 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Multiscale simulations of gas hydrates: from molecular mechanisms to mesoscale growth and macroscale flow and production predictions
Miguel
Pineda
 *a,
Anh
Phan
*b,
Michail
Stamatakis
*a,
Anh
Phan
*b,
Michail
Stamatakis
 a and
Alberto
Striolo
a and
Alberto
Striolo
 cd
cd
aDepartment of Chemistry, Inorganic Chemistry Laboratory, University of Oxford, South Parks Road, Oxford, OX1 3QR, UK. E-mail: miguel.pineda@chem.ox.ac.uk
bSchool of Chemistry and Chemical Engineering, Faculty of Engineering and Physical Sciences, University of Surrey, Guildford, Surrey GU2 7XH, UK. E-mail: a.phan@surrey.ac.uk
cSchool of Sustainable Chemical, Biological and Materials Engineering, The University of Oklahoma, Norman, OK 73019, USA
dMaterials Science and Engineering Program, The University of Oklahoma, Norman, OK 73019, USA
First published on 24th March 2026
Abstract
Gas hydrates—crystalline hydrogen-bonded water cages encapsulating gas molecules—are of major scientific and industrial interest for energy systems, safety and flow assurance, as well as for several emerging technologies. Predicting and controlling their formation, dissociation, and growth remains challenging due to coupled physical and chemical processes across multiple scales. This highlight article reviews recent advances in multiscale simulations of gas hydrates, from microscale mechanistic methods to mesoscale growth models and macroscale predictive simulators, and discusses how these approaches improve our ability to predict hydrate behaviour in natural and industrial settings. Current challenges are also discussed, together with suggestions for future research.
1. Introduction
Gas (clathrate) hydrates are non-stoichiometric crystalline structures in which water molecules form hydrogen-bonds to yield cages encapsulating gas molecules.1–6 These structures form under low-temperature and high-pressure conditions typically found in permafrost regions and in deep marine sediments along continental margins.7,8Due to their wide distribution and structural diversity,9 gas hydrates are considered a potential unconventional energy resource10,11 and play key roles in marine geohazards12 and climate processes.13 In industrial settings, hydrate formation can obstruct pipelines and equipment, posing significant flow-assurance challenges.14,15 Simultaneously, gas hydrates offer opportunities for gas storage and transport, including carbon dioxide (CO2) sequestration16,17 and hydrate-based hydrogen storage,18 and applications in wastewater treatment, seawater desalination,19,20 separations, and cold thermal energy storage and refrigeration technologies.21 Nevertheless, the practical deployment of applications based on hydrates remains limited because of an interplay of fundamental and technical challenges.
Toward overcoming these challenges, computational studies of gas hydrates provide an essential complement to experimental investigations because laboratory measurements are often limited by the pressure–temperature conditions required to form and stabilise hydrates, the difficulty of observing processes in buried marine sediments in the presence of various compounds, and the challenges of capturing nucleation and growth phenomena. The phenomena of interest evolve on diverse length- and time-scales, and various approaches focusing on specific aspects have been adopted: atomistic/molecular (micro)-scale models that describe cage formation and guest–host interactions,22,23 mesoscale approaches that explore hydrate growth and agglomeration,24,25 as well as macroscale models that address reservoir and flow assurance.14,26–28 Gas hydrate phenomena are inherently multiscale, and a holistic integration and interpretation of these three levels of description are necessary to fully understand these systems.25,29–32
This article highlights recent advances in modelling hydrate nucleation, growth, transport, and reservoir behaviour, and stresses the need for cross-scale integration. Accordingly, the reviewed efforts toward achieving such integration are based on using molecular insights to inform mesoscale models and applying mesoscale results to guide reservoir simulations. Overall, these efforts strive toward coherent and predictive hydrate modelling that advances fundamental understanding and guides technology development.
While progress has been made across quantum, molecular, and continuum modelling in recent years, these advances remain partially fragmented across scales. Bridging these scales through multiscale computational tools, achieving predictive accuracy under realistic conditions, and obtaining comprehensive experimental validation is necessary to leverage fundamental understanding to overcome practical technological challenges.
The rest of the article is structured as follows: section 2 reviews microscale computational approaches; in Sec. 3, mesoscale modelling strategies are discussed; and in Sec. 4, macroscale simulators for reservoir and flow assurance applications are examined. Finally, in Sec. 5, conclusions and an outlook are presented.
2. Microscale molecular simulations
Microscale molecular simulations resolve guest–host interactions and water hydrogen-bond networks at the atomic level, providing fundamental insight into hydrate structure, cage occupancy, nucleation, and early growth. Guest–host van der Waals (vdW) interactions36–40 stabilise the three main cage-based structures (see Fig. 1)—sI, sII, and sH—whose behaviour depends on cage geometry, connectivity, and guest occupancy.33 Density functional theory (DFT), grand canonical Monte Carlo (GCMC), and molecular dynamics (MD) simulations provide complementary perspectives on these processes, spanning electronic structure, thermodynamic stability, equilibrium cage occupancy, and non-equilibrium nucleation pathways. | ||
| Fig. 1 Three main hydrate structures—sI (A), sII (B), and sH (C) with different host water cages. Adapted from Hassanpouryouzband et al.2 Copyright © 2020 Royal Society of Chemistry. | ||
2.1. Density functional theory applications
DFT studies have advanced our understanding of the molecular and structural stability of gas hydrates. As a quantum-mechanical electronic-structure method, DFT determines the ground-state electron density and total energy of hydrate systems within the framework pioneered by Hohenberg and Kohn,41 enabling quantitative evaluation of hydrogen bonding, dispersion-driven guest–host interactions, cage-occupancy effects, and pressure-dependent lattice stability.By fitting equations of state to DFT energy–volume relations computed using the revised Perdew–Burke–Ernzerhof (revPBE) generalised-gradient approximation (GGA) exchange–correlation (XC) functional, Vlasic et al.42 showed that the bulk modulus of sII hydrates scales linearly with hydrogen-bond density and that larger guest molecules increase lattice volumes through van der Waals (vdW)-driven cage expansion. Daghash et al.43 performed extensive DFT calculations on sH hydrates using the revPBE GGA functional, complemented by vdW-DRSLL to assess dispersion effects, linking vibrational behaviour to elastic properties such as Young's modulus. The results highlighted an approximately linear dependence of elastic constants on pressure. Mathews et al.44 found that SCAN-based DFT with rVV10 dispersion corrections predicts the thermodynamic properties of sI hydrates more accurately at low temperatures than classical MD. Jendi et al.45 quantified the compressive and tensile strengths of sI methane (CH4) hydrates using the revPBE functional without an explicit vdW correction, while Zhu et al.46,47 used the revPBE functional with DFT-D2 dispersion corrections to confirm pressure-dependent elastic constants and the validity of the law of mixtures for sI CH4 hydrates. These studies commonly assume uniform force distribution within stable lattices, though hydrate mechanics depend on interactions among the hydrogen-bond network and the connectivity between large and small cages.
Zhu et al.33 combined DFT calculations (revPBE GGA with DFT-D2 vdW corrections) with a polyhedral composite framework to assess how cage occupancy governs pressure-induced failure of the hydrogen-bonded water lattice. Fig. 2A(left panel) shows lattice-volume changes from 0–7.6 GPa for four models: SELE (no cage occupancy), SOLE (small cages filled), SELO (large cages filled) and SOLO (all cages filled). At 0–4.5 GPa, all systems show similar, gradual volume reductions (<5%), indicating dominance of hydrogen-bond forces over guest-host interactions. The unit lattice volumes agree well with previous DFT work (e.g., SOLO with ∼1620 Å3vs. 1685 Å3 in Jendi et al.45). The SELE system fails at 4.6 GPa, marking the minimum compressive stability of the hydrogen-bonded lattice. Above 4.5 GPa, occupancy effects become pronounced. SOLO and SELO undergo major collapses at 7.3 and 6.4 GPa, respectively. SOLO is mechanically stronger than SELO, consistent with its fully occupied small cages. SELE and SOLE collapse earlier (4.6 and 4.9 GPa), indicating loss of structural integrity. The SOLO compressive limit (7.3 GPa) aligns with prior reports46,47 (∼7.5 GPa), supporting the accuracy of these results.33 By analogy with the law of mixtures—replacing volumetric fractions with small- and large-cage occupancies, Zhu et al. expressed compressive stability S (GPa) as: S (GPa) = 4.6 + 0.3Qs + 1.7Ql + 0.7QsQl, where Qs and Ql denote the fractional occupancies of small and large cages, respectively. The models predict stability limits to be in the pressure range 4.6 to 7.3 GPa, with SOLO being the most stable and SELE the least (see Fig. 2A, right panel).33
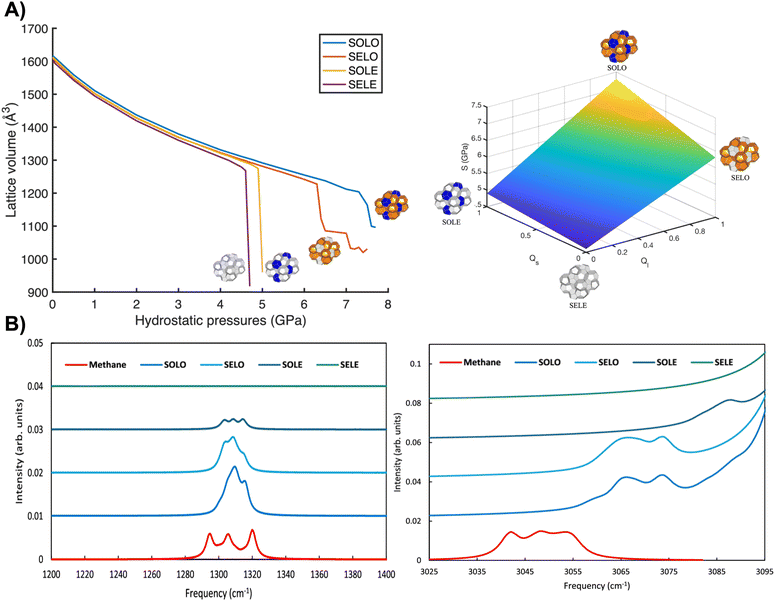 | ||
| Fig. 2 A) Lattice volume versus hydrostatic pressure for four occupancy configurations: blue, orange, yellow, and purple lines correspond to SOLO, SELO, SOLE, and SELE hydrates (left). The 3D surface shows compressive stability limits versus phase occupancy; Ql and Qs denote large- and small-cage occupancies (right). Adapted from Zhu et al.33 Copyright © 2023 Springer Nature. B) Methane C–H bending (left) and stretching (right) frequencies for different cage occupancies. Adapted from Schmidt et al.34 Copyright © 2025 American Chemical Society. | ||
Among experiments useful to validate the computational predictions, infrared (IR) spectroscopy has been widely used to probe CH4 hydrates properties. For example, the hydrogen-bond stretching frequency provides estimates of Young's modulus, while the number and frequencies of C–H stretching peaks reveal whether CH4 occupies small cages, large cages, or both.34,48,49 Computational work using VASP, SIESTA (Vibra), and CASTEP has reproduced hydrate vibrational spectra using either harmonic DFT-based calculations or classical MD simulations.50–53 Schmidt et al.34 used DFT with the rev-PBE-XC functional and DFT-D2 dispersion corrections to examine IR spectra of sI CH4 hydrates at different occupancies. CH4 exhibits two IR-active modes: C–H bending and asymmetric C–H stretching. Simulations reproduced these modes for both isolated CH4 and CH4 confined in hydrate cages. For isolated CH4, the modes appear at 1306 (bending) and 3048 (stretching) cm−1 (see Fig. 2B), matching experiments at 1306 and 3019 cm−1.34 The bending mode occurs at lower frequency than the bond stretching because angular deformation requires less energy than bond stretching. When encaged, both modes shift to higher frequencies, 1310 cm−1 for bending and 3074 and 3089 cm−1 for stretching, reflecting dispersion interactions with the surrounding water lattice. Splitting of each spectral band into several small peaks (10–15 cm−1 apart) likely arises from slight differences in degenerate vibrational modes. Distinct C–H stretching peaks correspond to CH4 in large (∼3074 cm−1) and small (∼3089 cm−1) cages, aligning with Raman results from Hansen and Berg.48 The higher frequency and lower intensity of the small-cage peak indicate stronger confinement and fewer molecules per cage. Changing occupancy primarily alters peak intensities rather than positions, making the relative intensity of the C–H bending mode a useful indicator of overall occupancy.34
Cabrera-Ramírez et al.35 investigated stability and occupancy patterns of sI CO2 hydrates using dispersion-corrected DFT-D methods, aimed at clarifying the transition from clathrate-like clusters to periodic crystals. They found that CO2 binds more strongly within the large T (51262) cage than the small D (512) cage (see Fig. 3A). This finding aligns with earlier studies based on both finite clathrate-like cluster models and periodic boundary simulations.54–59 Estimated binding energies were −6.740 kcal mol−1 in T cages and −4.835 kcal mol−1 in D cages, giving an ∼1.9 kcal mol−1 preference for large cages. Full occupancy of all eight cages stabilises the hydrate by ∼−51 kcal mol−1 relative to the empty lattice. Large-cage preference stems from the linear geometry of CO2: D cages impose more pronounced confinement and therefore stronger repulsive forces. Structurally, the D cage has twelve pentagons, while the T cage includes two hexagons and twelve pentagons, offering greater free volume. Single-crystal X-ray diffraction experiments at 173 K support these findings, showing full occupancy of T cages but partial filling of D cages.60
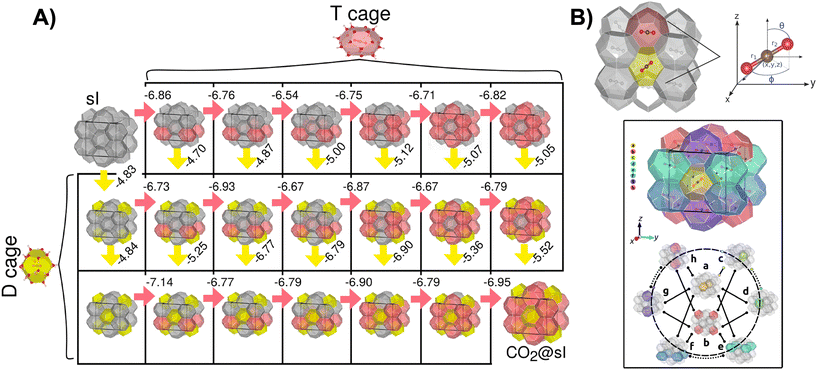 | ||
| Fig. 3 A) Progressive CO2 occupancy in sI hydrate, with red (T) and yellow (D) cavities marking cages filled at each step. Binding energies (in kcal mol−1 per added CO2) give the stabilization gained (negative) relative to the partially occupied structure and a free CO2. B) Top: sI CO2 unit cell with D (yellow) and T (pink) cages and the DFT coordinate sites. Bottom: fully occupied sI CO2 unit cell from periodic DFT showing DT (D–T), TT(6) (hexagon-sharing T–T), and TT(5) (pentagon-sharing T–T) connectivities. Cages a and b (D) connect to c–h (T) as DT pairs (solid); c–d, e–f, and g–h form TT(6) (dotted); other T–T pairs form TT(5) (dashed). Panels A) and B) adapted from Cabrera-Ramírez et al.35 Copyright © 2021 John Wiley and Sons. | ||
Recent experimental and theoretical studies54,55,57,59–62 show that CO2 molecules in sI clathrate cages adopt orientations allowing rotation. Cabrera-Ramírez et al.35 also analysed the relaxed sI CO2 structure and determined spatial orientations of guest CO2 molecules (see Fig. 3B, top panel). They identified eight unique CO2 positions: two in D cages and six in T cages (see Fig. 3B, bottom panel). In D cages, CO2 aligns near the cage centre, consistent with experimental observations from single-crystal X-ray diffraction and neutron scattering, as well as with theoretical data by others.54,55,57,59–62 In T cages, CO2 orientations display broader angular variation, with θ ranging between 75° to 100° and wide ϕ distributions, with small positional shifts.60,61 Cage connectivity governs intermolecular CO2–CO2 separations in the sI hydrate: distances are shortest in TT(6) (5.77–6.14 Å), intermediate in DT (6.39–6.82 Å), and longest in TT(5) (6.95–7.44 Å) (see Fig. 3B). Short TT(6) separations suggest stronger guest–guest interactions mediated by the host lattice, whereas TT(5) distances indicate predominantly dispersion-driven CO2–CO2 coupling. These results emphasise the combined role of cage geometry, guest orientation, and cage adjacency in stabilizing CO2-filled hydrates.
Valdés et al.72 analysed translational-rotational states of CO2 within sI hydrates using the multiconfiguration time-dependent Hartree method. They compared semiempirical and ab initio CO2–water potentials against DFT-D results and evaluated many-body influences on guest–host interactions.72 Distinct excitation patterns were found to depend on the interaction model, reflecting subtle changes in CO2 local structure within D and T cages. These results agree with neutron diffraction and 13C NMR data60,61,73 and predict previously unmeasured spectroscopic transitions, providing a benchmark for future studies.
The formation and stability of hydrates depend on fundamental guest–host and host–host interactions. Progress in this field requires a detailed understanding of these interactions and how they evolve under varying system conditions. From a theoretical standpoint, accurately capturing guest–host interactions is computationally demanding, as it necessitates a consistent treatment of both the covalent and extended hydrogen-bond networks of the water framework and the noncovalent interactions between guest species and the host lattice. Although DFT can effectively describe guest–host interactions and predict equilibrium structures and pressure responses with reasonable computational efficiency,33–35,55,72 the accuracy of both conventional and dispersion-corrected functionals in simultaneously representing hydrogen bonding and dispersion forces in gas hydrates must be carefully assessed. Consequently, systematic evaluations of key interaction effects, including cooperative guest–host interactions and guest–guest or cage–cage correlations, are necessary. Such understanding could help us better understand the thermal effects connected to the formation and dissolution of hydrates, one of the main challenges for the technological deployment of these systems. Capturing the full thermodynamic behaviour of clathrate hydrates requires explicit treatment of temperature- and pressure-dependent effects through molecular-scale simulations capable of resolving cavity occupancy and cooperative interactions.
2.2. Grand canonical Monte Carlo simulations
GCMC simulations are widely used to investigate guest adsorption and cage occupancy in gas hydrates under fixed temperature, volume, and chemical potential conditions.74–78 In this framework, particle insertion, deletion, translation, and rotation moves are performed to sample equilibrium configurations of guest molecules within a pre-defined (often rigid) hydrate lattice. GCMC calculations enable the efficient study of adsorption thermodynamics and cage occupancy distributions on much shorter timescales than MD, where evaluating occupancy requires guest diffusion through hydrate cages, a notoriously slow process. When combined with van der Waals–Platteeuw (vdWP) theory,6,79 GCMC enables the evaluation of hydrate phase equilibria across diverse compositions.To provide a fundamental underpinning for developing new processes that, for example, replace CH4 in natural hydrates with CO2, it is essential to understand hydrate stability as a function of guest type and occupancy.62,80–86 Phase-equilibria and composition-evolution studies,80,87,88 supported by spectroscopic evidence,62,82,84,86,89 show that CO2 preferentially replaces CH4 in the large sI cages due to size compatibility. However, the behaviour of mixed CO2–CH4 hydrates remains debated.
Qiu et al.63 combined GCMC with MD simulations to investigate the adsorption isotherms of pure and mixed CH4–CO2 hydrates using different guest and host models. Their CH4 cage occupancy results for sI and sII hydrates were compared with earlier stand-alone Monte Carlo (MC) simulation conducted at 270 K64–67 and with experiments at 268–271 K68–71 (see Fig. 4A). Simulations employing the TIP4P/Ice water model produced higher occupancies than those using TIP4P/Ew, owing to stronger host–guest interactions—overestimating small-cage occupancy but accurately capturing large-cage occupancy in the sI hydrate.63 Flexible hydrate models yield lower occupancies than rigid ones because cage distortion hindered guest entry. With flexible hydrate and united atoms (UA) CH4 models, Qiu et al.63 closely replicated the experimentally observed small-cage occupancies (Fig. 4A, right panel), though large-cage (Fig. 4A, left panel) values were slightly underestimated. Similar trends were observed with UA0.88 and all-atom (AA) CH4 models, which matched small-cage data from Klapproth et al.70 but underestimated total occupancies. These results highlight the strong dependence of hydrate occupancy on lattice and water models, with greater accuracy expected from rigid lattices and TIP4P/Ice model. The total occupancy obtained using TIP4PEw and DACNIS-UA90 models aligned more closely with experimental data than earlier TraPPE-UA predictions. Overall, both experimental and GCMC simulation results63–71 confirm that CH4 preferentially occupies large cages, consistent with DFT predictions linking cage occupancy to the stability of sI CH4 hydrates.33
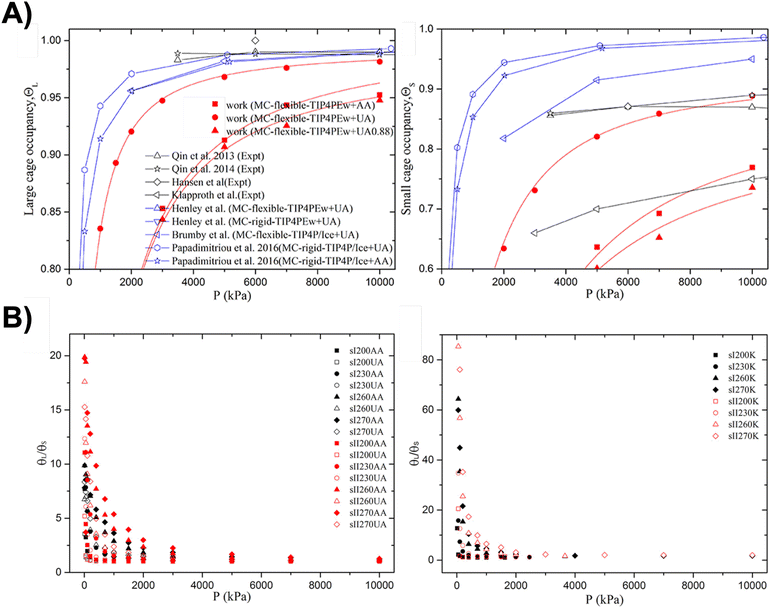 | ||
| Fig. 4 A) Pressure dependence of CH4 occupancy in large (left) and small (right) sI cages at ∼270 K. Red, blue, and black symbols correspond to Qiu et al.,63 previous simulations,64–67 and experiments.68–71 B) Temperature- and pressure-dependent occupancy ratios θL/θS for CH4 (left) and CO2 (right) in sI and sII hydrates; θL and θS denote fractions of large and small cages filled. Panels A) and B) adapted from Qiu et al.63 Copyright © 2018 American Chemical Society. | ||
Qiu et al.63 reported that in sI and sII hydrates, the occupancy ratio of CO2 and CH4 between large and small cages (θL/θS) decreases under higher pressures but increases with temperature (see Fig. 4B), consistent with both experiments91 and earlier simulations.65,92 The θL/θS ratio is higher in sII than in sI due to the greater size contrast between their cages. At sufficiently high pressures, θL/θS approaches 1.0 from above, indicating that small cages fill after large ones. For sI CH4 hydrate at 270 K and 6 MPa, calculated θL/θS values (1.40 and 1.16 for the AA and UA model, respectively) (see Fig. 4B, left panel) align well with experimental data (∼1.26 at 270 K84 and 1.13–1.15 at 268 K).68–71 For CO2, θL/θS is significantly larger at low pressures—about 3.13 and 5.13 for sI and sII at 270 K and 1.5 MPa (see Fig. 4B, right panel)—implying that large cages are preferentially occupied. These results agree reasonably with experimental (8.33 at 1.2 MPa and 273 K)93 and vdW–Platteeuw thermodynamic values (1.91 and 1.35),94 confirming that the GCMC + MD simulations reliably capture CO2 and CH4 adsorption behaviour in hydrates, while additionally resolving cage-specific occupancy, local heterogeneity, and molecular-scale adsorption mechanisms that are not directly accessible experimentally.
Unlike the classical vdWP theory, GCMC does not require assumptions regarding the number of guest molecules per cavity. In GCMC simulations, cavity occupancies emerge naturally from the calculations. In addition, GCMC explicitly accounts for guest–guest interactions, alongside guest–host interactions with the water lattice. Despite these advantages, GCMC cannot assess the thermodynamic stability of hydrate crystals, which must be assumed a priori. Moreover, predicted phase equilibria are highly sensitive to the choice of force fields, necessitating careful validation against experimental data. Finally, as a MC technique, GCMC provides not insight into hydrate nucleation and growth kinetics.
2.3. Molecular studies of hydrates nucleation and growth
By tracking the position over time of individual atoms and molecules, MD simulations reveal their assembly into hydrate structures, providing a powerful framework for understanding inhibitor effects and hydrate growth across environments. In classical MD, forces are described by empirical force fields, enabling nanosecond to microsecond simulations, whereas ab initio MD (AIMD) derives forces from DFT calculations, offering higher accuracy at substantially greater computational cost. Coarse-grained MD (CGMD) reduces molecular detail to access longer time and length scales. MD simulations provide insights into nucleation mechanisms,95,98–100 growth dynamics,97,101,102 and the influence of additives22,103–105 and confinement106,107—often beyond experimental reach.Several conceptual scenarios had been proposed to explain the nucleation mechanism. In 1991, Sloan and Fleyfel introduced the labile cluster hypothesis (LCH), which suggested that water molecules form transient, cage-like structures around guest molecules that subsequently merge to form hydrate crystals.108 Later classical MD and MC simulations demonstrated that isolated clathrate cages are rare, extremely short-lived, and energetically unstable in solution.109,110 Further studies showed that guest-filled cages are more likely to disintegrate than aggregate, and that fully enclosed polyhedral cages form only at very high guest concentrations with very low probability.109,110 Although cage stability can increase in the presence of multiple guest molecules, these findings indicate that hydrate nucleation does not proceed via isolated cages. Consequently, the local structuring hypothesis was proposed, suggesting that hydrate nucleation is governed by local concentration fluctuations that organise guest molecules into hydrate-like configurations. Achieving these configurations is the rate-limiting step in crystal formation. A historical overview of hydrate nucleation studies is provided in the work of Zhang et al.111
A pioneering MD simulation study of hydrate nucleation was conducted by Walsh et al.,95 who found that five water molecules and two CH4 molecules jointly organise into a stable pentagonal water ring that persists during transient cavity formation. This CH4–water cooperation facilitates further ordering and CH4 adsorption, in line with earlier works showing that CH4 stabilises hydrate faces.112,113 Under the super-saturated conditions considered in these simulations, a small (512) cage emerged at 1.225 μs (Fig. 5A), surrounded by a CH4 layer. Steric constraints prevent extended growth of face-sharing 512 cages until the central cage opens after ∼30 ns, enabling rearrangement and rapid expansion. Over ∼240 ns, the 512 cage evolved into a 51263 cage—a transitional motif connecting sI and sII hydrates. Similar rearrangements appear elsewhere, producing mixed sI–sII networks. Walsh et al.95 showed that 512 cages dominate early nucleation, followed by 51262 (sI) cages after ∼100 ns, consistent with the thermodynamic stability of sI hydrates. Larger 51264 (sII) cages remain rare,1 forming transiently due to adjacent small-cage patterns. The 51263 cages act as structural links, promoting sI formation from sII-like clusters (see Fig. 5A). The final hydrate contains mixed sI/sII motifs, consistent with experimental114–116 and simulation results117,118 showing that early nucleation generates heterogeneous structures stabilised by shared 512 cages. The apparent discrepancy with DFT studies highlighting large-cage occupancy as the primary stability factor33,35 likely reflects the non-equilibrium nature of MD simulations, which capture early-stage nucleation under highly supersaturated conditions.95
 | ||
| Fig. 5 A) Simulation snapshots and cage-type evolution during induction, nucleation, and growth; inset shows early nucleation. Violet, green, black, and yellow denote 512, 51262, 51263, 51264 cages. Adapted from Walsh et al.95 Copyright © 2009 AAAS. B) Interaction energy (Eint) of adsorbed water versus water count in small (blue) and large (red) cages. Adapted from Liu et al.96 Copyright © 2016 Elsevier. C) Temporal evolution of 512 (blue) and 51262 (red) cages in sI CH4 hydrate without additives (left) and with two SDS molecules (middle), and free-energy profiles (right) for CH4 transfer from hydrocarbon to aqueous phase. Systems: SDS at low (green) and high (red) coverages and without SDS (blue). Adapted from Phan et al.97 Copyright © 2023 Elsevier. | ||
Liu et al.96 used AIMD to examine the assembly of hydrogen-bonded cages in sI CH4 hydrates. They found that small cages originate from pentagonal water rings hosting one CH4 molecule, whereas large-cage precursors form from hexagonal water rings—consistent with results reported by Walsh et al.95 However, the formation mechanisms differ: small cages grow through ring expansion (ternary → quaternary → pentagonal), while large cages assemble through a layer-separated process, as surrounding water molecules are more strongly attracted to the large-cage precursor and experience less CH4 repulsion. These findings support a two-step formation process: (1) local water ordering forms precursors, and (2) CH4-driven restructuring produces hydrate cages. Small 512 cages preferentially form in early nucleation due to stronger interactions between adsorbed water and the cage structure (Fig. 5B). Large 51262 cages later dominate sI hydrates because of their higher energetic favourability.96
AIMD simulations are typically limited to short trajectories. It is therefore encouraging that, using non-equilibrium MD simulations, Phan et al.97 found that the formation rate of 51262 cages (∼43–51 cages per ns) is nearly three times higher than that of 512 cages (∼15 cages per ns). This corresponds to a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio of 512 to 51262 cages, indicating domination of the 51262 cages in the sI hydrate structure, in good agreement with the AIMD study conducted by Liu et al.96 Phan et al. demonstrated that it is possible to quantify the effect of chemical additives on hydrate growth, reporting that the introduction of two sodium dodecyl sulfate (SDS) molecules (0.06 molecules per nm2) significantly accelerates sI hydrate growth (Fig. 5C, left panel). This observation is consistent with experiments showing that low SDS concentrations enhance CH4 hydrate growth at CH4–water interfaces, increasing growth rates from 1.18 to 1.55 mm s−1 at 4 MPa and 273 K.119 Both simulations105 and experiments119 identify SDS as an efficient promoter at hydrocarbon–water boundaries. However, increasing SDS to twelve molecules (0.36 molecules per nm2) yields little additional acceleration.97 These results indicate that additives in curved interfaces influence CH4 solubility and thereby growth rates.22,100 Through thermodynamic analysis, Phan et al.97 proposed that combining potential-of-mean-force metrics—binding free energy (ΔGbind) and adsorption barrier (ΔGbarrier)—with interfacial deformation energy (ΔGdef) (Fig. 5C, right panel) enables prediction of additive performance, supporting the design of hydrate-based CH4 storage systems. Cai et al.120 further showed that SDS can form micellar aggregates with other chemicals, such as tetrahydrofuran (THF), leading to antagonistic effects on hydrate growth kinetics, in agreement with calorimetry experiments.
3 ratio of 512 to 51262 cages, indicating domination of the 51262 cages in the sI hydrate structure, in good agreement with the AIMD study conducted by Liu et al.96 Phan et al. demonstrated that it is possible to quantify the effect of chemical additives on hydrate growth, reporting that the introduction of two sodium dodecyl sulfate (SDS) molecules (0.06 molecules per nm2) significantly accelerates sI hydrate growth (Fig. 5C, left panel). This observation is consistent with experiments showing that low SDS concentrations enhance CH4 hydrate growth at CH4–water interfaces, increasing growth rates from 1.18 to 1.55 mm s−1 at 4 MPa and 273 K.119 Both simulations105 and experiments119 identify SDS as an efficient promoter at hydrocarbon–water boundaries. However, increasing SDS to twelve molecules (0.36 molecules per nm2) yields little additional acceleration.97 These results indicate that additives in curved interfaces influence CH4 solubility and thereby growth rates.22,100 Through thermodynamic analysis, Phan et al.97 proposed that combining potential-of-mean-force metrics—binding free energy (ΔGbind) and adsorption barrier (ΔGbarrier)—with interfacial deformation energy (ΔGdef) (Fig. 5C, right panel) enables prediction of additive performance, supporting the design of hydrate-based CH4 storage systems. Cai et al.120 further showed that SDS can form micellar aggregates with other chemicals, such as tetrahydrofuran (THF), leading to antagonistic effects on hydrate growth kinetics, in agreement with calorimetry experiments.
MD simulations with a hydrate seed enable the identification of the mechanisms of action of thermodynamic promoters. Using this approach, Economou and co-workers reproduced experimental properties of CH4 hydrates,121,122 while Phan et al.101 showed that THF promotes CO2 hydrates. Cai et al.123 further demonstrated that dioxane stabilizes sII hydrates in which CH4 can occupy both small and large cages, which grow via direct and indirect mechanisms and some cages remaining empty. These findings inform the potential of hydrate-based natural gas storage because sII hydrates offer larger cage where CH4 can be stored, compared to sI hydrates, reducing the amount of water necessary to store a given amount of CH4. However, CH4 alone does not stabilize sII hydrates - other guests are needed, and it is imperative to prevent those guests from occupying the large cages, which would defeat the purpose of enhancing CH4 storage capacity.
Returning to nucleation, previous simulations show that 512 cages form early in nucleation only for small guests.95,118,124–126 Larger species such as THF, propane, and ethane cannot enter these cages and form hydrates with only large cages filled. Jacobson et al.98 used CGMD to show that guest size governs nucleation pathways: small (S), medium (M), large (L), and extra-large (XL) guests stabilize sII, sI, psI, and psII structures, respectively (where the p designates a partially filled structure with empty 512 cages). Without guests, sII is more stable than sI due to reduced structural strain.127 Small guests occupy both cage types in sII; medium-sized guests favour sI, filling both 512 and 51262 cages; large guests occupy only the large cages of sI or sII; and XL guests occupy only the largest cages. Fig. 6A shows how guest size governs stabilization: S and M favour 512 cages, whereas M-sized (CH4-like) guests preferentially stabilize 51262 cages, in line with DFT calculations33–35 and AIMD simulations.96
 | ||
| Fig. 6 A) Normalized per–water–guest interaction energy versus the guest–water size parameter σws. Colours indicate 512 (green), 51262 (blue), 51263 (red), and 51264 (orange) cages. Vertical dashed lines mark the σws of the four model guests and their favoured cages. B) Stability domains of sI, sII, psI, and psII as functions of σwm (same as σws in A) and εwm (same as εws in A); nw is the water count per cage. C) Representative nuclei for S, M, and XL guests: S fills all cages, M mainly large, XL only large. Panels A), B), and C) adapted from Jacobson et al.98 Copyright © 2010 American Chemical Society. | ||
Jacobson et al.98,128 proposed an innovative method to compare the stability of various hydrate structures. As a first-order estimate of their free energies, the enthalpies of formation of sI, sII, psI, and psII hydrates were evaluated, and a stability phase diagram was obtained (see Fig. 6B).98 Direct melting simulations were then employed to refine these predictions. At approximately 50.7 MPa, the stable phases were identified as sII for S guests, sI for M guests, psI for L guests, and psII for XL guests. Only the latter two cases differ from the phase stability diagram predictions.128 S guests (e.g., Ar) fill both cages in sII; M guests (CH4, CO2) form sI with both cages filled;126,128 L guests such as trimethylene oxide occupy only large 51262 cages in sI;1 and XL guests like THF form sII with only large 51264 cages occupied.98 These results confirm that guest size is a key descriptor for hydrates stability. Guest size is also a key descriptor for the nucleation pathways. In fact, Jacobson et al.98 identified multiple competing nucleation pathways involving filled or empty 512 cages and various 5126n cages. For CH4, nucleation is dominated by filled 512 cages,95,124 whereas empty 512 cages provide secondary channels. When guests are too large to occupy the 512 cages, nucleation proceeds through combinations of vacant 512 cages and occupied 5126n cages; medium-sized guests mainly fill the large cages, while XL guests occupy only the largest ones (see Fig. 6C).98 Empty 512 cages are stabilized by nearby solvating guest molecules, requiring at least three; filled cages can form with minimal solvation. The abundance of guest-filled large cages in the critical nuclei arises from kinetic (hydration similarity) rather than thermodynamic preference.98 This highlights the central role of water reorganization in the first hydration shell. It should be noted that, to accelerate nucleation, gas hydrate MD simulations are often conducted under supercooled and/or supersaturated conditions, under which critical nuclei may differ in structure and composition from stable or metastable hydrate phases.98
Many hydrate-based technologies are limited by slow growth and dissolution kinetics, motivating the development of kinetic promoters. Among them, environmentally friendly compounds such as tryptophan significantly enhance CH4 hydrate growth by thickening the quasi-liquid water layer at the hydrate–liquid interface, thereby facilitating guest transport and incorporation,129 consistent with mechanisms proposed by Phan et al.130 One question to be addressed here is how to identify new effective promoters that are effective at low concentrations while being environmentally benign and economically affordable.
MD simulations can help address this quest, but their practical application remains limited by time- and length-scale constraints. As a result, many phenomena bridging microscopic and meso- or macro-scopic scales cannot be directly captured. In addition, simulations differ fundamentally from experiments in how temperature and pressure are controlled: thermostats and barostats rapidly remove the heat released during hydrate nucleation and growth, unlike experimental systems. Although microcanonical simulations can account for heat transfer, they cannot simultaneously control pressure and often suffer from excessive temperature rises due to small system sizes. Consequently, faithfully reproducing experimental hydrate formation via MD remains challenging. Furthermore, current force fields have limitations and reproduce only selected molecular properties; for example, while TIP4P/Ice accurately describes the triple point for water and hydrate phases, TIP4P/2005 better represents condensed water phases. While the discussion so far considered hydrates growth, to release the guests once trapped in the hydrate cages, it is necessary to dissolve the hydrates. The relevant mechanisms are different than those responsible for growth, and must be understood to facilitate practical applications.
Collectively, DFT, GCMC, and MD simulations form a coherent framework for understanding hydrate formation and stability. However, their predictive capability remains constrained by the accuracy of XC functionals in DFT and the fidelity of classical force fields in GCMC and MD simulations, as well as by the intrinsic difficulty of sampling rare events. Recent advances in machine learning (ML)-based force fields offer promising routes to improved accuracy at a reasonable computational cost. Parallel developments in ML-assisted DFT, particularly the construction of data-driven XC functionals, provide a promising route to improving the accuracy of DFT predictions for gas hydrate energetics, host–guest interactions, and phase stability.
3. Mesoscale gas-hydrate phenomena
Microscale processes control hydrate nucleation and early growth,4,22,23 whereas subsequent growth and environmental interactions are governed by mesoscale heat and mass transfer coupled with pore-scale dynamics.25 The mesoscale domain—typically spanning ∼100 nm to 100 μm and timescales from ms to s—bridges molecular phenomena with field-scale behaviour. At this scale, hydrate systems exhibit emergent behaviour arising from coupled transport and phase-change processes, necessitating spatially resolved continuum or stochastic modelling approaches. Mesoscale frameworks capture morphology evolution, transport behaviour, and particle-size dynamics. Cellular automata (CA), lattice Boltzmann modelling (LBM), phase-field (PF), and population balance (PB) methods each address complementary aspects of mesoscale hydrate behaviour.3.1. Cellular automata modelling
The CA method was one of the first spatially resolved simulation approaches developed to study solidification (e.g., dendritic growth and grain evolution).131 It represents complex systems as networks of cells whose state variables evolve according to deterministic or probabilistic rules and effectively captures emergent behaviour arising from local interactions and molecular phenomena such as those described above for hydrates.132Buanes et al.133,134 developed a probabilistic CA that was a hybrid between MC and CA methods to simulate the growth of CO2 hydrates, where cells switch between liquid and solid states based on minimization of free energy and probabilistic rules, with the diffusion of CO2 and heat treated explicitly. Their results showed that hydrate growth is primarily diffusion-limited, producing compact crystals at low supersaturation and dendritic structures at high supersaturation. The temperature effects were minor, except under large supersaturation conditions.
Pineda et al.24 extended the MC-CA framework to model sI CO2 hydrate growth from an already formed thin film into stagnant CO2-saturated water. Phase transitions, interfacial attachment kinetics, and diffusion are incorporated in the model (see Fig. 7A). The model predicts that low subcooling leads to planar fronts, while higher subcooling destabilizes growth through the Mullins–Sekerka instability135 and produces needle-like and dendritic morphologies (see Fig. 7B). These results are consistent with experiments.136 The framework by Pineda et al. was designed with computational efficiency in mind, making it well-suited for future integration into multiscale applications. This level of analysis seems very promising, and it would benefit from the direct inclusion of molecular phenomena - for example, what is the fate of the additives used to promote hydrate growth? Can this fate affect the macroscopic performance?
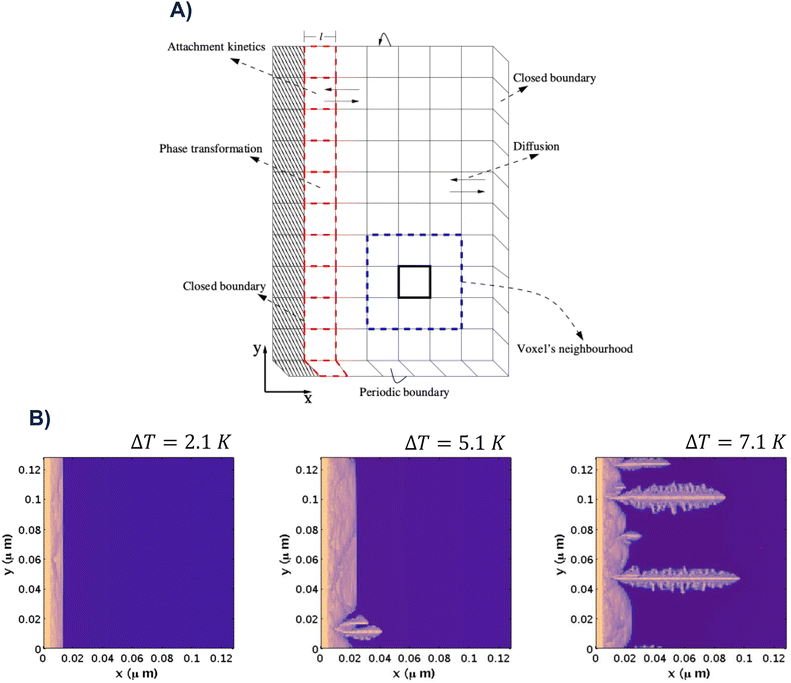 | ||
| Fig. 7 A) CA lattice of Pineda et al.24 composed of cubic voxels (V = l3): hydrate-crystal (diagonally hashed cells), hydrate–water interface (red dashed), and other voxels (black). The interface advances as voxels switch between liquid-like (water + dissolved CO2) and solid (CO2 hydrate). B) Spatiotemporal evolution of sI CO2 hydrate grown from a seeded thin film into CO2-saturated water at various subcoolings ΔT, all at t = 24 μs (common colour bars). Panels A) and B) adapted from Pineda et al.24 Copyright © 2023 The Authors. Published by the American Chemical Society under CC-BY 4.0. | ||
Jahangiri and Varaminian137 recently introduced a hybrid CA-finite difference (FD) model for THF hydrate formation at atmospheric pressure. The FD scheme captures transient heat transfer, while probabilistic CA rules govern local phase change. The model reproduces experimental temperature fields and growth kinetics and underscores the influence of surface cooling and initial solution temperature on hydrate growth.
The above examples show that CA modeling for gas hydrates is a promising and versatile approach, yet it faces important challenges. Its dependence on probabilistic transition rules based on free-energy differences or empirical acceptance functions may limit the accurate description of crystallization kinetics. Moreover, although CA models can reproduce key morphologies—such as planar growth fronts, needle-like structures, and dendritic instabilities—the validation of these predictions remains limited by the scarcity of well-controlled experimental data.136,138
3.2. Lattice Boltzmann modelling
The LBM approach was developed as a mesoscopic approach for simulating fluid flow and has since been extended to model multiphase phenomena. It recovers macroscopic hydrodynamic behaviour from simplified kinetic equations defined on a lattice, making it particularly well-suited systems in which the geometry, albeit complex, is well defined, as is the case for hydrates. LBM is especially advantageous for porous media applications, where pore-scale geometry controls multiphase flow and permeability evolution in hydrate-bearing sediments. The inherently local structure of the LBM enables efficient parallel implementation, offering significant computational benefits.140Back in 2004, Kang et al.141 first modelled crystal growth in supersaturated pure water as a first step towards simulating hydrate formation and decomposition. Although not applied to gas hydrates, the proposed two-dimensional (2D) LBM framework showed that the interaction between diffusion, surface reaction, and nucleation density governs the morphology and growth kinetics of crystals. Specifically, diffusion-controlled growth resulted in open dendritic (cluster-like) structures, while reaction-controlled growth produced compact, circular crystals.
Wang et al.142 applied a similar LBM framework directly to the formation of cyclopentane hydrates in brine. Cyclopentane offers promising applications in hydrate-based desalination because it can readily form hydrates in brine at atmospheric pressure and at temperatures above the freezing point of water.143 Their 2D-LBM kinetic model incorporated convection and diffusion of both guest molecules and salt ions, plus the salt exclusion effects and salinity-dependent kinetics, and predicted that hydrate morphology strongly depends on the diffusion length of the guest molecule, but only weakly on salinity. The predicted water conversion under a wide range of salinity conditions was found to be in good agreement with the experimental measurements.144
The evolution of permeability in hydrate-bearing sediments, which likely depends on the formation of hydrates within the pore network, is critically important for assessing natural gas production potential from hydrate deposits, as the presence, distribution, and dissociation of gas hydrates directly control gas and water flow through porous media, thereby governing reservoir productivity and stability. LBM studies have demonstrated that permeability is not solely determined by hydrate saturation, but depends also on the growth habit of the hydrate,145,146 wettability and microscale blocking effects,147 and the type of sediment.148 LBM simulations on microcomputed tomography images have confirmed that hydrate distribution can drastically alter flow pathways.149,150
Li et al.139 developed a coupled hydrodynamic-thermal LBM framework and showed that the evolution of permeability in hydrate-bearing sediments during the dissociation of hydrates strongly depends both on the hydrate morphology and the dissociation conditions. Their results, illustrated in Fig. 8, demonstrate that thermal stimulation enhances pore connectivity and leads to a nearly linear increase in permeability in heterogeneous sediments. This finding is relevant because hydrate dissociation is endothermic:139 thermal stimulation supplies the additional heat required to accelerate dissociation and to promote the early formation of interconnected flow pathways, which drive the changes in permeability.
 | ||
| Fig. 8 A) Grain-coating (left), pore-filling (middle), and dispersed (right) hydrate morphologies with sand grains (red), hydrate (green), and pore space (blue), all starting at 50.72% hydrate saturation in a 2.24 mm × 2.06 mm sample on a 224 × 206 lattice. B) LBM-predicted normalized permeability (Khyd,g) versus hydrate saturation (Shyd) for isothermal (left), depressurization (middle), and depressurization with thermal stimulation (right), with simulations run to full dissociation; each point shows saturation and corresponding permeability. Panels A) and B) adapted from Li et al.139 Copyright © 2024 Elsevier. | ||
LBM simulations have demonstrated that hydrate dissociation can be limited by either mass transfer or heat transfer, depending on local conditions.151–154 LBM investigations have also been extended to explicitly account for ice formation at sub-freezing temperatures,155 the dissociation of CO2 hydrates for energy storage applications,156 the impact of thermal fluid injection and multi-field coupling,157 as well as realistic hydrate dissociation pathways in rock matrices.158
The LBM is a powerful tool for hydrate research. However, it is inherently restricted to the pore scale and does not directly yield Darcy's law.159 Therefore, the development of robust and generic LBM frameworks to connect pore-scale insights with reservoir-scale predictions is crucial.
3.3. Phase-field modelling
The PF method is a leading computational approach for complex solidification and phase-transition phenomena. Based on a free-energy functional, it represents phase boundaries as diffuse interfaces of finite thickness and naturally captures their evolution when coupled to heat and mass transport, making it particularly suitable for modelling hydrate nucleation, growth, and dissociation processes.160In 2004 Kvamme et al.,161 implemented the PF method to investigate CO2 hydrate nucleation. An order parameter distinguishing solid hydrate from liquid water and a conserved field for the concentration of CO2 were introduced and coupled through a Helmholtz free-energy functional to represent a diffuse hydrate–liquid interface. Using thermodynamic and interfacial data from experiments and MD, the PF model allowed the calculation of the nucleation barrier and steady-state nucleation rates without adjustable parameters. The critical nuclei were found to be comparable in size to the thickness of the hydrate–liquid interface, indicating that the assumptions underlying classical droplet model, namely a sharp interface and bulk solid properties within the nucleus, break down at this scale, thereby rendering such models inconsistent for describing nucleation under these conditions.
Kvamme and co-authors published several other applications of PF to investigate CO2 and CH4 hydrates.162 These works demonstrated that the solute transport in water is the rate-limiting step for hydrate growth. It should be noted, however, that heat transport effects are not explicitly accounted for in these models, which assume isothermal conditions; consequently, the potential role of latent heat removal as a rate-limiting factor is not captured. At the same time, homogeneous nucleation was found to be unlikely because of large kinetic barriers in the formation of the complex crystal lattice. This observation suggests that homogeneous nucleation may be of limited relevance for practical applications, where heterogeneous nucleation is expected to dominate. In this context, identifying chemical structures or molecules that can act as nucleation sites may be more impactful for controlling hydrate formation. The conversion rates of CH4 hydrates to CO2 hydrates in the presence of liquid CO2 in typical underwater gas hydrate reservoirs were also estimated using PF simulations.163 Later extensions incorporated hydrodynamics and heat transport, showing that the rapid formation of new CO2 hydrates releases heat that accelerates CH4 dissociation.164 Furthermore, pore-scale modelling demonstrated that the thickness of water films around the hydrate strongly controls conversion rates.165 Finally, extensions incorporating hydrodynamics and gravity effects in PF descriptions addressed hydrate dissociation, reformation, and CH4 venting risks in natural reservoirs.166
Recently, Fu et al.167 developed a PF model to study the formation of CH4 hydrates at gas–liquid interfaces under non-equilibrium conditions. They constructed a Gibbs free energy functional for CH4–water mixtures that recovers the equilibrium phase diagram and incorporates it into a PF framework to simulate hydrate growth. Their simulations reveal that the direction of growth depends on the initial concentration of CH4 in water: supersaturated water leads to the growth of hydrate mainly in the liquid, while undersaturated water leads to the growth of hydrate in both the liquid and gas, which consumes a significant quantity of gas (see Fig. 9). Fu et al.170 demonstrated that xenon (Xe) and CH4 hydrates exhibit the same gas-to-liquid compositional gradient across their hydrate layers, providing evidence that Xe functions as a practical laboratory analogue for CH4 hydrate. This finding is important because Xe hydrate forms under milder pressure and temperature conditions, making it easier to study experimentally.
 | ||
| Fig. 9 A) Schematic of hydrate growth on a liquid–gas interface. 1D simulations (coordinate x is normalized) show hydrate phase-fraction profiles ϕs (gray) over time and the initial CH4 mass fraction χ = mCH4/(mCH4 + mH2O) (red) for B) supersaturated and C) undersaturated liquid. The black arrow marks the growth direction; the black dot the initial gas–liquid interface. Adapted from Fu et al.167 Copyright © 2018 American Physical Society. | ||
PF–LBM simulations of hydrate nucleation and growth in sand sediments reproduce realistic hydrate patterns and show how porosity, water saturation, two-phase CO2–water flow, and contact angle govern hydrate distribution and permeability. These studies also provide pore-scale mechanistic insight into hydrate-induced permeability reduction and multiphase transport.171–173
The PF method is accurate but it can become computationally cumbersome when it is used to address the challenges typically encountered when investigating hydrates. These challenges include non-equilibrium multiphase thermodynamics, coupled mass–heat–momentum transport, nanoscale interfacial dynamics and nucleation, ensuring consistency with experimental data. Because the model can be applied to large systems, it is natural to expect that eventually it should be adapted to describe the impact of reservoir heterogeneity. Furthermore, in principle, the free energy functional could be formulated to incorporate, in an effective manner, insights obtained from the methods discussed above (e.g., DFT, MD, and GCMC). Achieving such multiscale consistency remains challenging, but represents a promising direction for future research.
3.4. Population balance modelling
Understanding hydrate particle agglomeration is critical for flow assurance, where agglomeration must be prevented,14,174,175 as well as for emerging hydrate-based technologies, where agglomeration may be beneficial.2 It is not only important to understand how hydrates adhere to each other, but also how they adhere to metal surfaces, or the surfaces of other materials used in the various technologies of interest. To describe the dynamic evolution of hydrate particle populations, PB approaches are widely employed. At the core of these approaches lies the population balance equation (PBE), which describes the time evolution of the particle-size distribution by incorporating nucleation, growth, agglomeration, and breakage. The PBE is typically solved using the sectional method (SM), kinetic Monte Carlo (kMC), the direct quadrature method of moments (DQMoM), or moment-based methods (MM).176,177The first PBE applications to gas hydrates were carried out by Englezos et al.,178,179 who modelled nucleation and growth of hydrates formed from CH4, C2H6, or their mixtures in water under controlled, laboratory conditions. Using the MM to solve the PBE, they showed that the overall hydrate formation rate is governed by the total surface area available for gas incorporation. This framework linked microscopic events (nucleation and growth) to macroscopic behaviour such as gas consumption. Subsequent studies have also adopted PBE-based nucleation-and-growth models to describe hydrate formation kinetics.180,181
Herri et al.182 explicitly considered agglomeration and breakage in the PBE. Agglomeration was introduced to explain the experimentally observed crystallization behaviour of CH4 hydrate at low stirring rates. At the same time, mechanical erosion and breakage were included at high stirring rates, and the simulations with erosion achieved the best agreement with the experiments.
Balakin and co-authors applied the PBE formalism to study gas hydrate particles in pipelines.183–185 In 2010, they developed a PBE model to describe hydrate particle nucleation, growth, agglomeration, and breakage in turbulent flow, demonstrating that agglomeration and breakage have a strong influence on particle size.183 In 2011, they coupled the PBE with computational fluid dynamics (CFD), demonstrating that particle dynamics determines the behaviour of the deposition and that the coupled CFD–PBE model yields realistic predictions.184 In 2016, they presented a fully integrated CFD–PBM framework in which the particle size distribution and flow field evolved simultaneously, offering a realistic description of the hydrate agglomeration kinetics.185 The coupling of PBE with CFD or CFD-like approaches is a current open research area.186–189
The DQMoM was implemented by Sampaio et al.190 to solve a non-isothermal PBE for the formation and dissociation of gas hydrates. The modelling approach considered the mass and energy of the hydrate particles in conjunction with the gas–liquid balances and capture nucleation, growth, breakage, and agglomeration. The results showed that changes in the bulk suspension temperature, driven by slow heat transfer, strongly affect the kinetics.
Except for a few studies,185,189 most of the works mentioned above focus on hydrate–water systems. Fidel-Dufour et al.191 introduced the first PBE-based model for hydrate–oil–water systems. By tracking water droplets, hydrate particles, and agglomerates as number concentrations, their framework captured the characteristic rise and plateau of slurry viscosity during hydrate formation. Colombel et al.192 followed a similar approach to explore different agglomeration mechanisms in water-in-oil emulsions, comparing contact-induced agglomeration versus shear-limited agglomeration. They concluded that porous agglomerates are responsible for an increase in viscosity and for moving the system toward the plugging zone. Moreover, they found that contact-induced models explain the experimentally observed onset of viscosity rise, but shear-limited models better predict the experimentally reported final viscosity plateau.
Most recently, Bassani et al.31 presented a PBE framework to model the evolution of the hydrate particle/agglomerate size in a continuous flow of oil that includes nucleation, growth, and agglomeration processes simultaneously. A key aspect of the Bassani et al.'s model is the explicit representation of disruption and consolidation via a liquid-bridge mechanism, in which hydrate particles alternate between wet and dry states in response to water permeation and surface crystallisation. This wet/dry transition governs agglomeration efficiency and is itself controlled by subcooling: low subcooling favours wet, strongly agglomerating particles, while higher subcooling yields predominantly dry, weakly agglomerating particles. The model was found to be robust and predictive, offering quantitative predictions of stable agglomerate size (see Fig. 10).
 | ||
| Fig. 10 Evolution of particle/agglomerate radius during hydrate formation from the two theoretical cases of the Bassani et al. PBE model (solved via MM31), compared with experiments by Kakitani et al.168,169 solid lines indicate the full model; dashed lines indicate simplified versions. Experiments (circles) involve hydrate formation and agglomeration in oil-continuous systems with water and CH4 or CH4–C3H8 gas. Three subcooling-dependent behaviours occur: Trend 1 (low), Trend 2 (moderate), Trend 3 (high). Adapted with permission from Bassani et al.31 Copyright © 2020 American Chemical Society. | ||
PBE accuracy depends heavily on the agglomeration kernels, yet hydrate particle porosity, liquid coatings, and inhibitors often make standard kernels inadequate. Coupling PBEs with CFD is computationally costly, so PBE solutions must balance accuracy and tractability.
Taken together, CA, LBM, PF, and PBE frameworks establish the mesoscale link within a multiscale description of gas-hydrate systems, connecting molecular mechanisms to field-scale behaviour through morphology evolution, multiphase transport, and particle dynamics. Their predictive capability remains constrained by computational cost, parameter uncertainty, and the difficulty of consistently coupling heat transfer, mass transport, and phase change across scales. Advancing robust multiscale integration strategies and improving parameter calibration against molecular simulations and experiments are therefore essential for translating molecular-level insights from simulations to promote technological advancements.
4. Macroscale reservoir and flow assurance simulators
Macroscale gas hydrate models treat hydrates as a distributed phase within continuum frameworks, solving coupled multiphase flow, heat transfer, reaction kinetics, and geomechanics at the field scale.193 The goal is to predict hydrate formation, dissociation, and transport under geological and operating conditions. These simulations are crucial for (i) reservoir modelling, assessing hydrate-bearing sediments for gas production or storage,10,194–198 and (ii) flow assurance, evaluating and mitigating hydrate plugging in subsea piping systems.4.1. Reservoir production modelling
At the reservoir scale, hydrate-bearing sediments are treated as coupled thermal, hydraulic, chemical, and mechanical (THCM) systems.199,200 Numerous simulators predict gas production from hydrate dissociation under recovery strategies such as depressurization, thermal stimulation, chemical inhibition, and guest-molecule exchange.10 Tools for gas production include TOUGH + HYDRATE,201–205 HydrateResSim,206–209 and CMG STARS,210–212 while STOMP-HYDT-KE supports gas exchange and storage.26–28 Because comparisons with experimental data remain limited, several international efforts have benchmarked hydrate simulation codes.213,214 Reservoir-scale hydrate simulators solve continuum-scale partial differential equations using constitutive laws (e.g., Darcy and Fourier) and empirical correlations. Incorporating micro- and meso-scale insights remains difficult due to upscaling challenges, high computational cost, and limited experimental validation. Overcoming these barriers is crucial for achieving more predictive, physically consistent THCM models for gas hydrates.4.2. Flow assurance modelling
Flow assurance modelling predicts hydrate onset, growth, and transport in multiphase systems. It couples hydrate growth kinetics with multiphase flow simulators such as OLGA223 and LedaFlow.224OLGA applies the Colorado School of Mines Hydrate Kinetics (CSMHyK) model,225 where hydrate formation rates vary exponentially with temperature, together with the Camargo–Palermo model for agglomeration and slurry formation.226 LedaFlow uses a CSMHyK-like reaction model with a simplified subcooling-based kinetics and the SoFA viscosity model.227 Both simulators were developed and validated primarily for oil-dominated, water-in-oil systems, and require calibration for water- or gas-dominated conditions.228–230 Qu et al. showed that, with appropriate parameter tuning, the two simulators yield comparable predictions of hydrate transport behaviour.231
Since its integration into OLGA, CSMHyK has become a widely used hydrate model and has motivated newer transient simulators. Wang et al.215 developed a transient tool for predicting hydrate formation, growth, and plugging—similar to OLGA–CSMHyK but extended to oil- and water-dominated regimes, antiagglomerants (AAs)/thermodynamic hydrate inhibitors (THIs) tracking, and hydrate slurry viscosity and cohesion—and successfully reproduced plugging in a real subsea pipeline (see Fig. 11). CSMHyK describes hydrate nucleation as a deterministic event once a given subcooling condition is reached. Its predictions could become more realistic if the stochasticity typical of hydrate growth and the molecular-scale features of hydrate formation observed in MD simulations were incorporated, potentially through appropriate mesoscale models. Because hydrate plugging and slurry rheology are governed by particle agglomeration, consolidation, and breakage processes, further advances in flow assurance modelling are closely linked to improvements in the mechanistic description of hydrate particle properties.232 In particular, enhanced PB and mesoscale models that capture agglomeration kinetics and particle-scale interactions could significantly improve predictive reliability.
 | ||
| Fig. 11 Predicted transient hydrate slurry relative viscosity along a subsea pipeline at 49 vol% water cut with AAs, showing a sharp rise and plug formation between 3–8 km, matching field observations. Reproduced from Wang et al.215 Copyright © 2019 Elsevier. | ||
5. Conclusions and outlook
Computational approaches are essential for understanding gas hydrate behaviour across scales. This highlight article has reviewed key micro-, meso-, and macro-scale approaches and the distinct insights each offers at its characteristic length and timescales. As summarised in Table 1, modelling approaches at each scale face distinct yet interconnected challenges, ranging from rare-event sampling at the molecular level to reliable upscaling at larger scales. Advances in molecular simulations, mesoscale computational methods, and coupled continuum frameworks are progressively addressing these limitations.| Scale | Method | Key progress | Main challenges | Ref. |
|---|---|---|---|---|
| Quantum | DFT | Guest–host energetics; structural stability; cage occupancy; vibrational and elastic properties; spatial orientation of guest molecules | Functional accuracy (including dispersion) | 33–35, 42–44, 55, 216 |
| Molecular | GCMC | Adsorption isotherms; guest occupancy; spatial distributions; phase equilibria | Force-field sensitivity; limited transport properties; thermodynamic consistency limitations | 63, 74–78 |
| MD | Nucleation pathways; growth mechanisms; additive effects; confinement effects | Time/length-scale limitations; force-field limitations; ensemble and thermodynamic control constraints | 95, 96, 98, 101, 102, 110, 113, 217–221 | |
| Mesoscale | CA | Diffusion-limited growth; morphology evolution; MC-CA hybrid models; instability-driven growth; hybrid CA-FD thermal coupling | Empirical probabilistic rules; limited kinetic fidelity; scarce experimental validation | 24, 131–138 |
| LBM | Pore-scale multiphase flow; morphology-dependent permeability; coupled thermal–hydrodynamic dissociation; realistic pore geometry | Limited applicability to pore-scale; lack of direct Darcy-scale formulation; upscaling to reservoir scale | 139, 141, 142, 145, 149, 159 | |
| PF | Diffuse-interface nucleation; non-classical barriers; transport-limited growth; thermal coupling | Computational cost; thermodynamic complexity; parameter uncertainty | 161, 162, 164, 167, 222 | |
| PBE | Particle-size evolution; agglomeration and breakage modelling; CFD-PBE coupling; slurry rheology prediction | Kernel uncertainty; CFD coupling cost; accuracy-tractability trade-off | 31, 178, 184, 185 | |
| Macroscale | Reservoir modelling | THCM reservoir modelling; depressurization and thermal stimulation; guest-molecule exchange; benchmarked hydrate simulators | Empirical kinetics; upscaling limits; computational cost | 10, 26, 201, 206, 213 |
| Flow assurance modelling | Hydrate onset and growth prediction; OLGA/LedaFlow integration; CSMHyK kinetics; transient plugging modelling | Deterministic nucleation; empirical agglomeration; parameter calibration | 215, 223–225 |
At the microscale, DFT captures atomistic-level guest-host interactions and relative stability but is computationally expensive and essentially static, with accuracy strongly dependent on the chosen XC functional, including any applied dispersion corrections.233 GCMC and MD operate under realistic pressure–temperature conditions: GCMC yields equilibrium properties from the chosen interaction potential, whereas MD resolves dynamics but is constrained by timescale and force-field limitations. ML interatomic potentials provide a promising route to address these issues.234
At the mesoscale, CA provides fast heuristic descriptions of growth, while PF yields similar behaviour at much higher computational cost; PF can calibrate CA for large-scale studies.134 LBM resolves pore-scale flow but remains costly, and PBE predicts particle-size distributions and agglomeration while relying on empirical kernels. Interestingly, kMC—usually employed to solve PBEs—can also capture long-time guest diffusion in hydrate lattices via coarse-grained cage-to-cage hopping.235,236
Macroscale tools depend on simplified, highly upscaled models. CSMHyK, for instance, uses empirically tuned 1D formulations for oil-dominated systems with approximate chemistry, inhibitor, and plug-mechanics treatments requiring case-specific calibration. Thus, multiscale strategies are essential: micro- and meso-scale processes govern reservoir and pipeline behaviour, and macroscale simulators cannot be reliably predictive without them.
Ultimately, integrating multiscale simulation insights with molecular-scale experiments is crucial for controlling hydrate formation and growth, and ML techniques are likely to play an increasingly important role in future hydrate research.237–252
Author contributions
Miguel Pineda: writing – original draft preparation, reviewing and editing. Anh Phan: writing – original draft preparation, reviewing and editing. Michail Stamatakis: reviewing and editing. Alberto Striolo: reviewing and editing.Conflicts of interest
There are no conflicts of interest to declare.Data availability
No new data, software, code, or research results are presented in this highlight article. Figures are adapted or reproduced from previously published research papers and are appropriately cited in the corresponding figure captions.Acknowledgements
Financial support was generously provided by the UK EPSRC, under grant EP/T004282/1. Generous allocations of computing time were provided by ARCHER2, the UK National Supercomputing Service (https://www.archer2.ac.uk), via our membership of the UK's HEC Materials Chemistry Consortium, which is funded by EPSRC (EP/X035859). AS acknowledges financial support from the Asahi Glass Chair of Chemical Engineering at the University of Oklahoma.Notes and references
- E. D. J. Sloan and C. A. Koh, Clathrate Hydrates of Natural Gases, CRC Press, 2007 Search PubMed.
- A. Hassanpouryouzband, E. Joonaki, M. V. Farahani, S. Takeya, C. Ruppel, J. Yang, N. J. English, J. M. Schicks, K. Edlmann, H. Mehrabian, Z. M. Aman and B. Tohidi, Chem. Soc. Rev., 2020, 49, 5225–5309 RSC.
- C. I. Ratcliffe, Energy Fuels, 2022, 36, 10412–10429 CrossRef CAS.
- M. Khurana, Z. Yin and P. Linga, ACS Sustainable Chem. Eng., 2017, 5, 11176–11203 CrossRef CAS.
- W. F. Waite, J. C. Santamarina, D. D. Cortes, B. Dugan, D. N. Espinoza, J. Germaine, J. Jang, J. W. Jung, T. J. Kneafsey, H. Shin, K. Soga, W. J. Winters and T.-S. Yun, Rev. Geophys., 2009, 47, RG4003 CrossRef.
- T. Yagasaki, M. Matsumoto and H. Tanaka, J. Chem. Phys., 2025, 164, 020901 CrossRef.
- N. Wei, J. Pei, J. Zhao, L. Zhang, S. Zhou, P. Luo, H. Li and J. Wu, Front. Earth Sci., 2022, 10, 997337 CrossRef.
- R. Boswell and T. S. Collett, Energy Environ. Sci., 2011, 4, 1206–1215 RSC.
- T. Collett, J.-J. Bahk, R. Baker, R. Boswell, D. Divins, M. Frye, D. Goldberg, J. Husebø, C. Koh, M. Malone, M. Morell, G. Myers, C. Shipp and M. Torres, J. Chem. Eng. Data, 2014, 60, 319–329 CrossRef.
- Z. R. Chong, S. B. Yang, P. Babu, P. Linga and X.-S. Li, Appl. Energy, 2016, 162, 1633–1652 CrossRef.
- H. P. Veluswamy, ACS Appl. Energy Mater., 2024, 7, 11497–11515 CrossRef CAS.
- R. J. Davies, J. Yang, M. T. Ireland, C. Berndt, M. Á. Morales Maqueda and M. Huuse, Nat. Geosci., 2023, 17, 32–37 CrossRef.
- L. Ruffine, A. M. Tang, N. O'Neill, L. Toffin, J.-D. Paris, J. Yang, V. Georgiev, P. Fietzek, M. Giustiniani and U. Tinivella, Earth-Sci. Rev., 2023, 246, 104578 CrossRef CAS.
- P. Naeiji, M. Pan, J. M. Schicks and N. J. English, Energy Fuels, 2025, 39, 12001–12029 CrossRef CAS.
- Z. Xin, Z. Jiang, F. Wang and Y. Zhang, Chem. Eng. Sci., 2025, 306, 121290 CrossRef CAS.
- P. Wang, Y. Li, N. Sun, S. Han, X. Wang, Q. Su, Y. Li, J. He, X. Yu, S. Du, J. S. Francisco, J. Zhu and Y. Zhao, Chem. Rev., 2024, 124, 10363–10385 CrossRef CAS.
- S. K. Viswanadhan, A. Singh and H. P. Veluswamy, Gas Sci. Eng., 2024, 131, 205465 CrossRef CAS.
- N. N. Nguyen, Energy Fuels, 2023, 37, 9771–9789 CrossRef CAS.
- B. Lal and S. Nallakukkala, Gas Hydrate in Water Treatment: Technological, Economic, and Industrial Aspects, John Wiley & Sons, 2022 Search PubMed.
- S. M. Montazeri and G. Kolliopoulos, Desalination, 2022, 537, 115855 CrossRef CAS.
- X. Wang, F. Zhang and W. Lipiński, Sol. Energy, 2020, 211, 11–30 CrossRef CAS.
- A. Phan, Energy Fuels, 2025, 39, 10150–10164 CrossRef CAS.
- Y. Zhao, M. He, Y. Du and Z. Zhang, CrystEngComm, 2025, 27, 456 RSC.
- M. Pineda, A. Phan, C. A. Koh, A. Striolo and M. Stamatakis, Cryst. Growth Des., 2023, 23, 4222–4239 CrossRef CAS PubMed.
- C. L. Bassani, M. Engel and A. K. Sum, J. Chem. Phys., 2024, 160, 190901 CrossRef CAS PubMed.
- M. D. White, STOMP-HYDT-KE: A Numerical Simulator for the Production of Natural Gas Hydrate Using Guest Molecule Exchange with CO2 and N2, Pacific Northwest National Laboratory Technical Report PNNL-22130, 2012 Search PubMed.
- C. M. R. Yonkofski, J. A. Horner and M. D. White, J. Nat. Gas Sci. Eng., 2016, 35, 1480–1489 CrossRef CAS.
- A. Palodkar and A. K. Jana, Sci. Rep., 2019, 9, 18901 CrossRef CAS.
- C. L. Bassani, A. Melchuna, A. Cameirão, J.-M. Herri, A. K. Sum and R. E. Morales, Ind. Eng. Chem. Res., 2019, 58, 14446–14461 CrossRef CAS.
- C. L. Bassani, A. K. Sum, J.-M. Herri, R. E. Morales and A. Cameirão, Ind. Eng. Chem. Res., 2020, 59, 2123–2144 CrossRef CAS.
- C. L. Bassani, C. Kakitani, J. M. Herri, A. K. Sum, R. E. M. Morales and A. Cameirão, Ind. Eng. Chem. Res., 2020, 59, 15357–15377 CrossRef CAS.
- C. L. Bassani, J.-M. Herri, A. Cameirão, R. E. M. Morales and A. K. Sum, Ind. Eng. Chem. Res., 2021, 60, 14004–14012 CrossRef CAS.
- X. Zhu, A. Guerra, P. Servio and A. D. Rey, Sci. Rep., 2023, 13, 1907 CrossRef CAS PubMed.
- K. Schmidt, M. Mohamed, S. J. D. Sofla, A. Rey and P. Servio, Energy Fuels, 2025, 39, 9157–9169 CrossRef CAS.
- A. Cabrera-Ramírez, D. J. Arismendi-Arrieta, A. Valdés and R. Prosmiti, ChemPhysChem, 2021, 22, 359–369 CrossRef PubMed.
- J. Witte, N. Mardirossian, J. B. Neaton and M. Head-Gordon, J. Chem. Theory Comput., 2017, 13, 2043–2052 CrossRef CAS PubMed.
- J. Klimeš and A. Michaelides, J. Chem. Phys., 2012, 137, 120901 CrossRef PubMed.
- S. Grimme, A. Hansen, J. G. Brandenburg and C. Bannwarth, Chem. Rev., 2016, 116, 5105–5154 CrossRef CAS PubMed.
- J. Hermann, R. A. DiStasio Jr and A. Tkatchenko, Chem. Rev., 2017, 117, 4714–4758 CrossRef CAS PubMed.
- M. G. Medvedev, I. S. Bushmarinov, J. Sun, J. P. Perdew and K. A. Lyssenko, Science, 2017, 355, 49–52 CrossRef CAS PubMed.
- P. Hohenberg and W. Kohn, Phys. Rev. A, 1964, 136, B864–B871 CrossRef.
- T. M. Vlasic, P. Servio and A. D. Rey, AIP Adv., 2016, 6, 085317 CrossRef.
- S. M. Daghash, P. Servio and A. D. Rey, Mol. Simul., 2019, 45, 1524–1537 CrossRef CAS.
- S. L. Mathews, P. D. Servio and A. D. Rey, Cryst. Growth Des., 2020, 20, 5947–5955 CrossRef CAS.
- Z. M. Jendi, P. Servio and A. D. Rey, Cryst. Growth Des., 2015, 15, 5301–5309 CrossRef CAS.
- X. Zhu, A. D. Rey and P. Servio, Energy Fuels, 2022, 36, 10591–10600 CrossRef CAS.
- X. Zhu, A. D. Rey and P. Servio, Can. J. Chem. Eng., 2023, 101, 639–650 CrossRef CAS.
- S. B. Hansen and R. W. Berg, Appl. Spectrosc. Rev., 2009, 44, 168–179 CrossRef CAS.
- K. R. Ramya, G. V. Pavan Kumar and A. Venkatnathan, J. Chem. Phys., 2012, 136, 174305 CrossRef CAS PubMed.
- T. M. Vlasic, P. D. Servio and A. D. Rey, J. Phys. Chem. B, 2019, 123, 936–947 CrossRef CAS PubMed.
- H.-C. Wang, X.-L. Zhu, J.-W. Cao, X.-L. Qin, Y.-C. Yang, T.-X. Niu, Y.-B. Lu and P. Zhang, New J. Phys., 2020, 22, 093066 CrossRef CAS.
- J. M. Skelton, L. A. Burton, A. J. Jackson, F. Oba, S. C. Parker and A. Walsh, Phys. Chem. Chem. Phys., 2017, 19, 12452–12465 RSC.
- J. S. Tse, M. L. Klein and I. R. McDonald, J. Phys. Chem., 1983, 87, 4198–4203 CrossRef CAS.
- J. Jia, Y. Liang, T. Tsuji, S. Murata and T. Matsuoka, Sci. Rep., 2017, 7, 1290 CrossRef PubMed.
- A. Cabrera-Ramírez, D. J. Arismendi-Arrieta, A. Valdés and R. Prosmiti, ChemPhysChem, 2020, 21, 2618–2628 CrossRef PubMed.
- F. Izquierdo-Ruiz, A. Otero-de-la Roza, J. Contreras-García, O. Prieto-Ballesteros and J. Recio, Materials, 2016, 9, 777 CrossRef PubMed.
- A. Valdés, D. J. Arismendi-Arrieta and R. Prosmiti, J. Phys. Chem. C, 2015, 119, 3945–3956 CrossRef.
- M. Pérez-Rodríguez, A. Vidal-Vidal, J. M. Míguez, F. J. Blas, J.-P. Torré and M. M. Piñeiro, Phys. Chem. Chem. Phys., 2017, 19, 3384–3393 RSC.
- D. J. Arismendi-Arrieta, A. Valdés and R. Prosmiti, Chem. – Eur. J., 2018, 24, 9353–9363 CrossRef CAS PubMed.
- K. A. Udachin, C. I. Ratcliffe and J. A. Ripmeester, J. Phys. Chem. B, 2001, 105, 4200–4204 CrossRef CAS.
- S. Takeya, K. A. Udachin, I. L. Moudrakovski, R. Susilo and J. A. Ripmeester, J. Am. Chem. Soc., 2010, 132, 524–531 CrossRef CAS PubMed.
- Y. Park, D.-Y. Kim, J.-W. Lee, D.-G. Huh, K.-P. Park, J. Lee and H. Lee, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 12690–12694 CrossRef CAS PubMed.
- N. Qiu, X. Bai, N. Sun, X. Yu, L. Yang, Y. Li, M. Yang, Q. Huang and S. Du, J. Phys. Chem. B, 2018, 122, 9724–9737 CrossRef CAS PubMed.
- P. E. Brumby, D. Yuhara, D. T. Wu, A. K. Sum and K. Yasuoka, Fluid Phase Equilib., 2016, 413, 242–248 CrossRef CAS.
- N. I. Papadimitriou, I. N. Tsimpanogiannis, I. G. Economou and A. K. Stubos, J. Chem. Eng. Data, 2016, 61, 2886–2896 CrossRef CAS.
- N. I. Papadimitriou, I. N. Tsimpanogiannis, I. G. Economou and A. K. Stubos, J. Chem. Thermodyn., 2018, 117, 128–137 CrossRef CAS.
- H. Henley and A. Lucia, J. Nat. Gas Sci. Eng., 2015, 26, 446–452 CrossRef.
- J. Qin, C. Hartmann and W. Kuhs, Proceedings of the 8th International Conference on Gas Hydrates (ICGH-8), Beijing, China, 2015 Search PubMed.
- J. Qin and W. F. Kuhs, AIChE J., 2013, 59, 2155–2167 CrossRef CAS.
- A. Klapproth, E. Goreshnik, D. Staykova, H. Klein and W. F. Kuhs, Can. J. Phys., 2003, 81, 503–518 CrossRef CAS.
- T. C. Hansen, A. Falenty and W. F. Kuhs, J. Chem. Phys., 2016, 144, 054301 CrossRef CAS PubMed.
- A. Valdés, A. Cabrera-Ramírez and R. Prosmiti, J. Comput. Chem., 2023, 44, 1587–1598 CrossRef PubMed.
- S. Alavi, P. Dornan and T. K. Woo, ChemPhysChem, 2008, 9, 911–919 CrossRef CAS PubMed.
- I. N. Tsimpanogiannis and I. G. Economou, J. Supercrit. Fluids, 2018, 134, 51–60 CrossRef CAS.
- I. N. Tsimpanogiannis, I. G. Economou and A. K. Stubos, J. Chem. Eng. Data, 2020, 65, 1289–1299 CrossRef CAS.
- S. N. Chakraborty and L. D. Gelb, J. Phys. Chem. B, 2012, 116, 2183–2197 CrossRef CAS PubMed.
- N. I. Papadimitriou, I. N. Tsimpanogiannis, A. T. Papaioannou and A. K. Stubos, J. Phys. Chem. C, 2008, 112, 10294–10302 CrossRef CAS.
- M. H. Waage, T. J. H. Vlugt and S. Kjelstrup, J. Phys. Chem. B, 2017, 121, 7336–7350 CrossRef CAS PubMed.
- J. H. van der Waals and J. C. Platteeuw, in Clathrate Solutions, ed. I. Prigogine and S. A. Rice, John Wiley & Sons, Inc., 1958, vol. 2, pp. 1–57 Search PubMed.
- J. H. Yoon, T. Kawamura, Y. Yamamoto and T. Komai, J. Phys. Chem. A, 2004, 108, 5057–5059 CrossRef CAS.
- E. Kim, S. Lee, J. D. Lee and Y. Seo, Fuel, 2016, 164, 237–244 CrossRef CAS.
- S. Lee, S. Park, Y. Lee and Y. Seo, Chem. Eng. J., 2013, 225, 636–640 CrossRef CAS.
- S. Lee, Y. Lee, J. Lee, H. Lee and Y. Seo, Environ. Sci. Technol., 2013, 47, 13184–13190 CrossRef CAS PubMed.
- H. Lee, Y. Seo, Y.-T. Seo, I. L. Moudrakovski and J. A. Ripmeester, Angew. Chem., Int. Ed., 2003, 42, 5048–5051 CrossRef CAS PubMed.
- D.-L. Zhong, Z. Li, Y.-Y. Lu, J.-L. Wang and J. Yan, Appl. Energy, 2015, 158, 133–141 CrossRef CAS.
- M. Ota, K. Morohashi, Y. Abe, M. Watanabe, R. L. J. Smith and H. Inomata, Energy Convers. Manage., 2005, 46, 1680–1691 CrossRef CAS.
- Y.-T. Seo, H. Lee and J.-H. Yoon, J. Chem. Eng. Data, 2001, 46, 381–384 CrossRef CAS.
- Y.-T. Seo and H. Lee, J. Phys. Chem. B, 2001, 105, 10084–10090 CrossRef CAS.
- T. Uchida, R. Ohmura and A. Hori, J. Phys. Chem. C, 2008, 112, 4719–4724 CrossRef CAS.
- M. G. Martin, A. P. Thompson and T. M. Nenoff, J. Chem. Phys., 2001, 114, 7174–7181 CrossRef CAS.
- M. D. Jager, PhD thesis, Colorado School of Mines, 2001 Search PubMed.
- R. Sun and Z. Duan, Geochim. Cosmochim. Acta, 2005, 69, 4411–4424 CrossRef CAS.
- J. L. Cox, Natural gas hydrates: Properties, occurrence and recovery, Butterworth Publishers, Stoneham, MA, USA, 1982 Search PubMed.
- L. Ballard and E. D. Sloan Jr, Fluid Phase Equilib., 2004, 216, 257–270 CrossRef CAS.
- M. R. Walsh, C. A. Koh, E. D. Sloan, A. K. Sum and D. T. Wu, Science, 2009, 326, 1095–1098 CrossRef CAS PubMed.
- J. Liu, J. Hou, J. Xu, H. Liu, G. Chen and J. Zhang, Energy, 2017, 120, 698–704 CrossRef CAS.
- A. Phan, M. Stamatakis, C. A. Koh and A. Striolo, J. Colloid Interface Sci., 2023, 649, 185–193 CrossRef CAS PubMed.
- L. C. Jacobson, W. Hujo and V. Molinero, J. Phys. Chem. B, 2010, 114, 13796–13807 CrossRef CAS PubMed.
- L. C. Jacobson and V. Molinero, J. Am. Chem. Soc., 2011, 133, 6458–6463 CrossRef CAS PubMed.
- M. R. Walsh, G. T. Beckham, C. A. Koh, E. D. Sloan, D. T. Wu and A. K. Sum, J. Phys. Chem. C, 2011, 115, 21241–21248 CrossRef CAS.
- A. Phan, H. Schlösser and A. Striolo, Chem. Eng. J., 2021, 418, 129423 CrossRef CAS.
- A. Phan and A. Striolo, Energy Fuels, 2023, 37, 6002–6011 CrossRef CAS PubMed.
- A. Striolo, A. Phan and M. R. Walsh, Curr. Opin. Chem. Eng., 2019, 25, 57–66 CrossRef.
- A. Farhadian, A. Phan, Z. Taheri Rizi, A. Shaabani, E. Sadeh, M. Mohammad-Taheri, M. A. Aminolroayaei, A. Mohammadi, N. Sayyari and F. Wang, Green Chem., 2025, 27, 4523–4539 RSC.
- X. Cai, J. Worley, A. Phan, M. Salvalaglio, C. Koh and A. Striolo, J. Colloid Interface Sci., 2024, 658, 1–11 CrossRef CAS PubMed.
- Z. Zhang, P. G. Kusalik, N. Wu, C. Liu and Y. Zhang, Energy, 2022, 257, 124738 CrossRef CAS.
- K. B. Yu and A. O. Yazaydin, J. Phys. Chem. C, 2020, 124, 11015–11022 CrossRef CAS PubMed.
- E. D. Sloan Jr and F. Fleyfel, AIChE J., 1991, 37, 1281–1291 CrossRef.
- R. Radhakrishnan and B. L. Trout, J. Chem. Phys., 2002, 117, 1786–1796 CrossRef CAS.
- G. Guo, Y. Zhang, Y. J. Zhao, K. Refson and G. Shan, J. Chem. Phys., 2004, 121, 1542–1547 CrossRef CAS PubMed.
- Z. Zhang, N. Wu, C. Liu, X. Hao, Y. Zhang, K. Gao, B. Peng, W. Zheng, C. Tang and G. Guo, China Geol., 2022, 5, 330–344 CAS.
- E. A. Mastny, C. A. Miller and J. J. de Pablo, J. Chem. Phys., 2008, 129, 034701 CrossRef PubMed.
- G.-J. Guo, Y.-G. Zhang and H. Liu, J. Phys. Chem. C, 2007, 111, 2595–2606 CrossRef CAS.
- J. M. Schicks and J. A. Ripmeester, Angew. Chem., Int. Ed., 2004, 43, 3310–3313 CrossRef CAS PubMed.
- F. Fleyfel and J. P. Devlin, J. Phys. Chem., 1991, 95, 3811–3815 CrossRef CAS.
- D. K. Staykova, W. F. Kuhs, A. N. Salamatin and T. Hansen, J. Phys. Chem. B, 2003, 107, 10299–10311 CrossRef CAS.
- C. Moon, R. W. Hawtin and P. M. Rodger, Faraday Discuss., 2007, 136, 367–382 RSC.
- C. Moon, P. C. Taylor and P. M. Rodger, J. Am. Chem. Soc., 2003, 125, 4706–4707 CrossRef CAS PubMed.
- D. Daniel-David, F. Guerton, C. Dicharry, J.-P. Torré and D. Broseta, Chem. Eng. Sci., 2015, 132, 118–127 CrossRef CAS.
- X. Cai, J. Worley, A. Phan, M. Salvalaglio, C. A. Koh and A. Striolo, J. Colloid Interface Sci., 2024, 658, 1–11 CrossRef CAS PubMed.
- V. K. Michalis, J. Costandy, I. N. Tsimpanogiannis, A. K. Stubos and I. G. Economou, J. Chem. Phys., 2015, 142, 044501 CrossRef PubMed.
- I. N. Tsimpanogiannis, L. D. Peristeras, A. K. Stubos, I. G. Economou and V. K. Michalis, J. Chem. Phys., 2025, 163, 234502 CrossRef CAS PubMed.
- X. Cai, Y. Zhang, M. Salvalaglio, P. Linga and A. Striolo, Chem. Eng. J., 2026, 527, 171463 CrossRef CAS.
- R. W. Hawtin, D. Quigley and P. M. Rodger, Phys. Chem. Chem. Phys., 2008, 10, 4853–4864 RSC.
- J. Zhang, R. W. Hawtin, Y. Yang, E. Nakagava, M. Rivero, S. K. Choi and P. M. Rodger, J. Phys. Chem. B, 2008, 112, 10608–10618 CrossRef CAS PubMed.
- L. C. Jacobson, W. Hujo and V. Molinero, J. Am. Chem. Soc., 2010, 132, 11806–11811 CrossRef CAS PubMed.
- L. C. Jacobson, W. Hujo and V. Molinero, J. Phys. Chem. B, 2009, 113, 10298–10307 CrossRef CAS PubMed.
- L. C. Jacobson and V. Molinero, J. Phys. Chem. B, 2010, 114, 7302–7311 CrossRef CAS PubMed.
- X. Cai, M. Salvalaglio and A. Striolo, Proc. Natl. Acad. Sci. U. S. A., 2026, 123, e2521343123 CrossRef PubMed.
- A. Phan and A. Striolo, Energy Fuels, 2023, 37, 6002–6011 CrossRef CAS PubMed.
- K. Reuther and M. Rettenmayr, Comput. Mater. Sci., 2014, 95, 213–220 CrossRef.
- D. Raabe, Annu. Rev. Mater. Res., 2002, 32, 53–76 CrossRef CAS.
- T. Buanes, B. Kvamme and A. Svandal, J. Cryst. Growth, 2006, 287, 491–494 CrossRef CAS.
- T. Buanes, B. Kvamme and A. Svandal, J. Math. Chem., 2009, 46, 811–819 CrossRef CAS.
- D. V. Alexandrov and P. Galenko, J. Appl. Phys., 2024, 136, 055103 CrossRef CAS.
- L. Jiang, N. Xu, Q. Liu, Z. Cheng, Y. Liu and J. Zhao, Cryst. Growth Des., 2021, 20, 8148–8161 CrossRef.
- S. Jahangiri and F. Varaminian, J. Mol. Liq., 2024, 406, 125021 CrossRef CAS.
- L. Jiang, N. Xu, Q. Liu, Z. Cheng, Y. Liu and J. Zhao, Cryst. Growth Des., 2020, 20, 8148–8161 CrossRef CAS.
- Z. Li, L. Zhao, Z. Cao, S. Wu, B. Dindoruk and G. Quin, Geoenergy Sci. Eng., 2024, 241, 213066 CrossRef CAS.
- S. Chen and G. D. Doolen, Annu. Rev. Fluid Mech., 1998, 30, 339–364 CrossRef.
- Q. Kang, D. Zhang, P. C. Lichtner and I. N. Tsimpanogiannis, Geophys. Res. Lett., 2004, 31, L21604 CrossRef.
- Q. Wang, D. Han, Z. Wang, Q. Ma and D. Wang, Chem. Eng. J., 2016, 366, 133–140 CrossRef.
- D. Corak, T. Barth, S. Høiland, T. Skodvin, R. Larsen and T. Skjetne, Desalination, 2011, 278, 268–274 CrossRef CAS.
- G. Zylyftari, J. W. Lee and J. F. Morris, Chem. Eng. Sci., 2013, 95, 148–160 CrossRef CAS.
- D. H. Kang, K. Y. Yu, T. S. Kim and J. Jang, Geophys. Res. Lett., 2016, 43, 9018–9025 CrossRef.
- J. Hou, Y. Ji, K. Zhou, Y. Liu and B. Wei, Int. J. Heat Mass Transfer, 2018, 126, 416–424 CrossRef CAS.
- Y. Ji, T. J. Kneafsey, J. Hou, J. Zhao, C. Liu, T. Guo, B. Wei, E. Zhao and Y. Bai, Fuel, 2022, 321, 124013 CrossRef CAS.
- Y. Li, T. Xu, X. Xin, B. Yang, Y. Xia, Y. Zang, Y. Yuan and H. Zhu, J. Hydrol., 2024, 635, 131178 CrossRef CAS.
- X. Chen, R. Verma, D. Nicolas Espinoza and M. Prodanovíc, Water Resour. Res., 2018, 54, 600–608 CrossRef.
- J. Sun, Z. Zhang, D. Wang, C. Liu, H. Lu and K. Song, Mar. Pet. Geol., 2021, 123, 104749 CrossRef CAS.
- L. Zhang, C. Zhang, K. Zhang, L. Zhang, J. Yao, H. Sun and Y. Yang, Water Resour. Res., 2019, 55, 8422–8444 CrossRef CAS.
- J. Yang, X. Dai, Q. Xu, Z. Liu, C. Zan, W. Long and L. Shi, Chem. Eng. J., 2021, 423, 130226 CrossRef.
- J. Yang, Q. Xu, Z. Liu and L. Shi, Chem. Eng. J., 2022, 430, 132786 CrossRef CAS.
- J. Yang, Q. Xu, Z. Liu, L. Shi, T. Lei and K. H. Luo, Chem. Eng. Sci., 2023, 275, 118742 CrossRef CAS.
- J. Yang, B. Dong, W. Ping, F. Geng, L. Zhang, Y. Quin, C. Cheng and W. Li, Energy, 2023, 266, 126489 CrossRef.
- X. Li, W. Cui, T. Ma, Z. Ma, J. Liu and Q. Wang, Energy, 2023, 278, 127984 CrossRef CAS.
- Z. Li, Z. Wang and H. Ji, Water, 2024, 16, 2734 CrossRef CAS.
- J. Yang, Q. Xu, X. Kou, G. Wang, T. Lei, Y. Wang, X. Li and K. H. Luo, Innovation Energy, 2024, 1, 100015 CrossRef CAS.
- P. Hassard, I. Turner and D. Lester, Transp. Porous Media, 2023, 150, 675–707 CrossRef.
- I. Steinbach, Modell. Simul. Mater. Sci. Eng., 2009, 17, 073001 CrossRef.
- B. Kvamme, A. Graue, L. Gránásy, G. Tóth, T. Pusztai and G. Tegze, Phys. Chem. Chem. Phys., 2004, 6, 2327–2334 RSC.
- A. Svandal, B. Kvamme, L. Gránásy, T. Pusztai, T. Buanes and J. Hove, J. Cryst. Growth, 2006, 287, 486–490 CrossRef CAS.
- G. Tegze, L. Gránásy and B. Kvamme, Phys. Chem. Chem. Phys., 2007, 9, 3104–3111 RSC.
- B. Kvamme, M. Qasim, K. Baig, P.-F. Kivelä and J. Bauman, Int. J. Greenhouse Gas Control, 2014, 29, 263–278 CrossRef CAS.
- K. Baig, B. Kvamme, T. Kuznetsova and J. Bauman, AIChE J., 2015, 61, 3944–3957 CrossRef CAS.
- M. Qasim, B. Kvamme and K. Baig, Int. J. Geol., 2011, 5, 48–52 Search PubMed.
- X. Fu, L. Cueto-Felgueroso and R. Juanes, Phys. Rev. Lett., 2018, 120, 144501 CrossRef CAS PubMed.
- C. Kakitani, PhD thesis, Federal University of Technology–Paraná (UTFPR), Curitiba, Brazil, 2019 Search PubMed.
- C. Kakitani, D. C. Marques, M. A. M. Neto, A. Teixeira, L. S. Valim, R. E. M. Morales and A. K. Sum, Proceedings of the Offshore Technology Conference, Houston, Texas, USA, 2020, pp. 1–8 Search PubMed.
- X. Fu, W. F. Waite, L. Cueto-Felgueroso and R. Juanes, Geochem., Geophys., Geosyst., 2018, 20, 2462–2472 CrossRef.
- A. Fukumoto, K. Kamada, T. Sato, H. Oyama, H. Torii, F. Kiyono, J. Nagao, N. Temma and H. Narita, J. Nat. Gas Sci. Eng., 2018, 50, 269–281 CrossRef CAS.
- A. J. Yamaguchi, T. Sato, A. Nono, S. A. Scheidereit and T. Tobase, Mar. Syst. Ocean Technol., 2021, 16, 157–167 CrossRef.
- S. Lu, A. J. Yamaguchi, K. Kobayashi, T. Sato and T. Tobase, Int. J. Greenh. Gas Con., 2022, 121, 103789 CrossRef CAS.
- Y. Song, L. Yang, W. Zhao, J. Liu, M. Yang, Y. Li, Y. Liu and Q. Li, Renew. Sustain. Energy Rev., 2014, 31, 778–791 CrossRef CAS.
- S. Mokhatab, R. J. Wilkens and K. J. Leontaritis, Energy Sources, Part A, 2007, 29, 39–45 CrossRef CAS.
- J. Solsvik and H. A. Jakobsen, Chem. Soc. Rev., 2015, 36, 510–520 CAS.
- H. M. Omar and S. Rohani, Cryst. Growth Des., 2017, 17, 4028–4041 CrossRef CAS.
- P. Englezos, N. Kalogerakis, P. D. Dholabhai and P. R. Bishnoi, Chem. Eng. Sci., 1987, 42, 2647–2658 CrossRef CAS.
- P. Englezos, N. Kalogerakis, P. D. Dholabhai and P. R. Bishnoi, Chem. Eng. Sci., 1987, 42, 2659–2666 CrossRef CAS.
- B. ZareNezhad, M. Mottahedin and F. Varaminian, Chem. Eng. Sci., 2015, 137, 447–457 CrossRef CAS.
- M. S. Khatami and A. Shahsavand, Energy Fuels, 2018, 32, 8131–8144 CrossRef CAS.
- J. M. Herri, J. S. Pic, F. Gruy and M. Cournil, AIChE J., 1999, 45, 590 CrossRef CAS.
- B. V. Balakin, A. C. Hoffmann and P. Kosinski, AIChE J., 2010, 56, 2052 CrossRef CAS.
- B. V. Balakin, A. C. Hoffmann and P. Kosinski, Chem. Eng. Sci., 2011, 66, 755–765 CrossRef CAS.
- S. Balakin, B. V. Lo, P. Kosinski and A. C. Hoffmann, Chem. Eng. Sci., 2016, 153, 45–57 CrossRef.
- G. Song, Y. Li, W. Wang, K. Jiang, Z. Shi and S. Yao, J. Pet. Eng., 2018, 169, 393–404 CrossRef CAS.
- C. Yu, L. Wang, C. Han, Y. Jing, J. Li, W. Wang, M. Du, X. Song and L. Zhang, J. Mech. Sci. Technol., 2023, 36, 3477–3486 CrossRef.
- C. Yu, C. Han, L. Wang, J. Jing, Y. Li, W. Wang, M. Du and X. Song, J. Mech. Sci. Technol., 2023, 37, 5159–5171 CrossRef.
- Y. Gao, Y. Xu, K. Song, Q. Li, H. Yao, H. Chen, W. Wang and Y. Li, J. Pipeline Sci. Eng., 2023, 3, 100104 CrossRef.
- T. P. Sampaio, F. W. Tavares and P. L. C. Lage, Chem. Eng. Sci., 2017, 163, 234–254 CrossRef CAS.
- A. Fidel-Dufour, F. Gruy and J.-M. Herri, Chem. Eng. Sci., 2006, 61, 505–515 CrossRef CAS.
- E. E. Colombel, P. Gateau, L. L. Barré', F. F. Gruy and T. Palermo, Oil Gas Sci. Technol. Rev., 2009, 64, 629–636 CrossRef.
- J. K. S. Sayani, B. Lal and S. R. Pedapati, Arch. Comput. Method Eng., 2022, 29, 2171–2207 CrossRef.
- F. Li, Q. Yuan, T. Li, Z. Li, C. Sun and G. Chen, Chin. J. Chem. Eng., 2019, 27, 2062–2073 CrossRef CAS.
- Z. Yin, Z. R. Chong, H. K. Tan and P. Linga, Chin. J. Chem. Eng., 2016, 35, 1362–1387 CAS.
- M. Huang, L. Wu, F. Ning, J. Wang, X. Dou, L. Zhang, T. Liu and G. Jiang, Nat. Gas Ind. B, 2023, 10, 114–129 CrossRef.
- X. S. Li, C. G. Xu, Y. Zhang, X. K. Ruan, G. Li and Y. Wang, Appl. Energy, 2016, 172, 286–322 CrossRef CAS.
- X. Ruan, X.-S. Li and C. G. Xu, J. Nat. Gas Sci. Eng., 2021, 85, 103713 CrossRef CAS.
- R. Zhou, B. Bai, G. Cai and X. Chen, Comput. Geotech., 2024, 167, 106057 CrossRef.
- L. Zhang, Q. Wu, B. Li, Q. Hao and H. Zhang, Appl. Math. Model., 2024, 129, 88–111 CrossRef.
- G. J. Moridis, User's Manual for the HYDRATE v1.5 Option of TOUGH+ v1.5: A Code for the Simulation of System Behavior in Hydrate-Bearing Geologic Media, Lawrence Berkeley National Laboratory Technical Report LBNL-6869E, 2014 Search PubMed.
- L. Chen, Y. Feng, T. Kogawa, J. Okajima, A. Komiya and S. Maruyama, Energy, 2018, 143, 128–140 CrossRef CAS.
- S. Dhakal and I. Gupta, J. Nat. Gas Sci. Eng., 2020, 75, 103154 CrossRef CAS.
- N. Wei, C. Li, X. Zhao, H. Li, L. Zhang, J. Zhao, B. Kvamme and R. B. Coffin, Front. Earth Sci., 2024, 12, 1444690 CrossRef.
- Y. Liu, J. Liu, W. Jia, Y. Bai, J. Hou, H. Xu, E. Zhao, L. Chen, T. H. J. Guo, L. Zhang and E. Chuvilin, Nat. Gas Ind. B, 2024, 11, 274–290 CrossRef CAS.
- G. J. Moridis, M. B. Kowalsky and K. Pruess, HydrateResSim User's Manual: A Numerical Simulator for Modeling the Behavior of Hydrates in Geologic Media, Earth Sciences Division, Lawrence Berkeley National Laboratory, Berkeley, CA, 2005 Search PubMed.
- P. Zhang, S. Tian, Y. Zhang, G. Li, W. Zhang, W. A. Khan and L. Ma, Energy, 2021, 220, 119549 CrossRef.
- P. Zhang, Y. Zhang, W. Zhang and S. Tian, Energy, 2022, 238, 121738 CrossRef CAS.
- I. K. Gamwo and Y. Liu, Ind. Eng. Chem. Res., 2010, 49, 5231–5245 CrossRef CAS.
- S. Zhang, Y. Ma, Z. Xu, Y. Zhang, X. Liu, X. Zhong, G. Tu and C. Chen, Energy, 2024, 303, 131998 CrossRef CAS.
- J. Liu, Z. Xu, H. Li, X. Li and S. Fan, Energy Fuels, 2023, 37, 18996–19010 CrossRef.
- T. K. Lin, M. Dahyar, M. J. Lee and B. Z. Hsieh, J. Taiwan Inst. Chem. Eng., 2020, 114, 115–124 CrossRef CAS.
- J. W. Wilder, G. J. Moridis, S. J. Wilson, M. Kurihata, M. D. White, Y. Masuda, B. J. Anderson, T. S. Collet, R. B. Hunter, H. Narita, M. Pooladi-Darvish, K. Rose and R. Boswell, Proceedings of the 6th International Conference on Gas Hydrates (ICGH 2008), Vancouver, British Columbia, Canada, 2008, pp. 1–14 Search PubMed.
- M. D. White, T. J. Kneafsey, Y. Seol, W. F. Waite, S. Ushida, J. S. Lin, E. M. Myshakin, X. Gai, S. Gupta, M. T. Reagan, A. F. Queiruga, S. Kimoto and I. Participants, Mar. Pet. Geol., 2020, 120, 104566 CrossRef CAS.
- Y. Wang, C. A. Koh, J. White, Z. Patel and L. E. Zerpa, Fuel, 2019, 252, 458–468 CrossRef CAS.
- S. Patchkovskii and J. S. Tse, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 14645–14650 CrossRef CAS PubMed.
- L. C. Jacobson and V. Molinero, J. Am. Chem. Soc., 2011, 133, 6458–6463 CrossRef CAS PubMed.
- J. Vatamanu and P. G. Kusalik, J. Phys. Chem. B, 2008, 112, 2399–2404 CrossRef CAS PubMed.
- J. Vatamanu and P. G. Kusalik, Phys. Chem. Chem. Phys., 2010, 12, 15065–15072 RSC.
- M. Matsumoto, J. Phys. Chem. Lett., 2010, 1, 1552–1556 CrossRef CAS.
- S. J. Cox, D. J. F. Taylor, T. G. A. Youngs, A. K. Soper, T. S. Totton, R. G. Chapman, M. Arjmandi, M. G. Hodges, N. T. Skipper and A. Michaelides, J. Am. Chem. Soc., 2018, 140, 3277–3284 CrossRef CAS PubMed.
- F. Cheng, H. Lyu, W. Liu, X. Sun, Z. You, Y. Li and Y. Tian, Comput. Geotech., 2025, 188, 107532 CrossRef.
- D. Turner, J. Boxall, S. Yang, D. M. Kleehammer, C. A. Koh, K. T. Miller, E. D. Sloan, Z. Xu, P. Matthews and L. Talley, 5th International Conference on Gas Hydrates, Trondheim, Trondheim, Norway, 2005, pp. 1188–1197 Search PubMed.
- T. Danielson, K. Bansal, R. Hansen and E. Leporcher, Proceedings of the 12th International Conference on Multiphase Production Technology’05, Barcelona, Spain, 2005, pp. 477–492 Search PubMed.
- L. E. Zerpa, E. D. Sloan, A. K. Sum and C. A. Koh, J. Pet. Sci. Eng., 2012, 98-99, 122–129 CrossRef CAS.
- R. Camargo and T. Palermo, Proceedings of the fourth international conference on gas hydrates, Yokohama, Japan, 2002, pp. 880–885 Search PubMed.
- T. Palermo and E. Tournis, Ind. Eng. Chem. Res., 2015, 54, 4526–4534 CrossRef CAS.
- T. B. Charlton, M. Di Lorenzo, L. E. Zerpa, C. A. Koh, M. L. Johns, E. F. May and Z. M. Aman, Energy Fuels, 2018, 32, 1012–1023 CrossRef CAS.
- S. Zhang, L. Shang, L. Zhou and Z. Lv, Energy Fuels, 2022, 36, 1747–1775 CrossRef CAS.
- Y. Wang, C. A. Koh, J. A. Dapena and L. E. Zerpa, J. Nat. Gas Sci. Eng., 2018, 48, 126–134 CrossRef.
- A. Qu, L. F. R. Dalla, L. E. Zerpa, T. Parlermo, K. Mateen, A. Fidel-Dufour and C. A. Koh, Energies, 2024, 17, 6101 CrossRef.
- R. Castrejon-Miranda, J. G. Delgado-Linares and C. A. Koh, Fuel, 2026, 409, 137808 CrossRef CAS.
- S. J. Cox, M. D. Towler, D. Alfè and A. Michaelides, J. Chem. Phys., 2014, 140, 174703 CrossRef PubMed.
- I. Batatia, P. Benner, Y. Chiang, A. M. Elena, D. P. Kovács, J. Riebesell, X. R. Advincula, M. Asta, M. Avaylon, W. J. Baldwin, F. Berger, N. Bernstein, A. Bhowmik, F. Bigi, S. M. Blau, V. Cărare, M. Ceriotti, S. Chong, J. P. Darby, S. De, F. Della Pia, V. L. Deringer, R. Elijošius, Z. El-Machachi, E. Fako, F. Falcioni, A. C. Ferrari, J. L. A. Gardner, M. J. Gawkowski, A. Genreith-Schriever, J. George, R. E. A. Goodall, J. Grandel, C. P. Grey, P. Grigorev, S. Han, W. Handley, H. H. Heenen, K. Hermansson, C. H. Ho, S. Hofmann, C. Holm, J. Jaafar, K. S. Jakob, H. Jung, V. Kapil, A. D. Kaplan, N. Karimitari, J. R. Kermode, P. Kourtis, N. Kroupa, J. Kullgren, M. C. Kuner, D. Kuryla, G. Liepuoniutė, C. Lin, J. T. Margraf, I.-B. Magdău, A. Michaelides, J. H. Moore, A. A. Naik, S. P. Niblett, S. W. Norwood, N. O'Neill, C. Ortner, K. A. Persson, K. Reuter, A. S. Rosen, L. A. M. Rosset, L. L. Schaaf, C. Schran, B. X. Shi, E. Sivonxay, T. Stenczel, C. Sutton, V. Svahn, T. D. Swinburne, J. Tilly, C. van der Oord, S. Vargas, E. Varga-Umbrich, T. Vegge, M. Vondrák, Y. Wang, W. C. Witt, T. Wolf, F. Zills and G. Csányi, J. Chem. Phys., 2025, 163, 184110 CrossRef CAS PubMed.
- B. Peters, N. E. R. Zimmermann, G. T. Beckham, J. W. Tester and B. L. Trout, J. Am. Chem. Soc., 2008, 130, 17342–17350 CrossRef CAS PubMed.
- L.-S. Chu, D. T. Wu and S. T. Lin, J. Phys. Chem. C, 2019, 123, 11233–11243 CrossRef CAS.
- H. Xu, Z. Jiao, Z. Zhang, M. Huffman and Q. Wang, Comput. Chem. Eng., 2021, 151, 107358 CrossRef CAS.
- A. J. Yamaguchi, T. Sato, T. Tobase, X. Wei, L. Huang, J. Zhang, J. Bian and T.-Y. Liu, Proceedings of the ASME OMAE, 2022, p. V010T11A001 Search PubMed.
- Z. Yu and H. Tian, Energies, 2022, 15, 4719 CrossRef CAS.
- A. J. Yamaguchi, T. Sato, T. Tobase, X. Wei, L. Huang, J. Zhang, J. Bian and T.-Y. Liu, Fuel, 2023, 334, 126678 CrossRef.
- E. L. Gjelsvik, M. Fossen and K. Tøndel, Fuel, 2023, 334, 126696 CrossRef CAS.
- M. Hosseini and Y. Leonenko, Renew. Sustain. Energy Rev., 2023, 173, 113103 CrossRef CAS.
- D. Tian, S. Yang, Y. Gong, M. Geng, Y. Li and G. Hu, Geoenergy Sci. Eng., 2023, 223, 211564 CrossRef CAS.
- H. Osei, C. B. Bavoh and B. Lal, ACS Omega, 2024, 9, 4210–4228 CrossRef CAS PubMed.
- Y. Zhang, Z. Song, Y. Lin, Q. Shi, Y. Hao, Y. Fu, J. Wu and Z. Zhang, J. Phys.: Condens. Matter, 2024, 36, 015101 CrossRef CAS PubMed.
- V. Dhamu, M. Xiao, M. F. Qureshi and P. Linga, Energy, 2024, 313, 133802 CrossRef CAS.
- H. Li, H. Sun, J. Chen, B. Chen, D. Zhong and M. Yang, Energy Fuels, 2024, 38, 23320–23335 CrossRef CAS.
- G. S. Patel and A. K. Jana, Energy Fuels, 2024, 38, 16272–16285 CrossRef CAS.
- L. Wan, P. Cao and J. Sheng, Langmuir, 2024, 40, 22058–22067 CrossRef CAS PubMed.
- M. R. Atta, A. F. Al-Mahmodi, B. Lal, H. Abdulrab and S. F. Khor, Energy Fuels, 2025, 39, 18287–18310 CrossRef CAS.
- V. Dhamu, X. Mengqi, M. F. Qureshi, Z. Yin, A. K. Jana and P. Linga, Energy, 2024, 290, 129947 CrossRef CAS.
- A. N. Rehman, C. B. Bavoh, M. Y. Khan, M. Ansari and B. Lal, Fuel, 2025, 381, 133650 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |
