Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Spin-crossover iron(II) complexes featuring boronic acid and boronic ester groups: synthesis and magnetic properties
Minghui
Zhang
,
Frank W.
Heinemann
and
Marat M.
Khusniyarov
 *
*
Department of Chemistry and Pharmacy, Friedrich-Alexander University Erlangen-Nürnberg (FAU), Egerlandstraße 1, 91058, Germany. E-mail: marat.khusniyarov@fau.de
First published on 6th March 2026
Abstract
A series of bis(pyrazol-1-yl)pyridine (BPP) iron(II) complexes bearing either boronic acid or boronic monoester groups, with the general formula [FeII(BPP)2](ClO4)2, have been synthesized and characterized in the solid state. The boronic acid or boronic monoester substituents are introduced at the 4-position of the pyridine ring. Despite their close structural similarity, the complexes exhibit markedly different magnetic properties. Complex 1a, which contains boronic acid groups, undergoes an incomplete, thermally induced spin-crossover in the solid state. Conversion of the boronic acid groups into monomethyl or monoethyl esters stabilizes the high-spin state in 1b and 1c, respectively. Further substitution with electron-donating tert-butyl groups in 2b enforces a pure low-spin state.
Introduction
Spin-crossover (SCO) complexes are known to be reversibly switched between two spin states in response to external stimuli such as light, heat or chemical environments.1–4 Owing to their switching ability, SCO complexes have triggered significant interest over the past decades to develop responsive sensors,5 and functional parts for molecular spintronics,6 molecular electronics,7 and memory devices.8 A common class of SCO complexes comprises bis(pyrazol-1-yl)pyridine (1-BPP) based iron(II) complexes, which were developed and extensively studied by M. Halcrow and M. Ruben.9–13 These complexes not only show SCO behaviour near room temperature,14 but also feature 1-BPP ligands that can be readily functionalized at pyridine or pyrazole moieties, thus enabling the fine-tuning of the magnetic properties of corresponding metal complexes.13,15–17Various modifications of 1-BPP-based iron(II) complexes have been synthesized and characterized; however, complexes featuring boronic acid groups have not yet been reported. We are particularly interested in boronic acid substituents, because they have demonstrated their potential in sensing applications, stemming from the vacant p orbitals on the boron centre.18–22 The low-lying empty p orbitals can accept electrons from nucleophiles, resulting in the electron density enrichment of the boron centre as well as the geometry transformation from a trigonal planar to a tetrahedral structure.22 Besides the Lewis acid character, boronic acids can also interact with small anions through two hydroxyl groups in the manner of Brønsted acids.23 Moreover, boronic acids exhibit high affinity to biological molecules such as saccharides and dopamine, even under physiological conditions.21,24 Upon reaction with these analytes, boronic acid esters are formed, leading, for instance, to a detectable reduction of fluorescence emission compared with the original boronic acids.25
The aforementioned characteristics have promoted us to explore the possibility of integrating boronic acid moieties into 1-BPP-based iron(II) complexes and further adjust the SCO behaviour of such complexes. The boronic acid group has been chosen to be attached at the pyridine C4 position because of synthetic simplicity as well as the electron-withdrawing nature of boronic acid, which can potentially lead to a temperature-induced SCO.26 In this study, we demonstrate synthetic routes to 1-BPP-based iron(II) complexes functionalized with boronic acid groups and boronic esters. The first series of such complexes have been synthesized and their magnetic properties have been investigated.
Experimental section
All starting materials and solvents were used without further purification unless mentioned. Pure anhydrous solvents were collected from a Glass Contour solid-state solvent purification system (Irvine, CA). 4-Amino-2,6-bis(pyrazol-1-yl)pyridine, 4-bromo-2,6-bis(pyrazol-1-yl)pyridine, and 4-tert-butylpyrazole were synthesized following the literature reported methods.27–29Instrumentation
Magnetic susceptibility data on solid samples were collected with a Quantum Design MPMS 3 magnetometer. DC susceptibility data were collected in the temperature range of 2–350 K for powder samples restrained with a polycarbonate gel capsule in an applied magnetic field of 1 T at a heating/cooling rate of 2 K min−1 and at 5 K intervals.The magnetic susceptibility data were corrected for diamagnetism using the estimation χm,diamag = 0.5Mw × 10−6 cm3 mol−1 where Mw is the molar mass of compound.30
57Fe Mossbauer spectra were recorded on a WissEl Mossbauer spectrometer (MRG-500) in constant-acceleration mode. 57Co/Rh was used as the radiation source. The temperature of the samples was controlled by an MBBC-HE0106 Mossbauer He/N2 cryostat within an accuracy of ±0.3 K. Isomer shifts were determined relative to the α-iron at 298 K. The program MX4w was used for the quantitative analysis of spectra.31
The NMR spectra were recorded with a JEOL ECX400 in rotating 5 mm o.d. tubes and processed using Delta V4.0 software provided by JEOL Ltd.
Powder X-ray diffraction patterns were obtained using a Bruker D8 diffractometer with a Cu Kα radiation source and a LyneEye XE-T detector. The IR spectra were recorded on a Shimadzu IRAffinity-1 system.
Crystallography
A suitable single crystal was embedded in protective perfluoropolyalkyl ether oil on a microscope slide, and a single specimen was selected and subsequently transferred to the cold nitrogen gas stream of the diffractometer. Intensity data were collected using MoKα radiation (λ = 0.71073 Å) on a Bruker Kappa PHOTON II IμS Duo diffractometer equipped with QUAZAR focusing Montel optics at a temperature of 100 K. The data were corrected for Lorentz and polarization effects, and semi-empirical absorption corrections were performed on the basis of multiple scans using SADABS.32 The structures were solved by direct methods (SHELX XT)33 and refined by full-matrix least-squares procedures on F2 using SHELXL 2019/3.34 All non-hydrogen atoms were refined with anisotropic displacement parameters. The positions of the oxygen-bound hydrogen atoms were determined from a difference Fourier map and their positional parameters were either refined (for 1a, 1b, and 1c) or allowed to ride on their oxygen carrier atom (2b). All other hydrogen atoms were placed in optimized positions. The isotropic displacement parameters of all H atoms were tied to those of the corresponding carrier atoms by a factor of either 1.2 or 1.5. Olex2 was used to prepare the material for publication.35Synthesis
Elemental analysis calcd. for C19H26N6·0.3C6H14O3: C 65.97, H 8.04, N 21.19; found: C 66.19, H 8.01, N 22.06.1H NMR (400 MHz, CDCl3, RT) δ: 8.27 (s, 2H, H2), 7.61 (s, 2H, H1), 7.05 (s, 2H, H3), 4.45 (S, 2H, H5), 1.30 (s, 18H, H-7).13C NMR (400 MHz, CDCl3, RT) δ: 156.84 (C1), 151.59 (C3), 140.02 (C6), 135.01 (C4), 123.06 (C5), 94.60 (C6), 31.72 (C7), 29.70 (C8) ppm.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) as the eluent (Rf = 0.3), yielding a white solid after solvent removal and dried in vacuo. Yield: 2.14 g, 60.2%.
3) as the eluent (Rf = 0.3), yielding a white solid after solvent removal and dried in vacuo. Yield: 2.14 g, 60.2%.
Elemental analysis: calcd. for C19H24N6Br: C 56.72, H: 6.01, N 17.41; found: C: 56.65, H: 6.10, N: 16.94.1H NMR (400 MHz, CDCl3, RT) δ: 8.24 (s, 2H, H2), 7.96 (s, 2H, H1), 7.66 (s, 2H, H3), 1.35 (s, 18H, H4).13C NMR (400 MHz, CDCl3, RT) δ: 150.77, 141.18, 136.45, 135.96, 122.96, 112.06, 31.62, 29.76 ppm.
Elemental analysis: calcd. for C19H26BN5O2: C 62.14, H 7.14, N 19.07; found: C: 62.66, H: 7.14, N: 18.87.1H NMR (400 MHz, DMSO-d6, RT) δ: 8.73 (s, 2H, H2), 8.60 (s, 2H, H4), 8.12 (s, 2H. H1), 7.81 (s, 2H, H3) ppm, 1.33 (s, 18H, H5). 13C NMR (400 MHz, DMSO-d6, RT) δ: 149.33, 140.38, 134.98, 123.30, 113.11, 112.17, 31.42, 29.35 ppm.
Elemental analysis calcd. for C22H20N10O12B2Cl2Fe·0.1C4H8O: C: 34.84, H: 2.72, N: 18.14; found: C: 34.78, H: 2.92, N: 17.96.
Elemental analysis calcd. for C26H28O12N10B2Cl2Fe: C: 38.04; H: 3.44; N: 17.06; found C: 38.18; H: 3.51; N: 16.92.
Results and discussion
Synthesis
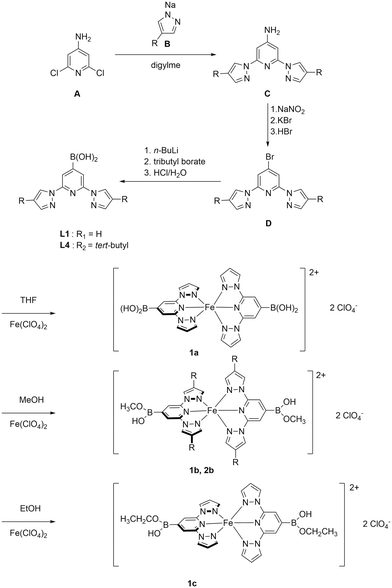
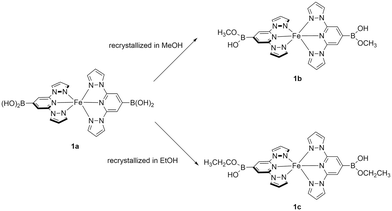
The novel boronic acid-featured BPP ligands were prepared following a multistep synthesis (Scheme 1). First, deprotonation of pyrazole with sodium hydride afforded the corresponding pyrazolate salt B, which was subsequently used to substitute chlorides on compound A, giving the chelating aniline C (yield 90%). A copper-free Sandmeyer reactions with C yielded a bromo-derivative D (yield 61%).28 Finally, the treatment of D with n-BuLi, the subsequent reaction with borate at low temperature, followed by acid-catalyzed hydrolysis produced the target tridentate ligand L1 as a white powder with a 72% yield. Ligand L4 was synthesized in a similar manner but starting with 4-tert-butylpyrazole, which was obtained by reacting tert-butylchloride and pyrazole in an autoclave at high pressure and high temperature for 2 days.29Complex 1a was prepared adopting literature-known methods.36 Solutions of L1 in THF and Fe(ClO4)2·6H2O were mixed and stirred at room temperature for 3 h. An orange solid was precipitated from the solution and was collected by filtration. Due to the poor solubility of 1a in common non-alcoholic solvents (note that 1a decomposes in DMSO or DMF), we have attempted to crystalize 1a in methanol and ethanol. Surprisingly, the obtained crystals 1b and 1c are modified complexes, where an esterification reaction took place giving methoxy and ethoxy monoesters, respectively (Scheme 2). The esterification of a single hydroxyl group of organic boronic acids in methanol is not uncommon.37–40 If one of the hydroxyl group binds with nearby atoms through hydrogen bonds, the remaining hydroxyl group can undergo methylation in methanol. In our case, one of the hydroxyl group binds with perchlorate (vide infra), facilitating the esterification of the other. However, esterification in ethanol was first observed to the best of our knowledge. Subsequently, we have explored the possibility of direct synthesis of 1b and 1c in methanol and ethanol, respectively (Scheme 1). Similar synthetic procedures to those used for 1a were performed, but in methanol and ethanol; 1b precipitated as a bright yellow powder from the reaction, whereas red crystalline materials of 1c were obtained via the slow diffusion of diethyl ether into the mother liquor.
In order to enhance the solubility of iron(II) complex 1a, a tert-butyl moiety was introduced at the 3-position of pyrazole, as in ligand L4 (Scheme 1). However, the solubility of the tert-butyl modified complex did not increase significantly, and we had to use alcoholic solutions here as well. The treatment of L4 with Fe(ClO4)2·6H2O in methanol leads to a red powder, which is proven to be 2b-a complex featuring monoesters at both ligands. Additionally, we have sought to react L1 with another commonly used iron precursor Fe(BF4)2. However, we are not able to purify this complex.
Crystal structures
Complex 1a crystallizes in the monoclinic C2/c space group with 4 molecules and 1 water molecule per unit cell (Fig. 1). At 100 K, complex 1a exhibits a distorted octahedral geometry, typically observed for this type of HS Fe(II) complex due to the Jahn–Teller effect.41 The dihedral angel (θ) between the two ligand planes is significantly distorted from the ideal 90° to 77.8°, and the N(pyridyl)–Fe–N(pyridyl) angle (ϕ) reaches 155.8. The Fe–N bond lengths are in the range 2.1370(11)–2.1716(11) Å, further denoting a HS Fe(II) centre.36 In the crystal structure, O–H⋯O hydrogen bonds are formed between boronic acid groups and nearby perchlorate anions. Each perchlorate anion bridges two boronic acid groups of neighbouring complex molecules within the lattice, thus leading to a polymeric chain structure along the crystallographic b axis. The introduction of an electron-withdrawing group at the pyridine C4 position is expected to favour the LS state according to Halcrow's work.26 Analogue complexes with electron-withdrawing substituents such as cyanide, carboxylic acid and aldehyde groups at this position stabilize LS Fe(II) state.12,42,43 However, our complex is a HS species. This discrepancy can be explained by the fact that the boronic acid group is less electron-withdrawing compared to the abovementioned substituents. Moreover, we assume that the highly distorted molecular structure of 1a is the result of strong intermolecular interactions in the cell, which ultimately leads to the stabilization of the HS state. | ||
| Fig. 1 Top: Molecular structure of 1a at 100 K; the H atoms are omitted for clarity. Bottom: Crystal packing of 1a showing a chain structure. | ||
Both mono-ester derivatives 1b and 1c crystallize in the C2/c space group with no solvent molecules in the unit cell (Fig. 2). The Fe(II) centres adopt HS configurations with typical long Fe–N bond lengths of 2.1269(16)–2.1873(18) Å and 2.1274(10)–2.1844(11) Å at 100 K, in 1b and 1c, respectively. Compared to the severely twisted molecular structure in 1a, the distortions in 1b and 1c are moderate, supported by the observed N(pyridyl)–Fe–N(pyridyl) angles (ϕ) of 162.9° and 174.7° approaching the undistorted 180° as well as dihedral angles (θ) of 86.4° and 87.9° approaching the ideal 90° in 1b and 1c, respectively. The reduced distortion is likely due to the substitution of one of the hydroxyl groups with a methoxyl or ethoxyl group, respectively, and the concomitant reduction of intermolecular interactions. Indeed, only one hydrogen bond is formed per perchlorate cation and the polymeric structures disappear in 1b and 1c.
 | ||
| Fig. 2 Molecular structures of 1b (top), 1c (middle) and 2b (bottom). The H atoms are omitted for clarity. | ||
Two polymorphs of 2b were observed from crystallization—red and yellow crystals, which is commonly observed for this class of compounds.44–48 Despite numerous attempts, the crystal structures of the yellow crystals could not be determined due to very poor diffraction of the crystallized samples. The red polymorph crystalized in the monoclinic P21/n space group and consists of two diethyl ether molecules per unit cell. Since the crystals degraded rapidly at room temperature due to solvent loss, we were able to determine the crystal structure only at low temperature. At 100 K, the Fe(II) centre reveals a low-spin (LS) Fe(II) state, as indicated by the shortened Fe–N bond lengths in the range of 1.899(21)–1.996(22) Å. The LS Fe(II) centre might be attributed to the electron-donating tert-butyl substituents at the pyridine 4-position strengthening the ligand field.26 Selected bond distances and angular parameters for the crystal structures of this work are summarized in Table 1. A detailed summary of the crystallographic data is listed in the SI.
| 1a | 1b | 1c | 2b | |
|---|---|---|---|---|
| Fe–N1 | 2.1716(11)–2.1716(12) | 2.1873(18)–2.1873(18) | 2.1844(11)–2.1844(11) | 1.981(21)–1.986(21) |
| Fe–N3 | 2.1370(11)–2.1716(11) | 2.1269(16)–2.1873(18) | 2.1274(10)–2.1844(11) | 1.899(21)–1.996(21) |
| Fe–N5 | 2.1635(11)–2.1636(11) | 2.1685(18)–2.1685(18) | 2.1943(11)–2.1943(11) | 1.976(22)–1.996(22) |
| ϕ | 155.8 | 162.9 | 174.7 | 177.7 |
| θ | 77.8 | 86.4 | 87.9 | 83.2 |
Magnetic properties
The magnetic properties, and consequently the spin states of the complexes, were investigated by SQUID magnetometry supported by 57Fe Mössbauer spectroscopy. Complex 1a undergoes partial, thermally induced SCO without hysteresis (Fig. 3). At room temperature (RT), 1a reveals a χT product of 1.70 cm3 mol−1 K (χ is the molar magnetic susceptibility), which is much lower than analogous pure HS Fe(II) complexes (3.3–3.8 cm3 mol−1 K).49 Upon heating to 350 K, χT increases to 2.35 cm3 mol−1 K, corresponding to a thermally driven conversion of LS Fe(II) to HS Fe(II). Upon cooling to low temperature, χT gradually decreases to 0.96 cm3 mol−1 K at around 100 K, reflecting a SCO from the HS to LS. A plateau at low temperature (30–100 K) suggests the presence of a residual HS fraction (around 25%). At 2 K, the χT product drops to 0.46 cm3 mol−1 K, which is ascribed to the zero-field splitting of the remaining HS fraction. When slowly heated to RT, the SCO behaviour of 1a follows the same behaviour as observed in the cooling mode, confirming reversible SCO without detectable hysteresis (SI). | ||
| Fig. 3 Variable-temperature χT products of 1a (top left), 1b (top right), 1c (bottom left) and 2b (bottom right) at an external magnetic field of 1 T. | ||
Note that the spin state of 1a determined from magnetic measurements (around 25% of the HS fraction at 100 K) is not in agreement with the crystallographic data, which indicates a pure HS state at 100 K. This is very likely due to the fact that magnetic measurements were performed on a powder sample, since we were not able to obtain enough crystalline materials for the magnetic measurements. So, the crystals and powder sample should represent two different phases. This has indeed been confirmed by measuring the powder XRD pattern of the powder sample, and comparing it with a simulated XRD pattern (see the SI).
Variable-temperature magnetic susceptibility measurements on complex 1b, 1c and 2b were performed from 300 to 2 K. The measurement reveals that 1b remains in the HS Fe(II) state at all temperatures, consistent with crystallographic data. At RT, the χT product of 3.78 cm3 mol−1 K is higher than the spin-only value expected for an HS Fe(II) centre (3.0 cm3 mol−1 K), but still coincides with values reported for similar HS Fe(II) complexes. Below 15 K, the χT product drops rapidly to 1.65 cm3 mol−1 K because of the zero-field splitting at the S = 2 state for the HS Fe(II) ion. The variable temperature data χT was fitted with the PHI program,50 which yields the g-value 2.24 and an axial zero-field splitting parameter D = 9.1 cm−1. Likewise, complex 1c reveals a pure HS Fe(II) state at all temperatures. At 300 K, the χT product of 1c is 3.38 cm3 mol−1 K, lower than 1b and remains constant until 60 K. Upon further cooling, the χT product decreases sharply to 1.45 cm3 mol−1 K at 2 K, again due to zero-field splitting. Fitting the variable temperature data yields g = 2.12 and a zero-field splitting parameter D = 7.0 cm−1.
Magnetic measurements were also performed for 2b over the 300–2 K range. Compound 2b displays a χT product of 0.33 cm3 mol−1 K at RT. The non-zero value is very likely due to minor paramagnetic impurities or a small fraction of the HS(II) species in the sample, which is very common for SCO Fe(II) species. Upon cooling, the χT product of 2b gradually approaches 0.0 cm3 mol−1 K at 4 K. The spin state of 2b determined from magnetic measurements is in full agreement with the crystallographic data.
Our magnetic measurements have revealed that complex 1a can potentially be used as a chemical sensor. At room temperature, 1a reveals a mixed HS/LS state. When the boronic acid group is converted to a monomethyl ester, transforming the complex into 1b, the material is switched to a pure HS state.
The boronic acid group can function as both a Lewis and a Brønsted acid, enabling interactions with small anions, diols, and various biological molecules.23 Therefore SCO complexes featuring boronic acid groups are attractive candidates for sensing applications in solution. Upon analyte binding, magnetic properties might change, which can be easily detected. In our case, complex 1a shows very poor solubility and limited stability toward non-alcoholic solvents and water, and thus of little use for such applications. However, a series of complexes in this report clearly demonstrate that modifications at the boronic acid moiety can induce SCO, highlighting the potential of this class of complexes in designing magnetic molecular sensors.
Mössbauer spectroscopy
Zero-field 57Fe Mössbauer spectroscopy was employed to confirm the oxidation and spin state of the iron centre in the complexes (Fig. 4). The Mössbauer spectrum of the SCO species 1a reveals the coexistence of two distinct Fe(II) species at 77 K. A major quadrupole doublet (75%) exhibits an isomer shift (δ) 0.38 mm s−1 and a quadrupole splitting (|ΔEQ|) of 0.69 mm s−1, in agreement with a LS Fe(II) state. A minor component (25%) exhibits an isomer shift of 1.18 mm s−1 and a quadrupole splitting of 3.03 mm s−1, which is characteristic of a HS Fe(II) state. These Mössbauer measurements are in line with magnetic measurements. | ||
| Fig. 4 Zero-field 57Fe Mössbauer spectra measured on 1a (top left), 1b (top right), 1c (bottom left) and 2b (bottom right) at 77 K. | ||
Complex 1b demonstrates (at 77 K) a single quadrupole doublet with an isomer shift of 1.14 mm s−1 and a quadrupole splitting of 2.82 mm s−1, which is in agreement with a pure HS Fe(II) state. The Mössbauer measurement results are consistent with the magnetic measurements. Complex 1c reveals one major HS Fe(II) component (90%) with an isomer shift of 1.12 mm s−1 and a quadrupole splitting of 3.42 mm s−1, along with a minor impurity (10%) with an isomer shift of 0.36 mm s−1 and a quadrupole splitting of 0.78 mm s−1. This impurity is identified as LS Fe(II) species, and despite many attempts to purify 1c, we were not able to remove it completely. A plausible reason might be that a monoethyl ester is less stable than a monomethyl ester, and it slowly decomposes to a boronic acid. Thus, we concluded that the impurity here might be 1a. The observed LS impurity can also explain the slightly lower χT product of 1c at RT compared with 1b.
The Mössbauer spectrum of 2b measured at 77 K reveals a single quadrupole doublet with an isomer shift of 0.37 mm s−1 and a quadrupole splitting of 0.72 mm s−1, identified as a LS Fe(II) species, which is in full agreement with the magnetic measurements. A summary of the parameters from Mössbauer measurements are listed in Table 2.
| 1a(LS) | 1a(HS) | 1b | 1c | 2b | |
|---|---|---|---|---|---|
| δ, mm s−1 | 0.38 | 1.18 | 1.14 | 1.12 | 0.37 |
| ∣ΔEQ∣, mm s−1 | 0.69 | 3.03 | 2.82 | 3.42 | 0.72 |
Conclusions
Four bis(pyrazol-1-yl)pyridine iron(II) complexes featuring boronic acid or boronic monoester groups have been synthesized and their magnetic properties have been investigated. Minor changes of the ligand substituents allowed us to control the magnetic properties by moving from a pure low-spin complex with tert-butyl substituents over spin-crossover species with boronic acid substituents to pure high-spin complexes featuring boronic monoester substituents. The change in magnetic properties induced by esterification of the boronic acid group highlights the potential of such complexes for chemosensing applications. The low solubility of the obtained complexes, however, prevents such studies. The work on more soluble analogues is currently in progress in our laboratory.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ce00062b.CCDC 2494923–2494926 contain the supplementary crystallographic data for this paper.51a–d
Acknowledgements
Dr. Andreas Scheurer is acknowledged for assistance with NMR measurements. Susanne Hoffmann is acknowledged for measuring powder XRDs. M. Z. is grateful to the China Scholarship Council for a stipend. M. M. K. thanks FAU Erlangen-Nürnberg and Prof. Karsten Meyer for providing access to spectroscopy facilities and continuous support. Deutsche Forschungsgemeinschaft is acknowledged for financial support (DFG Research Grants KH 279/6-1 and 279/8-1). This work is dedicated to Prof. Jörg Sundermeyer (Philipps-Universität Marburg) on the occasion of his retirement.Notes and references
- O. Kahn and C. Martinez, Science, 1998, 279, 44–48 CrossRef CAS.
- P. Gütlich and H. A. Goodwin, Spin crossover in transition metal compounds I-III, Topics in Current Chemistry, Springer-Verlag, Berlin Germany, 2004, vol. 233–235 Search PubMed.
- M. A. Halcrow, Spin-crossover materials: properties and applications, John Wiley & Sons, Ltd, 2013 Search PubMed.
- M. M. Khusniyarov, Chem. – Eur. J., 2016, 22, 15178–15191 CrossRef CAS.
- S. Venkataramani, U. Jana, M. Dommaschk, F. D. Sonnichsen, F. Tuczek and R. Herges, Science, 2011, 331, 445–448 CrossRef CAS PubMed.
- K. S. Kumar and M. Ruben, Angew. Chem., Int. Ed., 2021, 60, 7502–7521 CrossRef CAS.
- R. Torres-Cavanillas, M. Morant-Giner, G. Escorcia-Ariza, J. Dugay, J. Canet-Ferrer, S. Tatay, S. Cardona-Serra, M. Giménez-Marqués, M. Galbiati, A. Forment-Aliaga and E. Coronado, Nat. Chem., 2021, 13, 1101–1109 CrossRef CAS.
- G. Molnár, S. Rat, L. Salmon, W. Nicolazzi and A. Bousseksou, Adv. Mater., 2018, 30, 1703862 CrossRef.
- M. A. Halcrow, Coord. Chem. Rev., 2009, 253, 2493–2514 CrossRef CAS.
- M. A. Halcrow, New J. Chem., 2014, 38, 1868–1882 RSC.
- I. Šalitroš, O. Fuhr, R. Kruk, J. Pavlik, L. Pogány, B. Schäfer, M. Tatarko, R. Boča, W. Linert and M. Ruben, Eur. J. Inorg. Chem., 2013, 1049–1057 CrossRef.
- S. K. Kuppusamy, N. Del Giudice, B. Heinrich, L. Douce and M. Ruben, Dalton Trans., 2020, 49, 14258–14267 RSC.
- N. Suryadevara, A. Mizuno, L. Spieker, S. Salamon, S. Sleziona, A. Maas, E. Pollmann, B. Heinrich, M. Schleberger, H. Wende, S. K. Kuppusamy and M. Ruben, Chem. – Eur. J., 2022, 28, e202103853 CrossRef CAS PubMed.
- M. A. Halcrow, Coord. Chem. Rev., 2005, 249, 2880–2908 CrossRef CAS.
- L. J. K. Cook and M. A. Halcrow, Magnetochemistry, 2015, 1, 3–16 CrossRef.
- V. García-López, M. Palacios-Corella, V. Gironés-Pérez, C. Bartual-Murgui, J. A. Real, E. Pellegrin, J. Herrero-Martín, G. Aromí, M. Clemente-León and E. Coronado, Inorg. Chem., 2019, 58, 12199–12208 CrossRef PubMed.
- I. Galadzhun, R. Kulmaczewski, O. Cespedes, M. Yamada, N. Yoshinori, T. Konno and M. A. Halcrow, Inorg. Chem., 2018, 57, 13761–13771 CrossRef CAS.
- S. D. Bull, M. G. Davidson, J. M. Van den Elsen, J. S. Fossey, A. T. A. Jenkins, Y.-B. Jiang, Y. Kubo, F. Marken, K. Sakurai and J. Zhao, Acc. Chem. Res., 2013, 46, 312–326 CrossRef CAS.
- S. M. Butler, D. M. Beagan, W. Lewis, N. K. Szymczak and K. A. Jolliffe, Angew. Chem., Int. Ed., 2025, 64e202502582 Search PubMed.
- P. A. Gale, E. N. Howe and X. Wu, Chem, 2016, 1, 351–422 CAS.
- Z. Guo, I. Shin and J. Yoon, Chem. Commun., 2012, 48, 5956 RSC.
- R. Nishiyabu, Y. Kubo, T. D. James and J. S. Fossey, Chem. Commun., 2011, 47, 1106 RSC.
- M. A. Martínez-Aguirre and A. K. Yatsimirsky, J. Org. Chem., 2015, 80, 4985–4993 CrossRef PubMed.
- G. F. Whyte, R. Vilar and R. Woscholski, ChemBioChem, 2013, 6, 161–174 Search PubMed.
- L. R. Ortega-Valdovinos, J. Valdes-García, I. J. Bazany-Rodríguez, J. C. Lugo-González, A. Dorazco-González and A. K. Yatsimirsky, New J. Chem., 2021, 45, 15618–15628 RSC.
- L. J. K. Cook, R. Kulmaczewski, R. Mohammed, S. Dudley, S. A. Barrett, M. A. Little, R. J. Deeth and M. A. Halcrow, Angew. Chem., Int. Ed., 2016, 55, 4327–4331 CrossRef.
- E. Nuin, W. Bauer and A. Hirsch, Eur. J. Org. Chem., 2017, 790–798 CrossRef CAS.
- L. J. Kershaw Cook, H. J. Shepherd, T. P. Comyn, C. Baldé, O. Cespedes, G. Chastanet and M. A. Halcrow, Chem. – Eur. J., 2015, 21, 4805–4816 CrossRef CAS PubMed.
- A. L. Rheingold, L. M. Liable-Sands, J. A. Golan and S. Trofimenko, Eur. J. Inorg. Chem., 2003, 2767–2773 CrossRef CAS.
- O. Kahn, Molecular magnetism, VCH Publishers, Inc., USA, 1993 Search PubMed.
- E. Bill, MFIT, version 2.0, MPI for Chemical Energy Conversion, Mühlheim/Ruhr, Germany, 2011 Search PubMed.
- SADABS, Bruker AXS area detector scaling and absorption correction, Bruker AXS, Inc, Madison WI., USA, 2014 Search PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2008, 64, 112–122 CrossRef CAS PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8 Search PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- L. J. Cook, R. Kulmaczewski, O. Cespedes and M. A. Halcrow, Chem. – Eur. J., 2016, 22, 1789–1799 CrossRef CAS.
- C. J. Davis, P. T. Lewis, D. R. Billodeaux, F. R. Fronczek, J. O. Escobedo and R. M. Strongin, Org. Lett., 2001, 3, 2443–2445 CrossRef CAS.
- M. Schwalbe, P. Wrzolek, G. Lal and B. Braun, Eur. J. Inorg. Chem., 2014, 4209–4217 CrossRef CAS.
- J. Axthelm, H. Görls, U. S. Schubert and A. Schiller, J. Am. Chem. Soc., 2015, 137, 15402–15405 CrossRef CAS PubMed.
- W. Zhai, B. M. Chapin, A. Yoshizawa, H.-C. Wang, S. A. Hodge, T. D. James, E. V. Anslyn and J. S. Fossey, Org. Chem. Front., 2016, 3, 918–928 RSC.
- L. J. K. Cook, R. Mohammed, G. Sherborne, T. D. Roberts, S. Alvarez and M. A. Halcrow, Coord. Chem. Rev., 2015, 289, 2–12 CrossRef.
- A. Abhervé, M. Clemente-León, E. Coronado, C. J. Gómez-García and M. López-Jordà, Dalton Trans., 2014, 43, 9406–9409 RSC.
- Q. Yang, Y.-S. Meng, T. Liu and J. Tang, Dalton Trans., 2022, 51, 602–607 RSC.
- I. Šalitroš, J. Pavlik, R. Boča, O. Fuhr, C. Rajadurai and M. Ruben, CrystEngComm, 2010, 12, 2361 RSC.
- I. Šalitroš, O. Fuhr, A. Eichhöfer, R. Kruk, J. Pavlik, L. Dlháň, R. Boča and M. Ruben, Dalton Trans., 2012, 41, 5163 RSC.
- I. Šalitroš, L. Pogány, M. Ruben, R. Boča and W. Linert, Dalton Trans., 2014, 43, 16584–16587 RSC.
- I. Šalitroš, O. Fuhr and M. Ruben, Materials, 2016, 9, 585 CrossRef PubMed.
- S. K. Kuppusamy, A. Mizuno, L. Kämmerer, S. Salamon, B. Heinrich, C. Bailly, I. Salitros, H. Wende and M. Ruben, Dalton Trans., 2024, 53, 10851–10865 RSC.
- J. Elhaïk, D. J. Evans, C. A. Kilner and M. A. Halcrow, Dalton Trans., 2005, 1693–1700 RSC.
- N. F. Chilton, R. P. Anderson, L. D. Turner, A. Soncini and K. S. Murray, J. Comput. Chem., 2013, 34, 1164–1175 CrossRef CAS PubMed.
- (a) CCDC 2494923: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pr5d1; (b) CCDC 2494924: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pr5f2; (c) CCDC 2494925: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pr5g3; (d) CCDC 2494926: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pr5h4.
| This journal is © The Royal Society of Chemistry 2026 |