Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Plasma-assisted CH4 activation on Cu/CeO2 catalysts: insights into the effect of catalyst surface and vibrational excitation
Shangkun
Li†
 a,
Santu
Luo†
ab,
Rui
Liu
c,
Zhaolun
Cui
d,
Yanhui
Yi
a,
Santu
Luo†
ab,
Rui
Liu
c,
Zhaolun
Cui
d,
Yanhui
Yi
 c,
Erik C.
Neyts
c,
Erik C.
Neyts
 a,
Annemie
Bogaerts
a,
Annemie
Bogaerts
 a and
Nick
Gerrits
a and
Nick
Gerrits
 *ae
*ae
aResearch group PLASMANT and Center of Excellence PLASMA, Department of Chemistry, University of Antwerp, Universiteitsplein 1, 2610, Wilrijk, Belgium
bState Key Laboratory of Electrical Insulation and Power Equipment, Centre for Plasma Biomedicine, Xi’an Jiaotong University, Xi’an, 710049, P. R. China
cState Key Laboratory of Fine Chemicals, Frontier Science Center for Smart Materials, School of Chemical Engineering, Dalian University of Technology, Dalian, 116024, P. R. China
dSchool of Electric Power Engineering, South China University of Technology, Guangzhou, 510630, China
eLeiden Institute of Chemistry, Gorlaeus Laboratories, Leiden University, 2300 RA, Leiden, The Netherlands. E-mail: n.gerrits@lic.leidenuniv.nl
First published on 16th January 2026
Abstract
The lack of chemical understanding and efficient catalysts impedes the development of plasma–catalytic CH4 conversion. In this work, we employ density functional theory calculations to understand the effects of vibrational excitation on the dissociative chemisorption of both CH4 and CH3 on surfaces relevant for (plasma-assisted) catalysis, i.e., Cu(111), CeO2(111), and a single Cu atom supported on CeO2(111). The single-atom Cu catalyst (Cu1/CeO2(111)) shows the lowest energy barrier (0.35 eV) for CH4 dissociation among the three surfaces. The vibrational mode-specific reactivity of CH4 and CH3 is assessed using the sudden vector projection (SVP) model, in which the stretching mode of CH4 is dominant for CH4 dissociation on these three surfaces. Additionally, depending on the reaction mechanism of CH3 chemisorption and dissociation, either the stretching or bending modes are predicted to be more effective at promoting reactivity. Furthermore, vibrational efficacies for dissociative chemisorption of CH4 on the investigated catalyst surfaces are compared using a simplified model, which also employs SVP calculations, to reveal the importance of mode specificity and the structural dependence of the catalyst, offering valuable insights into catalyst design in heterogeneous and plasma catalysis.
Introduction
Methane (CH4) is a very stable molecule. Dissociative chemisorption of CH4 on catalyst surfaces is typically considered the rate-controlling step in CH4 conversion.1–3 Therefore, understanding and predicting C–H bond activation are key to comparing catalytic performances across various catalysts and for guiding the rational design of effective catalysts.4 Plasma, a partly ionized gas, is an alternative method to drive reactions, viz. with electrical energy instead of thermal energy. When coupled to catalytic materials, plasma catalysis offers a means to drive chemical reactions under ambient conditions, without the need for elevated temperatures or pressures.5–11 Typically, the function of a catalyst is to reduce the barrier height for the rate-controlling reaction or to regulate the product distribution. This clear-cut function could also apply to plasma catalysis.Specifically, reactants can be activated in the plasma before interaction with the catalyst takes place.5 Hence, energy can be converted from different internal degrees of freedom (DOFs) of the reactant into the reaction coordinate, effectively increasing the amount of energy available for the reaction. The vibrational modes of the reactants can be selectively activated by the reduced electric field (i.e., the ratio of the electric field to the gas number density) in non-equilibrium plasmas.6–8 The energy stored in specific vibrational modes can increase the vibrational temperature (Tvib > 1000 K), which may promote energy-efficient gas conversion in a plasma or on a catalyst.6–8 For example, plasma-induced excitations can play an important role in CH4/CO2 conversion and NH3 synthesis by plasma catalysis, as shown in both experimental and theoretical studies.12–17 Nozaki et al. studied the role of vibrational excitation of CH4 in plasma catalysis by emission spectroscopy and observed that the vibrational temperature of excited species largely increased with the packing bed temperature, when using Ni/SiO2 as a catalyst.12 Furthermore, the importance of the bending mode of vibrationally excited CO2 molecules on a Pd2Ga/SiO2 alloy catalyst system was investigated by Kim et al.13 These authors found that a lower effective barrier height, due to vibrational excitation, can significantly improve the reaction performance, in which the CO2 conversion is not only increased more than a factor of two but also breaks the thermodynamic equilibrium limitation compared to thermal conditions.13 Mehta et al. combined microkinetic modelling with experiments to investigate plasma–catalytic NH3 synthesis from N2 and H2. They suggested that vibrationally excited N2 can lower the barrier height in the catalyst–adsorption step and thereby increase the catalyst activity for metals that bind N2 weakly in thermal catalysis.14 In a follow-up study, Engelmann et al. specifically investigated the effect of various N2 vibrational energy distribution functions, including those characteristics for dielectric barrier discharge plasmas commonly used in plasma catalysis. They also compared the role of vibrationally excited N2 molecules vs. plasma-generated radicals.15 In another study, Engelmann et al. also investigated the reactions of vibrationally excited CH4 and hydrocarbon radicals on transition metal catalysts, revealing that vibrational excitations and plasma-generated radicals can impact the reaction rates and product selectivity.16 Finally, Michiels et al. also revealed by microkinetic modeling that vibrationally excited CO2, as well as radicals, can increase the turnover frequency of CH3OH formation in plasma–catalytic CO2 hydrogenation.17 It should be noted that in the aforementioned modelling studies, the plasma-induced vibrational excitations for the gas–surfaces reactions were always described by the Fridman–Macheret (F–M) α model, originally formulated for the gas-phase reaction of an atom with a diatomic molecule.18 Unfortunately, this model seems to be unable to capture the complexity of molecule–metal surface interactions arising from high-dimensional potential energy surfaces (PESs), which cause dynamical and state-specific effects in plasma catalysis.19–21
Quantum dynamics studies can provide high precision by incorporating all degrees of freedom and quantum effects. A lot of effort has been devoted to constructing a PES to describe the non-statistical nature of the dissociative chemisorption of CH4.22 In order to avoid the extreme, intractable computational cost of full-dimensional quantum dynamics, some approximate methods have been proposed, e.g., quasiclassical trajectory (QCT), and the reaction path Hamiltonian (RPH) approach.22 In the QCT approach, the quantum mechanical vibrational energy is imparted to each mode in the initial velocity setup by treating classical objects following Newton's laws of motion.23 QCT is fast and intuitive for large systems but fails to capture quantum tunneling or zero-point energy effects, leading to inaccuracies at low energies. Recently, Gerrits et al. proposed a new approach to more accurately predict the reactivity under catalytically relevant conditions (i.e., low translational and high vibrational energies in the molecule) by extending a ring polymer molecular dynamics (RPMD) approach to include surface atom motion.3 In contrast to the QCT approach, RPMD can include nuclear quantum effects, like tunneling, and remedy the artificial zero-point energy leakage of the molecule into the reaction coordinate for translational energies below the minimum barrier height. This RPMD method offers more accurate predictions of the experimental sticking probabilities (i.e., a measure of reactivity), which could also be employed for non-equilibrium conditions in, e.g., plasma catalysis.3
Besides, Bal et al. developed an indirect approach by implementing a bias potential in molecular dynamics (MD) simulations, in which the selected mode can be excited to higher temperatures, while all others remain at thermal equilibrium.21 Furthermore, different Ni surfaces and vibrational modes were investigated using the same approach to understand the impact of vibrational non-equilibrium on the dissociative chemisorption barrier of CH4.24 The effect of vibrational excitation on the free energy barrier was predicted to be larger on terrace sites than on surface steps. Also, even at a low vibrational temperature, high vibrational efficacies (i.e., the quantitative effect of vibrational energy on the reactivity compared to translational energy) were obtained. The efficacy of the symmetric stretch was greater than that of asymmetric stretches, which, in turn, was higher than that of the bending modes, in agreement with experiments.24
The RPH approach was also proposed by focusing on the minimum energy reaction path rather than the full-dimensional PES to understand the dynamics of vibrational mode-specific chemistry.25–27 Jackson et al. used this approach to understand the dissociative chemisorption of CH4 on a Ni(100) surface based on a harmonic expansion of the vibrational modes along the reaction path. Among the vibrational modes, the symmetric stretch (ν1) exhibited the highest efficacy, as it strongly couples to the reaction coordinate and softens at the transition state (TS).26 Likewise, Roy et al. reported the same trend that the symmetric stretching vibrational mode of CH4 on the Ni/Pt-bimetallic alloy exhibits the highest reactivity due to significant mode softening near the TS, lowering the effective barrier height.27
Indeed, dynamical simulations can provide a better understanding of the dynamical effects, including energy transfer between the molecule and the catalyst surface, thermal local barrier height modulation, and the bobsled effect.28–34 Interestingly, Jiang et al. proposed a sudden vector projection (SVP) model to qualitatively predict the vibrational state-specific efficacies in reactions involving polyatomic reactants, without requiring complex dynamical simulations.35–39 The SVP model assumes the energy transfer as occurring on a timescale too short for full intramolecular vibrational redistribution, which has been demonstrated to be valid in several gas phase reactions and gas–surface interactions.38–41 Jiang et al. compared the computed efficacies of vibrationally excited CH4 in the reaction on a rigid Ni(111) surface between MD simulations and the SVP model.35 In the SVP approach, the two stretching modes exhibited higher vibrational efficacy than the bending modes, in agreement with both theoretical and experimental observations.35,42,43
Recently, Gerrits and Bogaerts proposed an improved model to capture the effect of vibrational excitation on dissociative chemisorption rates in heterogeneous and plasma catalysis.19 Unlike the widely used F–M α model—which lacks vibrational mode specificity and very poorly matches MD and experimental molecular beam data—the new model combines the forward barrier height, “lateness” of the transition state geometry (ratio of the dissociating and equilibrium bond lengths), and the overlap of vibrational modes with the reaction coordinate using SVP calculations. This approach yields significantly better agreement with MD-derived vibrational efficacies (R2 = 0.52 vs. −0.35) at a similar computational cost, making it more suitable for microkinetic modeling of vibrationally excited molecule–metal surface reactions.19
The key to CH4 conversion lies in finding efficient catalytic systems with a controllable reaction kinetics process. Single-atom catalysts (SACs) have attracted wide attention as promising candidates for tackling challenges in energy conversion, environmental remediation, and chemical synthesis, owing to their maximum metal dispersion, precise control over catalytic sites, and enhanced reactivity and selectivity.44,45 Since the typical mean electron energy in a CH4 plasma is in the range of 1–5 eV, vibrational excitation of CH4 due to impacting electrons is assumed to be more prevalent than depositing the energy in other channels. The vibrational excitation of CH4, in turn, can enhance chemical reactivity of the molecule on a catalyst surface, compared to thermal (i.e., heterogeneous) catalysis.46–48 Thus, in this paper, we use density functional theory (DFT) calculations to investigate the performance of CH4 activation on a SAC, by comparing three typical surfaces, i.e., Cu(111), CeO2(111), and a single Cu atom supported on CeO2(111), denoted as Cu1/CeO2(111). The effects of vibrational mode-specificity of CH4 on the three surfaces are investigated to understand CH4 activation in plasma catalysis. We note that the F–M α model is currently used as a one-fits-all tool for quantitative prediction of the effect of vibrational excitation in microkinetic models. However, even for the dissociative chemisorption of diatomic molecules, it is inaccurate, where obviously no mode specificity is present.20 In this work, the qualitative comparison between the F–M α model and the alternative η model is also discussed, to indicate the importance of mode-specificity and the relationship between the structural dependence of the catalyst and vibrational efficacy.
Finally, we will also discuss the further dissociation of a CH3 radical from the plasma on the aforementioned three surfaces, because besides vibrational excitation, microkinetic models revealed that radical chemistry might also be important for plasma catalysis.11,15–17
Methodology
DFT calculations
Spin-polarized DFT calculations were performed using the Vienna ab initio simulation package (VASP, version 6.2.1).49–51 The Perdew–Burke–Ernzerhof (PBE) exchange–correlation density functional52 was employed and the projected augmented wave method was used to describe the core electrons.53,54 A cutoff energy of 500 eV was adopted throughout. To adequately describe the electron localization in the Ce 4f orbital, an on-site Coulomb repulsion was applied, as described by the Hubbard U parameter.55 The U value was set to 4.5 eV for the Ce 4f orbitals, providing sufficient localization of the electrons left on CeO2.56–58 The maximum force criterion for ionic convergence was set to 0.02 eV Å−1. van der Waals dispersion forces between adsorbates and surfaces were accounted for using the zero damping DFT-D3 method of Grimme.59 All configurations were optimized using the conjugate gradient algorithm. The climbing image nudged elastic band (CI-NEB) method and minimum-mode following dimer methods were adopted to determine the TS structures of elementary reaction steps as implemented in the VASP Transition State Tools package.60,61 All TSs were validated by vibrational frequency analysis.The projected crystal orbital Hamilton population (COHP) curves were calculated using LOBSTER to analyze the bonding and anti-bonding states,62,63 where the pbeVASPFit2015 basis set was used for the projection of wave functions. Bader charges were calculated for the electron population analysis.64
The adsorption energy, Eads, is defined as:
| Eads = Eadsorbate+surface − (Esurface + Eadsorbate) | (1) |
Here, Eadsorbate+surface, Esurface, and Eadsorbate are the total energies of the adsorbate on the slab, the clean slab, and the gaseous adsorbate, respectively.
Surface structures
The close-packed (111) surfaces of Cu and CeO2 are studied because they represent the most stable terminations observed experimentally. Furthermore, the CeO2(111) surface in particular has been widely used as a support for various metals in many theoretical studies.57,65,66 In addition, we also investigate a single Cu atom on CeO2(111), i.e., Cu1/CeO2(111), because single-atom catalysts are gaining attention for their maximal metal dispersion, precise site control, and improved reactivity and selectivity in energy, environmental, and chemical applications.44,45 These three specific surfaces help us theoretically understand the CH4 dissociation mechanism and its vibrational excitation due to their distinct TS structures. As shown in Fig. 1A, the Cu(111) surface was represented by a four-layer slab and a (5 × 5) supercell, with a vacuum gap of 15 Å. The bottom two layers were fixed at the ideal bulk positions, while the positions of all other atoms were allowed to relax. The surface model of CeO2(111) was represented by three O–Ce–O triple-layers with (4 × 4) supercells, again with a vacuum layer of 15 Å (Fig. 1B). The bottom two O–Ce–O layers were fixed, while the top O–Ce–O layers were allowed to relax. For a Cu atom adsorbed on CeO2(111), the hollow site (Fig. 1C) is the most stable adsorption site.67 A (2 × 2 × 1) γ-centered k-point mesh was used to sample the Brillouin zone for the Cu(111) surfaces, with a Methfessel–Paxton smearing width of 0.2 eV. For the surfaces of CeO2(111) and Cu1/CeO2(111), only the Γ-point sampling was used for k-point sampling and a Gaussian smearing width of 0.05 eV. Indeed, employing Γ-point sampling with the VASP_GAM version significantly reduces the computational cost, while the difference in total energy remains below 0.01 eV (Table S1). | ||
| Fig. 1 Top view (above) and side view (below) of the optimized structure for (A) Cu(111), (B) CeO2(111) and (C) Cu1/CeO2(111) (colour code: light blue: Cu; gold: Ce; red: O). | ||
CH4 vibrational excitation analysis
As explained in the introduction, we compare the efficacy of vibrationally exciting CH4 for promoting dissociation by using two methods. The first is the Fridman–Macheret (F–M) α model, which assesses the influence of plasma-induced vibrational excitations. This model was originally developed for reactions in the gas phase.18 However, it has also been used in gas–surface reactions to predict the enhancements in the dissociation rates by vibrational excitation, due to the lack of more accurate, feasible approaches available at that time.14–17 In the F–M α model, the efficacy is determined by the proportionality constant αFM:14–17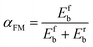 | (2) |
Here, Efb and Erb are the energy barriers for the forward and reverse reactions, respectively. If Efb is equal to zero, there is no enthalpy barrier, and thus, the reaction is diffusion-limited. In this case, αFM is equal to zero (i.e., the vibrationally excited levels play no role in enhancing the reaction). Vice versa, if Erb is equal to zero, the reaction is enthalpy-limited, and thus, the efficacy of the vibrationally excited levels to lower the reaction energy barrier is at maximum. In this case, αFM is equal to 1.
The other method is the alternative η model proposed by Gerrits and Bogaerts, which fits vibrational efficacies obtained from MD simulations by incorporating three variables,19i.e., (1) the forward barrier height (Efb), (2) the ratio of the dissociating bond length between the transition state (TS) and the reactant (RTS/Rgas), and (3) the SVP values, using the semi-empirical parameters of α1 = 0.008259 mol kJ−1, α2 = 2.4405, and α3 = 0.2032.
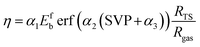 | (3) |
Specifically, the SVP values can be calculated by comparing the projection of a reactant normal mode onto the reaction coordinate at the transition state (TS). A larger SVP value indicates a stronger coupling between the reaction coordinate and the vibrational mode, thus resulting in more energy being available for the reaction.39
In this study, we will evaluate these two approaches to compare the impact of vibrational excitation of CH4 by calculating the forward rate constant kv:
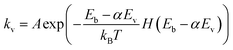 | (4) |
Results and discussion
CH4 and CH3 activation on the different catalyst surfaces
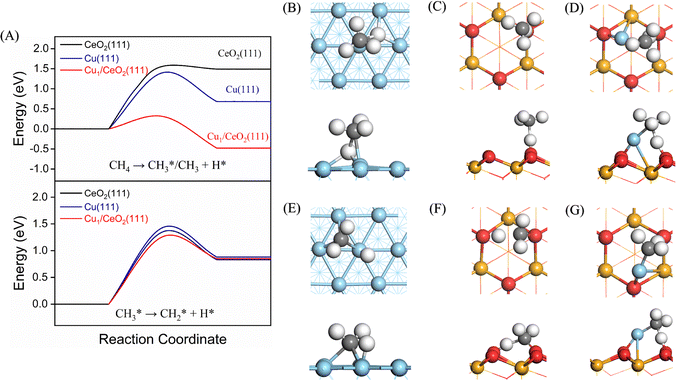
The three catalyst surfaces, i.e., Cu(111), CeO2(111), and Cu1/CeO2(111), were investigated to compare their ability for C–H activation. The optimized CH4 adsorption sites on these three surfaces are shown in Fig. S1–S3. We compare the CH4 orientation based on one of the H atoms towards the surfaces. On Cu(111) and CeO2(111), the most stable configuration corresponds to CH4 adsorbed at a hollow site, where three H atoms lie approximately parallel to the surface plane. The resulting adsorption energies are −0.26 eV and −0.22 eV, respectively. In contrast, on Cu1/CeO2(111), CH4 preferentially adsorbs at a bridge site because a single Cu atom already occupies the hollow site of CeO2(111), leading to a slightly increased adsorption energy of −0.28 eV. These results (Table 1) indicate that CH4 is only weakly physisorbed on all three surfaces. Although the orientation of CH4 varies noticeably among the different adsorption sites, the adsorption energy differences remain small. The dissociative chemisorption (Fig. 2A, upper panel) of CH4, i.e., CH4 → CH3* + H*, is endothermic on Cu(111) and CeO2(111), with a reaction energy of 0.68 eV and 1.41 eV, respectively. The energy barrier of CH4 on Cu(111) is 1.41 eV, which is similar to the result obtained using the optB86b-vdW functional.29,68 It should be mentioned that the barrier height is affected by the choice of density of functional (DF).29,69 For example, SRP32-vdW-DF1,28,31,70 which is a DF generally performing well for the dissociative chemisorption of CH4,71,72 yields a considerably higher energy barrier of 1.72 eV for CH4 on Cu(111).29 The variations in barrier heights obtained from DFs will be further discussed below in relation to the mode-specificity analysis.| Reaction | Surface | Barrier height (eV) | Reaction energy (eV) |
|---|---|---|---|
| CH4 → CH4* | Cu(111) | — | −0.26 |
| CH4 → CH4* | CeO2(111) | — | −0.22 |
| CH4 → CH4* | Cu1/CeO2(111) | — | −0.28 |
| CH4* → CH3* + H* | Cu(111) | 1.41 | 0.68 |
| CH4* → CH3* + H* | CeO2(111) | 1.56 | 1.49 |
| CH4* → CH3* + H* | Cu1/CeO2(111) | 0.35 | −0.49 |
| CH3 → CH3* | Cu(111) | — | −1.93 |
| CH3 → CH3* | CeO2(111) | — | −2.21 |
| CH3 → CH3* | Cu1/CeO2(111) | — | −2.16 |
| CH3* → CH2* + H* | Cu(111) | 1.43 | 0.88 |
| CH3* → CH2* + H* | CeO2(111) | 1.35 | 0.84 |
| CH3* → CH2* + H* | Cu1/CeO2(111) | 1.26 | 0.85 |
There is no evident difference in the barrier heights for C–H activation between the Cu(111) and CeO2(111) surfaces (i.e., 1.41 vs. 1.56 eV, respectively), although the geometries of the TSs are quite different. On Cu(111), the CH4 molecule can undergo dissociation to form Cu–CH3 as an intermediate, enabling direct cleavage of the C–H bond alongside the coordination of the CH3 group (Fig. 2B). However, this is not the case on CeO2(111). Indeed, we find that the vibrational direction of the imaginary frequency (Fig. S4) does not coincide with that of C–H bond dissociation, referred to as a “pseudo-transition state”,73 when we calculate the frequency of the TS structure of the O–CH3 intermediate formed. Potentially, this can lead to a very different mode-specificity for the different surfaces. Notably, the other distinct TS structure found on CeO2(111) corresponds to H abstraction from CH4 to form ˙CH3 radicals by electrophilic oxygen atoms from the catalyst surface (Fig. 2C).
The emergence of two distinct transition state (TS) geometries on the Cu(111) and CeO2(111) surfaces stems from different mechanisms: the first TS features a stabilized CH3 group by interacting with surface atoms, while a radical-like TS is formed instead when the geometric access or energetic favorability of CH3–surface interaction is hindered.4 Notably, the Cu1/CeO2(111) surface reduces the barrier height of CH4 dissociation to 0.35 eV, and the reaction becomes exothermic (reaction energy of −0.49 eV). Indeed, such a low barrier can also be obtained on the SAC Pd1/CeO2(111) with a similar exothermic process for dissociative chemisorption of CH4.74 Low-barrier catalysts for CH4 at a low temperature may avoid unwanted side reactions to selectively convert CH4 to value-added products.75 Additionally, the Cu–CH3 intermediate (Fig. 2D) can be formed in the TS for Cu1/CeO2(111), instead of the ˙CH3 radical being suspended in the gas phase, as observed for CeO2(111).
Next to dissociation on the surface, CH4 can also be already dissociated in the plasma before it interacts with the catalyst, i.e., CH4(g) → CH3(g) + H(g), e.g., upon electron impact or upon reaction with other molecules or radicals. The CH3 radicals produced in the gas phase can then adsorb on the catalyst surface. Therefore, we also compare the C–H bond dissociation of CH3 on the three surfaces. The adsorption energy of CH3 (Table 1) follows the order CeO2(111) (−2.21 eV) > Cu1/CeO2(111) (−2.16 eV) > Cu(111) (−1.93 eV), indicating that a metal oxide surface (e.g., CeO2) adsorbs CH3 more strongly. The structures of adsorbed CH3 for the three surfaces are shown in Fig. S5. Note that the adsorption energy of CH3 is an order of magnitude larger than that of CH4. However, the type of surface has only a limited effect on the barrier height for the dissociation of CH3 (Fig. 2A, lower panel), with a maximum barrier height of 1.43 eV on Cu(111), similar to the results obtained with the optB86b-vdW DF,68 and only slightly lower on CeO2(111) (1.35 eV) and on Cu1/CeO2(111) (1.26 eV). The TS structures of CH3 dissociation on the three surfaces are shown in Fig. 2E and F.
Electronic structure analysis
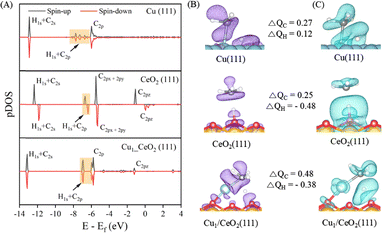
The different catalysts influence the barrier for CH4 dissociation, resulting in distinct TS structures and energies, while showing very similar behavior towards CH3 activation. To elucidate the effect of bonding on the dissociation of CH4, we investigate the projected density of states (pDOS) and the charge density differences of the CH4 TS on Cu(111), CeO2(111), and Cu1/CeO2(111). The pDOS of a gaseous CH4 molecule (Fig. S6) shows two clear peaks in which four H1s orbitals overlap with the C2s and C2p orbitals. The stability of CH4 in its tetrahedral structure arises from the formation of four equivalent C–H bonds, through sp3 hybridization between the C and H atoms.76 When CH4 is adsorbed on Cu(111), the H1s–C2p peak is split and a small peak appears nearby. Three peaks of H1s–C2p are observed if CH4 is adsorbed on Cu1/CeO2(111), whereas only one peak appears on CeO2(111). This means that the C2p orbital of CH4 can be split into C2px, C2py, and C2pz on Cu1/CeO2(111).In Fig. 3A, the overlaps between the H1s–C2s orbitals appear for the TS of CH4 on all three surfaces. No evident spin polarization is observed on either the Cu(111) or the Cu1/CeO2(111) surface. When CH4 dissociates on CeO2(111), there is a strong spin polarization because the surface oxygen atom can abstract the H atom, resulting in a radical-like TS structure (Fig. 2C). The pDOS of the H1s and C2p orbital overlap shows multiple peaks for the Cu(111) surface. The overlap is the largest on the Cu1/CeO2(111) surface, indicative of a strong interaction, and thus likely the origin of the comparatively low dissociation barrier for CH4.
In Fig. 3B, the electron density difference isosurfaces show that the density increases around the C atom in the order Cu1/CeO2(111) > Cu(111) > CeO2(111). The highest calculated Bader charge (i.e., 0.48 |e|, Table 2) is found for the Cu1/CeO2(111) surface, indicating the largest electron transfer. On Cu(111) (Fig. 3C), there is a slight electron density increase near the H atom; in this case, the Cu atom can act as an electron donor. However, the surface O atom from CeO2(111) attracts the H atom in CH4, which decreases the electron density near the H atom in the CH4 TS. Similarly, the electron density near H is also reduced on Cu1/CeO2(111). Overall, the pDOS analysis and electron density differences show that the CH4 TS interacts more strongly with Cu1/CeO2(111) than with the other surfaces, due to an increased electron transfer to and from the catalyst surface.
| Surfaces | R(C–H)/Å | Q/e | iCOHP | ||
|---|---|---|---|---|---|
| Initial state | Transition state | Q C | Q H | ||
| Cu(111) | 1.10 | 1.72 | 0.27 | 0.12 | −1.38 |
| CeO2(111) | 1.10 | 1.58 | 0.25 | −0.48 | −1.81 |
| Cu1/CeO2(111) | 1.10 | 1.39 | 0.48 | −0.38 | −3.55 |
Furthermore, the crystal orbital Hamiltonian population (COHP) is used to analyze the bonding interaction between atoms. The calculated projected COHPs (pCOHPs) were compared to consider the C–H bond interaction strength in the initial states (Fig. S7) and TSs (Fig. 4) of CH4 on Cu(111), CeO2(111), and Cu1/CeO2(111). The integral of the COHP up to the Fermi level (termed iCOHP) is used to qualitatively measure the strength of a chemical bond between two atoms. The more negative the iCOHP is, the stronger the bond is. As shown in Table 2, the largest value of iCOHP is found for the CH4 TS on the Cu1/CeO2(111) surface (with a value of −3.55), suggesting a strong bond. Correspondingly, the C–H bond length is the shortest among the three surfaces. The barrier height is lower if an intermediate (i.e., TS) is more stable. Therefore, the pCOHPs and iCOHPs results demonstrate that the TS on the Cu1/CeO2(111) surface is the most stable structure among the three surfaces, indicating the lowest barrier height (cf.Table 1).
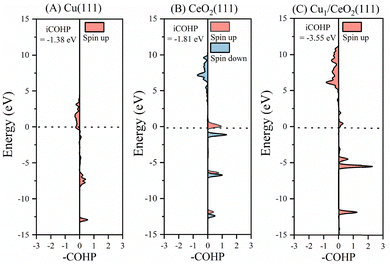 | ||
| Fig. 4 The crystal orbital Hamilton population (COHP) for the transition states of CH4 on (A) Cu(111), (B) CeO2(111), and (C) Cu1/CeO2(111). | ||
Mode specificity analysis by the SVP model
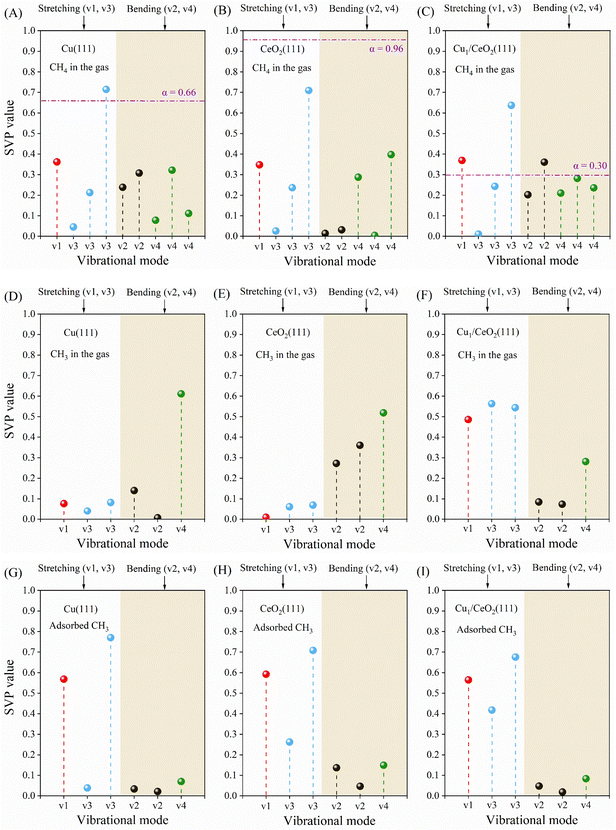
As mentioned in the introduction, vibrational excitation of CH4 can promote its reactivity and improve product selectivity compared to thermal conversion.15,46,77 The CH4 molecule has one C atom tetrahedrally bonded to four H atoms. Therefore, the total number of degrees of freedom is 15, i.e., 9 vibrational modes, 3 translation modes, and 3 rotational modes. Since some of the vibrational modes are degenerate, CH4 has 4 fundamental vibrational modes: the bending modes (ν2 and ν4), a symmetric stretching mode (ν1), and an asymmetric stretching mode (ν3). Here, the calculated vibrational frequencies of gaseous CH4 (Table S2) are in good agreement with experiments.47To investigate how these modes might affect CH4 dissociation on the three surfaces studied here, we use the SVP model to compare the gas phase vibrational modes with the reaction coordinate vector at the saddle point. In this study, CH4 in the gas phase is taken as the initial structure for the SVP calculation to mimic the process of the excited CH4 molecule reaching the surface under plasma conditions, rather than CH4 being physisorbed on the surface, which is directly followed by dissociative chemisorption. Note that activated dissociative chemisorption in general should be modelled as a direct reaction of the gaseous reactant with the surface without thermalization, instead of prior physisorption and thermalization of the adsorbate with the surface. The calculated frequencies for the SVP calculations are listed in Table S3. As shown in Fig. 5A, the SVP analysis shows that the stretching modes (v1, v3) of CH4 have the dominant contribution in the C–H bond dissociation of CH4 on the Cu(111) surface. The SVP value of the symmetric stretching mode (v1) on Cu(111) is 0.36, consistent with the value for CH4 + Ni(111).35 There are three degenerate asymmetric stretching modes (v3) with the highest SVP value of 0.72. However, the v3 mode is three-fold degenerate, thus the average SVP value of v3 over all three vectors is slightly lower than that of v1. This means that symmetric stretching has a larger contribution, in accordance with the experimental and theoretical results on Cu(111) and other metal surfaces.28,29,35,42,43
The SVP model (Fig. 5B–C) predicts similar results (v1 > v3) for CeO2(111) and Cu1/CeO2(111). Furthermore, the SVP values for the bending modes (i.e., v2 and v4) on Cu1/CeO2(111) and Cu(111) are similar. Interestingly, on CeO2(111), the v2 mode yields an SVP value of almost zero. This is caused by the distinct geometry structure of the TS on CeO2(111), in which the nearly perpendicular orientation of the TS structure (Fig. 2C) yields a limited projection of the bending mode onto the reaction coordinate, leading to the SVP value being close to zero.
Moreover, we compare the dependence of the SVP calculations for the choice of DFs on CH4 dissociation over three surfaces in Table S4. As expected, the choice of DF significantly affects the barrier height, as well as the reaction energy, since it is proportional to the barrier height.29 In particular, on Cu(111), the barrier height increases from 1.41 eV (PBE) to 1.92 eV (BEEF-vdW), indicating strong functional dependence. For Cu(111) and Cu1/CeO2(111) surfaces, however, the SVP values remain nearly constant, as the TS geometries and imaginary frequencies remain similar across different functionals, suggesting that the SVP and the shape of the PES are relatively insensitive to the choice of DF.78 However, the larger functional sensitivity observed for CeO2(111) can be attributed to distinct TS structures with ˙CH3 radical being suspended in the gas phase (Fig. 2C). For example, we employed DFT+U (U = 4.5 eV) to compare the SVP values on CeO2(111), in which PBE yields an SVP of 0.35, whereas SRP32-vdW-DF1 and BEEF-vdW give higher values of 0.43 and 0.46, respectively, indicating a modestly larger vibrational contribution by changing different treatments of DFs and dispersion interactions. Overall, comparisons among various DFs show that the variation in SVP values remains relatively small.
It should be mentioned that generally the SVP values of translation and rotation motion are less useful, possibly due to the complex nature of dynamical effects arising from these degrees of freedom. For example, the large rotational efficacy observed for HCl + Au(111) is due to the shape of the PES and concomitant dynamics, and, therefore, cannot be captured by the PES surrounding the TS.79 Likewise, the bobsled effect in late barrier systems is associated with translational motion and is caused by the curvature of the reaction path, again prior to reaching the TS.33 Thus, the static SVP calculation is more reliable for the vibrational modes. Additionally, the vibrational temperature in plasma can be much higher than the gas temperature, indicating a high prevalence of vibrationally excited reactants.80,81 Therefore, we mainly discuss the effects of vibrational modes by SVP in this paper.
Furthermore, the dissociation of the CH3 radical on the three surfaces is also investigated using the SVP model. Two options are considered: first, the CH3 radical might dissociate directly from the gas phase, without sufficient time to reorient the planar geometry to the more stable bent adsorption geometry. The chemisorption of CH3 (Fig. S8A) is highly exothermic, with an adsorption energy of 1.93 eV on Cu(111). The adsorption energy is 0.50 eV larger than the CH3 dissociation barrier (1.43 eV, Table 1). Similarly, the adsorption energies of CH3 on CeO2(111) and Cu1/CeO2(111) are also larger than the CH3 dissociation barrier (Fig. S8B and C). These results indicate that CH3 dissociation might occur directly during or after CH3 binds to the surface. Thus, we use the SVP model to understand the mode specificity of CH3 dissociation on the three surfaces. As shown in Fig. 5D and E, the bending modes (v2 and v4) of gaseous CH3 have a larger contribution to CH3 dissociation than the stretching modes (v1 and v3) on both Cu(111) and CeO2(111) because the bending modes align the planar gaseous CH3 radical with the bent configuration at the TS. In contrast, the stretching modes (Fig. 5F) still take a leading role in CH3 dissociation on Cu1/CeO2(111) because there is a large projection from the stretching vibration onto the reaction coordinate vector at the saddle point. The SVP values of CH3 dissociation are listed in Table S5.
Secondly, CH3 might adsorb without immediate subsequent dissociation, giving CH3 sufficient time to reorient from planar to bent. Furthermore, in that case, the large chemisorption energy needs to be dissipated. Several important dissipation channels might exist, such as vibrational excitation and phonons. Vibrational excitation could again lead to increased reactivity. Moreover, other plasma–catalytic processes might be able to vibrationally excite adsorbates like CH3. Energy transfer to the phonons, on the other hand, could reduce the reactivity, since there would be less energy available for the reaction. However, it is also likely that this process is relatively slow. In Fig. 5G–I, the stretching modes of adsorbed CH3 yield larger SVP values than the bending modes on all three surfaces, which is the opposite of when the gas phase vibrational modes are employed. In other words, there are large differences in vibrational efficacy (i.e., stretching vs. bending) for CH3 dissociation on Cu(111) and CeO2(111), depending on whether the TS is reached directly from the gaseous or chemisorbed state. The relevant state and concomitant reaction mechanism might be determined in future work using vibrational state-specific MD calculations and molecular beam experiments to investigate the radical chemistry in plasma catalysis.
Vibrational efficacy calculations
Vibrational efficacies are calculated using the F–M α model and the η model for the three surfaces. As shown in Table 3 and Fig. 5A–C, higher α values can be obtained on the Cu(111) and CeO2(111) surfaces when CH4 dissociation is endothermic for the F–M α model, while a lower α value (0.29) can be observed on Cu1/CeO2(111) when CH4 dissociation is exothermic. As for the η model, the efficacy of CH4 excitation is estimated by focusing on the dominant contribution from various vibrational modes, where symmetric stretching (v1) exhibited the highest SVP value on all three surfaces. Besides, the other two features, i.e., the forward barrier height (Efb) and the ratio of the dissociating bond length between the TS and the reactant (RTS/Rgas), have been incorporated to understand the vibrational state-specific efficacies.19 Evidently, the calculated vibrational efficacies η are much larger than the values predicted by the F–M α model on the Cu(111) and CeO2(111) surfaces. However, the difference between η and α are not so significant on Cu1/CeO2(111), since the forward barrier height is comparatively low. Generally, the η model deviates more strongly from the F–M α model when the absolute barrier height is large. Nevertheless, vibrational mode specific reactivity has been observed for low barrier heights as well, highlighting the importance of taking into account the coupling of specific vibrational modes to the reaction coordinate.19| Surfaces | Reaction type | α F–M | E fb (eV) | R TS/Rgas | SVP (= v1) | η | k η/kFM (500 K) |
|---|---|---|---|---|---|---|---|
| Cu(111) | Endothermic | 0.66 | 1.41 | 1.56 | 0.36 | 1.67 | 5846 |
| CeO2(111) | Endothermic | 0.96 | 1.56 | 1.44 | 0.35 | 1.69 | 243 |
| Cu1/CeO2(111) | Exothermic | 0.29 | 0.35 | 1.26 | 0.37 | 0.34 | 1.54 |
The integration of vibrational efficacy into existing kinetic models enables the prediction of gas–surface reaction rates dependent on the specific vibrational distribution and the evaluation of how vibrational non-equilibrium in a plasma affects the overall process at a macroscopic level.14–17 Here, we compare the ratio (kη/kFM) of forward rate constants between the η and F–M α model for the three surfaces at different temperatures (Fig. S9). The ratio disparities are greater at low temperatures and diminish as the temperature increases, indicating that the vibrational efficacy can significantly change rate coefficients at low temperatures. For example, the η model yields a rate coefficient that is 5846 times higher on Cu(111) and 243 times higher on CeO2(111) compared to those obtained from the F–M α model. In contrast, the difference between α and η is quite small on Cu1/CeO2(111), thus hardly affecting the reaction rate.
The main reason for the discrepancy between the F–M α model and the SVP model lies in the fact that CH4 dissociates differently on the three surfaces with the distinct structures of the TS. As for the F–M α model, the vibrational efficacy is solely computed based on the ratio between the forward and backward reaction barrier heights. This approach presents some critical limitations, including its oversimplified dependence on barrier height ratios without rigorous TS validation, its neglect of mode-specific vibrational effects, and failure to account for coupling between vibrational energy and various DOFs.19 Additionally, its arbitrary restriction (α ∈ [0, 1]) contradicts experimental observations where vibrational energy surpasses translational efficacy for dissociative chemisorption of CH4 on catalyst surfaces.19 Therefore, the prediction of vibrational efficacy by the F–M α model is less convincing for the gas–surface reactions. Vibrational efficacy predictions using the η model can be taken at comparable computational cost to understand the importance of mode specificity and the structural dependence of the catalyst. Notably, at low temperatures, the two models diverge significantly in their predictions of the reaction rate, implying that the role of vibrational excitation may be underestimated using the F–M α model, which merits further investigation through combined experimental and theoretical molecular beam studies. These observations were also made in an extensive investigation of the dissociative chemisorption of N2 on Ru(0001), by comparing various transition state theory-based models with MD simulations.20
Finally, we emphasize the importance of further validating the SVP results for plasma–catalytic CH4 activation. Performing dynamical calculations of CH4 dissociation on a realistic PES can offer more detailed insights into reactive collisions and capture the energy dependence of state-specific reaction probabilities.28,29 These insights might help to further develop and validate the SVP model and the fitted η approach to account for the effects of vibrational non-equilibrium in plasma catalysis.
Conclusions
We studied the dissociation of both CH4 and CH3 on Cu(111), CeO2(111), and Cu1/CeO2(111) with DFT calculations. We find that the TS structures for CH4 are different for the three surfaces. Compared to Cu(111) and CeO2(111), the single Cu atom supported on CeO2(111) can significantly reduce the barrier height (0.35 eV), which is attributed to an increased molecule–surface electron transfer and a more stable TS structure, indicated by electronic structure analysis. However, these effects are considerably less important for dissociating the chemisorbed CH3, where the barrier height is similar on all three investigated surfaces.The effect of vibrational excitation of CH4 and CH3 was investigated with the Fridman–Macheret α model and a novel, alternative approach that is fitted to vibrational efficacies and DFT results in the literature. Notably, the prediction of vibrational efficacy by the F–M α model seems to be less reliable due to its lack of mode specificity or structure dependence, potentially leading to an underestimation of the role of vibrational excitation at low temperatures. Furthermore, the SVP results indicate that the stretching modes of CH4 play a primary role in its dissociation on these three surfaces, in qualitative agreement with previous experimental and theoretical studies. The relative vibrational efficacies for CH3 dissociation of the stretching and bending modes show large differences, depending on the specific reaction dynamics. Future MD and molecular beam studies focusing on the vibrational efficacies can help elucidate the reaction mechanism, namely, whether the CH3 radical reacts directly from the gas phase or first reorients upon chemisorption. Moreover, the vibrational efficacies are compared between the F–M α model and the η model, which show differences of up to three orders of magnitude in computed reaction rates, in particular at low temperature. Future vibrational state-specific molecular beam studies should be able to validate our predictions regarding the dissociative chemisorption of vibrationally excited CH4 on catalyst surfaces.
Overall, these results offer valuable insights into catalytic C–H activation and the impact of vibrational excitation and may be of help in developing efficient catalysts for plasma–catalytic CH4 conversion. We hope that our study will inspire further exploration through high-dimensional (quantum) dynamical calculations and experiments, enriching our comprehension of reaction dynamics in plasma catalysis.
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. Shangkun Li: conceptualization, validation, formal analysis, resources, data curation, writing original draft, and writing – review and editing. Santu Luo: conceptualization, validation, formal analysis, resources, data curation, writing original draft, and writing – review and editing. Rui Liu: conceptualization, validation, and writing – review and editing. Zhaolun Cui: conceptualization, validation, and writing – review and editing. Yanhui Yi: conceptualization, validation, and writing – review and editing. Erik C. Neyts: formal analysis, resources, data curation, writing – original draft, writing – review and editing, and supervision. Annemie Bogaerts: formal analysis, resources, data curation, writing – original draft, writing – review and editing, supervision, and funding acquisition. Nick Gerrits: formal analysis, resources, data curation, writing – original draft, and writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting the results of this research article are available in the article and its supplementary information (SI). Supplementary information contains convergence and CPU timing benchmarks; depiction and several computational results of various adsorbed and transition states; vibrational mode analysis; dependence of barriers on the density functional; temperature dependence of kη/kFM. See DOI: https://doi.org/10.1039/d5cp03569d.Additional data can be made available, upon reasonable request.
Acknowledgements
The research was supported by the China Scholarship Council and the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation programme (Grant Agreement No. 810182 – SCOPE ERC Synergy project). We acknowledge financial support from the National Natural Science Foundation of China [22472018 and 22272015]. The computational resources and services used in this work were provided by the HPC core facility CalcUA of the Universiteit Antwerpen, and VSC (Flemish Supercomputer Center). We thank Dr Jintao Sun and Roel Michiels for their many valuable discussions.Notes and references
- P. Schwach, X. Pan and X. Bao, Chem. Rev., 2017, 117(13), 8497–8520 CrossRef CAS PubMed.
- S. Li, R. Ahmed, Y. Yi and A. Bogaerts, Catalysts, 2021, 11, 590 CrossRef CAS.
- N. Gerrits, B. Jackson and A. Bogaerts, J. Phys. Chem. Lett., 2024, 15(9), 2566–2572 CrossRef CAS PubMed.
- A. A. Latimer, A. R. Kulkarni, H. Aljama, J. H. Montoya, J. S. Yoo, C. Tsai, F. Abild-Pedersen, F. Studt and J. K. Norskov, Nat. Mater., 2017, 16(2), 225–229 CrossRef CAS PubMed.
- L. Lefferts, Angew. Chem., Int. Ed., 2024, 63(10), e202305322 CrossRef CAS PubMed.
- A. Bogaerts, Nat. Chem. Eng., 2025, 2, 336–340 CrossRef.
- A. Bogaerts, X. Tu, J. C. Whitehead, G. Centi, L. Lefferts, O. Guaitella, F. Azzolina-Jury, H.-H. Kim, A. B. Murphy, W. F. Schneider, T. Nozaki, J. C. Hicks, A. Rousseau, F. Thevenet, A. Khacef and M. Carreon, J. Phys. D: Appl. Phys., 2020, 53(44), 443001 CrossRef CAS.
- J. Van Turnhout, K. Rouwenhorst, L. Lefferts and A. Bogaerts, EES Catal., 2025, 3, 669–693 RSC.
- Y. Yi, S. Li, Z. Cui, Y. Hao, Y. Zhang, L. Wang, P. Liu, X. Tu, X. Xu, H. Guo and A. Bogaerts, Appl. Catal., B, 2021, 296, 120384 CrossRef CAS.
- E. C. Neyts, K. K. Ostrikov, M. K. Sunkara and A. Bogaerts, Chem. Rev., 2015, 115(24), 13408–13446 CrossRef CAS PubMed.
- B. Loenders, R. Michiels and A. Bogaerts, J. Energy Chem., 2023, 85, 501–533 CrossRef CAS.
- T. Nozaki, N. Muto, S. Kadio and K. Okazaki, Catal. Today, 2004, 89(1–2), 67–74 CrossRef CAS.
- D. Y. Kim, H. Ham, X. Chen, S. Liu, H. Xu, B. Lu, S. Furukawa, H. H. Kim, S. Takakusagi, K. Sasaki and T. Nozaki, J. Am. Chem. Soc., 2022, 144(31), 14140–14149 CrossRef CAS PubMed.
- P. Mehta, P. Barboun, F. A. Herrera, J. Kim, P. Rumbach, D. B. Go, J. C. Hicks and W. F. Schneider, Nat. Catal., 2018, 1(4), 269–275 CrossRef.
- Y. Engelmann, K. van’t Veer, Y. Gorbanev, E. C. Neyts, W. F. Schneider and A. Bogaerts, ACS Sustainable Chem. Eng., 2021, 9(39), 13151–13163 CrossRef CAS.
- Y. Engelmann, P. Mehta, E. C. Neyts, W. F. Schneider and A. Bogaerts, ACS Sustainable Chem. Eng., 2020, 8(15), 6043–6054 CrossRef CAS.
- R. Michiels, Y. Engelmann and A. Bogaerts, J. Phys. Chem. C, 2020, 124(47), 25859–25872 CrossRef CAS.
- A. Fridman, Plasma Chemistry, Cambridge Univ. Press, New York, NY, 2008 Search PubMed.
- N. Gerrits and A. Bogaerts, EES Catal., 2025, 3, 733–742 RSC.
- F. van den Bosch, N. Gerrits and J. Meyer, EES Catal., 2025, 3(6), 1257–1271 RSC.
- K. M. Bal and E. C. Neyts, J. Phys. D: Appl. Phys., 2021, 54(39), 394004 CrossRef CAS.
- S. Roy, N. K. J., N. Tiwari and A. K. Tiwari, Int. Rev. Phys. Chem., 2020, 39(3), 267–318 Search PubMed.
- M. Karplus, R. N. Porter and R. D. Sharma, J. Chem. Phys., 1965, 43(9), 3259–3287 CrossRef CAS.
- K. M. Bal, A. Bogaerts and E. C. Neyts, J. Phys. Chem. Lett., 2020, 11(2), 401–406 CrossRef CAS PubMed.
- W. H. Miller, N. C. Handy and J. E. Adams, J. Chem. Phys., 1980, 72, 99–112 CrossRef CAS.
- B. Jackson and S. Nave, J. Chem. Phys., 2011, 135, 114701 CrossRef PubMed.
- S. Roy and A. K. Tiwari, Phys. Chem. Chem. Phys., 2022, 24, 16596–16610 RSC.
- N. Gerrits, H. Chadwick and G. J. Kroes, J. Phys. Chem. C, 2019, 123(39), 24013–24023 CrossRef CAS PubMed.
- N. Gerrits, D. Migliorini and G. J. Kroes, J. Chem. Phys., 2018, 149(22), 224701 CrossRef PubMed.
- N. Gerrits, J. Geweke, E. W. F. Smeets, J. Voss, A. M. Wodtke and G. J. Kroes, J. Phys. Chem. C, 2020, 124(29), 15944–15960 CrossRef CAS.
- F. Nattino, C. Diaz, B. Jackson and G. J. Kroes, Phys. Rev. Lett., 2012, 108(23), 236104 CrossRef PubMed.
- B. Jackson, F. Nattino and G. J. Kroes, J. Chem. Phys., 2014, 141(5), 054102 CrossRef PubMed.
- X. Zhou, B. Jiang and H. Guo, J. Phys. Chem. C, 2019, 123(34), 20893–20902 CrossRef CAS.
- R. Moiraghi, A. Lozano, E. Peterson, A. Utz, W. Dong and H. F. Busnengo, J. Phys. Chem. Lett., 2020, 11(6), 2211–2218 CrossRef CAS PubMed.
- B. Jiang, R. Liu, J. Li, D. Xie, M. Yang and H. Guo, Chem. Sci., 2013, 4(8), 3249–3254 RSC.
- B. Jackson and S. Nave, J. Chem. Phys., 2011, 135(11), 114701 CrossRef PubMed.
- P. M. Hundt, M. E. van Reijzen, H. Ueta and R. D. Beck, J. Phys. Chem. Lett., 2014, 5(11), 1963–1967 CrossRef CAS PubMed.
- B. Jiang and H. Guo, J. Chem. Phys., 2013, 138(23), 234104 CrossRef PubMed.
- H. Guo and B. Jiang, Acc. Chem. Res., 2014, 47(12), 3679–3685 CrossRef CAS PubMed.
- F. Nattino, H. Ueta, H. Chadwick, M. E. van Reijzen, R. D. Beck, B. Jackson, M. C. van Hemert and G. J. Kroes, J. Phys. Chem. Lett., 2014, 5(8), 1294–1299 CrossRef CAS PubMed.
- H. Guo, A. Farjamnia and B. Jackson, J. Phys. Chem. Lett., 2016, 7(22), 4576–4584 CrossRef CAS PubMed.
- P. Maroni, D. C. Papageorgopoulos, M. Sacchi, T. T. Dang, R. D. Beck and T. R. Rizzo, Phys. Rev. Lett., 2005, 94(24), 246104 CrossRef.
- S. Nave and B. Jackson, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 81(23), 233408 CrossRef.
- A. Wang, J. Li and T. Zhang, Nat. Rev. Chem., 2018, 2(6), 65–81 CrossRef CAS.
- J.-C. Liu, Y. Tang, Y.-G. Wang, T. Zhang and J. Li, Natl. Sci. Rev., 2018, 5, 5 CrossRef.
- J. Sun, Q. Chen, Y. Guo, Z. Zhou and Y. Song, J. Energy Chem., 2020, 46, 133–143 CrossRef.
- T. Butterworth, A. van de Steeg, D. van den Bekerom, T. Minea, T. Righart, Q. Ong and G. van Rooij, Plasma Sources Sci. Technol., 2020, 29(9), 095007 CrossRef CAS.
- S. Li, J. Sun, Y. Gorbanev, K. van’t Veer, B. Loenders, Y. Yi, T. Kenis, Q. Chen and A. Bogaerts, ACS Sustainable Chem. Eng., 2023, 11(42), 15373–15384 CrossRef CAS.
- G. F. Kresse and J. Hafner, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47(1), 558–561 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- P. E. Blochl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50(24), 17953–17979 CrossRef PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Phys. Rev. B: Condens. Matter Mater. Phys., 1998, 57, 1505–1509 CrossRef CAS.
- Z.-Q. Huang, T. Zhang, C.-R. Chang and J. Li, ACS Catal., 2019, 9(6), 5523–5536 CrossRef CAS.
- L. Chen, I. A. W. Filot and E. J. M. Hensen, ACS Catal., 2023, 13(23), 15230–15247 CrossRef CAS.
- Z.-Q. Huang, L.-P. Liu, S. Qi, S. Zhang, Y. Qu and C.-R. Chang, ACS Catal., 2017, 8(1), 546–554 CrossRef.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132(15), 154104 CrossRef PubMed.
- G. Henkelman, B. P. Uberuaga and H. Jónsson, J. Chem. Phys., 2000, 113(22), 9901–9904 CrossRef CAS.
- G. Henkelman and H. Jónsson, J. Chem. Phys., 1999, 111(15), 7010–7022 CrossRef CAS.
- V. L. Deringer, A. L. Tchougreeff and R. Dronskowski, J. Phys. Chem. A, 2011, 115(21), 5461–5466 CrossRef CAS PubMed.
- S. Maintz, V. L. Deringer, A. L. Tchougreeff and R. Dronskowski, J. Comput. Chem., 2016, 37(11), 1030–1035 CrossRef CAS PubMed.
- W. Tang, E. Sanville and G. Henkelman, J. Phys.: Condens. Matter, 2009, 21(8), 084204 CrossRef CAS PubMed.
- Y. Zhang, S. Zhao, J. Feng, S. Song, W. Shi, D. Wang and H. Zhang, Chem, 2021, 7, 2022–2059 CAS.
- J. L. Chen, X. C. Jiang, L. Feng, J. Zhu, J. W. Zhao, J. X. Liu and W. X. Li, Nat. Commun., 2025, 16, 9144 CrossRef CAS PubMed.
- W. Ji, N. Wang, X. Chen, Q. Li, K. Lin, J. Deng, J. Chen and X. Xing, Inorg. Chem., 2022, 61(26), 10006–10014 CrossRef CAS PubMed.
- M. D. Marcinkowski, M. T. Darby, J. Liu, J. M. Wimble, F. R. Lucci, S. Lee, A. Michaelides, M. Flytzani-Stephanopoulos, M. Stamatakis and E. C. H. Sykes, Nat. Chem., 2018, 10(3), 325–332 CrossRef CAS PubMed.
- T. Tchakoua, N. Gerrits, E. W. F. Smeets and G. J. Kroes, J. Chem. Theory Comput., 2023, 19(1), 245–270 CrossRef CAS PubMed.
- H. Guo and B. Jackson, J. Chem. Phys., 2019, 150(20), 204703 CrossRef PubMed.
- N. Gerrits, Front. Chem., 2024, 12, 1481235 CrossRef CAS PubMed.
- H. Chadwick, D. Migliorini and G. J. Kroes, J. Chem. Phys., 2018, 149(4), 044701 CrossRef CAS PubMed.
- M. D. Krcha, A. D. Mayernick and M. J. Janik, J. Catal., 2012, 293, 103–115 CrossRef CAS.
- S. Tomar, B. S. Bhadoria, H. Jeong, J. H. Choi, S.-C. Lee and S. Bhattacharjee, J. Phys. Chem. C, 2024, 128, 8580–8589 CrossRef CAS.
- Z. Liang, T. Li, M. Kim, A. Asthagiri and J. F. Weaver, Science, 2017, 356, 299–303 CrossRef CAS PubMed.
- S. S. Sason, C. H. Phillippe, W. John and H. Sons, Chrom., 2009, 69(11–12), 1493 Search PubMed.
- P. Mehta, P. Barboun, D. B. Go, J. C. Hicks and W. F. Schneider, ACS Energy Lett., 2019, 4(5), 1115–1133 CrossRef CAS.
- A. D. Powell, N. Gerrits, T. Tchakoua, M. F. Somers, H. F. Busnengo, J. Meyer, G. Kroes and K. Doblhoff-Dier, J. Phys. Chem. Lett., 2024, 15, 307–315 CrossRef CAS PubMed.
- N. Gerrits, J. Geweke, D. J. Auerbach, R. D. Beck and G. J. Kroes, J. Phys. Chem. Lett., 2021, 12(30), 7252–7260 CrossRef CAS PubMed.
- J. C. Whitehead, J. Phys. D: Appl. Phys., 2016, 49(24), 243001 CrossRef.
- J. J. Qiao, Q. Yang, D. Z. Wang and Q. Xiong, Plasma Sources Sci. Technol., 2023, 32(11), 11LT01 CrossRef CAS.
Footnote |
| † S. Li and S. Luo contributed equally. |
| This journal is © the Owner Societies 2026 |