Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Tetrylidyne coordination modes: vinyl-type germa–ruthena anion [Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/h2_char_e001.gif) Ge:]− and its reaction with monofluorobenzene
Ge:]− and its reaction with monofluorobenzene
Frederic Seeger,
Liesa Renz and
Lars Wesemann *
*
Institut für Anorganische Chemie, Eberhard Karls Universität Tübingen, Auf der Morgenstelle 18, 72076 Tübingen, Germany. E-mail: lars.wesemann@uni-tuebingen.de
First published on 7th April 2026
Abstract
Ruthenium germyl complex [(Ph3P)ClRuGeCl2(C6H3-2-{η6-Trip}-6-Trip)] (1) is reduced with four electrons to yield the vinyl anion type complex [(Ph3P)Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) GeK(C6H3-2-{η6-Trip}-6-Trip)] (3a) exhibiting a novel coordination mode for a germylidyne ligand. Formation of a gold–germanium bond in reaction with [(Ph3P)AuCl] and reaction with monofluorobenzene is presented.
GeK(C6H3-2-{η6-Trip}-6-Trip)] (3a) exhibiting a novel coordination mode for a germylidyne ligand. Formation of a gold–germanium bond in reaction with [(Ph3P)AuCl] and reaction with monofluorobenzene is presented.
The higher homologues of the carbyne ligand [C–R] are referred to as tetrylidyne ligands [E–R] (E = Si–Pb), and the first tetrylidyne transition metal complex [Cp(CO)2Mo
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) GeAr′] was synthesized by Power and coworkers in 1996 [Ar′ = C6H3-2,6-(Mes)2, Mes = 2,4,6-C6H2Me3].1,2 Filippou et al. have further developed the family of tetrylidyne complexes and have presented a broad variety of examples of these low valent Group 14 element compounds.3–13 Heavy carbyne coordination chemistry has also been investigated by Hadlington, Hashimoto, Jones, Tilley, and Tobita, so that there are now numerous examples in the literature [(M
GeAr′] was synthesized by Power and coworkers in 1996 [Ar′ = C6H3-2,6-(Mes)2, Mes = 2,4,6-C6H2Me3].1,2 Filippou et al. have further developed the family of tetrylidyne complexes and have presented a broad variety of examples of these low valent Group 14 element compounds.3–13 Heavy carbyne coordination chemistry has also been investigated by Hadlington, Hashimoto, Jones, Tilley, and Tobita, so that there are now numerous examples in the literature [(M![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) E) M = Nb, Cr, Mo, W, Mn, Re, Fe, Co, Rh, Ir, Ni, Pt; E = Si, Ge, Sn, Pb].14–25 We have recently added the tetrylidynes (E = Ge, Sn, Pb) of Co, Rh and Ir to this family.23–25
E) M = Nb, Cr, Mo, W, Mn, Re, Fe, Co, Rh, Ir, Ni, Pt; E = Si, Ge, Sn, Pb].14–25 We have recently added the tetrylidynes (E = Ge, Sn, Pb) of Co, Rh and Ir to this family.23–25
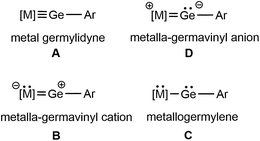
In heavy carbyne transition metal coordination compounds, the transition metal tetrylidynes, the tetrylidyne ligands build a linear triple bond [R–E![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) M] with transition metal fragments (Scheme 1A). This bond consists of a σ-bond, which results from donation of the Group 14 element lone pair to the transition metal and two π-bonds derived from transition metal d-orbital donation into empty Group 14 element p-orbitals.26–31 We have recently shown that after bromide abstraction from the tetrylene complexes [(Me3P)3HIrEBrTbb] (E = Ge, Sn, Tbb = 2,6-[CH(SiMe3)2]2-4-(t-Bu)C6H2) heavy metalla vinyl-cations [(Me3P)3HIr
M] with transition metal fragments (Scheme 1A). This bond consists of a σ-bond, which results from donation of the Group 14 element lone pair to the transition metal and two π-bonds derived from transition metal d-orbital donation into empty Group 14 element p-orbitals.26–31 We have recently shown that after bromide abstraction from the tetrylene complexes [(Me3P)3HIrEBrTbb] (E = Ge, Sn, Tbb = 2,6-[CH(SiMe3)2]2-4-(t-Bu)C6H2) heavy metalla vinyl-cations [(Me3P)3HIr![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) E–Tbb]+ (E = Ge, Sn) were obtained (Scheme 1B).32 In these cations the bonding of the [Sn–Tbb] ligand at the iridium fragment consists of a σ-donor interaction Sn → Ir and only one π-backbond from the iridium atom to the tin. Furthermore, there is an empty p-orbital at the tin atom and the cationic charge is primarily residing at the tin atom.32 Therefore we consider this bonding situation of the [Sn–Tbb] tetrylidyne ligand at iridium as a heavy metalla vinyl-cation (Scheme 1B).32 This vinyl-type coordination of [Sn–Tbb] was also observed in the cationic hydride abstraction product [Cp2Ti
E–Tbb]+ (E = Ge, Sn) were obtained (Scheme 1B).32 In these cations the bonding of the [Sn–Tbb] ligand at the iridium fragment consists of a σ-donor interaction Sn → Ir and only one π-backbond from the iridium atom to the tin. Furthermore, there is an empty p-orbital at the tin atom and the cationic charge is primarily residing at the tin atom.32 Therefore we consider this bonding situation of the [Sn–Tbb] tetrylidyne ligand at iridium as a heavy metalla vinyl-cation (Scheme 1B).32 This vinyl-type coordination of [Sn–Tbb] was also observed in the cationic hydride abstraction product [Cp2Ti![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) SnAr*(SnHAr*)][HB(C6F5)3] [(Ar* = C6H3-2,6-(Trip)2), Trip = 2,4,6-C6H2iPr3] and the iron complex [Cp*(iPr2MeP)Fe
SnAr*(SnHAr*)][HB(C6F5)3] [(Ar* = C6H3-2,6-(Trip)2), Trip = 2,4,6-C6H2iPr3] and the iron complex [Cp*(iPr2MeP)Fe![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) SnAr′][B(C6F5)4] shows also a comparable electronic situation.21,33,34 The metallotetrylene bonding mode (Scheme 1C) of the tetrylidyne ligands [E–R] is well known and was presented in the literature for the heavy elements of the Group 14 together with reactivity studies of the [M–Ë–R] unit (E = Si, Ge, Sn and Pb).2,18,21,35–42 Pandey, Frenking and coworker discussed in a comparative study the metal–germanium interaction in metallogermylenes and metal germylidyne complexes (Scheme 1A and C).29 We present in this manuscript with the germa–ruthena vinyl anion a hitherto unknown bonding mode for a germylidyne ligand [Ge–Ar*] (Scheme 1D) and show first reactions of this unprecedented [Ru
SnAr′][B(C6F5)4] shows also a comparable electronic situation.21,33,34 The metallotetrylene bonding mode (Scheme 1C) of the tetrylidyne ligands [E–R] is well known and was presented in the literature for the heavy elements of the Group 14 together with reactivity studies of the [M–Ë–R] unit (E = Si, Ge, Sn and Pb).2,18,21,35–42 Pandey, Frenking and coworker discussed in a comparative study the metal–germanium interaction in metallogermylenes and metal germylidyne complexes (Scheme 1A and C).29 We present in this manuscript with the germa–ruthena vinyl anion a hitherto unknown bonding mode for a germylidyne ligand [Ge–Ar*] (Scheme 1D) and show first reactions of this unprecedented [Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge:]− moiety.
Ge:]− moiety.
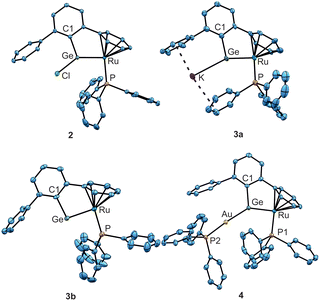
Starting with the ruthenium–germyl complex 1, which is synthesized following a literature procedure, the germylene coordination compound 2 was obtained straightforwardly by two-electron reduction of complex 1 (Scheme 2).43 The reduction gives a zero valent ruthenium complex coordinated by a chlorogermylene ligand. The ruthenium germylene coordination compound 2 (Fig. 1) shows a Ru–Ge bond length of 2.27500(18) Å, which is a very short bond between these elements and can be compared with the so far shortest Ru–Ge bond of 2.2821(6) Å in Cp*Ru(GeHTrip)H(PMeiPr2).44
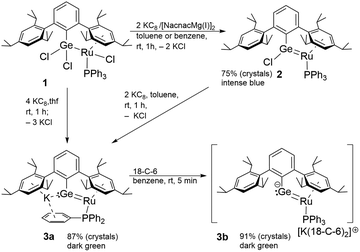 | ||
| Scheme 2 Reduction of germyl complex 1, synthesis of chloro germylene complex 2 and germa vinyl anion 3a and 3b. | ||
Other comparable Ru–Ge coordination compounds exhibit slightly longer Ru–Ge bond lengths [2.3162(2)–2.3579(3) Å].45–48 The germylene coordination in complex 2 features a typical σ-donor and π-acceptor interaction with the ruthenium atom. σ-Donation from a germanium s-orbital into a ruthenium d-orbital results in a σ-bond featuring polarization to the germanium atom (NLMO: atomic contributions Ru–Ge: 37, 58%, Table 1). π-Backdonation from a filled ruthenium d-orbital into an empty p-orbital of germanium was observed and this interaction is polarized to the ruthenium atom (NLMO: atomic contributions Ru–Ge: 71, 23%, Table 1).
| 2 | 3a | 3b | 4 | |
|---|---|---|---|---|
| Ru–Ge [Å] | 2.27271 | 2.34713 | 2.38133 | 2.30474 |
| q [e] Ru, Ge | –0.47, 0.96 | –0.47, 0.09 | –0.51, 0.32 | –0.43, 0.31 |
| Wiberg/Löwdin | 1.01/1.80 | 1.09/1.75 | 0.99/1.88 | 1.08/1.66 |
| σ-bond occ. | 1.8056 | 1.6821 | 1.3952 | 1.7537 |
| Ru–Ge% (NLMO) | 37, 58 | 55, 37 | 71, 23 | 46, 48 |
| π-bond occ. | 1.8652 | 1.8037 | 1.7936 | 1.8470 |
| Ru–Ge% (NLMO) | 71, 23 | 66, 24 | 71, 19 | 67, 26 |
Two-electron reduction of the zero valent ruthenium germylene complex 2 or four-electron reduction of germyl complex 1 with KC8 affords formation of a novel germa–ruthena vinyl anion 3a as a potassium salt (Scheme 2). After addition of crown ether 18-crown-6 (18-C-6) to compound 3a salt 3b was isolated featuring separation of the anion and cation in the solid-state structure. The germa vinyl anions 3a and 3b feature a longer Ru–Ge bond [3a: 2.3531(10), 3b: 2.3865(9) Å] (Fig. 1) in comparison to the chloro germylene complex 2. Solutions of 3a and 3b are thermally stable at rt and 3a was also heated to 100 °C without decomposing.
In Fig. 2 the natural localized molecular orbitals representing the s-orbital at germanium and the Ru–Ge interactions of compound 3b are shown (results for 3a were placed in the SI). Because of the two-electron reduction, in 3b a reduced natural charge of the germanium atom in comparison to the germylene complex 2 was observed (Table 1). The steric strain of the chelating ligand, which manifests itself in a small angle at germanium [3b: C1–Ge–Ru 97.1(2)°] (Fig. 1), enables the formation of a germanium s-orbital lone-pair (Fig. 2 left NLMO). Consequently, both the Ru–Ge σ-bond (Fig. 2 right NLMO) and the π-bond (Fig. 2, middle NLMO) consist of an interaction mainly between a Ge p-orbital and a Ru d-orbital. These Ru–Ge σ-bond and π-interactions in 3b feature both polarization of the bonds to the ruthenium atom (σ-bond: atomic contributions Ru–Ge 3b: 71, 23%), (π-bond atomic contributions Ru–Ge 3b: 71, 19%, Table 1). Thus, in anion 3b a germylidyne ligand [Ge–Ar*] coordinates at ruthenium via σ- and π-donation from ruthenium d-orbitals into empty germanium p-orbitals. The analysis of the electronic situation in anion 3b leads to the rationalization as a Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge double bond and a lone pair at germanium. We discuss this bonding motif, which is enforced by the steric strain of the chelat-forming ligand, as a novel bonding mode in tetrylidyne coordination chemistry (Scheme 1D). With the formal fragmentation of complex 3 into a (η6-C6H6)Ru(PR3) fragment being isolobal with :CH2 we propose that the tetrylidyne coordination complex 3 be designated as a heavy vinyl anion [Ru
Ge double bond and a lone pair at germanium. We discuss this bonding motif, which is enforced by the steric strain of the chelat-forming ligand, as a novel bonding mode in tetrylidyne coordination chemistry (Scheme 1D). With the formal fragmentation of complex 3 into a (η6-C6H6)Ru(PR3) fragment being isolobal with :CH2 we propose that the tetrylidyne coordination complex 3 be designated as a heavy vinyl anion [Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge:]−.
Ge:]−.
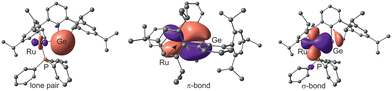 | ||
| Fig. 2 NLMOs representing the Ru–Ge interactions in 3b [lone pair: Ge 4s (83%) 4p (17%), Ru 5s (92%) 4d (7%), π-bond: Ge 4p (100%), Ru 4d (100%), σ-bond: Ge 4s (11%) 4p (88%), Ru 5s (3%) 4d (97%) orbital].59 | ||
This type of heavy metalla vinyl anion structural motif can be compared with carbene–CH-deprotonated carbene complexes [Li(dmp)][(Me3CCH2)3Ta![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(CMe3)] (dmp = N,N′-dimethylpiperazine).60
C(CMe3)] (dmp = N,N′-dimethylpiperazine).60
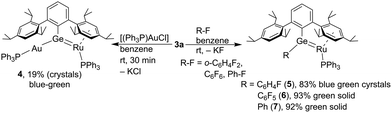
The chemical properties of the germa–ruthena vinyl anion 3a were evaluated in reaction with the gold electrophile [(Ph3P)AuCl] and fluorinated benzene derivatives. The [Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge:]− anion reacts as a nucleophile and substitutes the chloride ligand at the gold atom to give a [Ru
Ge:]− anion reacts as a nucleophile and substitutes the chloride ligand at the gold atom to give a [Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge–Au]-unit (Scheme 3). The Ge–Au bond length observed in the molecular structure of 4 (Fig. 1) of 2.4129(5) Å lies in the range of published interatomic distances between these elements [2.7587(7)–2.345(1) Å].61–64 The gold coordination compound 4 shows a longer Ru–Ge bond in comparison to the germylene complex 2 and the bond length can be compared with distances observed in germylene ruthenium coordination compounds [2.3162(2)–2.3579(3) Å].45–48 The germylene–gold coordination features a σ-bond and the Ru–Ge interaction consists of a σ- and π-bond. This type of germanium–gold coordination was previously reported treating a borylated silagermenide [K][(Me3Si)(NHB)Si
Ge–Au]-unit (Scheme 3). The Ge–Au bond length observed in the molecular structure of 4 (Fig. 1) of 2.4129(5) Å lies in the range of published interatomic distances between these elements [2.7587(7)–2.345(1) Å].61–64 The gold coordination compound 4 shows a longer Ru–Ge bond in comparison to the germylene complex 2 and the bond length can be compared with distances observed in germylene ruthenium coordination compounds [2.3162(2)–2.3579(3) Å].45–48 The germylene–gold coordination features a σ-bond and the Ru–Ge interaction consists of a σ- and π-bond. This type of germanium–gold coordination was previously reported treating a borylated silagermenide [K][(Me3Si)(NHB)Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) GeAr*] with two equivalents of gold electrophile [(Me2IPr)AuCl] at 80 °C to yield trans-[(Me2IPr)Au-(NHB)Si
GeAr*] with two equivalents of gold electrophile [(Me2IPr)AuCl] at 80 °C to yield trans-[(Me2IPr)Au-(NHB)Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) GeAr*Au(Me2IPr)] exhibiting an Au–Ge bond of 2.3919(4) Å [NHB = B(NMes)2(CH2)2, Me2IPr = 1,3-diisopropyl-4,5-dimethylimidazol-2-ylidene].64
GeAr*Au(Me2IPr)] exhibiting an Au–Ge bond of 2.3919(4) Å [NHB = B(NMes)2(CH2)2, Me2IPr = 1,3-diisopropyl-4,5-dimethylimidazol-2-ylidene].64
Optimization of the conditions for crystallization of potassium salts 3a and 3b prompted us to test o-difluorobenzene as solvent. Surprisingly, we observed the reaction of a C–F bond of the o-difluorobenzene solvent and formation of a Ge–C bond (Scheme 3). To evaluate the scope of this reaction we investigated the reaction of hexafluoro- and monofluorobenzene (Scheme 3). The halides were treated with the salt 3a at rt in benzene and the substitution reaction was monitored by NMR spectroscopy. Furthermore, addition of crown ether 18-C-6 to the mixture leads to shorter reaction times, which is probably due to complexation of the potassium cation resulting in separation of cation and anion of the germa–ruthena vinyl salt 3b.
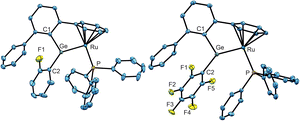
In the case of o-C6H4F2 and C6F6 we isolated the substitution products, and the molecular structures are shown in Fig. 3. Compounds 5 and 6 are diaryl germylene coordination compounds featuring a trigonal planar coordinated germanium atom and a short Ru–Ge bond of 5: 2.2855(3) and 6: 2.28155(18) Å, respectively. These Ru–Ge bonds are slightly longer than the Ru–Ge bond in 2. Reactivity studies of very stable C–F bonds65–67 is a research topic of high interest (C6H5F: C–F DH298 127.2 ± 0.7 kcal mol−1).65–73 In addition to the reactions of C–F bonds mediated by transition metals, it was also previously reported that compounds of main-group elements react with C–F bonds of fluorobenzenes. Low valent compounds of magnesium,74–76 boron,77,78 aluminium,79–84 gallium,85 silicon,86,87 germanium,88,89 tin,88 phosphorus90,91 and bismuth92 exhibit reactions with C–F units. The reaction of monofluorobenzene however was observed in comparison to the presented germa–ruthena vinyl anion 3b with examples of more electropositive elements shown in Scheme 4 E–I.74–76,83,84 Furthermore, C–F bond reactivity of monofluorobenzene was also observed by calcium and strontium hydrides and a magnesium dinitrogen complex.93–96
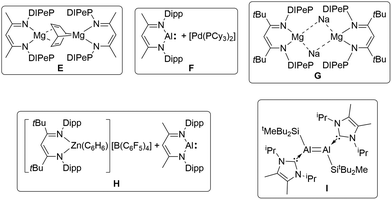 | ||
| Scheme 4 Monofluorobenzene C–F reaction with E (DIPeP = 2,6-(3-pentyl)phenyl),76 F (Dipp = 2,6-iPr2C6H3),84 G,74 H75 and I.83 | ||
To conclude, upon two electron reduction of an arylchlorogermylene ruthenium complex an anionic coordination compound [(Ph3P)Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif)
![[G with combining umlaut]](https://www.rsc.org/images/entities/char_0047_0308.gif) e(C6H3-2-{η6-Trip}-6-Trip)]− featuring a hitherto unknown coordination mode in heavy carbyne, tetrylidyne coordination chemistry was observed. This bonding mode expands in a systematic way the tetrylidyne ligand–transition metal interactions described in the literature so far. Because of the chelating terphenyl ligand enforcing a small C–Ge–Ru angle at germanium a vinyl anion type coordination compound [Ru
e(C6H3-2-{η6-Trip}-6-Trip)]− featuring a hitherto unknown coordination mode in heavy carbyne, tetrylidyne coordination chemistry was observed. This bonding mode expands in a systematic way the tetrylidyne ligand–transition metal interactions described in the literature so far. Because of the chelating terphenyl ligand enforcing a small C–Ge–Ru angle at germanium a vinyl anion type coordination compound [Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge:]− comprising a Ge s-orbital lone pair is isolated. In addition, both σ- and π-interactions are established in the Ru
Ge:]− comprising a Ge s-orbital lone pair is isolated. In addition, both σ- and π-interactions are established in the Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge double bond from orthogonal germanium p-orbitals and ruthenium d-orbitals. With the monofluorobenzene reaction of this germa–ruthena vinyl anion we are expanding the exclusive family of main-group element compounds that react with monofluorobenzene to include the least electropositive example. Furthermore, an example of the coordination chemistry of the vinyl anion leading to the formation of an [Au–Ge
Ge double bond from orthogonal germanium p-orbitals and ruthenium d-orbitals. With the monofluorobenzene reaction of this germa–ruthena vinyl anion we are expanding the exclusive family of main-group element compounds that react with monofluorobenzene to include the least electropositive example. Furthermore, an example of the coordination chemistry of the vinyl anion leading to the formation of an [Au–Ge![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ru] unit was characterized based on the reaction with a gold electrophile.
Ru] unit was characterized based on the reaction with a gold electrophile.
F. S. experiments, writing of manuscript, crystallographic investigations and SI, L. R. synthesis of 3a, L. W. supervision, funding acquisition, manuscript writing and review.
Conflicts of interest
There are no conflicts to declare.Data availability
Full experimental details are provided as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc01226d.CCDC 2512263–2512268 contain the supplementary crystallographic data for this paper.97a–f
Acknowledgements
The authors acknowledge support by the state of Baden-Württemberg through bwHPC and the German Research Foundation (DFG) through grant no. INST 40/575-1 FUGG (Justus 2 cluster).References
- R. S. Simons and P. P. Power, J. Am. Chem. Soc., 1996, 118, 11966–11967 CrossRef CAS.
- L. Pu, B. Twamley, S. T. Haubrich, M. M. Olmstead, B. V. Mork, R. S. Simons and P. P. Power, J. Am. Chem. Soc., 2000, 122, 650–656 CrossRef CAS.
- K. Tomer, G. Schnakenburg, U. Das and A. C. Filippou, Chem. Sci., 2025, 16, 7773–7793 RSC.
- P. Ghana, M. I. Arz, U. Chakraborty, G. Schnakenburg and A. C. Filippou, J. Am. Chem. Soc., 2018, 140, 7187–7198 CrossRef CAS PubMed.
- A. C. Filippou, D. Hoffmann and G. Schnakenburg, Chem. Sci., 2017, 8, 6290–6299 RSC.
- A. C. Filippou, P. Ghana, U. Chakraborty and G. Schnakenburg, J. Am. Chem. Soc., 2013, 135, 11525–11528 CrossRef CAS PubMed.
- A. C. Filippou, A. Barandov, G. Schnakenburg, B. Lewall, M. van Gastel and A. Marchanka, Angew. Chem., Int. Ed., 2012, 51, 789–793 CrossRef CAS PubMed.
- A. C. Filippou, O. Chernov, K. W. Stumpf and G. Schnakenburg, Angew. Chem., Int. Ed., 2010, 49, 3296–3300 CrossRef CAS PubMed.
- A. C. Filippou, N. Weidemann and G. Schnakenburg, Angew. Chem., Int. Ed., 2008, 47, 5799–5802 CrossRef CAS PubMed.
- A. C. Filippou, N. Weidemann, A. I. Philippopoulos and G. Schnakenburg, Angew. Chem., 2006, 118, 6133–6137 CrossRef.
- A. C. Filippou, N. Weidemann, G. Schnakenburg, H. Rohde and A. I. Philippopoulos, Angew. Chem., Int. Ed., 2004, 43, 6512–6516 CrossRef CAS PubMed.
- A. C. Filippou, P. Portius, A. I. Philippopoulos and H. Rohde, Angew. Chem., Int. Ed., 2003, 42, 445–447 CrossRef CAS PubMed.
- A. C. Filippou, A. I. Philippopoulos, P. Portius and D. U. Neumann, Angew. Chem., Int. Ed., 2000, 39, 2778–2781 CrossRef CAS PubMed.
- P. M. Keil and T. J. Hadlington, Chem. Commun., 2022, 58, 3011–3014 RSC.
- C. Fontanilla, Y. Shimizu, K. Nagata, S. Iwatsuki, A. Tanamura, S. Mori, T. P. Dhungana and H. Hashimoto, Dalton Trans., 2026, 55, 663–670 RSC.
- M. Matsuoka, K. Nagata, R. Ohno, T. Matsuo, H. Tobita and H. Hashimoto, Chem. – Eur. J., 2024, 30, e202303765 CrossRef CAS PubMed.
- K. Nagata, H. Omura, M. Matsuoka, H. Tobita and H. Hashimoto, Organometallics, 2023, 42, 1131–1138 CrossRef CAS.
- J. Hicks, T. J. Hadlington, C. Schenk, J. Li and C. Jones, Organometallics, 2013, 32, 323–329 CrossRef CAS.
- B. V. Mork and T. D. Tilley, Angew. Chem., Int. Ed., 2003, 42, 357–360 CrossRef CAS PubMed.
- P. G. Hayes, Z. Xu, C. Beddie, J. M. Keith, M. B. Hall and T. D. Tilley, J. Am. Chem. Soc., 2013, 135, 11780–11783 CrossRef CAS PubMed.
- R. C. Handford, M. A. Nesbit, P. W. Smith, R. D. Britt and T. D. Tilley, J. Am. Chem. Soc., 2022, 144, 358–367 CrossRef CAS PubMed.
- H. Hashimoto, T. Fukuda, H. Tobita, M. Ray and S. Sakaki, Angew. Chem., Int. Ed., 2012, 51, 2930–2933 CrossRef CAS PubMed.
- M. Widemann, K. Eichele, H. Schubert, C. P. Sindlinger, S. Klenner, R. Pöttgen and L. Wesemann, Angew. Chem., Int. Ed., 2021, 60, 5882–5889 CrossRef CAS PubMed.
- M. Auer, K. Zwettler, K. Eichele, H. Schubert, C. P. Sindlinger and L. Wesemann, Angew. Chem., Int. Ed., 2023, 62, e202305951 CrossRef CAS PubMed.
- L. G. Holzapfel, J. Manegold, S. F. Clewing, H. Schubert, K. Eichele and L. Wesemann, Angew. Chem., Int. Ed., 2025, 64, e202503930 CrossRef CAS PubMed.
- L. R. Maurer, J. Rump and A. C. Filippou, Inorganics, 2023, 11, 129 CrossRef CAS.
- H. Hashimoto and H. Tobita, Coord. Chem. Rev., 2018, 355, 362–379 CrossRef CAS.
- S. Saini, A. Agarwal and S. K. Bose, Dalton Trans., 2020, 49, 17055–17075 RSC.
- K. K. Pandey, M. Lein and G. Frenking, J. Am. Chem. Soc., 2003, 125, 1660–1668 CrossRef CAS PubMed.
- K. K. Pandey and A. Lledós, Inorg. Chem., 2009, 48, 2748–2759 CrossRef CAS PubMed.
- K. K. Pandey, P. Patidar and P. P. Power, Inorg. Chem., 2011, 50, 7080–7089 CrossRef CAS PubMed.
- M. Auer, J. Bolten, K. Eichele, H. Schubert, C. P. Sindlinger and L. Wesemann, Chem. Sci., 2023, 14, 514–524 RSC.
- J.-J. Maudrich, M. Widemann, F. Diab, R. H. Kern, P. Sirsch, C. P. Sindlinger, H. Schubert and L. Wesemann, Chem. – Eur. J., 2019, 25, 16081–16087 CrossRef CAS PubMed.
- Y. Liu, F. F. Westermair, I. Becker, S. Hauer, M. Bodensteiner, C. Hennig, G. Balázs, F. Meyer, R. M. Gschwind and R. Wolf, J. Am. Chem. Soc., 2025, 147, 7083–7093 CrossRef CAS PubMed.
- A. C. Filippou, B. Baars, O. Chernov, Y. N. Lebedev and G. Schnakenburg, Angew. Chem., Int. Ed., 2014, 53, 565–570 CrossRef CAS PubMed.
- B. E. Eichler, A. D. Phillips, S. T. Haubrich, B. V. Mork and P. P. Power, Organometallics, 2002, 21, 5622–5627 CrossRef CAS.
- L. Pu, P. P. Power, I. Boltes and R. Herbst-Irmer, Organometallics, 2000, 19, 352–356 CrossRef CAS.
- K. Inomata, T. Watanabe, Y. Miyazaki and H. Tobita, J. Am. Chem. Soc., 2015, 137, 11935–11937 CrossRef CAS PubMed.
- M. Widemann, S. Jeggle, M. Auer, K. Eichele, H. Schubert, C. P. Sindlinger and L. Wesemann, Chem. Sci., 2022, 13, 3999–4009 RSC.
- H.-J. Liu, J. Guihaumé, T. Davin, C. Raynaud, O. Eisenstein and T. D. Tilley, J. Am. Chem. Soc., 2014, 136, 13991–13994 CrossRef CAS PubMed.
- H.-J. Liu, M. S. Ziegler and T. D. Tilley, Angew. Chem., 2015, 127, 6722–6726 Search PubMed.
- M. Bamberg, T. Gasevic, J. Martinez Fernandez, T. G. Saint-Denis and T. D. Tilley, J. Am. Chem. Soc., 2025, 147, 38732–38742 Search PubMed.
- P. Nie, Q. Yu, H. Zhu and T.-B. Wen, Eur. J. Inorg. Chem., 2017, 4784–4796 CrossRef CAS.
- P. G. Hayes, R. Waterman, P. B. Glaser and T. D. Tilley, Organometallics, 2009, 28, 5082–5089 CrossRef CAS.
- K. A. Smart, E. Mothes-Martin, L. Vendier, R. N. Perutz, M. Grellier and S. Sabo-Etienne, Organometallics, 2015, 34, 4158–4163 CrossRef CAS.
- J. A. Cabeza, J. M. Fernandez-Colinas, P. Garcia-Alvarez and D. Polo, RSC Adv., 2014, 4, 31503–31506 RSC.
- M. C. Lipke, F. Neumeyer and T. D. Tilley, J. Am. Chem. Soc., 2014, 136, 6092–6102 CrossRef CAS PubMed.
- M. E. Fasulo and T. D. Tilley, Chem. Commun., 2012, 48, 7690–7692 RSC.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1606 Search PubMed.
- D. Andrae, U. Häussermann, M. Dolg, H. Stoll and H. Preuß, Theor. Chim. Acta, 1990, 77, 123–141 CrossRef CAS.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- F. Weigend, Phys. Chem. Chem. Phys., 2006, 8, 1057–1065 RSC.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 73–78 CAS.
- A. D. Becke, Phys. Rev. A: At., Mol., Opt. Phys., 1988, 38, 3098–3100 CrossRef CAS PubMed.
- J. P. Perdew and W. Yue, Phys. Rev. B: Condens. Matter Mater. Phys., 1986, 33, 8800–8802 CrossRef PubMed.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- B. Metz, H. Stoll and M. Dolg, J. Chem. Phys., 2000, 113, 2563–2569 CrossRef CAS.
- E. D. Glendening, J. K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, P. Karafiloglou, C. R. Landis and F. Weinhold, Journal, 2018 Search PubMed.
- G. A. Zhurko, CHEMCRAFT (https://www.chemcraftprog.com) Search PubMed.
- L. J. Guggenberger and R. R. Schrock, J. Am. Chem. Soc., 1975, 97, 2935 CrossRef CAS.
- W.-P. Leung, C.-W. So, K.-H. Chong, K.-W. Kan, H.-S. Chan and T. C. W. Mak, Organometallics, 2006, 25, 2851–2858 CrossRef CAS.
- N. Zhao, J. Zhang, Y. Yang, G. Chen, H. Zhu and H. W. Roesky, Organometallics, 2013, 32, 762–769 CrossRef CAS.
- Z. Dong, K. Bedbur, M. Schmidtmann and T. Müller, J. Am. Chem. Soc., 2018, 140, 3052–3060 CrossRef CAS PubMed.
- X.-Y. He, Q. Liang, Y. Mei and L. L. Liu, Angew. Chem., Int. Ed., 2025, 64, e202505940 CrossRef CAS PubMed.
- S. J. Blanksby and G. B. Ellison, Acc. Chem. Res., 2003, 36, 255–263 CrossRef CAS PubMed.
- S. A. Macgregor, D. McKay, J. A. Panetier and M. K. Whittlesey, Dalton Trans., 2013, 42, 7386–7395 RSC.
- G. Coates, F. Rekhroukh and M. R. Crimmin, Synlett, 2019, 2233–2246 CAS.
- O. Eisenstein, J. Milani and R. N. Perutz, Chem. Rev., 2017, 117, 8710–8753 CrossRef CAS PubMed.
- G. Coates, H. Y. Tan, C. Kalff, A. J. P. White and M. R. Crimmin, Angew. Chem., Int. Ed., 2019, 58, 12514–12518 CrossRef CAS PubMed.
- N. R. Judge, A. Logallo and E. Hevia, Chem. Sci., 2023, 14, 11617–11628 RSC.
- J. Wenz, C. A. Rettenmeier, H. Wadepohl and L. H. Gade, Chem. Commun., 2016, 52, 202–205 RSC.
- T. Ahrens, J. Kohlmann, M. Ahrens and T. Braun, Chem. Rev., 2015, 115, 931–972 CrossRef CAS PubMed.
- Q. Shen, Y.-G. Huang, C. Liu, J.-C. Xiao, Q.-Y. Chen and Y. Guo, J. Fluor. Chem., 2015, 179, 14–22 CrossRef CAS.
- B. Rösch, T. X. Gentner, J. Eyselein, J. Langer, H. Elsen and S. Harder, Nature, 2021, 592, 717–721 CrossRef PubMed.
- A. Friedrich, J. Eyselein, J. Langer, C. Färber and S. Harder, Angew. Chem., Int. Ed., 2021, 60, 16492–16499 CrossRef CAS PubMed.
- T. X. Gentner, B. Rösch, G. Ballmann, J. Langer, H. Elsen and S. Harder, Angew. Chem., Int. Ed., 2019, 58, 607–611 CrossRef CAS PubMed.
- C. D. Buch, A. Virovets, E. Peresypkina, B. Endeward, H.-W. Lerner, F. Fantuzzi, S. Yamaguchi and M. Wagner, J. Am. Chem. Soc., 2025, 147, 20071–20081 CrossRef CAS PubMed.
- H. Budy, S. E. Prey, C. D. Buch, M. Bolte, H.-W. Lerner and M. Wagner, Chem. Commun., 2022, 58, 254–257 RSC.
- M. R. Crimmin, M. J. Butler and A. J. P. White, Chem. Commun., 2015, 51, 15994–15996 RSC.
- T. Chu, Y. Boyko, I. Korobkov and G. I. Nikonov, Organometallics, 2015, 34, 5363–5365 CrossRef CAS.
- O. Kysliak, H. Görls and R. Kretschmer, Chem. Commun., 2020, 56, 7865–7868 RSC.
- S. Kurumada, S. Takamori and M. Yamashita, Nat. Chem., 2020, 12, 36–39 CrossRef CAS PubMed.
- X. Liu, S. Dong, J. Zhu and S. Inoue, J. Am. Chem. Soc., 2024, 146, 23591–23597 CrossRef CAS PubMed.
- F. Rekhroukh, W. Chen, R. K. Brown, A. J. P. White and M. R. Crimmin, Chem. Sci., 2020, 11, 7842–7849 RSC.
- O. Kysliak, H. Görls and R. Kretschmer, J. Am. Chem. Soc., 2021, 143, 142–148 CrossRef CAS PubMed.
- A. Jana, P. P. Samuel, G. Tavčar, H. W. Roesky and C. Schulzke, J. Am. Chem. Soc., 2010, 132, 10164–10170 CrossRef CAS PubMed.
- N. Tiessen, M. Keßler, B. Neumann, H.-G. Stammler and B. Hoge, Angew. Chem., Int. Ed., 2022, 61, e202116468 CrossRef CAS PubMed.
- P. P. Samuel, A. P. Singh, S. P. Sarish, J. Matussek, I. Objartel, H. W. Roesky and D. Stalke, Inorg. Chem., 2013, 52, 1544–1549 CrossRef CAS PubMed.
- R. Zitz, A. Pöcheim, J. Baumgartner and C. Marschner, Molecules, 2020, 25, 1322 CrossRef CAS PubMed.
- N. Gautam, S. P, K. Soni, S. Chakraborty, S. Maji, K. Bhattacharyya and S. K. Mandal, J. Am. Chem. Soc., 2025, 147, 23001–23013 CrossRef CAS PubMed.
- S. Lim and A. T. Radosevich, J. Am. Chem. Soc., 2020, 142, 16188–16193 CrossRef CAS PubMed.
- Y. Pang, M. Leutzsch, N. Nöthling, F. Katzenburg and J. Cornella, J. Am. Chem. Soc., 2021, 143, 12487–12493 CrossRef CAS PubMed.
- D. Schuhknecht, T. P. Spaniol, Y. Yang, L. Maron and J. Okuda, Inorg. Chem., 2020, 59, 9406–9415 CrossRef CAS PubMed.
- M. Wiesinger, B. Rösch, C. Knüpfer, J. Mai, J. Langer and S. Harder, Eur. J. Inorg. Chem., 2021, 3731–3741 CrossRef CAS.
- J. J. C. Struijs, M. A. Ellwanger, A. E. Crumpton, V. Gouverneur and S. Aldridge, Nat. Chem., 2024, 16, 1473–1480 CrossRef CAS PubMed.
- M. J. Evans, J. Mullins, R. Mondal and C. Jones, Chem. – Eur. J., 2024, 30, e202401005 CrossRef CAS PubMed.
- (a) CCDC 2512263: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qb6r1; (b) CCDC 2512264: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qb6s2; (c) CCDC 2512265: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qb6t3; (d) CCDC 2512266: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qb6v4; (e) CCDC 2512267: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qb6w5; (f) CCDC 2512268: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qb6x6.
| This journal is © The Royal Society of Chemistry 2026 |