Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Unlocking efficient near-infrared circularly polarized phosphorescence reaching 800 nm in cyclometalated Pt(II) complexes
Shilin
Gu
a,
Dongsheng
Li
a,
Deng
Long
 a,
Xinglin
Yu
a,
Wentao
Li
a,
Xinglin
Yu
a,
Wentao
Li
 *a,
Sihan
Ma
*a and
Peng
Tao
*a,
Sihan
Ma
*a and
Peng
Tao
 *b
*b
aInstitute of Advanced Optoelectronic Materials and Technology, College of Big Data and Information Engineering, Guizhou University, Guiyang 550025, P. R. China. E-mail: wtli@gzu.edu.cn; shma@gzu.edu.cn
bDepartment of Applied Biology and Chemical Technology, The Hong Kong Polytechnic University, Hung Hom, Hong Kong, P. R. China. E-mail: pengtao@polyu.edu.hk
First published on 22nd April 2026
Abstract
We report a promising strategy to design efficient NIR chiral Pt(II) complexes via ligand regulation and planar chirality formation, affording binuclear Pt(II) enantiomers that exhibit unprecedented NIR CPP reaching 800 nm, photoluminescence quantum yields of up to 35.4%, and luminescence dissymmetry factors of ∼3.9 × 10−3.
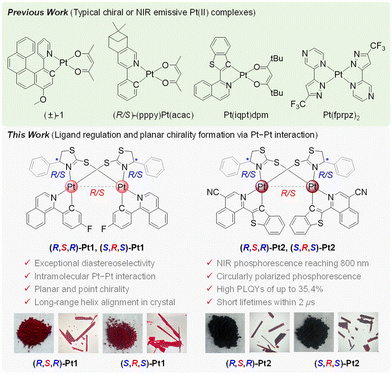
Near-infrared (NIR) circularly polarized (CP) luminescent (CPL) materials are promising for use in both biomedical engineering and optoelectronics.1 Traditional methods of generating CP light via polarizers are energy-intensive.1 Developing chiral luminescent materials that directly emit NIR CPL light is crucial. Integrating chirality into high-efficiency NIR emitters is therefore key to creating direct NIR CPL light sources with minimal energy loss. Phosphorescent transition-metal complexes (PTMCs) are particularly attractive because strong spin–orbit coupling (SOC) enables full utilization of both singlet and triplet excitons,2 resulting in near-unity internal quantum efficiency.3 Among PTMCs, Pt(II) complexes are promising candidates owing to the easy realization of long-wavelength emission.2,3 However, the traditional HC^N ligand-based Pt(II) complexes usually suffer from low photoluminescence (PL) quantum yields (PLQYs) in the NIR region due to the lack of strong intermolecular interactions, leading to inefficient intersystem crossing (ISC) from the singlet excited state (S1) to the triplet excited state (T1).4 Achieving Pt(II) complexes with high-efficiency near-infrared circularly polarized phosphorescence (CPP) remains a formidable challenge.1
The Pt–Pt interactions could promote the overlap of metal-centred orbitals, resulting in the formation of a low-energy triplet metal–metal-to-ligand charge transfer (3MMLCT) state, which typically serves as an energy trap.3c,5 The enhanced metal character strengthens SOC, facilitating both ISC from S1 to T1 and formally forbidden triplet-to-singlet radiative transition, thereby increasing the radiative decay rate.5 Meanwhile, the ordered molecular alignment induced by Pt–Pt interactions restricts ligand vibrations and rotations, effectively suppressing non-radiative decay pathways linked to molecular motion and significantly lowering the non-radiative decay rate.5 Thus, engineering Pt–Pt interactions offers a promising strategy to reduce non-radiative loss in low-energy NIR Pt(II) emitters by enhancing the radiative decay rate constant and ultimately improving their PLQYs. Furthermore, although Pt(II) complexes can be endowed with CPP upon the introduction of helical or point chirality,6 intramolecular Pt–Pt interactions could also provide an opportunity to generate planar chirality for realizing circularly polarized phosphorescence of Pt(II) complexes.5
In this communication, a promising strategy was proposed to design highly efficient NIR chiral Pt(II) complexes ((R,S,R)-Pt1, (S,R,S)-Pt1, (R,S,R)-Pt2, and (S,R,S)-Pt2) via rational ligand regulation and planar chirality formation (Scheme 1). Two isoquinoline derivatives (i.e., 1-(4-fluorophenyl)isoquinoline and 1-(benzo[b]thiophen-2-yl)isoquinoline-4-carbonitrile) with low triplet energy levels were selected as the cyclometalating ligands. The point chirality of the bridging ligand enables exceptional diastereoselectivity promoted by the intramolecular Pt–Pt interaction, affording pure binuclear Pt(II) enantiomers that exhibit unprecedented NIR CPP reaching 800 nm, notable PLQYs of up to 35.4%, short excited state lifetimes within 2 µs, and appreciable luminescence dissymmetry factors (glum) of up to ∼3.9 × 10−3. To the best of our knowledge, this is the first example of highly efficient NIR chiral binuclear Pt(II) complexes with CPP maxima reaching 800 nm.1
The target Pt(II) complexes were obtained as deep red or dark maroon solids by heating a chiral bridging ligand with the Pt(II) intermediate [Pt(F-piq)(F-Hpiq)Cl] or [Pt(CN-iqbt)(CN-Hiqbt)Cl] in the presence of K2CO3 at 80 °C in a sealed tube with CH2Cl2 under an argon atmosphere (Scheme S1). Notably, in all coordination reactions, only one enantiomerically pure product could be isolated, indicating nearly perfect diastereoselectivity (close to 100%). This high selectivity is likely driven by the point chirality of the chiral bridging ligand, which is in good agreement with previously reported observations.5b,c These Pt(II) enantiomers were well characterized by nuclear magnetic resonance (NMR) spectroscopy, elemental analysis, and mass spectrometry (MS). 1H, 13C{1H}, and 19F{1H} NMR spectra all exhibit distinct and well-resolved resonance signals. The typical chemical shifts of the 1H, 13C, and 19F nuclei are provided in Table S1. For each pair of enantiomers, identical NMR spectra are observed in CDCl3, which is attributed to their identical atomic connectivity and equivalent electronic environments surrounding the nuclei. Only one 19F resonance signal (∼−110.87 ppm) was observed for (R,S,R)-Pt1 and (S,R,S)-Pt1, implying the presence of molecular symmetry in binuclear complexes.
The Pt(II) enantiomers were unambiguously confirmed by single-crystal X-ray diffraction analysis (Table S2). The crystallographic data revealed that the Pt(II) centres adopt a slightly distorted square-planar geometry. In this configuration, the nitrogen atoms from both the cyclometalated and bridging ligands are coordinated in a trans arrangement, while the metalated carbon and the sulfur atom from the adjacent bridging ligand are coordinated in a cis manner, which aligns with previously reported complexes.5,7 The bond distances for Pt–C, Pt–N, and Pt–S fall within the ranges of 1.98–2.02 Å, 2.04–2.15 Å, and 2.28–2.30 Å, respectively (Table S3). Notably, the Pt–N bond lengths vary considerably depending on the N-donor strength of the ligands, with a significantly elongated Pt–N bond observed for the bridging ligand. Furthermore, the C–Pt–N bond angles are ∼80° (Table S3), indicating a minor deviation from ideal square-planar geometry, which can be attributed to the Jahn–Teller effect.5,7 As shown in Fig. 1 and Fig. S1, short intramolecular Pt–Pt distances were observed, measuring 2.922 and 2.910 Å for (R,S,R)/(S,R,S)-Pt1 enantiomers, and 2.941 and 2.948 Å for the (R,S,R)/(S,R,S)-Pt2 pair. The slightly longer Pt–Pt distances in the Pt2 complexes compared to those in the Pt1 pair suggest that the intramolecular Pt–Pt interactions can be modulated by the choice of cyclometalating ligands. The two cyclometalated ligands are oriented in a head-to-tail manner. In all crystal structures, intermolecular face-to-face π–π stacking interactions with distances ranging from 3.37 to 3.42 Å are observed between partially overlapping cyclometalated ligands of adjacent molecules (Fig. S1, S2 and Table S4). Additionally, various weak intermolecular contacts including S···H and F···H interactions with distances below 3 Å are present throughout the crystals, contributing to the formation of a long-range helical arrangement (Fig. S3 and Table S4).
To explore the optical properties of Pt(II) complexes, their ultraviolet (UV)–visible (vis) absorption spectra were measured in CH2Cl2 at 1.0 × 10−5 M at ambient temperature. As depicted in Fig. 2a, the complexes exhibit typical absorption profiles. An intense absorption band observed in the high-energy region (240–380 nm) originates from the π → π* transitions of the ligands.5 Conversely, the weaker and lower-energy absorption band spanning 380–700 nm is associated with MMLCT transitions (Table S5).5 To gain deeper insight into the electronic transition characteristics, time-dependent density functional theory (TD-DFT) calculations were performed.8 The simulated absorption profiles of the complexes in CH2Cl2 are in good agreement with the experimental spectra (Fig. S4). The optimized S0 and T1 geometries of the complexes and their Pt–Pt distances are shown in Fig. S5. For all Pt(II) complexes, the low-energy absorption bands predominantly arise from the transition between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), with a contribution exceeding 99% (Table S6). As depicted in Fig. 3, the HOMO electron density is mainly localized on the two Pt centres, whereas the LUMO is largely distributed across the cyclometalated ligands. This distribution further corroborates the assignment of the low-energy absorption to MMLCT transitions. As listed in Table S7, the Pt dπ orbital contributions to the HOMO and LUMO are 78.97% and 6.18% for (R,S,R)-Pt1, 79.10% and 6.00% for (S,R,S)-Pt1, 73.77% and 7.27% for (R,S,R)-Pt2, and 75.69% and 7.02% for (S,R,S)-Pt2, respectively. Meanwhile, the contributions from the π orbitals of the cyclometalated ligands to the HOMO and LUMO are 11.70% and 92.00% for (R,S,R)-Pt1, 11.39% and 92.25% for (S,R,S)-Pt1, 17.05% and 90.77% for (R,S,R)-Pt2, and 14.99% and 91.17% for (S,R,S)-Pt2, respectively. The TD-DFT-calculated HOMO–LUMO energy gaps are 2.99 and 2.94 eV for the Pt1 enantiomers, and 2.71 and 2.67 eV for the Pt2 pair.
 | ||
| Fig. 3 Calculated electron cloud distributions and the orbital energy levels of the LUMO and HOMO for Pt(II) complexes at their geometrical optimized ground state. | ||
Upon UV light excitation, all Pt(II) enantiomers exhibit intense NIR phosphorescence both in degassed CH2Cl2 and in poly(methyl methacrylate) (PMMA) films doped at 5 wt% under ambient conditions. As depicted in Fig. 2b and c, the Pt1 enantiomers show emission maxima at approximately 733 nm in degassed CH2Cl2 and around 709 nm in the PMMA matrix. In contrast, the Pt2 pair displays significantly red-shifted phosphorescence reaching 800 nm in both media, implying that the emission originates from individual molecules and the emission energies could be further decreased by lowering the triplet energy levels of cyclometalated ligands in the binuclear Pt(II) systems featuring strong intramolecular Pt–Pt interactions. Notably, the PLQYs of these complexes increase substantially upon dispersion in the rigid PMMA matrix, increasing from 0.178–0.21 in degassed CH2Cl2 to 0.285–0.354 in the doped films (Table S5). This enhancement indicates that non-radiative decay pathways are effectively suppressed in the rigid environment. Fig. 2d depicts the comparison of the PLQYs of the designed Pt(II) complexes in this work with those of the reported Ir(III) and Pt(II) complexes showing PL in the NIR region, highlighting the effectiveness of the molecular design strategy employed in this work. The emission is further characterized by large Stokes shifts and long-lived excited states with lifetimes ranging from 0.32 to 0.40 µs in degassed CH2Cl2 and from 1.25 to 1.84 µs in PMMA films (Fig. S6 and Table S5). These features are consistent with phosphorescence originating from triplet excited states.5 The observed long-wavelength NIR phosphorescence at room temperature can be ascribed to the intrinsically low triplet energy levels of the cyclometalated ligands, combined with the formation of triplet 3MMLCT states facilitated by strong intramolecular Pt–Pt and π–π interactions.
The electrochemical behaviour of the Pt(II) complexes was investigated by cyclic voltammetry in CH3CN (Fig. S7). Oxidation potentials centred on the Pt atoms were determined to be 0.61/0.60 eV for the Pt1 enantiomers and 0.89/0.88 eV for the Pt2 pair (Table S8). This assignment is supported by DFT calculations, which reveal that the HOMO is predominantly localized on the Pt centers (Pt dπ orbital contributions: 73.77–79.10%), with minimal ligand contribution. From the absorption edges, the HOMO and LUMO energy levels were calculated to be –4.90/–4.89 eV and –2.93/–2.92 eV for Pt1 enantiomers, and –5.18/–5.17 eV and –3.58/–3.57 eV for the Pt2 pair, respectively.
The chiroptical properties of the Pt(II) complexes were further evaluated in CH2Cl2 using circular dichroism (CD) and CPL spectroscopy. As shown in Fig. 4, mirror-image CD signals were observed for each enantiomeric pair as well as for the corresponding free chiral ligands. The experimental CD spectra closely matched the simulated results (Fig. S8 and Tables S9, S10), further confirming the absolute configurations. Based on TD-DFT calculations, the CD bands in the 470–700 nm region primarily arise from planar chirality.7 CPL spectra were acquired both in CH2Cl2 solution and in PMMA films. Mirror-image CPL responses were clearly observed for each pair of enantiomers (Fig. 4c and e). The glum values at the emission maxima were determined to be –2.5/2.5 × 10−3 in CH2Cl2 and –2.8/2.7 × 10−3 in PMMA for (R,S,R)/(S,R,S)-Pt1, and –2.1/2.1 × 10−3 in CH2Cl2 and –3.9/3.8 × 10−3 in PMMA for (R,S,R)/(S,R,S)-Pt2 (Table S5, Fig. 4 and Fig. S9). As far as we know, this is the first example of NIR chiral binuclear Pt(II) complexes with CPP maxima reaching 800 nm.1 For compounds with small molecular weight, glum is given by glum = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ|μe||μm|/(|μe|2 + |μm|2), where μe and μm are the electric and magnetic transition dipole moments and θ is the angle between them.1 The calculated |μe|/|μm| values (×10−20) are 3.9/0.006 for (R,S,R)-Pt1, 3.9/0.007 for (S,R,S)-Pt1, 2.9/0.002 for (R,S,R)-Pt2, and 3.0/0.002 for (S,R,S)-Pt2 (Table S10). The corresponding θ values are 45.4°, 44.2°, 2.4°, and 0.4°, respectively. Notably, the molecular structure exerts a much stronger influence on |μm| and θ than on |μe|. As expected, |μe| is substantially larger than |μm| for all complexes. The simulated glum values (3.3 × 10−3 for (R,S,R)-Pt1, –3.4 × 10−3 for (S,R,S)-Pt1, 1.8 × 10−3 for (R,S,R)-Pt2, and –1.6 × 10−3 for (S,R,S)-Pt2) are on the order of 10−3, consistent with typical values of reported analogues.1,9
θ|μe||μm|/(|μe|2 + |μm|2), where μe and μm are the electric and magnetic transition dipole moments and θ is the angle between them.1 The calculated |μe|/|μm| values (×10−20) are 3.9/0.006 for (R,S,R)-Pt1, 3.9/0.007 for (S,R,S)-Pt1, 2.9/0.002 for (R,S,R)-Pt2, and 3.0/0.002 for (S,R,S)-Pt2 (Table S10). The corresponding θ values are 45.4°, 44.2°, 2.4°, and 0.4°, respectively. Notably, the molecular structure exerts a much stronger influence on |μm| and θ than on |μe|. As expected, |μe| is substantially larger than |μm| for all complexes. The simulated glum values (3.3 × 10−3 for (R,S,R)-Pt1, –3.4 × 10−3 for (S,R,S)-Pt1, 1.8 × 10−3 for (R,S,R)-Pt2, and –1.6 × 10−3 for (S,R,S)-Pt2) are on the order of 10−3, consistent with typical values of reported analogues.1,9
To conclude, we have developed a series of chiral binuclear Pt(II) complexes achieving NIR CPP up to 800 nm through rational ligand regulation and planar chirality engineering. The intramolecular Pt–Pt interactions not only promote the formation of 3MMLCT states but also facilitate planar chirality. These complexes exhibit notable PLQYs (up to 35.4%), short lifetimes (<2 µs), and appreciable glum factors (∼3.9 × 10−3), representing the first example of NIR CPL Pt(II) emitters reaching 800 nm. These results demonstrate that this design strategy shows great potential for developing high-performance NIR CPL emitters for both biomedical and optoelectronic applications.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: synthesis, characterization and other data. See DOI: https://doi.org/10.1039/d6cc01319h.CCDC 2526096–2526099 contain the supplementary crystallographic data for this paper.10a–d
Acknowledgements
We acknowledge the financial support from the Guizhou University Talent Program (X2023159), the Guizhou Provincial Basic Research Program (Natural Science) (QKHJC MS[2025]606), the Guizhou Provincial Basic Research Program (Natural Science-Youth Guidance) (QKHJC2024Y100), the Youth Talent Growth Project of Guizhou Provincial Department of Education (QJJ2024023), the Guangdong Basic and Applied Basic Research Foundation (2026A1515011199), and the Startup Fund for RAPs under the Strategic Hiring Scheme (P0035922).References
- (a) N. Liang, C. Cao, Z. Xie, J. Liu, Y. Feng and C.-J. Yao, Mater. Today, 2024, 75, 309 CrossRef CAS; (b) X. Yang, X. Gao, Y.-X. Zheng, H. Kuang, C.-F. Chen, M. Liu, P. Duan and Z. Tang, CCS Chem., 2023, 5, 2760 CrossRef CAS.
- (a) K. Y. Zhang, Q. Yu, H. Wei, S. Liu, Q. Zhao and W. Huang, Chem. Rev., 2018, 118, 1770 CrossRef CAS PubMed; (b) P. Tao, X. Lü, G. Zhou and W.-Y. Wong, Acc. Mater. Res., 2022, 3, 830 CrossRef CAS; (c) P. Tao and W.-Y. Wong, Luminescent Transition-Metal Complexes and Their Applications in Electroluminescence, Comprehensive Inorganic Chemistry III, Elsevier, 3rd edn, 2023, vol. 8, pp. 2–79 Search PubMed; (d) S. Hattori and K. Shinozaki, ChemPhotoChem, 2025, 9, e202500041 CrossRef CAS.
- (a) P. Tao, J. Jin, X. Zheng, Y.-J. Pu and W.-Y. Wong, Matter, 2025, 8, 102142 CrossRef CAS; (b) S. N. T. Phan, N. B. Nguyen and T. S. Teets, Chem. Commun., 2025, 61, 17544 RSC; (c) K. T. Ly, R.-W. Chen-Cheng, H.-W. Lin, Y.-J. Shiau, S.-H. Liu, P.-T. Chou, C.-S. Tsao, Y.-C. Huang and Y. Chi, Nat. Photonics, 2017, 11, 63 CrossRef; (d) M. Wałęsa-Chorab, J. Photochem. Photobiol., C, 2024, 59, 100664 CrossRef; (e) H. J. Park, Crystals, 2025, 15, 273 CrossRef CAS.
- M. Penconi, M. Cazzaniga, S. Kesarkar, P. R. Mussini, D. Ceresoli and A. Bossi, Photochem. Photobiol. Sci., 2017, 16, 1220 CrossRef CAS PubMed.
- (a) M. Yoshida and M. Kato, Coord. Chem. Rev., 2018, 355, 101 CrossRef CAS; (b) J. Song, H. Xiao, B. Zhang, L. Qu, X. Zhou, P. Hu, Z.-X. Xu and H. Xiang, Angew. Chem., Int. Ed., 2023, 62, e202302011 CrossRef CAS PubMed; (c) J. Song, H. Xiao, L. Fang, L. Qu, X. Zhou, Z.-X. Xu, C. Yang and H. Xiang, J. Am. Chem. Soc., 2022, 144, 2233 CrossRef CAS PubMed.
- (a) J. R. Brandt, X. Wang, Y. Yang, A. J. Campbell and M. J. Fuchter, J. Am. Chem. Soc., 2016, 138, 9743 CrossRef CAS PubMed; (b) G.-Z. Lu, N. Su, Y. Li and Y.-X. Zheng, J. Organomet. Chem., 2017, 842, 39 CrossRef CAS.
- (a) B. Zhang, Q. Yang, B. Tu, W. Dong, X. Zhou, J. Song and H. Xiang, Org. Lett., 2025, 27, 6971 CrossRef CAS PubMed; (b) S. Gu, D. Li, D. Long, X. Yu, S. Ma, W. Li, J. Liu, P. Tao and W.-Y. Wong, Inorg. Chem., 2025, 64, 22513 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford, CT, p. 201 Search PubMed.
- (a) J. Han, S. Guo, H. Lu, S. Liu, Q. Zhao and W. Huang, Adv. Opt. Mater., 2018, 6, 1800538 CrossRef; (b) H. Lu, L. Di Bari and L. Favereau, Nat. Photonics, 2025, 19, 1041 CrossRef CAS; (c) F. J. Coughlin, M. S. Westrol, K. D. Oyler, N. Byrne, C. Kraml, E. Zysman-Colman, M. S. Lowry and S. Bernhard, Inorg. Chem., 2008, 47, 2039 CrossRef CAS PubMed; (d) Y. Deng, M. Wang, Y. Zhuang, S. Liu, W. Huang and Q. Zhao, Light: Sci. Appl., 2021, 10, 76 CrossRef CAS PubMed.
- (a) CCDC 2526096: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qslz3; (b) CCDC 2526097: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qsm05; (c) CCDC 2526098: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qsm16; (d) CCDC 2526099: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qsm27.
| This journal is © The Royal Society of Chemistry 2026 |