) is significantly shorter than that of the WCPMG spectrum of similar quality (21 min. compared to 4.3 hours) due to the shorter recycle delay in CP experiments afforded from the shorter T1(1H) relative to the T1(195Pt). Starting with a predicted value of ν1,I ≈ 44 kHz, that obtained from experimental optimizations is ν1,I = 42 kHz.The large span of the 195Pt NMR SSB manifold of cisplatin likely serves to reduce the offset dependency of the BRAIN-CP/WCPMG-MAS experiment and thus tuning ΩS/2π adjacent to any SSB should provide sufficient spin locking. To minimize the RF amplitude required for WURST excitation and refocusing pulses, ΩS/2π is set close to the center of the SSB manifold. Deviations from this central position would require an increase in the already demanding RF amplitude of νRF = 87 kHz. Importantly, the amplitude shape modulation parameter N is set to 2, which has been shown to greatly facilitate the excitation and refocusing capability of such pulses in applications to UW SSB manifolds.14
4.3 Wideline 1H–103Rh BRAIN-CP MAS experiments: a low-gamma application
103Rh presents a challenging case for acquisition of NMR signal due to its very low gyromagnetic ratio (γ = –0.8486 × 107 rad T−1 s−1). Accordingly, there are very few examples of 103Rh SSNMR spectra to date.60–64 Rh2Cl2(COD)2 has favorable properties for testing the applicability of BRAIN-CP/WCPMG-MAS for the acquisition of SSB manifolds of low-γ nuclides with small heteronuclear dipolar couplings, including a high wt% of Rh and short T1(1H), as estimated from an optimized recycle delay of 2 s. The 103Rh NMR SSB manifold (Fig. 9) is in agreement with the ideal simulated SSB manifold. Experimental acquisition was accomplished in 2.3 hours at νrot = 14 kHz using experimentally optimized matching conditions of ν1,S = 14 kHz and ν1,I = 35 kHz. For the same reasons as those outlined in Section 4.2, the transmitter was tuned near the center of the pattern. As observed in other spectra presented herein, the peaks present between SSBs are a result of the use of short τSE (see Section 3.4).5. A summary of guidelines for setting up BRAIN-CP/WCPMG-MAS experiments
Here, we provide a set of guidelines for preliminary setup of the BRAIN-CP/WCPMG-MAS pulse sequence. It is noted that as with conventional CP/MAS experiments, there is some degree of optimization necessary – but the following information provides a good starting point, at the very least.(i) The choice of MAS rate, νrot: based on this work, MAS rates between 10 and 25 kHz are recommended. Higher rates allow for more efficient 1H spin locking yet also decrease the number of SSBs and thus may introduce an offset dependency. Furthermore, higher MAS rates required higher S RF pulse amplitudes and thus may not be attainable for low-γ nuclei. MAS rates higher than 25 kHz were not explored, but could be the subject of future work.
(ii) Phase and amplitude of S3AP: the condition 2πΔ = ω1,S = ωrot (eqn (3)) should be fulfilled in order to provide efficient spin locking.
(iii) Contact pulse length, τCT: pulses on the order of several or tens of milliseconds are optimal. Intuition as to the relative magnitudes of the heteronuclear dipolar couplings can be used for starting points, as is done with conventional CP/MAS experiments.
(iv) Transmitter offset, ΩS: the exact value of ΩS should be determined through experimental optimizations, but its optimal values are predicted to normally occur halfway between the half-integer multiples of νrot. If the symmetry of the CS tensor is known, ΩS should be tuned adjacent to the SSBs of lower intensities. If δiso is known, ΩS can be preliminarily set adjacent to δiso, in either the positive or negative direction. For UW NMR spectra (e.g., spans in excess of 200 kHz), the choice of tuning to a specific SSB becomes less critical (Fig. 5). Nevertheless, a final optimization of ΩS in an interval of −ωrot/2 to +ωrot/2 in steps of 1 or 2 kHz around the initially chosen SSB should be conducted in order to optimize CP efficiency.
6. Conclusions
In this work, it is shown how the BRAIN-CP/WCPMG-MAS pulse sequence can be used to acquire wideline and UW spectra of spin-1/2 nuclides featuring SSB manifolds arising from CSA, under conditions of moderate MAS rates (10–25 kHz) and with S contact pulses with low RF amplitudes. Provided the condition νrot = ν1,S = Δ is met, both DQ and ZQ HH matching conditions can be satisfied when ΩS is set adjacent to any of the SSBs of the target NMR SSB manifold and ν1,I is within a few kHz of a half-integer multiple of νrot/2. Without knowledge of the exact resonance frequency of a SSB, experimental optimizations of ΩS and ν1,I are generally required. However, an intuition (i.e., from direct crystallographic information regarding the site symmetry and/or DFT calculations) about the axial symmetry of the CS tensor of the S nucleus can aid in the optimization of ΩS. The examples of applications of the BRAIN-CP/WCPMG-MAS sequence discussed herein feature nuclides with different gyromagnetic ratios and CS tensors of varying magnitude and symmetry, serving to demonstrate the robustness of this method. We hope that BRAIN-CP/WCPMG-MAS can aid in the routine acquisition of NMR spectra of spin-1/2 nuclides and certain quadrupolar nuclides (with small quadrupolar interactions) having anisotropically broadened patterns arising from CSA and possibly even Knight shift anisotropy. Furthermore, this robust method may even be useful in DNP experiments for the enhancement of wideline and UW NMR spectra, e.g., Pt catalytic centers on surfaces or a wide range of heterogeneous catalysts.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Pulse sequences used for collection of all data used for main text figures are available at https://github.com/rschurko. Representative setup files sufficient to reproduce the simulation results are provided in the supplementary information (SI). Supplementary information contains description of [Sn(HPDABA)]Cl2 co-crystal synthesis, additional pulse program schemes. Experimental parameters for all acquired BRAIN-CP/WCPMG-MAS and WCPMG-MAS spectra, analytic derivation of effective S nuclei nutation frequency in the jolting frame, supplementary figures (simulations and experiments), BRAIN-CP/WCPMG-MAS pulse sequence for SIMPSON simulations. See DOI: https://doi.org/10.1039/d5cp03931b.Acknowledgements
J. J. K. and R. W. S. would like to thank the Basic Energy Sciences Program in the Department of Energy (DE-SC0022310) for supporting this work. Early stages of these projects were supported by the National Science Foundation Chemical Measurement and Imaging Program, with partial co-funding from the Solid State and Materials Chemistry Program (NSF-2003854). Some NMR spectra were acquired at the National High Magnetic Field Laboratory, which is supported by the National Science Foundation (NSF/DMR-1644779, NSF/DMR-2128556) and the State of Florida. Some of the probe technologies used in this study were developed via resources provided by the National Resource for Advanced NMR Technology, an NIH RM1 center (RM1 GM148766). This study made use of NMRbox: National Center for Biomolecular NMR Data Processing and Analysis, a Biomedical Technology Research Resource (BTRR), which is supported by NIH grant P41GM111135 (NIGMS). J. J. K. and R. W. S. are grateful to Dr Zhehong Gan (NHMFL) for helpful discussions on spin dynamics. Jazmine Sanchez is thanked for synthesis of [Sn(HPDABA)]Cl2.References
- R. W. Schurko, Acc. Chem. Res., 2013, 46, 1985 CrossRef CAS PubMed.
- T. Kobayashi, F. A. Perras, T. W. Goh, T. L. Metz, W. Huang and M. Pruski, J. Phys. Chem. Lett., 2016, 7, 2322 CrossRef CAS PubMed.
- A. Venkatesh, M. P. Hanrahan and A. J. Rossini, Solid State Nucl. Magn. Reson., 2017, 84, 171 CrossRef CAS PubMed.
- A. J. Rossini, M. P. Hanrahan and M. Thuo, Phys. Chem. Chem. Phys., 2016, 18, 25284 RSC.
- L. A. O’Dell and R. W. Schurko, Chem. Phys. Lett., 2008, 464, 97 CrossRef.
- A. Tannús and M. Garwood, NMR Biomed., 1997, 10, 423 CrossRef.
- M. Garwood and L. DelaBarre, J. Magn. Reson., 2001, 153, 155 CrossRef CAS PubMed.
- J. M. Bohlen, M. Rey and G. Bodenhausen, J. Magn. Reson., 1989, 84, 191 Search PubMed.
- J. M. Böhlen, I. Burghardt, M. Rey and G. Bodenhausen, J. Magn. Reson., 1990, 90, 183 Search PubMed.
- V. L. Ermakov, J. M. Böhlen and G. Bodenhausen, J. Magn. Reson., Ser. A, 1993, 103, 226 CrossRef CAS.
- Ē. Kupče and R. Freeman, J. Magn. Reson., Ser. A, 1995, 115, 273 CrossRef.
- L. A. O’Dell, Solid State Nucl. Magn. Reson., 2013, 55–56, 28 CrossRef PubMed.
- A. W. MacGregor, L. A. O’Dell and R. W. Schurko, J. Magn. Reson., 2011, 208, 103 CrossRef CAS PubMed.
- J. Koppe, M. Bußkamp and M. R. Hansen, J. Phys. Chem. A, 2021, 125, 5643 CrossRef CAS PubMed.
- S. L. Veinberg, A. W. Lindquist, M. J. Jaroszewicz and R. W. Schurko, Solid State Nucl. Magn. Reson., 2017, 84, 45 CrossRef CAS PubMed.
- K. J. Harris, A. Lupulescu, B. E. G. Lucier, L. Frydman and R. W. Schurko, J. Magn. Reson., 2012, 224, 38 CrossRef CAS PubMed.
- A. R. Altenhof, S. Wi and R. W. Schurko, Magn. Reson. Chem., 2021, 59, 1009 CrossRef CAS PubMed.
- J. J. Kimball, A. R. Altenhof, M. J. Jaroszewicz and R. W. Schurko, J. Phys. Chem. A, 2023, 127, 9621 CrossRef CAS PubMed.
- S. Wi, Z. Gan, R. Schurko and L. Frydman, J. Chem. Phys., 2015, 142, 064201 CrossRef PubMed.
- S. Wi, R. Schurko and L. Frydman, J. Chem. Phys., 2017, 146, 43 CrossRef PubMed.
- S. Wi, R. W. Schurko and L. Frydman, Solid State Nucl. Magn. Reson., 2018, 94, 31 CrossRef CAS PubMed.
- D. Massiot, I. Farnan, N. Gautier, D. Trumeau, A. Trokiner and J. P. Coutures, Solid State Nucl. Magn. Reson., 1995, 4, 241 CrossRef CAS PubMed.
- I. Hung, A. J. Rossini and R. W. Schurko, J. Phys. Chem. A, 2004, 108, 7112 CrossRef CAS.
- A. J. Pell, G. Pintacuda and L. Emsley, J. Chem. Phys., 2011, 135, 144201 CrossRef PubMed.
- A. J. Pell, G. Kervern, L. Emsley, M. Deschamps, D. Massiot, P. J. Grandinetti and G. Pintacuda, J. Chem. Phys., 2011, 134, 1 CrossRef PubMed.
- A. J. Pell, K. J. Sanders, S. Wegner, G. Pintacuda and C. P. Grey, J. Chem. Phys., 2017, 146 Search PubMed.
- K. J. Sanders, A. J. Pell, S. Wegner, C. P. Grey and G. Pintacuda, Chem. Phys. Lett., 2018, 697, 29 CrossRef CAS.
- J. P. Carvalho and A. J. Pell, J. Magn. Reson., 2021, 324, 42 CrossRef PubMed.
- J. S. Frye and G. E. Maciel, J. Magn. Reson., 1982, 48, 125 CAS.
- H. J. Jakobsen, V. Langer, P. Daugaard and H. Bildsøe, A Device and Method for Magic-Angle Adjustment with Millidegree Accuracy using MAS of Quadrupolar Nuclei. In 31st Experimental NMR Conference, Asilomar, CA, 1990.
- A. R. Altenhof, A. W. Lindquist, L. D. D. Foster, S. T. Holmes and R. W. Schurko, J. Magn. Reson., 2019, 309, 106612 CrossRef CAS PubMed.
- R. K. Harris, E. D. Becker, S. M. Cabral De Menezes, R. Goodfellow and P. Granger, Concepts Magn. Reson., 2002, 14, 326 CrossRef CAS.
- J. C. Edwards and P. D. Ellis, Magn. Reson. Chem., 1990, 28, S59 CrossRef CAS.
- A. R. Altenhof, M. J. Jaroszewicz, A. W. Lindquist, L. D. D. Foster, S. L. Veinberg and R. W. Schurko, J. Phys. Chem. C, 2020, 124, 14730 CrossRef CAS.
- Y. Shrot and L. Frydman, J. Magn. Reson., 2005, 172, 179 CrossRef CAS PubMed.
- J. Koppe and M. R. Hansen, J. Phys. Chem. A, 2020, 124, 4314 CrossRef CAS PubMed.
- R. Bhattacharyya and L. Frydman, J. Chem. Phys., 2007, 127, 194503 CrossRef PubMed.
- F. H. Larsen, H. J. Jakobsen, P. D. Ellis and N. C. Nielsen, J. Phys. Chem. A, 1997, 101, 8597 CrossRef CAS.
- M. Bak, J. T. Rasmussen and N. C. Nielsen, J. Magn. Reson., 2000, 147, 296 CrossRef CAS PubMed.
- J. Herzfeld and A. E. Berger, J. Chem. Phys., 1980, 73, 6021 CrossRef CAS.
- N. C. Nielsen and M. Bak, J. Magn. Reson., 1997, 125, 132 CrossRef PubMed.
- M. W. Maciejewski, A. D. Schuyler, M. R. Gryk, I. I. Moraru, P. R. Romero, E. L. Ulrich, H. R. Eghbalnia, M. Livny, F. Delaglio and J. C. Hoch, Biophys. J., 2017, 112, 1529 CrossRef CAS PubMed.
- P. Caravatti, G. Bodenhausen and R. R. Ernst, J. Magn. Reson., 1983, 55, 88 CAS.
- J. Baum, R. Tycko and A. Pines, Phys. Rev. A: At., Mol., Opt. Phys., 1985, 32, 3435 CrossRef CAS PubMed.
- E. O. Stejskal, J. Schaefer and J. S. Waugh, J. Magn. Reson., 1977, 28, 105 CAS.
- M. Sardashti and G. E. Maciel, J. Magn. Reson., 1987, 72, 467 CAS.
- I. Hung and Z. Gan, J. Magn. Reson., 2015, 256, 23 CrossRef CAS PubMed.
- X. Wu and K. W. Zilm, J. Magn. Reson., Ser. A, 1993, 104, 154 CrossRef CAS.
- B. H. Meier, Chem. Phys. Lett., 1992, 188, 201 CrossRef CAS.
- Z. H. Gan and D. M. Grant, Chem. Phys. Lett., 1990, 168, 304 CrossRef CAS.
- J. Koppe, J. E. Frerichs and M. R. Hansen, J. Phys. Chem. Lett., 2023, 14, 10748 CrossRef CAS PubMed.
- M. Deschamps, Ultrafast Magic Angle Spinning Nuclear Magnetic Resonance, Annual Reports on NMR Spectroscopy, Elsevier Ltd, Amsterdam, 2014, vol. 81, pp. 109–144 Search PubMed.
- Y. Nishiyama, G. Hou, V. Agarwal, Y. Su and A. Ramamoorthy, Chem. Rev., 2023, 123, 918 CrossRef CAS PubMed.
- M. J. Jaroszewicz, L. Frydman and R. W. Schurko, J. Phys. Chem. A, 2017, 121, 51 CrossRef CAS PubMed.
- P. Paluch, A. G. M. Rankin, J. Trébosc, O. Lafon and J. P. Amoureux, Solid State Nucl. Magn. Reson., 2019, 100, 11 CrossRef CAS PubMed.
- A. Venkatesh, F. A. Perras and A. J. Rossini, J. Magn. Reson., 2021, 327, 106983 CrossRef CAS PubMed.
- Z. Wang, L. A. Völker, T. C. Robinson, N. Kaeffer, G. Menzildjian, R. Jabbour, A. Venkatesh, D. Gajan, A. J. Rossini and C. Copéret, et al. , J. Am. Chem. Soc., 2022, 144, 21530 CrossRef CAS PubMed.
- M. J. Jaroszewicz, A. R. Altenhof, R. W. Schurko and L. Frydman, J. Am. Chem. Soc., 2021, 143, 19778 CrossRef CAS PubMed.
- B. E. G. Lucier, A. R. Reidel and R. W. Schurko, Can. J. Chem., 2011, 89, 919 CrossRef CAS.
- B. L. Phillips, J. R. Houston, J. Feng and W. H. Casey, J. Am. Chem. Soc., 2006, 128, 3912 CrossRef CAS PubMed.
- H. Harbor-Collins, M. Sabba, G. Moustafa, B. Legrady, M. Soundararajan, M. Leutzsch and M. H. Levitt, J. Chem. Phys., 2023, 159, 104307 CrossRef CAS PubMed.
- S. T. Holmes, J. Schönzart, A. B. Philips, J. J. Kimball, S. Termos, A. R. Altenhof, Y. Xu, C. A. O’Keefe, J. Autschbach and R. W. Schurko, Chem. Sci., 2024, 15, 2181 RSC.
- B. A. Atterberry, P. Paluch, A. R. Lamkins, W. Huang and A. J. Rossini, J. Am. Chem. Soc., 2025, 147, 14411 CrossRef CAS PubMed.
- J. J. Kimball and R. W. Schurko, J. Phys. Chem. Lett., 2025, 16, 4596 CrossRef CAS PubMed.
Footnote |
| † This work is dedicated to the memory of Professor Robert W. Schurko, whose thoughtful insight and guidance were instrumental to the success of this project. |
| This journal is © the Owner Societies 2026 |









