Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Tetra-substituted BDPA radicals via click-chemistry and application to liquid-state DNP
Iram M.
Ahmad
 a,
Pralambika
Roy
a,
Pralambika
Roy
 a,
Andrei
Kuzhelev
a,
Andrei
Kuzhelev
 *b and
Snorri Th.
Sigurdsson
*b and
Snorri Th.
Sigurdsson
 *a
*a
aUniversity of Iceland, Department of Chemistry, Science Institute, Dunhaga 3, Reykjavik 107, Iceland. E-mail: snorrisi@hi.is
bInstitute of Physical and Theoretical Chemistry and Center for Biomolecular Magnetic Resonance (BMRZ), Goethe University, 60438, Frankfurt am Main, Germany
First published on 13th May 2026
Abstract
The 1,3-bisdiphenylene-2-phenylallyl (BDPA) radical is a promising polarizing agent for DNP NMR, but is limited by poor persistence. A divergent synthetic strategy, using copper(I)-catalyzed azide–alkyne cycloaddition, is presented for preparing tailored BDPA derivatives. A high-molecular-weight, sterically shielded BDPA-dendrimer showed improved persistence and the highest liquid-state DNP enhancement reported thus far.
Nuclear magnetic resonance (NMR) spectroscopy is an important analytical technique for investigating the structure and dynamics of biomolecules and materials.1–4 However, a major drawback of NMR is its low sensitivity, which is due to the small energy difference between the ground and excited states of nuclear spins in a magnetic field. Dynamic nuclear polarization (DNP) NMR has emerged as a powerful technique to overcome this drawback, by transferring the much higher polarization of unpaired electrons to the nuclei of interest.5–7 In DNP NMR, the sample is doped with a paramagnetic molecule, referred to as a polarizing agent, and irradiated with microwaves (µw) during signal acquisition.8,9 Persistent organic radicals are the most commonly used polarizing agents.
Bis-nitroxide biradicals are a class of polarizing agents that are fairly easy to synthesize and have extensively been used for DNP at magnetic fields of 9.4 and 14.1 T.10–14 AsymPol-POK (Fig. 1) is a prominent example for solid-state DNP in aqueous solutions, offering high DNP performance at these magnetic fields.12,15 With recent technological advances, DNP NMR has been extended to even higher magnetic fields (≥18.8 T) in order to improve both the signal-to-noise ratio and signal resolution.16 However, the performance of nitroxides drops at very high magnetic fields due to shorter electronic relaxation times and a concomitant broader EPR signal, which broadens linearly with the external magnetic field.16,17 This results in reduced excitation of the electronic spins and thus, lower DNP enhancement. Moreover, loss of nuclear polarization through depolarization becomes prominent for nitroxides at very high fields.18
 | ||
| Fig. 1 Persistent radicals used as polarizing agents for DNP NMR: AsymPol-POK, Finland trityl, OX063, BDPA and trimethylammonium BDPA (BDPA-NMe3). | ||
To overcome the drawbacks associated with nitroxides, carbon-based radicals like Finland trityl18–20 and 1,3-bisdiphenylene-2-phenylallyl (BDPA)21 (Fig. 1) have attracted attention as promising radicals for high field DNP-NMR. The isotropic g-values of carbon lead to a much lower or even non-existing depolarization.18,21 Furthermore, the narrow EPR line of carbon radicals in the solid-state and their long electron spin–lattice relaxation times (T1e) enable efficient saturation at comparatively lower microwave power.16,22
An advantage of the BDPA radical over the Finland trityl is its relative ease of synthesis.23,24 Moreover, its EPR signal is narrower and more isotropic than the Finland trityl radical.22 BDPA radicals have been used for DNP in both liquids and solids,22,25–31 where they exhibit multiple polarization pathways. Traditionally, the Overhauser effect (OE) was considered exclusive to liquids, relying on fast molecular motion to mediate electron-nuclear cross-relaxation.32,33 However, BDPA was shown to have the ability to operate in solids through the OE.26,27 This behaviour has been linked to its mixed-valence character and intramolecular charge-transfer dynamics.34 Furthermore, Kuzhelev et al. have also shown that BDPA monoradicals can polarize fluid lipid membranes and analytes in viscous solutions through the solid effect (SE) mechanism at high magnetic fields, previously only observed in solids.29,30 When molecular tumbling of an analyte in solution is reduced,30,35 electron-nuclear dipolar interactions slow down, which allows efficient polarization transfer and sizable signal enhancements.36 Such polarization can also be observed for large molecules (with a long rotational correlation time) in aqueous solutions.29,37 Thus, BDPA radicals exhibit versatile and interesting DNP properties for solid- and solution-state alike. Nonetheless, BDPA radicals have limitations.
Two major drawbacks of BDPA-based radicals have been their low persistence and limited solubility in aqueous solutions for use in structural biology.38 A new class of tetraalkylammonium BDPA derivatives has partially addressed these shortcomings by providing more persistent radicals with tuneable solubility.39 However, the water-soluble trimethylammonium BDPA derivative (BDPA-NMe3) (Fig. 1), has limited persistence in aqueous solutions, probably due to the tendency of tetraalkylammonium salts to aggregate in water,40–42 leading to dimerization.38,39 Attaching bulky substituents to BDPA should reduce its tendency to aggregate and thereby enhance its stability.43,44 Here we describe a convergent synthetic strategy that enables conjugation of a variety of different substituents to BDPA by Cu-catalyzed azide–alkyne cycloaddition (CuAAC) with a focus on the incorporation of hydrophilic and sterically demanding groups. This approach provides flexibility in preparing various substituted BDPA derivatives with tailored properties and complements the strategy of increasing the persistence of BDPA by changing the electronic properties of the aromatic rings.45,46 Of the BDPA derivatives described here, a BDPA-conjugated dendrimer was particularly promising, with improved persistence in water and the highest reported DNP enhancement thus far in viscous liquids.
The synthetic strategy was based on the known tetrabromo BDPA derivative 139 and its conversion to tetraazide BDPA 2 (Scheme 1). This tetraazide can be readily conjugated to various alkyne-bearing substituents by CuAAC, including hydrophilic and sterically demanding groups. We chose four different alkynes with distinct properties: propargyl alcohol (3a), which can be further derivatized; a polyhydroxyamide derivative (3b)47 to use in sorbitol-based glass matrices for DNP; a glucose derivative (3c)48 as a neutral and hydrophilic moiety that could provide some steric shielding; and a dendrimer alkyne (3d) to impart solubility and extensive steric effects (Scheme 1).
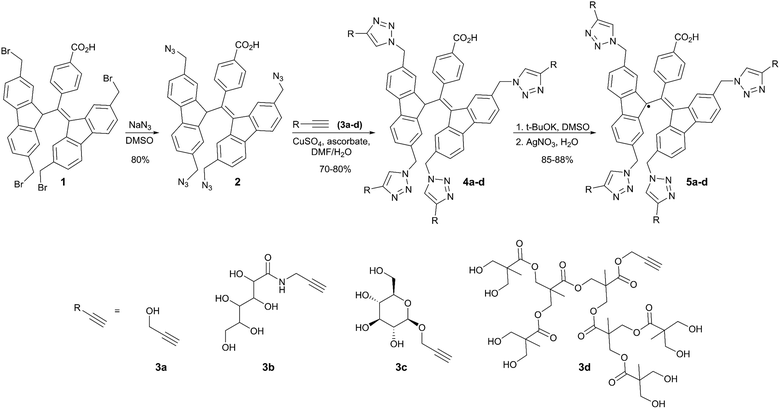 | ||
| Scheme 1 Synthesis of water-soluble BDPA radicals 5a–dvia CuAAC of tetraazide 2 and their corresponding alkyne substrates 3a–d. | ||
The synthesis began with the azidation of the tetrabromide 139 to give the tetraazide BDPA derivative 2 in excellent yield (Scheme 1). The click reaction of 2 with the alkynes (3a–d) was straightforward, however, the purification of BDPA derivatives 4a–d was challenging due to either limited solubility (4a and 4b) or very high polarity (4c and 4d). For derivatives 4a, 4b and 4c, precipitation with Et2O gave fairly pure products with good yields. Compound 4d was purified by flash column chromatography. The corresponding BDPA radicals 5a–d were prepared by treating derivatives 4a–d sequentially with t-BuOK and AgNO3 (Scheme 1). Since compound 4d was prone to hydrolysis in the presence of base, a shorter reaction time was used for the deprotonation with t-BuOK (see SI for details).
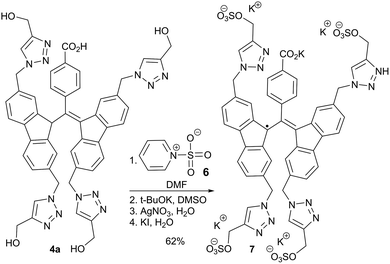
BDPA radicals 5a–d were all soluble in DMSO, while only 5c and 5d were soluble in water. On the other hand, compound 5b was unexpectedly insoluble in water, despite being a sugar-based derivative like 5c. The insolubility of 5b in water is likely due to a combination of intermolecular hydrogen bonding involving the carbohydrate moieties and π–π stacking of the BDPA cores. As anticipated, the tetrahydroxyl BDPA radical 5a exhibited negligible solubility in water. However, the hydroxyl groups can be readily phosphorylated12 or converted to sulfates to dramatically increase hydrophilicity.49 Thus, the tetrasulfate derivative of 4a was prepared and subsequently converted to the corresponding radical 7 in good overall yield (Scheme 2). The sulfate derivative 7 showed excellent solubility in water.
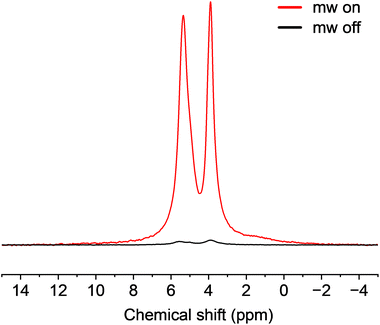
Based on its high molecular weight and good solubility in aqueous solutions, the BDPA-dendrimer radical (5d) was chosen for investigation of persistence and for evaluation as a polarizing agent. The persistence of 5d in DMSO and water was determined by monitoring the radical concentration as a function of time by UV-Vis spectroscopy (Fig. 2).39 Interestingly, the radical concentration 5d unexpectedly increased in DMSO during the first three days, before reaching a plateau (Fig. 2A). This indicated that the that the radical precursor 4d, which was still present in the sample of 5d, was converted to the radical under these conditions. DMSO plays a key role, facilitating formation of the BDPA anion (change in color), which then converts to the radical. The same behaviour was also observed when 4d was dissolved in DMSO, which ruled out possibility of oxidation of 5d by residual oxidizing agent from the previous step (Fig. S27). Once the anion had been converted to the radical, it remained persistent in DMSO for at least 25 days (Fig. 2A).39 The estimated half-life of 5d in water was ∼2 days, which was substantially longer that of BDPA-NMe3 (<24 h (Fig. 2B). Since the liquid DNP experiments were performed in glycerol (see below), the persistence of 5d in glycerol was also investigated (Fig. 2C); it showed a gradual degradation with an estimated half-life of ∼8 days. No detectable decrease in the DNP enhancement (see below) was observed during measurements at 315 K over a period of 4 h.
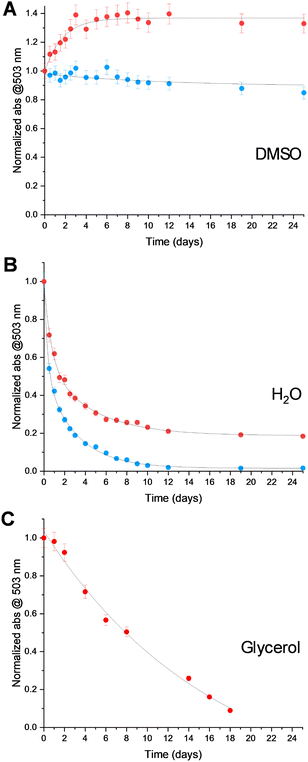 | ||
Fig. 2 Persistence of the trimethyl derivative of trialkylammonium BDPA (BDPA-NMe3) ( ) and BDPA-dendrimer 5d ( ) and BDPA-dendrimer 5d ( ) in DMSO (A) and H2O (B), as well as 5d in glycerol (C) at 23 °C, monitored by UV-Vis spectroscopy.39 The concentration of the radicals were 10 mM by weight, but the absolute radical concentration, determined by spin-counting was 7.5 mM and 5.6 mM for BDPA-NMe3 and BDPA-dendrimer 5d, respectively. ) in DMSO (A) and H2O (B), as well as 5d in glycerol (C) at 23 °C, monitored by UV-Vis spectroscopy.39 The concentration of the radicals were 10 mM by weight, but the absolute radical concentration, determined by spin-counting was 7.5 mM and 5.6 mM for BDPA-NMe3 and BDPA-dendrimer 5d, respectively. | ||
As mentioned above, DNP NMR in viscous liquids is emerging as a valuable approach for solution-state NMR studies at high magnetic fields and room temperature.35 When molecular tumbling of an analyte in solution is reduced, for example in viscous solvents,30,35 electron-nuclear dipolar interactions slow down, which allows efficient polarization transfer and sizable signal enhancements.36 Such polarization can also be observed for large molecules (with a long rotational correlation time) in aqueous solutions.29,37 Carbon-based radicals like BDPA-NMe3 and OX063 (Fig. 1) give enhancements through the SE pathway using this approach, correlating with molecular weight, with OX063 (MW = 1360.8) showing the highest DNP performance.30 Given the high molecular weight of BDPA-dendrimer 5d (MW = 4157.2), we investigated its DNP performance. At 9.4 T and 315 K, 5d produced a 1H DNP enhancement of 40 ± 5 at a concentration of 20 mM in glycerol (Fig. 3), a roughly twofold increase in DNP enhancement relative to OX063 (ε = 20 ± 3, 20 mM).30 DNP performance was also evaluated at radical concentrations of 10 and 40 mM for 5d, giving DNP enhancements of 18 ± 3 and 57 ± 6, respectively (Table S1). Although high viscosity leads to an increase in transverse relaxation rates and subsequent line broadening, using a viscous solvent is not a requirement for the DNP mechanism itself. Efficient polarization of small molecules, like ATP, can be achieved at viscosities only six times that of water, while larger biomolecules (∼15 kDa) possess sufficiently long rotational correlation times to exhibit sizable enhancements in pure aqueous solutions.37 Glycerol was chosen here to enable a direct and quantitative comparison with the trityl-based radical OX063.30
In summary, we have developed a versatile click-based approach to prepare tetrasubstituted BDPA radicals, giving access to BDPA derivatives with tuneable size, solubility and stability. Among the four derivatives, BDPA-dendrimer 5d stood out, showing improved persistence in both water and DMSO. Importantly, 5d exhibited the best DNP performance reported thus far for 1H DNP NMR in viscous liquids, with enhancements of 57 at 40 mM, making the BDPA-dendrimer a promising polarizing agent for liquid-state DNP NMR.
Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article are available at Zenodo.org at https://doi.org/10.5281/zenodo.17975646. The supporting data has been provided as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc01089j.Acknowledgements
We thank Dr S. Jonsdottir and Dr Gunnar W. Reginsson for assistance with collecting analytical data for structural characterization of new compounds and members of the Sigurdsson research group for helpful discussions. S. Th. S. acknowledges financial support from the Icelandic Research Fund (239662). I. M. A. acknowledges the Aðalsteinn Kristjánsson Memorial Fund for providing the doctoral research fellowship. A. K. acknowledges the financial support from the European Union (ERC, LiquidStateDNP, 101219713). Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or the European Research Council. Neither the European Union nor the granting authority can be held responsible for them.References
- A. G. M. Rankin, J. Trébosc, F. Pourpoint, J. P. Amoureux and O. Lafon, Solid State Nucl. Magn. Reson., 2019, 101, 116–143 CrossRef CAS PubMed.
- A. R. Camacho-Zarco, V. Schnapka, S. Guseva, A. Abyzov, W. Adamski, S. Milles, M. R. Jensen, L. Zidek, N. Salvi and M. Blackledge, Chem. Rev., 2022, 122, 9331–9356 CrossRef CAS PubMed.
- M. J. Zheng, Y. Y. Chu, Q. Wang, Y. X. Wang, J. Xu and F. Deng, Prog. Nucl. Magn. Reson. Spectrosc., 2024, 140, 1–41 CrossRef.
- X. Lu, W. Myint, C. S. Muli, M. Larion, H. Matsuo and K. J. Walters, J. Mol. Biol., 2025, 437, 169302 CrossRef CAS PubMed.
- A. S. Lilly Thankamony, J. J. Wittmann, M. Kaushik and B. Corzilius, Prog. Nucl. Magn. Reson. Spectrosc., 2017, 102–103, 120–195 CrossRef CAS.
- B. Corzilius, Annu. Rev. Phys. Chem., 2020, 71, 143–170 CrossRef CAS PubMed.
- T. Biedenbänder, V. Aladin, S. Saeidpour and B. Corzilius, Chem. Rev., 2022, 122, 9738–9794 CrossRef.
- M. Rosay, M. Blank and F. Engelke, J. Magn. Reson., 2016, 264, 88–98 CrossRef CAS PubMed.
- T. Maly and T. J. Keller, Eur. Phys. J. A, 2025, 61, 18 CrossRef CAS.
- C. Sauvée, M. Rosay, G. Casano, F. Aussenac, R. T. Weber, O. Ouari and P. Tordo, Angew. Chem., Int. Ed., 2013, 52, 10858–10861 CrossRef.
- A. Zagdoun, G. Casano, O. Ouari, M. Schwarzwälder, A. J. Rossini, F. Aussenac, M. Yulikov, G. Jeschke, C. Copéret, A. Lesage, P. Tordo and L. Emsley, J. Am. Chem. Soc., 2013, 135, 12790–12797 CrossRef CAS PubMed.
- F. Mentink-Vigier, I. Marin-Montesinos, A. P. Jagtap, T. Halbritter, J. van Tol, S. Hediger, D. Lee, S. T. Sigurdsson and G. De Paëpe, J. Am. Chem. Soc., 2018, 140, 11013–11019 CrossRef CAS PubMed.
- M.-A. Geiger, A. P. Jagtap, M. Kaushik, H. Sun, D. Stöppler, S. T. Sigurdsson, B. Corzilius and H. Oschkinat, Chem. – Eur. J., 2018, 24, 13485–13494 CrossRef CAS PubMed.
- A. Lund, G. Casano, G. Menzildjian, M. Kaushik, G. Stevanato, M. Yulikov, R. Jabbour, D. Wisser, M. Renom-Carrasco, C. Thieuleux, F. Bernada, H. Karoui, D. Siri, M. Rosay, I. V. Sergeyev, D. Gajan, M. Lelli, L. Emsley, O. Ouari and A. Lesage, Chem. Sci., 2020, 11, 2810–2818 RSC.
- T. Maly, G. T. Debelouchina, V. S. Bajaj, K. N. Hu, C. G. Joo, M. L. Mak-Jurkauskas, J. R. Sirigiri, P. C. van der Wel, J. Herzfeld, R. J. Temkin and R. G. Griffin, J. Chem. Phys., 2008, 128, 052211 CrossRef PubMed.
- G. Menzildjian, J. Schlagnitweit, G. Casano, O. Ouari, D. Gajan and A. Lesage, Chem. Sci., 2023, 14, 6120–6148 RSC.
- G. Mathies, M. A. Caporini, V. K. Michaelis, Y. Liu, K. N. Hu, D. Mance, J. L. Zweier, M. Rosay, M. Baldus and R. G. Griffin, Angew. Chem., Int. Ed., 2015, 54, 11770–11774 CrossRef CAS PubMed.
- F. Mentink-Vigier, G. Mathies, Y. Liu, A.-L. Barra, M. A. Caporini, D. Lee, S. Hediger, R. G. Griffin and G. De Paëpe, Chem. Sci., 2017, 8, 8150–8163 RSC.
- Y. Liu, F. A. Villamena, A. Rockenbauer, Y. Song and J. L. Zweier, J. Am. Chem. Soc., 2013, 135, 2350–2356 CrossRef CAS PubMed.
- K. Sato, R. Hirao, I. Timofeev, O. Krumkacheva, E. Zaytseva, O. Rogozhnikova, V. M. Tormyshev, D. Trukhin, E. Bagryanskaya, T. Gutmann, V. Klimavicius, G. Buntkowsky, K. Sugisaki, S. Nakazawa, H. Matsuoka, K. Toyota, D. Shiomi and T. Takui, J. Phys. Chem. A, 2019, 123, 7507–7517 CrossRef CAS PubMed.
- D. Wisser, G. Karthikeyan, A. Lund, G. Casano, H. Karoui, M. Yulikov, G. Menzildjian, A. C. Pinon, A. Purea, F. Engelke, S. R. Chaudhari, D. Kubicki, A. J. Rossini, I. B. Moroz, D. Gajan, C. Copéret, G. Jeschke, M. Lelli, L. Emsley, A. Lesage and O. Ouari, J. Am. Chem. Soc., 2018, 140, 13340–13349 CrossRef CAS PubMed.
- O. Haze, B. Corzilius, A. A. Smith, R. G. Griffin and T. M. Swager, J. Am. Chem. Soc., 2012, 134, 14287–14290 CrossRef CAS PubMed.
- T. J. Reddy, T. Iwama, H. J. Halpern and V. H. Rawal, J. Org. Chem., 2002, 67, 4635–4639 CrossRef CAS.
- E. L. Dane, T. Maly, G. T. Debelouchina, R. G. Griffin and T. M. Swager, Org. Lett., 2009, 11, 1871–1874 CrossRef CAS.
- L. Lumata, S. J. Ratnakar, A. Jindal, M. Merritt, A. Comment, C. Malloy, A. D. Sherry and Z. Kovacs, Chem. – Eur. J., 2011, 17, 10825–10827 CrossRef CAS PubMed.
- T. V. Can, M. A. Caporini, F. Mentink-Vigier, B. Corzilius, J. J. Walish, M. Rosay, W. E. Maas, M. Baldus, S. Vega, T. M. Swager and R. G. Griffin, J. Chem. Phys., 2014, 141, 064202 CrossRef CAS PubMed.
- S. R. Chaudhari, D. Wisser, A. C. Pinon, P. Berruyer, D. Gajan, P. Tordo, O. Ouari, C. Reiter, F. Engelke, C. Copéret, M. Lelli, A. Lesage and L. Emsley, J. Am. Chem. Soc., 2017, 139, 10609–10612 CrossRef CAS.
- A. Radaelli, H. A. I. Yoshihara, H. Nonaka, S. Sando, J. H. Ardenkjaer-Larsen, R. Gruetter and A. Capozzi, J. Phys. Chem. Lett., 2020, 11, 6873–6879 CrossRef CAS PubMed.
- A. A. Kuzhelev, D. Dai, V. Denysenkov and T. F. Prisner, J. Am. Chem. Soc., 2022, 144, 1164–1168 CrossRef CAS.
- A. A. Kuzhelev, V. Denysenkov, I. M. Ahmad, O. Y. Rogozhnikova, D. V. Trukhin, E. G. Bagryanskaya, V. M. Tormyshev, S. T. Sigurdsson and T. F. Prisner, J. Am. Chem. Soc., 2023, 145, 10268–10274 CrossRef CAS PubMed.
- M. Levien, L. M. Yang, A. van der Ham, M. Reinhard, M. John, A. Purea, J. Ganz, T. Marquardsen, I. Tkach, T. Orlando and M. Bennati, Nat. Commun., 2024, 15, 5904 CrossRef CAS PubMed.
- N. M. Loening, M. Rosay, V. Weis and R. G. Griffin, J. Am. Chem. Soc., 2002, 124, 8808–8809 CrossRef CAS PubMed.
- C. Griesinger, M. Bennati, H. M. Vieth, C. Luchinat, G. Parigi, P. Höfer, F. Engelke, S. J. Glaser, V. Denysenkov and T. F. Prisner, Prog. Nucl. Magn. Reson. Spectrosc., 2012, 64, 4–28 CrossRef CAS.
- S. Pylaeva, P. Marx, G. Singh, T. D. Kuhne, M. Roemelt and H. Elgabarty, J. Phys. Chem. A, 2021, 125, 867–874 CrossRef CAS.
- D. H. Dai, V. Denysenkov, E. G. Bagryanskaya, V. M. Tormyshev, T. F. Prisner and A. A. Kuzhelev, J. Phys. Chem. Lett., 2023, 14, 7059–7064 CrossRef CAS.
- D. Sezer, Magn. Reson., 2023, 4, 153–174 CrossRef CAS PubMed.
- A. Kuzhelev, Anal. Chem., 2025, 97, 14890–14893 CrossRef CAS PubMed.
- S. Mandal and S. T. Sigurdsson, Chem. – Eur. J., 2020, 26, 7486–7491 CrossRef CAS PubMed.
- S. Mandal and S. T. Sigurdsson, Chem. Commun., 2020, 56, 13121–13124 RSC.
- T. Singh and A. Kumar, J. Phys. Chem. B, 2007, 111, 7843–7851 CrossRef CAS PubMed.
- R. Atkin and G. G. Warr, J. Phys. Chem. B, 2008, 112, 4164–4166 CrossRef CAS.
- R. Dutta, S. Kundu and N. Sarkar, Biophys. Rev., 2018, 10, 861–871 CrossRef CAS PubMed.
- Y. P. Liu, F. A. Villamena and J. L. Zweier, Chem. Commun., 2008, 4336–4338, 10.1039/b807406b.
- W. Liu, J. Nie, X. Tan, H. Liu, N. Yu, G. Han, Y. Zhu, F. A. Villamena, Y. Song, J. L. Zweier and Y. Liu, J. Org. Chem., 2017, 82, 588–596 CrossRef CAS.
- A. R. Birge, M. J. Piper, K. J. Painter and G. T. Sazama, Results Chem., 2022, 4, 100393 CrossRef CAS.
- H. Hamamoto, D. Shimizu and K. Matsuda, Chem. – Eur. J., 2024, 30, e202401353 CrossRef CAS PubMed.
- E. H. Ryu and Y. Zhao, Org. Lett., 2005, 7, 1035–1037 CrossRef CAS PubMed.
- A. L. M. Morotti, K. L. Lang, I. Carvalho, E. P. Schenkel and L. S. C. Bernardes, Tetrahedron Lett., 2015, 56, 303–307 CrossRef CAS.
- J. A. Alshehri and A. M. Jones, Essays Biochem., 2024, 68, 449–466 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |