Open Access Article
Open Access ArticleStringent selection on kinetics of condensation reactions: early steps in chemical evolution
Pau
Capera-Aragones
abc,
Kavita
Matange
ab,
Vahab
Rajaei
ab,
Yuval
Pinter
 d,
Anton S
Petrov
d,
Anton S
Petrov
 ab,
Loren Dean
Williams
ab,
Loren Dean
Williams
 *ab and
Moran
Frenkel-Pinter
*ab and
Moran
Frenkel-Pinter
 *acef
*acef
aNASA Center for Integration of the Origins of Life, Georgia Institute of Technology, Atlanta, GA 30332-0400, USA
bSchool of Chemistry and Biochemistry, Georgia Institute of Technology, 315 Ferst Drive NW, Atlanta, GA 30332-0400, USA. E-mail: loren.williams@chemistry.gatech.edu; Tel: (+1) (404) 385-6258
cInstitute of Chemistry, The Hebrew University of Jerusalem, Edmond J. Safra Campus, Jerusalem 9190401, Israel. E-mail: moran.fp@mail.huji.ac.il; Tel: (+972)-2-6584171
dFaculty of Computer and Information Science, Ben-Gurion University of the Negev, Israel
eBlue Marble Space Institute of Science, Seattle, WA 98104, USA
fThe Casali Center of Applied Chemistry, The Hebrew University of Jerusalem, Jerusalem 9190401, Israel
First published on 27th January 2026
Abstract
The emergence of chemical selectivity poses a central challenge in origins-of-life research. As demonstrated by analyses of asteroid and meteorite samples, abiotic chemistry is incredibly messy. Experiments show that even limited sets of reactive species can undergo vast numbers of distinct chemical transformations, leading to a combinatorial explosion of products. These explosions arise from the numerous ways in which reactants in mixtures can combine, generating large and chemically diverse ensembles that reduce or even preclude the possibility of productive pathways of chemical evolution. However, recent empirical studies have demonstrated that chemical systems can exhibit combinatorial compression – a marked reduction in product diversity relative to combinatorial expectations. This selection is observed under conditions of low water activity, such as in the dry phase of wet–dry cycling experiments. Here, we integrate transition-state theory with computer simulations to demonstrate that experimentally observed combinatorial compression is a consequence of kinetic selection in condensation–dehydration reactions. Kinetic selection depends on several key factors: (i) chemical connectivity, where multiple species can react with each other; (ii) at least one particularly reactive species – termed a “kinetic compressor”; and (iii) appropriate temperature, concentrations, and reaction times. We find that small differences in activation free energies, on the order of just ∼3 kcal mol−1, can dominate a kinetic landscape, dramatically limiting product distributions. Connected systems can favor a narrow subset of products, suggesting selection mechanisms in prebiotic contexts. Our results provide mechanistic insight into combinatorial compression, establish a quantitative framework for exploring the emergence of stringent chemical selectivity, and can guide future experimental efforts in chemical evolution.
Introduction
Geochemical and environmental processes on the ancient Earth gave rise to biology by processes that are poorly understood. Increasing complexity of large molecules,1 accompanied by a reduction in chemical diversity, ultimately favored a small set of building blocks that now compose the universal biopolymers – RNA, DNA, proteins, and glycans.2 Recognizing that spontaneous, unguided processes in natural environments produced the remarkable molecules of life can inspire exploration of new frontiers in chemistry.Key insights into the origins of biopolymers have come from considering the effects of oscillating water activity. The rotation of Earth (diurnal cycling) induces daily wet and dry phases, with surfaces wetting at night and drying during the day.3,4 Such wet–dry cycling causes systematic alterations in the driving forces of reactions involving water as a reactant or product. Dry phases promote condensation–dehydration reactions and the formation of oligomers, while wet phases favor hydrolysis and the breakdown of oligomers.
It has been demonstrated that when diverse mixtures of mercapto acids, hydroxy acids, and amino acids are concentrated at low water activity, they can form interconnected reaction networks with emergent behaviors.5–13 Emergent behaviors are those not predicted by the properties of isolated components.14 One such emergent behavior is combinatorial compression. Experiments have shown that wet–dry cycling at modest temperatures imposes chemical selection, favoring a limited set of products from many possible reaction outcomes.12,13
Combinatorial compression is the production of few product types, representing only a small fraction of what is theoretically possible (Fig. 1). Under experimental conditions of combinatorial compression, reinitiation of a reaction with additional (new) reactants led to the disappearance of previous products (product subtraction) and the appearance of new products, without a combinatorial increase in the total number of products (Fig. 1). Addition of new reactants causes product identities to shift but does not significantly increase product counts. This effect is observed at relatively low temperatures (45 °C) but not at higher temperatures (85 °C), where selection collapses and products proliferate (combinatorial explosion15–19). As the reaction temperature increases, a system transitions from combinatorial compression to combinatorial explosion.12
 | ||
| Fig. 1 A schematic representation of combinatorial compression in a UV-vis HPLC chromatogram of products of dry-down reactions as previously published.12 Each peak here corresponds to a dimer product. Top: two monomers A and B combine to form four dimers: AB, BA, AA, and BB. Middle: three monomers A, B, and C combine to form nine dimers under conditions of combinatorial explosion. Bottom: three monomers, A, B, and C, combine to form four dimers under conditions of combinatorial compression. Here, combinatorial compression is represented by the absence of dimers AB, BA, AC, CA, and CC. Product subtraction is indicated by vertical dashed lines. | ||
Although combinatorial compression has been observed experimentally,12 it has not previously been addressed theoretically, and its mechanistic basis remained unknown. Here, we integrate transition state theory with computer simulations and demonstrate that combinatorial compression is a consequence of kinetic selection in condensation–dehydration reactions and depends on several key factors: (i) chemical connectivity, where multiple species can react with each other; (ii) a particularly reactive species – termed a ‘compressor’; and (iii) appropriate temperature, concentrations and reaction time. We find that small differences in activation free energies, on the order of just 3–5 kcal mol−1, can dominate a kinetic landscape, dramatically limiting product distributions. The terms compressor and chemical connectivity are defined more fully in subsequent sections. A mechanistic explanation for combinatorial compression enables new experimental and theoretical strategies for driving the evolution of complex chemical mixtures. These findings have implications for the origins of life, systems chemistry, and dynamic combinatorial chemistry.20–23
Results
Combinatorial compression under kinetic control
| Mi + Mj ⇌ Dij + W |
The kinetic and thermodynamic parameters (Table 1) are similar to those of known prebiotic reactions24 but do not represent exact values for any specific system. To capture the inherent variability of chemical processes in mixtures, each parameter is defined by a mean and standard deviation. We examine system behavior across a range of parameter values to identify conditions that produce combinatorial compression.
| Parameter | Value |
|---|---|
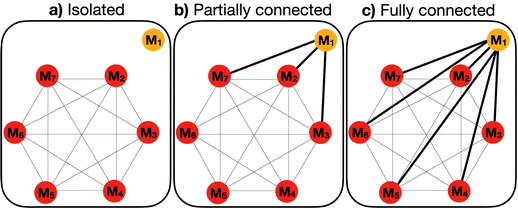
| a These simulations take place in the dry phase where the concentration of water is low and the concentration of monomers is high. For the compressor, a range of initial concentrations were investigated. In some cases, the system was fed with a compressor, by holding its concentration constant at 0.05 M. b The mean activation reaction free energy, and the standard enthalpy, entropy, and free energy of dimerization for reactions that do not involve the kinetic compressor. The free energy of reactions varies with temperature. c The standard deviation of the activation reaction free energy, and the standard enthalpy, entropy, and free energy of reactions that do not involve the kinetic compressor. The standard deviation is defined by a uniform random distribution within an interval centered on the mean. For the kinetic compressor, no variation is applied. d The concentration threshold is the minimum concentration required for a monomer or dimer to be considered present in the reaction mixture. e All monomers are assumed to be fully connected – they can react with each other – except for the kinetic and thermodynamic compressors, whose connectivity varies among three defined states: fully connected, partially connected, and isolated. A fully connected compressor can react with all other monomers to form dimers; a partially connected compressor reacts with only a subset of monomers; and an isolated compressor does not react with any. f Previous experiments have shown that this temperature is appropriate for combinatorial compression.12 For some simulations, the variation of temperature is investigated. g Previous experiments have shown that this time interval is appropriate for combinatorial compression.12 h The mean activation free energy for dimerization reactions involving the kinetic compressor. For some simulations, this parameter is varied. i The mean standard enthalpy of reactions involving the thermodynamic compressor. For some simulations, this parameter is varied. j The mean of the free energy of reactions involving the thermodynamic compressor. This parameter varies with ΔH°(rxn) and the temperature. | |
| Number of monomers | 12 |
| [Water] | 0.5 Ma |
| [Monomer]initial | 0.05 Ma |
| [Dimer]initial | 0 M |
| Mean ΔG‡ | 25 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
| Standard deviation of ΔG‡ [σ(ΔG‡)] | 0.29 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
| Mean ΔH0rxn | 8 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
| Standard deviation of ΔH0rxn [σ(ΔH0rxn)] | 0.29 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
| Mean ΔS0rxn | 0.033 kcal mol−1 K−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
| Standard deviation of ΔS0rxn [σ(ΔS0rxn)] | 0 kcal mol−1 K−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
| Mean ΔGrxn | −2.4 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
| Standard deviation of ΔGrxn [σ(ΔGrxn)] | 0.29 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
| Concentration threshold | 10−4 Md |
| Connectivity | Fully connectede |
| Temperature | 40 °Cf |
| Time interval | 1000 sg |
| Kinetic compression | |
| [Kinetic compressor]initial | 2.5 Ma |
Mean ΔG‡kinetic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) compressor compressor |
20 kcal mol−1 (ΔΔG‡ = −5 kcal mol−1)h |
σ(ΔG‡kinetic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) compressor) compressor) |
0 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
| Thermodynamic compression | |
| [Thermodynamic compressor]initial | 25 M![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Mean ΔH0rxn,thermodynamic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) compressor compressor |
0 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i i |
σ(ΔH0rxn,thermodynamic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) compressor) compressor) |
0 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
Mean ΔGrxn,thermodynamic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) compressor compressor |
−10.4 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j (ΔΔGrxn = −8 kcal mol−1) j (ΔΔGrxn = −8 kcal mol−1) |
σ(ΔGrxn,thermodynamic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) compressor) compressor) |
0 kcal mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
The simulations are run in two modes: kinetic (finite time intervals) and thermodynamic (equilibrium, no time constraint). We assume homogeneous systems with negligible diffusion limitations,25 constant activity coefficients, and constant pH. Temperature dependencies of enthalpy and entropy are neglected, and entropy contributions are assumed to be small, such that reaction free energies are predominantly determined by enthalpy. At most one compressor species is present in any simulation, though future work will explore multiple compressors. Subsequent studies will expand the parameter space explored here.
We compared computation with experiment to test the utility of our model and to define the rules governing combinatorial compression. We observed in simulations that combinatorial compression depends on the thermodynamic and kinetic properties of reactants and products, concentrations, and on temperature and reaction time.
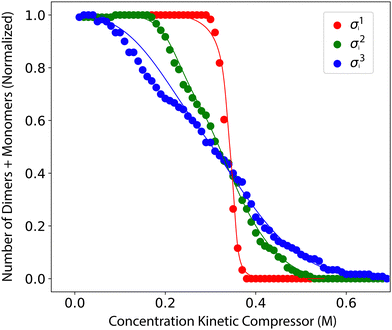 | ||
| Fig. 4 The number of dimers plus monomers above the concentration threshold depends on the concentration of the kinetic compressor and σ(ΔG‡), the standard deviation of the activation free energies. Parameters for this simulation are as indicated in Table 1, except for σ(ΔG‡), which is set to three different values: σ1i(ΔG‡) = 0.04 (red line), σ2i(ΔG‡) = 0.21 (green), or σ3i(ΔG‡) = 0.34 (blue) kcal mol−1. Results are normalized from 0 to 1; values reaching the minimum appear as zero. | ||
The concentration of the kinetic compressor required to convert a system from combinatorial explosion to compression depends on the extent of connectivity, ΔΔG‡, the temperature, and the number and concentration of monomers. Maintaining kinetic compression as the total concentration of non-compressing monomers increases requires a corresponding increase in the concentration of the kinetic compressor. This dependence arises because reaction fluxes are governed by aggregate encounter rates. The compressor competes not with a single species, but with the entire pool of non-compressing reactants; the total rate at which these species either react with the compressor or with each other scales with the sum of their concentrations. As a result, the effectiveness of kinetic compression is controlled by the ratio between the compressor concentration and the total concentration of competing reactants, rather than by any individual concentration alone. For the conditions in Table 1, an initial concentration of kinetic compressor of >0.3 M is required to observe kinetic compression (Fig. 4).
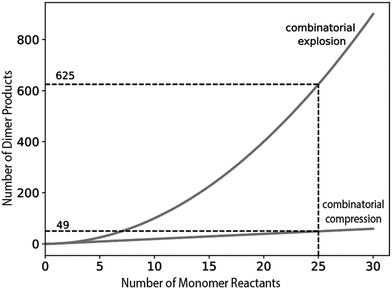 | ||
| Fig. 5 In the absence of a kinetic compressor, the number of product dimers above the concentration threshold increases exponentially with the number of reactant monomers. In the presence of a kinetic compressor, the number of dimers above the threshold increases linearly with the number of monomers. The reaction conditions are shown in Table 1. | ||
The simulations demonstrate how under specific conditions, addition of new monomers can systematically add new dimers and subtract other dimers from the product mixture. Fig. 6a shows that in the absence of a kinetic compressor in a highly connected system, all dimers rise above the concentration threshold. This simulation corresponds to a combinatorial explosion, as observed in Fig. 5. However, reinitiating the reaction in the presence of a kinetic compressor leads to the emergence of a new set of dimers and the suppression of the previous set. The newly formed dimers contain the kinetic compressor linked to other monomers and are produced via fast reactions with low activation free energies. In contrast, the suppressed dimers arise from pairs of monomers that do not include the kinetic compressor and react with higher activation free energies. As a result, this latter set of dimers does not exceed the concentration threshold at the time point of measurement (Fig. 6b). Chemical selection based on reaction rates therefore stems, in part, from the rapid consumption of certain monomers by the kinetic compressor. These quickly consumed reactants are unavailable for other reactions, subtracting products from the reaction mixture. Our simulations demonstrate that product subtraction by a kinetic compressor is more pronounced when the kinetic compressor reacts at lower reaction free energy (greater negative ΔΔG‡) (Fig. S1). We observe resilience in production of some dimers that are not connected to the compressor and are not subtracted by the compressor.
 | ||
| Fig. 6 Time evolution of monomer (red lines) and dimer (blue lines) concentrations in the (a) absence of a kinetic compressor and (b) presence of a kinetic compressor (orange line). All monomers are fully connected except for the kinetic compressor, which is partially connected, meaning that it can only react with a subset of monomers. Dimers that contain the kinetic compressor form quickly (green lines). Dimers that do not contain the kinetic compressor form slowly (blue lines). The kinetic compressor delays the rise of some products (subtracted products in b). The vertical dashed line indicates the time point at which measurements are made. Parameters for this simulation are shown in Table 1. | ||
The balance between combinatorial compression and explosion depends on chemical connectivity, ΔΔG‡, concentrations, time, and temperature. Fig. 7 shows that when ΔΔG‡ is close to zero and connectivity is low, all dimers rise above the concentration threshold, even at low temperature. In this exploded state, 12 reactants combine to form 144 dimers. In contrast, when ΔΔG‡ is large in magnitude and connectivity is high, many dimers remain below the threshold. In this compressed state, 12 monomers, including the kinetic compressor, form only 23 dimers. Kinetic compression is observed only when the system is under kinetic control and is pronounced at relatively low temperatures and short reaction times. Indeed, our previous experiments demonstrate that modest increases in temperature cause the number of products to explode.12
 | ||
| Fig. 7 Effects of connectivity and ΔΔG‡ on the reaction of monomers to dimers. The numbers of dimers and monomers above the concentration threshold at time = 1000 s are determined by chemical connectivity and ΔΔG‡. (a) Chemical connectivity of the compressor is varied from isolated, to partially connected (the kinetic compressor reacts with 3 out of 12 monomers), to fully connected (the kinetic compressor reacts with 12 out of 12 monomers). The ΔΔG‡ of the kinetic compressor varies from 0 kcal mol−1 (no compression), to −3 kcal mol−1, to −5 kcal mol−1 (strong compression). The time point at which concentrations are determined is indicated by a vertical dashed line at time = 1000 s. (b) A summary showing how concentrations of dimers are influenced by connectivity and ΔΔG‡. Parameters for this simulation are shown in Table 1 except for variation of ΔΔG‡ and connectivity of the compressor. The concentration of the kinetic compressor is kept constant at 0.05 M through feeding. | ||
Combinatorial compression under thermodynamic control
Here, we describe simulations of combinatorial compression in connected systems that have reached equilibrium. A thermodynamic compressor (Mtc) must be highly connected, at high concentration, and have a free energy of reaction (ΔGrxn) greater in magnitude than the free energies of reaction of other monomers (ΔΔGrxn ≈ −1 to −8 kcal mol−1, Fig. 8a). Hence, the reaction of the thermodynamic compressor has greater driving force than the reactions of other monomers.20–23 | ||
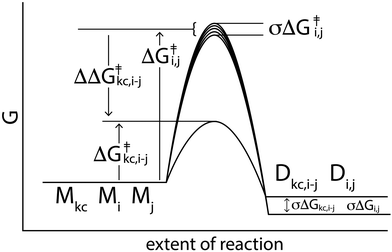
| Fig. 8 Effect of connectivity and ΔΔGrxn in the reaction of monomers to dimers. (a) A schematic of a reaction coordinate indicating relative free energies of reaction for Mi + Mj → Di,j and Mi + Mtc → Di,tc, where Mtc is the thermodynamic compressor. (b) The number of dimers and monomers above the concentration threshold at equilibrium depends on the concentration of the thermodynamic compressor and the standard deviation of the free energies of reaction. Parameters for this simulation are as indicated in Table 1, except the standard deviation in free energies of reaction, which is set to three different values: σi(ΔGrxn) = 0.04 (red line), 0.21 (green), or 0.34 (blue) kcal mol−1. The time interval is 109 seconds, which allows equilibrium. Results are normalized from 0 to 1; values reaching the minimum appear as zero. (c) Changes in concentrations of dimers over time and at equilibrium are influenced by connectivity and ΔΔGrxn. Chemical connectivity varies from isolated, to ‘partially connected’ in which the thermodynamic compressor is connected to 3 out of 12 monomers, to fully connected. The ΔΔGrxn for reactions of the thermodynamic compressor varies from −2.8 kcal mol−1 (ΔΔGrxn = 0 kcal mol−1), to −3.8 kcal mol−1 (ΔΔGrxn = −1 kcal mol−1), to −10.8 kcal mol−1 (ΔΔGrxn = −8 kcal mol−1). The concentration of the thermodynamic compressor is held constant at 0.05 M. (d) The number of dimers and monomers above the concentration threshold is determined by ΔΔGrxn and chemical connectivity of the compressor. The concentration of thermodynamic compressor is kept constant at 0.05M through feeding in (c) and (d). | ||
In our equilibrium simulations, the ΔH0 for reactions between monomers is 8 kcal mol−1, except for reactions involving the thermodynamic compressor, for which it is 0 kcal mol−1 (Fig. 8a). The ΔS0 is 0.033 kcal mol−1 K−1 for all reactions. ΔG = ΔH0 − TΔS0 at 40 °C is −10.4 kcal mol−1 for reactions with the thermodynamic compressor and −2.4 kcal mol−1 for all other reactions. The value of ΔG(rxn) changes over the course of the reaction as concentrations of reactants and products change, but is generally slightly less than 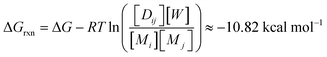 for the thermodynamic compressor and ≈−2.8 kcal mol−1 for all other dimerization reactions. Other reaction conditions are specified in Table 1. The balance between compression and explosion at equilibrium depends on reaction free energies (Fig. 8a), concentrations (Fig. 8b), and chemical connectivity (Fig. 8c and d). Fig. 8d shows that increasing the connectivity or increasing the magnitude of ΔΔGrxn of one monomer causes the number of detectable dimers to decrease from 144 to 23. Thermodynamic compression requires relatively high temperatures or long reaction times.
for the thermodynamic compressor and ≈−2.8 kcal mol−1 for all other dimerization reactions. Other reaction conditions are specified in Table 1. The balance between compression and explosion at equilibrium depends on reaction free energies (Fig. 8a), concentrations (Fig. 8b), and chemical connectivity (Fig. 8c and d). Fig. 8d shows that increasing the connectivity or increasing the magnitude of ΔΔGrxn of one monomer causes the number of detectable dimers to decrease from 144 to 23. Thermodynamic compression requires relatively high temperatures or long reaction times.
In comparison to kinetic control, under thermodynamic control, the transition from combinatorial explosion to compression requires a higher concentration of compressor. In the kinetics simulation, the transition from explosion to compression occurs with an initial concentration of kinetic compressor of around 10 times the concentration of other monomers (Fig. 4). In the thermodynamics simulation, the transition from explosion to compression occurs at an initial concentration of the thermodynamic compressor of around 100 times the concentration of other monomers (5 M, Fig. 8b). In both the kinetic and thermodynamic realms, the number of products varies inversely with the concentration of the compressor. In the simulations in Fig. 8c and d, the thermodynamic compressor is fed to the reaction continuously, such that it is maintained at a constant concentration as it is consumed by the reactions. This protocol is followed to avoid the high concentrations of thermodynamic compressor required for compression, which gives a kinetic advantage to the thermodynamic compressor that obscures the true effect of a thermodynamic compression.
Thermodynamic and kinetic compression have fundamentally different origins and are observed under strikingly different conditions: thermodynamic control for thermodynamic compression versus kinetic control for kinetic compression. Kinetic compression appears to be more relevant than thermodynamic compression to selection during chemical evolution, according to recent experiments.12
Systems with both kinetic and thermodynamic compression
To understand how temperature and time affect combinatorial compression, we simulated evolution of a combined system with both kinetic and thermodynamic compression. These simulations employed both a kinetic and a thermodynamic compressor. The two compressors are distinct monomers. Monomers in the systems are fully connected. Feeding (continuous addition) maintained the constant concentration of both the kinetic and the thermodynamic compressors (Fig. 9 and 10). The simulations show that the mixture is kinetically compressed at low temperature, under kinetic control, with under 20% of the normalized monomers and dimers above the threshold after a given time interval. The system is combinatorially exploded at intermediate temperatures, with 100% of possible monomers and dimers over the concentration threshold. The system is thermodynamically compressed at high temperatures, under thermodynamic control, with under 18% of the normalized monomers and dimers over the concentration threshold (Fig. 9a). A set of dimers dominates the reaction under kinetic control. A different set of dimers dominates the system under thermodynamic control. Combinatorial compression at low temperatures produced dimers containing the kinetic compressor. Combinatorial compression at high temperatures produces dimers containing the thermodynamic compressor. No single dimer dominates the mixture in both realms. These compositions are confirmed by independent simulations of a system with only a thermodynamic compressor, in which combinatorial compression is observed only at high temperatures, and simulations of a system with only a kinetic compressor, in which combinatorial compression is observed only at low temperatures (Fig. S2). At a fixed temperature, the system transitions over time from kinetic compression to combinatorial explosion to thermodynamic compression (Fig. 9b). The balance between combinatorial compression and explosion is dependent on the reaction time, in a manner similar to the temperature (Fig. 9a). However, this relationship may not hold for reactions in which entropy makes a major contribution to the free energy of reactions or when enthalpy and entropy exhibit a strong temperature dependence. | ||
Fig. 9 Kinetic and thermodynamic compression. (a) Normalized number of monomers plus dimers versus temperature at a constant time of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 seconds. (b) Normalized number of monomers plus dimers versus time at a constant temperature of 40 °C. Both panels show transitions from kinetic compression to explosion to thermodynamic compression. The concentrations of both kinetic and thermodynamic compressors are constant at 0.05 M. Mean activation free energies are 26 kcal mol−1 (ref. 21) (red lines), 25 kcal mol−1 (ref. 20) (green lines), and 24 kcal mol−1 (ref. 19) (blue lines). Other parameters for these simulations are shown in Table 1. 000 seconds. (b) Normalized number of monomers plus dimers versus time at a constant temperature of 40 °C. Both panels show transitions from kinetic compression to explosion to thermodynamic compression. The concentrations of both kinetic and thermodynamic compressors are constant at 0.05 M. Mean activation free energies are 26 kcal mol−1 (ref. 21) (red lines), 25 kcal mol−1 (ref. 20) (green lines), and 24 kcal mol−1 (ref. 19) (blue lines). Other parameters for these simulations are shown in Table 1. | ||
 | ||
| Fig. 10 Normalized number of monomers and dimers as a function of the temperature and reaction time. Three states are identified: thermodynamic compression, combinatorial explosion, and kinetic compression. In addition, at very short reaction times and low temperatures, we observe the case in which initial components do not react significantly and persist over the course of the simulation. Parameters for this simulation are set as shown in Table 1, except for the concentrations of both kinetic and thermodynamic compressors are constant at 0.05 M. The temperature ranges from −50 °C to 200 °C, and reaction times range from 10−2 s to 108 s. | ||
The temperature and time required for the transition from combinatorial compression to explosion in the combined system are dependent on the activation energies (Fig. 9). For higher activation energies, higher temperatures or longer reaction times are required to observe the kinetic-related transition and the thermodynamic-related transition. The slope of the transition in Fig. 9 depends on the variability in activation energies (Fig. S2).
Giving the system either more time to react or increasing the temperature can drive a system from kinetic compression to combinatorial explosion, followed by a thermodynamic compression. Fig. 10 shows the plot of the normalized number of monomers and dimers as a function of the reaction time and temperature realms of kinetic compression, combinatorial explosion, and thermodynamic compression. The pseudo-symmetry across the diagonal (from top-left to bottom-right) indicates a rough equivalence of the effects of temperature and reaction time. There are subtle but important distinctions between the effects of the reaction time and the effects of temperature. A linear increase in temperature is equivalent to an exponential increase in the reaction time (see the logarithmic scale of the reaction time axis in Fig. 9 and 10). Moreover, at sufficiently low temperatures, some reactions do not occur for thermodynamic reasons. There is no combinatorial explosion at very low temperatures (Fig. 10). This behavior has no equivalence in reaction times; decreasing reaction times cannot affect the driving force (thermodynamics) of the reaction.
Thermodynamic compression is independent of initial conditions. However, kinetic compression requires initial conditions with monomers only, or semi-continuous dissociation of dimers (such as hydrolysis through dry-wet cycling) or feeding with new monomers. We plot the dependence of the states identified in Fig. 10 as a function of the number of monomer types included in the mixture, the concentration threshold for dimers, and the variability in free activation energies and enthalpies (Fig. S3). In all cases, we identify the states of combinatorial explosion and kinetic and thermodynamic compression. The transitions between these states are sharper for small variability of activation energies and enthalpies. The state of explosion is more pronounced for a low concentration threshold. In addition, we plot the dependence of the thermodynamic and kinetic compression on the initial concentration of reactants (Fig. S4). It is evident that thermodynamic compression is independent of initial concentrations whereas kinetic compression is dependent.
Discussion
Combinatorial compression under kinetic control
Using basic chemical principles and simulations, we have recapitulated experimental phenomena characteristic of chemical evolution. Stringent selection and compression under kinetic control are characterized by (i) production of fewer products than expected from the combinatorics of the reactants, (ii) loss of some products upon addition of new reactants, and (iii) temperature-dependent transitions between compression and explosion. These simulations of a single drying step are consistent with acute selection based on reaction kinetics within complex chemical ensembles. We can now explain the underlying principles of chemical selection and combinatorial compression. The simulations indicate that selection and combinatorial compression under kinetic control require the following:• Connection. Many reactants must form chemical connections to many other reactants.
• High reactivity. One (or a few) reactants must be more reactive and at a sufficient concentration and react faster than others.
• Temperature. The temperature must be appropriate to the activation energies and the reaction time.
The results are relevant to known condensation–dehydration reactions. Mixtures of mercapto acids, hydroxy acids, and amino acids form highly connected systems and react to form thioester, ester, and amide bonds, with activation free energies in the range of 20–35 kcal mol−1. These reactions are favorable during drying.5–13
The results of the simulations are general and are not dependent on specific chemical identities. Kinetic compression depends on activation energies, concentrations, connection, time and temperature. For example, a reactant with ΔG‡ = 20 kcal mol−1 might be kinetically dominant in a mixture with other reactants with ΔG‡ = 25 kcal mol−1 but not in a mixture with reactants with ΔG‡ = 20 kcal mol−1. A reactant at 0.25 M can act as a kinetic compressor if the sum of other concentrations is 0.1 M, but not if the sum of other concentrations is 0.5 M. A kinetically dominant reactant with ΔG‡ = 20 kcal mol−1 causes combinatorial compression at 45 °C while a dominant reactant with ΔG‡ = 30 kcal mol−1 would cause combinatorial compression at higher temperatures and/or longer reaction times.
Given sufficient time, a kinetically compressed system can combinatorically explode. However, kinetic compression can persist indefinitely, if a system is prevented from reaching equilibrium. Continuously changing conditions can preclude equilibrium. Wet–dry cycling, feeding of reactants, and formation of kinetically trapped products can prevent equilibrium.
Chemical evolution and stringency of selection
Selection during wet–dry cycling can be remarkably stringent, imposing sharp constraints on reaction trajectories under specific physicochemical conditions. In particular, complex chemical systems that combine cyclic environmental forcing with appropriate kinetic asymmetries can sustain high concentrations of selected products over time and, as a result, evolve chemically. While such stringent selection does not arise across the full parameter space, our results indicate that it emerges robustly within a defined and physically plausible subset of conditions, suggesting that chemical selection and combinatorial compression may represent necessary ingredients for chemical evolution.Our experiments and simulations indicate that small differences in activation free energies can control and direct product formation. We find that a modest difference of just 3 kcal mol−1, equivalent to approximately five times the thermal energy at 40 °C, is sufficient to direct and focus the flow of chemical reactions. Under the conditions of the dry phase, where condensation reactions dominate, small energetic differences can sculpt the kinetic landscape, resulting in distinct and reproducible product distributions.
The combined results suggest that production of certain products in complex chemical systems is possible from real-time selection based on inherent chemical properties and environmental conditions. Complex chemical systems are sensitive to internal and external parameters in the absence of enzymes or other mechanisms of biological control. The dry phase of wet–dry cycling acts as a stringent kinetic filter, favoring synthetic pathways with the lowest effective activation barriers. Minimal differences in reactivity can be magnified into large differences in chemical outcomes, contributing to the selectivity.
We hypothesize that additional selective pressures, acting in concert with kinetic compression, contributed to the emergence of complex oligomers and polymers during the origins of life.26 The data support a model in which molecular ensembles evolve in response to dynamic, continuously shifting selective pressures.12,27 Selection during wet–dry cycling can involve solubility in water (wet phase); intrinsic condensation kinetics (dry phase); resistance to hydrolysis (wet phase); catalytic enhancement of condensation, such as through ester–amide exchange (dry phase);10,11,28 molecular recalcitrance (wet phase);1,29 and, ultimately, autocatalytic cycles.30,31 In this model, kinetic trapping during the dry phase can drive the formation of large molecules. As the complexity of individual molecules increases, additional selective forces are likely to emerge, including deep kinetic traps formed by recalcitrant assemblies29 and the development of autocatalytic behavior.30,31
The results of our simulations here can guide the design of new experiments concerning chemical evolution. It appears that conditions expected on the early Earth can enable kinetic compression and chemical evolution. Moreover, we believe that kinetic compression is a universal phenomenon that might have broad implications in chemical and related fields.
An acute culling of chemical diversity occurred during the transition from abiotic chemistry to biochemistry. Abiotic chemical systems exhibit far greater molecular diversity than biochemical systems.2,32 During the origins of life, a net decrease in the overall chemical diversity of the evolving ensemble was accompanied by an increase in the size and complexity of individual molecules. Combinatorial compression, based on selection operating during the kinetics of condensation reactions, may have served as a key mechanism for molecular culling during the origins of life. Other compressive phenomena include the action of borate,33 the effect of metal ions and minerals,34–37 and the convergence to a single genetic code.38
Combinatorial compression under thermodynamic control
In addition to kinetic compression, in this work, we identified a phenomenon we call thermodynamic compression, based on selection on thermodynamics of reaction. In this realm, the most stable products are selected over other products.A system that is thermodynamically compressed does not explore new chemical spaces and does not undergo chemical evolution. Thermodynamic compression does not explain the experiments in ref. 12, and it is reasonable to assume that it did not drive prebiotic chemical evolution because (i) life is far from equilibrium, (ii) equilibrium has intrinsic ‘sink-like’ behavior that challenges continuous chemical change,39 and (iii) long times are required to reach a thermodynamic compression.
Kinetic and thermodynamic compression are fundamentally different phenomena, driven by different factors and occurring on different timescales. Kinetic compression arises under short reaction times or when the system is coupled to processes (e.g., dry–wet cycling) that prevent it from reaching equilibrium. In contrast, thermodynamic compression requires prolonged reaction times and occurs as a system approaches equilibrium. Kinetic compression is a transient state that emerges before the full set of possible products is realized, whereas thermodynamic compression reflects a distribution at equilibrium.
Combinatorial compression reflects chemical selection of few products from a large possibility space. In prior work, chemical selection has been observed across multiple scales by various mechanisms. Examples of systems that harvest environmental energy and invest it in chemical reactions include in vivo biochemistry and in vitro condensation reactions under wet–dry cycling conditions. Wet–dry cycling uses kinetic control and energy dissipation to select for complex oligomers.12,13 Similarly, stellar nucleosynthesis selects for stable configurations of protons and neutrons.40 Mineral evolution selects for locally stable arrangements of chemical elements.41 Biology uses Darwinian evolution to select for persistent biomolecules.
Author contributions
PCA, LDW, and MFP conceptualized the research. Computer simulations were conducted by PCA. ASP and YP contributed to improving the organization and clarity of the code. The manuscript was written and edited by all authors. All authors have approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The Python code developed to perform computer simulations of this work can be found at https://github.com/HUJI-MFP/Compression/tree/main. Using this code, all results shown in this manuscript are reproducible.Supplementary information (SI), including methods and supplementary figures, is available. See DOI: https://doi.org/10.1039/d5cp03057a.
Acknowledgements
We thank Prof. Doron Lancet for helpful discussions. This research was funded by the European Union to MFP (ERC, Sweet_Evo, 101163270), the Azrieli Foundation Early Career Faculty Grant, the Israel Science Foundation grant (1611/22), the Minerva Foundation, the FEBS Foundation Excellence Award to MFP, and the National Aeronautics Space Agency (grant no. 80NSSC24K0344). Views and opinions expressed are, however, those of the author only and do not necessarily reflect those of the European Union or the European Research Council. Neither the European Union nor the granting authority can be held responsible for them.References
- K. Matange, E. Marland, M. Frenkel-Pinter and L. D. Williams, Biological Polymers: Evolution, Function, and Significance, Acc. Chem. Res., 2025, 58, 659–672 CrossRef CAS PubMed.
- D. L. Nelson, A. L. Lehninger and M. M. Cox, Principles of Biochemistry, Macmillan, 8th edn, 2021 Search PubMed.
- I. Mamajanov, P. J. MacDonald, J. Ying, D. M. Duncanson, G. R. Dowdy and C. A. Walker, et al., Ester formation and hydrolysis during wet–dry cycles: generation of far-from-equilibrium polymers in a model prebiotic reaction, Macromolecules, 2014, 47(4), 1334–1343 CrossRef CAS.
- B. Damer and D. Deamer, The hot spring hypothesis for an origin of life, Astrobiology, 2020, 20(4), 429–452 CrossRef PubMed.
- S.-S. Yu, M. D. Solano, M. K. Blanchard, M. T. Soper-Hopper, R. Krishnamurthy and F. M. Fernández, et al., Elongation of model prebiotic proto-peptides by continuous monomer feeding, Macromolecules, 2017, 50(23), 9286–9294 CrossRef CAS.
- Z. Li, L. Li, K. R. McKenna, M. Schmidt, P. Pollet and L. Gelbaum, et al., The oligomerization of glucose under plausible prebiotic conditions, Origins Life Evol. Biospheres, 2019, 49(4), 225–240 CrossRef CAS PubMed.
- M. Rodriguez-Garcia, A. J. Surman, G. J. Cooper, I. Suárez-Marina, Z. Hosni and M. P. Lee, et al., Formation of oligopeptides in high yield under simple programmable conditions, Nat. Commun., 2015, 6(1), 8385 CrossRef CAS PubMed.
- M. Frenkel-Pinter, J. W. Haynes, A. S. Petrov, B. T. Burcar, R. Krishnamurthy and N. V. Hud, et al., Selective incorporation of proteinaceous over nonproteinaceous cationic amino acids in model prebiotic oligomerization reactions, Proc. Natl. Acad. Sci. U. S. A., 2019, 116(33), 16338–16346 CrossRef CAS PubMed.
- T. Z. Jia, M. Caudan and I. Mamajanov, Origin of species before origin of life: the role of speciation in chemical evolution, Life, 2021, 11(2), 154 CrossRef PubMed.
- J. G. Forsythe, S. S. Yu, I. Mamajanov, M. A. Grover, R. Krishnamurthy and F. M. Fernández, et al., Ester-mediated amide bond formation driven by wet–dry cycles: a possible path to polypeptides on the prebiotic earth, Angew. Chem., Int. Ed., 2015, 54(34), 9871–9875 CrossRef CAS PubMed.
- M. C, M. Frenkel-Pinter, K. H. Smith, V. F. Rivera-Santana, A. B. Sargon and K. C. Jacobson, et al., Water-Based Dynamic Depsipeptide Chemistry: Building Block Recycling and Oligomer Distribution Control Using Hydration–Dehydration Cycles, JACS Au, 2022, 2(6), 1395 CrossRef CAS PubMed.
- K. Matange, V. Rajaei, P. Capera-Aragones, J. T. Costner, A. Robertson and J. S. Kim, et al., Evolution of complex chemical mixtures reveals combinatorial compression and population synchronicity, Nat. Chem., 2025, 17, 590–597 Search PubMed.
- J. G. Forsythe, A. S. Petrov, W. C. Millar, S. S. Yu, R. Krishnamurthy and M. A. Grover, et al., Surveying the sequence diversity of model prebiotic peptides by mass spectrometry, Proc. Natl. Acad. Sci. U. S. A., 2017, 114(37), E7652–E7659 CrossRef CAS PubMed.
- in A survey of emergent behavior and its impacts in agent-based systems, ed. Z. Li, C. H. Sim and M. Y. H. Low, 2006 4th IEEE international conference on industrial informatics, 2006, IEEE.
- N. Nogal, M. Sanz-Sánchez, S. Vela-Gallego, K. Ruiz-Mirazo and A. de la Escosura, The protometabolic nature of prebiotic chemistry, Chem. Soc. Rev., 2023, 52(21), 7359–7388 Search PubMed.
- S. Asche, G. J. T. Cooper, G. Keenan, C. Mathis and L. Cronin, A robotic prebiotic chemist probes long term reactions of complexifying mixtures, Nat. Commun., 2021, 12(1), 3547 Search PubMed.
- N. Guttenberg, N. Virgo, K. Chandru, C. Scharf and I. Mamajanov, Bulk measurements of messy chemistries are needed for a theory of the origins of life, Philos. Trans. R. Soc., A, 2017, 375(2109), 20160347 Search PubMed.
- L. E. Orgel, Prebiotic chemistry and the origin of the RNA world, Crit. Rev. Biochem. Mol. Biol., 2004, 39(2), 99–123 Search PubMed.
- P. Schuster, Taming combinatorial explosion, Proc. Natl. Acad. Sci. U. S. A., 2000, 97(14), 7678–7680 Search PubMed.
- P. T. Corbett, J. Leclaire, L. Vial, K. R. West, J.-L. Wietor and J. K. Sanders, et al., Dynamic combinatorial chemistry, Chem. Rev., 2006, 106(9), 3652–3711 Search PubMed.
- Y. Cao, J. Yang, D. Eichin, F. Zhao, D. Qi and L. Kahari, et al., Self-synthesizing nanorods from dynamic combinatorial libraries against drug resistant cancer, Angew. Chem., Int. Ed., 2021, 60(6), 3062–3070 Search PubMed.
- Z. Lei, H. Chen, S. Huang, L. J. Wayment, Q. Xu and W. Zhang, New advances in covalent network polymers via dynamic covalent chemistry, Chem. Rev., 2024, 124(12), 7829–7906 CrossRef CAS PubMed.
- A. Sood, P. K. Mandal, J. Ottelé, J. Wu, M. Eleveld and J. Hatai, et al., Simultaneous Formation of a Foldamer and a Self-Replicator by Out-of-Equilibrium Dynamic Covalent Chemistry, J. Am. Chem. Soc., 2024, 146(49), 33386–33394 Search PubMed.
- S.-S. Yu, R. Krishnamurthy, F. M. Fernández, N. V. Hud, F. J. Schork and M. A. Grover, Kinetics of prebiotic depsipeptide formation from the ester–amide exchange reaction, Phys. Chem. Chem. Phys., 2016, 18(41), 28441–28450 Search PubMed.
- P. G. Higgs, The effect of limited diffusion and wet–dry cycling on reversible polymerization reactions: implications for prebiotic synthesis of nucleic acids, Life, 2016, 6(2), 24 Search PubMed.
- G. Ashkenasy, T. M. Hermans, S. Otto and A. F. Taylor, Systems chemistry, Chem. Soc. Rev., 2017, 46(9), 2543–2554 RSC.
- K. Matange, V. Rajaei, G. Schuster, N. V. Hud, C. Menor-Salvan and P. Capera-Aragonès, et al., Origins of Life: Chemistry and Evolution, ChemRxiv, 2026 DOI:10.26434/chemrxiv-2023-1jrcq-v4.
- M. Frenkel-Pinter, M. Bouza, F. M. Fernández, L. J. Leman, L. D. Williams and N. V. Hud, et al., Thioesters provide a plausible prebiotic path to proto-peptides, Nat. Commun., 2022, 13(1), 2569 Search PubMed.
- R. Edri, S. Fisher, C. Menor-Salvan, L. D. Williams and M. Frenkel-Pinter, Assembly-Driven Protection from Hydrolysis as Key Selective Force during Chemical Evolution, FEBS Lett., 2023, 597, 2879–2896 Search PubMed.
- W. Hordijk, J. Hein and M. Steel, Autocatalytic sets and the origin of life, Entropy, 2010, 12(7), 1733–1742 Search PubMed.
- L. Vincent, M. Berg, M. Krismer, S. T. Saghafi, J. Cosby and T. Sankari, et al., Chemical ecosystem selection on mineral surfaces reveals long-term dynamics consistent with the spontaneous emergence of mutual catalysis, Life, 2019, 9(4), 80 Search PubMed.
- D. P. Glavin, J. P. Dworkin, C. M. O. D. Alexander, J. C. Aponte, A. A. Baczynski and J. J. Barnes, et al., Abundant ammonia and nitrogen-rich soluble organic matter in samples from asteroid (101955) Bennu, Nat. Astron., 2025, 1–12 Search PubMed.
- S. Benner, Borate Minerals Stabilize Ribose, Science, 2004, 303, 196 Search PubMed.
- S. Colón-Santos, G. J. Cooper and L. Cronin, Taming the combinatorial explosion of the formose reaction via recursion within mineral environments, ChemSystemsChem, 2019, 1(3), e1900014 CrossRef.
- K. Chandru, C. Potiszil and T. Z. Jia, Alternative Pathways in Astrobiology: Reviewing and Synthesizing Contingency and Non-Biomolecular Origins of Terrestrial and Extraterrestrial Life, Life, 2024, 14(9), 1069 CrossRef PubMed.
- R. M. Hazen, T. R. Filley and G. A. Goodfriend, Selective adsorption of L-and D-amino acids on calcite: Implications for biochemical homochirality, Proc. Natl. Acad. Sci. U. S. A., 2001, 98(10), 5487–5490 Search PubMed.
- A. J. Surman, M. Rodriguez-Garcia, Y. M. Abul-Haija, G. J. Cooper, P. S. Gromski and R. Turk-MacLeod, et al., Environmental control programs the emergence of distinct functional ensembles from unconstrained chemical reactions, Proc. Natl. Acad. Sci. U. S. A., 2019, 116(12), 5387–5392 Search PubMed.
- K. Vetsigian, C. Woese and N. Goldenfeld, Collective evolution and the genetic code, Proc. Natl. Acad. Sci. U. S. A., 2006, 103(28), 10696–10701 Search PubMed.
- S. A. Benner, H.-J. Kim and M. A. Carrigan, Asphalt, water, and the prebiotic synthesis of ribose, ribonucleosides, and RNA, Acc. Chem. Res., 2012, 45(12), 2025–2034 Search PubMed.
- M. L. Wong, C. E. Cleland, D. Arend Jr, S. Bartlett, H. J. Cleaves and H. Demarest, et al., On the roles of function and selection in evolving systems, Proc. Natl. Acad. Sci. U. S. A., 2023, 120(43), e2310223120 Search PubMed.
- R. M. Hazen and J. M. Ferry, Mineral evolution: Mineralogy in the fourth dimension, Elements, 2010, 6(1), 9–12 Search PubMed.
| This journal is © the Owner Societies 2026 |