Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Wadsley vanadium oxides
Andrzej
Grzechnik
 *ab and
Karen
Friese
bc
*ab and
Karen
Friese
bc
aJülich Centre for Neutron Science-4 (JCNS-4), Forschungszentrum Jülich GmbH, Jülich D-52425, Germany. E-mail: a.grzechnik@fz-juelich.de
bInstitute of Crystallography, RWTH Aachen University, Aachen 52056, Germany
cJülich Centre for Neutron Science-2 (JCNS-2), Forschungszentrum Jülich GmbH, Jülich D-52425, Germany
First published on 11th March 2026
Abstract
Wadsley vanadium oxides form a homologous series VnO2n+1 between the end members α-V2O5 and VO2(B). They exhibit a variety of crystal structures due to different polyhedra around the V4+ and V5+ cations and polyhedral connectivity. Their structural features as well as transport and magnetic properties are reviewed here. The Wadsley vanadium oxides are oxygen deficient with different ordered vacancies in the oxygen cubic close-packing array. The rutile-type structure, with the oxygen hexagonal close packing, is found in V6O13 (n = 6) obtained at high pressures and high temperatures, which unlike its α polymorph stable at atmospheric conditions, does not undergo a metal–insulator phase transition. This finding opens a possibility of synthesizing new materials with unexpected properties in the system V2O5–VO2 by varying compositions and exerting extreme conditions.
 Karen Friese | Karen Friese studied mineralogy (Diploma) at the University of Hamburg, where she also obtained her PhD. Her work is focused on crystal structure determination of functional materials. |
Introduction
Experimentally determined equilibrium phases in the central part of the V–O phase diagram (VOx, 1.5 ≤ x ≤ 2.5) at atmospheric pressure are V2O3, VnO2n−1 (n = 3 ÷ 8), VO2, V3O7, V6O13, and V2O5, with V2O3, VO2, and V2O5 exhibiting very narrow composition ranges.1 The theoretically calculated phase diagram includes only V2O3, V3O5, VO2, V3O7, and V2O5.2 The synthesis, functionalities, and applications of the VOx oxides are extensively reviewed in ref. 2–5.The oxides VnO2n−1 (n = 3 ÷ 9) form a Magnéli homologous series, VnO2n−1 = V2O3 + (n − 2) VO2.6,7 The end members of this series are corundum-type V2O3 and rutile-type VO2.8 The corundum structure (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c, Z = 6) is a hexagonal close-packed array of oxygen atoms, in which the V3+ cations occupy two-thirds of the octahedral sites. It is built of pairs of VO6 octahedra sharing faces along the c direction. Such pairs form chains by edge-sharing in the plane perpendicular to c. These chains are linked to others by edge sharing in a three-dimensional network. The rutile structure (phase R, P42/mnm, Z = 2) is a distorted hexagonal close-packed oxygen array with the V4+ cations occupying one half of the octahedral sites. Chains of edge-sharing VO6 octahedra along the c direction connect with each other in a three-dimensional network. Magnéli phases could be derived from the parent rutile structure by removing an oxygen layer at every nth vanadium layer in the direction perpendicular to the (211) plane of the parent rutile structure.8 With respect to the composition of vanadium dioxide, they are anion deficient and can be expressed as VO2−y. They order antiferromagnetically and, apart from V7O13, undergo metal–insulator phase transitions (MIT) accompanied by structural transformations.6–10Magnéli phases are also known for titanium, niobium, and tungsten oxides.11
c, Z = 6) is a hexagonal close-packed array of oxygen atoms, in which the V3+ cations occupy two-thirds of the octahedral sites. It is built of pairs of VO6 octahedra sharing faces along the c direction. Such pairs form chains by edge-sharing in the plane perpendicular to c. These chains are linked to others by edge sharing in a three-dimensional network. The rutile structure (phase R, P42/mnm, Z = 2) is a distorted hexagonal close-packed oxygen array with the V4+ cations occupying one half of the octahedral sites. Chains of edge-sharing VO6 octahedra along the c direction connect with each other in a three-dimensional network. Magnéli phases could be derived from the parent rutile structure by removing an oxygen layer at every nth vanadium layer in the direction perpendicular to the (211) plane of the parent rutile structure.8 With respect to the composition of vanadium dioxide, they are anion deficient and can be expressed as VO2−y. They order antiferromagnetically and, apart from V7O13, undergo metal–insulator phase transitions (MIT) accompanied by structural transformations.6–10Magnéli phases are also known for titanium, niobium, and tungsten oxides.11
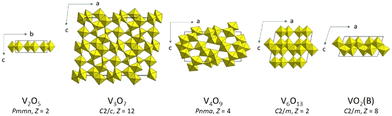
The emphasis of this article is on the homologous series of vanadium oxides VnO2n+1 predicted by A. D. Wadsley12 in the system V2O5–VO2. Its formula can be written as VnO2n+1 = V2O5 + (n − 2) VO2 for 2 ≤ n. The end members of this homologous series are α-V2O5 (Pmmn, Z = 2) and VO2(B) (C2/m, Z = 8).8 With respect to the chemical composition of VO2, the Wadsley oxides are cation deficient, V1−zO2. They exhibit a variety of crystal structures due to different coordination polyhedra around the V atoms (tetrahedra, trigonal prisms, square pyramids, or octahedra) and different polyhedral connectivities. The V5+ cations favor the polyhedra with low coordination numbers, while the V4+ cations are five- or six-fold coordinated to the oxygen atoms.13 Compared to the Magnéli phases, the polymorphism in the Wadsley series is altogether richer at ambient pressure. It offers a possibility to study the structure–property relationship in correlation with phase transitions including those induced at extreme conditions. In the following, the crystal structures and physical properties of the Wadsley homologous series VnO2n+1 and of the relevant polymorphs of vanadium dioxide are reviewed. It is then indicated how high-pressure phase transitions and synthesis could lead to new materials in the V2O5–VO2 system.
Rutile-type and VO2(S) vanadium dioxides
The first-order MIT in stoichiometric vanadium dioxide occurs at TMIT ≈ 341 K.14–17 It is associated with a magnetic susceptibility drop and a change in thermochromic properties. The V3+ and V5+ cations co-exist in the high-temperature metallic phase R due to charge fluctuations of the V4+ cations.16 The V–V distance in the octahedral chains is equal to the c lattice parameter (Fig. 1). The low-temperature insulating phase M1 (P21/c, Z = 4) is made of VO6 chains but with V–V dimers in a zigzag pattern due to displacement of the V atoms from the ideal-rutile positions.18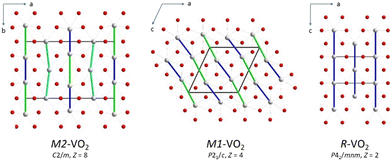 | ||
| Fig. 1 Interatomic connectivity in VO2 polymorphs. The V–V distances in the octahedral chains are drawn as thick green, blue, and cyan lines. | ||
Doping with low-valence cations (e.g., Al3+, Cr3+, Fe3+) stabilizes additional insulating polymorphs M2 (C2/m, Z = 8) and T (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 4). In M2, there are alternating short and long V–V distances in the linear octahedral chain and equidistant V–V distances in the zigzag octahedral chain (Fig. 1).19,20 T is a distorted variant of M2 due to linearity breaking and pairing of the V atoms in the zigzag chain.20 M1 and M2 can transform into each other, with T as an intermediate.19 M2 can also be an intermediate in the M1 → R transition.21–23 The three insulating phases may coexist and form domains.24 Their stabilities are affected by electric field, strain, or pressure.14,16,25,26 The high-valence dopants (e.g., Nb5+, Mo6+, W6+) lower TMIT, while the low-valence dopants (e.g., Al3+, Cr3+, Fe3+) increase it.14–16 The charge is compensated by the presence of the V3+ or V5+ cations, respectively. In non-stoichiometric undoped vanadium dioxide, TMIT decreases in VO2−y with increasing y, while it increases in V1−zO2 with increasing z.15–17 The crystallographic data across MIT for different non-stoichiometries are lacking.16
, Z = 4). In M2, there are alternating short and long V–V distances in the linear octahedral chain and equidistant V–V distances in the zigzag octahedral chain (Fig. 1).19,20 T is a distorted variant of M2 due to linearity breaking and pairing of the V atoms in the zigzag chain.20 M1 and M2 can transform into each other, with T as an intermediate.19 M2 can also be an intermediate in the M1 → R transition.21–23 The three insulating phases may coexist and form domains.24 Their stabilities are affected by electric field, strain, or pressure.14,16,25,26 The high-valence dopants (e.g., Nb5+, Mo6+, W6+) lower TMIT, while the low-valence dopants (e.g., Al3+, Cr3+, Fe3+) increase it.14–16 The charge is compensated by the presence of the V3+ or V5+ cations, respectively. In non-stoichiometric undoped vanadium dioxide, TMIT decreases in VO2−y with increasing y, while it increases in V1−zO2 with increasing z.15–17 The crystallographic data across MIT for different non-stoichiometries are lacking.16
In addition to the R and M1 polymorphs, insulating M1′ (monoclinic), metallic X (triclinic) and O (Pnnm, Z = 4) phases are identified in the pressure–temperature phase diagram of VO2.27 At room temperature, M1 transforms to isostructural M1′ at 13.9 GPa.26 At 383 K, R undergoes a phase transition to O above 13.7 GPa.28 Upon further compression, both M1′ and O transform into X above 34.3 and 38.3 GPa at room temperature and 383 K, respectively. X, which is postulated to be metallic above about 35 GPa,28–31 co-exists with M1′ in the pressure range 32–42 GPa.
Insulating VO2(S) (P3, Z = 7), synthesized from a mixture of V2O3 and V2O5 at 2–7 GPa and 873–1173 K, is built of isolated and strongly distorted edge-sharing octahedral VO6 trimers arranged in two different layers.32 The trimers in every second layer are linked via edge-sharing with another octahedron. Upon heating, the product transforms to M1 below 4 GPa and to M2 at higher pressures.
Cation-deficient V1−zO2 phases with a distorted rutile structure (P2/m, Z = 2) are synthesized at 6.5 GPa and 1273 K by substituting 2 wt% and 10 wt% of V2O5 into VO2.33,34 Their compositions could also be written as V0.995O2 and V0.976O2, respectively. The V–V distances in both octahedral chains are equidistant. The resistivity data show that V0.995O2 and V0.976O2 undergo MIT at 353 K and 361 K, respectively.33 No structural details of the metallic phases in both materials are provided.
A summary of the compounds discussed in this section, including their space group symmetries, synthesis methods, V–V distances, and properties, is provided in Table S1 in the SI. Further details on polymorphism and physical properties of vanadium dioxide can be found in the ref. 2–5 and 14–17.
α-V2O5 and VO2(B) end members of the Wadsley homologous series VnO2n+1
α-V2O5
α-V2O5 (Pmmn, Z = 2) is built of ribbons of edge-sharing VO5 square pyramids or very distorted VO6 octahedra when an additional long V–O distance is considered (Fig. 2).13 The smallest building block in the idealized octahedral structure is a doublet of edge-sharing octahedra in the ribbons along the a axis. These ribbons are linked with the adjacent ones by corners to form layers. In each ribbon, there is a zigzag chain of equidistant V–V atoms.35 The layers share corners in a three-dimensional structure.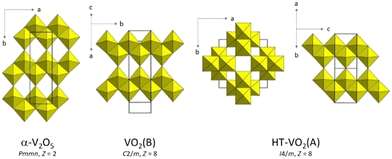 | ||
| Fig. 2 Octahedral connectivity in the idealized α-V2O5 structure with distorted VO6 octahedra, single layer in VO2(B), and high-temperature phase of VO2(A). | ||
α-V2O5 amorphizes above about 7.3 GPa.36,37 The polymorph that crystallizes from the amorphous material at high pressures and high temperatures is δ-V2O5 (C2/c, Z = 4) as evidenced by in situ angle-dispersive synchrotron powder diffraction in large-volume multi-anvil press.37 It is built of slabs connected with each other by octahedral corners.36–39 Each slab is made of edge-sharing VO6 doublets linked with the others by corners. The single slab is in fact a building block of the rutile type (Fig. 3). In β-V2O5 (P21/m, Z = 2), obtained at lower pressures than δ-V2O5,38,39 there are two symmetry non-equivalent V positions V1 and V2. The octahedra V1O6 form isolated stripes of the α-V2O5 type along the b direction. The V2O6 octahedra are linked by corners to form chains along b. The stripes and chains share edges within one layer. The phase boundaries between α-V2O5 and the recovered β-V2O5 and δ-V2O5 products from high pressures and temperatures are drawn in ref. 39.
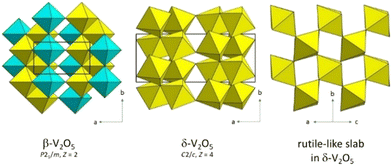 | ||
| Fig. 3 High-pressure high temperature phases of V2O5. The V1O6 and V2O6 octahedra in β-V2O5 are drawn yellow and cyan, respectively. | ||
VO2(B)
VO2(B) is synthesized by reducing V2O5 in a sulphur or hydrogen atmosphere.41 Its crystal structure (C2/m, Z = 8) consists of sheets of edge- and corner-sharing distorted VO6 octahedra. It could be derived from the one of α-V2O5 by a crystallographic shear when every second oxygen plane is removed, and the adjacent blocks are shifted by the vector ⅙[103]. In an alternative description, each sheet in VO2(B) could be separated into two layers of the idealized α-V2O5 type,13 which share octahedral edges with each other (Fig. 2).VO2(B) as a bulk, nanorod, or thin film material undergoes a first-order isostructural phase transition in the range 180–300 K (ref. 42–46) with co-existing low-temperature (insulating and magnetically ordered), intermediate-temperature (insulating), and high-temperature (presumably metallic) phases.45 The transition is associated with partial V4+–V4+ pairing.42 When heated in argon, it transforms to R.43 When heated under reducing conditions, it turns into corundum. Annealing in vacuum yields M1.47
Under uniaxial tensile strain, VO2(B) undergoes MIT at room temperature.48 The transition along the b axis is gradual, while it is abrupt along the a axis. Insulating domains develop with increasing tensile strain along the b axis. They are due to the V–V dimerization within the distorted zigzag chains of vanadium atoms in the octahedral ribbons (Fig. 2).
The structural phase transition VO2(B) → VO2(A) is induced by uniaxial compressing and grinding VO2(B) powders at room temperature and by subsequent heat treatment.49,50 In nanobelts, the sequence of the phase transitions is VO2(B) → VO2(A)HT → VO2(A)LT, where VO2(A)LT and VO2(A)HT are the low- (P4/ncc, Z = 16) and high-temperature (I4/m, Z = 8) phases of VO2(A), respectively.51 In the low-temperature structure below 435 K, the V atoms form zigzags with alternating short and long V–V distances in the c direction due to V4+–V4+ pairing. At higher temperatures, they form straight lines of equidistant V atoms. VO2(A) has a lower formation energy compared to VO2(B).52 On annealing, VO2(A) transforms to M1.3
The difference between VO2(B) and both phases of VO2(A) is the connectivity of the VO6 octahedra (Fig. 2). According to the structure interpretation in ref. 48–51, the ribbons in VO2(B) are formed by octahedral doublets that share edges with the adjacent doublets. In VO2(A), the neighbouring doublets are rotated by 90° with respect to each other and share edges. The resulting chains share corners in the three-dimensional framework. Our description of the low- and high-temperature VO2(A) structures, which is an alternative to the one in ref. 49–51, is that they consist of slabs of edge-sharing octahedra stacked along the [110] or [1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0] directions. Each slab is then formed by two edge-sharing layers that are formed by ribbons that are nearly exactly like those in α-V2O5 and VO2(B) (Fig. 2). In other words, α-V2O5, VO2(B), and VO2(A) have the same building unit that is the single layer of the idealized α-V2O5 octahedral type.13 The differences arise from the stacking of these layers due to corner-sharing in α-V2O5 and different schemes of octahedral edge-sharing in VO2(B) and VO2(A).
0] directions. Each slab is then formed by two edge-sharing layers that are formed by ribbons that are nearly exactly like those in α-V2O5 and VO2(B) (Fig. 2). In other words, α-V2O5, VO2(B), and VO2(A) have the same building unit that is the single layer of the idealized α-V2O5 octahedral type.13 The differences arise from the stacking of these layers due to corner-sharing in α-V2O5 and different schemes of octahedral edge-sharing in VO2(B) and VO2(A).
At room temperature, VO2(B) nanosheets and nanobelts become amorphous above 21 GPa (ref. 53) and 30 GPa,54 respectively, due to a dynamically hindered transformation to M1.53 When annealed at atmospheric pressure, the recovered material, produced during amorphization of the nanosheets, recrystallizes back to VO2(B). VO2(A) becomes metallic at about 28 GPa and amorphizes at 32 GPa at room temperature.55
Wadsley homologous series VnO2n+1
Known Wadsley VnO2n+1 phases are V3O7 (V0.86O2), V4O9 (V0.89O2), and V6O13 (V0.92O2) (Fig. 4). The stable compounds in the experimentally determined V–O phase diagram are V3O7 and V6O13,1 while thermodynamical calculations find only V3O7 as a stable phase.2 The difficulty extending the series using various synthesis methods at ambient conditions is due to different chemical and structural characteristics of the V4+ and V5+ cations. The unifying description of the crystal structures for n = 2, 3, and 6 as well as for α-V2O5 and VO2(B) is presented in ref. 8. These materials have oxygen-deficient structures deduced from VOx (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, Z = 4), where x ≈ 1, by introducing different ordered vacancies in the oxygen close-packing array. Symmetry breaking and collapse of the fcc layers along the cubic c axis induces shear deformations. The considerations in ref. 8 do not include the analysis of the structures of V4O9 determined in ref. 56 and 57. A classification of the polyhedral connectivities is given in ref. 13. A summary of the compounds discussed in this section, including α-V2O5 as well as VO2(B) and VO2(A), is provided in Table S2 in the SI.
m, Z = 4), where x ≈ 1, by introducing different ordered vacancies in the oxygen close-packing array. Symmetry breaking and collapse of the fcc layers along the cubic c axis induces shear deformations. The considerations in ref. 8 do not include the analysis of the structures of V4O9 determined in ref. 56 and 57. A classification of the polyhedral connectivities is given in ref. 13. A summary of the compounds discussed in this section, including α-V2O5 as well as VO2(B) and VO2(A), is provided in Table S2 in the SI.
V3O7
V3O7 (C2/c, Z = 12), which is an insulator and uniaxial ferromagnet,58,59 is synthesized from a mixture of V2O3 and V2O5.60 The structure is built of distorted VO6 octahedra and VO5 polyhedra joined by corner- and edge-sharing to form a three-dimensional framework (Fig. 4). The VO5 polyhedra could be interpreted either as distorted trigonal bipyramids or square pyramids. The V4+ cations are situated in the octahedra, while the V5+ cations are in the VO5 polyhedra.Related to VO2(S)32 is the cation-deficient insulating oxide V3.047O7 (P3, Z = 2) obtained by reduction of V2O5 at 5 GPa and 1073 K.61 It has a composition very close to V3O7 and is reported to have the Al4Ta3O13(OH) simpsonite-type structure. The structure of V3.047O7 is described as a stacking of two octahedral layers along the c direction. One of them has isolated triplets of edge-sharing VO6 octahedra. In the other, the triplets are connected to each other via additional bridging octahedra around partially occupied vanadium sites. Zibrov et al.61 expected that a new phase of V3O7 with the simpsonite structure could also be synthesized from a mixture of V2O3 and V2O5 at the same pressure–temperature conditions. However, their synthesis was not successful.
V4O9
Three different V4O9 products, depending on the synthesis procedures, are reported in the literature. The phase prepared by decomposing V3O7 in supercritical water at 873 K and 0.2 GPa (Pnma, Z = 4) is built of highly distorted edge-sharing octahedra in zigzag ribbons running along the b direction (Fig. 4).56 The ribbons are joined by corners to form a three-dimensional framework. V4O9 could also be grown topotactically on single crystals of α-V2O5 by reducing them in a sulphur atmosphere.57 The resulting crystal structure can then be considered a distorted superstructure of α-V2O5. The third product (Cmcm, Z = 8) can be obtained with a soft chemistry method also by reducing V2O5 with sulphur.62 It is composed of pairs of distorted edge-sharing octahedra and square pyramids linked by corner sharing with tetrahedra. The V atoms in the octahedra and pyramids have the valence 4+, while they have the valence 5+ in the tetrahedra. Such a structure cannot be explained with the shear concept that relies on the presence of oxygen-plane faults. This product is a spin-½ one-dimensional antiferromagnetic system with a spin-gap ground state.62α-V6O13
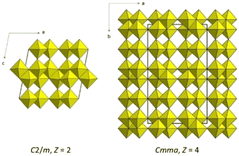
α-V6O13 (C2/m, Z = 2), obtained by reacting a mixture of V2O3 and V2O5 in an evacuated quartz glass tube at 873 K,63–65 is composed of single and double layers formed by edge- and corner-sharing distorted VO6 octahedra. The crystal structure can be described as built of alternating VO2(B)- and idealized α-V2O5-like layers stacked along the c axis (Fig. 2, 4, and 5). One of the sites for the vanadium atoms in the double layer is preferentially occupied by the V5+ cations.66 In the idealized α-V2O5, the shear planes separate narrow slabs of the ReO3 type.65 The structure of α-V6O13 is formed when another set of shear planes, perpendicular to the first, is introduced in α-V2O5 resulting in the loss of oxygen. The relationship between α-V2O5 and α-V6O13 is also defined in ref. 67. Removing all the atoms on every third (001) oxygen-atom plane in α-V2O5 and introducing crystallographic shear ⅙[10![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ] leads to the structure of α-V6O13. Such a mechanism of transformations yields the double layer of the VO2(B) type. The shear in β-V6O13 (Cmma, Z = 4), which is obtained by exposure of α-V6O13 to an electron beam67 and by hydrothermal synthesis,68 is ½[0
] leads to the structure of α-V6O13. Such a mechanism of transformations yields the double layer of the VO2(B) type. The shear in β-V6O13 (Cmma, Z = 4), which is obtained by exposure of α-V6O13 to an electron beam67 and by hydrothermal synthesis,68 is ½[0![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif)
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ]. The difference between the α and β phases of V6O13 is clearly visible in Fig. 5: the double layer in β is like the slab in VO2(A) (Fig. 2). Worth noting is the fact that a pseudosymmetry search69 in β-V6O13 with respect to the minimal supergroups of Cmma results in the structure with space group Fmmm (Z = 4). This transformation does not involve any atomic displacements indicating that the reported space group symmetry Cmma67 is too low.
]. The difference between the α and β phases of V6O13 is clearly visible in Fig. 5: the double layer in β is like the slab in VO2(A) (Fig. 2). Worth noting is the fact that a pseudosymmetry search69 in β-V6O13 with respect to the minimal supergroups of Cmma results in the structure with space group Fmmm (Z = 4). This transformation does not involve any atomic displacements indicating that the reported space group symmetry Cmma67 is too low.
α-V6O13 is a paramagnetic metal at ambient conditions. It undergoes MIT at TMIT = 150 K but remains paramagnetic down to 55 K.70–75TMIT decreases to 100 K at 1.4 GPa.74 The low-temperature crystal structure is disputed as C2 (ref. 74) and P21/a76 space groups are initially proposed. The most recent study reports the structure in space group Pc.70 All these models exhibit a high degree of pseudosymmetry with respect to space group C2/m and distortions from the parent phase are very small. In Pc, all the atoms are displaced from the mirror plane, but the structural changes appear mainly in the single layer. The charge transfer between the vanadium atoms in the single and double layers takes place during this phase transition. Some of the vanadium atoms in the single layer acquire more pentavalent character, while the others develop the tetravalent one. The model in space group Pc is then used to explain the changes of various physical properties across MIT in all subsequent studies.71–74 At TN ≈ 55 K, α-V6O13 becomes antiferromagnetically ordered.74 However, the crystal and magnetic structures below TN have not been reported yet.
At room temperature, α-V6O13 is structurally stable to at least 9.4 GPa.37 Single-crystals of rutile-related V0.92O2 can be grown at 10 GPa and 1273 K from a polycrystalline α-V6O13 starting material.65In situ synchrotron measurements reveal that this new phase starts to crystallize above 500 K in the pressure range 4–17.5 GPa and is recovered to ambient conditions. The characteristic feature of its crystal structure (C2/m, Z = 4) is the presence of disorder affecting the V atoms, which occupy two split atom positions V1 and V2 (Fig. 6). The V1 atoms in one of the octahedral chains are displaced along the b axis, while the V2 atoms in the other are four-fold split in the (b, c) plane. This results in two zigzag V–V chains: one with equidistant V1 atoms and the other with short and long V2–V2 distances. There is no indication for V4+ and V5+ charge ordering and separation. Disregarding the V split positions, the average structure (P2/m, Z = 2) of this new phase is like the one for the V1−zO2 material obtained by Chamberland.33,34 Pseudo-symmetry considerations69 indicate that rutile-related V0.92O2 is a variant of insulating M2 (Fig. 1).65
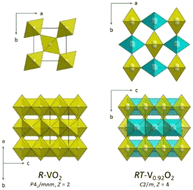 | ||
| Fig. 6 Comparison of the ideal rutile and V0.92O2 crystal structures. The V1O6 and V2O6 octahedra are drawn yellow and cyan, respectively. | ||
V0.92O2 is metastable when recovered to ambient conditions. It starts to decompose at 470 K and atmospheric pressure.77 At about 350 K, the material undergoes a first-order phase transition, in which the low- and high-temperature phases have the same average structure (P2/m, Z = 2). The temperature for this transformation correlates well with those in V0.995O2 and V0.976O2.33 V0.92O2 above the phase transition forms a modulated structure in super-space group X2/m(0β0)s0 (Z = 2) with the centering vector X(½, 0, ½, ½). The structure is remarkable as it is mainly the modulation of the displacement parameters of the vanadium atoms that dominate the incommensurate phase. Both structures of V0.92O2 have one important feature common to all the known (non-)stoichiometric VO2 phases: the hexagonal close-packing oxygen sublattice is rigid while the cation sublattice is flexible, allowing for various schemes of cation (dis)order.
The first-order phase transition to the incommensurate phase in V0.92O2 is also visible in the temperature-dependent resistivity data measured on a single crystal on heating and cooling in the range 275–400 K.77 On heating, the onset of the phase transition is at about 330 K. A hysteresis of about 10 K is observed on cooling the crystal from 400 K down to room temperature. Both phases of V0.92O2 are insulating. Such a behaviour is different from that of (nearly) stoichiometric VO2, in which the high-temperature phases are metallic.16,32
On compression at room temperature, V0.92O2 reaches the ideal rutile structure at about 5.0 GPa.77 This transformation is also of the first-order character. It demonstrates that the structural behavior of the cation deficient vanadium dioxide at extreme conditions is distinctly different from that of stoichiometric VO2.26–31
Conclusions and outlook
Ribbons of edge-sharing VO6 octahedral doublets could be traced in the crystal structures along the Wadsley series at atmospheric pressure. They are a basic building unit in idealized α-V2O5, VO2(B), and related VO2(A). The connectivity of the rutile type, with chains of edge-sharing octahedra joined with each other by corners, can be found in the materials synthesized at high pressures. It is partially realized in δ-V2O5 (ref. 37–40) and fully achieved in V0.92O2 (ref. 65 and 77) obtained from V6O13. This could imply that vanadium oxides with the Wadsley stoichiometries VnO2n+1 and high VO2 content (at least n ≥ 6) would tend to transform to the rutile type at extreme conditions. The crystal structures of the Magnéli phases at ambient are also derived from the rutile one. The rutile structure would then be an aristotype for both cation and anion deficient vanadium dioxides at various pressures and temperatures. The rigidity of the hexagonal close-packing oxygen sublattice and different occupational schemes of the V3+, V4+, and V5+ cations could also result in the occurrence of incommensurate phases in non-stoichiometric VO2.Metallic properties in all the rutile-type as well as Wadsley phases are observed in the compounds with no V–V dimerization along the octahedral chains (see also Tables S1 and S2 in the SI). The application of pressure in the synthesis could yield materials with shorter V–V distances but would not necessarily break the dimers. Based on the current data available for the V2O5–VO2 system, it is not possible to establish any relation of the crystal structures and properties with the synthesis conditions without considering the compositional variations. V0.92O2, which is insulating, contains 35 wt% of V2O5.65,77 It demonstrates the capacity of the rutile-type framework to accommodate a wide range of VO2–V2O5 compositions. Varying stoichiometries and pressure–temperature conditions could then yield V1−zO2 materials with transport properties ranging from metallic to insulating. Since the compound with 10 wt% of V2O5 (V0.976O2) is indeed metallic,33 it remains to be seen for which higher V2O5 contents V1−zO2 oxides would become insulating. In addition, 35 wt% of V2O5 does not need to be a compositional limit for the stability of the rutile-related structures. We conclude then that materials engineering by varying compositions in the system V2O5–VO2 and exerting high pressures and temperatures offers a possibility to synthesize new materials with unexpected properties.
Previous studies show that TMIT decreases in the anion-deficient VO2−y, while it increases in the cation-deficient V1−zO2.14,16,33 For small deviations from the exact VO2 stoichiometry, the high-temperature phase is supposed to have the ideal rutile structure. However, this has not been clearly demonstrated either for the compounds synthesized at elevated pressures reported in ref. 33 nor for several materials, which can already be obtained at atmospheric conditions.16 Further detailed investigations of the structural and electronic (in)stabilities in the non-stoichiometric vanadium dioxides are then warranted as the occurrence of the ideal R and metallic phases in the V1−zO2 materials is not demonstrated.
Author contributions
Both authors contributed equally to this work.Conflicts of interest
No conflicts of interest.Data availability
All data discussed in the manuscript are available upon request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ce01171j.
Acknowledgements
This work is supported by the Helmholtz InnoPool Project MATHIPE (MATerials under HIgh PrEssure).Notes and references
- H. A. Wriedt, Bull. Alloy Phase Diagrams, 1989, 10, 271–277 CrossRef CAS.
- P. Hu, P. Hu, T. D. Vu, M. Li, S. Wang, Y. Ke, X. Zeng, L. Mai and Y. Long, Chem. Rev., 2023, 123, 4353–4415 CrossRef CAS PubMed.
- M. Li, S. Magdassi, Y. F. Gao and Y. Long, Small, 2017, 13, 1701147 CrossRef PubMed.
- O. Monfort and P. Petrisková, Processes, 2021, 9, 214 CrossRef CAS.
- N. Vishwakarma, A. A. Remadevi, D. Kumar, A. Solanki, A. S. Rana and A. Srivastava, J. Appl. Phys., 2024, 135, 025303 CrossRef CAS.
- U. Schwingenschlögl and V. Eyert, Ann. Phys., 2004, 13, 475–510 CrossRef.
- J. M. Allred and R. J. Cava, J. Solid State Chem., 2013, 198, 10–17 CrossRef CAS.
- H. Katzke, P. Tolédano and W. Depmeier, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 68, 024109 CrossRef.
- S. Åsbrink, Acta Crystallogr., Sect. B, 1980, 36, 1332–1339 CrossRef.
- S.-H. Hong and S. Åsbrink, Acta Crystallogr., Sect. B, 1982, 38, 713–719 CrossRef.
- X. Wu, H. Wang and Y. Wang, Materials, 2023, 16, 6874 CrossRef CAS PubMed.
- A. D. Wadsley, Acta Crystallogr., 1957, 10, 261–267 CrossRef CAS.
- P. Y. Zavalij and M. S. Whittigham, Acta Crystallogr., Sect. B: Struct. Sci., 1999, 55, 627–663 CrossRef PubMed.
- J.-P. Pouget, C. R. Phys., 2021, 22, 37–87 CrossRef CAS.
- Y. Xue and S. Yin, Nanoscale, 2022, 14, 1054–1097 RSC.
- S. Joshi, N. Smieszek and V. Chakrapani, Sci. Rep., 2023, 10, 17121 CrossRef PubMed.
- H. Sim, K.-Y. Doh, Y. Park, K. Song, G.-Y. Kim, J. Son, D. Lee and S.-Y. Choi, Small, 2024, 20, 2402260 CrossRef CAS PubMed.
- J. M. Longo and P. Kierkegaard, Acta Chem. Scand., 1970, 24, 420–426 CrossRef CAS.
- M. Marezio, D. B. McWhan, J. P. Remeika and P. D. Dernier, Phys. Rev. B: Solid State, 1972, 5, 2541–2551 CrossRef.
- M. Ghedira, H. Vincent, M. Marezio and J. C. Launay, J. Solid State Chem., 1977, 22, 423–438 CrossRef CAS.
- M. Liu, S. Xie, L. Wei, M. Galluzzi, Y. Li, Q. Wang, X. Zhou, Y. Wang and J. Li, Acta Mater., 2020, 195, 720–727 CrossRef CAS.
- Y. Bleu, F. Bourquard, K. Misdanitis, A. Poulet, A.-S. Loir, F. Garrelie and C. Donnet, Mater. Today Commun., 2023, 35, 105564 CrossRef CAS.
- D. K. Manousou, S. B. Atata, Y. J. Sohn, P. Tsipas, A. Grzechnik, M. Calamiotou, K. Friese and S. Gardelis, J. Alloys Compd., 2025, 1024, 18081 CrossRef.
- P. Lu, J. Zhou, X. Liu, Z. Zhang, F. Xu, L. Zhang, X. Mou, J. Feng, Y. Gao and J. Zhao, J. Appl. Crystallogr., 2014, 47, 732–738 CrossRef CAS.
- Z. Shao, X. Cao, H. Luo and P. Jin, NPG Asia Mater., 2018, 10, 581–605 CrossRef.
- P. Bouvier, L. Bussmann, D. Machon, I. Breslavetz, G. Garbarino, P. Strobel and V. Dmitriev, Phys. Rev. B, 2023, 108, 144106 CrossRef CAS.
- Y. Chen, S. Zhang, F. Ke, C. Ko, S. Lee, K. Liu, B. Chen, J. W. Ager, R. Jeanloz, V. Eyert and J. Wu, Nano Lett., 2017, 17, 2512–2516 CrossRef CAS PubMed.
- L. Bai, Q. Li, S. A. Corr, Y. Meng, C. Park, S. V. Sinogeikin, C. Ko, J. Wu and G. Shen, Phys. Rev. B: Condens. Matter Mater. Phys., 2015, 91, 104110 CrossRef.
- H. Zhang, Q. Li, B. Cheng, Z. Guan, R. Liu, B. Liu, Z. Liu, X. Li, T. Cui and B. Liu, RSC Adv., 2016, 6, 104949–104954 RSC.
- Q. Li, H. Zhang, C. Lin, F. Tian, J. S. Smith, C. Park, B. Liu and G. Shen, J. Alloys Compd., 2017, 709, 260–266 CrossRef CAS.
- H. Zhang, Q. Li, F. Wang, R. Liu, Y. Mao, Z. Liu, X. Li, K. Yang, T. Cui and B. Liu, J. Phys. Chem. C, 2019, 123, 955–962 CrossRef CAS.
- I. P. Zibrov, V. P. Filonenko, V. A. Sidorov, N. M. Chtchelkatchev and M. V. Magnitskaya, Materialia, 2022, 23, 101456 CrossRef CAS.
- B. L. Chamberland, J. Solid State Chem., 1973, 7, 377–384 CrossRef CAS.
- J. Galy and G. Miehe, Solid State Sci., 1999, 1, 433–448 CrossRef CAS.
- R. Enjalbert and J. Galy, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1986, 42, 1467–1469 CrossRef.
- A. Grzechnik, Chem. Mater., 1998, 10, 2505–2509 CrossRef CAS.
- B. V. Hakala, D. K. Manousou, K. Glazyrin, W. A. Crichton, K. Friese and A. Grzechnik, J. Alloys Compd., 2022, 911, 164966 CrossRef CAS.
- V. P. Filonenko, M. Sundberg, P. E. Werner and I. P. Zibrov, Acta Crystallogr., Sect. B: Struct. Sci., 2004, 60, 375–381 CrossRef CAS PubMed.
- P. Balog, D. Orosel, Z. Cancarevic, C. Schön and M. Jansen, J. Alloys Compd., 2007, 429, 87–98 CrossRef CAS.
- I. P. Zibrov, V. P. Filonenko, S. G. Lyapin and V. A. Sidorov, High Pressure Res., 2013, 33, 399–408 CrossRef CAS.
- F. Théobald, R. Cabala and J. Bernard, J. Solid State Chem., 1976, 17, 431–438 CrossRef.
- Y. Oka, T. Yao, N. Yamamoto, Y. Ueda and A. Hayashi, J. Solid State Chem., 1993, 105, 271–278 CrossRef CAS.
- S. A. Corr, M. Grossman, Y. Shi, K. R. Heier, G. D. Stucky and R. Seshadri, J. Mater. Chem., 2009, 19, 4362–4367 RSC.
- J. Lourembam, A. Srivastava, C. La-o-vorakiat, H. Rotella, T. Venkatesan and E. E. M. Chia, Sci. Rep., 2015, 5, 9182 CrossRef CAS PubMed.
- S. R. Popuri, A. Artemenko, R. Decourt, M. Josse, U. C. Chung, D. Michau, M. Maglione, A. Villesuzanne and M. Pollet, J. Phys. Chem. C, 2015, 119, 25085–25092 CrossRef CAS.
- J. Lourembam, A. Srivastava, C. La-o-vorakiat, L. Cheng, T. Venkatesan and E. E. M. Chia, Sci. Rep., 2016, 6, 25538 CrossRef CAS PubMed.
- S. R. Popuri, M. Miclau, A. Artemenko, C. Labrugere, A. Villesuzanne and M. Pollet, Inorg. Chem., 2013, 52, 4780–4785 CrossRef CAS PubMed.
- Z. Ma, S. Yan, Z. Liu, T. Xu, F. Chen, S. Chen, T. Cao, L. Sun, B. Chen, S.-J. Liang and F. Miao, Chin. Phys. B, 2024, 33, 067103 CrossRef CAS.
- Y. Oka, T. Yao and N. Yamamoto, J. Solid State Chem., 1990, 86, 116–124 CrossRef CAS.
- Y. Oka, T. Yao and N. Yamamoto, J. Mater. Chem., 1991, 1, 815–818 RSC.
- Y. Oka, S. Sato, T. Yao and N. Yamamoto, J. Solid State Chem., 1998, 141, 594–598 CrossRef CAS.
- S. Zhang, B. Shang, J. Yang, W. Yan, S. Wei and Y. Xie, Phys. Chem. Chem. Phys., 2011, 13, 15873–15881 RSC.
- Y. Wang, J. Zhu, W. Yang, T. Wen, M. Pravica, Z. Liu, M. Hou, Y. Fei, L. Kang, Z. Lin, C. Jin and Y. Zhao, Nat. Commun., 2016, 7, 12214 CrossRef CAS PubMed.
- B. Cheng, H. Zhang, Q. Li, J. Liu and B. Liu, Inorganics, 2022, 10, 122 CrossRef CAS.
- B. Cheng, Q. Li, H. Zhang, R. Liu, B. Liu, Z. Yao, T. Cui, J. Liu, Z. Liu, B. Sundqvist and B. Liu, Phys. Rev. B, 2016, 93, 184109 CrossRef.
- K.-A. Wilhelmi and K. Waltersson, Acta Chem. Scand., 1970, 24, 3409–3411 CrossRef CAS.
- G. Grymonprez, L. Fiermans and J. Vennik, Acta Crystallogr., Sect. A, 1977, 33, 834–837 CrossRef.
- A. Heidemann, K. Kosuge, Y. Ueda and S. Kachi, Phys. Status Solidi A, 1977, 39, K37 CrossRef CAS.
- H. Nishihara, Y. Ueda, K. Kosuge, H. Yasuoka and S. Kachi, J. Phys. Soc. Jpn., 1979, 47, 790 CrossRef CAS.
- K. Waltersson, B. Forslund and K.-A. Wilhelmi, Acta Crystallogr., Sect. B, 1974, 30, 2644–2652 CrossRef.
- I. P. Zibrov, V. P. Filonenko, V. A. Sidorov and S. G. Lyapin, Inorg. Mater., 2016, 52, 902–908 CrossRef CAS.
- S. Yamazaki, C. Li, K. Ohoyama, M. Nishi, M. Ichikara, H. Ueda and Y. Ueda, J. Solid State Chem., 2010, 183, 1496–1503 CrossRef CAS.
- K. Kawashima, Y. Ueda, K. Kosuge and S. Kachi, J. Cryst. Growth, 1974, 26, 321–322 CrossRef CAS.
- Y. Ueda, K. Kosuge and S. Kachi, Mater. Res. Bull., 1976, 11, 293–298 CrossRef CAS.
- A. Grzechnik, B. V. Hakala, S. Kurig, N. Walte, N. Tsujino, S. Kakizawa, Y. Higo, D. Zagorac, J. Zagorac, R. Dronskowski, J. C. Schön and K. Friese, Cryst. Growth Des., 2024, 24, 5582–5592 CrossRef CAS.
- K.-A. Wilhelmi, K. Waltersson and L. Kihlborg, Acta Chem. Scand., 1971, 25, 2675–2687 CrossRef CAS.
- T. Ohno, Y. Nakamura and S. Nagakura, J. Solid State Chem., 1985, 56, 318–324 CrossRef CAS.
- Y. Zhang and C. Meng, Mater. Lett., 2015, 160, 404–407 CrossRef CAS.
- C. Capillas, E. S. Tasci, G. de la Flor, D. Orobengoa, J. M. Perez-Mato and M. I. Aroyo, Z. Kristallogr., 2011, 226, 186–196 CrossRef CAS.
- J. Höwing, T. Gustafsson and J. O. Thomas, Acta Crystallogr., Sect. B: Struct. Sci., 2003, 59, 747–752 CrossRef PubMed.
- Y. Shimizu, S. Aoyama, T. Jinno, M. Itoh and Y. Ueda, Phys. Rev. Lett., 2015, 114, 166403 CrossRef PubMed.
- M. Onoda, T. Ohki and Y. Uchida, J. Phys.: Condens. Matter, 2004, 16, 7863–7871 CrossRef CAS.
- T. Toriyama, T. Nakayama, T. Konishi and Y. Ohta, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 90, 085131 CrossRef CAS.
- I. V. Leonov, JETP Lett., 2022, 116, 884–890 CrossRef CAS.
- P. D. Dernier, Mater. Res. Bull., 1974, 9, 955–964 CrossRef CAS.
- I. Kawada, I. Motohiko, M. Saeki, N. Kimizuka, M. Nakano-Onoda and K. Kato, Acta Crystallogr., Sect. B, 1978, 34, 1037–1039 CrossRef.
- A. Grzechnik, V. Petříček, P. Reiss, P. Zakalek, D. Chernyshov and K. Friese, IUCrJ, 2026, 13, 116–125 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |