Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mechanochemically-synthesised AIE-active receptors for selective ceftazidime sensing in high-water-content matrices
Krzysztof Melcer a,
Tim David
a,
Tim David b,
Bernd M. Schmidt
b,
Bernd M. Schmidt b and
Artur Kasprzak
b and
Artur Kasprzak *a
*a
aFaculty of Chemistry, Warsaw University of Technology, Noakowskiego 3, 00-664 Warsaw, Poland. E-mail: artur.kasprzak@pw.edu.pl
bInstitut für Organische Chemie und Makromolekulare Chemie Heinrich-Heine-Universität Düsseldorf Universitätsstraße 1, D-40225 Düsseldorf, Germany
First published on 21st April 2026
Abstract
Mechanochemical synthesis of water-insoluble, AIE-active dendritic receptors enable selective fluorescence detection of ceftazidime in up to 99 vol% water. Selectivity over structurally related β-lactams was confirmed by titrations, DFT, and NMR studies. Detection remains reliable in tap water, artesian groundwater, and seawater, with a limit of detection (LOD) as low as 1.1 µM.
Mechanochemistry has emerged as a powerful non-conventional tool in modern organic synthesis, enabling sustainable synthetic protocols and access to compounds that are difficult or even inaccessible by classical in-solution methods.1–7 Molecular receptors capable of selectively interacting with guest molecules through non-covalent interactions are highly relevant in both fundamental and applied sciences.8–12 Among these, receptors featuring aggregation-induced emission (AIE) are particularly attractive, offering enhanced luminescence in aggregated states while simultaneously offering improved sensing performance, including sensitivity and selectivity, especially in aqueous media that remain challenging for conventional organic receptors.13–19 Although mechanochemistry has been explored for the synthesis of AIE-active receptors,20,21 the reported examples remain limited to a narrow set of molecular classes (such as azines,22–25 imides,26 and amides27,28) used for the detection of a limited number of analytes (such as inorganic ions,22,27,28 hydrazine,22,26 hydrogen peroxide,24 and nitroaromatic compounds23). Nevertheless, in these studies, the advantageous properties of the receptors in the aggregated state, particularly the enhancement of detection sensitivity relative to solution-phase behaviour, have been clearly demonstrated.
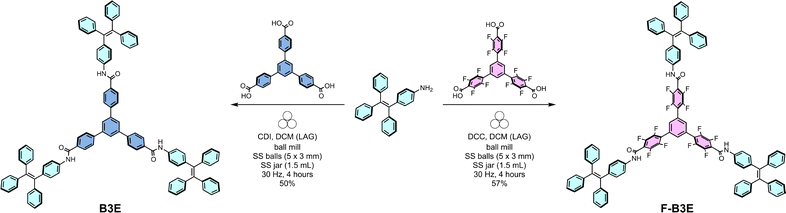
Herein, we report the mechanochemical synthesis of novel dendritic amide receptors, B3E and F-B3E, composed of 1,1,2,2-tetraphenylethene (TPE) and 1,3,5-triphenylbenzene (TPB) cores, with F-B3E bearing twelve fluorine substituents at the TPB core (Fig. 1). Introducing fluorinated substituents into AIE luminogens enables modification of their optical properties,29–31 with examples where up to four fluorine substituents alter absorption and emission maxima.29,32 Both receptors exhibit strong AIE behaviour in aqueous media and selectively detect the β-lactam antibiotic ceftazidime (Cef) in aqueous solutions containing up to 99 vol% water, as established by spectrofluorimetric studies. Currently established tools allow detection of Cef with LOD values down to 0.55 nM for electrochemical sensors33 and at the micromolar level for fluorescence-based sensors.34–36 The performance was further validated in tap water, artesian groundwater, and seawater, establishing practical applicability for environmental antibiotic monitoring using supramolecular systems.
Preliminary attempts to synthesise the target molecules in solution and via sonochemistry yielded undesired mixed products. The mechanochemical approach allowed the synthesis of compounds B3E and F-B3E, giving access to dendritic-like triamides under non-conventional conditions, including the trifold coupling of the near-perfluorinated tricarboxylic acid 2, a particularly challenging substrate, due to the electronic effect of the fluorine atoms on the reactivity of the carboxylic groups. Notably, the introduction of 12 fluorine atoms into the receptor scaffold might influence physicochemical properties, potential shifts in UV-vis absorption and fluorescence emission maxima, and a significant change of the fluorescence quantum yield. Tricarboxylic acid 1 or 2, amine 3, and a coupling agent, N,N′-dicyclohexylcarbodiimide (DCC) or carbonyldiimidazole (CDI), were ground for 4 hours with DCM (liquid-assisted grinding (LAG), η = 0.51–0.91 µL mg−1) in a stainless steel (SS) jar with SS balls. Isolation of pure products required purification by column chromatography and preparative thin-layer chromatography (PTLC), yielding B3E and F-B3E in 50% and 57% yields, respectively. Notably, solution-phase amide couplings of analogous hydrogenated substrates rarely exceed 50% per side.37
Structure and purity were confirmed by 1H NMR, {1H}13C NMR, 19F NMR (for F-B3E), 1H–1H COSY NMR, and ESI-HRMS (for full compound characterisation data, see SI, Sections S1–S3). The optical properties of B3E and F-B3E were studied using UV-vis and fluorescence spectroscopy. UV-vis measurements in THF solution and in the aggregated state (water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) THF 95
THF 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 v/v) revealed a shared solution-phase absorption maximum (λmax) at 235 nm for both receptors. Upon aggregation, B3E showed a redshift to 261 nm accompanied by a 2-fold increase in the molar absorption coefficient (ε) value for aggregates (εsolution = 6.9 × 104 dm3 mol−1 cm−1, εaggregates = 1.4 × 105 dm3 mol−1 cm−1). F-B3E shifted to 250 nm with a 10-fold decrease in ε value (εsolution = 2.6 × 105 dm3 mol−1 cm−1, εaggregates = 2.7 × 104 dm3 mol−1 cm−1), suggesting distinct aggregation behaviour between the two receptors (SI, Section S5).
5 v/v) revealed a shared solution-phase absorption maximum (λmax) at 235 nm for both receptors. Upon aggregation, B3E showed a redshift to 261 nm accompanied by a 2-fold increase in the molar absorption coefficient (ε) value for aggregates (εsolution = 6.9 × 104 dm3 mol−1 cm−1, εaggregates = 1.4 × 105 dm3 mol−1 cm−1). F-B3E shifted to 250 nm with a 10-fold decrease in ε value (εsolution = 2.6 × 105 dm3 mol−1 cm−1, εaggregates = 2.7 × 104 dm3 mol−1 cm−1), suggesting distinct aggregation behaviour between the two receptors (SI, Section S5).
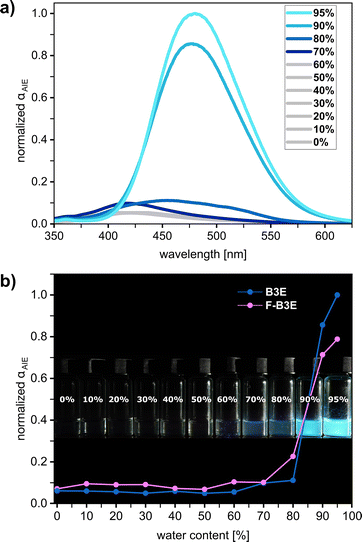
AIE behaviour was investigated by measuring fluorescence spectra in water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) THF systems with increasing water content (Fig. 2; SI, Section S5.2). For B3E, emission onset occurred at 70% vol., with intensity rising sharply at 90% vol. and reaching a maximum at 95%. F-B3E showed a more gradual response, with a modest intensity increase from 60–70% vol. water content that continued to increase until 95% vol. In both cases, aggregation was accompanied by a redshift in emission maximum (λem = 480 nm and 470 nm for B3E and F-B3E, respectively, redshift range 42–84 nm). The AIE enhancement ratio (αAIE)38 was 16.3 and 11.1 for B3E and F-B3E, respectively. Fluorescence quantum yield (ΦF) was measured in dissolved and aggregated states and increased approximately 5-fold upon aggregation for both receptors ΦF value of B3E (0.059) and F-B3E (0.109) in the aggregated state (SI, Section S5.1). The size of aggregates in different solvent systems was analysed using dynamic light scattering (DLS) (refer to SI, Section S5.3). DLS analysis revealed a 10-fold decrease in mean aggregate size with increasing water content for B3E and a 2.6-fold decrease for F-B3E. This tendency suggests a correlation between a decrease in aggregate size and an increase in fluorescence intensity, allowing to connect the process of disaggregation with a plausible mechanism of fluorescence quenching (SI, Section S5.3).
THF systems with increasing water content (Fig. 2; SI, Section S5.2). For B3E, emission onset occurred at 70% vol., with intensity rising sharply at 90% vol. and reaching a maximum at 95%. F-B3E showed a more gradual response, with a modest intensity increase from 60–70% vol. water content that continued to increase until 95% vol. In both cases, aggregation was accompanied by a redshift in emission maximum (λem = 480 nm and 470 nm for B3E and F-B3E, respectively, redshift range 42–84 nm). The AIE enhancement ratio (αAIE)38 was 16.3 and 11.1 for B3E and F-B3E, respectively. Fluorescence quantum yield (ΦF) was measured in dissolved and aggregated states and increased approximately 5-fold upon aggregation for both receptors ΦF value of B3E (0.059) and F-B3E (0.109) in the aggregated state (SI, Section S5.1). The size of aggregates in different solvent systems was analysed using dynamic light scattering (DLS) (refer to SI, Section S5.3). DLS analysis revealed a 10-fold decrease in mean aggregate size with increasing water content for B3E and a 2.6-fold decrease for F-B3E. This tendency suggests a correlation between a decrease in aggregate size and an increase in fluorescence intensity, allowing to connect the process of disaggregation with a plausible mechanism of fluorescence quenching (SI, Section S5.3).
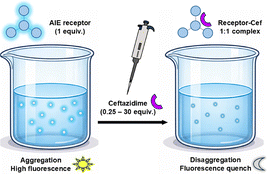
Detection of β-lactam antibiotics, which are widely used in the treatment of bacterial infections in humans, in high-water-content matrices represents a significant analytical challenge for environmental protection and monitoring.39 Unlike conventional organic receptors, which suffer from poor aqueous solubility and aggregation-caused quenching (ACQ), AIE-active receptors inherently overcome both limitations, making B3E and F-B3E well-suited candidates for antibiotic sensing in aqueous media.
To assess sensing performance, spectrofluorometric titrations were conducted with B3E and F-B3E against a series of β-lactam antibiotics, namely Amoxicillin (Amox), Penicillin G (Pen), Ampicillin (Ampc), and Ceftazidime (Cef), in water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) THF (95
THF (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
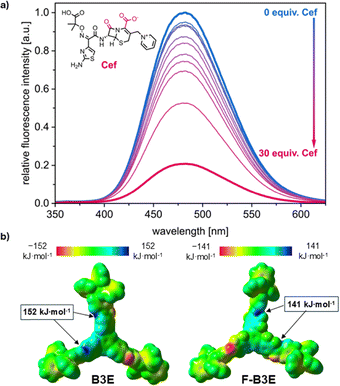
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 v/v) using ultrapure water and 0–30 equiv. of antibiotics (SI, Section S4). These antibiotics were carefully chosen due to their structural similarity and potential for binding with amide-bonding-containing molecules (refer to SI, Section S4, for structures). Both receptors responded selectively to Cef, with a 72–75% decrease in fluorescence intensity (Fig. 3a), while Pen, Ampc, and Amox produced no significant response (refer to SI, Section S6.1 for spectra). Selectivity was further confirmed by a competition experiment in which the addition of 1 equiv. Ampc did not affect the B3E–Cef titration curve. Analysis of the titration curves with the Bindfit40,41 tool indicated a 1
5 v/v) using ultrapure water and 0–30 equiv. of antibiotics (SI, Section S4). These antibiotics were carefully chosen due to their structural similarity and potential for binding with amide-bonding-containing molecules (refer to SI, Section S4, for structures). Both receptors responded selectively to Cef, with a 72–75% decrease in fluorescence intensity (Fig. 3a), while Pen, Ampc, and Amox produced no significant response (refer to SI, Section S6.1 for spectra). Selectivity was further confirmed by a competition experiment in which the addition of 1 equiv. Ampc did not affect the B3E–Cef titration curve. Analysis of the titration curves with the Bindfit40,41 tool indicated a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry for the Cef-receptor complexes, with association constant (Ka) values of 2.844 × 103 M−1 and 3.571 × 103 M−1 for B3E and F-B3E, respectively. Stern–Volmer constants (KS–V) of 104 M−1 and limit of detection (LOD) values in the micromolar range further confirm excellent detection parameters (Table 1; see SI, Section S6.1 for spectra and plots and Section S7 for all values).
1 stoichiometry for the Cef-receptor complexes, with association constant (Ka) values of 2.844 × 103 M−1 and 3.571 × 103 M−1 for B3E and F-B3E, respectively. Stern–Volmer constants (KS–V) of 104 M−1 and limit of detection (LOD) values in the micromolar range further confirm excellent detection parameters (Table 1; see SI, Section S6.1 for spectra and plots and Section S7 for all values).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) THF = 99
THF = 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v; full data for titrations with pure H2O are presented in Table S10, SI)
1 v/v; full data for titrations with pure H2O are presented in Table S10, SI)
| Solvent–receptor system | Ks–v × 103 [M−1] | LOD [µM] | |
|---|---|---|---|
| Tap water | B3E | 3.369 ± 0.153 | 7.1 |
| F-B3E | 3.159 ± 0.133 | 6.5 | |
| Seawater (filtered) | B3E | 4.070 ± 0.077 | 2.9 |
| F-B3E | 5.411 ± 0.228 | 6.5 | |
| Seawater (non-filtered) | B3E | 4.101 ± 0.178 | 6.7 |
| F-B3E | 4.346 ± 0.152 | 5.4 | |
| Artesian groundwater | B3E | 4.115 ± 0.179 | 6.4 |
| F-B3E | 2.423 ± 0.198 | 12.7 | |
The effect of water content on receptor performance was investigated by conducting titrations at 80, 95, and 99 vol% water (SI, Section S6.1). Increasing water content to 99 vol% maintained KS–V values comparable to those at 95 vol%, while delivering the lowest LOD observed across the entire study (1.1 µM for B3E), demonstrating effective Cef detection with minimal amount of organic solvent in the detection medium. Buffer experiments (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 buffer
1 buffer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) THF v
THF v![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v) revealed the KS–V value peaked at pH = 7.4 for both receptors, with only modest differences across acidic (pH = 5.14) and basic (pH = 8.29) conditions (ΔKS–V values ≤0.6 × 103 M−1). LOD values remained in the ranges of 2.3–7.5 µM and 6.3–9.4 µM for B3E and F-B3E, respectively, across all pH conditions, with B3E outperforming F-B3E under non-neutral conditions (SI, Section S6.1 and S7).
v) revealed the KS–V value peaked at pH = 7.4 for both receptors, with only modest differences across acidic (pH = 5.14) and basic (pH = 8.29) conditions (ΔKS–V values ≤0.6 × 103 M−1). LOD values remained in the ranges of 2.3–7.5 µM and 6.3–9.4 µM for B3E and F-B3E, respectively, across all pH conditions, with B3E outperforming F-B3E under non-neutral conditions (SI, Section S6.1 and S7).
To demonstrate real-world applicability, titrations were performed in tap water, artesian groundwater (manganese-rich), and seawater (sodium-rich), with seawater tested both filtered (0.2 µm PTFE) and unfiltered (mean particle size 841 nm, polymodal distribution; SI, Section S7 for full ICP-MS characterisation). In all matrices, both receptors successfully detected Cef, with KS–V and LOD values remaining comparable to those obtained in ultrapure water. B3E demonstrated greater robustness across environmental matrices, with KS–V values showing less variation than F-B3E and LOD values ranging from 2.9–7.1 µM, compared to 5.4–12.7 µM for F-B3E (Table 1, SI S7). In addition, competitive experiments using other classes of antibiotics (tetracycline, lincosamide, rifamycin) in the form of real-world pharmaceutical products did not affect the Cef sensing, further evidencing the potential of obtained receptors (refer to SI, Section S6.1, for the full data on titrations).
DFT calculations (B3LYP42/6-31g(d,p)43) were performed to optimise the structures of B3E and F-B3E and identify potential binding sites (Fig. 3b; SI, Section S4). Both receptors feature regions of high electrostatic surface potential (ESP), centred on the amide nitrogen atoms (ca. 152 and 141 kJ mol−1 for B3E and F-B3E, respectively), with the positive potential in B3E more delocalised across the TPB core compared to the more localised distribution in F-B3E. Complementarily, ESP maps of the tested β-lactam antibiotics revealed a uniquely concentrated negative potential on the carboxylic group of Cef, which also possesses the highest number of hydrogen bond acceptors in the series (12, versus 5–7 for Pen, Ampc, and Amox; SI, Section S4), rationalising the observed selectivity.
1H NMR titration experiments in DMSO-d6 confirmed that the amide N–H protons of both receptors shift upon Cef addition, while aromatic proton signals remain unchanged, identifying the amide moiety as the primary binding site (SI, Section S6.2). 19F NMR titration of F-B3E showed no fluorine signal shifts, ruling out fluorine atom participation in binding. Reverse titration (adding B3E to the Cef solution) produced an upfield shift of the pyridinium cation ring protons of Cef, consistent with receptor binding at the carboxylic group disrupting the internal ion pair. DLS measurements in the presence of 1 equiv. Cef showed a reduction in mean aggregate size of 11% and 49% for B3E and F-B3E, respectively (SI, Section S6.3), indicating that fluorescence quenching arises from a combination of direct amide–carboxylate binding and Cef-induced disaggregation (Fig. 4).
In conclusion, a mechanochemical synthesis afforded two novel AIE-active dendritic receptors, B3E and F-B3E, capable of selectively detecting ceftazidime in aqueous media containing up to 99 vol% water, enabled by their AIE properties that conventional water-insoluble receptors cannot match. Notably, introducing 12 fluorine atoms to an AIE-active molecule (F-B3E) led to a 2-fold increase in the fluorescence quantum yield. Detection parameters were robust across ultrapure water, buffer solutions, and real environmental matrices (tap water, artesian groundwater, and seawater), with KS-V and Ka at the 103 M−1 level and LOD as low as 1.1 µM. Beyond the immediate analytical application, this work establishes mechanochemical synthesis as a powerful green strategy for accessing structurally complex, functional AIE-active receptors that are difficult or inaccessible by conventional solution-phase routes. We anticipate that this approach will inspire broader exploration of mechanochemically synthesised supramolecular sensing systems, opening new avenues in sustainable materials chemistry, environmental monitoring, and the development of next-generation receptors for biorelevant analyte detection in complex aqueous matrices.
K. M. performed synthesis, characterisation and receptor experiments, analysed the data and wrote the manuscript and SI drafts with A. K. T. D. synthesised compound 1; A. K. conceived and supervised the project, analysed the data, performed DFT studies and wrote the respective part of SI, provided funding acquisition, reviewed and edited the manuscript and supporting information drafts together with K. M., T. D. and B. M. S., as well as led the correspondence with the editor and reviewers. All authors discussed the results and commented on the manuscript.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: materials and methods, experimental section, compounds characterization data, data on receptor studies, DFT calculations. See DOI: https://doi.org/10.1039/d6cc01502f.Acknowledgements
We acknowledge support from the POB Technologie Materiałowe (CPR-IDUB/37/Z01/POB5/2024; A. K.), the National Science Centre, Poland (OPUS, 2021/43/B/ST4/00114 (A. K.), and the Warsaw University of Technology (WUT, statutory support; A. K.). This work was supported by the Jürgen Manchot Foundation (PhD fellowship to T. D.). B. M. S. acknowledges the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) SCHM 3101/6. The DFT studies were performed with support of the Interdisciplinary Centre for Mathematical and Computational Modelling University of Warsaw (ICM UW) under computational allocation no. G101-2385 (A. K.). The number of hydrogen bonding donors/acceptors for antibiotics was computed with ADMET-predictor™ (Simulations Plus, University +License).44References
- S. L. James and T. Friščić, Chem. Soc. Rev., 2013, 42, 7494 RSC.
- J. F. Reynes, F. Leon and F. García, ACS Org. Inorg. Au, 2024, 4, 432–470 CrossRef CAS PubMed.
- T. Friščić, C. Mottillo and H. M. Titi, Angew. Chem., Int. Ed., 2020, 59, 1018–1029 CrossRef PubMed.
- J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13–19 CrossRef PubMed.
- T. Stolar, J. Alić, L. Casali, N. Gugin, M. Baláž, A. A. L. Michalchuk and F. Emmerling, Chem, 2026, 102880 CAS.
- S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. M. Harris, G. Hyett, W. Jones, A. Krebs, J. Mack, L. Maini, A. G. Orpen, I. P. Parkin, W. C. Shearouse, J. W. Steed and D. C. Waddell, Chem. Soc. Rev., 2012, 41, 413–447 RSC.
- T. Kunde, T. Pausch, P. A. Guńka, M. Krzyżanowski, A. Kasprzak and B. M. Schmidt, Chem. Sci., 2022, 13, 2877–2883 RSC.
- J. H. Hartley, T. D. James and C. J. Ward, J. Chem. Soc., Perkin Trans. 1, 2000, 3155–3184 RSC.
- L. Chen, S. N. Berry, X. Wu, E. N. W. Howe and P. A. Gale, Chem, 2020, 6, 61–141 CAS.
- P. A. Gale and C. Caltagirone, Chem. Soc. Rev., 2015, 44, 4212–4227 RSC.
- K. Maslowska-Jarzyna, E. York, E. Feo, R. M. Maklad, G. Bao, M. Fares and P. A. Gale, Chem, 2025, 11, 102695 CAS.
- S. K. Kim and J. L. Sessler, Chem. Soc. Rev., 2010, 39, 3784 RSC.
- R. T. K. Kwok, C. W. T. Leung, J. W. Y. Lam and B. Z. Tang, Chem. Soc. Rev., 2015, 44, 4228–4238 RSC.
- F.-Y. Ye, M. Hu and Y.-S. Zheng, Coord. Chem. Rev., 2023, 493, 215328 CrossRef CAS.
- Y. Hong, J. W. Y. Lam and B. Z. Tang, Chem. Commun., 2009, 4332 RSC.
- C. Zhu, R. T. K. Kwok, J. W. Y. Lam and B. Z. Tang, ACS Appl. Bio Mater., 2018, 1, 1768–1786 CrossRef CAS PubMed.
- S. Kulczyk and A. Kasprzak, Analysis Sensing, 2025, e202500173 Search PubMed.
- M. Gao and B. Z. Tang, ACS Sens., 2017, 2, 1382–1399 CrossRef CAS PubMed.
- M. H. Chua, B. Y. K. Hui, K. L. O. Chin, Q. Zhu, X. Liu and J. Xu, Mater. Chem. Front., 2023, 7, 5561–5660 RSC.
- J. S. Cyniak and A. Kasprzak, ACS Omega, 2024, 9, 48870–48883 CrossRef CAS PubMed.
- M. Banerjee, A. A. Bhosle, A. Chatterjee and S. Saha, J. Org. Chem., 2021, 86, 13911–13923 CrossRef CAS PubMed.
- A. A. Bhosle, M. Banerjee, S. Saha, S. Garg, S. Ghosh and A. Chatterjee, Sens. Actuators, B, 2023, 397, 134661 CrossRef CAS.
- A. A. Bhosle, M. Banerjee, S. D. Hiremath, A. C. Bhasikuttan and A. Chatterjee, Chem. – Asian J., 2023, 18, e202300048 CrossRef CAS PubMed.
- A. A. Bhosle, M. Banerjee, V. Gupta, S. Ghosh, A. C. Bhasikuttan and A. Chatterjee, New J. Chem., 2022, 46, 18961–18972 RSC.
- A. A. Bhosle, S. D. Hiremath, A. C. Bhasikuttan, M. Banerjee and A. Chatterjee, J. Photochem. Photobiol., A, 2021, 413, 113265 CrossRef CAS.
- S. D. Hiremath, R. U. Gawas, D. Das, V. G. Naik, A. A. Bhosle, V. P. Murali, K. K. Maiti, R. Acharya, M. Banerjee and A. Chatterjee, RSC Adv., 2021, 11, 21269–21278 RSC.
- J. S. Cyniak and A. Kasprzak, RSC Adv., 2024, 14, 13227–13236 RSC.
- J. S. Cyniak, K. Melcer and A. Kasprzak, Chem. Eur. J., 2025, 31, e02859 CrossRef CAS PubMed.
- A. Nitti, E. Tatsi, E. Magnani, G. Mattioli, F. Porcelli, C. Botta, G. Griffini and D. Pasini, Chem. Commun., 2026, 62, 4320–4324 RSC.
- P. Zheng, Y. Ding, A. Abdurahman, J. Zhu, L. Tian, Y. Liu, Q. Liu, L. Wang, D. Wang and B. Z. Tang, Sci. China: Chem., 2025, 68, 3086–3099 CrossRef CAS.
- X. Lv, Y. Bao, H. Zhou and Y. Qu, Dyes Pigm., 2025, 233, 112525 CrossRef CAS.
- H. Zhang, Y. Nie, J. Miao, D. Zhang, Y. Li, G. Liu, G. Sun and X. Jiang, J. Mater. Chem. C, 2019, 7, 3306–3314 RSC.
- M. Torkashvand, M. B. Gholivand and G. Malekzadeh, Sens. Actuators, B, 2016, 231, 759–767 CrossRef CAS.
- J. Hu, Y. Ma and S. Wu, Spectrochim. Acta, Part A, 2025, 327, 125341 CrossRef CAS PubMed.
- S. Fathi, Russ. J. Electrochem., 2014, 50, 468–475 CrossRef CAS.
- A. de, H. Moreno and H. R. N. Salgado, J. AOAC Int., 2009, 92, 820–823 CrossRef.
- L. Zhang, S. Jiang, Z. Yi, M. Peng, F. Wang and Y. Tian, Chem. Commun., 2026, 62, 3526–3530 RSC.
- R. Hu, N. L. C. Leung and B. Z. Tang, Chem. Soc. Rev., 2014, 43, 4494–4562 RSC.
- K. M. Sta Ana, J. Madriaga and M. P. Espino, Environ. Pollut., 2021, 275, 116624 CrossRef CAS PubMed.
- D. Brynn Hibbert and P. Thordarson, Chem. Commun., 2016, 52, 12792–12805 RSC.
- P. Thordarson, Chem. Soc. Rev., 2011, 40, 1305–1323 RSC.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- J. S. Binkley, J. A. Pople and W. J. Hehre, J. Am. Chem. Soc., 1980, 102, 939–947 CrossRef CAS.
- ADMET Predictor, https://www.simulations-plus.com.
| This journal is © The Royal Society of Chemistry 2026 |