Open Access Article
Open Access ArticleThiosulfonate synthesis via halothiolation of aryne intermediates followed by oxidative S-sulfonylation
Yusei Yamamoto,
Shinya Tabata,
Koyo Numata and
Suguru Yoshida *
*
Department of Biological Science and Technology, Faculty of Advanced Engineering, Tokyo University of Science, 6-3-1 Niijuku, Katsushika-ku, Tokyo 125-8585, Japan. E-mail: s-yoshida@rs.tus.ac.jp
First published on 20th April 2026
Abstract
An efficient method for preparing aryl thiosulfonates through aryne intermediates is disclosed. Following aryne halothiolation with potassium xanthate, the resulting aryl xanthates were transformed into thiosulfonates using iodine and sodium sulfinates. The versatility of thiosulfonates together with divergent aryne precursors enabled the access to a wide range of highly functionalized organosulfur compounds.
Thiosulfonates are promising bench-stable sulfur surrogates owing to their good electrophilicity, which enables the synthesis of a wide range of sulfides in an odorless manner (Fig. 1A).1,2 For example, carbanions readily react with thiosulfonates to afford various sulfides with liberation of water-soluble sulfinate anions.2a,2b,2l We have developed efficient methods for the preparation of sulfides from thiosulfonates and aryl- and alkenylborons catalyzed by copper2d,2g,2k or rhodium.2e In addition, alkynyl sulfides were also synthesized from thiosulfonates and terminal alkynes with copper catalysis.2i
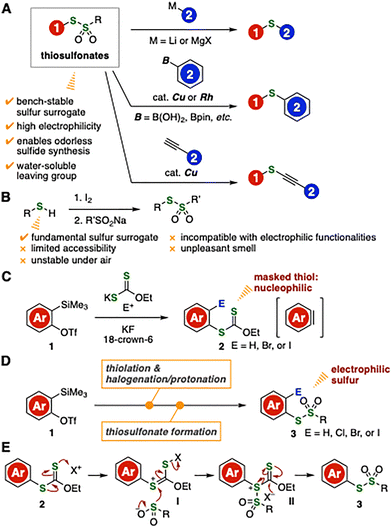 | ||
| Fig. 1 (A) Transformations of thiosulfonates. (B) Synthesis of thiosulfonates from thiols. (C) Our previous study. (D) This work. (E) Working hypothesis. | ||
In view of the versatility of thiosulfonates, their limited availability should be addressed to improve access to diverse organosulfur compounds.3 In particular, conventional thiol-based syntheses are limited in the scope of accessible thiosulfonates owing to the instability of thiols under air, their incompatibility with electrophilic functionalities, and their unpleasant odor (Fig. 1B). We herein disclose an efficient method for the synthesis of a wide variety of aromatic thiosulfonates from o-silylaryl triflates via halothiolation of aryne intermediates followed by oxidative S-sulfonylation.
Our recent studies on organosulfur chemistry involving transformations of aryne intermediates have enabled the efficient synthesis of aryl xanthates bearing a masked thiol functionality from o-silylaryl triflates, potassium xanthates, and electrophiles such as pentafluorophenyl bromide (Fig. 1C).4,5 Building on this novel transformation, we envisioned a stepwise thiosulfonate synthesis from o-silylaryl triflates involving aryne halothiolation followed by oxidative S-sulfonylation of the resulting aryl xanthates, thereby enabling umpolung of the masked thiol functionality to an electrophilic sulfur surrogate (Fig. 1D). In light of our previous study on the direct oxidative transformation of thioesters into sulfinate esters, we hypothesized that oxidation of aryl xanthates followed by addition of metal sulfinates would promote thiosulfonate formation owing to the electron-rich nature of the xanthate moiety (Fig. 1E).6 Compared with the previous hydrolysis–oxidation approach,4 the present strategy offers a significant advantage by avoiding thiols, which often exhibit poor functional-group compatibility due to their high nucleophilicity.
We first examined the oxidation of aryl xanthate 2a with an equimolar amount of iodine in dichloromethane at room temperature, followed by the addition of sodium sulfinate (Table 1, entry 1). Under these conditions, 3a was obtained in moderate yield. Subsequent optimization of the reaction conditions enabled more efficient thiosulfonate formation. Increasing the amount of iodine slightly improved the yield of 3a (entry 2). The use of other oxidants including N-iodo-, N-bromo-, and N-chlorosuccinimide decreased the yields of 3a (entries 3–5). While thiosulfonate synthesis in ethanol or THF resulted in lower efficiencies (entries 6 and 7), we succeeded in the preparation of 3a in good yield when using acetonitrile (entry 8). Finally, treatment of 2a with iodine in acetonitrile at 80 °C followed by the addition of sodium p-toluenesulfinate at room temperature provided thiosulfonate 3a in high yield (entry 9).
With the optimized conditions in hand, we synthesized a wide variety of thiosulfonates 3 from aryl xanthates 2 prepared by our previously reported aryne chemistry (Fig. 2). For example, 3b was obtained from O-ethyl S-phenyl xanthate (2b) in good yield. In the case of electron-deficient 3,4-difluorophenyl-substituted xanthate 2c, we achieved the synthesis of 3c in moderate yield. Also, oxidation–S-sulfonylation of electron-rich aromatic xanthates also proceeded smoothly, affording 3d–3f in high yields. Notably, we accomplished the synthesis of 3g in good yield without oxidation of the 4-methoxyphenylthio group. This result demonstrates that hydrothiolation of 3-sulfanylbenzyne followed by oxidative conversion enabled the selective synthesis of 3-sulfanylphenyl-substituted thiosulfonates.7 Unfortunately, attempts to synthesize 3h from O-ethyl S-(3-morpholinophenyl) xanthate, prepared from 3-morpholinobenzyne precursor,8 were unsuccessful, and cyclized product 4 was obtained instead. It is also worth noting that the oxidation–S-sulfonylation took place efficiently with o-bromo-substituted aryl xanthates without affecting the ortho substituents, to afford thiosulfonates 5b, 5c, and 5f.
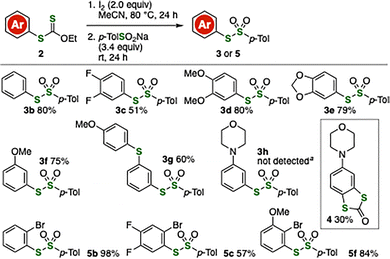 | ||
Fig. 2 Synthesis of various thiosulfonates from aryl xanthates 2. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I2 (4.0 equiv) was employed and 2nd step was conducted at 80 °C. See the SI for details. I2 (4.0 equiv) was employed and 2nd step was conducted at 80 °C. See the SI for details. | ||
Then, efficient preparation of S-aryl thiosulfonates 3, 5, 9, and 10 was achieved from o-silylaryl triflates 1 as aryne precursors (Fig. 3). For instance, a mixture of thiosulfonates 3i and 3i′ (= 3f) was synthesized by hydrothiolation of 4-methoxybenzyne (step A) followed by oxidation–S-sulfonylation (step B).9 Of note, the synthesis of thiosulfonate 3j bearing an ester moiety was realized by hydrothiolation of the corresponding aryne intermediate followed by oxidative S-sulfonylation. Because ester functionalities are often susceptible to hydrolysis under basic conditions, this result supports the advantage of the present thiol-free protocol for the preparation of thiosulfonates bearing electrophilic functional groups such as esters. We accomplished the synthesis of 5a through bromothiolation of 3,4-dimethylbenzyne followed by oxidation–S-sulfonylation developed in this study. Furthermore, chlorothiolation of aryne intermediates was achieved using tetrachloromethane as an electrophilic chlorine source. Subsequent conversion of the resulting xanthates afforded 9a and 9b in good yields. Preparation of 10a, 10b, and 10d–10g was accomplished by iodothiolation of arynes using 1-iodo-2-phenylacetylene, followed by oxidation with iodine and subsequent addition of sodium sulfinate. These results clearly demonstrate that a wide range of o-halogen-substituted aryl thiosulfonates can be synthesized through aryne intermediates, benefiting from the recent great achievements in readily accessible aryne precursors.10,11
