Open Access Article
Open Access ArticleZn-flux-enabled synthesis of orthorhombic kagome YbFe6Ge6: Yb reduction and magnetic behavior
Rahul
Meduri
a,
Grant R.
Wilkinson
b,
Muhammad Z.
Idrees
c,
Kaya
Wei
 d,
Sebastian A.
Stoian
d,
Sebastian A.
Stoian
 c,
Henry S.
La Pierre
c,
Henry S.
La Pierre
 b,
Gregory T.
McCandless
a and
Julia Y.
Chan
b,
Gregory T.
McCandless
a and
Julia Y.
Chan
 *a
*a
aDepartment of Chemistry and Biochemistry, Baylor University, Waco, TX 76798, USA. E-mail: Julia_Chan@baylor.edu
bSchool of Chemistry and Biochemistry, Georgia Institute of Technology, Atlanta, GA 30332, USA
cDepartment of Chemistry, University of Idaho, Moscow, ID 83844, USA
dNational High Magnetic Field Laboratory, Tallahassee, FL 32310, USA
First published on 4th March 2026
Abstract
Single crystals of an orthorhombic polymorph of kagome metal YbFe6Ge6 were grown from Zn flux. This Zn-flux-stabilized phase exhibits mixed Yb valency and magnetic behavior that differs from its hexagonal counterpart, underscoring the profound influence of flux selection on both structure and physical properties in intermetallic materials.
Flux growth synthesis is an efficient method for producing bulk single-crystalline materials.1,2 The technique not only enables the discovery of compounds within complex phase spaces, but also facilitates the growth of large single crystals suitable for physical measurements. Flux growth leverages eutectic points in a mixture of elements, often utilizing a low-melting metal or compound to reduce the overall melting point of a reaction. The technique permits synthesis with refractory elements to be more accessible, thus making it particularly valuable for the exploratory growth of intermetallic phases. Conventional synthesis methods for intermetallic compounds typically promote the formation of thermodynamically stable phases within a given composition space, thereby limiting access to metastable or otherwise unexplored structural regions that require kinetic or non-equilibrium control.3 Flux growth can circumvent this by altering reaction pathways through adjustable experimental parameters intrinsic to the technique. For example, the formation of specific products can be guided by tuning the cooling rate or by selecting the temperature at which the reaction is removed from the furnace and the excess flux is removed. This level of control was demonstrated through the investigation of the Ce–Pd–Ga phase space where the use excess Ga flux lead to the discovery of a family of structurally related compounds.2,4 A range of related structures were obtained by tuning the heating parameters, which illustrate the diverse parametric control of flux growth methods. In the synthesis of the Prn+1ConGe3n+1 homologous series,5 the concentration of flux used directly affected which n-members and related phases crystallized from the melt. This study reinforced the notion that flux concentration itself serves as a valuable tuning parameter for directing phase formation.6 Harnessing control over structural formation within a phase space is also evident in the stabilization of the two polymorphs of Pr2Co3Ge5, one orthorhombic7 and one monoclinic, the latter displaying nearly Pr4+ character, which is rare for Pr intermetallic compounds.8
In this communication, we demonstrate how the choice of flux can decisively influence phase formation in intermetallic synthesis. YbFe6Ge6 is a member of the broader family of rare-earth kagome metals.9,10 The HfFe6Ge6-type structure (h-YbFe6Ge6, SG: P6/mmm) is routinely grown from a Sn flux and hosts a low temperature spin reorientation near 63 K,11,12 a feature associated with an anomalous Hall effect.13 The Y0.5Co3Ge3-type structure (y-YbFe6Ge6, SG:P6/mmm) can also be obtained through conventional arc-melting and annealing at 900 °C and does not possess the spin reorientation present in the ordered variant.14 Here, we show that loading Yb, Fe, Ge with an excess of Zn flux, yields an orthorhombic polymorph of YbFe6Ge6 (o-YbFe6Ge6, SG: Cmcm) that adopts the TbFe6Sn6 structure type and, exhibits differing magnetic properties than its hexagonal counterpart. Energy-dispersive X-ray spectroscopy confirms Zn incorporation into the structure while powder diffraction indicates a preferential Zn site occupation within the Fe-based kagome slabs (Fig. S2). For simplicity, we refer to Zn-flux-stabilized orthorhombic phase, YbFe5.52Zn0.48Ge6, as o-YbFe6Ge6 throughout this communication. Single crystals of o-YbFe6Ge6 were grown out of excess Zn flux with sizes up to 5 mm, adopting a hexagonal prism morphology (Fig. S1, inset). Synthesis conditions and structure determination using powder X-ray diffraction, single crystal X-ray diffraction and energy dispersive X-ray spectroscopy are provided in the SI.
Fig. 1 shows the unit cell of o-YbFe6Ge6 (a) and the pattering scheme of the Yb sublattice (b). Crystallographic data and refinement parameters for o-YbFe6Ge6 are given in Table S2 and atomic coordinates of o-YbFe6Ge6 are given in Table S3. Crystallographic comparison with h-YbFe6Ge6 was performed using the CIF from our previous report, as the reactant stoichiometry offers a more meaningful comparison to the orthorhombic variant under similar synthesis conditions.15 Both the h-YbFe6Ge6 and o-YbFe6Ge6 structures consist of alternating honeycomb and kagome slabs that are stacked in a staggered fashion, forming channels which house the Yb and the Ge–Ge dimer (Fig. S3a and b). In h-YbFe6Ge6, Yb occupies every other honeycomb slab along the c-axis. Structural complexity increases in o-YbFe6Ge6 due to differences in Yb ordering, which represents the main feature of distinction between the two structure types. Assuming the absence of the disordered sites, in h-YbFe6Ge6, the Yb atoms centered within the honeycomb hexagons are adjacent to one another in the ab plane. In contrast, o-YbFe6Ge6 exhibits a staggered Yb arrangement along the b-axis, such that every other pair of Yb atoms is adjacent. Unlike h-YbFe6Ge6, where Yb occupies every other honeycomb layer along the c-axis, the orthorhombic variant possesses Yb in each honeycomb layer along the a-axis, resulting in a more intricate ordering scheme. The reason behind the specific ordering was discussed in detail by Fredrickson et al.16 It is seen that with larger rare earth elements occupying the hexagons of the honeycomb layer (or other constituent elements that surround the rare earth), they are increasingly unable to order adjacent to one another because of increased steric interactions, leading to a staggered ordering of the rare earth element. One detour from the hexagonal to orthorhombic transition is seen with the hexagonal Y0.5Co3Ge3 structure type; that harbors large amounts of positional disorder of Yb and Ge dimers, resulting in a reduction of the hexagonal HfFe6Ge6-type cell by c/2. Our previous report of Yb0.5(Co1−xFex)3Ge3 explores the structural transition from a Y0.5Co3Ge3/CoSn hybrid structure to the HfFe6Ge6 structure type as a function of Fe substitution.15
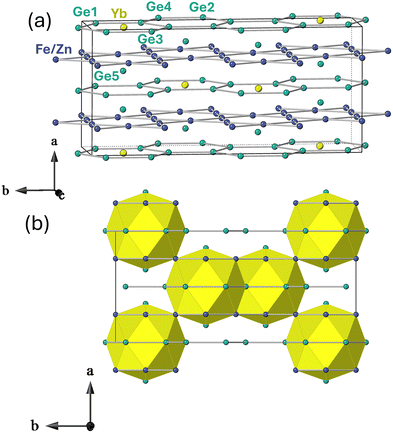 | ||
| Fig. 1 Unit cell (a) and pattering scheme (b) along the b-direction of o-YbFe6Ge6, disordered sites omitted for clarity. | ||
Fig. S4c and d show the honeycomb sublattice of the h-YbFe6Ge6 and o-YbFe6Ge6 structures, respectively with distinct interatomic distances highlighted to show how the perfect hexagonal slab in the h-YbFe6Ge6 structure differs from the irregular hexagons present in o-YbFe6Ge6. Fig. S4e and Fig. S4f show the areas of the h-YbFe6Ge6 and o-YbFe6Ge6 hexagons, respectively. Fig. S3c and d show the patterning scheme of the Ln environment in h-YbFe6Ge6 and o-YbFe6Ge6 which differs based on the occupancy of the lanthanide in the honeycomb net. Using the rationale that unfavorable steric environments can drive an orthorhombic distortion, we propose that Zn incorporation on the Fe sites reduces the local volume surrounding the Yb atoms in a manner reminiscent trends in of our previous doping study.15 Another possibility we considered is that Yb in o-YbFe6Ge6 may not be strictly trivalent; partial Yb2+ character could increase the average ionic radius and therefore influence the observed structural distortion. We also note that differences in synthesis conditions between h-YbFe6Ge6 and o-YbFe6Ge6 may play a role. Synthesis-dependent polymorphism has been documented in YFe6Sn6 and DyFe6Sn6,17 and studies of FeGe also previously showed that the annealing conditions affected the structural ordering,18 thus in our Yb0.5(Co1−xFex)3Ge3 report, an annealing step of 72 h was performed to ensure structural consistency in the doped series; such post-growth annealing was not performed here. When attempting to use the same heating profile as our previous report, the orthorhombic polymorph consistently forms with the only difference in structure being the amount of positional disorder present, which could be due to either the change in cooling rate or the time dwelled.
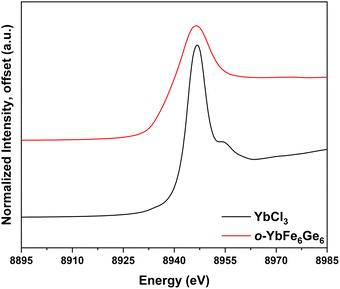
X-ray absorption spectroscopy (XAS) performed at the Yb L3-edge was used to quantify the Yb oxidation state in o-YbFe6Ge6 (see SI for measurement details). The resulting normalized spectra for o-YbFe6Ge6 and the known +3 oxidation state reference YbCl3 are shown in Fig. 2. Measurement of the edge energy via derivative analysis (Fig. S5c), reveals a small redshift in the edge energy from 8944.11 eV for YbCl3 to 8942.68 eV for o-YbFe6Ge6 (ΔE = −1.43 eV) consistent with an oxidation state assignment Yb3+ for the YbFe6Ge6 analyte.19 In addition to this main feature, the derivative of the spectrum displayed an additional, lower energy feature that contributes to the broad nature of the primary peak. Peak fitting analysis (see SI) was performed to deconvolute the spectrum. This revealed a secondary, lower intensity peak centered at 8938.27(5) eV, redshifted ∼6.6 eV from the main peak at 8944.846(16) eV. While the higher energy peak is consistent with Yb3+, the lower energy peak is consistent with a small percentage of Yb2+ character in the compound and this intermediate-valence effect has been observed in other Yb-containing intermetallic compounds.20–23 The appearance of Yb2+ character in these systems is associated with coupling of the conduction band electrons to the 4f hole of Yb3+, which itself lies near the Fermi energy. The increase of valency in the kagome sublattice with Zn substitution can also be contributing to the reduction of Yb. To estimate the relative Yb2+/3+ character of o-YbFe6Ge6, the ratio of the peak area of the Yb3+ feature to the sum of the area of both peaks, denoted nf (eqn (S1)), is calculated and found to be 0.79(8), which is consistent with the assignment of a formal Yb3+ oxidation state, with relatively minor amounts of Yb2+ character and an effective oxidation state (eqn (S2)) of +2.79(8) at the measurement temperature of 7 K.
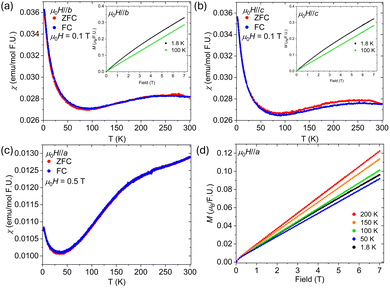
Fig. 3a and b presents the anisotropic temperature-dependent magnetic susceptibility measurements when the applied magnetic field is parallel to the b-direction and c-direction (μ0H‖b and μ0H‖c), respectively. The data for both field orientations are largely comparable, showing a broad hump beginning near 300 K, which suggests possible spin canting or fluctuations of the Fe moments down to approximately 100 K, where the emergence of Yb paramagnetism becomes apparent. The magnetization versus applied magnetic field measurements (inset) at T = 100 K and T = 1.8 K also exhibit similar behavior, characterized by an almost linear field dependence. These similarities for fields applied parallel to the b- and c-directions can be attributed to the fact that in both cases the field lies parallel to the kagome planes. Fig. 3c and d present the temperature-dependent magnetic susceptibility and the field-dependent magnetization, respectively, when the field is applied parallel to the a-direction (μ0H‖a). The susceptibility exhibits a gradual decrease from 300 K to about 50 K where a paramagnetic upturn is observed. Field-dependent magnetization measurements were collected at several temperatures to examine how changes in magnetic field strength influence the downturn observed in the magnetic susceptibility. The 200 K isotherm exhibits the largest magnetization across the measured field range. As the temperature decreases, the overall magnetization systematically diminishes until 50 K, where the onset of the Yb paramagnetic contribution becomes apparent, consistent with the susceptibility data. At 1.8 K, the magnetization increases slightly relative to the 50 K isotherm, reflecting the growing Yb paramagnetism at low temperatures. LnFe6Ge6 members (Ln = Gd-Lu) adopt an antiferromagnetic ordering of Fe moments along the kagome stacking axis at approximately 480–490 K.9 In compounds adopting the HfFe6Ge6 structure type, this axis corresponds to the c-axis, whereas in those with the TbFe6Sn6 structure type, it corresponds to the a-axis (Fig. 1). Therefore, in o-YbFe6Ge6 (TbFe6Sn6-type), the a-axis can be considered the magnetic “hard axis,” consistent with the orientation of the antiferromagnetically ordered Fe moments. Direct confirmation of the Fe-sublattice magnetic transition would require high-temperature magnetic susceptibility measurements, as the transition is expected to occur well above room temperature. This interpretation is further supported by the reduced magnitude of the susceptibility for μ0H‖a compared to μ0H‖b and μ0H‖c. Reliable Curie–Weiss fits could not be obtained because the Fe sublattice likely orders above 300 K, preventing access to a true paramagnetic regime for the Fe moments. The paramagnetic upturn observed below ∼50 K is attributed to the emergence of localized Yb3+ moments, which begin to dominate the magnetic response as thermal fluctuations decrease. At higher temperatures, the broad behavior of the magnetic susceptibility is governed primarily by Fe-based interactions, potentially involving weak spin canting or short-range correlations within the kagome planes. This is most likely a result of Zn incorporation into the Fe lattice, which diminishes the long-range ordering normally exhibited in a pure Fe kagome slab. As the temperature decreases, the Yb3+ ions display Curie–Weiss-like behavior, producing the rapid increase in susceptibility. This temperature evolution reflects a crossover from itinerant Fe magnetism at high temperatures to localized rare-earth paramagnetism at low temperatures, consistent with the onset of Yb-moment polarization in applied magnetic fields.
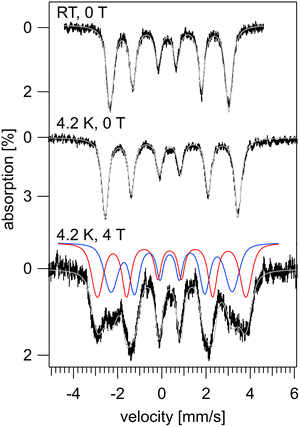
The temperature dependent, zero-field 57Fe Mössbauer spectra recorded for o-YbFe6Ge6 exhibit a single well defined six-line pattern, see Fig. 4 and Fig. S7. These spectra demonstrate that there is a spontaneous magnetic field acting on the 57Fe nuclei which reveals that the iron sublattice is magnetically ordered. Inspection of Fig. 4, Fig. S8, and Table S4 shows that the hyperfine field increases from Beff = 16.6 T at room temperature to 18.6 T at 4.2 K. These values are ∼10% larger than those reported for h- and y-type YbFe6Ge6.11,14,24,25 Assuming that the 11.39 T µB−1 conversion factor determined at 150 K for h-YbFe6Ge6 from its combined neutron diffraction and 57Fe Mössbauer spectroscopic investigation is still valid, these values suggests that for o-YbFe6Ge6 the iron-based magnetic moment increases from 1.46µB at room temperature to 1.63µB at 4.2 K. Importantly, lowering the temperature below 80 K does not lead to a broadening or a splitting of the observed resonances. This indicates that, just like for y-YbFe6Ge6, the spin reorientation transition observed at ∼63 K for h-YbFe6Ge6 is suppressed by the increased disorder of the Yb sites. Field dependent data up to 8 T was also collected (Fig. S9). These spectra suggest that above 6 T, the onset of a metamagnetic transition may occur even with no obvious features in the magnetization.
Several factors may account for the differences in magnetic behavior. (1) In the h-YbFe6Ge6 polymorph, XAS measurements confirm that Yb is trivalent. However, it is seen here that the o-YbFe6Ge6 polymorph has mixed valency that could potentially alter Yb–Fe coupling. (2) Partial substitution of Fe by Zn within the kagome lattice suppresses magnetic exchange among Fe atoms, thereby weakening the Yb–Fe coupling through alloy dilution. (3) The magnetic distinctions may also arise from differences in the structural ordering of the Yb sublattice. h-YbFe6Ge6 features a well-ordered triangular arrangement of Yb atoms, while the highly disordered y-YbFe6Ge6 contains a random Yb distribution and is reported to lack the spin reorientation present in the ordered variant.14 The specific patterning scheme of Yb atoms we observe in o-YbFe6Ge6 may similarly disrupt Yb-Fe exchange interactions. Given that the temperature dependent Mössbauer spectra manifests similar behavior as the y-YbFe6Ge6, the origin in the differences in magnetism seem to strongly point to the pattern of the Yb sublattice. In conclusion, the reduction of Yb from a Zn flux growth leads to a change in physical properties and structure in o-YbFe6Ge6 when compared to the previously reported h-YbFe6Ge6. Clarifying the mechanism of growth and its influence on the contrasting physical properties of h-YbFe6Ge6 and o-YbFe6Ge6 will require the use of in situ diffraction and spectroscopic methods and are a prospect of future work. The present study underscores the critical role of parametric control of the flux growth method as well as its significance in enabling the synthesis of previously inaccessible phases.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included as part of the supplementary information (SI). Supplementary information: additional experimental details, XRD, crystallographic data, XAS analysis, and additional Mössbauer spectra. See DOI: https://doi.org/10.1039/d6cc00958a.CCDC 2528289 contains the supplementary crystallographic data for this paper.26
Acknowledgements
R. M. acknowledges NSF DMR-2505304 and Welch AA-2056-20240404 and J. Y. C. acknowledges DOE DE-SC0022854 for partial support of this work. M. Z. I. and S. A. S. acknowledge NSF: CHE 2441886. A portion of this work was performed at the National High Magnetic Field Laboratory, which is supported by National Science Foundation Cooperative Agreement No. DMR-2128556 and the State of Florida. Use of the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, is supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences under Contract No. DE-AC02-76SF00515. The SSRL Structural Molecular Biology Program is supported by the DOE Office of Biological and Environmental Research, and by the National Institutes of Health, National Institute of General Medical Sciences (P30GM133894). G. R. W. was supported by the National Science Foundation Graduate Research Fellowship under Grant No. DGE-2039655. The spectroscopic studies of H. S. L. were supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Heavy Element Chemistry program under Award Number DE-SC0019385. Any opinion, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the NSF, NIGMS, or NIH.References
- P. C. Canfield and Z. Fisk, Growth of Single Crystals From Metallic Fluxes, Philos. Mag. B, 1992, 65, 1117–1123 CAS.
- W. A. Phelan, M. C. Menard, M. J. Kangas, G. T. McCandless, B. L. Drake and J. Y. Chan, Adventures in Crystal Growth: Synthesis and Characterization of Single Crystals of Complex Intermetallic Compounds, Chem. Mater., 2012, 24, 409–420 CrossRef CAS.
- M. G. Kanatzidis, R. Pöttgen and W. Jeitschko, The Metal Flux: A Preparative Tool for the Exploration of Intermetallic Compounds, Angew. Chem., Int. Ed., 2005, 44, 6996–7023 CrossRef CAS PubMed.
- S. E. Latturner, Clusters, Assemble: Growth of Intermetallic Compounds from Metal Flux Reactions, Acc. Chem. Res., 2018, 51, 40–48 CrossRef CAS PubMed.
- T. M. Kyrk, M. Bravo, G. T. McCandless, S. H. Lapidus and J. Y. Chan, Investigating the An+1BnX3n+1 Homologous Series: A New Platform for Studying Magnetic Praseodymium Based Intermetallics, ACS Omega, 2022, 7, 19048–19057 CrossRef CAS PubMed.
- T. M. Kyrk, M. G. Anderson, G. T. McCandless, S. H. Lapidus and J. Y. Chan, Influence of “Non-Interacting” Flux Concentration in Metallic Flux Growth Synthesis, Chem. Mater., 2025, 37, 2603–2610 CrossRef CAS.
- T. M. Kyrk, E. R. Kennedy, J. Galeano-Cabral, K. Wei, G. T. McCandless, M. C. Scott, R. E. Baumbach and J. Y. Chan, Anisotropic Magnetic and Transport Properties of Orthorhombic o-Pr2Co3Ge5, J. Phys.: Mater., 2022, 5, 044007 CAS.
- T. M. Kyrk, E. R. Kennedy, J. Galeano-Cabral, G. T. McCandless, M. C. Scott, R. E. Baumbach and J. Y. Chan, Much More to Explore with an Oxidation State of Nearly Four: Pr Valence Instability in Intermetallic m-Pr2Co3Ge5, Sci. Adv., 2024, 10, eadl2818 CrossRef CAS PubMed.
- G. Venturini, R. Welter and B. Malaman, Crystallographic Data and Magnetic Properties of RT6Ge6 compounds (R = Sc, Y, Nd, Sm, Gd-Lu; T = Mn, Fe), J. Alloys Compd., 1992, 185, 99–107 CrossRef CAS.
- G. Venturini, Filling the CoSn Host-Cell: The HfFe6Ge6-type and the Related Structures, Z. Kristallogr. - New Cryst. Struct., 2006, 221, 511–520 CrossRef CAS.
- T. Mazet and B. Malaman, Evidence of Spin Reorientation in YbFe6Ge6 from Neutron Diffraction and 57Fe Mössbauer Experiments, J. Phys.: Condens. Matter, 2000, 12, 1085–1095 CrossRef CAS.
- M. A. Avila, T. Takabatake, Y. Takahashi, S. L. Bud’ko and P. C. Canfield, Direct Observation of Fe spin Reorientation in Single-Crystalline YbFe6Ge6, J. Phys.: Condens. Matter, 2005, 17, 6969–6979 CrossRef CAS.
- W. Yao, S. Liu, H. Kikuchi, H. Ishikawa, Ø. S. Fjellvåg, D. W. Tam, F. Ye, D. L. Abernathy, G. D. A. Wood, D. Adroja, C.-M. Wu, C.-L. Huang, B. Gao, Y. Xie, Y. Gao, K. Rao, E. Morosan, K. Kindo, T. Masuda, K. Hashimoto, T. Shibauchi and P. Dai, Anomalous Electrical Transport in the Kagome Magnet YbFe6Ge6, Phys. Rev. Lett., 2025, 134, 186501 CrossRef CAS PubMed.
- J. M. Cadogan and D. H. Ryan, A Study on the Magnetic Behaviour of Polymorphic YbFe6Ge6, J. Phys.: Condens. Matter, 2010, 22, 016009 CrossRef CAS PubMed.
- R. Meduri, M. A. Plata, G. T. McCandless, B. C. Schundelmier, M. Ghafoor, K. Wei and J. Y. Chan, Evolution of Structural Order and Magnetic Anisotropy in Yb0.5(Co1−xFex)3Ge3 through Doping of a Kagome Lattice, Chem. Mater., 2025, 37, 2302–2313 CrossRef CAS.
- D. C. Fredrickson, S. Lidin, G. Venturini, B. Malaman and J. Christensen, Origins of Superstructure Ordering and Incommensurability in Stuffed CoSn-Type Phases, J. Am. Chem. Soc., 2008, 130, 8195–8214 CrossRef CAS PubMed.
- B. C. El Idrissi, G. Venturini and B. Malaman, Crystal Structures of RFe6Sn6 (R = Sc, Y, Gd-Tm, Lu) Rare-Earth Iron Stannides, Mater. Res. Bull., 1991, 26, 1331–1338 CrossRef CAS.
- X. Wu, X. Mi, L. Zhang, C.-W. Wang, N. Maraytta, X. Zhou, M. He, M. Merz, Y. Chai and A. Wang, Annealing-Tunable Charge Density Wave in the Magnetic Kagome Material FeGe, Phys. Rev. Lett., 2024, 132, 256501 CrossRef CAS PubMed.
- M. E. Fieser, M. G. Ferrier, J. Su, E. Batista, S. K. Cary, J. W. Engle, W. J. Evans, J. S. Lezama Pacheco, S. A. Kozimor, A. C. Olson, A. J. Ryan, B. W. Stein, G. L. Wagner, D. H. Woen, T. Vitova and P. Yang, Evaluating the electronic structure of formal LnII ions in LnII(C5H4SiMe3)31− using XANES spectroscopy and DFT calculations, Chem. Sci., 2017, 8, 6076–6091 RSC.
- J. Yamaguchi, A. Sekiyama, S. Imada, H. Fujiwara, M. Yano, T. Miyamachi, G. Funabashi, M. Obara, A. Higashiya, K. Tamasaku, M. Yabashi, T. Ishikawa, F. Iga, T. Takabatake and S. Suga, Kondo Lattice Effects and the Collapse of Lattice Coherence in Yb1−xLuxB12 Studied by Hard X-ray Photoelectron Spectroscopy, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 79, 125121 CrossRef.
- M. Okawa, M. Matsunami, K. Ishizaka, R. Eguchi, M. Taguchi, A. Chainani, Y. Takata, M. Yabashi, K. Tamasaku, Y. Nishino, T. Ishikawa, K. Kuga, N. Horie, S. Nakatsuji and S. Shin, Strong Valence Fluctuation in the Quantum Critical Heavy Fermion Superconductor ß-YbAlB4 A Hard X-Ray Photoemission Study, Phys. Rev. Lett., 2010, 104, 247201 CrossRef CAS PubMed.
- Y. Utsumi, H. Sato, S. Ohara, T. Yamashita, K. Mimura, S. Motonami, K. Shimada, S. Ueda, K. Kobayashi, H. Yamaoka, N. Tsujii, N. Hiraoka, H. Namatame and M. Taniguchi, Electronic Structure of Kondo Lattice Compounds YbNi3X9 (X = Al, Ga) Studied by Hard X-ray Spectroscopy, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 86, 115114 CrossRef.
- A. He, E. L. K. Wille, L. M. Moreau, S. M. Thomas, J. M. Lawrence, E. D. Bauer, C. H. Booth and S. M. Kauzlarich, Intermediate Yb Valence in the Zintl Phases Yb14MSb11 (M = Zn, Mn, Mg): XANES, Magnetism, and Heat Capacity, Phys. Rev. Mater., 2020, 4, 114407 CrossRef CAS.
- D. H. Ryan and J. M. Cadogan, Observation of Independent Iron and Rare-Earth Ordering in RFe6Ge6 (R = Y, Gd–Lu) Compounds, J. Appl. Phys., 1996, 79, 6004–6006 CrossRef CAS.
- D. H. Ryan, J. M. Cadogan and R. Gagnon, The Spin-Reorientation Transition in H-type YbFe6Ge6, J. Phys.: Conf. Ser., 2010, 217, 012124 CrossRef.
- CCDC 2528289: Experimental Crystal Structure Determination, 2026 DOI:10.25505/fiz.icsd.cc2qvwq6.
| This journal is © The Royal Society of Chemistry 2026 |