Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
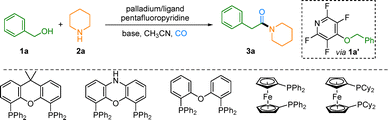
Direct carbonylative amidation of benzylic alcohols with alkylamines via palladium catalyzed C–O bond activation
Zhiping Yin *a,
Heng Lia,
Xiaowen Qina,
Mengyang Liua,
Chang-Sheng Wang
*a,
Heng Lia,
Xiaowen Qina,
Mengyang Liua,
Chang-Sheng Wang b and
Xiao-Feng Wu
b and
Xiao-Feng Wu *c
*c
aSchool of Pharmacy, Jiangsu University, Zhenjiang 212013, China. E-mail: zhiping_yin@ujs.edu.cn
bCollege of Biotechnology and Pharmaceutical Engineering, Nanjing Tech University, 30 Puzhu Road S., Nanjing 211816, PR China
cDalian National Laboratory for Clean Energy, Dalian Institute of Chemical Physics, Chinese Academy of Science, Dalian 116023, Liaoning, China. E-mail: xwu2020@dicp.ac.cn
First published on 17th March 2026
Abstract
A palladium-catalyzed direct carbonylative amidation that enables the efficient coupling of benzylic alcohols with alkylamines under mild conditions has been developed. Key to the success is the use of pentafluoropyridine as an activating group, which facilitates selective alcohol activation and suppresses undesired side reactions.
Amide bond formation stands as a cornerstone – and a persistent challenge – in synthetic chemistry, particularly in the context of modern atom- and step-economical synthesis.1 This is especially relevant for medicinally privileged structures, such as the benzylamide motifs found in widely prescribed Z-drugs like zolpidem and alpidem, where efficient and modular access to this scaffold is highly desirable.2 While classical condensation methods relying on pre-activated carboxylic derivatives remain indispensable in both academic and industrial settings, their dependence on stoichiometric coupling agents and the concomitant generation of chemical waste imposes clear limitations in terms of sustainability and operational simplicity. Following the pioneering work on transition-metal-catalyzed carbonylative coupling by R. F. Heck, catalytic carbonylation has emerged as a powerful synthetic platform.3 Among its most notable advances is the three-component coupling of aryl (pseudo)halides, carbon monoxide, and amines, which offers an efficient and direct route to amide bonds.4 Despite substantial progress, these methods universally require pre-functionalized electrophilic partners, which introduces additional synthetic steps and constrains the modularity of the approach.
In contrast, alcohols are traditionally employed as nucleophiles or solvents. Realizing the direct carbonylative amidation of alcohols with amines via catalytic CO insertion would represent a paradigm shift, transitioning from the conventional logic of pre-functionalization and coupling to a strategy of direct assembly from simple and abundant feedstocks. This transformation demands both activation of the robust C–O bond and a formal polarity inversion at the alcohol's α-carbon to generate an electrophilic acyl equivalent.5 Achieving such an inversion with high selectivity under catalytic control is essential to suppress competing pathways, particularly the challenge of differentiating two inherently nucleophilic species within a single catalytic manifold. Our group previously demonstrated that nitroarenes could serve as a nitrogen source in the conversion of alcohols to amides, albeit through a distinct redox-neutral C–N coupling pathway limited to aromatic amines.6 Despite their ready availability, the direct use of alcohols and aliphatic amines in carbonylative couplings has been hindered by the difficulty of the reduction of alkyl nitro compounds. While transition-metal catalyzed carbonylations of benzyl alcohols to phenylacetic acid derivatives have been known for decades (typically proceeding through halide intermediates),7 Wu's pioneering work showed that dimethyl carbonate could act as a benign activator, avoiding halides entirely.8 However, the strategic merger of alcohol C–O activation with aliphatic amines for direct amide synthesis remains an open frontier – one that offers considerable potential for more sustainable and efficient synthetic platforms.
Building on our group's sustained interest in nitrogen-heterocycle synthesis,9 we herein report a catalytic system that directly addresses this challenge: the deoxygenative carbonylation of benzylic alcohols with aliphatic amines. Using a tailored palladium catalyst, we achieve selective activation of the robust benzylic C–O bond, controlled CO insertion, and efficient interception by the amine nucleophile. This strategy effectively reverses the inherent polarity of the alcohol, enabling its use as a latent electrophile in a direct, one-pot coupling to amides. The method operates under mild conditions and provides a modular and step-economical route to a range of functionally diverse benzylic amides.
Our investigation began with benzyl alcohol (1a), pentafluoropyridine, and piperidine (2a) as model substrates in acetonitrile under a carbon monoxide atmosphere (5 bar), with Pd(OAc)2/XantPhos as the catalytic system. Recognizing the crucial role of a base in neutralizing the HF byproduct generated during the formation of the key intermediate 4-(benzyloxy)-2,3,5,6-tetrafluoropyridine (1a′) from 1a and pentafluoropyridine, we systematically evaluated a variety of bases (Table 1, entries 1–5). The results indicated that KOH afforded the optimal yield (37%), whereas other tested bases (e.g., NaH, t-BuOK, Et3N, etc.) provided only trace amounts of product or no reaction. Consequently, KOH was established as the most effective base for this transformation and served as the benchmark condition for subsequent optimization. A preliminary investigation into the loading of KOH revealed that decreasing its amount to 1.5 equivalents led to a decreased yield of 28% (Table 1, entry 6). We then optimized the stoichiometry of the three coupling partners while keeping other conditions fixed. A molar ratio of 1a:pentafluoropyridine:2a = 1.0:1.5:2.5 provided the best result (56% yield, entry 7), whereas a reversed ratio (2.0:1.5:1.0) significantly reduced the yield to 21% (entry 8), highlighting the importance of a balanced stoichiometry. Subsequently, a systematic screening of ligands within the palladium catalytic system was conducted. Under otherwise fixed conditions, the influence of various phosphine ligands, including XantPhos and DPEPhos, was examined, and the properties of the ligand were proven crucial (Table 1, entries 9–14). Among them, DCyPFc (1,1′-bis(dicyclohexylphosphino)ferrocene) delivered the best catalytic performance, affording the product in 86% yield (Table 1, entry 13). Notably, the stability of the ligand was not influenced in the presence of pentafluoropyridine, which was confirmed by 31P NMR. With DCyPFc identified as the optimal ligand, we further evaluated the activity of different palladium catalyst precursors. PdCl2 demonstrated the highest catalytic efficiency, elevating the yield to 91% (Table 1, entry 17). The target product 3a was ultimately obtained in an isolated yield of 78%. Finally, solvent screening confirmed acetonitrile as optimal; alternative solvents such as toluene, DMF, and DCE were less effective (see the SI, Section 3.5). These optimized conditions (PdCl2/DCyPFc, KOH, CH3CN, 5 bar CO) were used for all subsequent substrate-scope investigations.
| Entry | Catalyst/ligand | Bite angle | Base | Yieldb (%) |
|---|---|---|---|---|
a Reaction conditions: Step 1: 1a (0.1 mmol, 1.0 equivalent, 10 µL), pentafluoropyridine (0.2 mmol, 2.0 equivalent, 22 µL), base 2.5 equivalent, CH3CN (1 mL), N2, r.t. and 5 h; Step 2: 2a (0.2 mmol, 2.0 equivalent, 20 µL), palladium catalysts (5 mol%), ligand (10 mol%), CO (5 bar), 110 °C and 20 h. N.R. = no reaction.b GC yields of mixture 3a were determined by using dodecane as the internal standard.c KOH (8.4 mg, 0.15 mmol, 1.5 equivalent).d 1.0 equivalent = 0.1 mmol, 1a: pentafluoropyridine: 2a = 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 eq.e 1.0 equivalent = 0.1 mmol, 1a: pentafluoropyridine: 2a = 2.0 2.5 eq.e 1.0 equivalent = 0.1 mmol, 1a: pentafluoropyridine: 2a = 2.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 eq.f Isolated yield is given in parentheses. TFP = Tri-2-furanylphosphine. 1.0 eq.f Isolated yield is given in parentheses. TFP = Tri-2-furanylphosphine. |
||||
| 1 | Pd(OAc)2/XantPhos | 108.11 | KOH | 37 |
| 2 | Pd(OAc)2/XantPhos | 108.11 | NaOH | 26 |
| 3 | Pd(OAc)2/XantPhos | 108.11 | NaH | Trace |
| 4 | Pd(OAc)2/XantPhos | 108.11 | t-BuOK | 8 |
| 5 | Pd(OAc)2/XantPhos | 108.11 | Et3N | Trace |
| 6c | Pd(OAc)2/XantPhos | 108.11 | KOH | 28 |
| 7d | Pd(OAc)2/XantPhos | 108.11 | KOH | 56 |
| 8e | Pd(OAc)2/XantPhos | 108.11 | KOH | 21 |
| 9d | Pd(OAc)2/DPEPhos | 103.93 | KOH | 63 |
| 10d | Pd(OAc)2/N-Xantphos | 114.2 | KOH | 41 |
| 11d | Pd(OAc)2/PPh3 | — | KOH | N.R. |
| 12d | Pd(OAc)2/TFP | — | KOH | Trace |
| 13d | Pd(OAc)2/DCyPFc | 99.18 | KOH | 86 |
| 14d | Pd(OAc)2/DPPF | 99.18 | KOH | 79 |
| 15d | Pd(PPh3)4/DCyPFc | 99.18 | KOH | 86 |
| 16d | Pd(PhCN)2Cl2/DCyPFc | 99.18 | KOH | 71 |
| 17d | PdCl2/DCyPFc | 99.18 | KOH | 91 (78f) |
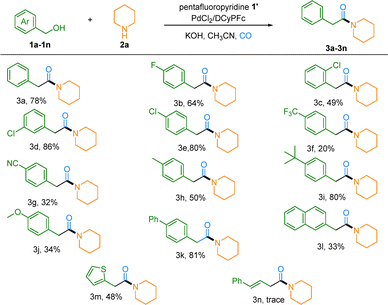
Under the optimized conditions, the substrate scope of this carbonylative amidation was systematically examined. As illustrated in Table 2, the reaction exhibited broad compatibility with a wide range of benzylic alcohols. Electron-deficient substrates bearing halogen (F and Cl), trifluoromethyl, or cyano groups reacted effectively, affording the corresponding amides in 20–86% yield, though the trifluoromethyl- and cyano-substituted derivatives exhibited moderate reactivity (Table 2, 3f and 3g). The decreased yield was due to the decreased stability of the corresponding palladium intermediate. Electron-rich benzylic alcohols with methyl, tert-butyl, methoxy, or phenyl substituents were also well tolerated, delivering products in 34–81% yield (Table 2, 3h–3k). The method further extended to other aromatic systems such as naphthalene and thiophene, which provided moderate yields (33–48%, 3l and 3m). It must be noted that trace or no target product was observed for (E)-3-phenylprop-2-en-1-ol and pyridin-3-ylmethanol (3n), likely due to the inherent strength of their C–O bonds and the potential coordination of the pyridine moiety under the reaction conditions (see the SI, Section 2.3, for failed examples, including aliphatic alcohols).
| a Reaction conditions: Step 1: 1a-1n (0.2 mmol, 1.0 equivalent), pentafluoropyridine (0.3mmol, 1.5 equivalent, 33 µL), KOH (0.5 mmol, 2.5 equivalent, 28 mg), CH3CN (2 mL), N2, r.t. and 5h; Step 2: 2a (0.5 mmol, 2.5 equivalent, 49 µL,), PdCl2 (5 mol%, 1.8 mg), DCyPFc (10 mol%, 11.6 mg), CO (5 bar), 110 °C and 20 h. |
|---|
 |
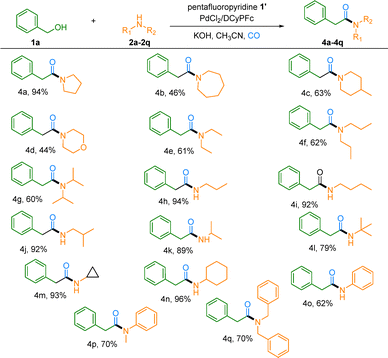
We then evaluated the generality of this transformation with respect to various alkylamines. A wide range of both primary and secondary alkylamines proved to be competent coupling partners, delivering the corresponding amides in 44–96% yield (Table 3, 4a–4q). Cyclic secondary amines of varying ring sizes performed effectively: five- and seven-membered ring analogues afforded the desired amides in 94% and 46% yield, respectively. Similarly, substituted piperidines such as 4-methylpiperidine and morpholine were well tolerated, giving the products in 44–63% yield (Table 3, 4c and 4d). Acyclic secondary amines also participated smoothly. Diethylamine, dipropylamine, and even the more sterically hindered diisopropylamine all provided the corresponding amides in >60% yield (Table 3, 4e–4g). Among primary alkylamines, linear analogues including propylamine, butylamine, and isobutylamine reacted with high efficiency (93–94% yield, Table 3, 4h–4j). Notably, more hindered primary amines such as isopropylamine and tert-butylamine remained viable substrates, furnishing the products in 79–89% yield, further highlighting the practical utility of this method (Table 3, 4k and 4l). The protocol also exhibited excellent compatibility with strained and alicyclic amines, with cyclopropylamine and cyclohexylamine delivering amides in up to 96% yield (Table 3, 4m and 4n). Furthermore, various benzylamines and aromatic amines proved to be valuable substrates, providing the corresponding products in 62–70% yield (Table 3, 4o–4q).
| a Reaction conditions: Step 1: 1a (0.2 mmol, 1.0 equivalent, 21 µL), pentafluoropyridine (0.3 mmol, 1.5 equivalent, 33 µL), KOH (0.5 mmol, 2.5 equivalent, 28 mg), CH3CN (2 mL), N2, r.t. and 5 h; Step 2: 2a–2q (0.5 mmol, 2.5 equivalent), PdCl2 (5 mol%, 1.8 mg), DCyPFc (10 mol%, 11.6 mg), CO (5 bar), 110 °C and 20 h. |
|---|
 |
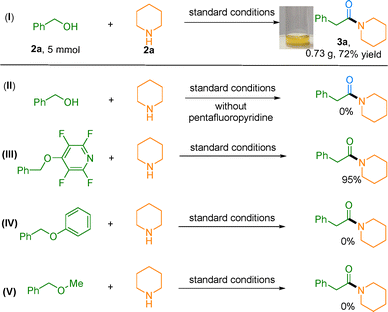
To assess practical applicability, we performed a gram-scale synthesis (5 mmol) under a 5 bar CO atmosphere. The reaction proceeded smoothly, delivering the desired product in 72% isolated yield (Scheme 1, I), demonstrating the scalability of this method. Mechanistic investigations through control experiments provided further insight. Omitting pentafluoropyridine under standard conditions completely inhibited product formation, indicating its essential role in C–O bond activation. This finding was corroborated by the isolation and structural confirmation of intermediate 1a'; when subjected to the standard reaction conditions, the purified intermediate cleanly furnished the target product in 95% yield (Scheme 1, II, III). Significantly, alternative C–O activation strategies failed to generate 3a (Scheme 1, IV, V), highlighting the distinctive efficiency of the pentafluoropyridine-enabled pathway. Together, these results provide important insights into the proposed catalytic cycle.
Based on experimental evidence and literature precedents,6,10 we propose a plausible catalytic cycle for this direct C–O activation carbonylation (Fig. 1). The reaction commences with the in situ formation of activated intermediate 1a′ from benzyl alcohol and pentafluoropyridine, which undergoes oxidative addition with Pd(0) to afford the benzylpalladium(II) species A. Following CO coordination and migratory insertion, the acylpalladium complex C is generated. Subsequent ligand exchange with piperidine furnishes intermediate D, from which reductive elimination delivers the amide product and regenerates the active Pd(0) catalyst for the next catalytic cycle. This mechanistic proposal aligns with all experimental observations and offers a coherent rationale for this unprecedented deoxygenative carbonylation reaction.
In summary, we have developed a palladium-catalyzed system that achieves the direct carbonylative amidation of benzylic alcohols with alkylamines. This transformation effectively reverses the inherent polarity of the alcohol, activating it as an electrophilic coupling partner, while avoiding the need for prefunctionalized substrates or stoichiometric activators. The protocol is operationally simple, scalable, and compatible with a wide range of functional groups, including those present in pharmaceutically relevant motifs. By providing a concise and sustainable route to benzylic amides from abundant feedstocks, this work not only advances the field of catalytic C–O bond functionalization but also establishes a practical platform for the modular synthesis of amide-containing compounds in organic and medicinal chemistry.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc01058j.Acknowledgements
We acknowledge the financial support from the Senior Talent Foundation of Jiangsu University (4111290008), the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (23KJB150007), and the Natural Science Foundation of Jiangsu Province (BK20250875). We also appreciate the general support of Professor Weiping Zheng at Jiangsu University.References
- (a) V. R. Pattabiraman and J. W. Bode, Nature, 2011, 480, 471–479 Search PubMed; (b) S. Kumari, A. V. Carmona, A. K. Tiwari and P. C. Trippier, J. Med. Chem., 2020, 63, 12290–12358 CrossRef CAS PubMed; (c) B. T. Matsuo, P. H. R. Oliveira, E. F. Pissinati, K. B. Vega, I. S. de Jesus, J. T. M. Correia and M. Paixao, Chem. Commun., 2022, 58, 8322–8339 RSC.
- (a) S. Z. Langer, S. Arbilla, J. Benavides and B. Scatton, Adv. Biochem. Psychopharmacol., 1990, 46, 61–72 CAS; (b) D. Aakash, B. R. Kaur, K. Ramanjot, K. Sanjiv, J. U. Kumar, S. Harinder, B. Sandeep, K. Dinesh and D. P. Kishore, Curr. Top. Med. Chem., 2017, 17, 238–250 Search PubMed; (c) N. N. Zhang, Y. L. Zhao, D. M. Fan, J. B. Xiao, K. W. Cheng and M. F. Wang, Food Hydrocolloids, 2020, 108, 106073 CrossRef CAS.
- (a) H. Konishi and K. Manabe, Tetrahedron Lett., 2019, 60, 151147 CrossRef CAS; (b) A. Schoenberg and R. F. Heck, J. Org. Chem., 1974, 39, 3327–3331 CrossRef CAS; (c) A. Schoenberg, I. Bartoletti and R. F. Heck, J. Org. Chem., 1974, 39, 3318–3326 CrossRef CAS.
- (a) J. Cao, Z.-J. Zheng, Z. Xu and L.-W. Xu, Coord. Chem. Rev., 2017, 336, 43–53 CrossRef CAS; (b) B. Gabriele, R. Mancuso and G. Salerno, Eur. J. Org. Chem., 2012, 6825–6839 CrossRef CAS; (c) Z.-P. Bao, L.-C. Wang and X.-F. Wu, ACS Catal., 2025, 15, 19580–19606 CrossRef CAS.
- (a) P. Villo, A. Shatskiy, M. D. Kärkäs and H. Lundberg, Angew. Chem., Int. Ed., 2023, 62, e202211952 CrossRef CAS PubMed; (b) E. Bisz and M. Szostak, ChemSusChem, 2017, 10, 3964–3981 CrossRef CAS PubMed; (c) J. Cornella, C. Zarate and R. Martin, Chem. Soc. Rev., 2014, 43, 8081–8097 RSC; (d) T. B. Boit, A. S. Bulger, J. E. Dander and N. K. Garg, ACS Catal., 2020, 10, 12109–12126 CrossRef CAS PubMed; (e) S.-Q. Zhang and X. Hong, Acc. Chem. Res., 2021, 54, 2158–2171 CrossRef CAS PubMed.
- X. W. Qin, H. Li, S. H. Sun, S. Y. Shen, T. F. Xu, X. F. Wu and Z. P. Yin, Org. Lett., 2025, 27, 12524–12529 CrossRef CAS PubMed.
- (a) J. Duan, J. Jiang, J. Gong, Q. Fan and D. Jiang, J. Mol. Catal. A: Chem., 2000, 159, 89–96 CrossRef CAS; (b) S. Jayasree, A. Seayad and R. V. Chaudhari, Org. Lett., 2000, 2, 203–206 CrossRef CAS PubMed; (c) S. Sabater, M. Menche, T. Ghosh, S. Krieg, K. S. L. Rück, R. Paciello, A. Schäfer, P. Comba, A. S. K. Hashmi and T. Schaub, Organometallics, 2020, 39, 870–880 CrossRef CAS; (d) Y. Zhu, D. Chen, M. Luo and H. Xia, Chin. J. Chem., 2024, 42, 2765–2772 CrossRef CAS; (e) Y. Zhang, Q. Cao, Y. Xi, X. Wu, J. Qu and Y. Chen, J. Am. Chem. Soc., 2024, 146, 7971–7978 CrossRef CAS PubMed.
- Y. Li, Z. Wang and X.-F. Wu, Green Chem., 2018, 20, 969–972 RSC.
- (a) Z. Yin, J. Xu and X.-F. Wu, ACS Catal., 2020, 10, 6510–6531 CrossRef CAS; (b) Z. Yin, W. Shi and X.-F. Wu, J. Org. Chem., 2023, 8, 4975–4994 CrossRef PubMed; (c) I. A. Khan, J. Luo, H. B. Shi, Y. Zou, A. Khan, Z. S. Zhu, W. M. Xu, D. Y. Wang and M. Huang, Food Chem., 2022, 368, 130845 CrossRef CAS PubMed; (d) N. N. Liang, B. Q. Shi, X. T. Hu, W. T. Li, X. W. Huang, Z. H. Li, X. N. Zhang, X. B. Zou and J. Y. Shi, Food Chem., 2024, 460, 140570 CrossRef CAS PubMed; (e) T. An, C. Liu, W. Yuan, X. Qin and Z. Yin, Chem. Commun., 2024, 60, 3389–3392 RSC; (f) C. Liu, T. An, W. Yuan, H. Dai, X. Liang and Z. Yin, Chem. Commun., 2023, 59, 12891–12894 RSC.
- Y. Jiang, Y. Wei, Q.-Y. Zhou, G.-Q. Sun, X.-P. Fu, N. Levin, Y. Zhang, W.-Q. Liu, N. Song, S. Mohammed, B. G. Davis and M. J. Koh, Nature, 2024, 631, 319–327 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |