Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Exploration of naphtho[c]dithiophenes: synthesis and optical and electrochemical properties of naphtho[1,2-c:5,6-c′]dithiophenes
Yuki Okazaki,
Shogo Amimoto,
Kumpei Kozuka,
Satoru Maekawa,
Keiichi Imato and
Yousuke Ooyama
and
Yousuke Ooyama *
*
Applied Chemistry Program, Graduate School of Advanced Science and Engineering, Hiroshima University, 1-4-1 Kagamiyama, Higashi-Hiroshima 739-8527, Japan. E-mail: yooyama@hiroshima-u.ac.jp
First published on 27th April 2026
Abstract
An efficient synthetic method for naphtho[1,2-c:5,6-c′]dithiophenes and their tetrasubstituted derivatives has been developed and their photophysical and electrochemical properties were investigated. The naphtho[c]dithiophene skeleton as a new thiophene-fused polycyclic aromatic system can be used as a π-building block in emitters, semiconductors and photosensitizers for organic optoelectronic devices.
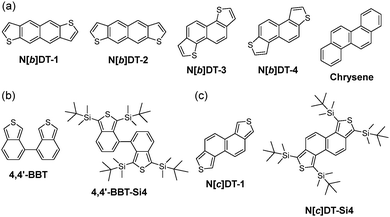
Thiophene-fused polycyclic aromatic systems are of scientific and practical interest in synthetic organic chemistry, photochemistry and electrochemistry, and serve as key constituents of emitters, semiconductors and photosensitizers for organic optoelectronic devices.1–5 In particular, benzo[b]thiophenes and naphtho[b]dithiophenes, including naphtho[2,3-b:6,7-b′]dithiophene, naphtho[2,3-b:7,6-b′]dithiophene, naphtho[1,2-b:5,6-b′]dithiophene, and naphtho[2,1-b:6,5-b′]dithiophene, exhibiting high charge transport properties are widely used in organic field-effect transistors (OFETs), organic photovoltaics (OPVs), and organic light-emitting diodes (OLEDs) (Fig. 1a).6–15 Meanwhile, benzo[c]thiophenes, 1,1′- and 4,4′-bibenzo[c]thiophenes and fused-bibenzo[c]thiophenes have received considerable attention in recent years not only due to the challenges in developing facile and efficient synthetic methods, but also due to their attractive photophysical and electrochemical properties, which make them promising fluorophores and photosensitizers for highly efficient bioimaging, phototheranostics, and optoelectronic devices (Fig. 1b).16–30
 | ||
| Fig. 1 (a) Chemical structures of naphtho[b]dithiophenes (ref. 10) and chrysene. (b) Previous work (ref. 25) for 4,4′-bibenzo[c]thiophenes and (c) this work for naphtho[c]dithiophenes. | ||
Inspired by the strategic research for the development and practical application of thiophene-fused polycyclic aromatic systems, which are attracting increasing attention, we devised a strategy to construct naphtho[c]dithiophenes and to investigate their photophysical and electrochemical properties. Herein, we report an efficient synthetic method for naphtho[1,2-c:5,6-c′]dithiophene (N[c]DT-1) and its tetrasubstituted derivative (N[c]DT-Si4) with four tert-butyldimethylsilyl groups on two thiophene rings (Fig. 1c) and investigate their photophysical and electrochemical properties using photoabsorption and fluorescence spectroscopies, cyclic voltammetry (CV), and density functional theory (DFT) calculations. It was found that N[c]DT-1 exhibits intense vibronic-structured photoabsorption and fluorescence bands in the long-wavelength region and a high HOMO energy level and a low LUMO energy level, compared to its structural isomers naphtho[b]dithiophenes, naphtho[1,2-b:5,6-b′]dithiophene (N[b]DT-3) and naphtho[2,1-b:6,5-b′]dithiophene (N[b]DT-4) as well as isoelectronic chrysene with N[c]DT-1. Indeed, this work is the first to report the synthesis and photophysical and electrochemical characteristics of naphtho[c]dithiophenes.
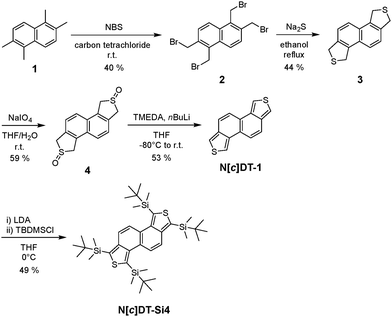
N[c]DT-1 and its tetrasilyl-substituted derivative (N[c]DT-Si4) were synthesized according to a stepwise synthetic protocol (Scheme 1). The starting compound 1, 1,2,5,6-tetramethylnaphthalene, was prepared through the Wolff–Kishner reduction of 2,6-dimethylnaphthalene-1,5-dicarbaldehyde, which was prepared through Bodroux–Chichibabin aldehyde synthesis using 1,5-dibromo-2,6-dimethylnaphthalene (Scheme S1, SI), because commercially available compound 1 is very expensive. The bromination of 1 with N-bromosuccinimide (NBS) gave compound 2 with a yield of 40%. Tetrahydronaphtho[1,2-c:5,6-c′]dithiophene 3 was obtained in a yield of 44% by the reaction of 2 with sodium sulfide. The oxidation of 3 with sodium periodate gave sulfoxide 4 with a moderate yield (59%), which is a key intermediate in the synthesis of naphtho[1,2-c:5,6-c′]dithiophenes. Indeed, naphtho[1,2-c:5,6-c′]dithiophene (N[c]DT-1) was successfully prepared in a yield of 53% or 30% by treatment of 4 with tetramethylethylenediamine (TMEDA) and then nBuLi or with lithium hexamethyldisilazide (LHMDS) (see the SI). Furthermore, the tetrasilyl-substituted derivative (N[c]DT-Si4) was obtained in a yield of 49% by the reaction of N[c]DT-1 with lithium diisopropylamide (LDA), followed by treatment with tert-butyldimethylsilyl chloride (TBDMSCl) as an electrophile. N[c]DT-1 and N[c]DT-Si4 were successfully characterized by FTIR, 1H and 13C NMR measurements and HRMS analysis. Therefore, this result proposes a facile and stepwise synthetic method for the introduction of various substituents, including bromo, stannyl, and boronic acid functional groups for the Stille or Suzuki coupling reaction, into the thiophene rings of the naphtho[1,2-c:5,6-c′]dithiophene skeleton.
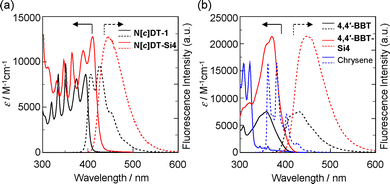
The photoabsorption and fluorescence spectra of N[c]DT-1 and N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4 as non-fused bibenzo[c]thiophenes, and isoelectronic chrysene with N[c]DT-1 in toluene are shown in Fig. 2, and their photophysical data along with those of N[b]DT-3 and N[b]DT-4 are summarized in Table 1. N[c]DT-1 and N[c]DT-Si4 exhibit vibronic-structured photoabsorption bands similar to N[b]DT-3, N[b]DT-4 and chrysene, although 4,4′-BBT and 4,4′-BBT-Si4 exhibit broad photoabsorption bands. The photoabsorption maximum wavelength (λabsmax = 395 nm) of N[c]DT-1 showed bathochromic shifts by 36 nm, 75 nm, 45 nm and 73 nm, compared to those of 4,4′-BBT, N[b]DT-3, N[b]DT-4 and chrysene, respectively. Furthermore, similar to 4,4′-BBT and 4,4′-BBT-Si4, N[c]DT-Si4 exhibits an intense photoabsorption band (λabsmax = 411 nm) with a relatively high molar absorption coefficient (εmax = 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 M−1 cm−1) at a longer wavelength by 16 nm, compared to N[c]DT-1 (εmax = 8600 M−1 cm−1). The corresponding fluorescence band of N[c]DT-1 has a vibrational structure, similar to that of chrysene, while that of N[c]DT-Si4 is broadened, similar to those of 4,4′-BBT and 4,4′-BBT-Si4 (Fig. 2). It is worth noting here that the fluorescence maximum wavelength (λflmax = 407 nm) of N[c]DT-1 appeared at a shorter wavelength by 38 nm, 3 nm and 44 nm, respectively, compared to those (445 nm, 410 nm and 451 nm, respectively) of N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4. Thus, the Stokes shift (7.46 × 102 cm−1) of N[c]DT-1 is significantly smaller than those (1.85 × 103 cm−1, 3.46 × 103 cm−1, 4.78 × 103 cm−1, and 3.43 × 103 cm−1, respectively) of N[c]DT-Si4, 4,4′-BBT, 4,4′-BBT-Si4, and chrysene. Obviously, this result indicates that N[c]DT-1 has a rigid and expanded π-conjugation structure, compared to 4,4′-BBT. Meanwhile, for N[c]DT-Si4, the broad fluorescence band and a relatively large Stokes shift (SS) value are due to the rotatable and flexible tert-butyldimethylsilyl group. However, the fluorescence quantum yield (Φfl = 0.20) of N[c]DT-1 is higher than those (0.10) of N[c]DT-Si4 and chrysene, but is lower than those (0.41 and 0.36, respectively) of 4,4′-BBT and 4,4′-BBT-Si4. Time-resolved fluorescence spectroscopy demonstrated that the fluorescence lifetimes (τfl = 2.93 ns and 1.00 ns) of N[c]DT-1 and N[c]DT-Si4 are a little shorter than those (3.46 ns and 3.59 ns) of 4,4′-BBT and 4,4′-BBT-Si4, but much shorter than that (12.1 ns) of chrysene. The radiative rate constant (kr = 6.8 × 107 s−1) for N[c]DT-1 is about one-half those (1.0–1.1 × 108 s−1) for N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4, but the kr value (8.2 × 106 s−1) for chrysene is much smaller than those for the other four compounds. In contrast, the nonradiative rate constant (knr = 9.0 × 108 s−1) for N[c]DT-Si4 is significantly larger than those (7.4 × 107–2.7 × 108 s−1) for the other four compounds. Thus, the ratio (knr/kr = 9.0) of the nonradiative constant to the radiative constant for N[c]DT-Si4 and chrysene is larger than those (4.0, 1.5 and 1.7, respectively) for N[c]DT-1, 4,4′-BBT and 4,4′-BBT-Si4, indicating that the lower Φfl values of N[c]DT-Si4 and chrysene are mainly due to the larger knr value and smaller kr value, respectively, compared to the Φfl values of N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4. Consequently, these facts for the naphtho[1,2-c:5,6-c′]dithiophene skeleton compared to the 4,4′-bibenzo[c]thiophene skeleton, that is, the vibronic-structured photoabsorption and fluorescence bands in the long-wavelength region, the mirror symmetry relationship, and the small Stokes shift value indicate that naphtho[c]dithiophene has a rigid and expanded π-conjugation structure.
700 M−1 cm−1) at a longer wavelength by 16 nm, compared to N[c]DT-1 (εmax = 8600 M−1 cm−1). The corresponding fluorescence band of N[c]DT-1 has a vibrational structure, similar to that of chrysene, while that of N[c]DT-Si4 is broadened, similar to those of 4,4′-BBT and 4,4′-BBT-Si4 (Fig. 2). It is worth noting here that the fluorescence maximum wavelength (λflmax = 407 nm) of N[c]DT-1 appeared at a shorter wavelength by 38 nm, 3 nm and 44 nm, respectively, compared to those (445 nm, 410 nm and 451 nm, respectively) of N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4. Thus, the Stokes shift (7.46 × 102 cm−1) of N[c]DT-1 is significantly smaller than those (1.85 × 103 cm−1, 3.46 × 103 cm−1, 4.78 × 103 cm−1, and 3.43 × 103 cm−1, respectively) of N[c]DT-Si4, 4,4′-BBT, 4,4′-BBT-Si4, and chrysene. Obviously, this result indicates that N[c]DT-1 has a rigid and expanded π-conjugation structure, compared to 4,4′-BBT. Meanwhile, for N[c]DT-Si4, the broad fluorescence band and a relatively large Stokes shift (SS) value are due to the rotatable and flexible tert-butyldimethylsilyl group. However, the fluorescence quantum yield (Φfl = 0.20) of N[c]DT-1 is higher than those (0.10) of N[c]DT-Si4 and chrysene, but is lower than those (0.41 and 0.36, respectively) of 4,4′-BBT and 4,4′-BBT-Si4. Time-resolved fluorescence spectroscopy demonstrated that the fluorescence lifetimes (τfl = 2.93 ns and 1.00 ns) of N[c]DT-1 and N[c]DT-Si4 are a little shorter than those (3.46 ns and 3.59 ns) of 4,4′-BBT and 4,4′-BBT-Si4, but much shorter than that (12.1 ns) of chrysene. The radiative rate constant (kr = 6.8 × 107 s−1) for N[c]DT-1 is about one-half those (1.0–1.1 × 108 s−1) for N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4, but the kr value (8.2 × 106 s−1) for chrysene is much smaller than those for the other four compounds. In contrast, the nonradiative rate constant (knr = 9.0 × 108 s−1) for N[c]DT-Si4 is significantly larger than those (7.4 × 107–2.7 × 108 s−1) for the other four compounds. Thus, the ratio (knr/kr = 9.0) of the nonradiative constant to the radiative constant for N[c]DT-Si4 and chrysene is larger than those (4.0, 1.5 and 1.7, respectively) for N[c]DT-1, 4,4′-BBT and 4,4′-BBT-Si4, indicating that the lower Φfl values of N[c]DT-Si4 and chrysene are mainly due to the larger knr value and smaller kr value, respectively, compared to the Φfl values of N[c]DT-Si4, 4,4′-BBT and 4,4′-BBT-Si4. Consequently, these facts for the naphtho[1,2-c:5,6-c′]dithiophene skeleton compared to the 4,4′-bibenzo[c]thiophene skeleton, that is, the vibronic-structured photoabsorption and fluorescence bands in the long-wavelength region, the mirror symmetry relationship, and the small Stokes shift value indicate that naphtho[c]dithiophene has a rigid and expanded π-conjugation structure.
| Dye | λabsmax/nm (εmax/M−1 cm−1) | λflmax/nm (Φfl)f | SS/cm−1 | τflg/ns | krh/s−1 | knri/s−1 | knr/kr | Eoxonset![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j/V j/V |
Eoptg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k/eV k/eV |
HOMOl/eV | LUMOl/eV |
|---|---|---|---|---|---|---|---|---|---|---|---|
| a In toluene.b In dichloromethane.c The longest wavelength maximum of the vibronic-structured photoabsorption band.d The shortest wavelength maximum of the vibronic-structured fluorescence band.e Photoabsorption edge.f Fluorescence quantum yields (Φfl) were determined by using a calibrated integrating sphere system (λex = 366 nm, 366 nm, 359 nm, 371 nm, and 318 nm for N[c]DT-1, N[c]DT-Si4, 4,4′-BBT, 4,4′-BBT-Si4, and chrysene, respectively).g Fluorescence lifetime.h Radiative rate constant (kr = Φfl/τfl).i Nonradiative rate constant (knr = (1 − Φfl)/τfl).j Onset potentials (Eoxonset) versus Fc/Fc+ of the oxidation potential.k Optical energy gaps (Eoptg) of N[c]DT-1, N[c]DT-Si4, 4,4′-BBT, and 4,4′-BBT-Si4 were determined from the intersection (400 nm, 422 nm, 393 nm, and 408 nm, respectively) of photoabsorption and fluorescence spectra in toluene. Eoptg values of N[b]DT-3, N[b]DT-4, and chrysene were determined from the photoabsorption edge (320 nm, 350 nm, and 334 respectively).l Versus the vacuum level.m Previous work (ref. 25).n Ref. 10.o No data.p See Fig. S22 for the cyclic voltammogram, SI. | |||||||||||
| N[c]DT-1 | 395c (8600)a | 407d (0.20)a | 7.46 × 102 | 2.93a | 6.8 × 107a | 2.7 × 108a | 4.0a | 0.50 | 3.10a | −5.30 | −2.20 |
| N[c]DT-Si4 | 411c (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700)a 700)a |
445 (0.10)a | 1.85 × 103 | 1.00a | 1.0 × 108a | 9.0 × 108a | 9.0a | 0.59 | 2.94a | −5.39 | −2.45 |
| 4,4′-BBTm | 359 (7500)a | 410 (0.41)a | 3.46 × 103 | 3.46a | 1.1 × 108a | 1.7 × 108a | 1.5a | 0.75m | 3.16a | −5.55 | −2.39 |
| 4,4′-BBT-Si4m | 371 (21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300)a 300)a |
451 (0.36)a | 4.78 × 103 | 3.59a | 1.0 × 108a | 1.7 × 108a | 1.7a | 0.54m | 3.04a | −5.34 | −2.30 |
| N[b]DT-3n | 320e (—o)b | —o | —o | —o | —o | —o | —o | —o | 3.90b | −5.80 | −1.90 |
| N[b]DT-4n | 350e (—o)b | —o | —o | —o | —o | —o | —o | —o | 3.50b | −5.70 | −2.20 |
| Chrysene | 322c (16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400)a 400)a |
362d (0.10)a | 3.43 × 103 | 12.1a | 8.2 × 106a | 7.4 × 107a | 9.0a | 1.00p | 3.71a | −5.80 | −2.09 |
The electrochemical properties of N[c]DT-1 and N[c]DT-Si4 were determined using CV in DMF containing 0.1 M tetrabutylammonium perchlorate (Bu4NClO4). The potentials were internally referenced to ferrocene/ferrocenium (Fc/Fc+). The cyclic voltammograms of the two compounds are shown in Fig. 3, and their electrochemical data and the HOMO and LUMO energy levels are summarized in Table 1. The irreversible oxidation wave was observed at 0.68 V for N[c]DT-1 and 0.76 V for N[c]DT-Si4, while any obvious reduction wave did not appear within the potential window (−1.5 V to 0 V versus Fc/Fc+). Thus, the oxidation wave for N[c]DT-1 is slightly cathodically shifted by ca. 0.10 V, compared to that for N[c]DT-Si4. The HOMO energy levels (–[Eoxonset + 4.8] eV) versus the vacuum level were estimated from the onset potentials (Eoxonset = 0.50 V for N[c]DT-1 and 0.59 V for N[c]DT-Si4) of the oxidation waves, and the LUMO energy levels were estimated from the Eoxonset and intersections (optical energy gap: Eoptg = 3.10 eV for N[c]DT-1 and 2.94 eV for N[c]DT-Si4) of the photoabsorption and fluorescence spectra in toluene. The HOMO energy level (−5.30 eV) of N[c]DT-1 is significantly higher than those (−5.55 eV, −5.80 eV, −5.70 eV, and −5.80 eV) of 4,4′-BBT, N[b]DT-3, N[b]DT-4 and chrysene. Meanwhile, the LUMO energy level (−2.20 eV) of N[c]DT-1 is equivalent to that of N[b]DT-4, but is lower than those (−1.90 eV and −2.09 V) of N[b]DT-3 and chrysene and is somewhat higher than that (−2.39 eV) of 4,4′-BBT. Consequently, it was revealed that the bathochromic shift of the photoabsorption band for N[c]DT-1 relative to 4,4′-BBT, N[b]DT-3, N[b]DT-4 and chrysene is attributed not only to destabilization of the HOMO energy level but also to stabilization of the LUMO energy level due to the naphtho[c]dithiophene skeleton, resulting in a decrease in the HOMO–LUMO band gap. Interestingly, the HOMO and LUMO energy levels of N[c]DT-Si4 are lower than those of N[c]DT-1, but the lowering of the LUMO energy level is larger than that of the HOMO energy level, whereas the HOMO and LUMO energy levels of 4,4′-BBT-Si4 are higher that those of 4,4′-BBT, but the rise of the HOMO energy level is larger than that of the LUMO energy level, resulting in a decrease in the HOMO–LUMO band gap, that is, a bathochromic shift of the photoabsorption band.
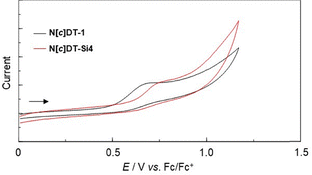 | ||
| Fig. 3 Cyclic voltammograms of N[c]DT-1 and N[c]DT-Si4 in DMF containing 0.1 M Bu4NClO4 at a scan rate of 100 mV s−1. The arrow denotes the direction of the potential scan. | ||
To examine the electronic structures of naphtho[1,2-c:5,6-c′]dithiophenes, the molecular orbitals of N[c]DT-1 and N[c]DT-Si4 were calculated using DFT at the B3LYP/6-31G(d,p) level (Fig. S23). Similar to chrysene, the HOMO and the LUMO of N[c]DT-1 and N[c]DT-Si4 are delocalized over the whole molecule. The DFT calculations demonstrated that the HOMO and LUMO energy levels of N[c]DT-1 are higher and lower than those of 4,4′-BBT, N[b]DT-3, N[b]DT-4 and chrysene, respectively, similar to the experimental results, resulting in a decrease in the HOMO–LUMO band gap. The fact that the HOMO–LUMO band gaps of N[b]DT-3, N[b]DT-4 and chrysene are wider than that of N[c]DT-1 can be understood from Clar's aromatic π-sextet rule, which states that the HOMO–LUMO band gap widens as the number of aromatic π-sextet in polycyclic aromatic hydrocarbon increases; N[b]DT-3, N[b]DT-4 and chrysene have four aromatic π-sextets, while N[c]DT-1 has two aromatic π-sextets.31 In addition, as in the case of 4,4′-BBT and 4,4′-BBT-Si4, the LUMO energy level of N[c]DT-Si4 is similar to that of N[c]DT-1, whereas the HOMO energy level of N[c]DT-Si4 is higher than that of N[c]DT-1, leading to a decrease in the HOMO–LUMO band gap. This result suggests that the destabilization of the HOMO energy level is due to the introduction of the electron-donating tert-butyldimethylsilyl group into the thiophene ring, although the change in the HOMO energy level from N[c]DT-1 to N[c]DT-Si4 according to the DFT calculations is opposite to the experimental results. Furthermore, the time-dependent density functional theory (TD-DFT) calculations were performed to elucidate the photophysical properties of naphtho[1,2-c:5,6-c′]dithiophenes (Fig. S24). The calculated λabs-calcdmax of the seven compounds appears in longer wavelength regions in the order of N[b]DT-3 (308 nm) < N[b]DT-4 (317 nm) ≈ chrysene (322 nm) < 4,4′-BBT (345 nm) < 4,4′-BBT-Si4 (356 nm) < N[c]DT-1 (402 nm) < N[c]DT-Si4 (428 nm), and the calculated εcalcd increases in the order of N[c]DT-1 (4200 M−1 cm−1) < chrysene (5300 M−1 cm−1) < N[b]DT-3 (6100 M−1 cm−1) < N[c]DT-Si4 (6700 M−1 cm−1) < 4,4′-BBT (7800 M−1 cm−1) < N[b]DT-4 (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 M−1 cm−1) < 4,4′-BBT-Si4 (15
200 M−1 cm−1) < 4,4′-BBT-Si4 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1). The S0 → S1 transitions are mainly attributed to the transitions from the HOMO to the LUMO (99% for N[c]DT-1, 99% for N[c]DT-Si4, 67% for 4,4′-BBT, 68% for 4,4′-BBT-Si4, 55% for N[b]DT-3, and 89% for N[b]DT-4, 52% for chrysene). Indeed, the TD-DFT calculations are in good agreement with the experimental results regarding the bathochromic shift of the photoabsorption band from N[b]DT-3 to N[c]DT-Si4, although there are differences in the εcalcd values between the experimental and TD-DFT calculation results.
600 M−1 cm−1). The S0 → S1 transitions are mainly attributed to the transitions from the HOMO to the LUMO (99% for N[c]DT-1, 99% for N[c]DT-Si4, 67% for 4,4′-BBT, 68% for 4,4′-BBT-Si4, 55% for N[b]DT-3, and 89% for N[b]DT-4, 52% for chrysene). Indeed, the TD-DFT calculations are in good agreement with the experimental results regarding the bathochromic shift of the photoabsorption band from N[b]DT-3 to N[c]DT-Si4, although there are differences in the εcalcd values between the experimental and TD-DFT calculation results.
In conclusion, in order to construct naphtho[c]dithiophenes and to investigate their optical and electrochemical properties, we developed an efficient synthetic method for naphtho[1,2-c:5,6-c′]dithiophene (N[c]DT-1) and its tetrasubstituted derivative (N[c]DT-Si4) with four tert-butyldimethylsilyl groups on two thiophene rings for the first time. Furthermore, this facile synthetic method will allow the introduction of various substituents, including bromo, stannyl, and boronic acid functional groups for the Stille or Suzuki coupling reaction, into the thiophene rings of the naphtho[1,2-c:5,6-c′]dithiophene skeleton. The photophysical and electrochemical measurement, and density functional theory calculations revealed that naphtho[1,2-c:5,6-c′]dithiophenes possess intense vibronic-structured photoabsorption and fluorescence bands in the long-wavelength region and a high HOMO energy level and a low LUMO energy level, compared to their structural isomers naphtho[b]dithiophenes, naphtho[1,2-b:5,6-b′]dithiophene and naphtho[2,1-b:6,5-b′]dithiophene as well as isoelectronic chrysene with naphtho[1,2-c:5,6-c′]dithiophenes. Consequently, this work demonstrates that the naphtho[1,2-c:5,6-c′]dithiophene skeleton with the above advantageous properties would be used as a π-building block in emitters, semiconductors and photosensitizers for organic optoelectronic devices, and thus will open up research and development of new thiophene-fused polycyclic aromatic systems.
Prof. Yousuke Ooyama conceived the project. Dr Keiichi Imato directed the experimental work. Yuki Okazaki performed most of the experiments. Shogo Amimoto, Kumpei Kozuka, and Satoru Maekawa assisted with the experiment. The manuscript was written with contributions from all authors.
Conflicts of interest
The authors declare that there are no conflicts of interest.Data availability
The data that support the findings of this work are available in the supplementary information (SI). Supplementary information: details of the experimental methods, and additional figures and tables. See DOI: https://doi.org/10.1039/d6cc01367h.Acknowledgements
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers 25K01808 and 25K22857.References
- L. Guo, L. Wu, T. Jia, H. Zhang, J. Song, X. Xie, M. H. Jee, H. Ma, S. Liu, G. Lu, H. Y. Woo, Z. Wang, F. Gao and Y. Sun, Angew. Chem., Int. Ed., 2025, 64, e202516421 CrossRef CAS PubMed.
- D. Zhang, X. Zheng, Y. Zhao, C. Zhao, F. Huang and Y. He, J. Phys. Chem. C, 2024, 128, 7377–7387 CrossRef CAS.
- D. Lee, J. Moon, S. Kim and J. Y. Lee, Adv. Mater., 2026, e21668 CrossRef CAS PubMed.
- E. B. A. Adusei, S. Ibrahim, K. Jenneker, C. D. Goldsmith, D. Dragoi, M. Zeller and Z. J. Kinney, J. Org. Chem., 2026, 91, 394–400 CrossRef CAS PubMed.
- K. Kawabata, K. Mashimo and K. Takimiya, Chem. Mater., 2024, 36, 11920–11933 CrossRef CAS.
- K. Haase, J. P. Andrade, M. Hambsch, V. Sethumadhavan, W. He, T. Michinobu, P. Sonar and S. C. B. Mannsfeld, Adv. Electron. Mater., 2025, 11, e00375 CrossRef CAS.
- I. Osaka, T. Kakara, N. Takemura, T. Koganezawa and K. Takimiya, J. Am. Chem. Soc., 2013, 135, 8834–8837 CrossRef CAS PubMed.
- I. Osaka, T. Abe, S. Shinamura and K. Takimiya, J. Am. Chem. Soc., 2011, 133, 6852–6860 CrossRef CAS PubMed.
- S. Shinamura, E. Miyazaki and K. Takimiya, J. Org. Chem., 2010, 75, 1228–1234 CrossRef CAS PubMed.
- S. Shinamura, I. Osaka, E. Miyazaki, A. Nakao, M. Yamagishi, J. Takeya and K. Takimiya, J. Am. Chem. Soc., 2011, 133, 5024–5035 CrossRef CAS PubMed.
- M. E. Cinar and T. Ozturk, Chem. Rev., 2015, 115, 3036–3140 CrossRef CAS PubMed.
- J. Y. Kim, D. Yokoyama and C. Adachi, J. Phys. Chem. C, 2012, 116, 8699–8706 CrossRef CAS.
- H. Usta, D. Kim, R. Ozdemir, Y. Zorlu, S. Kim, M. C. R. Delgado, A. Harbuzaru, S. Kim, G. Demirel, J. Hong, Y.-G. Ha, K. Cho, A. Facchetti and M.-G. Kim, Chem. Mater., 2019, 31, 5254–5263 CrossRef CAS.
- H. Tan, H. Tan, X. Zheng, J. Yang, J. Yu and W. Zhu, J. Mater. Chem. C, 2020, 8, 3183–3191 RSC.
- M. Liang and J. Chen, Chem. Soc. Rev., 2013, 42, 3453–3488 RSC.
- P. Amaladass, R. Dhanusuraman, A. Lazer, D. John, K. Thangaraju and V. Dhayalan, Asian J. Org. Chem., 2025, 14, e202500085 CrossRef CAS.
- Y. Ooyama and K. Imato, Tetrahedron Lett., 2025, 155, 155393 CrossRef CAS.
- K.-W. Lee, Y. Cao, W.-C. Wei, J.-H. Tan, Y. Wan, Z. Feng, Y. Zhang, Y. Liu, X. Zheng, C. Cao, H. Chen, P. Wang, S. Li, K.-T. Wong and C.-S. Lee, Adv. Mater., 2023, 35, 2211632 CrossRef CAS PubMed.
- D. Yan, M. Wang, Q. Wu, N. Niu, M. Li, R. Song, J. Rao, M. Kang, Z. Zhang, F. Zhou, D. Wang and B. Z. Tang, Angew. Chem., Int. Ed., 2022, 61, e202202614 CrossRef CAS PubMed.
- D. Yan, W. Xie, J. Zhang, L. Wang, D. Wang and B. Z. Tang, Angew. Chem., Int. Ed., 2021, 60, 26769–26776 CrossRef CAS PubMed.
- Y. Wan, G. Lu, W.-C. Wei, Y.-H. Huang, S. Le, J.-X. Chen, X. Cui, Y.-F. Xio, X. Li, Y. Liu, X.-M. Meng, P. Wang, H.-Y. Xie, J. Zhang, K.-T. Wong and C.-S. Lee, ACS Nano, 2020, 14, 9917–9928 CrossRef CAS PubMed.
- X. Chen, D. Zhang, Y. He, M. U. Ali, Y. Wu, C. Zhao, P. Wu, C. Yan, F. Wudl and H. Meng, Mater. Chem. Front., 2020, 4, 3578–3584 RSC.
- K. Yamamoto, Y. Ie, M. Nitani, N. Tohnai, F. Kakiuchi, K. Zhang, W. Pisula, K. Asadi, P. W. M. Blom and Y. Aso, J. Mater. Chem. C, 2018, 6, 7493–7500 RSC.
- Y.-C. Hu, Z.-L. Lin, T.-C. Huang, J.-W. Lee, W.-C. Wei, T.-Y. Ko, C.-Y. Lo, D.-G. Chen, P.-T. Chou, W.-Y. Hung and K.-T. Wong, Mater. Chem. Front., 2020, 4, 2029–2039 RSC.
- K. Obayashi, K. Imato, S. Aoyama, T. Enoki, S. Akiyama, M. Ishida, S. Suga, K. Mitsudo and Y. Ooyama, RSC Adv., 2021, 11, 18870–18880 RSC.
- Y. Hara, K. Kozuka, K. Imato, S. Akiyama, M. Ishida and Y. Ooyama, New J. Chem., 2024, 48, 9890–9898 RSC.
- T. Higashino, Y. Hara, K. Imato, S. Akiyama, M. Ishida and Y. Ooyama, New J. Chem., 2023, 47, 9555–9559 RSC.
- K. Obayashi, S. Miho, M. Yasui, K. Imato, S. Akiyama, M. Ishida and Y. Ooyama, New J. Chem., 2021, 45, 17085–17094 RSC.
- K. Obayashi, T. Higashino, K. Imato and Y. Ooyama, New J. Chem., 2021, 45, 13258–13261 RSC.
- Y. Ooyama, T. Enoki, S. Aoyama and J. Ohshita, Org. Biomol. Chem., 2017, 15, 7302–7307 RSC.
- E. Clar, The Aromatic Sextet, John Wiley & Sons, 1972 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |