Open Access Article
Open Access ArticleSupramolecular glass formation by arene–perfluoroarene pairing of three-dimensional macrocycles
Katsuto Onishi a,
Shunsuke Ohtani
a,
Shunsuke Ohtani *a,
Kenichi Kato
*a,
Kenichi Kato a,
Shigehisa Akine
a,
Shigehisa Akine bc and
Tomoki Ogoshi
bc and
Tomoki Ogoshi *ab
*ab
aDepartment of Synthetic Chemistry and Biological Chemistry, Graduate School of Engineering, Kyoto University, Katsura, Nishikyo-ku, Kyoto 615-8510, Japan. E-mail: otani.shunsuke.6k@kyoto-u.ac.jp; ogoshi.tomoki.3s@kyoto-u.ac.jp
bWPI Nano Life Science Institute (WPI-NanoLSI), Kanazawa University, Kakuma-machi, Kanazawa, 920-1192, Japan
cGraduate School of Natural Science and Technology, Kanazawa University, Kakuma-machi, Kanazawa, 920-1192, Japan
First published on 23rd April 2026
Abstract
We report supramolecular glass formation by mixing two crystalline pillar[6]arenes bearing benzyl and pentafluorobenzyl substituents, even though neither compound exhibited a melting point. The glass formation was attributed to arene–perfluoroarene interactions and the sterically bulky, three-dimensional pillar[6]arene framework that hinders efficient packing.
Molecular glasses, consisting of low-molecular-weight organic compounds, generally exhibit superior transparency, homogeneity, and isotropic properties compared with most polymer glasses.1 Owing to these properties, they have attracted attention for applications such as organic light-emitting diodes and resist materials.2
The most common method for preparing glass materials is melting, followed by cooling. Melting effectively disrupts ordered molecular packing, which suppresses crystallization (Fig. 1a). Therefore, compounds that cannot be melted are fundamentally unsuitable for molecular glass formation.
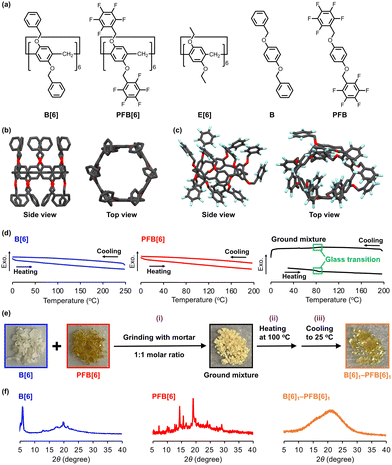
Here, we discovered that mixing two crystalline pillar[6]arenes, one having benzyl groups (B[6]) and the other having pentafluorobenzyl groups (PFB[6]), neither of which exhibits a melting point, results in the formation of a molecular glass (Fig. 1b). The B[6] and PFB[6] ground powder mixture had a glass transition and was converted into a transparent film upon heating above and then cooling below the glass transition temperature (Tg). To the best of our knowledge, this is the first example of a molecular glass prepared from two crystalline, low-molecular-weight, organic molecules, neither of which has a melting point. We attributed the glass formation to two factors: the sterically bulky pillar[6]arene scaffold and intermolecular arene–perfluoroarene (AP) interactions between the benzyl and pentafluorobenzyl groups on the two components. Strong interactions between electron-rich aromatic and electron-deficient perfluoroaromatic groups3 enhance miscibility, while the three-dimensional, distorted pillar[6]arenes4 hinder efficient packing and thus suppress crystallization.
First, we synthesized two pillar[6]arene derivatives with benzyl groups B[6] and pentafluorobenzyl groups PFB[6] (Fig. 2a). Perethylated pillar[6]arene (E[6]) and unit models of B[6] and PFB[6] without macrocyclic structures (B and PFB) were also prepared as reference compounds (Fig. 2a). All new compounds were characterized with 1H, 13C and 19F NMR spectroscopy and mass spectrometry (see supplementary information (SI)).
To obtain the structural information, single crystals of B[6] and PFB[6] were obtained (Fig. 2b, c, and Fig. S13). B[6] formed a highly symmetric pillar-shaped structure. In contrast, PFB[6] had a distorted conformation where orientations of the six units were not aligned. The PFB[6] distortion could be attributed to repulsive interactions among the electron-rich fluorine atoms in the pentafluorobenzyl groups.
Next, thermal properties of B[6] and PFB[6] were examined with thermogravimetry-differential thermal analysis (TG-DTA) and differential scanning calorimetry (DSC). The former indicated onsets of decomposition temperatures at approximately 250 °C and 200 °C for B[6] and PFB[6], respectively (Fig. S14). In the DSC curves, there were no peaks until the decomposition temperatures of B[6] and PFB[6] (Fig. 2d). These results indicated that neither B[6] nor PFB[6] underwent a melting or a glass transition prior to the thermal decomposition. This could be attributed to the high molecular rigidity arising from the presence of multiple aromatic rings in both structures. In contrast, the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 B[6]/PFB[6] ground mixture exhibited a Tg at 82 °C in the first cycle (Fig. 2d). The second cycle also exhibited the Tg at the same points during heating and cooling (Fig. S15). Hence, the ground mixture could be vitrified by heating above and then cooling below Tg.
1 B[6]/PFB[6] ground mixture exhibited a Tg at 82 °C in the first cycle (Fig. 2d). The second cycle also exhibited the Tg at the same points during heating and cooling (Fig. S15). Hence, the ground mixture could be vitrified by heating above and then cooling below Tg.
Accordingly, we prepared a molecular glass by mixing B[6] and PFB[6] in the following steps: (i) Physically mixing B[6] and PFB[6] in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio via grinding with an agate mortar, (ii) heating the mixture at 100 °C for 10 min, and (iii) cooling to 25 °C (Fig. 2e). The powder mixture changed into a transparent viscous state upon heating in step (ii). After cooling in step (iii), the viscous state did not revert to the pristine powder, but instead formed a new transparent film (denoted as B[6]1–PFB[6]1, Fig. 2e). Powder X-ray diffraction (PXRD) analysis of B[6]1–PFB[6]1 revealed only amorphous halos with no Bragg diffractions in contrast to the numerous sharp peaks of B[6] and PFB[6] (Fig. 2f). We also measured transmittance of B[6]1–PFB[6]1 by the UV-vis spectroscopy, resulting in a transmittance of 80% (Fig. S16). These results clearly demonstrate the formation of a molecular glass by mixing two crystalline, low-molecular-weight, organic compounds that had no melting points.
1 molar ratio via grinding with an agate mortar, (ii) heating the mixture at 100 °C for 10 min, and (iii) cooling to 25 °C (Fig. 2e). The powder mixture changed into a transparent viscous state upon heating in step (ii). After cooling in step (iii), the viscous state did not revert to the pristine powder, but instead formed a new transparent film (denoted as B[6]1–PFB[6]1, Fig. 2e). Powder X-ray diffraction (PXRD) analysis of B[6]1–PFB[6]1 revealed only amorphous halos with no Bragg diffractions in contrast to the numerous sharp peaks of B[6] and PFB[6] (Fig. 2f). We also measured transmittance of B[6]1–PFB[6]1 by the UV-vis spectroscopy, resulting in a transmittance of 80% (Fig. S16). These results clearly demonstrate the formation of a molecular glass by mixing two crystalline, low-molecular-weight, organic compounds that had no melting points.
The morphology of B[6]1–PFB[6]1 was investigated via scanning electron microscopy (SEM). The images showed a smooth surface with no grain boundaries at the nanometre scale, in contrast to the significant gaps and grain boundary features in B[6] and PFB[6] images (Fig. 3a). SEM-energy dispersive X-ray (EDX) spectroscopy in elemental mappings of fluorine species indicated that PFB[6] was uniformly distributed throughout B[6]1–PFB[6]1 (Fig. 3b). These results clearly demonstrated homogeneous mixing of B[6] and PFB[6] at the nanoscale (see Note S1 and Fig. S17 for the porosity of B[6]1–PFB[6]1).
 | ||
| Fig. 3 (a) SEM images of B[6], PFB[6] and B[6]1–PFB[6]1. (b) SEM-EDX elemental mapping of fluorine species on B[6]1–PFB[6]1. | ||
We also prepared mixtures with other B[6]:PFB[6] molar ratios (B[6]1–PFB[6]2 and B[6]2–PFB[6]1) using the same procedure as for the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 sample B[6]1–PFB[6]1. In contrast to B[6]1–PFB[6]1, both B[6]1–PFB[6]2 and B[6]2–PFB[6]1 were opaque solids (Fig. S18). PXRD peaks in B[6]1–PFB[6]2 were similar to those for PFB[6] (Fig. S19). Similarly, the B[6]2–PFB[6]1 peaks closely resembled those of B[6] (Fig. S20). These results indicated that B[6]1–PFB[6]2 and B[6]2–PFB[6]1 contained crystalline domains derived predominantly from the higher-ratio components in each mixture, suggesting that only the 1
1 sample B[6]1–PFB[6]1. In contrast to B[6]1–PFB[6]1, both B[6]1–PFB[6]2 and B[6]2–PFB[6]1 were opaque solids (Fig. S18). PXRD peaks in B[6]1–PFB[6]2 were similar to those for PFB[6] (Fig. S19). Similarly, the B[6]2–PFB[6]1 peaks closely resembled those of B[6] (Fig. S20). These results indicated that B[6]1–PFB[6]2 and B[6]2–PFB[6]1 contained crystalline domains derived predominantly from the higher-ratio components in each mixture, suggesting that only the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio yielded a uniform glass material.
1 molar ratio yielded a uniform glass material.
To identify the factors governing formation of the B[6]1–PFB[6]1 glass state, we performed control experiments on various pairs of B[6], PFB[6] and reference compounds E[6], B and PFB (Fig. 2a). Mixing was conducted via the three-step procedure described above.5a The resulting samples were denoted as B1–PFB1, B[6]1–PFB6, PFB[6]1–B6, B[6]1–E[6]1, and PFB[6]1–E[6]1. For B[6]1–PFB6 and PFB[6]1–B6, six molar equivalents of PFB and B were added per macrocycle.5b
B1–PFB1 was a powder and showed different diffraction peaks from those of the B and PFB constituents (Fig. S21), indicating that it was a co-crystal instead of a molecular glass. This suggested that AP interactions attributed to the relatively planar B and PFB structures. Similarly, the opaque B[6]1–PFB6 and PFB[6]1–B6 solids exhibited many sharp PXRD peaks (Fig. S22). These results demonstrated that three-dimensional scaffolds of pillar[6]arenes in the B[6] and PFB[6] constituents were important in preventing formation of crystalline assemblies. B[6]1–E[6]1 and PFB[6]1–E[6]1 were also powders, and their PXRD patterns were superposed on the those of the constituents B[6] and E[6], and PFB[6] and E[6] (Fig. S23). These results indicated that both benzyl and pentafluorobenzyl groups, as well as the sterically bulky pillar[6]arene skeleton, were necessary for molecular glass formation.
In our previous study, the introduction of fluoroalkyl groups into pillar[n]arene scaffolds enabled glass formation through a melt-cooling process.6 In contrast, this work demonstrates a distinct mechanism for supramolecular glass formation via mixing of two crystalline components, neither of which exhibits a melting transition (see Note S2 and Fig. S24 and S25 for information about pillar[5]arene derivatives).
To verify AP interactions in B[6]1–PFB[6]1, we acquired Fourier transform infrared (FT-IR) spectra for B[6], PFB[6] and B[6]1–PFB[6]1 (Fig. 4a). B[6] bands at 694 cm−1 and 733 cm−1, attributable to C–H out-of-plane bending of monosubstituted benzene rings, shifted to 699 cm−1 and 737 cm−1 in B[6]1–PFB[6]1, respectively (Fig. 4b). Additionally, the C–F stretching band of the pentafluorobenzyl groups in PFB[6] shifted from 976 cm−1 to 979 cm−1 in B[6]1–PFB[6]1 (Fig. 4c). These shifts indicated that benzyl groups in B[6] and pentafluorobenzyl groups in PFB[6] participated in AP interactions upon glass formation.
 | ||
| Fig. 4 (a) FT-IR spectra of B[6], PFB[6] and B[6]1–PFB[6]1. Enlarged views of (a) in the (b) C–H out-of-plane bending and (c) C–F stretching regions. | ||
Furthermore, TG curve for B[6]1–PFB[6]1 exhibited only one degradation step, with the decomposition onset temperature at approximately 230 °C, which was between those of B[6] and PFB[6] (Fig. S26 and S27). This suggested that B[6]1–PFB[6]1 uniformly decomposed as a consequence of the intimate mixing. Interestingly, over 40% of the B[6]1–PFB[6]1 weight remained even after heating to 900 °C, in contrast to the absence of residual mass for B[6] and PFB[6], indicating strong AP interactions between B[6] and PFB[6] in B[6]1–PFB[6]1. Taken together, the FT-IR and TGA results both verified that AP interactions were formed and that the equimolar glass was both compositionally homogeneous and thermally robust.
In general, amorphous mixtures remain stable against crystallization (devitrification) when maintained approximately 50 K below their Tg. This is because the mobilities of the component molecules are greatly reduced.7 In this context, the stability of B[6]1–PFB[6]1, which exhibited a Tg at 82 °C, was examined at room temperature. The appearances, PXRD patterns and transmittances did not change even after 6 months in air (Fig. S28), indicating that the glass state of B[6]1–PFB[6]1 was stable at room temperature for at least half a year.
Previous studies have reported that assemblies composed of arene and perfluoroarene exhibit room-temperature phosphorescence (RTP) because of accelerated intersystem crossing with enhanced spin–orbit coupling via AP interactions.8 However, the effect has been predominantly observed in crystalline materials. Motivated by these studies, we examined the optical properties of glassy B[6]1–PFB[6]1. Kubelka–Munk transformed diffuse reflectance (DR) spectrum of B[6]1–PFB[6]1 revealed a broad absorption band with a maximum at 344 nm (Fig. S29). Upon excitation at 344 nm at room temperature, B[6]1–PFB[6]1 gave a single phosphorescence band at 562 nm with a lifetime of 70 ms (Fig. 5a, b and see Note S3 and S30, S31 for further phosphorescence measurements under controlled atmosphere and variable temperature conditions). In contrast, there were no peaks in the spectra for B[6] or PFB[6] (Fig. S32). Although the RTP of B[6]1–PFB[6]1 was too weak to be visible at room temperature, yellow-green emission was confirmed upon cooling to 77 K (Fig. 5c). The co-crystal of unit models B1–PFB1 also emitted RTP at 559 nm with a lifetime of 82 ms (Fig. S33). Therefore, AP interactions were essential for RTP emission in this system. RTP-active molecular glasses are rare,9 and, to the best of our knowledge, this is the first example of exploiting AP interactions to form a molecular glass that exhibits RTP.
In conclusion, we have successfully prepared a molecular glass by mixing two types of pillar[6]arene derivatives: one bearing benzyl groups and the other bearing pentafluorobenzyl groups. Remarkably, even though neither crystalline constituent exhibited a distinct Tm, the glass readily formed via simple physical mixing, followed by mild thermal treatment. Comprehensive investigations revealed that the glass formation was driven by two key factors: the sterically bulky architecture of the pillar[6]arene skeleton and intermolecular AP interactions.
The AP interactions enabled RTP, which is rare in molecular glasses. Because of their high optical transparency, suppression of self-quenching and light scattering and excellent processability into thin films and coatings, this molecular glass is a promising RTP emitter. Overall, our findings provide new design principles for molecular glasses and highlight their potential as materials for practical applications.
Tomoki Ogoshi and Shunsuke Ohtani conceptualized and supervised the work., Katsuto Onishi and Shigehisa Akine designed and performed experiments. The first draft of the manuscript was written by Katsuto Onishi, Shunsuke Ohtani, and Tomoki Ogoshi. All authors analyzed and discussed the results, and co-wrote the paper.
Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article have been included in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc01163b.CCDC 2531301 and 2531501 contain the supplementary crystallographic data for this paper.10a,b
Acknowledgements
This work was supported by JST CREST Grant Number JPMJCR24S6 (T. O.), Japan, JSPS KAKENHI Grant Numbers JP25K18086 (Early-Career Scientists, S. O.), JP22K19063 (Challenging Research (Exploratory), T. O.), JP25H00896 (Scientific Research (A), T. O.), JP24K21247 (Challenging Research (Pioneering), T. O.), JST SPRING, Grant Number JPMJSP2110 (K. O.), JSPS Research Fellowship for Young Scientists: JP25KJ1616 (K. O.), and the MEXT World Premier International Research Center Initiative (WPI), Japan. Dr Takeshi Yamamoto and Prof. Dr Michinori Suginome (Kyoto University) for PXRD measurement. We thank Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.References
- (a) Y. Shirota, J. Mater. Chem., 2000, 10, 1–25 RSC; (b) Y. Shirota, J. Mater. Chem., 2005, 15, 75–93 RSC.
- (a) L.-H. Chan, R.-Ho Lee, C.-F. Hsieh, H.-C. Yeh and C.-T. Chen, J. Am. Chem. Soc., 2002, 124, 6469–6479 CrossRef CAS PubMed; (b) A. D. Silva, N. M. Felix and C. K. Ober, Adv. Mater., 2008, 20, 3355–3361 CrossRef; (c) J. Chen, Q. Hao, S. Wang, S. Li, T. Yu, Y. Zeng, J. Zhao, S. Yang, Y. Wu, C. Xue, G. Yang and Y. Li, Acs. Appl. Polym. Mater., 2019, 1, 526–534 CrossRef CAS; (d) D. Umeyama, S. Horike, M. Inukai, T. Itakura and S. Kitagawa, J. Am. Chem. Soc., 2015, 137, 864–870 CrossRef CAS PubMed; (e) T. D. Bennett and S. Horike, Nat. Rev. Mater., 2018, 3, 431–440 CrossRef; (f) N. Ma, H. Ando, R. Ma and T. Nakanishi, Small, 2026, e14459 CrossRef CAS PubMed; (g) X. Jiang, X. Cui, A. J. E. Duncan, L. Li, R. P. Hughes, R. J. Staples, E. V. Alexandrov, D. M. Proserpio, Y. Wu and C. Ke, J. Am. Chem. Soc., 2019, 141, 10915–10923 CrossRef CAS PubMed.
- (a) O. R. Lozman, R. J. Bushby and J. G. Vinter, J. Chem. Soc., Perkin Trans., 2001, 2, 1446–1452 RSC; (b) S. Lorenzo, G. R. Lewis and I. Dance, New J. Chem., 2000, 24, 295–304 RSC; (c) J. Hernández-Trujillo, F. Colmenares, G. Cuevas and M. Costas, Chem. Phys. Lett., 1997, 265, 503–507 CrossRef; (d) A. P. West, Jr., S. Mecozzi and D. A. Dougherty, J. Phys. Org. Chem., 1997, 10, 347–350 CrossRef; (e) Z. Huang, X. Chen, G. Wu, P. Metrangolo, D. Whitaker, J. A. McCune and O. A. Scherman, J. Am. Chem. Soc., 2020, 142, 7356–7361 CrossRef CAS PubMed; (f) H. Zhang, J. Han, X. Jin and P. Duan, Angew. Chem., Int. Ed., 2021, 60, 4575–4580 CrossRef CAS PubMed; (g) W. Wu, C. Ye, J. Qin and Z. Li, ACS Appl. Mater. Interfaces, 2013, 5, 7033–7041 CrossRef CAS PubMed; (h) X. Ji, H. Wang, H. Wang, T. Zhao, Z. A. Page, N. M. Khashab and J. L. Sessler, Angew. Chem., Int. Ed., 2020, 59, 23402–23412 CrossRef CAS PubMed.
- (a) T. Ogoshi, T. Yamagishi and Y. Nakamoto, Chem. Rev., 2016, 116, 7937–8002 CrossRef CAS PubMed; (b) K. Jie, Y. Zhou, E. Li, Z. Li, R. Zhao and F. Huang, J. Am. Chem. Soc., 2017, 139, 15320–15323 CrossRef CAS PubMed; (c) T. Ogoshi, K. Saito, R. Sueto, R. Kojima, Y. Hamada, S. Akine, A. M. P. Moeljadi, H. Hirao, T. Kakuta and T. Yamagishi, Angew. Chem., Int. Ed., 2018, 57, 1592–1595 CrossRef CAS PubMed; (d) K. Jie, M. Liu, Y. Zhou, M. A. Little, A. Pulido, S. Y. Chong, A. Stephenson, A. R. Hughes, F. Sakakibara, T. Ogoshi, F. Blanc, G. M. Day, F. Huang and A. I. Cooper, J. Am. Chem. Soc., 2018, 140, 6921–6930 CrossRef CAS PubMed; (e) K. Jie, Y. Zhou, E. Li and F. Huang, Acc. Chem. Res., 2018, 51, 2064–2072 CrossRef CAS PubMed; (f) K. Jie, Y. Zhou, E. Li, R. Zhao and F. Huang, Angew. Chem., Int. Ed., 2018, 57, 12845–12849 CrossRef CAS PubMed.
-
(a) The heating temperatures in the step (ii) were set between the melting points and the thermal decomposition temperatures of the components;
(b) In the preparation of mixtures of B[6] and PFB, and PFB[6] and B, the number of moles of benzyl and pentafluorobenzyl groups were equalized to provide proper comparisons with B[6]1–PFB[6]1. To this end, the corresponding pillar[6]arenes and unit models were mixed in a 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 molar ratio in both cases.
6 molar ratio in both cases. - K. Onishi, S. Ohtani, K. Kato, S. Fa, Y. Sakata, S. Akine, M. Ogasawara, H. Asakawa, S. Nagano, Y. Takashima, M. Mizuno and T. Ogoshi, Chem. Sci., 2022, 13, 4082–4087 RSC.
- (a) L. Yu, Adv. Drug Delivery Rev., 2001, 48, 27–42 CrossRef CAS PubMed; (b) S. V. Bhujbal, B. Mitra, U. Jain, Y. Gong, A. Agrawal, S. Karki, L. S. Taylor, S. Kumar and Q. T. Zhou, Acta Pharm. Sin. B, 2021, 11, 2505–2536 CrossRef CAS PubMed.
- (a) Q. Zhang, Y. Fan, Q. Liao, C. Zhong, Q. Li and Z. Li, Sci. China: Chem., 2022, 65, 918–925 CrossRef CAS; (b) X. Pang, H. Wang, W. Wang and W. J. Jin, Cryst. Growth Des., 2015, 15, 4938–4945 CrossRef CAS; (c) Q. Liao, Q. Li and Z. Li, Adv. Mater., 2025, 37, 2306617 CrossRef CAS PubMed.
- (a) J. J. Flynn, Z. M. Marsh, D. M. Krein, S. M. Wolf, J. E. Haley, E. S. Vasquez, T. M. Cooper, N. P. Godman and T. A. Grusenmeyer, ChemPhotoChem, 2022, 6, e202200134 CrossRef CAS; (b) M. Zhang, X. Lan, M. Ding, C. Han, X. W. Liu, Z. Meng, Z.-Q. Yu and Z. An, Angew. Chem., Int. Ed., 2025, 64, e202415250 CrossRef CAS PubMed.
- (a) CCDC 2531301: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qz0wm; (b) CCDC 2531501: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qz7b9.
| This journal is © The Royal Society of Chemistry 2026 |