Open Access Article
Open Access ArticleA dual-metal strategy for N-heterocycle coordination using nickel and aluminum
Gabriel J. Jobin and
Marcus W. Drover *
*
Department of Chemistry, Western University, 1151 Richmond St, London, ON N8K 3G6, Canada. E-mail: marcus.drover@uwo.ca
First published on 7th May 2026
Abstract
We present a new ambiphilic P–Al ligand that preorganizes N-heterocycles for nickel coordination, affording well-defined co-activated complexes that are commonly proposed, but rarely observed, intermediates in N-heterocycle upgrading.
Nitrogen-containing heterocycles are ubiquitous in biologically active molecules, comprising approximately 82% of FDA-approved drugs from 2013–23.1,2 The selective functionalization of these privileged motifs remains a central objective in synthetic chemistry, particularly due to the difficulty of achieving site-selective C–H bond activation.3,4 Transition metal-catalyzed C–H bond activation offers a promising avenue, yet the inherent Lewis basicity of N-heterocycles often leads to unproductive or poorly selective outcomes.5
A detailed understanding of substrate coordination behavior is critical for expanding the toolkit of heterocycle functionalization strategies. Key mechanistic questions: how is substrate activated? what is the role of the ligand? how do geometric constraints govern product formation? lie at the heart of ligand and catalyst design. One emerging strategy involves heterobimetallic complexes, in which a main-group Lewis acid cooperatively engages a substrate alongside a transition metal catalyst.1,6–24 In such systems, the Lewis acid (e.g., a trivalent aluminum species) serves to anchor the N-heterocycle in a defined orientation, thereby facilitating selective coordination by an adjacent transition metal.9,10,25
Progress in this area has been marked by the use of trivalent aluminum additives as components of “catalytic cocktails” where Al and a transition metal (often nickel) work in synergy to co-activate a given substrate (Scheme 1a).15,26,27 While mixtures of Al, Ni, and L-type donor ligands such as carbenes and phosphines have been proven adept for N-heterocycle upgrading catalysis, examples where substrate has been unambiguously characterized in the coordination spheres of both metals are rare.13 A notable exception is the 2010 work of Yap and co-workers, who reported the crystallographic characterization of a Ni(0)–Al(III) η2-pyridine intermediate, displaying a formal N–Al dative interaction with Ni(0) coordinating to the pyridine π-system, (Scheme 1b). This dual activation prevents the more common coordination mode associated with pyridine(N) acting as a σ-donor to Ni, and was shown to be catalytically relevant, serving as an intermediate in the selective para-alkynylation of heteroaryl substrates (Scheme 1b).13
 | ||
| Scheme 1 (a) {Ni,Al} Co-catalysis for pyridine upgrading reactivity; (b) an example of a co-activated intermediate. | ||
While ill-defined mixtures of {Al, Ni, and L-type ligands} have seen application in N-heterocycle functionalization, bringing all three components (Ni(Ln), Al, substrate) together comes at a significant entropic cost, often requiring high concentrations of aluminum additive. Ambiphilic ligands comprising a donor (such as a phosphine) and pendent Lewis acid reduce the barrier for substrate activation.6,28 For {Ni,Al} heterometallics, so-called ambiphilic ligands have shown improved selectivity and reactivity, due to synergistic directing effects.9,29 Implementation of such scaffolds has enabled catalytic transformations not readily accessible using traditional ligand architectures, including unconventional C3-selective pyridine olefination.7
Here, we report the isolation and characterization of a novel aluminum–phosphorus ambiphilic ligand that enables cooperative η2-pyridine/quinoline coordination. This distinctive binding motif arises from a cooperative interaction between a secondary-sphere aluminum(III) Lewis acid and a nickel(0) center. The emergence of this motif provides key structural insight into the nature of substrate activation within a bimetallic system.30,31 Using this pair, dual activation of the heterocycle is achieved using a nickel–diphosphine template32 featuring a secondary coordination sphere Lewis acid.
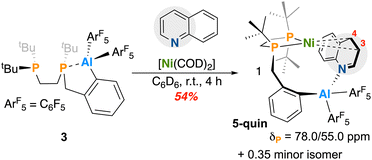
The phosphine electrophile, 1 was selected as a suitable host for introduction of both phosphine and alane functionality. The polarized P–I bound in 1 readily reacts with o-bromobenzylmagnesium bromide at −78 °C to afford 2, bearing three bulky tert-butyl groups and an ortho-benzyl bromide site amenable to Lewis acid introduction (Scheme 1).33 Perfluoroaryl rings significantly enhance Lewis acidity, and so {Cl–Al(C6F5)2}2 was selected as the alane partner of choice. To facilitate lithium halogen exchange, 2 was reacted with 1 equiv. of n-BuLi and stirred at room temperature. Addition of 0.5 equiv. {Cl–Al(C6F5)2}2 quenched reaction color and after filtration, 3 was isolated as a colorless oil in 81% yield. It is important to note that despite repeated purification attempts, including variation of solvent systems and crystallization conditions, compound 3 could not be isolated in 100% analytically pure form and contained some –P(tBu)2-containing impurities (Fig. S3), however, it remains a valuable precursor to coordination complexes (vide infra), whose distinct solubility profiles enabled more straightforward purification. By atmospheric pressure chemical ionization mass spectrometry (APCI-MS), a [M]+ signal at m/z = 713.211 (calcd. = 713.210 for C33H38AlF10P2) of the appropriate isotope pattern was observed. By 31P{1H} NMR spectroscopy, two doublets in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio at δP = 37.4 and 1.13 ppm (JP,P = 39.3 Hz) were additionally seen for 3; notably, the upfield shifted signal is significantly broadened due to the presence of an intramolecular P–Al interaction (owing to the quadrupolar nature of 27Al nucleus (I = 5/2)). 19F{1H} NMR spectroscopy further supported adduct formation, with the distance between para- and meta-aryl fluorine signals being close (Δp,m = 8.2 ppm).34 Formation of a five membered –P–(C)3–Al ring is additionally shown by significant splitting of the benzylic CH2 protons, which appear as two doublets of doublets at δH = 3.21 and 2.63 ppm (2JH,H = 16.4 Hz). These peaks are significantly shifted from precursor 2, which has overlapping benzylic signals at δH = 2.93 ppm.33
1 ratio at δP = 37.4 and 1.13 ppm (JP,P = 39.3 Hz) were additionally seen for 3; notably, the upfield shifted signal is significantly broadened due to the presence of an intramolecular P–Al interaction (owing to the quadrupolar nature of 27Al nucleus (I = 5/2)). 19F{1H} NMR spectroscopy further supported adduct formation, with the distance between para- and meta-aryl fluorine signals being close (Δp,m = 8.2 ppm).34 Formation of a five membered –P–(C)3–Al ring is additionally shown by significant splitting of the benzylic CH2 protons, which appear as two doublets of doublets at δH = 3.21 and 2.63 ppm (2JH,H = 16.4 Hz). These peaks are significantly shifted from precursor 2, which has overlapping benzylic signals at δH = 2.93 ppm.33
The Lewis acid–base interaction in 3 is highly stabilizing. Computational analysis of the ring-opened and -closed forms reveals an energy difference of ΔG° = −22.6 kcal mol−1 in favor of the ring-closed product. Taking advantage of the ability of aluminum to host coordination numbers greater than four, we wondered if the ring system in 3 could be ‘primed’ for metal-induced ring-opening through addition of a target N-heterocycle, granting a five-coordinate aluminum adduct.
In a representative reaction, complex 3 was reacted with pyridine. Upon treatment of 3 with 1.5 equivs. pyridine, two new signals at δP = 37.3 and −1.10 ppm were observed by 31P{1H} NMR spectroscopy; for the signal ascribed to the P–(C)3–Al arm at δP = −1.10 ppm, a slight shift from 1.12 ppm is consistent with a change in aluminum coordination environment to give 3-pyr (Scheme 2). Broadness of this 31P NMR spectroscopic signal (despite the addition of excess heterocycle) and the persistence of diastereotopic CH2 groups by 1H NMR spectroscopy, supports a maintained P–Al interaction in 3-pyr. Notably, dissolution of 3 in the weaker donor solvent, THF results in similarly shifted 31P NMR resonances (δP = 36.6 and −3.4 ppm), suggestive of adduct formation to give 3-THF. For THF, this reaction was found to be reversible on exposure to vacuum, returning complex 3; this is not the case for pyridine as 3-pyr persists despite the introduction of vacuum. Extended exposure of 3 to excess pyridine leads to decomposition, affording aluminum-free products (a diphosphine ligand containing a benzyl pendent group) via cleavage of the Al–C bond.
 | ||
| Scheme 2 Synthetic pathway towards aluminum-functionalized diphosphine 2 and its coordination to nickel(0). ΔG° value calculated using DLPNO-CCSD(T) (see SI). | ||
Adduct formation to give 3-pyr and 3-THF is thermodynamically downhill (ΔG° = −26.4 kcal mol−1 for pyridine (3-pyr) and ΔG° = −22.4 kcal mol−1 for THF (3-THF)). The optimized structure of both depicts a distorted trigonal bipyramidal alumatrane35–37 where three X-type ligands are arranged equatorially, with two dative donors occupying both axial coordination sites. Upon adduct formation, the P–Al bond length elongates from 2.432 Å to 2.621 and 2.593 Å, for 3-pyr and 3-THF, respectively. This outcome supports our hypothesis: addition of an L-type donor to 3 weakens the P–Al interaction.
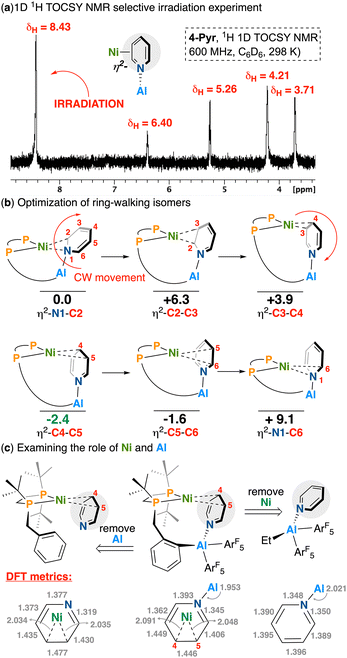
We next studied metalation. As implied by the stability of 3, its combination with [Ni(COD)2] (COD = 1,5-cyclooctadiene) in C6D6 resulted in null reactivity. Given the propensity of 3 to coordinate weak donors such as THF, however, we wondered whether ring-opening could be donor-induced. In accord, the abovementioned reaction was repeated in the presence of 1 equiv. of pyridine, giving 3-pyr in situ. Addition of [Ni(COD)]2 to this mixture generated two signals at δP = 79.7 and 56.7 ppm (JP,P = 58.5 Hz) by 31P{1H} NMR spectroscopy. Pyridine coordination and COD dissocation at nickel was confirmed by LIFDI-MS where a [M]+ signal was observed at m/z = 849.179 (calcd. = 849.180 for C38H42NAlF10NiP2+) of the appropriate isotope pattern. This compound was assigned as 4-pyr, whose chemical shifts are close to a related η2-bonded aryl Ni diphosphine complex synthesized by Fryzuk, which features 31P peaks at δP = 79.5 and 40.2 ppm (JP,P = 102.9 Hz).33 Of note, 4-pyr can exist as one of six possible isomers: two of which are η2-N,C and four of which are η2-C,C bound. These isomeric possibilities are discussed in detail below.
Upon work-up, the 1H NMR spectrum of 4-pyr displayed broadened peaks at δH = 4.21 and 3.71 ppm, both significantly upfield of typical pyridine 1H environments (δH = 6.66–8.53 ppm in C6D6).38 Three additional resonances (five total) include a multiplet at δH = 5.26 and broadened downfield-shifted signals at 6.40 and 8.43 ppm, which exhibit clear coupling by 1H–1H COSY NMR spectroscopy. These shifts, assigned to a major pyridine-bound isomer, are partnered with a second, minor secondary component having similar chemical shifts, suggesting the presence of isomers (vide supra) – this is shown in the 1D TOCSY NMR spectrum depicted in Scheme 3a. Proving these five signals are pyridine derived, use of pyridine-d5 gives 4-pyr-d5, having the same 31P signatures though the five pyridine 1H NMR signals described previously are expectedly absent.
Altogether, these features are consistent with a high degree of nickel (dπ)-to-pyridine (CC)π* back-donation, an interaction that is mediated by aluminum. This supposition is further buttressed by a lack of observed exchange between 4-pyr-d5 and free pyridine. It is worth underscoring that despite the ubiquity of heterocycle upgrading reactions using nickel and their linked intermediacy in pyridine functionalization reactions – examples of late metal η2-pyridine complexes such as these are exceptionally rare.
Despite our best efforts, X-ray quality crystals of 4-pyr could not be obtained, prompting the use of computational chemistry to assess structural possibilities. Although this rules out an unambiguous assignment, our NMR spectroscopic (symmetry/chemical shifts) and computational data (energies) suggest the η2-C4-C5 and η2-C3-C4 geometries to be most likely (Scheme 3b). Indeed, given the inherent asymmetry found in the Ni(diphosphine) framework, six pyridine-bound isomers are all possible, each of which reveals significant disruption of pyridine aromaticity. By arbitrarily assigning the η2-N1-C2 geometry as having an energy of 0 kcal mol−1, movement in a clockwise geometry cycles through 5/6 possible isomers with the η2-C4–C5 isomer being the global minimum.39–41 For the η2-C4–C5 isomer, the Ni-bound C–C bond is elongated to 1.446 Å (Scheme 3b). In contrast, optimized structures of both free pyridine and its adduct with Al(C6F5)2Et display uniform bond lengths consistent with aromatic character (pyridine: C–N = 1.342 Å, C–C = 1.398, 1.395 Å; pyridine–Al(C6F5)2Et: C–N = 1.35 Å, C–C = 1.39, 1.40 Å). To evaluate the individual contributions of Ni and Al to substrate activation, we also optimized a structure in which pyridine is η2-C4–C5-bound to Ni in the absence of Al. While this species also shows dearomatization, its C–N bonds are shorter (1.377, 1.319 Å) compared to those in η2-C4–C5 4-pyr (1.393, 1.345 Å), suggesting a cooperative activation effect from both metal centers.
Expanding scope to a less symmetric heterocycle, we next assessed the reactivity of 3 with quinoline. Unlike pyridine, quinoline could give a series of two isomers that differ in the position of the quinoline N-heterocycle (whether it is syn- or anti-) in relation to the P-Al-containing arm (Scheme 4). By arbitrarily assigning the syn-η2-N1–C2 geometry as having an energy of 0 kcal mol−1, clockwise movement from syn-η2-N1-C2 through to syn-η2-C4-C5 shows syn-η2-N1-C2 to be a minimum, whereas the syn-η2-C4-C5 isomer was the global maximum (+ 10 kcal mol−1); η2-C5-C6 and η2-C6-N1 isomers were not modelled for either syn- or anti- as these include an interaction between Ni and a ring junction carbon (which would, in principle, dearomatize the adjoining benzene ring, incurring an energetic penalty), as was observed from the η2-C4-C5 isomer.
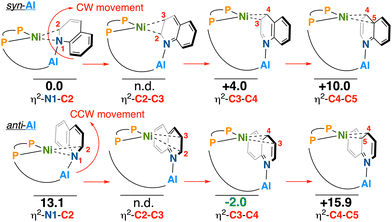 | ||
| Scheme 4 Optimization of quinoline ring-walking isomers. ΔG° values in kcal mol−1, calculated using DLPNO-CCSD(T) (see SI). n.d. = not determined/located. | ||
The syn-η2-C2-C3 was not located as it converges to the more stable syn-η2-C1-C2 isomer. Efforts to obtain a structure for the anti-η2-C2-C3 isomer (Scheme 4) resulted in the most stable anti-η2-C3-C4 isomer (−2.0 kcal mol−1), though the syn-η2-N1-C2 and syn-η2-C4-C5 were calculated to be high in energy (+13.1 and +15.9 kcal mol−1, respectively). Repeated convergence to the anti-η2-C3-C4 isomer structure suggests this coordination mode is a dominant minimum on the potential energy surface.42
In practice, reaction of 3, [Ni(COD)2], and quinoline resulted in an instant color change from yellow to red. By 31P{1H} NMR spectroscopy, two major doublets at δP = 78.0 and 55.0 ppm (JP,P = 44.0 Hz) were revealed, assigned as an isomer of 5-quin; a minor product was also observed at δP = 86.3 and 60.9 ppm (JP,P = 39.0 Hz) (Scheme 5). Atmospheric pressure chemical ionization mass spectrometry (LIFDI-MS) confirmed quinoline inclusion, giving an [M]+ signal at m/z = 899.195 (calcd. = 899.195 for C42H44AlF10NNiP2). Further support for the structure of 5-quin is provided from 1H NMR spectroscopy, which displays two broadened upfield-shifted signals corresponding to activated quinoline at δH = 4.73 and 3.86 ppm. These resonances exhibit clear through-bond coupling by 1H–1H COSY NMR spectroscopy and show 31P coupling to the P(tBu)(BnAl) arm by 1H–31P HMBC NMR spectroscopy. Given the similarity of the 1H chemical shifts in the two isomers, including δH = 4.90 and 3.92 ppm for the minor, this isomer likely arises by the benzenoid ring orienting on either side of the molecule.
A new ambiphilic diphosphine ligand featuring a dative P–Al interaction was synthesized. Although the interaction is strong, addition of an N-heterocycle weakens the Al-centered Lewis adduct, enabling metalation with Ni(0). The resulting complexes exhibit η2-coordination of the N-heterocycle to nickel. These results demonstrate a new approach for cooperative binding and activation of valuable N-heterocycles.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, spectroscopic data, and computational methods. See DOI: https://doi.org/10.1039/d6cc01343k.Acknowledgements
The authors are grateful to Western University, the Council of Ontario Universities for a John C. Polanyi award to M. W. D., the Canadian Foundation for Innovation (LOF-212442), and the Natural Sciences and Engineering Research Council of Canada (Discovery Grant, RGPIN-2020-04480 (M. W. D.), Discovery Launch Supplement, DGECR-2020-00183), and graduate award (CGRS-D to G. J.) for funding.References
- V. T. Nguyen, R. N. Sladek, Y. Cao, N. Bhuvanesh, J. Zhou and O. V. Ozerov, J. Am. Chem. Soc., 2024, 146, 31281–31294 CrossRef CAS PubMed.
- C. M. Marshall, J. G. Federice, C. N. Bell, P. B. Cox and J. T. Njardarson, J. Med. Chem., 2024, 67, 11622–11655 CrossRef CAS PubMed.
- H. Cao, Q. Cheng and A. Studer, Angew. Chem., Int. Ed., 2023, 62, e202302941 CrossRef CAS PubMed.
- S. Maity, A. Bera, A. Bhattacharjya and P. Maity, Org. Biomol. Chem., 2023, 21, 5671–5690 RSC.
- S. M. Khake and N. Chatani, Chemistry, 2020, 6, 1056–1081 CrossRef CAS.
- A. D. Dilinaer, G. J. Jobin and M. W. Drover, Dalton Trans., 2024, 53, 13298–13307 RSC.
- T. Zhang, Y.-X. Luan, N. Y. S. Lam, J.-F. Li, Y. Li, M. Ye and J.-Q. Yu, Nat. Chem., 2021, 13, 1207–1213 CrossRef CAS PubMed.
- J.-F. Li, D. Pan, H.-R. Wang, T. Zhang, Y. Li, G. Huang and M. Ye, J. Am. Chem. Soc., 2022, 144, 18810–18816 CrossRef CAS PubMed.
- Y.-X. Luan and M. Ye, Chem. Commun., 2022, 58, 12260–12273 RSC.
- Y. Nakao, K. S. Kanyiva and T. Hiyama, J. Am. Chem. Soc., 2008, 130, 2448–2449 CrossRef CAS PubMed.
- M. M. Shoshani, Cell Rep. Phys. Sci., 2023, 4, 101213 CrossRef CAS.
- V. Singh, Y. Nakao, S. Sakaki and M. M. Deshmukh, J. Org. Chem., 2017, 82, 289–301 CrossRef CAS PubMed.
- C.-C. Tsai, W.-C. Shih, C.-H. Fang, C.-Y. Li, T.-G. Ong and G. P. A. Yap, J. Am. Chem. Soc., 2010, 132, 11887–11889 CrossRef CAS PubMed.
- N. Hara, N. Uemura and Y. Nakao, Chem. Commun., 2021, 57, 5957–5960 RSC.
- Y. Kuroda, K. Park, Y. Shimazaki, R.-L. Zhong, S. Sakaki and Y. Nakao, Angew. Chem., Int. Ed., 2023, 62, e202300704 CrossRef CAS PubMed.
- H. Gao, L. Hu, Y. Hu, X. Lv, Y.-B. Wu and G. Lu, Catal. Sci. Technol., 2021, 11, 4417–4428 RSC.
- B. Jiang, J.-M. Liu and S.-L. Shi, ACS Catal., 2023, 13, 6068–6075 CrossRef CAS.
- J.-F. Li, W.-W. Xu, R.-H. Wang, Y. Li, G. Yin and M. Ye, Nat. Commun., 2021, 12, 3070 CrossRef CAS PubMed.
- J.-B. Ma, X. Zhao, D. Zhang and S.-L. Shi, J. Am. Chem. Soc., 2022, 144, 13643–13651 CrossRef CAS PubMed.
- W.-C. Shih and O. V. Ozerov, J. Am. Chem. Soc., 2017, 139, 17297–17300 CrossRef CAS PubMed.
- W.-C. Shih, W.-C. Chen, Y.-C. Lai, M.-S. Yu, J.-J. Ho, G. P. A. Yap and T.-G. Ong, Org. Lett., 2012, 14, 2046–2049 CrossRef CAS PubMed.
- R. P. Singh, S. Sinhababu and N. P. Mankad, ACS Catal., 2023, 13, 12519–12542 CrossRef CAS.
- Y.-X. Wang, S.-L. Qi, Y.-X. Luan, X.-W. Han, S. Wang, H. Chen and M. Ye, J. Am. Chem. Soc., 2018, 140, 5360–5364 CrossRef CAS PubMed.
- J. Zhou and B.-F. Shi, Transition-Metal-Catalyzed C–H Functionalization of Heterocycles, 2023, pp. 357–392 Search PubMed.
- Y. Nakao, Y. Yamada, N. Kashihara and T. Hiyama, J. Am. Chem. Soc., 2010, 132, 13666–13668 CrossRef CAS PubMed.
- J. Becica and G. E. Dobereiner, Org. Biomol. Chem., 2019, 17, 2055–2069 RSC.
- A. Bhunia, K. Bergander and A. Studer, J. Am. Chem. Soc., 2018, 140, 16353–16359 CrossRef CAS PubMed.
- G. Bouhadir and D. Bourissou, Chem. Soc. Rev., 2016, 45, 1065–1079 RSC.
- Y. Zhang, Q. Ni, L. Jiang, R. Cao and L. Qiu, Eur. J. Org. Chem., 2025, e202401100 CrossRef CAS.
- G. Song, B. Wang, M. Nishiura and Z. Hou, Chem. – Eur. J., 2015, 21, 8394–8398 CrossRef CAS PubMed.
- Q. Sun, P. Chen, Y. Wang, Y. Luo, D. Yuan and Y. Yao, Inorg. Chem., 2018, 57, 11788–11800 CrossRef CAS PubMed.
- A. C. Zimmerman and M. D. Fryzuk, Organometallics, 2019, 38, 3671–3679 CrossRef CAS.
- A. C. Zimmerman and M. D. Fryzuk, Organometallics, 2018, 37, 2305–2318 CrossRef CAS.
- N. A. Shcherbina, I. V. Kazakov, A. V. Pomogaeva, A. S. Lisovenko, M. A. Kryukova, D. A. Doinikov, N. Yu. Gugin, Y. V. Kondrat’ev and A. Y. Timoshkin, J. Organomet. Chem., 2021, 948, 121909 CrossRef CAS.
- A. K. Phukan and A. K. Guha, Inorg. Chem., 2011, 50, 1361–1367 CrossRef CAS PubMed.
- W. Su, Y. Kim, A. Ellern, I. A. Guzei and J. G. Verkade, J. Am. Chem. Soc., 2006, 128, 13727–13735 CrossRef CAS PubMed.
- W. Su, J. Kobayashi, A. Ellern, T. Kawashima and J. G. Verkade, Inorg. Chem., 2007, 46, 7953–7959 CrossRef CAS PubMed.
- G. R. Fulmer, A. J. M. Miller, N. H. Sherden, H. E. Gottlieb, A. Nudelman, B. M. Stoltz, J. E. Bercaw and K. I. Goldberg, Organometallics, 2010, 29, 2176–2179 CrossRef CAS.
- A. Nova, M. Reinhold, R. N. Perutz, S. A. Macgregor and J. E. McGrady, Organometallics, 2010, 29, 1824–1831 CrossRef CAS.
- S. A. Johnson, N. M. Mroz, R. Valdizon and S. Murray, Organometallics, 2011, 30, 441–457 CrossRef CAS.
- T. A. Ateşin, T. Li, S. Lachaize, J. J. García and W. D. Jones, Organometallics, 2008, 27, 3811–3817 CrossRef.
- J. J. Garcia, N. M. Brunkan and W. D. Jones, J. Am. Chem. Soc., 2002, 124, 9547–9555 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |