Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Tunable solvent-induced gelation of dipeptide-based gelators: exploring the role of solvent and acid concentration
Henna
Rahkola
 a,
Efstratios D.
Sitsanidis
a,
Efstratios D.
Sitsanidis
 a,
Romain
Chevigny†
a,
Romain
Chevigny†
 a and
Maija
Nissinen
a and
Maija
Nissinen
 *a
*a
aDepartment of Chemistry, Nanoscience Center, University of Jyväskylä, P.O. Box 35, FI-40014 JYU, Finland. E-mail: maija.nissinen@jyu.fi
bCurrent: Université de Lorraine, CNRS, IJL, F-54000 Nancy, France
First published on 20th January 2026
Abstract
The tunability of the solvent-induced gelation mechanism using tert-butyl (tBu) containing solvents and two tBu-protected dipeptide precursor gelators (Boc-Phe-Phe-OtBu – 1 and Boc-Leu-Phe-OtBu – 2) is reported. Gelation behaviour, network morphology, material stability, and gelators’ structures can be adjusted by both the solvent type and acid concentration. While tert-butyl chloroacetate (tBuClOAc) enables rapid gelation, tert-butyl methyl ether (tBuOMe), acting as a solvent with two leaving groups, promotes the in situ formation of two different gelators and prolongs the gelation time. Gel-to-sol transition temperature (Tgel–sol), NMR, HR-MS, ATR-FTIR and TEM analyses revealed that both the solvent type and acid concentration influenced the precursor gelator-to-gelator conversion efficiency, as well as the secondary structure (β-sheet and helical-like motif) and morphology of the resulting gels. This study highlights the adaptability of solvent-induced gelation across different solvent environments. In addition, the findings demonstrate that the solvent type and acid loading are powerful tools for tuning the properties of peptide-based supramolecular organogels with potential applications in biomedical and materials science.
Introduction
Low-molecular-weight gelators (LMWGs) are a diverse and useful class of compounds that can self-assemble, forming, for example, supramolecular gels.1–3 Particularly, amino acid- and peptide-based LMWGs have attracted attention due to their simple structures, biocompatibility, biodegradability, and natural abundance. Their chemical and physical properties can be easily modified by altering the amino acid sequence or utilising various functionalisation options, for example, by the addition of protective groups at the C- and N-termini. This further enhances their potential as gelator building blocks and for applications in various fields.4,5 By controlling the gelator structure, the solvent, and the gelation conditions, the desired properties of the gels can be tuned to a considerable extent.The self-assembly of gelator peptides is a spontaneous, thermodynamically and kinetically driven process based on the development of intermolecular non-covalent interactions,6 such as π–π stacking, hydrogen bonding, van der Waals forces and electrostatic interactions.2,3,7,8 Due to the weak and dynamic nature of these interactions, self-assembled structures (and their corresponding macroscopic materials) can be modified by altering several parameters, including pH, temperature, the presence of counterions, concentration, solvent type, and the type of gelation trigger.9–11 By controlling the kinetic parameters (e.g., heating/cooling rates, solvent exchange or evaporation rates, agitation-shear forces, and diffusion rates), inherently dynamic metastable materials can be created.6
Additionally, in dissipative self-assembled (DSA) systems, non-gelator precursor molecules are activated into gelators by external energy input (e.g., light, enzymes, or chemical fuels), leading to their assembly into ordered structures. Upon energy depletion, inhibition or intentional deactivation, the formed structures collapse, and initial precursors are formed.12–14 These chemically fuelled self-assembled systems are thermodynamically unfavourable and thus require a continuous external energy input to maintain their out-of-equilibrium state.15,16 In contrast, in-equilibrium self-assembled systems form stable and permanent structures that do not require continuous energy input. Although they may pass through dynamic, non-equilibrium intermediate states during formation, they eventually reach a thermodynamically favourable structure. Systems as such can exhibit continuous internal interconversion of states; however, once the system has reached equilibrium (or a favourable thermodynamic state), the forward and reverse processes occur at equal rates and do not affect the system's balance and stability.13,15
Transient supramolecular materials created by DSA have been extensively studied. Due to their limited lifetimes, such materials are attractive for applications in drug delivery platforms and self-abolishing systems.17 Properties including viscoelasticity, thermoreversibility, and dynamic reversibility enable gels to perform unique functions such as self-healing, responsiveness to external stimuli, and tolerance/adaptability to environmental changes. These properties are important for applications in pharmaceuticals (e.g., drug delivery), the food industry, and cosmetics.8,18
The solvent environment of the gel material can be, for example, water (hydrogels), an organic solvent (organogels), or a mixture of the two.8 In general, solvents do not participate actively in the self-assembly of gelator molecules but are instead encapsulated within the resulting gel network. Nevertheless, the solvent may affect the properties of the material, and in this case, its role is as important as the structure of the gelator. In some cases, the solvent can initiate the gelation process (e.g., solvent exchange-triggered gelation):11,19 the gelator solution is diluted with a non-solvent, and a balance is sought between the solubility and aggregation of the gelator molecules.
Usually, different solvents affect the gelation outcome primarily due to changes in solubility or being unsuitable for gel formation. Consequently, gelator–gelator and gelator–solvent interactions have a fundamental impact on self-assembly per se and on the properties of the resulting macroscopic gel material.20–24 Stable, in-equilibrium gels form when intermolecular interactions, solubility parameters, and the forces driving fibrillar aggregation are in a delicate balance.22,24,25 Reduced solvent–gelator interactions promote fibrillar growth, resulting in thinner, more flexible, and entangled fibres, whereas stronger gelator–gelator interactions favour the formation of thicker and shorter fibres.20,21 Fibres can further interact, leading to the formation of bundles and ribbons.24 Thus, the gelation process can be tailored by solvent properties, such as polarity and the ability to form weak interactions, including hydrogen bonds and π–π interactions.11,20,22,23,26
The choice of solvent can alter gel properties, such as mechanical strength, thermoreversibility, and thermal stability.8,18,19 The boiling and freezing points of organic solvents can also be used to control the operating temperature range of organogels. For instance, if high heat tolerance is required, using a solvent with a higher boiling point is sensible.8,18 Conversely, low boiling points cause solvents to evaporate more readily from the gel matrix, leading to gel shrinkage. Although this may be unfavourable for mechanical durability and thermal stability, it can be advantageous in applications where solvent removal is required.8 The solvent can also influence the system's morphology,9,26 for example, solvent-induced morphological changes can be seen as a structural transition. Indeed, Li et al.26 observed that a toluene-based organogel transforms from fibrous to microcrystalline morphology upon the addition of ethanol as a co-solvent.
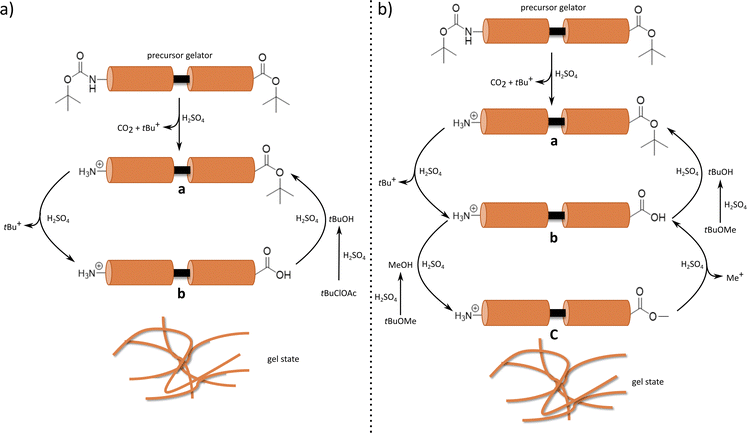
Recently, we introduced the concept of solvent-induced gelation (Scheme 1), in which the solvent (tert-butyl acetate, tBuOAc) is chemically active and participates in the formation of supramolecular organogels from various di- and tripeptides.27,28 This process occurs when the solvent and precursor gelators share the same protective group. Gelation proceeds through [regio]selective and irreversible deprotection of the N-Boc-protected peptide precursor gelator. Concurrently, tert-butyl (tBu) ester at the C-terminus undergoes reversible deprotection in the presence of tBuOAc (primary solvent) and sulfuric acid (accelerator). This reaction pathway yields two activated gelators (a and b) that interconvert via a hydrolysis–esterification cycle, leading to the formation of an organogel. At the same time, hydrolysis of the primary solvent (tBuOAc) produces an alcohol (secondary solvent; tert-butyl alcohol, tBuOH), providing tBu+ cations in the system and simultaneously acting antagonistically toward gelation. As the tBuOH concentration increases over time, it induces a gel-to-sol transition, resulting in the gradual dissolution of the organogel.
Based on previous research, we hypothesise that this gelation mechanism requires the presence of a common protective group in both the solvent and the precursor gelator.28 To further explore the generalisability of this solvent-induced process, we investigated gelation in other tBu-group-containing solvents and examined the effect of the amount of acid in these systems.
Results and discussion
Gel fabrication and gelation outcome
Based on our previous studies,28 two dipeptide precursor gelators were selected for the solvent effect studies: Boc-Phe-Phe-OtBu (1, gel system I) and Boc-Leu-Phe-OtBu (2, gel system II) (Fig. 1). Four tBu-group-containing solvents, tert-butyl chloroacetate (tBuClOAc), tert-butyl methyl ether (tBuOMe), tert-butyl acetoacetate (tBuAcAc), and tert-butyl formate (tBuOCHO), were chosen to investigate the gelation mechanism and self-assembly behaviour. Additionally, varying amounts of sulfuric acid (H2SO4; 1.0, 0.5 and 0.18 eq.) were used to assess the impact of acid concentration on gelation. This design allows us to examine whether shared functional motifs between the solvent and gelator contribute to the solvent-induced gelation, and how acid concentrations adjust the rate and extent of in situ precursor gelator activation, govern the efficiency of gel formation, and influence gel stability. | ||
| Fig. 1 The structures of precursor gelators 1 and 2, monoprotected gelators 1a–1c and 2a–2c and deprotected gelators 1b–2b. | ||
All gel samples were prepared following the same protocol (experimental and S3.1, SI) using a constant precursor gelator concentration of 50 mM throughout the study. In addition, control gelation experiments were performed using the identified gelator molecules (1a–c and 2a–c) alone to determine which of the in situ formed gelators can act as organogelators independently (Table S2, SI). The gelation outcome was evaluated using the vial-inversion method. Samples that “solidified” without any detectable free gravitational flow upon vial inversion were classified as self-supporting gels (SSGs). Those exhibiting “half-solidified” or weakly gel-like precipitate, along with flowable characteristics, were designated as partial gels (PGs), while samples showing no visible change were classified as solutions (Sol) (Table 1 and Table S1, SI). It is noteworthy that precursor gelator 1 is not soluble in tBuOMe, resulting in the formation of heterogeneous gels (Fig. S5, SI). However, upon heating these gels to their gel-to-sol phase transition temperature (Tgel–sol) and subsequently cooling to room temperature, homogeneous gels were obtained.
| Precursor gelator | Acid (eq.) | tBuClOAc | T gel–sol (°C) | tBuOMe | T gel–sol (°C) |
|---|---|---|---|---|---|
| a Measured from PG. b Lifespan: 2 days. | |||||
| Boc-Phe-Phe-OtBu (1) | 1.0 | Sol | — | SSG, transparent/opaque | 55–60 |
| 0.5 | SSG, opaque | 45–50 | SSG, transparent/opaque | 60–65 | |
| 0.18 | SSG, opaque | 55–60 | SSG, transparent/opaque | 60–65 | |
| Boc-Leu-Phe-OtBu (2) | 1.0 | SSG, opaqueb | 40–45 | SSG, transparent | 55–60 |
| 0.5 | SSG, transparent | 50–55 | SSG/PG, transparent | 60–65a | |
| 0.18 | SSG, transparent | 55–60 | PG, transparent | 60–65a | |
Gelation was not observed in tBuAcAc and tBuOCHO solvents under the tested conditions (Table S1, SI). Therefore, these were excluded from further study. In contrast, both gel systems I and II formed SSGs or PGs in tBuClOAc and tBuOMe, except for the sample with the highest amount of acid (1.0 eq.) in gel system I in tBuClOAc. The resulting organogels remained stable at room temperature for several months, except for gel system II in tBuClOAc and 1.0 eq. of acid, which reverted to a sol within two days after gelation. A similar transient behaviour was observed in our previous studies.27,28 To evaluate whether the transient property of this gel system is caused by the formation of sufficient tBuOH to dissolve the gel system, the effect of tBuOH was tested on the highest-equivalent gel systems (S3.2, SI). After material formation, tBuOH (500 µL) was added on top of the materials. Here, it was found that precursor gelator 2-based systems dissolved within 1 day, whereas precursor gelator 1-based systems did not dissolve completely with the same amount of tBuOH, but required additional tBuOH (500 µL) to dissolve. This indicates that precursor gelator 2-based systems are more susceptible to tBuOH than precursor gelator 1-based systems, which partly explains why only gel system II in tBuClOAc with 1.0 eq. of acid exhibited transient behaviour.
In tBuClOAc, all gels formed within one day, regardless of the precursor gelator or acid concentration. In contrast, gelation in tBuOMe was significantly slower than in tBuClOAc and the previously reported tBuOAc,28 with gelation times ranging from 5 to 14 days for both gel systems I and II. The amount of acid also influenced the gelation behaviour. Lower acid concentrations led to longer gelation times and outcomes ranging from SSGs to PGs. As the acid effect was particularly pronounced for gel system II in tBuOMe, higher acid equivalents (1.5 and 2.0 eq.) were tested, both of which produced SSGs (Fig. S5, SI) and reduced the gelation time to 4 days. These results demonstrate that both the solvent type and the acid concentration can be used to adjust gelation time and outcome.
The difference in gelation behaviour can be explained by the pH of the samples, which was assessed by three parallel pH measurements of solvent and precursor gelator mixtures (Table S3, SI). The data show that the initial pH (before acid addition) of the tBuClOAc mixtures is lower than that of the tBuOMe mixtures for both precursor gelators 1 and 2. Therefore, the same amount of acid affects the two systems differently due to these distinct starting conditions, leading to a significantly lower final pH in tBuClOAc mixtures. This is reflected in the final gelation results, which show that SSGs form in tBuClOAc with lower acid amounts than in tBuOMe, and the gelation process occurs more rapidly.
The gels’ Tgel–sol were measured by controlled heating of the gels (in triplicate) and assessed by the vial inversion method to observe the phase changes (S3.4, SI). The measurements confirmed that the organogels are thermoreversible, as they transitioned to the sol state upon slow heating and reformed upon cooling to room temperature.
The solvent was found to affect the Tgel–sol values (Table 1): slightly higher transition temperatures were observed in tBuOMe compared to tBuClOAc. In addition, Tgel–sol increased as the amount of acid decreased in both gel systems. This trend is likely due to the presence of residual precursor gelators in samples with lower acid content, which have higher melting points than the individual gelators and may reinforce the gel network, thereby raising the transition temperature.
NMR studies
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2b with a ratio of 0.50
2b with a ratio of 0.50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00, Fig. 2b; 1a
1.00, Fig. 2b; 1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1b with a ratio of 1.00
1b with a ratio of 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.26). Their presence in the gel system is further corroborated by a singlet at ∼1.3 ppm corresponding to the tert-butyl group of gelator 1a or 2a and a peak at 8.05 ppm assigned to the protonated NH2 (NH3+). The ratio of gelators varies dynamically within the systems. This is evident from the NMR spectra measured on different gel batches on the day of gel formation, in which the gelator ratio differs among batches (Fig. S28, SI).
0.26). Their presence in the gel system is further corroborated by a singlet at ∼1.3 ppm corresponding to the tert-butyl group of gelator 1a or 2a and a peak at 8.05 ppm assigned to the protonated NH2 (NH3+). The ratio of gelators varies dynamically within the systems. This is evident from the NMR spectra measured on different gel batches on the day of gel formation, in which the gelator ratio differs among batches (Fig. S28, SI).
 | ||
| Fig. 2 1H NMR spectra of (a) gel system II in tBuClOAc and (b) gel system I in tBuOMe. In all samples 1.0 eq. of acid was used. | ||
In our previous studies, the formation of tBuOH as a result of solvent decomposition was shown to be crucial for the gelation mechanism and gel stability, specifically through the hydrolysis/esterification cycle, as well as for the subsequent collapse of the gel.27,28
Although gel systems are dried under vacuum, their drying may be more or less incomplete, as solvent peaks are clearly visible in gel NMR spectra (S4, SI). Therefore, for example, tBuOH should also be detectable in the NMR spectra. To our surprise, no tBuOH was observed in the tBuClOAc samples. To determine whether tBuOH forms, we first tested whether the solvent alone undergoes acid-catalysed decomposition. The reaction was monitored by NMR spectroscopy, with spectra measured before acid addition and at regular time intervals thereafter. Solvent decomposition was clearly observed, confirming the release of the tBu+ cation. However, tBuOH was not detected in either the xerogels or the solvent (S7, SI). The same test was also performed using tBuOMe as the solvent, which showed solvent decomposition. Interestingly, although tBuOH was not detected in the solvent spectra, a broad peak at ∼6.75 ppm (Fig. 2b), corresponding to the hydroxyl group of tBuOH, was detected in the gel samples prepared in tBuOMe. This suggests that, although tBuOH may also form in solution, its concentration is likely below the detection limit, or that the hydroxyl resonance is broadened beyond observation due to rapid proton exchange. In the gel state, however, supramolecular confinement may stabilize and enrich tBuOH, giving rise to the detectable broad resonance at ∼6.75 ppm in the case of tBuOMe systems.
It should be noted that the formation of the third gelator occurs somewhat randomly, mirroring the variability observed in the ratios of the other two gelators. The use of a solvent capable of generating two leaving groups may partially explain this variability in gelation outcomes and the prolonged gelation time. These findings suggest that solvents with multiple potential leaving groups provide greater tunability in solvent-induced gelation systems and indicate that the leaving groups of the precursor gelator and the solvent do not necessarily need to match.
In contrast, gels formed in tBuClOAc at the same acid concentrations do not consistently display these peaks (Fig. S6, S8, S12 and S14, SI). This difference arises from variations in the initial pH of the precursor gelator solutions; for example, at the lower final pH (after acid addition), Boc deprotection is more complete. Nevertheless, more sensitive HR-MS can still detect residual precursor gelators in the systems, even when NMR does not clearly reveal their presence (S4.1 and S4.2, SI).
Assessing the secondary structure of gels by ATR-FTIR
Infrared (IR) spectroscopy of xerogel samples was conducted to assess the self-assembly and supramolecular interactions in the corresponding gels (Fig. 4). Particular attention was given to the amide I region (1700–1600 cm−1), where absorption bands arise primarily from C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations with minor contributions from N–H plane bending. Vibrations in this region are sensitive to C
O stretching vibrations with minor contributions from N–H plane bending. Vibrations in this region are sensitive to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯H–N hydrogen bond formation and are directly influenced by the resulting secondary structure.29,30
O⋯H–N hydrogen bond formation and are directly influenced by the resulting secondary structure.29,30
In gel system I with tBuClOAc and 0.5 eq. of acid (Fig. 4a, red solid line), a single band at 1666 cm−1, corresponding to a β-sheet structure, is observed. In contrast, at 0.18 eq. of acid (solid blue), two weak bands attributed to β-sheet structures appear at 1681 cm−1 and 1667 cm−1, together with a peak at 1654 cm−1 indicative of a helical-like assembly. For the xerogels of gel system I prepared from tBuOMe (dotted lines), both helical-like and β-sheet structures are present for all acid concentrations. The helical-like band consistently appears around 1650 cm−1, regardless of acid equivalency. However, the β-sheet bands show minor variations as the acid equivalency changes. The band appears broader at higher acid concentrations (1.0 and 0.5 eq.) compared to the lowest acid concentration (0.18 eq.), and their positions shift irregularly with acid concentration (1.0 eq.: 1673 cm−1; 0.5 eq.: 1666 cm−1 and 0.18 eq.: 1684 cm−1).
For gel system II, the secondary structures of xerogels in tBuClOAc vary with acid concentration. The xerogel formed with 1.0 eq. of acid (solid black, Fig. 4b) shows a broad band at 1671 cm−1 associated with β-sheet formation. At 0.5 eq. of acid (solid red), only a broad peak (1660 cm−1) related to a helical-like structure is observed. In the sample prepared with the lowest acid content (0.18 eq.), bands corresponding to both β-sheet (1684 cm−1) and helical-like structures (1654 cm−1) appear. For xerogels of gel system II prepared with tBuOMe, only a peak corresponding to a β-sheet around 1671 cm−1 is observed at higher acid concentrations (1.0 and 0.5 eq.). In contrast, at 0.18 eq. the dominant structure is helical, as indicated by a peak at 1657 cm−1.
As expected from the literature,31–33 β-sheet structures are found in these xerogels in both solvents. The position and number of β-sheet bands vary depending on the type of β-sheet (parallel or antiparallel), the number of strands, and the degree of twisting within the sheets.29 Notably, short helices or helical-like structures with fewer than six residues typically do not exhibit the characteristic α-helix absorption. However, several bands can still arise in the amide I region due to shorter helical-like conformations.29 In our previous studies, helical-like structures were frequently observed.27,28 In addition, detailed investigations of a related gel system revealed a distinct helical-like band, although the β-sheet remained as the dominant nanoscale structure.34 The helical-like features observed in the current study are likely due to the coexistence of multiple gelators, including the precursor gelator, and their respective distribution within the samples. This also accounts for the variation in secondary structure peaks, particularly in response to acid concentration.
Morphological features
To investigate the effects of solvent and the type of precursor gelator on the morphology of the gel network, the gel systems were imaged using transmission electron microscopy (TEM, Fig. 5 and Fig. S40a–c, SI), atomic force microscopy (AFM, Fig. S40d, SI) and scanning electron microscopy (SEM, Fig. S41, SI) Imaging was performed on gels prepared with 0.5 eq. of acid and diluted before drying at room temperature (standard protocol, S1, SI). However, the exceptions are systems imaged with SEM that were vacuum-dried, and the samples were prepared by dipping the grid directly into the gel (S1, SI). | ||
| Fig. 5 TEM images of 0.5 eq. of gel systems in tBuClOAc: (a) gel system I and (b) gel system II, and in tBuOMe: (c) gel system I and (d) gel system II. Scale bars are shown in the image. | ||
In the case of gel system I in tBuClOAc, the standard protocol was modified because the gel matrix collapsed under the standard conditions (Fig. S40a, SI). To address this issue, the gel was transferred directly onto the chip and allowed to dry at room temperature. This approach resulted in a scaffold-like structure composed of shorter, flattened ribbons connected to wider, flatter bundles, forming a porous network (Fig. 5a), as confirmed by TEM. A similar porous network can be observed in the TEM image (Fig. S41a2, SI) of the 0.18 eq. system. Furthermore, a distinct ribbon structure is observed (Fig. S41a1 and a2, SI).
Using the standard sample-preparation protocol, gel system I in tBuOMe (Fig. 5c) produced a dense, fibrous network of thin, long, entangled fibres. Additionally, needle-like structures were observed (Fig. S40c, SI). Also, TEM images (Fig. S41b1, b2, SI) show similar needle-like structures that begin to intertwine together to form a larger bundle-like structure. In gel system II in tBuClOAc (Fig. 5b), long, straight fibres were observed alongside thin, intertwined individual fibres. Furthermore, long, narrow, separate, and breached single fibres as well as flat ribbons were present (Fig. S40b, SI). Gel system II in tBuOMe exhibited elongated, straight fibres (Fig. 5d), together with bundles and flat ribbons (Fig. S40d, SI).
The morphologies of gel system II were broadly similar across solvents, except that in the tBuClOAc system (Fig. 5b), single entangled fibres were also observed, a feature likewise characteristic of gel system I in tBuOMe (Fig. 5c). The two gel system I samples cannot be directly compared due to differences in sample preparation. However, the divergent behaviour during preparation suggests inherent differences between the systems. Indeed, Phe-derivatives are well known as self-assembling molecules.31,35 Even small changes in molecular structure or environmental conditions can lead to significant morphological differences.36 In the present case, the environment is influenced by the distribution of active gelators (which cannot be precisely controlled), acid content, and the choice of solvent, all of which contribute to the resulting gel morphology.
Conclusions
This study demonstrates that the unique solvent-induced gelation mechanism can be adapted to different solvent environments and that the acid concentration affects the gelation process. The gelation time, final outcome, and supramolecular structure of the resulting materials can be tuned by varying both the solvent and the acid concentration. In tBuClOAc, the gelation time remained constant regardless of the acid amount, whereas in tBuOMe, the gelation time increased as the acid concentration decreased. Acid concentration also determined the outcome of gelation in tBuOMe, influencing whether a self-supporting or partial gel was obtained, and affecting the efficiency of precursor gelator-to-gelator conversion. These effects were consistent across both dipeptide systems, highlighting the critical role of solvent and acid, independent of gelator structure.Switching from the previously studied tBuOAc to tBuClOAc and tBuOMe impacted gel stability and transient behaviour, with the new systems producing stable gels to date. Notably, tBuOMe acted as a “solvent with two leaving groups” as evidenced by NMR spectroscopy. These findings show that solvent-induced systems are adaptable and that the leaving groups in the solvent and in the precursor gelator do not necessarily need to match. However, the presence of multiple leaving groups may prolong gelation time and favour the formation of partial gels.
Both gel systems exhibited diverse supramolecular structures in the solvents studied. β-sheet structures were the dominant secondary motif in ATR-FTIR spectra. In tBuClOAc, helical-like structures emerged at lower acid concentrations, independent of the precursor gelator. By contrast, secondary structure formation in tBuOMe was less systematic, likely due to the dual leaving groups. Notably, tBuClOAc exhibited more pronounced structural changes with varying acid concentrations compared to tBuOMe.
Overall, this study provides new insights into the tunability of solvent-induced gelation systems. Deliberate manipulation of ester groups in precursor gelators, together with variation in solvent leaving groups, offers promising strategies for tailoring gelation behaviour, material stability, and mechanical properties. Furthermore, the scaffold-like architectures suggest potential applications in areas such as cell culture scaffolds or drug delivery platforms.
Experimental
Synthesis of precursor gelators and the corresponding active gelators
Compounds 1–3, 1a and 2a were synthesised according to previous protocols.28 As an exception, NaCl was used in the separation of water phases to prevent suspension formation. In addition, two water washes were added at the end of the purification process (S2.1, SI).Gelation protocol
The precursor gelator (1: 23.5 mg, 0.05 M; or 2: 21.7 mg, 0.05 M) was suspended in the corresponding organic solvent (1 mL) and sonicated until dissolution or a fine suspension formed, before adding concentrated H2SO4 (1.0, 0.5, or 0.18 eq.). The mixture was gently swirled and left at room temperature for gelation to occur. Gelation was confirmed by the vial-inversion test.Author contributions
H. R.: conceptualisation, formal analysis, investigation, validation, visualisation, writing – original draft, review and editing. E. D. S.: conceptualisation, supervision, writing – review and editing. R. C.: conceptualisation, writing – review and editing. M. N.: supervision, validation, writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included as part of the supplementary information (SI). Supplementary information: synthesis and characterisation of 2c and 3. Gelation experiments and NMR spectra of gels and degraded solvents, ATR-FTIR spectra of xerogels, AFM, TEM and SEM images. See DOI: https://doi.org/10.1039/d5cp03810c.The original files are available from the corresponding author upon request.
Acknowledgements
E. D. S. acknowledges the Jane and Aatos Erkko Foundation for supporting the work. Dr Andreas Johansson is thanked for operating the TEM and SEM, and MSc Veera Nikkola, and MSc Jenni Rive for preliminary gelation tests.References
- P. Terech and R. G. Weiss, Chem. Rev., 1997, 97, 3133–3160 CrossRef CAS PubMed.
- G. Yu, X. Yan, C. Han and F. Huang, Chem. Soc. Rev., 2013, 42, 6697–6722 RSC.
- D. Giuri, F. Cenciarelli and C. Tomasini, J. Pept. Sci., 2024, 30, e3643 CrossRef CAS PubMed.
- S. Fleming and R. V. Ulijn, Chem. Soc. Rev., 2014, 43, 8150–8177 RSC.
- T. Das, M. Häring, D. Haldar and D. Díaz Díaz, Biomater. Sci., 2018, 6, 38–59 RSC.
- J. Wang, K. Liu, R. Xing and X. Yan, Chem. Soc. Rev., 2016, 45, 5589–5604 RSC.
- T. Li, X.-M. Lu, M.-R. Zhang, K. Hu and Z. Li, Bioactive Mater., 2022, 11, 268–282 CrossRef CAS PubMed.
- M. A. Kuzina, D. D. Kartsev, A. V. Stratonovich and P. A. Levkin, Adv. Funct. Mater., 2023, 33, 2301421 CrossRef CAS.
- J. Kim, T. H. Han, Y. Kim, J. S. Park, J. Choi, D. G. Churchill, S. O. Kim and H. Ihee, Adv. Mater., 2010, 22, 583–587 CrossRef CAS PubMed.
- Y. Wang, W. Qi, R. Huang, X. Yang, M. Wang, R. Su and Z. He, J. Am. Chem. Soc., 2015, 137, 7869–7880 CrossRef CAS PubMed.
- C. Zhao, Y. Wang, B. Shi, M. Li, W. Yan and H. Yang, Langmuir, 2022, 38, 7965–7975 CrossRef CAS PubMed.
- J. Boekhoven, A. M. Brizard, K. N. K. Kowlgi, G. J. M. Koper, R. Eelkema and J. H. van Esch, Angew. Chem., 2010, 122, 4935–4938 CrossRef.
- S. A. P. Van Rossum, M. Tena-Solsona, J. H. Van Esch, R. Eelkema and J. Boekhoven, Chem. Soc. Rev., 2017, 46, 5519–5535 RSC.
- B. Rieß, C. Wanzke, M. Tena-Solsona, R. K. Grötsch, C. Maity and J. Boekhoven, Soft Matter, 2018, 14, 4852–4859 RSC.
- M. Fialkowski, K. J. M. Bishop, R. Klajn, S. K. Smoukov, C. J. Campbell and B. A. Grzybowski, J. Phys. Chem. B, 2006, 110, 2482–2496 CrossRef CAS PubMed.
- G. Ragazzon and L. J. Prins, Nat. Nanotechnol., 2018, 13, 882–889 CrossRef CAS PubMed.
- B. Rieß and J. Boekhoven, ChemNanoMat, 2018, 4, 710–719 CrossRef.
- L. Zeng, X. Lin, P. Li, F.-Q. Liu, H. Guo and W.-H. Li, Prog. Org. Coat., 2021, 159, 106417 CrossRef CAS.
- J. Raeburn, C. Mendoza-Cuenca, B. N. Cattoz, M. A. Little, A. E. Terry, A. Zamith Cardoso, P. C. Griffiths and D. J. Adams, Soft Matter, 2015, 11, 927–935 RSC.
- G. Zhu and J. S. Dordick, Chem. Mater., 2006, 18, 5988–5995 CrossRef CAS.
- S. Wu, J. Gao, T. J. Emge and M. A. Rogers, Soft Matter, 2013, 9, 5942–5950 RSC.
- D. M. Zurcher and A. J. McNeil, J. Org. Chem., 2015, 80, 2473–2478 CrossRef CAS PubMed.
- Y. Lan, M. G. Corradini, R. G. Weiss, S. R. Raghavan and M. A. Rogers, Chem. Soc. Rev., 2015, 44, 6035–6058 RSC.
- E. R. Draper and D. J. Adams, Chem, 2017, 3, 390–410 CAS.
- M. L. Muro-Small, J. Chen and A. J. McNeil, Langmuir, 2011, 27, 13248–13253 CrossRef CAS PubMed.
- P. Zhu, X. Yan, Y. Su, Y. Yang and J. Li, Chem. – Eur. J., 2010, 16, 3176–3183 CrossRef CAS PubMed.
- R. Chevigny, J. Schirmer, C. C. Piras, A. Johansson, E. Kalenius, D. K. Smith, M. Pettersson, E. D. Sitsanidis and M. Nissinen, Chem. Commun., 2021, 57, 10375–10378 RSC.
- R. Chevigny, H. Rahkola, E. D. Sitsanidis, E. Korhonen, J. R. Hiscock, M. Pettersson and M. Nissinen, Chem. Mater., 2024, 36, 407–416 CrossRef CAS PubMed.
- A. Barth, Biochim. Biophys. Acta, Bioenerg., 2007, 1767, 1073–1101 CrossRef CAS PubMed.
- J. Kong and S. Yu, Acta Biochim. Biophys. Sin., 2007, 39, 549–559 CrossRef CAS PubMed.
- X. Yan, P. Zhu and J. Li, Chem. Soc. Rev., 2010, 39, 1877–1890 RSC.
- X. Yan, Y. Cui, Q. He, K. Wang and J. Li, Chem. Mater., 2008, 20, 1522–1526 CrossRef CAS.
- S. Marchesan, L. Waddington, C. D. Easton, D. A. Winkler, L. Goodall, J. Forsythe and P. G. Hartley, Nanoscale, 2012, 4, 6752–6760 RSC.
- R. Chevigny, E. D. Sitsanidis, J. Schirmer, E. Hulkko, P. Myllyperkiö, M. Nissinen and M. Pettersson, Chem. – Eur. J., 2023, 29, e202300155 CrossRef CAS PubMed.
- P. Sahandi Zangabad, Z. Abousalman Rezvani, Z. Tong, L. Esser, R. B. Vasani and N. H. Voelcker, ACS Appl. Bio Mater., 2023, 6, 3532–3554 CrossRef CAS PubMed.
- S. Marchesan, A. V. Vargiu and K. E. Styan, Molecules, 2015, 20, 19775–19788 CrossRef CAS PubMed.
Footnote |
| † Current address: Université de Lorraine, CNRS, IJL, F-54000 Nancy, France. |
| This journal is © the Owner Societies 2026 |