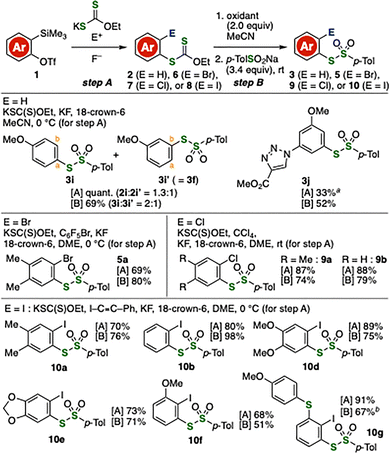 | ||
Fig. 3 Synthesis of various thiosulfonates from o-silylaryl triflates 1. DME = 1,2-dimethoxyethane. See the SI for details. a 1H NMR yield. b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Step B was performed at 80 °C. Step B was performed at 80 °C. | ||
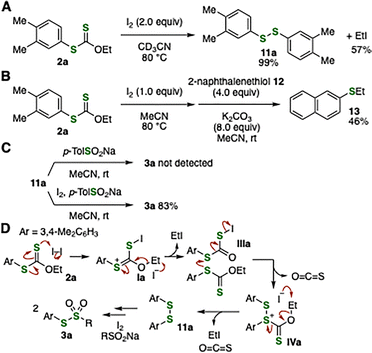
To clarify the reaction mechanism of thiosulfonate formation, we conducted a series of control experiments (Fig. 4A–D). Treatment of aryl xanthate 2a with iodine in acetonitrile-d3 at 80 °C furnished disulfide 11a and iodoethane, as identified by 1H and 13C NMR analysis of the crude reaction mixture (Fig. 4A). The formation of iodoethane was further supported by an experiment in which 2-naphthalenethiol (12) was added, affording sulfide 13 (Fig. 4B). While 3a was not observed when sodium p-toluenesulfinate was added to disulfide 11a in acetonitrile (Fig. 4C, upper), treatment of 11a with sodium sulfinate in the presence of iodine afforded 3a in high yield (Fig. 4C, lower), consistent with the report by Fujiki and coworkers.3b These control experiments indicate that residual iodine plays a crucial role in the S-sulfonylation.
Control experiments shown in Fig. 4A–C support the following plausible mechanism for thiosulfonate formation from 2a (Fig. 4D). First, oxidation of 2a with iodine generates disulfide 11a, presumably via Ia, which undergoes deethylation promoted by iodide. This novel oxidation of xanthates realized an umpolung transformation of masked thiols into electrophilic sulfur surrogates. In the synthesis of 3h, which bears a strong electron-donating morpholino group, competing intramolecular electrophilic aromatic substitution of Ia may result in the formation of side-product 4. Second, thiosulfonate formation proceeded from 11a with sodium p-toluenesulfinate and an additional equivalent of iodine, according to the previous report by Fujiki.3b Thus, we have established not only a practical and efficient method for the synthesis of thiosulfonates from aryne precursors, but also a novel iodine-mediated oxidative transformation of aryl xanthates 2. In contrast to previous oxidative transformations of xanthates leading to S-oxidation or thiocarbonate formation,12 the present finding enables thiosulfonate synthesis by S-sulfonylation of disulfides.
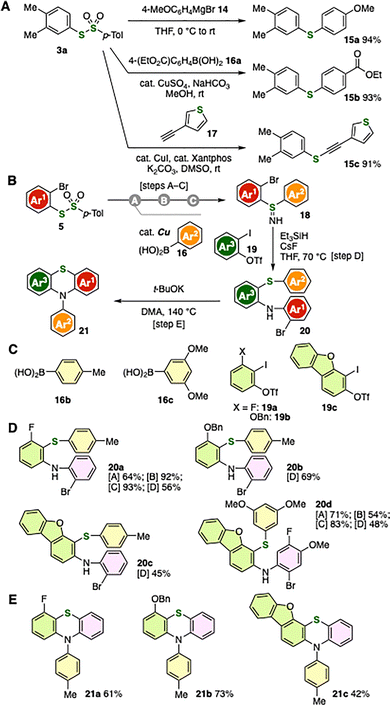
The good versatility of thiosulfonates was demonstrated by odorless sulfide synthesis (Fig. 5A). Treatment of 3a with 4-methoxyphenylmagnesium bromide provided 15a in high yield (Fig. 5A, upper).2b Preparation of 15b bearing an ester moiety was achieved by copper-catalyzed S-arylation with 16a under mild conditions (Fig. 5A, middle), following our recently developed protocol.2d In addition, alkynyl sulfide 15c was efficiently synthesized from 3a and terminal alkyne 17 in the presence of a catalytic amount of copper iodide and Xantphos (Fig. 5A, lower).2i These results clearly demonstrate that a broad range of sulfides can be accessed from o-silylaryl triflates via thiolation of arynes, oxidative conversion to thiosulfonates, and subsequent S-arylation with diverse reaction partners, including organomagnesium reagents, organoborons, and terminal alkynes.
Thiosulfonate synthesis via aryne halothiolation followed by oxidation–S-sulfonylation enabled access to highly functionalized o-arylamino-substituted diaryl sulfides 20 in combination with our aryne-based organosulfur chemistry (Fig. 5B–E). Specifically, we accomplished the preparation of 18 through copper-catalyzed S-arylation of 5 followed by S-imidation and subsequent hydrolysis. Sulfilimines then served as aminothiolation reagents for aryne intermediates generated from 19 under carbanion-free conditions using triethylsilane and cesium fluoride, affording sulfides 20a–20d while retaining fluoro, bromo, methyl, benzyloxy, methoxy, and dibenzofuran moieties (Fig. 5B and D).13,14 For example, following the synthesis of S-(2-bromophenyl)-S-(4-tolyl)sulfilimine (18a) from 5b and arylboronic acid 16b, we succeeded in preparing 21a–21c by migrative aminothiolation of arynes generated from 19a–19c in moderate to good yields. It is worth noting that sulfide 20d possessing fluoro, bromo, and methoxy substituents was synthesized via bromothiolation of 4,5-difluorobenzyne from 4,5-difluoro-2-silylphenyl triflate, where methoxy group was selectively introduced during the hydrolysis in sulfilimine synthesis under basic conditions.15 Moreover, we achieved the synthesis of 21a–21c by t-BuOK-facilitated migrative N-arylation from arylthio group followed by ring-closing S-arylation through C–Br cleavage (Fig. 5B and E).16 Notably, a pentacyclic scaffold was constructed in 21c from 19c through aryne aminothiolation, t-BuOK-promoted migrative N-arylation, and subsequent ring closure.
In conclusion, we have established an efficient method for synthesizing aryl thiosulfonates from o-silylaryl triflates. A key finding is an umpolung transformation in which aryl xanthates are readily oxidized with iodine to afford the corresponding thiosulfonates via disulfide intermediates. The versatility of thiosulfonates, together with our divergent aryne-based toolbox, enables access to highly functionalized sulfides involving phenothiazine derivatives. Further studies in our group are underway, including detailed mechanistic investigations supported by theoretical calculations and applications to the synthesis of bioactive organosulfur compounds.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, characterization for new compounds including NMR spectra. See DOI: https://doi.org/10.1039/d6cc01087c.Acknowledgements
The authors thank Central Glass Co., Ltd. for providing Tf2O. This work was supported by JSPS KAKENHI Grant Number JP22H02086 (S. Y.); and Tokuyama Science Foundation (S. Y.).References
- For reviews of thiosulfonate chemistry, see: (a) P. Mampuys, C. R. McElroy, J. H. Clark, R. V. A. Orru and B. U. W. Maes, Adv. Synth. Catal., 2020, 362, 3 CrossRef CAS; (b) S. Yu, Z. Chen, Q. Chen, S. Lin, J. He, G. Tao and Z. Wang, Chin. J. Org. Chem., 2022, 42, 2322 CrossRef CAS; (c) D. Ge, J.-W. Chen, P. Xu, J. Pan and X.-Q. Chu, Chin. Chem. Lett., 2022, 33, 4732 CrossRef CAS.
- (a) M. Balasubramanian and V. Baliah, J. Chem. Soc., 1954, 1844 RSC; (b) S. H. Wunderlich and P. Knochel, Angew. Chem., Int. Ed., 2007, 46, 7685 CrossRef CAS PubMed; (c) P. Saravanan and P. Anbarasan, Org. Lett., 2014, 16, 848 CrossRef CAS PubMed; (d) S. Yoshida, Y. Sugimura, Y. Hazama, Y. Nishiyama, T. Yano, S. Shimizu and T. Hosoya, Chem. Commun., 2015, 51, 16613 RSC; (e) K. Kanemoto, Y. Sugimura, S. Shimizu, S. Yoshida and T. Hosoya, Chem. Commun., 2017, 53, 10640 RSC; (f) Y. Fang, T. Rogge, L. Ackermann, S.-Y. Wang and S.-J. Ji, Nat. Commun., 2018, 9, 2240 CrossRef PubMed; (g) K. Kanemoto, S. Yoshida and T. Hosoya, Chem. Lett., 2018, 47, 85 CrossRef CAS; (h) S. Huang, N. Thirupathi, C.-H. Tung and Z. Xu, J. Org. Chem., 2018, 83, 9449 CrossRef CAS PubMed; (i) K. Kanemoto, S. Yoshida and T. Hosoya, Org. Lett., 2019, 21, 3172 CrossRef CAS PubMed; (j) K. Kanemoto, Y. Sakata, T. Hosoya and S. Yoshida, Chem. Lett., 2020, 49, 593 CrossRef CAS; (k) K. Mutsuura, Y. Sakata, K. Uchida, T. Hosoya and S. Yoshida, Chem. Lett., 2020, 49, 753 CrossRef; (l) A. Kobayashi, T. Matsuzawa, T. Hosoya and S. Yoshida, Chem. Lett., 2021, 50, 1624 CrossRef CAS; (m) K. Gadde, P. Mampuys, A. Guidetti, H. Y. V. Ching, W. A. Herrebout, S. Van Doorslaer, K. A. Tehrani and B. U. W. Maes, ACS Catal., 2020, 10, 8765 CrossRef CAS; (n) J. Luo, M. Lin, D. Xia, G. Du, Z. Cai, B. Dai and L. He, Org. Chem. Front., 2023, 10, 1224 RSC; (o) J. Mindner, S. Rombach and D. B. Werz, Org. Lett., 2024, 26, 2124 CrossRef CAS PubMed.
- (a) F. Freeman and C. N. Angeletakis, J. Org. Chem., 1985, 50, 793 CrossRef CAS; (b) K. Fujiki, N. Tanifuji, Y. Sasaki and T. Yokoyama, Synthesis, 2002, 343 CrossRef CAS; (c) K. Bahrami, M. M. Khodaei and D. Khaledian, Tetrahedron Lett., 2012, 53, 354 CrossRef CAS; (d) H. T. Pham, N.-L. T. Nguyen, F. Duus and T. X. T. Luu, Phosphorus, Sulfur Silicon Relat. Elem., 2015, 190, 1934 CrossRef CAS; (e) P. Natarajan, Tetrahedron Lett., 2015, 56, 4131 CrossRef CAS; (f) Y. Zheng, F.-L. Qing, Y. Huang and X.-H. Xu, Adv. Synth. Catal., 2016, 358, 3477 CrossRef CAS; (g) L. Liu, B. Luo and C. Wang, Eur. J. Org. Chem., 2021, 5880 CrossRef CAS; (h) J. Strehl and G. Hilt, Eur. J. Org. Chem., 2022, 35 Search PubMed; (i) L. Sancineto, J. K. Kibambe, C. Scimmi and C. Santi, ARKIVOC, 2023, 135 Search PubMed.
- S. Tabata and S. Yoshida, Org. Lett., 2024, 26, 3816 CrossRef CAS PubMed.
- For selected papers of functionalization of arynes with sulfur functional groups, see: (a) M. Pawliczek, L. K. B. Garve and D. B. Werz, Chem. Commun., 2015, 51, 9165 RSC; (b) H.-Y. Li, L.-J. Xing, M.-M. Lou, H. Wang, R.-H. Liu and B. Wang, Org. Lett., 2015, 17(5), 1098 CrossRef CAS PubMed; (c) X. Wu, M. Chen, F.-S. He and J. Wu, Org. Lett., 2023, 25, 5157 CrossRef CAS PubMed; (d) Y. Guo, Z. Zhuang, X. Feng, Q. Ma, N. Li, C. Jin, H. Yoshida and J. Tan, Org. Lett., 2023, 25, 7192 CrossRef CAS PubMed.
- K. Nakamura, Y. Kumagai, A. Kobayashi and S. Yoshida, Org. Biomol. Chem., 2023, 21, 6886 RSC.
- Y. Nakamura, Y. Miyata, K. Uchida, S. Yoshida and T. Hosoya, Org. Lett., 2019, 21, 5252 CrossRef CAS PubMed.
- (a) S. Yoshida, Y. Nakamura, K. Uchida, Y. Hazama and T. Hosoya, Org. Lett., 2016, 18, 6212 CrossRef CAS PubMed; (b) Y. Nakamura, S. Ozawa, S. Yoshida and T. Hosoya, Chem. Lett., 2019, 48, 1296 CrossRef CAS.
- We attempted a one-pot synthesis of thiosulfonate 3d from 4,5-di(methoxy)-2-(trimethylsilyl)phenyl triflate by aryne hydrothiolation with potassium xanthate in acetonitrile followed by oxidative S-sulfonylation. However, the desired product 3d was not obtained under these conditions.
- Modern Aryne Chemistry, ed. A. T. Biju, Wiley-VCH, Weinheim, 2021 Search PubMed.
- For selected reviews of aryne chemistry, see: (a) P. M. Tadross and B. M. Stoltz, Chem. Rev., 2012, 112, 3550 CrossRef CAS PubMed; (b) A. E. Goetz, T. K. Shah and N. K. Garg, Chem. Commun., 2015, 51, 34 RSC; (c) J.-A. García-López and M. F. Greaney, Chem. Soc. Rev., 2016, 45, 6766 RSC; (d) F. I. M. Idiris and C. R. Jones, Org. Biomol. Chem., 2017, 15, 9044 RSC; (e) H. Takikawa, A. Nishii, T. Sakai and K. Suzuki, Chem. Soc. Rev., 2018, 47, 8030 RSC; (f) T. Roy and A. T. Biju, Chem. Commun., 2018, 54, 2580 RSC; (g) D. B. Werz and A. T. Biju, Angew. Chem., Int. Ed., 2020, 59, 3385 CrossRef CAS PubMed; (h) J. Shi, L. Li and Y. Li, Chem. Rev., 2021, 121, 3892 CrossRef CAS PubMed; (i) H. Hazarika and P. Gogoi, Org. Biomol. Chem., 2021, 19, 8466 RSC; (j) R. Zhang, X. Peng and J. Tan, Synthesis, 2022, 5064 CAS; (k) N. Kim, M. Choi, S.-E. Suh and D. M. Chenoweth, Chem. Rev., 2024, 124, 11435 CrossRef CAS PubMed; (l) S. Tabata and S. Yoshida, Tetrahedron Chem., 2025, 14, 100136 CrossRef CAS.
- (a) B. G. Cox and R. Natarajan, J. Chem. Soc., Perkin Trans. 2, 1977, 113 RSC; (b) E. Silvester, D. Truccolo and F. P. Hao, J. Chem. Soc., Perkin Trans. 2, 2002, 1562 RSC.
- (a) S. Yoshida, T. Yano, Y. Misawa, Y. Sugimura, K. Igawa, S. Shimizu, K. Tomooka and T. Hosoya, J. Am. Chem. Soc., 2015, 137, 14071 CrossRef CAS PubMed; (b) S. Yoshida, T. Kuribara, T. Morita, T. Matsuzawa, K. Morimoto, T. Kobayashi and T. Hosoya, RSC Adv., 2018, 8, 21754 RSC; (c) T. Matsuzawa, K. Uchida, S. Yoshida and T. Hosoya, Chem. Lett., 2018, 47, 825 CrossRef CAS.
- M. Minoshima, K. Uchida, Y. Nakamura, T. Hosoya and S. Yoshida, Org. Lett., 2021, 23, 1868 CrossRef CAS PubMed.
- J. R. Rodriguez, J. Agejas and A. B. Bueno, Tetrahedron Lett., 2006, 47, 5661 CrossRef CAS.
- T. Matsuzawa, T. Hosoya and S. Yoshida, Org. Lett., 2021, 23, 2347 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |