The crystal engineering foundations of the MOF Nobel Prize
Nathaniel L.
Rosi
 a,
Shuhei
Furukawa
a,
Shuhei
Furukawa
 b,
Stuart R.
Batten
b,
Stuart R.
Batten
 c and
Christian J.
Doonan
c and
Christian J.
Doonan
 d
d
aCovestro Professor of Chemistry, Department of Chemistry 907a CHVRN (Chevron Science Center), University of Pittsburgh, USA. E-mail: nrosi@pitt.edu
bInstitute for Integrated Cell-Material Sciences (WPI-iCeMS), Kyoto University, iCeMS Research Building, Yoshida, Sakyo-ku, Kyoto 606-8501, Japan. E-mail: shuhei.furukawa@icems.kyoto-u.ac.jp
cSchool of Chemistry, Monash University, 19 Rainforest Walk, Victoria 3800, Australia. E-mail: stuart.batten@monash.edu
dSchool of Physics, Chemistry and Earth Sciences, University of Adelaide, Australia. E-mail: christian.doonan@adelaide.edu.au
Nathaniel Rosi (The Yaghi Laboratory)
I entered the University of Michigan as a graduate student in fall 1999. Omar was 34 years old and earlier that year had transitioned from Arizona State University. Moving with him were several graduate students, notably Hailian Li and Theresa Reineke, and postdoc/senior researcher Mohamed Eddaoudi. Postdocs Banglin Chen and Jaheon Kim arrived shortly thereafter. I joined the lab in early December, and there was a lot I didn't know. I didn't know what a MOF was until ∼October 1999, when Omar gave his recruiting talk to 1st-year graduate students. MOFs were not part of the chemistry lexicon as they are today: only folks in Yaghi's group used the acronym, and even then we didn't say ‘MOF’ but rather sounded out each letter (e.g., M-O-F). I also had no inkling of the import of my decision to join Omar's group and what it meant to work shoulder to shoulder with such a tremendous group of colleagues, many of whom have become lifelong friends.At that time, the group exclusively focused on MOFs with carboxylate linkages. MOF-2, the layered square-grid framework with Zn(II)-carboxylate paddle-wheel vertices, had been reported a year earlier.4 It was the first MOF or coordination polymer to exhibit a low-pressure Type I gas sorption isotherm, the essential stamp of permanent porosity. Approximately two weeks before I officially joined the lab, Omar, with Hailian, Mohamed, and Prof. Mike O'Keeffe, published a paper that would change history and define a major inflection point in the field.1 I remember staring at a glossy reprint with a cubic molecular lattice set against a black background. Those indigo Zn(II) tetrahedra, the terephthalate linkages, and that glowing yellow ball at the center: the MOF-5 structure in all its glory (Fig. 1)! While this iconic image alone likely attracted hundreds of new researchers into the field, it was the sorption data on the following pages that blew the roof off: SL = 2900 m2 g−1. MOFs and coordination polymers were no longer merely elegant molecular lattice-works and structural curiosities. Now, among their ranks was one of the most porous materials on the planet and any spoken-of potential was not only plausible but imminently achievable.
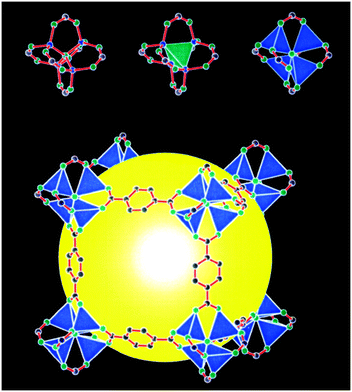 | ||
| Fig. 1 The crystal structure of MOF-5, as depicted in the seminal 1999 Nature paper. Reproduced from ref. 1 with permission. | ||
A key non-trivial advance that led to the discovery of MOF-5, and subsequently the vast libraries of structurally robust, porous MOFs, was the transition from single metal ion nodes to metal-carboxylate cluster secondary building units (SBUs).5 In those days, group efforts were focused on identifying synthetic protocols that could predictably deliver SBUs of prescribed geometry, a prerequisite for designing and achieving robust MOFs. We were interested in design, targeting specific structures, and identifying strategies for overcoming the shortcomings of earlier prototype materials (e.g., preventing things that might preclude porosity, like interpenetration or structural collapse). Indeed, the focus was on establishing the foundation, both intellectual and practical, of a new class of porous materials. Along the way, however, I and other graduate students were sometimes frustrated when MOFs we discovered weren’t celebrated as ‘publication material’ (even if they were!). But for Omar, the MOF structure needed to teach us something new that could guide the field forward. He had a tremendous knack for identifying a MOF’s distinguishing features and articulating their relevance. He would always emphasize that we must patiently study the structure to learn what it was trying to teach us.
After MOF-5 was published, Mohamed led experimental efforts toward devising a general synthesis for reliably producing MOFs with the basic Zn(II) acetate SBU, regardless of linker length, functionality, or geometry. Then, only minor synthetic adjustments were needed to crystallize a diverse collection of new MOFs, which included the IRMOF series reported in 2002,6 as well as MOF-177 reported in 2004.7 Numerous other iterations followed in subsequent years. The IRMOFs were especially significant. The series most impactfully demonstrated the modularity of MOF chemistry, that building blocks could be interchanged to afford a vast family of similar structures with different functionality, porosity, and properties. I vividly remember Omar coming around lab with his Oxford English dictionary opened to the ‘R’ section. He pointed out the word ‘reticular’ to us, and soon thereafter the series of ‘MOF-5 like’ structures became isoreticular (IR)-MOFs 1–16. When the Science paper was published in 2002, Mike O'Keeffe came into town and we all shared a nice dinner at a boutique restaurant in Ann Arbor to celebrate this special achievement. I recall that it was around this time that representatives from BASF, notably Dr. Ulrich Mueller, first visited the lab in Michigan, setting in motion the early steps toward MOF commercialization. I remember Omar telling us that folks at BASF referred to our materials as ‘MOFs’ (one syllable) instead of ‘M-O-Fs’ (three syllables). ‘MOF’ was certainly more economical and flowed off the tongue more easily, and from then on that is how we referred to them.
In parallel with IRMOF development, research at this time also laid the foundation for libraries of structures based on the Cu-carboxylate ‘paddle-wheel’ SBU.8 Groundbreaking achievements from Banglin Chen included preparation of MOF-11, permanently porous and replete with open-metal sites,9 and MOF-14, a highly porous structure consisting of two interwoven nets.10 Mohamed's syntheses of MOP-1 based on the truncated cuboctahedron (MOP = metal–organic polyhedra)11 and MOF-101, based on the NbO net12 proved definitively that structure design was possible. With conditions for producing the ‘square’ SBU clearly articulated, structure elaboration by increasing the length and branching of the linkers became a clear area of opportunity for the field in general.
Discrete SBUs were not the only focus. While tuning IRMOF syntheses, we identified MOFs with infinite rod-like SBUs.13 Studying these structures with Omar revealed something new: rod SBUs resulted in tightly packed ‘walls’ of linkers that prevented interpenetration. Thus, for the first time, MOF interpenetration could be decoupled from linker length, an important advance that ultimately led to MOF families with some of the largest pore diameters.14 We published a diverse collection of rod-based MOFs in 2005.15 Worth noting is MOF-74, prepared by a quiet hero in the Yaghi lab, Jaheon Kim. Jaheon made a lot of important synthetic advances and was also the lab's master crystallographer. MOF-74, of course, has become a staple compound in the MOF literature.
At the outset, I mentioned that there was a lot I didn't know about MOFs when I joined Omar's lab. This hasn't changed, even 25 years later (sigh). While the Nobel Prize in Chemistry very aptly recognizes this broad and monumental branch of chemistry, MOFs are still relatively unexplored. There's so much that we don't know—new rooms to discover and new spaces to mine, all within a virtually unlimited landscape of structure and composition. What new MOFs are yet to be discovered by the next generation of researchers? More importantly, what lessons are woven into their structures, waiting to be revealed through patient study?
Shuhei Furukawa, (The Kitagawa Lab)
I joined Susumu Kitagawa's laboratory as a fourth-year undergraduate student in 1999, one of the first students in the new Kitagawa Lab at Kyoto University—just six months after he had moved from Tokyo Metropolitan University. At that time, the lab was mainly exploring assembled metal complexes for their electronic properties, such as conductivity, magnetism, and redox chemistry, and I was encouraged to pursue research in this direction.16–18 Only about one-third of the students were studying porous coordination polymers or metal–organic frameworks (MOFs), even several years after the publication of the now-famous 1997 Angewandte Chemie paper,19 which would later be recognized as a landmark in the history of MOF chemistry.Perhaps due to Kitagawa's relaxed, open, and encouraging personality, the lab soon became a lively place full of self-motivated students. Around 2002, one of them, Ryo Kitaura, published a groundbreaking paper in Science20 in collaboration with researchers at SPring-8, a synchrotron radiation facility, reporting the first structural determination of a gas-adsorbed MOF phase. Following that achievement, the “porous team” in the Kitagawa Lab began investigating every MOF they had, aiming to determine their gas-adsorbed structures at SPring-8.
Among those who joined this effort was Hirotoshi Sakamoto. He chose to study the compound [Co2(4,4′-bipyridine)3(NO3)4]—one of those fragile pink crystals that looked beautiful under the microscope but behaved badly whenever you tried to study them (Fig. 2). I still remember Sakamoto reporting that every powder X-ray diffraction measurement ended in frustration: the pattern no longer matched the expected structure. The crystal had quietly transformed into a different phase before revealing its true form.
 | ||
| Fig. 2 The crystal structure of [Co2(4,4′-bipyridine)3(NO3)4], originally published in the 1997 Angew. Chem. paper. | ||
That compound, of course, was the very same one described in the 1997 paper—the tongue-and-groove framework that adsorbed gases such as methane and oxygen.19 At the time, it provided the first clear evidence that a coordination polymer could act like a zeolite, with channels that permitted gas uptake. The sorption experiments were carried out at room temperature and under high pressure, demonstrating that even fragile molecular frameworks could behave as porous solids.
Two decades later, science—and Sakamoto—came full circle. In 2022, after years of independent research, he returned to Kyoto University as a Senior Lecturer in the Kitagawa Lab. He happened to become my next-door office neighbor again. By then, the field had completely transformed: MOFs and “soft porous crystals” filled journals and conference programs, and techniques once unimaginable—such as in situ single-crystal X-ray diffraction under gas atmospheres—had become routine. The 1997 paper had grown iconic, yet one detail remained unsolved: it lacked sorption data under low-temperature, ambient-pressure conditions—the gold standard for proving permanent porosity.
One afternoon, Sakamoto said quietly, “I want to redo the experiment I couldn't finish twenty years ago”. It was said with the calm determination that only comes from long acquaintance with failure—and patience.
He reconstructed the synthesis from scratch, meticulously controlling humidity, stoichiometry, and solvent diffusion. He soon realized that the cobalt compound was sensitive to moisture and easily transformed during powder X-ray diffraction in open air. Extra care was therefore required to reproduce the 1997 results. With his colleague Ken-ichi Otake, he conducted in situ single-crystal X-ray diffraction experiments while introducing CO2 at low temperature. What they discovered was remarkable.
In their 2024 Communications Materials paper,21 Sakamoto, Otake, and Kitagawa described how CO2 molecules entered the narrow, corrugated channels by squeezing through windows smaller than themselves. The cobalt framework flexed locally, with the bipyridine ligands rotating just enough to open transient passages. At higher pressures, a collective deformation propagated through the lattice—a global “breathing” transition. What had once appeared to be a static, permanently porous framework was revealed to be dynamically alive.
Kitagawa first presented this discovery publicly in 2023 at the Nobel Symposium on MOFs in Karlskoga, Sweden—before the paper was published. I still remember how the audience responded with genuine excitement.
For me, the most touching part of this story was not the mechanism itself but what it symbolized. During our student days, that cobalt framework had resisted every attempt to be understood. Yet it had quietly kept its secret all along—it could adsorb gases even at low temperature. The true reward was to prove that the original 1997 material, the oldest gas-adsorbing MOF, was also inherently soft. The circle had finally closed after twenty-seven years.
Sakamoto's return reminded me that science does not always progress in a straight line. Some experiments simply wait for their time—for better instruments, steadier hands, or the right person to try again. His perseverance turned a half-forgotten compound into a story that now connects generations of chemists. He even wrote about this journey in a Nature Communities post titled “Born to be Soft”,22 giving a behind-the-scenes look at the rediscovery.
When the 2025 Nobel Prize in Chemistry was awarded to the pioneers of MOFs, I was overjoyed—for three reasons. My former supervisor, Kitagawa, received the prize; the entire MOF field was recognized as a legitimate and vibrant scientific discipline; and Sakamoto's 2024 Communications Materials paper was cited in the Nobel Foundation's official Scientific Background on MOFs.23
This story shows that, in science, patience and persistence can create room for discovery—just as MOFs have literally opened new spaces for chemistry.
Stuart Batten (The Robson Laboratory)
I joined Richard Robson's lab as an Honours student in 1990, the year after his seminal 1989 communication established the field,24 and the same year the follow-up full paper25 was published. I initially wanted to work on what I would later learn are known as “Robson macrocycles”,26 but Richard mentioned that he had a new line of research he'd just started, and that he was keen for me to work on that instead. “Sure” I said, in the process becoming the first student in his group to work on this area (though there was already a couple of postdocs and research assistants starting to work on it, notably including Brendan Abrahams, who is still collaborating with Richard today, more than 35 years later). It sounded like it might be an interesting thing to work on.The next year I was joined by a couple of fellow starting PhD students, but Richard's group was always a relatively small group by international standards, and funding was modest and sporadic. Notably, all the chemistry in the initial paper was done by Richard himself, in a small laboratory he had next to his office, in which he could often be found.
The initial papers by Richard foresaw with amazing clarity, even at the very beginning, much of the future of the field, including very open structures, diffusion of molecular and ionic species in and out of the materials, molecular sieving properties, porous materials with high thermal, chemical and mechanical stability, post-synthetic modification, pore size and environment control, and heterogenous catalysis. He would later even predict the possibility of COFs.27 However, in the early years the focus was purely on formulating and testing design principles, and getting proof of concept results. It is important to remember that just a year earlier John Maddox had written in Nature that “One of the continuing scandals in the physical sciences is that it remains in general impossible to predict the structure of even the simplest crystalline solids from a knowledge of their chemical composition”.28 So this work was some of the first steps in truly crystal engineering designed crystal structures. While coordination polymers had a long history in the literature, this was the first time they were to be constructed using a pre-designed topological approach.
The main aim of this early work was to design coordination polymers by combining bridging ligands with carefully designed connectivities with transition metals judiciously chosen for their likely preferred coordination geometries. It was, essentially, a retrosynthetic approach. A targeted net was broken down into nodes and linkers, and then appropriate metals and ligands chosen and combined. For example, the diamond net contains tetrahedral nodes linked together; this could be mimicked by either tetrahedral metals linked by linear bridging ligands (e.g. Cd(CN)2) or by combining tetrahedral Cu(I) metals with large tetrahedral bridging ligands (Fig. 3).25
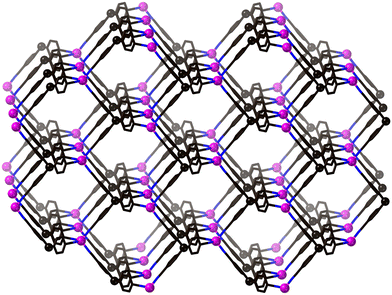 | ||
| Fig. 3 The structure that launched a new field of chemistry.25 | ||
To test the general applicability of the design strategy, we targeted a number of simple nets with a variety of node connectivities. Particularly useful were the works on nets by AF Wells,29 which gave us a range of simple template nets to try, and Richard's office soon filled with a menagerie of 3D models, many hanging from the ceiling. We soon expanded the family of structures to include rutile networks combining octahedral and trigonal components,30 PtS (tetrahedral and square planar),31 square grids,32 α-Po (now better known as pcu)33 and a variety of unexpected topologies.34 Three-connecting nodes give us the chiral (10,3)-a net (srs),35 as well as the (10,3)-b (ths) net of Zn3(tpt)2Cl6.36 This later structure was notable in that the 3D model of this structure clearly showed that the underlying net was flexible and could easily expand or contract its channels, and the iodide analogue was later used by Fujita as the basis of his very clever ‘crystal sponge’ technique.37
Given that the initial aim was to see how far we could push this design approach, rather than focusing on highly stable materials, the use of weak coordination bonds was important as it facilitated reversibility in the components coming together and greatly improved our chances of getting suitable single crystals. There was a real concern that rapid precipitation of amorphous products would be the main inevitable outcome otherwise. So monodentate nitrile, pyridyl, and metallocyano ligands, either neutral or anionic, were the favoured building blocks. Once the general net-based approach was validated, the group would later move to bidentate bridging ligands to improve stability.38 Nonetheless, early papers still managed to demonstrate ion exchange,24,25 solvent exchange and solvent-induced structural transformations,39 and even post-synthetic modification.40
Crystals were typically grown by slow diffusion of solutions of the two components at room temperature. Every structure was genuinely beautiful. While common now, these types of structures were essentially completely unknown and unseen until then. New nets were common, and interpenetration, poorly appreciated at the time, was an increasingly important phenomenon and consideration.36
The crystallography was obviously key, but it was also incredibly challenging. Richard is not a crystallographer, so collaboration with crystallographers was vital. Bernard Hoskins was utterly central to all this initial work, and his role was later assumed by Brendan Abrahams after Bernard's retirement. Many of the structures (including the initial trailblazer) had large cavities full of highly disordered solvent and counterions, which meant the sensitive crystals had to be sealed in Lindemann tubes in a solvent atmosphere, and data collected at room temperature (we did not have cryogenic cooling of crystals in the early days). Data was collected with point-detector laboratory diffractometers (up to two-week data collections). The highly disordered contents of the frameworks could also not be modelled. No solvent masking techniques were available in the software, so structures with high R-values were common, which created difficulties with referees not used to such values for chemical crystal structures (as opposed to macromolecular structures). Primitive graphical packages with no colour, real-time rotation or easy reduction to topological representation provided their own challenges in analysing the structures, particularly those with interpenetrating networks. Indeed, one of the early structures was only understood after Bernard plotted out the coordinates on graph paper across multiple unit cells.
For a number of years we were basically the only group attempting this kind of chemistry, and going to conferences as the lone voice on coordination polymers was common.34 But eventually other groups around the world started to join us, first slowly, then at a rush, and now there are whole conferences and journals devoted to the field, which I still find quite astonishing.
References
- H. Li, M. O'Keeffe, M. Eddaoudi and O. M. Yaghi, Nature, 1999, 402, 276–279, DOI:10.1038/46248.
- J. Lin, T. T. T. Nguyen, R. Vaidhyanathan, J. Burner, J. M. Taylor, H. Durekova, F. Akhtar, R. K. Mah, O. Ghaffari-Nik, S. Marx, N. Fylstra, S. S. Iremonger, K. W. Dawson, P. Sarkar, P. Hovington, A. Rajendran, T. K. Woo and G. K. H. Shimizu, Science, 2021, 374(6574), 1464–1469, DOI:10.1126/science.abi7281.
- H. Kim, S. Yang, S. R. Rao, S. Narayanan, E. A. Kapustin, H. Furukawa, A. S. Umans, O. M. Yaghi and E. N. Wang, Science, 2017, 356(6336), 430–434, DOI:10.1126/science.aam8743.
- H. Li, M. Eddaoudi, T. L. Groy and O. M. Yaghi, J. Am. Chem. Soc., 1998, 120(33), 8571–8572, DOI:10.1021/ja981669x.
- M. Eddaoudi, D. B. Moler, H. Li, B. Chen, T. M. Reineke, M. O'Keeffe and O. M. Yaghi, Acc. Chem. Res., 2001, 34(4), 319–330, DOI:10.1021/ar000034b.
- M. Eddaoudi, J. Kim, N. Rosi, D. Vodak, J. Wachter, M. O'Keeffe and O. M. Yaghi, Science, 2002, 295(5554), 469–472, DOI:10.1126/science.1067208.
- H. K. Chae, D. Y. Siberio-Pérez, J. Kim, Y. B. Go, M. Eddaoudi, A. J. Matzger, M. O'Keeffe and O. M. Yaghi, Nature, 2004, 427, 523–527, DOI:10.1038/nature02311.
- M. Eddaoudi, J. Kim, D. Vodak and O. M. Yaghi, Proc. Natl. Acad. Sci. U. S. A., 2002, 99(8), 4900–4904, DOI:10.1073/pnas.082051899.
- B. Chen, M. Eddaoudi, T. M. Reineke, J. W. Kampf, M. O'Keeffe and O. M. Yaghi, J. Am. Chem. Soc., 2000, 122(46), 11559–11560, DOI:10.1021/ja003159k.
- B. Chen, M. Eddaoudi, S. T. Hyde, M. O'Keeffe and O. M. Yaghi, Science, 2001, 291(5506), 1021–1023, DOI:10.1126/science.1056598.
- M. Eddaoudi, J. Kim, J. B. Wachter, H. K. Chae, M. O'Keeffe and O. M. Yaghi, J. Am. Chem. Soc., 2001, 123(18), 4368–4369, DOI:10.1021/ja0104352.
- M. Eddaoudi, J. Kim and M. O'Keeffe, J. Am. Chem. Soc., 2002, 124(3), 376–377, DOI:10.1021/ja017154e.
- N. L. Rosi, M. Eddaoudi, J. Kim, M. O’Keeffe and O. M. Yaghi, Angew. Chem., Int. Ed., 2002, 41, 284–287, DOI:10.1002/1521-3773(20020118)41:2<284::AID-ANIE284>3.0.CO;2-M.
- H. Deng, S. Grunder, K. E. Cordova, C. Valente, H. Furukawa, M. Hmadeh, F. Gándara, A. C. Whalley, Z. Liu, S. Asahina, H. Kazumori, M. O'Keeffe, O. Terasaki, J. F. Stoddart and O. M. Yaghi, Science, 2012, 336, 1018–1023, DOI:10.1126/science.1220131.
- N. L. Rosi, J. Kim, M. Eddaoudi, B. Chen, M. O'Keeffe and O. M. Yaghi, J. Am. Chem. Soc., 2005, 127(5), 1504–1518, DOI:10.1021/ja045123o.
- S. Furukawa and S. Kitagawa, Inorg. Chem., 2004, 43, 6464–6472, DOI:10.1021/ic0493752.
- S. Furukawa, M. Ohba and S. Kitagawa, Chem. Commun., 2005, 865–867, 10.1039/B415843A.
- S. Furukawa, T. Okubo, S. Masaoka, D. Tanaka, H.-C. Chang and S. Kitagawa, Angew. Chem., Int. Ed., 2005, 44, 2700–2704, DOI:10.1002/anie.200462962.
- M. Kondo, T. Yoshitomi, H. Matsuzaka, S. Kitagawa and K. Seki, Angew. Chem., Int. Ed. Engl., 1997, 36, 1725–1727, DOI:10.1002/anie.199717251.
- R. Kitaura, S. Kitagawa, Y. Kubota, T. C. Kobayashi, K. Kindo, Y. Mita, A. Matsuo, M. Kobayashi, H.-C. Chang, T. C. Ozawa, M. Suzuki, M. Sakata and M. Takata, Science, 2002, 298, 2358–2361, DOI:10.1126/science.1078481.
- H. Sakamoto, K.-I. Otake and S. Kitagawa, Commun. Mater., 2024, 5, 171, DOI:10.1038/s43246-024-00609-x.
- H. Sakamoto, Born to be Soft: An Overlooked Insight on the Oldest Gas-Adsorbing Porous Coordination Polymer, Springer Nature Communities, 2024, https://communities.springernature.com/posts/born-to-be-soft-an-overlooked-insight-on-the-oldest-gas-adsorbing-porous-coordination-polymer Search PubMed.
- The Nobel Foundation, Scientific Background on the Nobel Prize in Chemistry 2025: Metal–Organic Frameworks (MOFs) – New Room for Chemistry, The Royal Swedish Academy of Sciences, 2025, https://www.nobelprize.org/prizes/chemistry/2025/advanced-information Search PubMed.
- B. F. Hoskins and R. Robson, J. Am. Chem. Soc., 1989, 111, 5962–5964, DOI:10.1021/ja00197a079.
- B. F. Hoskins and R. Robson, J. Am. Chem. Soc., 1990, 112, 1546–1554, DOI:10.1021/ja00160a038.
- N. H. Pilkington and R. Robson, Aust. J. Chem., 1970, 23, 2225–2236, DOI:10.1071/CH9702225.
- R. Robson, J. Chem. Soc., Dalton Trans., 2000, 3735–3744, 10.1039/B003591M.
- J. Maddox, Nature, 1998, 335, 201, DOI:10.1038/335201a0.
- A. F. Wells, Three-Dimensional Nets and Polyhedra, Wiley-Interscience, New York, 1977 Search PubMed.
- S. R. Batten, B. F. Hoskins and R. Robson, J. Chem. Soc., Chem. Commun., 1991, 445–447, 10.1039/C39910000445.
- (a) R. W. Gable, B. F. Hoskins and R. Robson, J. Chem. Soc., Chem. Commun., 1990, 762–763, 10.1039/C39900000762; (b) B. F. Abrahams, B. F. Hoskins, D. M. Michael and R. Robson, Nature, 1994, 369, 727–729, DOI:10.1038/369727a0.
- R. W. Gable, B. F. Hoskins and R. Robson, J. Chem. Soc., Chem. Commun., 1990, 1677–1678, 10.1039/C39900001677.
- B. F. Hoskins, R. Robson and D. A. Slizys, Angew. Chem., Int. Ed. Engl., 1997, 36, 2752–2755, DOI:10.1002/anie.199727521.
- R. Robson, B. F. Abrahams, S. R. Batten, R. W. Gable, B. F. Hoskins and J. Liu, in Supramolecular Architecture: Synthetic Control in Thin Films and Solids, ACS Symposium Series, ed. T. Bein, Am. Chem. Soc., Washington DC, 1992, ch. 19, vol. 499, pp. 256–273 Search PubMed.
- B. F. Abrahams, S. R. Batten, H. Hamit, B. F. Hoskins and R. Robson, Chem. Commun., 1996, 1313–1314, 10.1039/CC9960001313.
- S. R. Batten and R. Robson, Angew. Chem., Int. Ed., 1998, 37, 1460–1494, DOI:10.1002/(SICI)1521-3773(19980619)37:11<1460::AID-ANIE1460>3.0.CO;2-Z.
- Y. Inokuma, S. Yoshioka, J. Ariyoshi, T. Arai, Y. Hitora, K. Takada, S. Matsunaga, K. Rissanen and M. Fujita, Nature, 2013, 495, 461–466, DOI:10.1038/nature11990.
- B. F. Abrahams, P. A. Jackson and R. Robson, Angew. Chem., Int. Ed., 1998, 37, 2656–2659, DOI:10.1002/(SICI)1521-3773(19981016)37:19<2656::AID-ANIE2656>3.0.CO;2-M.
- B. F. Abrahams, M. J. Hardie, B. F. Hoskins, R. Robson and G. A. Williams, J. Am. Chem. Soc., 1992, 114, 10641–10643, DOI:10.1021/ja00052a073.
- B. F. Abrahams, M. J. Hardie, B. F. Hoskins, R. Robson and E. E. Sutherland, J. Chem. Soc., Chem. Commun., 1994, 1049–1050, 10.1039/C39940001049.
| This journal is © The Royal Society of Chemistry 2026 |
