 Open Access Article
Open Access ArticleReview of supra-carbon dots: from functional modification to biological applications
Xue
Wu†
a,
Yupeng
Liu†
ab,
Caishi
Huang
*c and
Songnan
Qu
 *ab
*ab
aJoint Key Laboratory of Ministry of Education, Institute of Applied Physics and Materials Engineering (IAPME), University of Macau, Taipa, Macau SAR 999078, China. E-mail: songnanqu@um.edu.mo
bZhuhai UM Science and Technology Research Institute, University of Macau, Taipa, Macau SAR 999078, China
cFaculty of Health Sciences, University of Macau, Macau SAR, 999078, PR China. E-mail: cshuang@um.edu.mo
First published on 24th March 2026
Abstract
Carbon dots (CDs) tend to assemble and aggregate into supra-CDs due to surface energy-driven forces, electrostatic interactions, and hydrogen bonding. To better utilize the assembly process, external modulation strategies such as protonation regulation, nanoconfinement, solvent polarity control, and directed hydrogen bonding have been employed to design ordered structures. This review summarizes the assembly structures and functional modifications of supra-carbon dots, focusing on strategies to overcome limitations such as rapid renal clearance, shallow tissue penetration of luminescence, and restricted photothermal/photodynamic efficacy. Recent advances in the biological applications of supra-CDs, including bioimaging, drug loading, photothermal therapy, and photodynamic therapy, are systematically discussed. By integrating nanoscale properties with microscale/macroscale functionalities, supra-CDs demonstrate enhanced performance in diagnostics and therapeutics, offering a promising platform for future biomedical applications.
1. Introduction
Nanomedicine, as a pivotal frontier in 21st-century medical technology, offers revolutionary strategies for disease diagnosis and treatment by leveraging the unique physicochemical properties of nanoscale materials.1,2 Traditional nanocarriers, such as liposomes and polymeric nanoparticles, can deliver drugs via the enhanced permeability and retention (EPR) effect or through active targeting. However, they still commonly face several challenges, including insufficient physical stability, limited drug loading efficiency, unpredictable in vivo behavior, and potential biosafety concerns.3–5Carbon dots (CDs) are characterized by excellent water solubility, facile surface functionalization, low cytotoxicity, and high biocompatibility, making them promising candidates for biomedical applications.6,7 Their tunable fluorescence, straightforward synthesis routes, and abundant carbon sources further enhance their potential in bioimaging, drug delivery, photodynamic therapy (PDT) and photothermal therapy (PTT). However, the small size of CDs, while beneficial for tissue penetration, also leads to rapid renal clearance and limited tumor-targeting efficiency, thereby restricting their therapeutic efficacy. Moreover, most CDs exhibit emission in the short-wavelength region, which suffers from poor tissue penetration—highlighting the need to develop long-wavelength emissive CDs for deep-tissue imaging.8–10
Supra-carbon dots (supra-CDs) are CD assemblies with diverse architectures, formed through the non-covalent or covalent assembly of individual CDs.11–13 Early research primarily focused on suppressing CD aggregation due to the associated aggregation-caused quenching (ACQ) effect. However, with advances in chemistry and nanoscience, it has been recognized that precisely controlling the assembly process of CDs can lead to ordered structures that exhibit new functionalities not present in discrete CDs.14
A major research focus regarding supra-CDs involves functional modifications to overcome the limitations of conventional CDs in biomedical applications. For instance, optimizing the size and surface chemistry of supra-CDs can balance tumour accumulation efficiency and clearance kinetics. Studies indicate that supra-CD particles in the 50–100 nm range avoid rapid renal filtration while promoting enrichment in tumor tissues via the EPR effect.15 In terms of optical performance, supra-CDs can achieve significant absorption redshift through J-type aggregation. For example, platinum(IV)-coordinated supra-CDs (HS-Pt-CDs) developed by Qu and collaborators exhibited a redshift of 240 nm (from 423 nm to 670 nm), conferring near-infrared (NIR) photocatalytic properties and markedly improved tissue penetration.16 Moreover, supra-CDs demonstrate substantially enhanced photothermal conversion efficiency (PTCE) and reactive oxygen species (ROS) generation, leading to qualitative improvements in therapeutic outcomes. One study reported that pH-triggered supra-CDs achieved a PTCE of more than 50% under 650 nm irradiation, far exceeding that of most individual CDs.17–19
Despite significant progress, several challenges and research gaps remain in this field. First, the precise assembly dynamics and structure–function relationships of supra-CDs are not fully understood, hindering accurate prediction and control of their properties. Second, many studies focus on concentration-dependent self-assembled supra-CDs, yet the lack of effective regulation strategies often results in poor stability and uncontrollable behaviours in complex biological environments.20,21 The development of precise modulation strategies to enhance the adaptability of supra-CDs under physiological conditions remains an urgent issue for advancing real-world nanomedicine. Therefore, a comprehensive review of the current literature on supra-CDs is warranted to elucidate the fundamental principles governing the assembly of CDs into supra-CD structures, thereby informing the future design of multifunctional supra-CDs, particularly those intended for biological applications.
Based on the current situation, this review systematically summarizes the intrinsic driving forces and external modulation strategies for CD assembly, outlines the correlation between assembly structures and functions, and highlights recent advances in bioimaging, therapy, and theragnostics using assembled CDs. Finally, challenges and future directions in the field are discussed. In summary, by integrating nanoscale properties with micro/macroscopic functionalities, supra-CDs represent a versatile biomedical platform with great potential in diagnosis, therapy, and theragnostic integration.
2. Inherent driving forces
A fundamental prerequisite for fully utilizing supra-CDs is a deep understanding of the intrinsic driving forces that trigger CD aggregation. These forces primarily originate from the nanoscale characteristics of CDs and their rich surface chemistry, mainly including surface energy-driven effects, electrostatic interactions, and hydrogen bonding (Fig. 1).2.1 Surface energy-driven assembly
Surface energy-driven assembly represents one of the most basic forms. Due to the nano-size effect, individual CDs possess an extremely high specific surface area and surface energy, rendering them thermodynamically unstable.22 To reduce the total energy of the system, CDs tend to aggregate, thereby minimizing the exposed surface area and achieving a more stable state.23 This surface energy-driven process often does not require specific functional groups and exhibits a certain universality, but it is poorly controllable and prone to forming random aggregates.24,252.2 Electrostatic interactions
Electrostatic interactions also provide a significant driving force for CD assembly. The surfaces of CDs typically contain both positively charged groups (e.g., amino groups, –NH2) and negatively charged groups (e.g., carboxyl groups, –COOH), exhibiting amphoteric ionic characteristics.26,27 When oppositely charged groups are present on adjacent CDs, the electrostatic attraction between them can effectively overcome interparticle repulsion and serve as the primary driving force for assembly. For instance, Li et al. synthesized carbon nanodots (CNDs) from citric acid and urea via a hydrothermal method. Adjacent CNDs are supposed to assemble via possible interparticle electrostatic interactions and hydrogen bonding between the amino groups as positive charge centres and the carboxyl/pyridinic groups as negative charge centres. The IR spectra reveal that the characteristic absorption peaks of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, C
N, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and amino groups shifted to smaller wavenumbers in the supra-CNDs relative to the CNDs, indicating the formation of interparticle hydrogen bonds. These possible interparticle electrostatic interactions and hydrogen bonding could stabilize the assemblies and support the formation of supra-CNDs.17 In another study, Li et al. constructed hierarchical assemblies of CDs (HA-CDs) by assembling monodispersed ultraviolet-absorbing CDs. The resulting HA-CDs exhibit good hydrophilicity and achieve a superior PTCE of up to 84% under simulated solar irradiation.28 This mode of assembly is sensitive to environmental conditions, offering possibilities for a certain degree of regulation.
O and amino groups shifted to smaller wavenumbers in the supra-CNDs relative to the CNDs, indicating the formation of interparticle hydrogen bonds. These possible interparticle electrostatic interactions and hydrogen bonding could stabilize the assemblies and support the formation of supra-CNDs.17 In another study, Li et al. constructed hierarchical assemblies of CDs (HA-CDs) by assembling monodispersed ultraviolet-absorbing CDs. The resulting HA-CDs exhibit good hydrophilicity and achieve a superior PTCE of up to 84% under simulated solar irradiation.28 This mode of assembly is sensitive to environmental conditions, offering possibilities for a certain degree of regulation.
2.3 Hydrogen bonding
Among the various intrinsic driving forces, hydrogen bonding stands out as the most common and critical due to its directionality and saturation.29,30 During synthesis, CDs typically accumulate abundant oxygen- and nitrogen-containing functional groups, such as hydroxyl (–OH), carboxyl (–COOH), and amino (–NH2) groups, which act as excellent hydrogen bond donors and acceptors, creating inherent conditions for the formation of an intensive hydrogen-bonding network between CDs. The advantage of hydrogen bond-driven assembly lies in its moderate strength: it effectively guides the directional aggregation of CDs into structurally stable superstructures while allowing reversible disassembly under external stimuli (e.g., temperature, pH), which is crucial for constructing smart responsive materials.31 For example, Lin et al. synthesized chiral carbon dots (Ch-CDs) using R-/S-α-phenylethylamine and 3,4,9,10-perylenetetracarboxylic dianhydride as precursors. The Ch-CDs exhibit concentration-dependent self-assembly, attributed to directional hydrogen bonds provided by surface molecules.32 Similarly, Wang et al. synthesized carbonized polymer dots (CPDs) using trimeric acid (TA) and m-phenylenediamine (m-PD) as precursors via an airflow-assisted melt polymerization (AMP) method. The self-assembly, driven by supramolecular interactions such as hydrogen bonding and π–π stacking, significantly enhanced the emission of the CPDs.33 Therefore, hydrogen bonding not only acts as an adhesive in non-covalent assembly but also often serves as a prelude and template for covalent assembly, decisively influencing the structural order of the final supra-CDs. In summary, hydrogen bond-driven assembly is one of the most promising intrinsic strategies for achieving controllable and ordered CD assembly.These innate internal driving forces act synergistically or competitively to determine the likelihood of CD aggregation, also explaining the concentration-dependent assembly phenomena observed in many CDs. However, assembly processes relying solely on intrinsic forces are typically stochastic, lacking precise spatial and structural control, leading to significant polydispersity in the morphology, size, and structure of the resulting products. Particularly in aqueous environments, the abundance of water molecules competes with CD surface groups in forming hydrogen bonds, severely disrupting the intrinsic hydrogen-bonding network between CDs and limiting the formation of ordered hierarchical structures. While more defined assemblies might be achieved in organic solvents, this greatly restricts their direct application in biomedical fields. Consequently, relying exclusively on intrinsic driving forces makes it challenging to meet the demands for controllable CD assembly and functional applications, underscoring the urgent need to introduce external stimuli for precise regulation, guiding CDs along predefined pathways to assemble into advanced materials with specific functions and structures.
3. External modulation strategies
The intrinsic tendency of CDs to undergo assembly and aggregation, driven by surface energy, electrostatic interactions, and hydrogen bonding, often leads to unpredictable or disordered structures. To harness this propensity for creating functional architectures, precise external modulation strategies are paramount. These strategies allow for the rational design of supra-CDs with tailored optical properties, enhanced stability, and specific morphological characteristics. This section delves into the principal external modulation techniques—protonation regulation, nanoconfinement, solvent engineering, directed hydrogen bonding, and surface functional group control—highlighting their mechanisms, advantages, and profound implications for advancing biological applications in nanomedicine.3.1 Protonation regulation
Protonation regulation is a highly effective and reversible strategy for controlling the assembly state of CDs by altering their surface charge and intermolecular interactions through pH changes.34 This approach typically involves the protonation of basic surface functional groups (e.g., amino groups) or, as notably demonstrated, the protonation of acidic groups like phosphates under acidic conditions.35,36 For instance, CDs synthesized from levofloxacin and phosphoric acid possess surface phosphate groups (Fig. 2a). Under neutral conditions, these CDs exist in a dispersed molecular state, exhibiting green room-temperature phosphorescence (RTP). Upon acidic treatment, the phosphate groups undergo protonation, which significantly reduces electrostatic repulsion between CD particles and, more importantly, enhances the hydrogen bonding capability of the surface. This protonation-triggered self-assembly leads to a redshift in phosphorescence emission from green to the NIR region (e.g., 725 nm), a phenomenon attributed to the elongation of the π-conjugated core and an increased density of delocalized π-electron clouds, which reduces the bandgap (Fig. 2b).37,38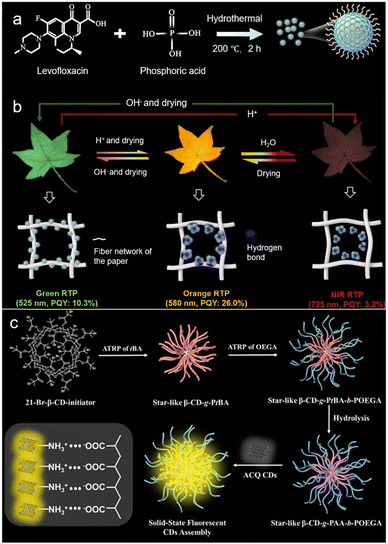 | ||
| Fig. 2 (a) Schematic diagram of CDs’ synthesis processes. (b) Schematic of the mechanism of redshift in the luminescence region induced by water and acid. Reproduced with permission from ref. 37. Copyright 2025, Wiley-VCH. (c) Schematic of the polymer unimolecular micelles directed nanoconfined self-assembly of ACQ CDs with star-like hydrophilic polymer β-CD-g-PAA-b-POEGA as template scaffolds. Reproduced with permission.41 Copyright 2025 The Authors. | ||
The NIR emission absorbance and excitation make protonation-regulated supra-CDs exceptionally promising for biosensing and imaging. The mildly acidic microenvironment of tumors (pH ∼ 6.5–7.0) or inflammatory sites can serve as an endogenous trigger for the assembly of pH-sensitive supra-CDs. The CDs designed by Shen can self-assemble into aggregated states under the regulation of pH values (ranging from approximately 11.0 to approximately 5.0), thus demonstrating a high absorption rate in the visible to NIR region. Surface protonation in an acidic environment may generate electrostatic attraction, eventually causing these CDs to aggregate and form larger aggregates (SCDs).39 In addition, PH-induced hydrophilic CDs’ aggregation can also be used for fluorescence detection of acidic amino acids and intracellular pH imaging.40
3.2 Nanoconfinement effects
Nanoconfinement involves directing the assembly of CDs within spatially restricted environments, such as the pores of templates or the cores of unimolecular micelles. This strategy physically separates individual CD units, preventing the uncontrolled aggregation that typically leads to fluorescence quenching. A sophisticated example utilizes β-cyclodextrin-grafted-poly(acrylic acid)-block-poly(oligo(ethylene glycol)acrylate) (β-CD-g-PAA-b-POEGA) unimolecular micelles as nanoreactors. Within these nanoreactors, the PAA chains act as both electrostatic binders and physical spacers, facilitating a high-density yet isolated assembly of CDs. This nanoconfined aggregation effectively prevents direct π–π stacking between adjacent CDs while simultaneously enhancing emission through the restriction of intramolecular motion (RIM) (Fig. 2c).41,42In biomedicine, the nanoconfinement approach is directly translatable to designing robust nanoplatforms for diagnosis and theragnostic. Yu et al. transformed CDs into metal–organic frameworks (MOFs) via Fe3+-carbenicillin (CARB) coordination. In the acidic microenvironment caused by infection, the dissociation of CDs to restore fluorescence enables real-time detection of bacteria.43 DNA, serving as a scaffold, can be used to arrange carbon layers in homogeneous, rapidly synthesized assemblies, expanding the range of emission properties that all-carbon hydrocarbons can achieve in water, making it a highly regarded biomaterial.44–47
3.3 Solvent engineering
Solvent engineering exploits the polarity and solvation power of the solvent medium to balance intermolecular forces, thereby controlling the kinetics and thermodynamics of CD assembly.47 By systematically varying the solvent composition, such as the water fraction in acetonitrile–water (CH3CN–H2O) mixtures, the aggregation behaviour of CDs can be precisely programmed. A high-quality solvent (e.g., pure DMSO) solvates the CD surface groups effectively, keeping the particles dispersed. As the solvent polarity increases (e.g., by adding water), the solvation shell is destabilized, driving the CDs together through hydrophobic interactions (Fig. 3a). Time-dependent spectral evolution in solvents like methanol reveals a progressive shift in fluorescence from blue (characteristic of dispersed particles) to red, indicating controlled, stepwise aggregation at the sub-nanoscale (Fig. 3b and c).49 Large assemblies of CDs, formed via the precipitation of the CDs from aqueous solutions by the addition of N,N-dimethyl methanamide (DMF), can come together to form regular, well-ordered structures whilst still maintaining their PL properties.50 The solvent method for regulating the assembly of CDs is hindered in the application of biological existence. Effectively ensuring the water solubility and stability of CD assemblies is key.29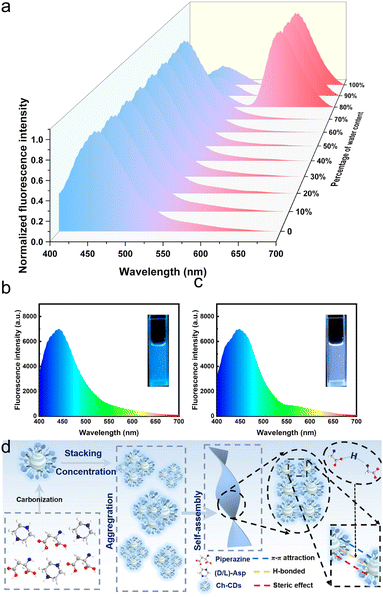 | ||
| Fig. 3 (a) Fluorescence emission spectra of as-prepared CDs-0 solution (0.1 mg mL−1) with varying ratios of water. Fluorescence emission spectrum and fluorescence image of CDs-0 dispersed in methanol (0.1 mg mL−1) (b) at first and (c) after 10 min. Reproduced with permission from ref. 49. Copyright 2025, Wiley-VCH. (d) Schematic of the assembly process of Ch-CDs. Reproduced with permission from ref. 51. Copyright 2025, Wiley-VCH. | ||
3.4 Directed hydrogen bonding
Moving beyond random interactions, directed hydrogen bonding involves the strategic design of CD surface chemistry to pre-program specific, directional hydrogen-bonding patterns. This biomimetic approach ensures that CDs assemble into well-defined, ordered superstructures. A prime example is the use of chiral precursors like D- or L-aspartic acid during CD synthesis. The chiral functional groups grafted onto the CD surface impose a specific spatial orientation that guides the assembly process. Through directional hydrogen bonds formed by these amino acid residues, along with contributions from van der Waals forces and core interactions, CDs can be induced to assemble into sophisticated architectures such as curved nanoribbons, which exhibit emergent properties like circularly polarized luminescence (CPL) (Fig. 3d).51 Surface-modified ligands represent a feasible strategy for the ordered assembly of CDs. Utilizing the hydrophobic interactions of aromatic ligands to drive the directional assembly of CDs can endow the assembled structures with classical aggregation-induced emission characteristics.52In addition, several other methods have been employed to modulate the assembly process. For instance, Lin et al. introduced coordination-induced self-assembly technology by leveraging the coordination interaction between zirconium oxo clusters and carboxyl groups on CDs, enabling efficient enrichment of CDs.53 Wang et al. precisely controlled the ratio of surface functional groups to direct AIE CDs into either highly aggregated or weakly aggregated states. These distinct aggregation states alter the intermolecular energy gaps through spatial configuration changes, thereby enabling fine-tuning of the nuances in fluorescence properties.49
External modulation strategies serve as a valuable tool for enhancing and expanding the functionalities of simple CDs into complex functional materials known as supra-CDs. By integrating nanoscale properties into micro- and macro-scale functions, supra-CDs offer a promising and versatile platform for the next generation of diagnostic and therapeutic agents.
4. Performance-oriented improvement
Individual CDs face challenges in biomedical applications, such as rapid renal clearance, insufficient tissue penetration depth, and limited therapeutic efficacy. Constructing supra-CDs through precise assembly strategies can effectively overcome these bottlenecks, enabling performance-oriented optimization and functional enhancement. This section will systematically elaborate on the performance-oriented improvement of key properties of supra-CDs.4.1 Biocompatibility: aqueous solubility optimization and size control
The assembly process of supra-CDs does not merely enhance aqueous solubility. By meticulously balancing the hydrophilic and hydrophobic groups on their surface, good aqueous dispersibility can be maintained. For instance, amphiphilic molecules such as F127 have been employed to modify near-infrared-emitting carbon dots (NECDs), leading to the formation of stable NECDs-assemblies (NECDs-A) with an average diameter of about 200 nm and improved dispersibility in physiological environments (Fig. 4a).54 Similarly, Jia et al. utilized DSPE-PEG to cooperatively self-assemble with magneto fluorescent Mn-CDs, thereby improving their water solubility and biocompatibility.55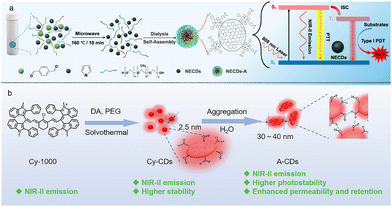 | ||
| Fig. 4 (a) The schematic of the synthesis process of NECDs-A. Reproduced with permission.54 Copyright 2018 Elsevier B.V. (b) Preparation and aggregation of Cy-CDs. Reproduced with permission from ref. 56. Copyright 2025, Wiley-VCH. | ||
More importantly, assembly strategies allow for precise regulation of the size of supra-CDs. Studies have shown that controlling the size of supra-CDs within the range of 50–100 nanometres can avoid the rapid renal clearance issue associated with individual CDs being too small, while simultaneously leveraging the enhanced permeability and retention (EPR) effect of solid tumors. This significantly increases their accumulation at tumor sites, thereby improving targeting efficiency. For example, polyline-modified NIR-emitting CDs assemblies (plys-CDs) exhibit a size of approximately 40 ± 8 nm, which falls within the optimal range for effective EPR-mediated tumor targeting.14 Likewise, cyanine dye-derived NIR-II Cy-CDs (2.5 nm) can spontaneously aggregate via hydrogen-bonding interactions into larger assemblies (A-CDs) of 30–40 nm, endowing them with excellent tumor-targeting ability and stability under varying pH conditions (Fig. 4b).56 In another study, lecithin-derived self-assembled carbon dots (LDSCD) were developed to address the limitation of small-sized CDs (<10 nm) that are rapidly excreted from cells. The assembled structures show higher retention in cancer cells and enhanced phototherapeutic efficacy.57 Thus, through rational assembly design, supra-CDs can be tailored to possess both superior aqueous compatibility and optimized dimensions for enhanced biomedical performance.
4.2 Optical properties: fluorescence redshift and enhancement
Supra-CDs assemblies exhibit a qualitative leap in optical performance compared to individual CDs. The enhanced properties primarily stem from precise morphological and electronic structural modulations achieved through the assembly process.58,59 Firstly, supra-CDs can achieve significant red shifts in both absorption and emission spectra. This phenomenon largely relies on the expansion of π-conjugated domains within the assembly structure. For instance, under the guidance of directional hydrogen bonds, CD particles undergo ordered self-assembly, promoting through-space conjugation (TSC) between particles, and resulting in a narrowed energy band gap, which leads to a red-shifted emission (Fig. 5a–d).29 Similarly, the concentration-induced self-assembly of CDs derived from carbon spheres and sucrose leads to the formation of H- and J-type aggregates, which introduce new excitation bands at longer wavelengths (e.g., 465 and 520 nm) and cause the photoluminescence behaviours to be tunable from green to red emission.60 Yang et al. designed and synthesized a relatively flexible molecule (1, N4,N7-di(quinolin-2-yl)benzo[c][1,2,5]thiadiazole-4,7-diamine, Scheme S1), which contains a flat π-conjugated benzothiadiazole (BTD, chromophore) unit and two terminal units of aminoquinoline (4AQ). As intermolecular π–π interactions are the main force driving the self-assembly of 1, studying crystalline organic nanodots composed of 1 can provide a simplified model to simulate the structure of CDs. Crystalline organic nanodots showed an obviously aggregation-induced luminescence redshift as well as luminescence enhancement, which provides a new idea for exploring the fluorescence mechanism of CDs.61 This capability to shift the fluorescence emission of the CDs into the NIR region is crucial for enhancing their penetration capability in biological tissues, laying the foundation for deep-tissue imaging and therapy.62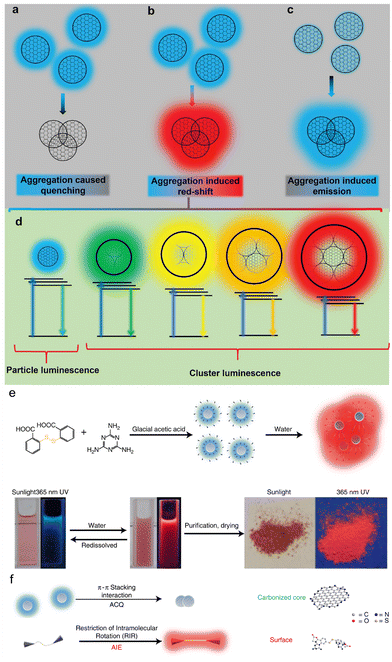 | ||
| Fig. 5 Schemes showing (a) classic aggregation-caused photoluminescence quenching; (b) aggregation-induced photoluminescence shift; (c) classic aggregation-induced photoluminescence emission; and (d) aggregation-induced particle and cluster luminescence. Reproduced with permission from ref. 29. Copyright 2025, Wiley-VCH. (e) H-CDs’ formation, structure, fluorescence, and appearance in different states. (f) Fluorescence principle and proposed structure of H-CD's core and surface (the colors of glowing edges represent the color of their fluorescence). H-CD, hydrophobic CDs. Reproduced with permission.64 Copyright 2024 The Authors. | ||
On the other hand, overcoming the common issue of aggregation-caused quenching is vital for practical applications. Supra-CDs address this through strategies such as the restriction of intramolecular motion (RIM), which can effectively suppress non-radiative decay pathways.63 A representative example is hydrophobic CDs (H-CDs) obtained via a one-pot solvothermal method, which exhibit blue emission in the dispersed state. Upon aggregation induced by water addition, the π–π stacking interactions within the carbonized cores quench the blue emission, while the restriction of intramolecular rotation around surface disulfide bonds turns on red fluorescence, conforming to the aggregation-induced emission phenomenon (Fig. 5e and f).64,65 This AIE effect can be systematically tailored; research on AIE CDs has demonstrated that increasing the nitrogen content in the precursor can enhance intramolecular charge transfer (ICT) efficiency, leading to a fluorescence change from green to red and an increase in quantum yield. Furthermore, the assembly process itself can create novel optical properties. The construction of CD-based supramolecular aggregates in an antisolvent like ethanol can lead to a decreased energy gap between the ground and excited states, contributing to a new long-wavelength, excitation-dependent emission band.48 Even model systems, such as crystalline organic nanodots composed of a flexible π-conjugated molecule, exhibit aggregation-induced luminescence redshift and enhancement, providing simplified insights into the mechanism behind the optical enhancement in supra-CDs.61 Fluorescence-enhanced carbon dots (named hr-CDs) were prepared from sustainable hydrogenated rosin, using a simple hydrothermal method in a water solvent. With the increase in the concentration of hr-CDs aqueous solutions, the distance between the carbon cores decreased, which resulted in the formation of J aggregates and the enhanced blue fluorescence emission.66 Therefore, by leveraging these assembly strategies, supra-CDs substantially improve their luminescence intensity, stability, and functionality for advanced optoelectronic and biomedical applications.
4.3 Endowment and augmentation of therapeutic: PTT and PDT
One of the most significant advantages of supra-CDs lies in their ability to integrate and enhance multiple therapeutic functions. In photothermal therapy, the ordered assembly structure can promote non-radiative transitions, enabling supra-CDs to exhibit a much higher PTCE than individual CDs. In photodynamic therapy, close packing facilitates electron transfer and energy transfer, thereby efficiently generating reactive oxygen species (ROS). Furthermore, by introducing specific functional units (e.g., photocatalytic centres), supra-CDs can be endowed with new properties such as photocatalysis, enabling the construction of integrated diagnostic and therapeutic platforms.A key strategy for enhancing these photophysical processes involves optimizing the intermolecular interactions within the assembled structure. For example, Ding et al. introduced oxygen and nitrogen co-doped atoms into CDs, which adjusted the non-coplanar molecular angles and promoted favourable π–π stacking interactions. This directed the self-assembly of adjacent nanoparticles into metal-free rigid clusters with a gear-like meshing pattern. As a result, intramolecular motion was effectively constrained, vibration coupling was reduced, and non-radiative transition losses were minimized, thereby ensuring efficient NIR emission from the CD clusters even at high concentrations.67 Such precise control over the assembly geometry not only strengthens the optical performance but also provides a structural basis for enhancing energy and electron transfer in therapeutic applications.
Moreover, the dense packing and regular organization of CDs in supra-assemblies’ favor exciton delocalization and facilitate charge carrier migration, which is critical both for ROS generation in PDT and for photocatalytic processes. By carefully designing the doping elements and assembly conditions, it is possible to tailor the electronic structure of supra-CDs, thereby augmenting their catalytic and therapeutic efficacy for combined cancer treatment and beyond.
In summary, through the synergistic regulation and internal interactions (e.g., hydrogen bonding, electrostatic interactions), supra-CDs have achieved performance-oriented improvements in aqueous solubility, size, optical properties, and therapeutic functions, demonstrating significant potential for biomedical applications.
5. Biomedical applications
Supra-CDs demonstrate significant potential in the biomedical field due to their tunable physicochemical properties, excellent optical characteristics, and good biocompatibility. In recent years, researchers have successfully applied supra-CDs in various cutting-edge areas, including bioimaging, cancer therapy, drug delivery, biosensing, and antibacterial applications, thereby advancing the development of theragnostic.68,69 This section will systematically review the latest research progress in the biomedical applications of supra-CDs.5.1 Bioimaging
Bioimaging is a key technology for the visual observation of the interior of living organisms. The emission wavelengths of individual CDs are mostly concentrated in the visible or first NIR window (NIR-I, 750–900 nm), which presents challenges such as limited tissue penetration depth and strong autofluorescence interference. The supramolecular assembly strategies of supra-CDs offer an effective approach to address these issues. For instance, CDs have been functionalized and assembled to achieve targeted and enhanced imaging capabilities. One study reports yellow fluorescent, rare-earth-doped aggregation-induced emission CDs that exhibit excellent biocompatibility and precise targeting for long-term imaging of zebrafish embryos and specific cellular structures like the nucleus in HeLa cells.70,71 Similarly, assemblies formed from amphiphilic CDs enable ultrafast, wash-free cellular imaging and specific organelle targeting, such as mitochondria (Fig. 6a).72 Furthermore, the emission of CDs can be significantly amplified through strategic assembly with other nanomaterials. In one approach, hydrophobically tailored CDs were conjugated with organic nanoparticles, creating nanohybrids with enhanced AIE properties suitable for long-term cellular imaging due to their superior brightness and biocompatibility.73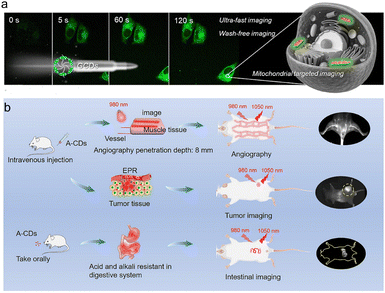 | ||
| Fig. 6 (a) Amphiphilic CDs for ultrafast and wash-free mitochondria-targeted imaging. Reproduced with permission.72 Copyright 2025, American Chemical Society. (b) A-CDs’ applications in mouse angiography, tumor imaging and gastrointestinal motility dynamic imaging. Reproduced with permission from ref. 56. Copyright 2025, Wiley-VCH. | ||
Through meticulously designed assemblies, researchers have successfully achieved a red shift in the luminescence of CDs from the visible range to the second NIR window (NIR-II, 1000–1700 nm). NIR-II imaging offers reduced tissue scattering, weaker autofluorescence, and greater penetration depth, providing biological images with a higher signal-to-noise ratio and resolution. For example, a novel cyanine dye-derived CD was developed with emission at 1044 nm in aqueous solution. These CDs can spontaneously aggregate in aqueous solution via hydrogen bonding, forming larger supra-CD structures that exhibit excellent tumor-targeting ability and resistance to environmental changes, enabling high-resolution NIR-II imaging of vasculature, tumors, and dynamic physiological processes in vivo (Fig. 6b).56 More importantly, these NIR-II luminescent supra-CDs often concurrently possess photodynamic or photothermal therapeutic functions, laying a solid foundation for achieving “theragnostic.” In surgical navigation, NIR afterglow imaging technology based on supra-CDs can guide surgeons to precisely resect tumor margins, significantly improving the thoroughness and safety of surgery.
5.2 Cancer therapy
The application of supra-CDs in cancer therapy is particularly remarkable, as they can not only serve as agents for monotherapy but also be used to construct platforms for diversified synergistic therapies.74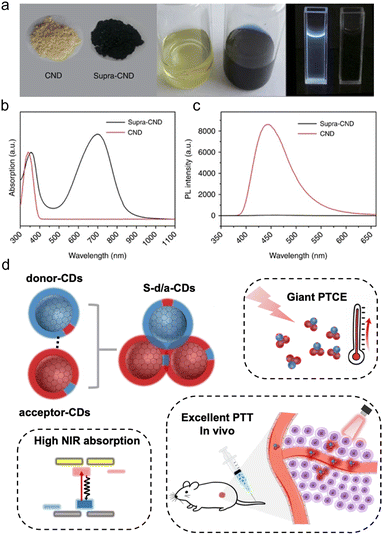 | ||
Fig. 7 (a) Optical images of CNDs (left) and supra-CNDs (right). (b) The absorption spectra, and (c) the fluorescence spectra of CND and supra-CND (0.28 mg mL−1) excited at 340![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nm. Reproduced with permission from ref. 17. Copyright 2016. The Authors. (d) Rationally assembling different surface functionalized CDs for enhanced NIR tumor photothermal therapy. Reproduced with permission from ref. 75. Copyright 2025, College of Chemistry and Molecular Engineering, Peking University. Published by Elsevier B.V. All rights are reserved. nm. Reproduced with permission from ref. 17. Copyright 2016. The Authors. (d) Rationally assembling different surface functionalized CDs for enhanced NIR tumor photothermal therapy. Reproduced with permission from ref. 75. Copyright 2025, College of Chemistry and Molecular Engineering, Peking University. Published by Elsevier B.V. All rights are reserved. | ||
More advanced designs involve the precise integration of therapeutic functions at the nano structural level. A representative example is a novel class of supra-CDs constructed by fusing red-emissive CDs with an electron-accepting molecule, 2,3-dicyanohydroquinone (DCHQ). This fusion creates a unique donor–acceptor nanostructure, where the assembled CD core acts as the electron donor and the DCHQ-derived interface serves as the electron acceptor. Upon light irradiation, this configuration enables highly effective photoinduced charge separation. Electrons transfer to the acceptor site to generate superoxide radicals, while the remaining holes within the CDs can directly oxidize cellular components. This dual mechanism results in exceptional cell-killing efficacy. Furthermore, the optimized particle size (∼20 nm) of these supra-CDs enhances tumor accumulation via the enhanced permeability and retention effect, leading to satisfactory anti-tumor PDT outcomes in vivo (Fig. 8a).81
 | ||
| Fig. 8 (a) Donor–acceptor type supra-carbon-dots with long lifetime photogenerated radicals boosting tumor photodynamic therapy. Reproduced with permission.81 Copyright 2024 The Authors. (b) A structures and photocatalytic behaviours of J-type aggregates in HS-Pt-CDs under NIR irradiation. (c) Schematic illustration of photocatalytic HS-Pt-CDs that promote the NIR-triggering Pt(II) species release and ROS to trigger tumor pyroptosis to prevent cancer metastasis. Reproduced with permission.16 Copyright 2025 The Authors. | ||
5.3 Drug delivery and controlled release
Supra-CDs inherently possess a large specific surface area and abundant functional groups, making them ideal drug carriers. Through methods such as π–π stacking, electrostatic adsorption, or covalent bonding, chemotherapeutic drugs, nucleic acid drugs, or protein drugs can be efficiently loaded onto supra-CDs. For instance, CDs with distinct surface functional groups (e.g., carboxyl or amino groups) can be covalently conjugated to chemotherapeutic agents such as doxorubicin (DOX), forming versatile nanocarriers that leverage the properties of their precursors for enhanced drug delivery.86,87 Moreover, amphiphilic CDs can self-assemble into cluster structures under ultrasound, enabling efficient encapsulation of DOX and subsequent surface modification with targeting ligands (e.g., cRGD peptides). These nanocomposites exhibit dual passive and active tumor-targeting capabilities, leading to timely drug release, significant antitumor efficacy, and reduced systemic toxicity in malignant glioma models (Fig. 9a).88 | ||
| Fig. 9 (a) Illustration of the construction of CDs-cluster-DOX/cRGD-PAA-OA nanocomposites via a self-assembly strategy for cancer imaging and treatment. Reproduced with permission.88 Copyright 2018, Wiley-VCH GmbH. (b) The proposed fluorescence principles of hydrogen bonding cross-linking and breaking. Reproduced with permission.95 Copyright 2021 Elsevier B.V. (c) Schematic illustration of aggregated CDs-loaded macrophages for eliminating bacteria and stimulating anti-inflammation macrophages. Reproduced with permission.97 Copyright 2022 The Authors. | ||
Beyond small-molecule drugs, supra-CDs also serve as promising vectors for nucleic acid delivery. Polyethyleneimine-modified CDs (PEI-CDs) can self-assemble with DNA into compact, stable nanospheres, protecting genetic material from enzymatic degradation while achieving high loading capacity. The incorporation of Mg2+ further enhances stability and facilitates biological imaging applications, demonstrating the potential of supra-CD-based assemblies for gene therapy and theragnostics.89
The design of supra-CD carriers often incorporates stimuli-responsive elements to enable controlled release at the target site. By exploiting tumor-specific conditions such as acidic pH, elevated glutathione levels, or enzymatic activity, these systems can achieve precise drug liberation, thereby maximizing therapeutic efficacy while minimizing off-target effects. Such advanced control over drug release kinetics underscores the potential of supra-CDs to advance precision medicine in oncology and beyond.
5.4 Biosensing
The fluorescence properties (e.g., intensity, wavelength) of supra-CDs can change in response to alterations in their immediate microenvironment (e.g., pH, ion concentration, and presence of specific biomolecules). Leveraging this characteristic, highly sensitive biosensors can be constructed.90–92 For instance, the co-assembly of peptides with carbon nanodots (CNDs) exploits non-covalent interactions to form structures that integrate the molecular recognition capability of peptides with the catalytic activity of CNDs. The catalytic activity of the synthesized carbon nanodots (CNDs) is dependent on pH, temperature, and the concentration of H2O2. For example, the UV-vis absorption signal of the solution after incubation with the electrodes increases as the pH rises from 2.0 to 4.0. Such systems have been applied for the highly sensitive detection of biomarkers like transglutaminase 2 (TG2), achieving a detection limit as low as 0.25 pg mL−1.93,94 Similarly, hydrogen-bonded self-assembled carbon dots (HB-CDs) exhibit tunable fluorescence emissions, where the introduction of hydrogen-bond-breaking agents induces a shift from yellow to blue fluorescence, demonstrating dual-mode emission behavior attributable to the coexistence of J-aggregated and dispersed states of HB-CDs. This responsive fluorescence enables the design of sensors for detecting subtle changes in the microenvironment (Fig. 9b).95,965.5 Antibacterial applications
Supra-CDs also show promising applications in the antibacterial field, particularly through their ability to generate reactive oxygen species that disrupt bacterial membranes and, when combined with photothermal effects, effectively eliminate drug-resistant bacteria. For example, macrophages loaded with aggregated carbon dots (ACDs) exhibit enhanced targeting and bactericidal activity against multidrug-resistant pathogens while simultaneously modulating immune responses by promoting anti-inflammatory macrophage polarization, thereby improving survival outcomes in sepsis models (Fig. 9c).97 Similarly, kanamycin sulfate-derived carbon dots (KCDs) self-assemble with cationic guar gum to form injectable, self-healing hydrogels (CG-KCDs) that, under light irradiation, penetrate and disrupt bacterial biofilms via ROS-mediated mechanisms, offering a versatile platform for treating wound infections.98 Beyond hydrogels, supra-CDs can be integrated into smart wound dressings, such as those embedding CDs in metal–organic frameworks (MOFs) functionalized with antibiotics. These systems respond to acidic infection microenvironments by concurrently releasing drugs and restoring fluorescence, enabling real-time monitoring and potent antibacterial action (>99.99% efficacy).43 Furthermore, advanced nanoplatforms combining arginine-functionalized CDs with bimetallic components demonstrate pH-responsive enzymatic activity—generating bactericidal ROS under acidic conditions while detoxifying excess radicals at neutral pH—thus synergizing photodynamic and chemodynamical therapies to resolve inflammation and promote tissue regeneration.99 These innovations highlight the potential of supra-CD-based materials for designing antibacterial coatings to prevent implant-related infections or formulating hydrogel dressings for managing infected wounds.In conclusion, the biomedical applications of supra-CDs are rapidly evolving from single-mode imaging or therapy towards integrated theragnostic platforms that combine multimodal imaging-guided synergistic therapy and real-time monitoring.100 These innovative applications fully demonstrate the great potential of supra-CDs as a new generation of nano biomaterials. In the future, with a deeper understanding of the assembly mechanisms and structure–activity relationships of supra-CDs, along with systematic evaluation of their biodistribution, metabolic pathways, and long-term safety, supra-CDs are expected to achieve breakthroughs in clinical translation, making important contributions to human health.
6. Conclusions and outlook
This review examines supra-CDs, focusing on their assembly, functional optimization, and biomedical uses. Supra-CDs overcome key limitations of individual CDs via covalent or non-covalent assembly. Size control to 50–100 nm reduces renal clearance and improves tumor targeting via the EPR effect. Assembly-induced J-aggregation, through-space conjugation, and restriction of intramolecular motion cause a fluorescence redshift to the NIR-II window, overcoming ACQ and enabling deep-tissue imaging and theragnostic. The dense structure enhances non-radiative transitions and electron transfer, boosting PTCE and ROS generation for photothermal and photodynamic therapy. Furthermore, supra-CDs act as versatile platforms for drug delivery, biosensing, and antibacterial applications, allowing controlled release via pH or enzyme triggers and showing utility in cancer therapy, infection control, and real-time monitoring. In summary, supra-CDs integrate nanoscale and macroscopic traits into a “diagnosis-therapy” platform, signalling a paradigm shift for carbon nanomaterials in biomedicine.101Despite significant advancements in supra-CDs, several challenges persist in their assembly, functional optimization, and biomedical applications. A key issue is the incomplete understanding of the precise assembly dynamics and structure–function relationships, which hinders the accurate prediction and control of their properties. Relying solely on intrinsic driving forces often leads to stochastic assembly processes, resulting in polydisperse structures with poor stability in physiological environments. Moreover, the lack of effective external regulation strategies makes it difficult to achieve controllable assembly under complex biological conditions, limiting their adaptability and performance in vivo. Furthermore, many of the precise external control methods, while excellent for achieving tailored structures and functions at the milligram to gram scale, may face significant challenges in terms of cost-effectiveness, batch-to-batch reproducibility, and process control when scaled to the kilogram levels required for clinical trials and eventual production.
Looking ahead, future research on supra-CDs should prioritize elucidating the fundamental mechanisms of assembly to establish clear structure–property correlations. Developing advanced modulation strategies, such as stimuli-responsive systems, could enable precise control over assembly in biological settings, enhancing stability and functionality.102–104 Integrating supra-CDs with emerging technologies like immunotherapy or gene-editing tools may lead to multifunctional theragnostic platforms for complex diseases. Furthermore, interdisciplinary collaborations will be crucial for conducting thorough preclinical studies on safety and metabolism, alongside exploring sustainable production routes, to accelerate the clinical translation of supra-CD-based innovations.
Author contributions
Xue Wu: writing – original draft, writing – review & editing, investigation, conceptualization. Yupeng Liu: writing – review & editing, funding acquisition. Caishi Huang: writing – review & editing. Songnan Qu: writing – review & editing, conceptualization, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was financially supported by the Science and Technology Development Fund of Macau SAR (0139/2022/A3 and 0002/2024/TFP), the National Natural Science Foundation of China (52502204), and Research Grant (MYRG-GRG2025-00148-IAPME and MYRG-CRG2025-00011-FHS), UM Postdoctoral Fellowship (Class A) from the University of Macau.References
- N. Feliu and W. J. Parak, Science, 2024, 384(6694), 385–386, DOI:10.1126/science.abq3711.
- J. O. Winter, Science, 2022, 377(6604), 371–372, DOI:10.1126/science.add3666.
- Y. Wang, Z. Xiao and Z. Wang, et al. , Nat. Rev. Bioeng., 2025, 3(4), 333–348, DOI:10.1038/s44222-024-00264-4.
- V. F. Gomerdinger, N. Nabar and P. T. Hammond, Nat. Rev. Cancer, 2025, 25(9), 657–683, DOI:10.1038/s41568-025-00847-2.
- P. Zhu, Y. Liu and Y. Tang, et al. , Chin. Chem. Lett., 2024, 4, 108689, DOI:10.1016/j.cclet.2023.108689.
- H. Song, Y. Ru and J. Yu, et al. , Sci. China Chem., 2026, 69(2), 539–618, DOI:10.1007/s11426-025-3195-y.
- B. Wang and S. Lu, Matter, 2022, 5(1), 110–149, DOI:10.1016/j.matt.2021.10.016.
- Y. Jiang, T. Zhao, W. Xu and Z. Peng, Carbon, 2024, 219, 118838, DOI:10.1016/j.carbon.2024.118838.
- L. Đorđević, F. Arcudi, M. Cacioppo and M. Prato, Nat. Nanotechnol., 2022, 17(2), 112–130, DOI:10.1038/s41565-021-01051-7.
- Y. Liu, H. Wang and S. Qu, Chin. Chem. Lett., 2025, 36(5), 110618, DOI:10.1016/j.cclet.2024.110618.
- Y. Ru, G. I. N. Waterhouse and S. Lu, Aggregate, 2022, 3(6), e296, DOI:10.1002/agt2.296.
- C. Kang, S. Tao, F. Yang and B. Yang, Aggregate, 2022, 3(2), e169, DOI:10.1002/agt2.169.
- L. L. Mokoloko, R. P. Forbes and N. J. Coville, Nanomaterials, 2022, 12(15), 2515, DOI:10.3390/nano12152515.
- L. Ai, Y. Pei and Z. Song, et al. , Adv. Sci., 2023, 10(12), 2207688, DOI:10.1002/advs.202207688.
- Z. Li, H. Cheng and B. Wang, et al. , J. Colloid Interface Sci., 2024, 668, 132–141, DOI:10.1016/j.jcis.2024.04.153.
- D. Guo, Y. Hou and Q. Xu, et al. , Light: Sci. Appl., 2025, 14(1), 163, DOI:10.1038/s41377-025-01834-w.
- D. Li, D. Han and S.-N. Qu, et al. , Light: Sci. Appl., 2016, 5(7), e16120, DOI:10.1038/lsa.2016.120.
- Q. Hou, C. Xue and N. Li, et al. , Carbon, 2019, 149, 556–563, DOI:10.1016/j.carbon.2019.04.083.
- F. Shan, L. Yan and Z. Wei, et al. , Nano Energy, 2025, 140, 111053, DOI:10.1016/j.nanoen.2025.111053.
- Q. Xia, Y. Zhang and H. Zhang, et al. , J. Mater. Chem. B, 2022, 10(39), 8046–8057, 10.1039/D2TB01363K.
- X. Gao, L. Wang, C. Sun and N. Zhou, Molecules, 2022, 27(5), 1690, DOI:10.3390/molecules27051690.
- Y. Xia, T. D. Nguyen and M. Yang, et al. , Nat. Nanotechnol., 2011, 6(9), 580–587, DOI:10.1038/nnano.2011.121.
- D. A. Sousa, M. N. Berberan-Santos and J. V. Prata, Chem. – Eur. J., 2024, 30(3), e202302955, DOI:10.1002/chem.202302955.
- X. Sun, H. Wang, J. Qi, S. Zhou and H. Li, Dyes Pigm., 2021, 190, 109287, DOI:10.1016/j.dyepig.2021.109287.
- F. Arcudi and L. Đorđević, Small, 2023, 19(31), 2300906, DOI:10.1002/smll.202300906.
- X. Cao, R. Lv and Y. Wei, Small, 2025, 21(29), 2501531, DOI:10.1002/smll.202501531.
- M. Li, Q. Shi, N. Song, Z. Chen, L. Wang and T. D. James, Adv. Opt. Mater., 2023, 11(15), 2203079, DOI:10.1002/adom.202203079.
- L. Li, D. Li and Y. Qu, et al. , Adv. Sci., 2025, 12(18), 2417457, DOI:10.1002/advs.202417457.
- C. Ji, F. Zeng and W. Xu, et al. , Adv. Mater., 2025, 37(4), 2414450, DOI:10.1002/adma.202414450.
- M. Cai, Y. Qiu, F. Li, S. Cai and Z. Cai, ACS Appl. Mater. Interfaces, 2024, 16(35), 46609–46618, DOI:10.1021/acsami.4c09567.
- P. K. Kundu, D. Samanta and R. Leizrowice, et al. , Nat. Chem., 2015, 7(8), 646–652, DOI:10.1038/nchem.2303.
- L. Ai, H. Wang, B. Wang, S. Liu, H. Song and S. Lu, Adv. Mater., 2024, 36(48), 2410094, DOI:10.1002/adma.202410094.
- Y. Wang, Y. Qin and F. Wang, et al. , Adv. Sci., 2024, 11(38), 2405043, DOI:10.1002/advs.202405043.
- F. Eweje, V. Ibrahim and A. Shajii, et al. , Nat. Biotechnol., 2025 DOI:10.1038/s41587-025-02664-2.
- S. Lu, R. Cong and S. Zhu, et al. , ACS Appl. Mater. Interfaces, 2016, 8(6), 4062–4068, DOI:10.1021/acsami.5b11579.
- T.-H. Chen and W.-L. Tseng, Anal. Chem., 2017, 89(21), 11348–11356, DOI:10.1021/acs.analchem.7b02193.
- J. Tan, C. Lu and X. Gu, et al. , Adv. Funct. Mater., 2025, e23830, DOI:10.1002/adfm.202523830.
- J.-L. Zhao, Q.-Y. Luo and Q. Ruan, et al. , Chem. Mater., 2021, 33(15), 6091–6098, DOI:10.1021/acs.chemmater.1c01620.
- Y. Shen, X. Zhang and L. Liang, et al. , Carbon, 2020, 156, 558–567, DOI:10.1016/j.carbon.2019.09.079.
- Y. Z. Yang, N. Xiao, S. G. Liu, L. Han, N. B. Li and H. Q. Luo, Mater. Sci. Eng., C, 2020, 108, 110401, DOI:10.1016/j.msec.2019.110401.
- J. Hao, W. Zhang and Y. Li, et al. , Adv. Sci., 2025, 12(27), 2503317, DOI:10.1002/advs.202503317.
- J. H. Swisher, L. Jibril, S. H. Petrosko and C. A. Mirkin, Nat. Rev. Mater., 2022, 7(6), 428–448, DOI:10.1038/s41578-021-00402-z.
- Z. Yu, C. Deng and L. Meng, et al. , Adv. Funct. Mater., 2025, e20327, DOI:10.1002/adfm.202520327.
- K. M. Chan, W. Xu, H. Kwon, A. M. Kietrys and E. T. Kool, J. Am. Chem. Soc., 2017, 139(37), 13147–13155, DOI:10.1021/jacs.7b07420.
- W. B. Rogers, W. M. Shih and V. N. Manoharan, Nat. Rev. Mater., 2016, 1(3), 16008, DOI:10.1038/natrevmats.2016.8.
- J. S. Kahn, B. Minevich and A. Michelson, et al. , Nat. Mater., 2025, 24(8), 1273–1282, DOI:10.1038/s41563-025-02263-1.
- C. Rossi-Gendron, F. El Fakih and L. Bourdon, et al. , Nat. Nanotechnol., 2023, 18(11), 1311–1318, DOI:10.1038/s41565-023-01468-2.
- R.-W. Song, C.-L. Shen and G.-S. Zheng, et al. , Nano Lett., 2023, 23(24), 11669–11677, DOI:10.1021/acs.nanolett.3c03529.
- S. Wu, L. Song and X. Zhu, et al. , Adv. Mater., 2025, e19060, DOI:10.1002/adma.202519060.
- S. Hu, Y. Ding and Q. Chang, et al. , Nanoscale, 2015, 7(10), 4372–4376, 10.1039/C4NR07119K.
- H. Wang, S. Liu and Z. Shen, et al. , Adv. Opt. Mater., 2025, 13(19), 2500408, DOI:10.1002/adom.202500408.
- L. Ai, W. Xiang and Z. W. Li, et al. , Angew. Chem., 2024, 136(49), e202410988, DOI:10.1002/anie.202410988.
- Z. Lin, W. Cheng, C. Liu, M. Zhao, S. Ding and Z. Deng, Talanta, 2023, 254, 124101, DOI:10.1016/j.talanta.2022.124101.
- X. Xue, J. Li and T. Chen, et al. , Chem. Eng. J., 2025, 505, 159356, DOI:10.1016/j.cej.2025.159356.
- Q. Jia, J. Ge and W. Liu, et al. , Adv. Mater., 2018, 30(13), 1706090, DOI:10.1002/adma.201706090.
- S. Guo, W. Liu and C. Lv, et al. , Adv. Funct. Mater., 2025, e23460, DOI:10.1002/adfm.202523460.
- P. S. Reddy, D. Balasaheb Kadam, S. Thakkellapati, M. Verma, A. M. Raichur and L. Narashimhan Ramana, Microchem. J., 2024, 198, 110144, DOI:10.1016/j.microc.2024.110144.
- Y. Liu, Z. Huang and X. Wang, et al. , Adv. Funct. Mater., 2024, 35(17), 2420587, DOI:10.1002/adfm.202420587.
- Y. Liu, H. Wang and S. Qu, Chin. Chem. Lett., 2024, 36, 110618, DOI:10.1016/j.cclet.2024.110618.
- T. Song, Y. Zhao, K. Matras-Postolek and P. Yang, J. Lumin., 2019, 212, 69–75, DOI:10.1016/j.jlumin.2019.04.025.
- J. Yang, L. Guo and X. Yong, et al. , Angew. Chem., Int. Ed., 2022, 61(33), e202207817, DOI:10.1002/anie.202207817.
- X. Huang, X. Jin and H. Bai, et al. , Adv. Mater., 2025, 37(28), 2418335, DOI:10.1002/adma.202418335.
- J. Zhang, M. Zheng, F. Zhang, B. Xu, W. Tian and Z. Xie, Chem. Mater., 2016, 28(23), 8825–8833, DOI:10.1021/acs.chemmater.6b04894.
- H. Yang, Y. Liu and Z. Guo, et al. , Nat. Commun., 2019, 10(1), 1789, DOI:10.1038/s41467-019-09830-6.
- S. Wu, H. Shi and W. Lu, et al. , Angew. Chem., Int. Ed., 2021, 60(40), 21890–21898, DOI:10.1002/anie.202107281.
- J. Zhou, M. Ge and Y. Han, et al. , ACS Omega, 2020, 5(20), 11842–11848, DOI:10.1021/acsomega.0c01527.
- S. Ding, S. Liu and Y. Yang, et al. , Sci. China Chem., 2025, 68(5), 1980–1990, DOI:10.1007/s11426-024-2308-3.
- J. Ren, L. Chen and J. Du, et al. , Chem. Eng. J., 2024, 502, 157825, DOI:10.1016/j.cej.2024.157825.
- Y. Zhang, L. Chen and B. Liu, et al. , Adv. Funct. Mater., 2024, 34, 2315366, DOI:10.1002/adfm.202315366.
- X. Kou, Y. Cong, W.-F. Dong and L. Li, Mater. Des., 2024, 240, 112855, DOI:10.1016/j.matdes.2024.112855.
- G. A. Alfani, Z. Cui, Z. Zhao, Z. Qiu, P. Alam and B. Z. Tang, Chem. Commun., 2025, 61(91), 17770–17786, 10.1039/D5CC04673D.
- F. Shan, J. Zhang and C. Liao, et al. , ACS Nano, 2025, 19(21), 20205–20214, DOI:10.1021/acsnano.5c05934.
- P. Choudhury and P. K. Das, Langmuir, 2019, 35(32), 10582–10595, DOI:10.1021/acs.langmuir.9b01631.
- N. Gao, W. Yang and H. Nie, et al. , Biosens. Bioelectron., 2017, 96, 300–307, DOI:10.1016/j.bios.2017.05.019.
- X. Wu, Y. Liu and B. Wang, et al. , Acta Phys.-Chim. Sin., 2025, 41, 100109, DOI:10.1016/j.actphy.2025.100109.
- Q. Duan, S. Si and S. Sang, et al. , Colloids Surf., A, 2022, 648, 129346, DOI:10.1016/j.colsurfa.2022.129346.
- J. Phipps, W. Fan, M. Kapoor, D. Huynh and S. Ma, Chem. Commun., 2026, 62, 3194–3209, 10.1039/D5CC06306J.
- N.-N. Chai, H.-X. Wang, C.-X. Hu, Q. Wang and H.-L. Zhang, J. Mater. Chem. A, 2015, 3(32), 16613–16620, 10.1039/C5TA03649F.
- Y. He, J. He and Z. Yu, et al. , J. Mater. Chem. C, 2018, 6(10), 2495–2501, 10.1039/C8TC00182K.
- C.-L. Shen, Q. Lou and K.-K. Liu, et al. , Carbon, 2023, 213, 118217, DOI:10.1016/j.carbon.2023.118217.
- X. Zhang, L. Li and B. Wang, et al. , Angew. Chem., Int. Ed., 2024, 63(49), e202410522, DOI:10.1002/anie.202410522.
- T. Chen, X. Xue and J. Li, et al. , Acta Phys.-Chim. Sin., 2025, 41, 100113, DOI:10.1016/j.actphy.2025.100113.
- S. W. Linderman, L. DeRidder and L. Sanjurjo, et al. , Nat. Rev. Clin. Oncol., 2025, 22(4), 262–282, DOI:10.1038/s41571-025-01000-6.
- Z. Yan, H. Cheng and Y. Liu, et al. , Adv. Funct. Mater., 2025, e22706, DOI:10.1002/adfm.202522706.
- C. Shen, T. Jiang and Q. Lou, et al. , SmartMat, 2022, 3(2), 269–285, DOI:10.1002/smm2.1099.
- Y. Zhou, K. J. Mintz and L. Cheng, et al. , J. Colloid Interface Sci., 2020, 576, 412–425, DOI:10.1016/j.jcis.2020.05.005.
- Z. Li, R. Zhao, Q. Pei, Z. Xie and M. Zheng, Adv. Sci., 2025, 12(33), e03883, DOI:10.1002/advs.202503883.
- T. Deng, R. Zhang and J. Wang, et al. , Part. Part. Syst. Charact., 2018, 35(9), 1800190, DOI:10.1002/ppsc.201800190.
- Q. Chen, X. Xia and Z. Liang, et al. , Small, 2024, 20(25), 2310728, DOI:10.1002/smll.202310728.
- S. Zong, J. Zhang, X. Yin, J. Li and S. Qu, Nano Lett., 2024, 24(6), 1859–1866, DOI:10.1021/acs.nanolett.3c03501.
- Y. Tang, P. Zhu, Q. Xu and J. Wang, et al. , Microchem. J., 2024, 201, 110536, DOI:10.1016/j.microc.2024.110536.
- G. Yi, Z. Tao, W. Fan, H. Zhou, Q. Zhuang and Y. Wang, ACS Sustainable Chem. Eng., 2024, 12(4), 1378–1387, DOI:10.1021/acssuschemeng.3c04729.
- B. Wang, Y. Chen and Y. Wu, et al. , Biosens. Bioelectron., 2016, 78, 23–30, DOI:10.1016/j.bios.2015.11.015.
- Y. Han, Y. Zhang and S. Wu, et al. , ACS Appl. Mater. Interfaces, 2021, 13(31), 36919–36925, DOI:10.1021/acsami.1c10326.
- J. Bai, G. Yuan and Z. Huang, et al. , Inorg. Chem. Commun., 2021, 126, 108500, DOI:10.1016/j.inoche.2021.108500.
- L. Yu, Q. Jiang and X. Yang, et al. , Microchim. Acta, 2025, 192(5), 282, DOI:10.1007/s00604-025-07119-8.
- L. Li, L. Chen and Y. Lu, et al. , Aggregate, 2023, 4(1), e200, DOI:10.1002/agt2.200.
- Z. Wang, P. Zhang and C. Yin, et al. , Adv. Funct. Mater., 2023, 33(29), 2300341, DOI:10.1002/adfm.202300341.
- N. Li, Y. Cao and W. Shi, et al. , Chem. Eng. J., 2025, 526, 170815, DOI:10.1016/j.cej.2025.170815.
- Z. Wang, J. Du and X. Leng, et al. , Adv. Funct. Mater., 2025, 35(50), e10476, DOI:10.1002/adfm.202510476.
- Q. Chen, X. Yang and Y. Yu, et al. , Small, 2025, 21(8), 2408980, DOI:10.1002/smll.202408980.
- A. Detappe and F. André, Nat. Rev. Bioeng., 2026, 4(1), 9–10, DOI:10.1038/s44222-025-00392-5.
- Y. Ren, Z. Zhou and I. Harley, et al. , Nat. Synth., 2025, 4(6), 673–683, DOI:10.1038/s44160-025-00751-5.
- C. Wang, M. Chen and M. Zhang, et al. , Nat. Biomed. Eng., 2025 DOI:10.1038/s41551-025-01560-z.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |





