Open Access Article
Open Access ArticleNitrogen editing of aromatic rings: from skeletal editing to fragment editing
Hikaru
Nakahara
and
Junichiro
Yamaguchi
 *
*
Department of Applied Chemistry, Waseda University, 513 Wasedatsurumakicho, Shinjuku, Tokyo 162-0041, Japan. E-mail: junyamaguchi@waseda.jp
First published on 22nd April 2026
Abstract
Direct conversion of aromatic rings into heteroaromatic frameworks represents an attractive strategy for tuning molecular properties in pharmaceuticals and functional materials. In recent years, the concept of molecular editing has emerged as a powerful approach that enables such transformations without reconstructing the entire aromatic framework. In this Feature Article, we discuss two complementary strategies for nitrogen incorporation into aromatic systems based on different editing units: atom-level skeletal editing and aromatic-unit fragment editing. Skeletal editing modifies aromatic frameworks through single-atom transformations such as carbon-to-nitrogen transmutation and ring-expansion-type nitrogen insertion. In contrast, fragment editing reorganizes molecular structures through exchange of aromatic fragments enabled by reversible bond activation processes, including single-bond metathesis-type reactions, shuttle-type reactions, and heteroaromatic swapping reactions. By comparing these approaches from the perspective of molecular design, this review highlights how aromatic identities can be reorganized through distinct editing units and discusses future opportunities for fragment-level editing of heteroaromatic frameworks.
1. Introduction
Aromatic compounds are central structural motifs in molecular design and play essential roles in pharmaceuticals and functional materials. In particular, nitrogen-containing heteroaromatic rings are widely used in medicinal chemistry because incorporation of nitrogen atoms into an aromatic framework strongly influences electronic properties, polarity, hydrogen-bonding ability, and metabolic stability (Fig. 1A).1 Heteroaromatic systems are also important in materials science, where precise control of π-electron density, charge transport, and coordination ability is essential for the development of organic semiconductors and light-emitting materials.2 Replacing a carbon atom in benzene with nitrogen significantly alters the electronic distribution of the aromatic system, transforming a π-electron-rich arene into a more electron-deficient heteroaromatic ring. Increasing the number of nitrogen atoms further changes molecular properties such as lipophilicity and dipole moment. Even the simple conversion of benzene into pyridine can lead to pronounced differences in physicochemical behaviour and biological activity. In this sense, nitrogen incorporation into aromatic rings is not merely a substituent modification but a fundamental design element that redefines the electronic structure of the molecular framework.Traditionally, nitrogen-containing heteroaromatic compounds have been synthesized mainly through de novo construction of heteroaromatic frameworks using condensation reactions, cyclizations, or transition-metal-catalyzed annulations.3 Although these methods provide efficient access to diverse heterocycles, they generally require dedicated precursors and reconstruction of the aromatic framework from simpler building blocks. Cross-coupling reactions, on the other hand, are highly effective for the diversification of aromatic compounds but primarily modify peripheral substituents rather than the identity of the aromatic core itself. Consequently, the direct conversion of an existing arene into a heteroaromatic system while retaining its substitution pattern has historically been difficult.
In recent years, the concept of molecular editing, in which structural modifications are directly applied to existing molecules, has rapidly emerged in synthetic chemistry (Fig. 1B, left).4 Among these approaches, skeletal editing has attracted considerable attention as a strategy for reorganizing molecular frameworks through insertion, deletion, or replacement of single atoms. In aromatic systems, skeletal editing enables nitrogen incorporation through transformations that replace a carbon atom with nitrogen or embed nitrogen within an existing aromatic framework while preserving the overall ring topology. Such transformations allow the electronic properties of aromatic rings to be tuned at the atomic level while maintaining the original substitution pattern.
From another perspective, strategies distinct from single-atom editing have also emerged to alter the identity of aromatic rings at the fragment level. Recent developments in single-bond metathesis and shuttle catalysis,5 as well as heteroaromatic swapping reactions,6 enable the introduction of nitrogen-containing aromatic rings through the exchange of entire aromatic fragments. These fragment editing strategies differ fundamentally from single-atom transformations in that they operate at the level of aromatic units rather than individual atoms (Fig. 1B, right). As a result, fragment exchange often involves the reorganization of substitution patterns or bonding connectivity within the aromatic framework, providing greater flexibility in structural redesign. In this sense, fragment editing can be viewed as a complementary strategy to skeletal editing, enabling exploration of a broader molecular design space through the interchange of aromatic identities.
Thus, nitrogen incorporation into aromatic rings can be achieved through two conceptually distinct approaches based on different editing units. One is skeletal editing, in which the electronic structure of an aromatic framework is modified at the atomic level while preserving the overall molecular skeleton. The other is fragment editing, in which the identity of an aromatic fragment is exchanged to expand structural diversity. The former is characterized by minimal structural perturbation and high topological fidelity, whereas the latter often involves reorganization of substitution patterns or replacement of aromatic ring types, thereby enabling access to a broader design space. These two strategies are therefore complementary and should be applied according to the objectives of molecular design.
Conversely, skeletal editing can also proceed in the opposite direction. Numerous studies have demonstrated the removal of nitrogen atoms from heteroaromatic frameworks, reconstructing carbocyclic aromatic systems through denitrogenative skeletal editing or nitrogen extrusion reactions (Fig. 1C).7 These reactions are conceptually intriguing, as they reorganize aromatic frameworks through selective C–N bond cleavage. However, despite the increasing number of such studies, methods that directly introduce nitrogen atoms into existing aromatic rings remain comparatively limited. A similar trend is observed in fragment editing strategies. Although aromatic fragment exchange reactions have rapidly developed in recent years, most reported examples involve exchanges between carbocyclic arenes, whereas examples involving nitrogen-containing heteroarenes remain relatively rare. One possible reason is that these transformations often rely on transition-metal or photoredox catalysis, where coordination of heteroaromatic nitrogen atoms can lead to catalyst deactivation. While both skeletal and fragment editing enable powerful molecular reorganization, limitations remain. Although direct comparison is not always straightforward due to their distinct conceptual foundations, skeletal editing often requires activated substrates and faces selectivity challenges, whereas fragment editing relies on suitable functional handles or partners. Both strategies also show limitations in functional group compatibility and scalability, highlighting their complementary nature and the need for further development.
In this Feature Article, we present a complementary perspective to existing reviews on skeletal editing by organizing skeletal editing and fragment editing in parallel and re-examining the conversion of aromatic rings into heteroaromatic systems from the standpoint of molecular design. As a Feature Article, the present work aims to highlight emerging concepts from our perspective, including developments related to heteroaromatic swapping. In this context, we propose that heteroaromatic interconversion can be approached through two distinct yet complementary directions: atom-level skeletal editing, which enables direct C-to-N transmutation within an aromatic framework, and fragment-level editing, which enables exchange of entire aromatic units. Although many current fragment editing reactions do not explicitly introduce nitrogen atoms, they establish key mechanistic principles for heteroaromatic interconversion through reversible bond reorganization. Rather than providing an exhaustive survey of reaction mechanisms or historical developments, this article focuses on conceptual frameworks for nitrogen incorporation into aromatic rings.
2. Skeletal editing
2.1. From arenes to heteroarenes
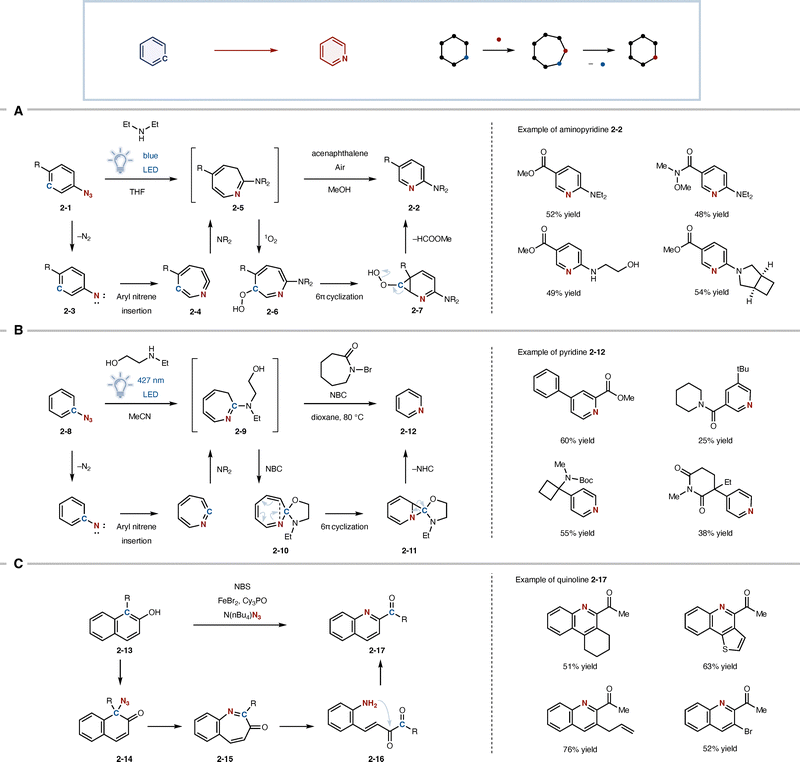
Skeletal editing provides a direct strategy for introducing nitrogen atoms into aromatic rings through atom-level modification of the molecular framework. Early attempts to achieve such transformations can be traced back to studies on the photolysis of aryl azides. In this context, “nitrogen scanning” refers to a molecular editing strategy in which a carbon atom at a defined position within an aromatic framework is selectively replaced by nitrogen, enabling systematic exploration of positional effects on molecular properties.In the 1970s, Sundberg and co-workers reported that photolysis of phenyl azides 2-1 in amine solvents produced small amounts of 2-aminopyridine derivatives 2-2 among complex mixtures of products.8 This transformation was proposed to proceed through the generation of an aryl nitrene 2-3, followed by nitrogen incorporation into the aromatic framework and formation of a seven-membered azepine intermediate 2-4, which subsequently undergoes carbon extrusion to afford a pyridine skeleton. Although the reaction proceeds in low yield and generates multiple pyridine isomers together with various side products, it is historically significant because it demonstrated that skeletal conversion from benzene to pyridine is, in principle, possible.
A significant advance toward practical skeletal editing was reported by Burns and co-workers in 2022 (Fig. 2A).9 In their study, attention was focused on the 3H-azepine intermediate 2-5 generated from aryl azides 2-1. By coupling this intermediate with oxidative carbon extrusion process mediated by singlet oxygen, the authors achieved the conversion of arenes into 2-aminopyridines 2-2via2-5 to 2-7. A key feature of this strategy is the exploitation of the photosensitizing properties of the azepine intermediate 2-5, allowing the reaction to proceed under carefully designed photoirradiation conditions in the presence of molecular oxygen. Importantly, the development and widespread use of LED light sources has greatly facilitated such photochemical transformations, providing practical and tunable irradiation conditions compared with traditional high-pressure mercury lamps. In this transformation, one carbon atom of the benzene ring is formally extruded, while the nitrogen atom remains embedded in the ring, leading to reconstruction of a pyridine framework.
Building on these developments, a more design-oriented approach was introduced by Levin and co-workers in 2023 (Fig. 2B).10 In this work, the authors explicitly aimed to implement nitrogen scanning as a practical molecular editing strategy and achieved ipso-selective nitrene internalization, in which the site of azide installation directly determines the position of nitrogen incorporation. Mechanistically, the transformation proceeds through an azepine intermediate 2-9 generated from the aryl azide 2-8, followed by a distinctive rearrangement involving spirocyclization and carbon extrusion (2-10 and 2-11). A key role is played by N-bromocaprolactam (NBC), which promotes the formation and controlled rearrangement of the azepine intermediate 2-9, thereby enabling selective ipso carbon extrusion. This sequence selectively removes the ipso carbon atom while internalizing the nitrogen atom at the same position. As a result, a specific carbon atom in an aromatic ring can be selectively converted into nitrogen, enabling the formation of a single pyridine isomer 2-12 while avoiding mixtures of regioisomers. In this design, control over the site of carbon extrusion is central to the reaction concept, representing not merely nitrogen insertion but a site-selective carbon-to-nitrogen transmutation of aromatic rings.
Wei and co-workers reported a related skeletal editing strategy using arenol scaffolds rather than aryl azides (Fig. 2C).11,12 In this approach, dearomative azidation of arenol 2-13 first generates an azide intermediate 2-14, which undergoes ring expansion (2-15) followed by ring opening to form aniline intermediate 2-16. Subsequent cyclization and rearomatization ultimately furnish quinoline derivative 2-17, corresponding to a formal C-to-N transmutation of the aromatic framework. This transformation proceeds through a ring expansion–contraction pathway, in which the aromatic framework is temporarily dearomatized to enable skeletal reorganization. Notably, this strategy is applicable not only to simple arenes but also to polycyclic aromatic systems, highlighting the potential of dearomatization-based skeletal editing for the modification of more complex aromatic architectures. Conceptually, this approach differs from aryl-azide-based nitrene internalization strategies in that it temporarily disrupts aromatic stability to enable skeletal reorganization.
Although not discussed in detail in this Review, Wang and Luan and their co-workers have also reported a related transformation in which nitrogen incorporation into naphthols triggers ring expansion and ring opening to afford isoquinoline derivatives.13 Using a catalytic system based on AgOTf and PhI![[quadruple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e003.gif) NTs, the reaction outcome can be tuned simply by modifying the reaction conditions, enabling access to different products. Although this strategy differs mechanistically from nitrene internalization pathways that proceed through aryl azide intermediates, both approaches share a common conceptual feature in that control of aromatic stability plays a central role in enabling skeletal editing. These studies collectively highlight the expanding design space for nitrogen-incorporating skeletal editing of aromatic frameworks.
NTs, the reaction outcome can be tuned simply by modifying the reaction conditions, enabling access to different products. Although this strategy differs mechanistically from nitrene internalization pathways that proceed through aryl azide intermediates, both approaches share a common conceptual feature in that control of aromatic stability plays a central role in enabling skeletal editing. These studies collectively highlight the expanding design space for nitrogen-incorporating skeletal editing of aromatic frameworks.
Taken together, these studies illustrate that conversion of benzene into pyridine is not merely a single reaction example but rather the outcome of optimizing multiple design factors, including control over the carbon extrusion site, modes of nitrogen internalization, and preservation of skeletal topology. From the early demonstration of feasibility by Sundberg, through the oxygen-driven carbon extrusion reported by Burns, the establishment of ipso-selective nitrogen incorporation by Levin, and the ring expansion–contraction transmutation strategy developed by Wei, the concept of nitrogen incorporation into aromatic rings has gradually evolved through successive refinements. Although these approaches all fall within the category of skeletal editing, in which the identity of an aromatic ring is redefined at the single-atom level, they employ diverse strategies such as site-selective carbon extrusion and intermediate design. Together, these advances highlight the rapidly expanding design space for nitrogen incorporation into aromatic frameworks.
2.2. From heteroarenes to heteroarenes (C-to-N transmutation)
In addition to the conversion of arenes into heteroarenes, another emerging concept is C-to-N transmutation within existing heteroaromatic frameworks. In this strategy, a carbon atom embedded in a heteroaromatic ring is selectively replaced by a nitrogen atom. This transformation represents a form of single-atom skeletal editing, in which part of the carbon framework is replaced by nitrogen. However, a distinctive feature of this approach is that the starting materials are already nitrogen-containing heteroarenes. As a result, the transformation can be viewed not simply as heteroatom incorporation but rather as interconversion between different heteroaromatic frameworks, enabling more precise tuning of electronic properties and polarity distributions within aromatic systems.A representative example illustrating this concept was reported by Levin and co-workers in 2023, demonstrating a C-to-N transmutation that converts quinoline into quinazoline (Fig. 3A).14 Upon irradiation at 390 nm, quinoline N-oxide 3-1 undergoes a rearrangement to generate a benzoxazepine intermediate 3-2. Subsequent ozone oxidation in the presence of pyridine converts this intermediate into an imidic anhydride 3-4, while pyridine serves to reduce the transiently formed ozonide species 3-3. The resulting intermediate 3-4 then undergoes recyclization with an ammonia-derived nitrogen nucleophile, ultimately furnishing the quinazoline framework 3-5. In this transformation, carbon extrusion and nitrogen incorporation occur at the same position, such that the C3 carbon atom of quinoline is replaced by nitrogen without significantly altering the overall skeletal topology of the heteroaromatic system.
Studer and co-workers reported a C-to-N swapping strategy starting from indole and benzofuran frameworks 3-6, enabling divergent access to multiple heteroaromatic scaffolds such as indazole, benzimidazole, benzisoxazole, and benzoxazole (Fig. 3B).15 A key feature of this transformation is the use of N-nitrosomorpholine (3-7), which undergoes photolytic N–N bond cleavage to generate a morpholinium radical cation together with a nitroso radical. The morpholinium radical cation first reacts with the substrate at the C2 position, generating a radical at C3 (3-8) that subsequently recombines with the nitroso radical to afford nitroso intermediate 3-9. Driven by aminal ring opening, cleavage of the C2–C3 bond then generates an oxime-like intermediate 3-10, which undergoes rearrangement and re-cyclization to construct new nitrogen-containing aromatic frameworks. Notably, the single intermediate 3-10 can diverge into different products depending on the reaction conditions: benzisoxazole (benzimidazole) 3-11 is obtained under PPh3/DEAD conditions, whereas benzoxazole (benzimidazole) 3-12 is formed under MsCl/NEt3 conditions. Around the same time, Morandi and co-workers independently reported a chemodivergent C-to-N swapping of benzofuran, in which an oxime intermediate formed after photooxidative cleavage can be selectively converted into benzoxazole or benzisoxazole depending on the reaction conditions.16 Thus, simple changes in reaction conditions enable divergent product formation, demonstrating that C-to-N transmutation can serve not only as a one-to-one atomic substitution but also as an entry point to skeletal diversification of heteroaromatic systems.
In 2025, a direct conversion of pyridines into pyridazines was reported (Fig. 3C).17 In this study, 2-chloropyridine (3-13) was first transformed into a 2-azido-N-aminopyridinium intermediate (3-15) through sequential amination and azidation steps. The amination reagent bears a Boc-protected amino group, which undergoes Boc deprotection under TFA conditions, allowing the resulting NH2+ species to act as the aminating unit. Subsequent treatment of 3-14 with NaN3/PPTS affords intermediate 3-15, where PPTS serves as a buffering agent because the intermediate is sensitive to strongly basic conditions. Upon photoirradiation at 390 nm, 3-15 undergoes a rearrangement that replaces a carbon atom in the pyridine ring with nitrogen, providing access to unsymmetrical pyridazine 3-16 frameworks that are otherwise difficult to synthesize. Although pyridine and pyridazine differ by only a single atom, their conventional synthetic strategies are fundamentally distinct. This transformation therefore demonstrates that existing pyridine frameworks can be edited into pyridazines, offering a molecular editing route that complements traditional de novo heterocycle synthesis.
Hong and co-workers also reported an alternative skeletal editing strategy for C-to-N transmutation, in which pyridines are converted into pyridazines via N-amination and oxidative ring remodeling through a diazatriene intermediate.18 Although not discussed in detail here, this study further highlights the expanding scope of heteroaromatic skeletal editing.
Taken together, these studies highlight a common conceptual theme: the selective replacement of a carbon atom within an existing heteroaromatic framework by nitrogen to generate a new heteroaromatic ring. Similar to arene-to-heteroarene conversion, C-to-N transmutation represents a form of single-atom skeletal editing, in which the molecular framework is modified at the atomic level. The principal difference lies in the nature of the starting scaffold, and consequently in the reaction design and mechanistic strategies employed. At the same time, transformations within heteroaromatic systems introduce additional considerations, including electronic interactions with pre-existing heteroatoms and the preservation of aromatic stabilization. As a result, C-to-N transmutation provides a distinct molecular editing strategy that enables modulation of the electronic structure of heteroaromatic frameworks without extensive reconstruction of the molecular skeleton.
2.3. From heteroarenes to heteroarenes (ring expansion)
While C-to-N transmutation directly replaces a carbon atom within an existing aromatic framework with nitrogen, thereby redefining the electronic structure and molecular polarity of the ring, another emerging concept is ring-expansion-type nitrogen insertion.19 In this strategy, nitrogen incorporation occurs concomitantly with an increase in ring size. In contrast to simple single-atom substitution, this transformation proceeds through cleavage and reorganization of carbon–carbon bonds, followed by formation of new N–C bonds, ultimately reconstructing the molecular framework itself. In this sense, ring-expansion type nitrogen insertion can be viewed as a complementary skeletal editing strategy, in which the identity of the aromatic framework is redefined through changes in ring topology rather than simple atomic substitution.A representative example was reported by Morandi and co-workers in 2022 (Fig. 4).20 Using an electrophilic iodinitrene species generated in situ from ammonium carbamate and a hypervalent iodine reagent, they achieved single-atom insertion into TBS-protected indoles 4-1, affording quinazoline or quinoxaline derivatives 4-3. The reaction proceeds through a stepwise (2+1) cycloaddition to generate an aziridine-like intermediate 4-2, followed by iodobenzene elimination and rearomatization, ultimately resulting in ring expansion of the heteroaromatic framework. Importantly, the TBS group acts as a reversible and controllable protecting group, suppressing undesired overreaction at the indole nitrogen while enabling efficient release of the final products.
 | ||
| Fig. 4 Ring-expansion nitrogen insertion for heteroarene-to-heteroarene skeletal editing (Morandi, Sharma, and Ackerman). | ||
In the following year, the same group reported a related protocol employing LiHMDS as the nitrogen source, enabling the direct conversion of unprotected indoles and pyrroles into quinazoline and pyrimidine frameworks.21 In this system, LiHMDS functions both as a base and as a nitrogen donor, eliminating the need for prior protection of the indole nitrogen and improving the overall synthetic practicality of the transformation.
Related approaches have also been developed by other groups. Sharma and co-workers reported a ring-expansion nitrogen insertion using sulfenyl nitrene species, enabling the conversion of pyrroles, indoles, and imidazoles into pyrimidines, quinazolines, and triazines, respectively.22 In addition, Ackermann and co-workers achieved nitrogen insertion into indole frameworks through an electrochemical strategy driven by the oxygen reduction reaction (ORR).23
Taken together, these studies demonstrate that ring-expansion-type nitrogen insertion provides a powerful complement to C-to-N transmutation, enabling the conversion of heteroarenes into new heteroaromatic frameworks through topology-altering skeletal editing.
3. Fragment editing
This section discusses fragment-level approaches as a complementary direction, providing a conceptual framework for reprogramming aromatic identity at the level of molecular fragments.3.1. Single-bond metathesis
In contrast to skeletal editing, which modifies molecular frameworks through atom-level transformations, fragment editing operates at the level of molecular fragments, enabling the exchange of entire aromatic units between molecules. Unlike conventional cross-coupling reactions that modify substituents around an aromatic scaffold, fragment editing alters the identity of the aromatic unit itself. A central conceptual foundation of fragment editing is single-bond metathesis, in which σ bonds between two substrates are reversibly cleaved and reformed to exchange functional groups or aromatic fragments.Unlike cross-coupling reactions, where bond formation proceeds directionally through the combination of a nucleophile and an electrophile, single-bond metathesis involves oxidative addition of two σ bonds to a metal catalyst, followed by fragment exchange at the metal center and subsequent reductive elimination to furnish the exchanged products.24 For this strategy to operate effectively, several conditions must be satisfied: (i) the catalyst must be capable of reversible oxidative addition to two different σ bonds; (ii) crossover between the resulting oxidative addition complexes must be possible; and (iii) an appropriate thermodynamic driving force must be designed to direct the equilibrium toward the desired products. In addition, when nitrogen-containing heteroarenes are involved, the catalyst must tolerate their coordinating ability to avoid catalyst deactivation.
A representative example that clearly illustrates this concept is the functional group metathesis between aryl thioethers 16 and aryl nitriles 17 reported by Morandi and co-workers (Fig. 5A).25 The Ni(cod)2/dcype (1,2-Bis(dicyclohexylphosphino)ethane) catalytic system is capable of oxidative addition to both C(sp2)–S from 5-1 and C(sp2)–CN bonds from 5-2, allowing Ar–Ni–SR 5-3 and Ar–Ni–CN 5-4 intermediates to coexist at the catalytic center. Subsequent aryl exchange followed by reductive elimination from 5-7 and 5-8 releases the metathesis products 5-5 and 5-6. This transformation is intrinsically reversible and operates under thermodynamic control. By tuning the electronic properties of the nitrile “donor”, the authors designed the exchanged combination to be thermodynamically favorable, thereby directing the equilibrium toward the desired products. In this sense, the key principle of the method lies in accepting equilibrium as an inherent feature and controlling its position through electronic design. Although the reaction mainly involves exchange between aromatic rings, several nitrogen-containing heteroaromatic substrates are also tolerated, demonstrating that the catalytic system can operate without deactivation by coordinating heteroarenes.
In contrast to Morandi's strategy, which controls the equilibrium position through electronic design, Yamaguchi and co-workers reported around the same time an exchange reaction based on a funneling strategy that exploits the intrinsic reactivity of the products (Fig. 5B).26 Using a Ni/dcypt (3,4-bis(dicyclohexylphosphino)thiophene) catalytic system,27 functional group exchange was realized between pyridyl sulfide 5-7 and aromatic ester 5-8. In this transformation, the C–S bond of 5-7 and the C–O bond of 5-8 each undergo oxidative addition to nickel, generating Ar–Ni–SR 5-9 and Ar–Ni–OAr 5-10 intermediates that subsequently undergo ligand exchange at the metal center. The metathesis products are then released through reductive elimination.
A key feature of this system is that reductive elimination at the C2 position of the pyridine ring (5-14) proceeds faster than other possible pathways,28 effectively shifting the equilibrium toward the product side. In this sense, the reaction operates through a funneling mechanism, in which the products are selectively accumulated. As a result, the reaction furnishes thiosulfide 5-11 and pyridyl ether 5-12. Although the process is therefore not a perfectly symmetrical exchange reaction, it proceeds efficiently regardless of the electronic nature of the aromatic ester substrate 5-8.
In contrast, Xie and co-workers reported a related thioester transfer reaction in 2022 using a Ni/4,7-dimethyl-1,10-phenanthroline catalytic system (Fig. 5C).29 In this transformation, the C(acyl)–S bond of an aromatic thioester 5-15 and the C–Br bond of an aryl halide 5-16 are activated, leading to the formation of an aryl-substituted thioester 5-17. A distinctive feature of this system is that the aryl thioester 5-15 bearing a strongly electron-deficient aromatic ring, typically substituted with a CF3 group, undergoes rapid oxidative addition of the C(acyl)–S bond followed by decarbonylation. These steps proceed essentially irreversibly and therefore serve as the driving force of the reaction, allowing the transformation to proceed in an apparently unidirectional manner despite its formal equilibrium nature.
It should be noted that, although not discussed in detail here because the focus of this section is on arene-to-heteroarene exchange reactions, a related transformation using esters instead of thioesters was also reported by Yamaguchi and co-workers.30
In 2024, Yamaguchi and Yatabe reported an exchange reaction of diaryl thioethers (C–S bond metathesis) using supported Au–Pd alloy nanoparticles (Fig. 5D).31 The authors had previously reported a similar transformation using a Pd/PCy3 catalyst system and proposed that nanoparticles were the active species, although the possibility of a homogeneous catalytic pathway could not be excluded.32 In the present study, they demonstrated that the reaction proceeds under heterogeneous conditions using supported Au–Pd alloy nanoparticles. Because both diaryl thioethers 5-18 and 5-19, as well as the product 5-20, possess very similar chemical properties, the transformation proceeds as a reversible equilibrium process, and an excess amount of one of the starting materials is required to drive the reaction. Only a single example involving a nitrogen-containing heteroarene was reported.
Finally, although somewhat earlier, an example involving exchange between ketones and esters has also been reported. Arisawa and co-workers demonstrated that reversible acyl transfer occurs between benzyl ketones 5-21 and thioesters 5-22 using a RhH(CO)(PPh3)3/dppBz catalytic system (Fig. 5E).33 The reaction proceeds under thermodynamic control, as the product distribution varies depending on the substrate ratio. The acyl exchange was proposed to proceed through (1) stabilization of the metal center by the benzylic aryl group, (2) formation of an acyl–rhodium intermediate, and (3) reductive elimination to generate a new C–C bond.
Taken together, these studies illustrate the fundamental design principles underlying single-bond metathesis in fragment editing. In contrast to directional cross-coupling reactions, single-bond metathesis operates through reversible activation of σ bonds followed by fragment exchange and reductive elimination. Consequently, many of these transformations proceed under thermodynamic control. Various strategies have therefore been developed to direct the equilibrium toward desired products, including electronic tuning of substrates, irreversible steps such as decarbonylation, and funneling mechanisms that selectively stabilize specific products. These studies collectively establish single-bond metathesis as a central and versatile platform for fragment-level reorganization of aromatic frameworks.
3.2. Ketone fragment editing
In the context of fragment editing of ketone frameworks, heteroaromatic swapping reactions based on mechanisms distinct from transition-metal-catalyzed single-bond metathesis have also recently been reported. These approaches highlight that fragment-level reorganization can be achieved through mechanistically distinct activation modes beyond σ-bond metathesis. Li and co-workers described a strategy in which ketones 6-1 are converted into radical species through cooperative catalysis by an N-heterocyclic carbene (NHC) and a photoredox catalyst, enabling C–C bond reorganization (Fig. 6A).34 In this transformation, aromatic ketone 6-1 first reacts with aminobenzamide to form aminal 6-2. Upon photoirradiation, the excited photoredox catalyst (4CzIPN*) induces a single-electron transfer (SET) from 6-2, generating an alkyl (R) radical.35 Meanwhile, aromatic N-acylsuccinimide 6-3 reacts with the NHC catalyst to form an acyl azolium intermediate, which is subsequently reduced by the radical anion of 4CzIPN to produce a ketyl radical 6-4. Radical–radical coupling between these species ultimately furnishes the product aromatic ketone 6-5.In this approach, the key mechanistic feature is the involvement of radical intermediates rather than the conventional oxidative addition pathway. Consequently, the driving force of the transformation arises not from metal–carbon bond formation but from radical stabilization and recombination processes.
Yamaguchi and co-workers reported a heteroaromatic swapping reaction between aromatic ketones 6-6 and heteroaromatic esters 6-7 that proceeds through a Claisen/retro-Claisen mechanism (Fig. 6B).6,36
In this transformation, no transition-metal catalyst is required. Instead, the reaction is initiated by base-mediated enolate formation from aromatic ketone 6-6, which reacts with heteroaromatic ester 6-7 to generate a 1,3-diketone intermediate 6-8 through a Claisen-type condensation. The resulting diketone intermediate 6-8 then undergoes selective retro-Claisen cleavage, in which the C–C bond adjacent to the carbonyl group is broken to release the exchanged fragments. This process ultimately furnishes heteroaromatic ketone 6-9 together with the corresponding ester 6-10, thereby achieving heteroaromatic fragment exchange.
The thermodynamic preference for these products has been rationalized by the electronic stability differences between the enolate and ester components in the product pair, as well as by additional stabilization arising from aggregation of the sodium enolate species. Accordingly, the reaction operates under thermodynamic control, and fragment exchange proceeds through a reversible Claisen/retro-Claisen equilibrium that funnels the system toward the more stable product combination. This example demonstrates that classical carbonyl chemistry can be reinterpreted as a platform for fragment editing through controlled bond reorganization.
These studies demonstrate that the C–C(O) bond of ketones is not inherently inert. By selecting an appropriate activation mode—such as oxidative addition, radical generation, or enolate formation—ketone frameworks can be reorganized at the fragment level. Importantly, in all of these reactions the key design considerations are which bond is selectively cleaved under particular electronic conditions and whether the transformation proceeds through an equilibrium or irreversible pathway. Thus, ketone fragment editing provides a mechanistically diverse platform that complements single-bond metathesis-based strategies for heteroaromatic interconversion. These factors ultimately determine the product distribution and the feasibility of heteroaromatic incorporation.
3.3. Emerging opportunities for fragment editing
Beyond the single-bond metathesis and ketone-based fragment editing strategies described above, recent studies have demonstrated that reversible C–C bond “activation” can also enable fragment transfer between molecules.37 These developments suggest that fragment editing is not limited to specific bond classes but can, in principle, be generalized through reversible bond reorganization. Although these transformations have not yet been applied to heteroaromatic swapping, they provide important conceptual insights into how aromatic fragments can be redistributed through catalytic bond activation.For example, Dong and co-workers reported an orthogonal cross-coupling strategy enabled by intermolecular metathesis of unstrained C(aryl)–C(aryl) single bonds (Fig. 7A).38 In this transformation, a transition-metal catalyst activates an aryl–aryl σ bond 7-2 through oxidative addition, generating an aryl–metal intermediate that can subsequently exchange with another aryl fragment 7-1. Reductive elimination then furnishes cross-coupled biaryl products 7-3. Importantly, the study demonstrated that even relatively strong C(aryl)–C(aryl) bonds can be reversibly cleaved and recombined under catalytic conditions, thereby enabling redistribution of aromatic fragments through a metathesis-type process.
Morandi and co-workers later developed a shuttle arylation reaction based on Rh(I)-catalyzed reversible C–C bond activation of unstrained alcohol derivatives (Fig. 7B).39 In this system, Rh(I) inserts into a benzylic C–C bond of 7-5 adjacent to an alcohol-derived leaving group to generate an aryl–rhodium intermediate. The aryl fragment can then be transferred to another substrate 7-4 through a shuttle-type mechanism, effectively enabling intermolecular aryl transfer to generate product 7-6. Because the C–C bond activation step is reversible, the reaction allows catalytic redistribution of aryl fragments between molecules.
More recently, Zuo and co-workers reported a catalytic deracemization of alcohols (±)-7-9 that proceeds through reversible bond cleavage and recombination processes (Fig. 7C).40 In this strategy, a single catalyst system mediates both bond cleavage and reformation events, allowing repeated interconversion between enantiomeric intermediates 7-9 and 7-11. Iterative cleavage and recombination ultimately lead to amplification of stereochemical purity. Although the transformation is not designed for fragment exchange, it highlights how reversible bond activation can enable dynamic redistribution of molecular fragments under catalytic control.
Taken together, these studies illustrate that reversible C–C bond activation provides a powerful conceptual platform for fragment editing. Although heteroaromatic interconversion has not yet been realized using these strategies, several plausible scenarios can be envisaged. For example, extension to heteroaromatic substrates with relatively low coordinating ability, such as thiophene- or furan-derived systems, may enable C–C bond metathesis without catalyst deactivation. In addition, heteroaromatic ketone or alcohol derivatives bearing less strongly coordinating nitrogen-containing rings may also be compatible with reversible bond activation processes. Furthermore, tuning the electronic properties of heteroaromatic substrates, for instance by employing more electron-deficient systems, may facilitate selective bond cleavage and recombination. Collectively, these considerations suggest that rational substrate and catalyst design could enable heteroaromatic swapping reactions based on reversible σ-bond reorganization in the future.
4. Conclusions and outlook
Nitrogen incorporation into aromatic frameworks represents a powerful strategy for tuning molecular properties in pharmaceuticals, functional materials, and molecular design. Recent advances in molecular editing have enabled the direct transformation of aromatic systems without complete reconstruction of the molecular framework. As discussed in this Feature Article, nitrogen incorporation can be broadly achieved through two complementary strategies: skeletal editing and fragment editing. Skeletal editing operates at the atomic level through insertion, deletion, or replacement of individual atoms, whereas fragment editing reorganizes molecular structures through exchange of entire aromatic units. Together, these approaches provide a versatile platform for reprogramming aromatic frameworks with distinct levels of structural reorganization.From a practical perspective, skeletal and fragment editing strategies offer complementary advantages and should be selected based on the desired transformation. Skeletal editing is particularly suitable when precise atom-level modification of an aromatic framework is required, such as site-selective C-to-N transmutation while preserving the overall substitution pattern. In contrast, fragment editing is more effective for reorganizing molecular structures at a larger scale through exchange of aromatic units, enabling access to structural motifs that are difficult to obtain through direct atomic modification. Despite recent advances, both skeletal and fragment editing strategies still face important limitations. Skeletal editing often requires specific substrate preactivation and may be constrained by challenges in achieving general site-selectivity, particularly in complex molecular settings. In addition, the involvement of highly reactive intermediates can limit functional group compatibility and overall efficiency. To date, many transformations have been demonstrated primarily on relatively simple substrates, and their applicability to structurally complex molecules bearing multiple functional groups remains unclear. In parallel, classical skeletal rearrangements have seen increasing application in natural product synthesis in recent years.41 In contrast, many of the recently developed skeletal editing reactions are still at an early stage, and further conceptual and methodological advances are required to fully realize their potential.
Fragment editing, on the other hand, typically relies on reversible bond activation processes and is therefore often governed by thermodynamic constraints, which can limit substrate scope or require the use of excess reagents. From a catalytic perspective, these transformations require simultaneous activation of two distinct substrates, posing an inherent challenge for catalyst design. In the case of ketone fragment editing, the current strategies are largely limited to ketone-based systems, and analogous transformations involving other functional groups remain underexplored. Furthermore, examples involving nitrogen-containing heteroarenes remain relatively limited, highlighting issues associated with catalyst compatibility and coordination effects. These challenges underscore the need for new catalyst systems capable of selectively engaging multiple substrates while tolerating heteroaromatic functionalities.
Looking forward, addressing these challenges will be essential for advancing nitrogen editing of aromatic systems. In particular, achieving predictable site-selectivity in complex molecules remains a central goal, especially for applications in late-stage functionalization and drug discovery. The development of catalytic systems that are compatible with heteroaromatic substrates will also be critical for expanding the scope of fragment editing. In addition, improvements in efficiency, scalability, and operational simplicity are required for practical applications. Future progress may also arise from integrating skeletal and fragment editing strategies, as well as from combining reversible bond activation with selective nitrogen incorporation processes, thereby enabling new approaches to heteroaromatic framework editing. Such integration may ultimately allow programmable and predictable editing of aromatic frameworks across multiple structural scales.
Taken together, these developments position nitrogen editing as an emerging paradigm for the direct reprogramming of aromatic frameworks. Continued advances along both skeletal and fragment editing directions are expected to unlock new opportunities for molecular design across synthetic chemistry, materials science, and drug discovery.
Author contributions
H. N. conceived the concept of this review in discussion with J. Y. and prepared the figures. J. Y. wrote the main manuscript and guided its overall direction. H. N. and J. Y. revised the manuscript and finalized it for submission.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by JSPS KAKENHI Grant Number JP21H05213 (Digi-TOS), and JP25K01775 (to J.Y.). This work was partly supported by JST CREST Grant Number JPMJCR24T3 (to J.Y.).References
- (a) D. J. St. Jean and C. Fotsch, J. Med. Chem., 2012, 55, 6002–6020 CrossRef CAS PubMed; (b) L. D. Pennington and D. T. Moustakas, J. Med. Chem., 2017, 60, 3552–3579 CrossRef CAS PubMed; (c) Y. Hu, D. Stumpfe and J. Bajorath, J. Med. Chem., 2017, 60, 1238–1246 CrossRef CAS PubMed; (d) Y. Ling, Z.-Y. Hao, D. Liang, C.-L. Zhang, Y.-F. Liu and Y. Wang, Drug Des., Dev. Ther., 2021, 15, 4289–4338 CrossRef CAS PubMed; (e) L. D. Pennington, P. N. Collier and E. Comer, Med. Chem. Res., 2023, 32, 1278–1293 CrossRef CAS; (f) C. M. Marshall, J. G. Federice, C. N. Bell, P. B. Cox and J. T. Njardarson, J. Med. Chem., 2024, 67, 11622–11655 CrossRef CAS PubMed.
- (a) X. Guo, A. Facchetti and T. J. Marks, Chem. Rev., 2014, 114, 8943–9021 CrossRef CAS PubMed; (b) J. E. Anthony, Chem. Rev., 2006, 106, 5028–5048 CrossRef CAS PubMed; (c) A. Narita, X.-Y. Wang, X. Feng and K. Müllen, Chem. Soc. Rev., 2015, 44, 6616–6643 RSC; (d) X. Guo, M. Baumgarten and K. Müllen, Prog. Polym. Sci., 2013, 38, 1832–1908 CrossRef CAS; (e) J. Chen, K. Yang, X. Zhou and X. Guo, Chem. – Asian J., 2018, 13, 2587–2600 CrossRef CAS PubMed; (f) C. Kaes, A. Katz and M. W. Hosseini, Chem. Rev., 2000, 100, 3553–3590 CrossRef CAS PubMed; (g) X. Yu, C.-Y. Tsai, Y.-W. Chu, C.-C. Chueh and Z. Li, JACS Au, 2025, 5, 3483–3490 CrossRef PubMed.
- (a) J. A. Joule and K. Mills, Heterocycl. Chem., Wiley, Chichester, UK, 5th edn, 2010 Search PubMed; (b) ed. A. R. Katritzky, C. A. Ramsden, E. F. V. Scriven and R. J. K. Taylor, Comprehensive Heterocyclic Chemistry III, Elsevier, Oxford, UK, 2008 Search PubMed; (c) ed. E. F. V. Scriven, Pyridines: From Lab to Production, Elsevier, Amsterdam, 2013 Search PubMed; (d) X.-F. Wu, Transition Metal-Catalyzed Pyridine Synthesis, Elsevier, Amsterdam, 2016 Search PubMed; (e) L.-C. Campeau and K. Fagnou, Chem. Soc. Rev., 2007, 36, 1058–1068 RSC.
- (a) J. Jurczyk, J. Woo, S. F. Kim, B. D. Dherange, R. Sarpong and M. D. Levin, Nat. Synth., 2022, 1, 352–364 CrossRef CAS PubMed; (b) B. W. Joynson and L. T. Ball, Helvetica Chim. Acta, 2023, 106, e202200182 CrossRef CAS; (c) L. Ding, Y. Fan and H. Lu, Chem. Soc. Rev., 2025, 54, 8145–8169 RSC; (d) R. Sharma, M. Arisawa, S. Takizawa and M. S. H. Salem, Org. Chem. Front., 2025, 12, 1633–1670 RSC; (e) F.-P. Wu, J. L. Tyler and F. Glorius, Acc. Chem. Res., 2025, 58, 893–906 CrossRef CAS PubMed; (f) X. Bi, H. Wei, H. Lu and S. Song, CCS Chem., 2026, 8, 607–692 CrossRef; (g) D. Das, A. Mukherjee and S. Rej, Green Chem., 2026, 28, 4409–4448 RSC; (h) A. Kachore, V. Aggarwal, E. Bala, H. Singh, M. Selvaraj, M. A. Assiri, Saima, R. Kumar and P. K. Verma, Eur. J. Org. Chem., 2026, e202501188 CrossRef CAS; (i) S. F. Kim, C. Amber, G. L. Bartholomew and R. Sarpong, Acc. Chem. Res., 2025, 58, 1786–1800 CrossRef CAS PubMed; (j) R. Al-Ahmad and M. Dai, Advancing Total Synthesis Through Skeletal Editing, Acc. Chem. Res., 2025, 58(9), 1392–1406 CrossRef CAS PubMed; (k) M. D. Levin, R. Sarpong and A. E. Wendlandt, Acc. Chem. Res., 2025, 58, 1725–1726 CrossRef CAS PubMed.
- (a) B. N. Bhawal and B. Morandi, ACS Catal., 2016, 6, 7528–7535 CrossRef CAS; (b) B. N. Bhawal and B. Morandi, Chem. Eur. J., 2017, 23, 12004–12013 CrossRef CAS PubMed; (c) J. Zhang, M. Zhang and M. Oestreich, Chem Catal., 2024, 4, 100962 Search PubMed; (d) M. Kubo and J. Yamaguchi, Acc. Chem. Res., 2024, 57, 1747–1760 CrossRef CAS PubMed.
- H. Nakahara, R. Shirai, Y. Nishimoto, D. Yokogawa and J. Yamaguchi, Nat. Commun., 2025, 16, 8998 CrossRef CAS PubMed.
- Representative examples, See (a) Q. Cheng, D. Bhattacharya, M. Haring, H. Cao, C. Mück-Lichtenfeld and A. Studer, Nat. Chem., 2024, 16, 741–748 CrossRef CAS PubMed; (b) A. Conboy and M. F. Greaney, Chem, 2024, 10, 1940–1949 CrossRef CAS; (c) T. Morofuji, K. Inagawa and N. Kano, Org. Lett., 2021, 23, 6126–6130 CrossRef CAS PubMed; (d) F.-G. Zhang, Z. Chen, X. Tang and J.-A. Ma, Chem. Rev., 2021, 121, 14555–14593 CrossRef CAS PubMed; (e) S. Suzuki, Y. Segawa, K. Itami and J. Yamaguchi, Nat. Chem., 2015, 7, 227–233 CrossRef CAS PubMed; (f) A. R. Fout, B. C. Bailey, J. Tomaszewski and D. J. Mindiola, J. Am. Chem. Soc., 2007, 129, 12640–12641 CrossRef CAS PubMed.
- R. J. Sundberg, S. R. Suter and M. Brenner, J. Am. Chem. Soc., 1972, 94, 513–520 CrossRef CAS.
- S. C. Patel and N. Z. Burns, J. Am. Chem. Soc., 2022, 144, 17797–17802 CrossRef CAS PubMed.
- T. J. Pearson, R. Shimazumi, J. L. Driscoll, B. D. Dherange, D.-I. Park and M. D. Levin, Science, 2023, 381, 1474–1479 CrossRef CAS PubMed.
- H. Lu, Y. Zhang, X.-H. Wang, R. Zhang, P.-F. Xu and H. Wei, Nat. Commun., 2024, 15, 3772 CrossRef CAS PubMed.
- H. Lu, J. Chang and H. Wei, Acc. Chem. Res., 2025, 58, 933–946 CrossRef CAS PubMed.
- L. Li, D. Wang, Y. Zhang, J. Liu, H. Wang and X. Luan, Org. Lett., 2024, 26, 4910–4915 CrossRef CAS PubMed.
- J. Woo, C. Stein, A. H. Christian and M. D. Levin, Carbon-to-Nitrogen Single-Atom Transmutation of Azaarenes, Nature, 2023, 623(7985), 77–82 CrossRef CAS PubMed.
- Z. Wang, P. Xu, S.-M. Guo, C. G. Daniliuc and A. Studer, Nature, 2025, 642, 92–98 CrossRef CAS PubMed.
- A.-S. K. Paschke, S. Schiele, C. Pinard, F. Sandrini and B. Morandi, Chem. Sci., 2025, 16, 11464–11467 RSC.
- M. Puriņš, H. Nakahara and M. D. Levin, Science, 2025, 389, 295–298 CrossRef PubMed.
- W. Choi, A. Jang and S. Hong, J. Am. Chem. Soc., 2025, 147, 42042 CrossRef CAS PubMed.
- S. Liu, H. Li, Y. Chen, H. Bi and D. Mo, Chem. Eur. J., 2026, e00014 CrossRef PubMed.
- J. C. Reisenbauer, O. Green, A. Franchino, P. Finkelstein and B. Morandi, Science, 2022, 377, 1104–1109 CrossRef CAS PubMed.
- J. C. Reisenbauer, A.-S. K. Paschke, J. Krizic, B. B. Botlik, P. Finkelstein and B. Morandi, Org. Lett., 2023, 25, 8419–8423 CrossRef CAS PubMed.
- B. Ghosh, P. Kafle, R. Mukherjee, R. Welles, D. Herndon, K. M. Nicholas, Y. Shao and I. Sharma, Science, 2025, 387, 102–107 CrossRef CAS PubMed.
- B. Zhang, S. L. Homölle, T. Bauch, J. C. A. Oliveira, S. Warratz, B. Yuan, X. Gou and L. Ackermann, Angew. Chem., Int. Ed., 2024, 63, e202407384 CrossRef CAS PubMed.
- (a) Y. H. Lee and B. Morandi, Nat. Chem., 2018, 10, 1016–1022 CrossRef CAS PubMed; (b) M. D. L. H. Macias and B. A. Arndtsen, J. Am. Chem. Soc., 2018, 140, 10140–10144 CrossRef PubMed; (c) B. Mouhsine, M. Norlöff, J. Ghouilem, A. Sallustrau, F. Taran and D. Audisio, J. Am. Chem. Soc., 2024, 146, 8343–8351 CrossRef CAS PubMed; (d) H. Tanaka, E. Moriya, Y. Onozawa and J. Yamaguchi, JACS Au, 2026, 6, 1269–1279 CrossRef CAS PubMed.
- T. Delcaillau, P. Boehm and B. Morandi, J. Am. Chem. Soc., 2021, 143, 3723–3728 CrossRef CAS PubMed.
- (a) R. Isshiki, M. B. Kurosawa, K. Muto and J. Yamaguchi, J. Am. Chem. Soc., 2021, 143, 10333–10340 CrossRef CAS PubMed; (b) K. Muto, R. Isshiki, M. B. Kurosawa and J. Yamaguchi, Trends Chem., 2022, 5, 102–103 CrossRef.
- R. Takise, K. Muto, J. Yamaguchi and K. Itami, Angew. Chem., Int. Ed., 2014, 53, 6791–6794 CrossRef CAS PubMed.
- R. Takise, R. Isshiki, K. Muto, K. Itami and J. Yamaguchi, J. Am. Chem. Soc., 2017, 139, 3340–3343 CrossRef CAS PubMed.
- X. Wu, J. Li, S. Xia, C. Zhu and J. Xie, J. Org. Chem., 2022, 87, 10003–10017 CrossRef CAS PubMed.
- R. Isshiki, N. Inayama, K. Muto and J. Yamaguchi, ACS Catal., 2020, 10, 3490–3494 CrossRef CAS.
- T. Matsuyama, T. Yatabe, T. Yabe and K. Yamaguchi, Chem. Sci., 2024, 15, 11884–11889 RSC.
- T. Matsuyama, T. Yatabe, T. Yabe and K. Yamaguchi, Catal. Sci. Technol., 2023, 14, 76–82 RSC.
- M. Arisawa, M. Kuwajima, F. Toriyama, G. Li and M. Yamaguchi, Org. Lett., 2012, 14, 3804–3807 CrossRef CAS PubMed.
- Q.-Z. Li, M.-H. He, R. Zeng, Y.-Y. Lei, Z.-Y. Yu, M. Jiang, X. Zhang and J.-L. Li, J. Am. Chem. Soc., 2024, 146, 22829–22839 CrossRef CAS PubMed.
- F. Cong, R. S. Mega, J. Chen, C. S. Day and R. Martin, Angew. Chem., Int. Ed., 2023, 62, e202214633 CrossRef CAS PubMed.
- H. Nakahara, R. Isshiki, M. Kubo, K. Iizumi, K. Muto and J. Yamaguchi, Chem, 2024, 10, 2916–2930 CAS.
- B. Rybtchinski and D. Milstein, Angew. Chem., Int. Ed., 1999, 38, 870–883 CrossRef PubMed.
- J. Zhu, R. Zhang and G. Dong, Nat. Chem., 2021, 13, 836–842 CrossRef CAS PubMed.
- M. D. R. Lutz, V. C. M. Gasser and B. Morandi, Chem, 2021, 7, 1108–1119 CAS.
- L. Wen, J. Ding, L. Duan, S. Wang, Q. An, H. Wang and Z. Zuo, Science, 2023, 382, 458–464 CrossRef CAS PubMed.
- (a) S. F. Kim, C. Amber, G. L. Bartholomew and R. Sarpong, Acc. Chem. Res., 2025, 58, 1786–1800 CrossRef CAS PubMed; (b) R. Al-Ahmad and M. Dai, Acc. Chem. Res., 2025, 58, 1392–1406 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |