Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis of bisenarsan; an organoarsenic metabolite from streptomycetes
Ihor Kholomieiev ,
Jeya Prathap Kaniraj*,
Rafael Tarozo and
Adriaan J. Minnaard
,
Jeya Prathap Kaniraj*,
Rafael Tarozo and
Adriaan J. Minnaard *
*
Stratingh Institute for Chemistry, University of Groningen, Nijenborgh 7, NL-9747 AG, Groningen, The Netherlands. E-mail: j.p.kaniraj@rug.nl; a.j.minnaard@rug.nl
First published on 20th March 2026
Abstract
The first synthesis of bisenarsan; the (4S,6S,Z)-2,4,6-trimethylnon-2-enoic acid ester of (2-hydroxyethyl)arsonic acid, is described. Its absolute configuration is experimentally determined by preparing both enantiomers and comparing these to the natural isolate. The synthesis comprises 10 steps and required the development of an improved, versatile (2-hydroxyethyl)arsonic acid building block. The availability of bisenarsan together with the acquired insight into organoarsenic chemistry supports studies on the unique bioactivities of arsenic-containing secondary metabolites.
Out of over 400
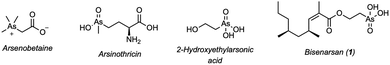
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 natural products reported (through 2022), only approximately 300 contain arsenic.1,2 Although rare, these organoarsenicals exhibit a considerable structural, biological, and biosynthetic diversity. Many of them are formed via methylation of inorganic As(III), ultimately providing a detoxification pathway for inorganic arsenic. These methylated organoarsenicals serve as precursors to most other organoarsenic natural products, such as arsenobetaine (Fig. 1), a nontoxic compound found to be the major arsenic-containing species in a variety of marine animals and terrestrial fungi.3–6 The antibiotic arsinothricin, isolated from B. gladioli, is instead believed to be formed by reductive alkylation of inorganic As(III) with the amino acid residue, followed by methylation and oxidation.7,8
000 natural products reported (through 2022), only approximately 300 contain arsenic.1,2 Although rare, these organoarsenicals exhibit a considerable structural, biological, and biosynthetic diversity. Many of them are formed via methylation of inorganic As(III), ultimately providing a detoxification pathway for inorganic arsenic. These methylated organoarsenicals serve as precursors to most other organoarsenic natural products, such as arsenobetaine (Fig. 1), a nontoxic compound found to be the major arsenic-containing species in a variety of marine animals and terrestrial fungi.3–6 The antibiotic arsinothricin, isolated from B. gladioli, is instead believed to be formed by reductive alkylation of inorganic As(III) with the amino acid residue, followed by methylation and oxidation.7,8
An intriguing natural product the biosynthesis of which is not initiated by reductive methylation is bisenarsan (1), isolated from Streptomyces lividans in 2023 by Hoshino et al.2 Bisenarsan is the first secondary metabolite isolated from actinomycetes, synthesized from (2-hydroxyethyl)arsonic acid, which is an unprecedented moiety in natural products.2 The C-As bond in bisenarsan is believed to be formed by an intramolecular rearrangement of arsenoenolpyruvate, an arsenic analogue of phosphoenolpyruvate. The rearranged organoarsenic product then undergoes enzymatic decarboxylation, reduction and O-acylation.9 The structure of bisenarsan, including the configuration of the alkene and relative stereochemistry of the methyl branches, was established using mass spectroscopy and detailed NMR studies, while the absolute stereochemistry was predicted based on bioinformatics analysis.
Although the biological role of bisenarsan is still unclear, it was proposed that this natural product acts as a protoxin, forming toxic (2-hydroxyethyl)arsonic acid after hydrolysis. It is possible that this mechanism allows Streptomyces lividans to target organisms possessing esterases that facilitate this reaction, although no such strains have been identified.2
As the number of organoarsenic natural products is so limited, synthetic methods in this field have been underexplored, although the isolation of arsenicin10,11 and arsinothricin12–14 has elicited important developments in this area. Together with the unique structure of bisenarsan and its unknown biological role, this was reason enough to prepare this natural product. Furthermore, to experimentally determine its absolute configuration, it was necessary to prepare both of its enantiomers.
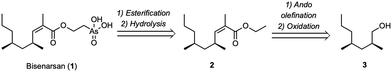
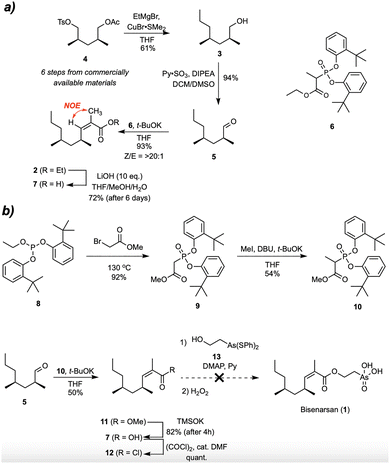
Bisenarsan is characterized by three stereogenic elements; two methyl-branched stereocenters and a trisubstituted Z-alkene. We envisioned that it could be prepared by esterification of a protected (2-hydroxyethyl)arsonic acid derivative with the corresponding acyl chloride. The required acyl chloride could be prepared from the known (2S,4S)-2,4-dimethylheptan-1-ol 3 using a highly stereoselective Ando olefination reaction, leading to ester 2, followed by hydrolysis and activation (Fig. 2).
To this end, known tosylate 4, prepared via enzymatic desymmetrization of readily available 2,4-dimethylpentane-1,5-diol,15 was treated with excess ethylmagnesium bromide and substoichiometric CuBr·SMe2, to provide alcohol 3 in 61% yield (Fig. 3a). 3 was converted to the corresponding aldehyde 5 under Parikh-Doering conditions in 94% yield, and reacting 5 with the known phosphonate 6 and t-BuOK resulted in a highly Z-selective (Z/E > 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 by 1H NMR) Ando olefination that gave ester 2 in an excellent 93% yield. Hydrolysis of ester 2 turned out to be sluggish, and the reaction using aqueous lithium hydroxide took 6 d, providing acid 7 in 72% yield. This decreased reactivity of branched unsaturated esters is known in literature and also observed with esters of the related angelic acid.16
1 by 1H NMR) Ando olefination that gave ester 2 in an excellent 93% yield. Hydrolysis of ester 2 turned out to be sluggish, and the reaction using aqueous lithium hydroxide took 6 d, providing acid 7 in 72% yield. This decreased reactivity of branched unsaturated esters is known in literature and also observed with esters of the related angelic acid.16
 | ||
| Fig. 3 (a) The first-generation synthetic route. (b) The optimised synthetic route to carboxylic acid 7 and the unsuccessful esterification. | ||
To facilitate hydrolysis towards 7, we sought to prepare its corresponding methyl ester instead. Michaelis–Arbuzov reaction of phosphite 8 and methyl bromoacetate yielded phosphonate 9 in 92% yield, which was then mono-methylated using the procedure developed by Bressin and coworkers17 to give the desired Ando reagent 10 (Fig. 3b). Olefination of the aldehyde 5 with this reagent gave methyl ester 11, which was converted to 7 by TMSOK-promoted demethylation with an improved yield and reaction time (82%, 4 h) compared to the hydrolysis of 2. To prepare bisarsenan, activated 7 had to be coupled to (2-hydroxyethyl)arsonic acid. However, to avoid interference of the arsonic acid function and allow its solubilization in organic solvents, this group has to be masked. As shown by Ioannou et al.,18,19 this is commonly done by treatment of (2-hydroxyethyl)arsonic acid with an excess of thiophenol (not shown), which converts the arsonic acid group to a rather labile diphenyl dithioarsonite group in 13. Carboxylic acid 7 was converted to the corresponding acyl chloride 12 ((COCl)2, cat. DMF), but the esterification (pyridine, DMAP)18,20 with 13 did not lead to the expected product. When an equimolar or excess amount of 13 was employed, decomposition of the acyl chloride took place, while with an excess of the acyl chloride the reaction did not proceed to completion even after a prolonged time at elevated temperature. Referring to the earlier noted lack of reactivity of the carboxylic function in 7, we attempted to prepare the corresponding acyl fluoride (not shown), which was expected to make the carbonyl group more accessible owing to the smaller atomic radius of fluorine compared to that of chlorine.21 Disappointingly, treating carboxylic acid 7 with NMe4SCF322 yielded an inseparable mixture of products arising, inter alia, from Z/E-isomerisation, which has also been observed for derivatives of angelic acid.16 These results prompted us to revise our synthetic approach.
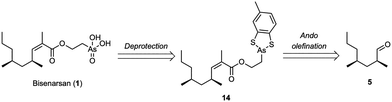
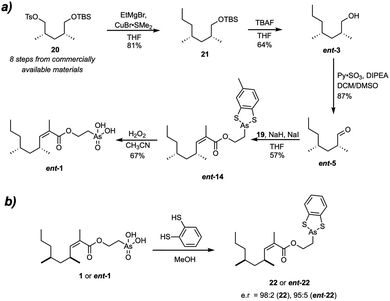
To circumvent the reluctant hydrolysis and esterification reactions altogether, we explored the design of a novel Ando reagent already containing the masked arsonic acid. In addition this approach would lead to a shorter and more convergent route. The diphenyl dithioarsonite group, however, was considered unsuitable for this application because it is incompatible with the basic conditions of the olefination step. Furthermore, its instability on silica is undesirable. We envisioned that a cyclic dithiocatechol-type arsonite would be more stable, in analogy with the chelate effect in transition metal complexes. With this in mind, it was proposed to prepare bisenarsan by deprotection of the ester 14, which would in turn be prepared by olefination of aldehyde 5 with a suitable Ando reagent (Fig. 4).
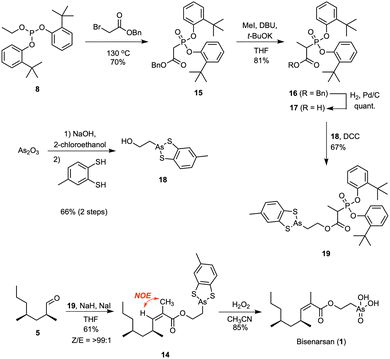
The synthesis of the required Ando reagent 19 commenced with the Michaelis-Arbuzov reaction between benzyl bromoacetate and phosphite 8, which yielded phosphonate 15 in 70% yield (Fig. 5). Methylation yielded benzyl ester 16 in 81% yield, which was quantitatively converted to the corresponding carboxylic acid 17 by hydrogenolysis. The protected (2-hydroxyethyl)arsonic acid fragment 18 could be prepared on gram scale in a two-step procedure. Treating arsenic trioxide (As2O3) with NaOH and 2-chloroethanol provided (2-hydroxyethyl)arsonic acid,18 which was reacted in situ with toluene-3,4-dithiol, a less expensive derivative of benzene-1,2-dithiol. Alcohol 18 prepared this way in 66% yield could be purified on silica and stored in air for weeks without noticeable decomposition. Since purification of (2-hydroxyethyl)arsonic acid requires tedious ion exchange chromatography, and its dithiophenol derivative 13 decomposes on silica, this procedure represents a significant improvement in the synthesis of (2-hydroxyethyl)arsonic acid derivatives.
Alcohol 18 was esterified with carboxylic acid 17 using DCC without an additional catalyst to give the phosphonate 19 in 67% yield. The remarkable activity of carboxylic acids substituted with an electron-withdrawing group in the α-position towards DCC-mediated esterifications is rationalised by the intermediacy of a corresponding ketene.23 Subsequent olefination of the aldehyde 5 with 19 provided the desired ester 14 in a satisfying 61% yield (Z/E > 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 by 1H NMR). Finally, treatment of 14 with aqueous H2O2 in CH3CN under optimised conditions removed the dithiocatechol protecting group and oxidized the arsonite to the corresponding arsonic acid, providing (S,S)-bisenarsan 1 in 85% yield. Careful comparison of the NMR spectra and mass spectral data showed that the synthetic material was identical to the natural product (see SI).
1 by 1H NMR). Finally, treatment of 14 with aqueous H2O2 in CH3CN under optimised conditions removed the dithiocatechol protecting group and oxidized the arsonite to the corresponding arsonic acid, providing (S,S)-bisenarsan 1 in 85% yield. Careful comparison of the NMR spectra and mass spectral data showed that the synthetic material was identical to the natural product (see SI).
To experimentally determine the absolute configuration of natural bisenarsan, it was necessary to also synthesise the other enantiomer, ent-1. To this end, tosylate 20 was reacted with ethylmagnesium bromide using Corey-House conditions identical to those employed in synthesis of 3, yielding silyl ether 21 in 81% yield (Fig. 6a). Deprotection of 21 with TBAF provided ent-5 in 64% yield. ent-5 was then converted to (R,R)-configured ent-1 in the same way as its enantiomer.
With both enantiomers of synthetic bisenarsan in hand, together with sub-mg quantities of the natural isolate, we turned our attention to the determination of its absolute stereochemistry. The conventional way to determine the stereochemistry of methyl-branched polyketides is the conversion to their methyl esters, followed by analysis with chiral GC.15 With the reluctance of the ester group in bisenarsan towards hydrolysis in mind, we chose another approach. A dithioarsonite of bisarsenan, like 14, could be suitable for analysis by chiral HPLC using UV-detection. As 14 itself is chiral at arsenic, a phenomenon that became immediately apparent upon analysis by chiral HPLC, the derivatization of synthetic and natural bisenarsan was carried out with benzene-1,2-dithiol. Indeed, upon conversion of 1 and ent-1 to 22 and ent-22, respectively (Fig. 6b), baseline resolution was obtained using a Chiralpak AD-H column, with n-heptane/i-PrOH: 99.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 as mobile phase. Conversion of 50 µg of the natural isolate unambiguously showed the 4S,6S-stereochemistry of bisenarsan, thereby confirming the prediction made using bioinformatics analysis2 (See Fig. S1–S4). The enantiomeric purity of the synthesised products was similarly assessed (e.r. = 98
0.5 as mobile phase. Conversion of 50 µg of the natural isolate unambiguously showed the 4S,6S-stereochemistry of bisenarsan, thereby confirming the prediction made using bioinformatics analysis2 (See Fig. S1–S4). The enantiomeric purity of the synthesised products was similarly assessed (e.r. = 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 for 22 and 95
2 for 22 and 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 for ent-22) by utilising chiral HPLC of 22 and ent-22 (Fig. S2 and S3). To our knowledge, this is the first report using the benzene-1,2-dithiol derivatisation of arsonic acids for chromatographic analysis.
5 for ent-22) by utilising chiral HPLC of 22 and ent-22 (Fig. S2 and S3). To our knowledge, this is the first report using the benzene-1,2-dithiol derivatisation of arsonic acids for chromatographic analysis.
In conclusion, the first synthesis of bisenarsan has been accomplished in 10 steps. The synthesis led to an improved synthesis of a masked (2-hydroxyethyl)arsonic acid building block and an extension of the use of Ando olefination reagents. Utilising a benzene-1,2-dithiol derivatisation reaction, the synthetic products and the natural isolate were analysed by chiral HPLC to confirm the absolute stereochemistry of bisenarsan. These acquired tools will be helpful in the synthesis of newly discovered organoarsenic metabolites.
JPK and AJM conceptualised the study, IK and RT carried out the investigation and data analysis. The manuscript was drafted by IK and edited by all authors. All authors approved the final version of the manuscript.
The authors thank Prof. Dr Onaka and Prof. Dr Hoshino for kindly providing a sample of the natural bisenarsan and raw NMR data.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article, such as experimental procedures and NMR spectra have been included in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc01090c.References
- M. Sorokina and C. Steinbeck, J. Cheminf., 2020, 12, 20 CAS.
- S. Hoshino, S. Ijichi, S. Asamizu and H. Onaka, J. Am. Chem. Soc., 2023, 145, 17863–17871 CrossRef CAS PubMed.
- X.-M. Xue, J. Ye, G. Raber, K. A. Francesconi, G. Li, H. Gao, Y. Yan, C. Rensing and Y.-G. Zhu, Environ. Sci. Technol., 2017, 51, 1224–1230 CrossRef CAS PubMed.
- J. Edmonds and K. Francesconi, Nature, 1981, 289, 602–604 CrossRef CAS.
- W. R. Cullen and K. J. Reimer, Chem. Rev., 1989, 89, 713–764 CrossRef CAS.
- Z. Ålejkovec, A. R. Byrne, T. Stijve, W. Goessler and K. J. Irgolic, Appl. Organomet. Chem., 1997, 11, 673–682 CrossRef.
- M. Kuramata, F. Sakakibara, R. Kataoka, K. Yamazaki, K. Baba, M. Ishizaka, S. Hiradate, T. Kamo and S. Ishikawa, Environ. Chem., 2016, 13, 723–731 CrossRef CAS.
- Y. Yao, J. He, F. Chen and M. Dong, J. Am. Chem. Soc., 2024, 146, 21214–21219 CrossRef PubMed.
- S. Hoshino, S. Ijichi and H. Onaka, Chem. Pharm. Bull., 2025, 73, 698–706 CrossRef CAS PubMed.
- D. Lu, A. D. Rae, G. Salem, M. L. Weir, A. C. Willis and S. B. Wild, Organometallics, 2010, 29, 32–33 CrossRef CAS.
- D. Lu, M. L. Coote, J. Ho, N. L. Kilah, C.-Y. Lin, G. Salem, M. L. Weir, A. C. Willis, S. B. Wild and P. J. Dilda, Organometallics, 2012, 31, 1808–1816 CrossRef CAS.
- A. H. Howlader, S. H. Suzol, V. S. Nadar, A. E. Galván, A. Nedovic, P. Cudic, B. P. Rosen, M. Yoshinaga and S. F. Wnuk, RSC Adv., 2021, 11, 35600–35606 RSC.
- H. Li, F. Chen, W. Ding and Q. Zhang, ACS Catal., 2025, 15, 494–501 CrossRef CAS.
- S. H. Suzol, A. Hasan Howlader, A. E. Galván, M. Radhakrishnan, S. F. Wnuk, B. P. Rosen and M. Yoshinaga, J. Nat. Prod., 2020, 83, 2809–2813 CrossRef CAS PubMed.
- Z. Lin, J. P. Kaniraj, M. Holzheimer, J. Nigou, M. Gilleron, J. Hekelaar and A. J. Minnaard, Angew. Chem., Int. Ed., 2024, 63, e202318582 CrossRef CAS PubMed.
- X. Liang, G. Grue-Sørensen, A. K. Petersen and T. Högberg, Synlett, 2012, 2647–2652 CrossRef.
- R. K. Bressin, J. L. Driscoll, Y. Wang and K. Koide, Org. Process Res. Dev., 2019, 23, 274–277 CrossRef CAS.
- S. Mourtas, K. Papadia, G. G. Kordopati, P. V. Ioannou, S. G. Antimisiaris and G. M. Tsivgoulis, Pharmaceutics, 2022, 14, 1649 CrossRef CAS PubMed.
- P. V. Ioannou, Chem. Phys. Lipids, 2002, 117, 7–18 CrossRef CAS PubMed.
- P. V. Ioannou and G. M. Tsivgoulis, Chem. Phys. Lipids, 2010, 163, 51–55 CrossRef CAS PubMed.
- C. Bonnefoy, A. Gallego, C. Delobel, B. Raynal, M. Decourt, E. Chefdeville, G. Hanquet, A. Panossian, F. R. Leroux, F. Toulgoat and T. Billard, Eur. J. Org. Chem., 2024, e202400142 CrossRef CAS.
- T. Scattolin, K. Deckers and F. Schoenebeck, Org. Lett., 2017, 19, 5740–5743 CrossRef CAS PubMed.
- M. Nahmany and A. Melman, Org. Lett., 2001, 3, 3733–3735 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |