Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Desaturative arylaldehyde synthesis by synergistic carbocatalysis and organocatalysis
Naokatsu Kannari*ab,
Ivan Curicb,
Nina Placeb,
Lukas Endersb,
Shotaro Kawasakia,
Anna Lenarda *b and
Juho Helaja
*b and
Juho Helaja *b
*b
aDivision of Materials and Environment, Graduate School of Science and Technology, Gunma University, 1-5-1, Tenjin-cho, Kiryu, Gunma 376-8515, Japan. E-mail: nkannari@gunma-u.ac.jp
bDepartment of Chemistry, University of Helsinki, A. I. Virtasen aukio 1, P.O. Box 55, Helsinki 00014, Finland. E-mail: anna.lenarda@helsinki.fi; juho.helaja@helsinki.fi
First published on 7th April 2026
Abstract
A metal-free cooperative carbocatalyst–organocatalyst system promotes the dehydrogenative aromatization of 3-cyclohexene aldehydes to aromatic aldehydes. Quinone-rich, oxidized activated carbon and secondary amines act synergistically, providing a structurally diverse substrate scope. Mechanistic studies indicate a predominantly ionic hydride-transfer pathway and identify pore-blocking as the main cause of catalyst deactivation.
Carbon materials are widely employed as supports for metal catalysts owing to their thermal robustness, chemical stability, and high surface areas. Beyond this role, appropriately engineered carbons can display intrinsic catalytic activity, particularly when modified through heteroatom doping.1–3 These materials, termed carbocatalysts, have emerged as sustainable and cost-effective alternatives to metal-based systems. Their activity is commonly associated with structural defects and heteroatom-containing functional groups at graphitic edges. Quinone-type moieties have been reported to play a central role in several oxidative dehydrogenation (ODH) processes ranging from industrial production of commodity chemicals4 (e.g. styrene from ethylbenzene) to liquid-phase coupling and aromatization reactions.1,5–7
Our group has previously developed oxygen-rich carbon materials for the ODH aromatization of unsaturated carbocycles and carboheterocycles to access (hetero)aryls6,7 and heterobiaryls.8
Aromatic aldehydes constitute a high-value class of compounds used extensively in pharmaceuticals, agrochemicals, fragrances, and fine chemicals.9 Conventional synthetic routes often rely on stoichiometric oxidants or precious metals, motivating the development of greener alternatives.10
Catalytic dehydrogenative aromatization represents a distinct desaturative approach in which redox-active enamines, generated from non-aromatic precursors, undergo sequential oxidation and dehydrogenation to afford functionalized arenes.11 In this context, Leonori and co-workers introduced a dual photoredox-cobalt catalytic method approach for the synthesis of anilines from cyclohexanone and amine building blocks.12 This work enabled the development of related enamine-aromatization approaches to aryl amines under electro-13 photo14 and Pd-15 catalysis conditions.
Building on this concept Lenarda et al.16 recently reported a carbocatalyzed condensation–oxidative aromatization cascade affording diarylamines from partially unsaturated cyclic ketones, demonstrating cooperative metal-free redox activity of quinones and other oxygenated surface functionalities on porous carbon.
Leonori et al. subsequently extended enamine-mediated desaturation to aldehydes trough a triple catalytic system combining Ir-based photoredox catalysis, organocatalytic enamine activation, and cobalt co-catalysis.17 In this system, enamine formation with a secondary amine (e.g., morpholine) enables single electron transfer (SET) oxidation followed by hydrogen atom transfer (HAT) dehydrogenation, mediated, respectively, by the photoredox and cobalt catalysts (Fig. 1).
 | ||
| Fig. 1 Dehydrogenative aromatization cascades of aldehydes utilizing enamine, Ir-photoredox, and cobalt vs. quinone-based redox carbocatalysis with proposed mechanism for the amine-mediated step. | ||
Guided by this insight, we envisioned that quinone-rich oxidized carbon materials could, in combination with a secondary amine co-catalyst, promote the ODH step of this reaction. We therefore initiated our study using HNO3-oxidized activated carbon (oACHNO3), prepared from commercial activated carbon (AC) following a protocol established in our laboratory,6 as the carbocatalyst. X-ray photoelectron spectroscopy (XPS) and temperature-programmed desorption (TPD) analyses confirmed the presence of abundant oxygenated functionalities, including quinone moieties, in agreement with previous reports (Fig. S1, S2, and Tables S1, S2), on a 635 m2 g−1 surface area characterized by an extended porous network (Table S9).6
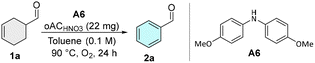
At first, we evaluated different secondary amines as organo-co-catalysts using the dehydrogenative aromatization of Cyclohex-3-ene-1-carbaldehyde (1a) as a probe reaction, in conditions previously optimized for oAC catalyzed ODH aromatization reactions.6
Morpholine (A1), previously employed in photoredox-mediated cyclohexene dehydrogenation,17 afforded only a marginal yield increase of 5% relative to the amine-free control (Table 1, entries 3 and 4), and phenoxazine (A2) and phenothiazine (A3) were similarly ineffective. In contrast, diphenylamine-derived catalysts showed markedly higher activity, with diphenylamine (A4), bis(4-methylphenyl)amine (A5), and bis(4-methoxyphenyl)amine (A6) delivering progressively higher yields, of 37%, 47% and 70% respectively. This trend suggests an enamine-mediated activation pathway in which more electron-rich diarylamines generate enamines better matched to the redox properties of the quinone sites on oACHNO3 (Fig. 1).
| Entry | Variation of reaction conditions | Yield of 2a (%) | Conversion of 1a (%) |
|---|---|---|---|
| 1 | None | 70 | 90 |
| 2 | no oACHNO3, A6 | 0 | 4 |
| 3 | oACHNO3, no A6 | 2 | 56 |
| 4 | A1 (morpholine) instead of A6 | 5 | 51 |
| 5 | A2 (phenoxazine) instead of A6 | 3 | 34 |
| 6 | A3 (phenothiazine) instead of A6 | 4 | 38 |
| 7 | A4 (diphenylamine) instead of A6 | 37 | 60 |
| 8 | A5 (bis(4-methylphenyl)amine) instead of A6 | 47 | 69 |
| 9 | ACdm as catalyst | 51 | 63 |
| 10 | AC as catalyst | 29 | 38 |
| 11 | HT-oACHNO3 as catalyst | 14 | 43 |
| 12 | HT-ACdm as catalyst | 9 | 23 |
| 13 | Anthraquinone (0.5 equiv.) as catalyst | 2 | 6 |
| 14 | PQ (0.5 equiv.) as catalyst | 7 | 17 |
After optimization of the reaction conditions (Table S4), the influence of AC surface oxidation on catalytic performance was examined using A6 as the amine co-catalyst (Table 1). HNO3-oxidized activated carbon (oACHNO3) afforded high aromatization efficiency (70% yield), whereas ACdm and untreated AC showed markedly lower activity (entries 9 and 10). This enhancement is consistent with the increased concentration of quinone-type functionalities introduced by nitric acid oxidation. Notably, untreated ACs retained moderate activity, likely due to native quinone groups inherently present in commercial activated carbons.18
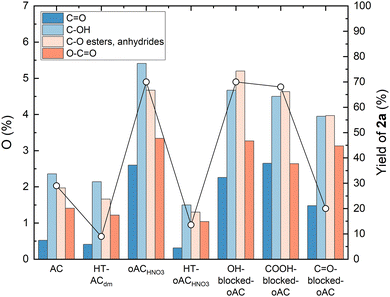
Thermal removal of oxygen functionalities by high-temperature treatment (HT-ACdm and HT-oACHNO3, entries 11 and 12) led to a pronounced loss of activity, corroborating the essential role of oxygenated active sites. In particular, XPS O1s deconvolution reveals a clear correlation between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O content and product yield (Fig. 2), suggesting the critical role of these functionalities in the catalytic process. In contrast, molecular quinones displayed only modest catalytic activity (entries 13 and 14), with 0.5 equiv. of 9,10-phenanthrenequinone (PQ) affording a remarkably low yield of 7%, only slightly higher than the one obtain in absence of catalyst (entry 2). These results suggest that efficient catalysis requires not only functional redox centres but also the presence of an extended, porous carbon framework, which appears to play a crucial electronic and structural role.
O content and product yield (Fig. 2), suggesting the critical role of these functionalities in the catalytic process. In contrast, molecular quinones displayed only modest catalytic activity (entries 13 and 14), with 0.5 equiv. of 9,10-phenanthrenequinone (PQ) affording a remarkably low yield of 7%, only slightly higher than the one obtain in absence of catalyst (entry 2). These results suggest that efficient catalysis requires not only functional redox centres but also the presence of an extended, porous carbon framework, which appears to play a crucial electronic and structural role.
While the correlation presented in Fig. 2 suggests that quinone sites on the carbon play a crucial role in the catalytic activity, it is acknowledged that XPS peak deconvolution inherently involves overlapping components. Therefore, The role of different functional groups as the catalytically active sites was further probed through selective surface-group blocking experiments, using previously reported procedures (Fig. S3).16 Blocking –OH and –COOH groups had a negligible effect on catalytic performance, affording product yields of 70% and 68%, respectively, comparable to that obtained with untreated oACHNO3 (70%). In contrast, selective masking of carbonyl groups led to a sharp decrease in activity, with the yield dropping to 20%. XPS analysis confirmed the selective modification of the targeted oxygen functionalities (Fig. 2 and Tables S5, S6, Fig. S4). These results provide further substance to our hypothesis of quinone groups being the active sites responsible for dehydrogenative aromatization catalysis.
With the catalytic system established, we next examined the substrate scope for the dehydrogenative aromatization of cyclohexene aldehydes (Fig. 3). Yields are reported as isolated unless otherwise stated, subject to purification constraints. Under the standard conditions (toluene, 90 °C), 2,4-dimethylcyclohex-3-ene-1-carbaldehyde (2b) afforded the aromatic aldehyde in 36% yield after 24 h, indicating modest steric inhibition relative to 1a. In contrast, aryl-fused substrates were efficiently converted: the biphenyl-derived aldehyde 2c was obtained in 89% yield after 48 h, while a naphthyl analogue 2d furnished 79% yield under identical conditions. Both electron-withdrawing and electron-donating substituents were tolerated, with 4′-CF3 (77%, 2e), 3′,4′-dimethoxy (89%, 2f), and 3′,5′-dimethyl (49% after 72 h, 2g) derivatives providing the corresponding aldehydes. Strongly electron-donating substituents led to diminished performance, as observed for the 4′-dimethylamino substrate (49%, 2h). A heteroaromatic thiophene-containing substrate was also proved compatible, affording 4-(thiophen-3-yl)benzaldehyde 2i in 53% yield. Overall, the reaction tolerates a broad range of electronic environments, with highest efficiency observed for biphenyl-derived systems, consistent with an enamine oxidation pathway modulated by substrate electronics.
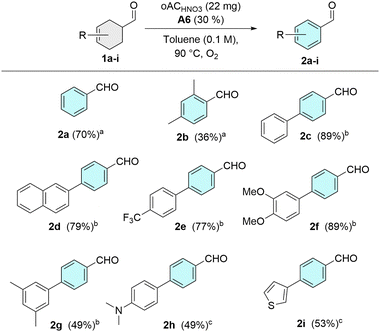 | ||
| Fig. 3 a24 h, GC yield, b48 h, isolated yield, c48 h, NMR yield. Scope study for the aromatization of cyclohexene-3-aldehydes. | ||
The durability of the carbocatalyst was evaluated through recycling experiments using 1a as a model substrate. Activity progressively decreased over successive cycles, indicating catalyst deactivation (Fig. S5a). Amine recovery experiments showed significant retention of the secondary amine on the catalyst, suggesting surface adsorption as a primary deactivation pathway (Fig. S5b). Consistently, N2 physisorption revealed loss of surface area and microporosity upon reuse (Fig. S7 and Table S9), consistent with pore blocking by adsorbed amines.
Post-reaction XPS analysis showed no significant changes in overall surface composition were observed, although a slight decrease in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O content was detected (Tables S7 and S8). Combined with the BET data, these results are consistent with reversible deactivation via adsorption of amine species on active sites and within the porous structure.
O content was detected (Tables S7 and S8). Combined with the BET data, these results are consistent with reversible deactivation via adsorption of amine species on active sites and within the porous structure.
Importantly, catalyst deactivation was found to be reversible; washing the spent material with acetic acid restores catalytic activity and textural properties close of the fresh catalyst (Fig. S6 and Table S7). These indicates that deactivation primarily arises from reversible amine adsorption, which blocks quinone active sites and the porous carbon framework.
Additional evidence for pore-blocking-induced deactivation was obtained from catalytic tests in the presence of pyrene, employed as an inert polycyclic aromatic probe to occupy pore volume. Consistent with prior reports,16 pyrene was selected for its strong π–π affinity for carbon surfaces and its ability to physically reside within micropores without participating in the reaction. The addition of two equivalents of pyrene reduced the yield to 53%, consistent with progressive pore occupation limiting access of the substrate, intermediates, and organocatalyst. Similar experiments performed under Ar in presence of non-aromatic probes resulted in lowered yields (see SI).
The critical role of porosity was further supported by experiment using reduced graphene oxide (rGO), a commonly reported carbocatalyst for oxidative dehydrogenation.3,16 Despite its high oxygen content, rGO afforded only 1% yield under the optimized conditions, indicating that surface oxygen functionalities alone are insufficient for efficient catalysis (see SI). Together with the pyrene-blocking and recycling studies, these results suggest pore accessibility as critical for catalytic activity.
Collectively, these results indicate that quinone active sites and an accessible porous carbon framework must operate cooperatively to enable efficient enamine oxidation and aromatization.
PQ is frequently employed as a molecular model for quinone-mediated dehydrogenative carbocatalysis on oxidized carbon materials.19
To gain theoretical insight, DFT calculations were performed on the dehydrogenative aromatization of enamines 1a-A4 and 1a-A6 mediated by PQ (Table S11 and Fig. S8). The reaction proceeds via an initial hydride abstraction from the doubly allylic C2 position, followed by deprotonation and a second hydride abstraction to afford the aromatic aldehyde. However, the computed barriers for the first hydride abstraction are relatively high (30.0 and 29.0 kcal mol−1), indicating that PQ alone does not fully capture the experimentally observed reactivity of oACHNO3.
To refine the model and better account for the extended π-conjugation effects, coronenequinone (CQ) was next examined (Table S12 and Fig. S10). Although the reaction mechanism remains unchanged, the larger π-system lowers the initial activation barriers to 24.5 and 21.7 kcal mol−1. Incorporation of a carboxylic acid group onto CQ (CQCA), mimicking nitric acid–oxidized carbon, further reduces these values to 23.6 and 19.9 kcal mol−1 (Fig. 4), and accurately reproduces the experimental enamine reactivity trend. Notably, the CO2H substituent acts as an electron withdrawing group, rendering the π-surface of CACQ more electron-deficient and thereby strengthening its interaction with the enamine.
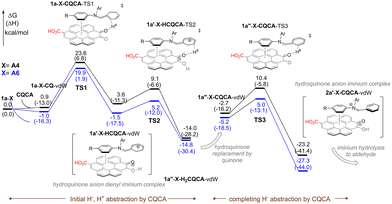 | ||
| Fig. 4 Gibbs free-energy profiles for CQCA-mediated aromatization of enamines 1a-A4 and 1a-A6. M06-2X/6-311++G(d,p)//M06-2X/6-31+G(d,p), CPCM(toluene), GD3. | ||
Noncovalent interaction (NCI) analysis reveals stabilizing π–π interactions between the enamine and the CQ–CO2H surface, while HOMO analysis indicates greater delocalization for the more reactive 1a-A6 system (see SI, NCI plots section). Moreover, DFT calculations predict that the morpholine-derived enamine (1a-Morp) is similarly reactive to 1a-A4 (Fig. S9 and S11); its poor experimental performance likely arises from inefficient enamine formation under the acidic oACHNO3 carbocatalyst conditions. This interpretation is supported by its strong H-bonding interaction with the CO2H site of model compound CACQ (see Fig. S12 and Table S15).
Although TEMPO-like radical traps substantially suppress reactivity (Table S10), computational analysis shows that SET and HAT pathways between enamines and quinone models are thermodynamically unfavourable (Tables S13 and S14), suggesting a predominantly ionic hydride-transfer mechanism,20 with radical species contributing only to catalyst regeneration, as supported by the moderate yield obtained in the tests performed under Ar atmosphere (see Table S4).21 Nevertheless, based on computations with model compounds alone, a radical component in the catalytic cycle cannot be fully excluded.
In summary, we report a metal-free, cooperative carbocatalyst–organocatalyst strategy for the dehydrogenative aromatization of cyclohexene aldehydes to aromatic aldehydes under mild conditions. Quinone-rich oxidized activated carbon acts as the redox-active catalyst, while secondary amines enable formation of reactive enamine intermediates. The method displays a diverse substrate scope, efficiently converting aryl-fused, biphenyl-derived, heteroaromatic cyclohexene aldehydes bearing both electron-donating and electron-withdrawing substituent. Experimental and computational studies suggest quinone surface sites and carbon porosity as key determinants of activity, with catalyst deactivation likely arising from reversible pore blocking by amines. Mechanistic investigations support a predominant ionic hydride-transfer pathway. Overall, this work expands the scope of carbocatalysis and underscores the potential cooperative catalyst design in sustainable oxidation chemistry.
N. K.: writing – original draft preparation, conceptualization, investigation (catalyst development and catalyst testing), analysis of results, resources and funding acquisition, I. C.: investigation (synthetic work and catalyst testing), N. P.: investigation (synthetic work), L. E.: investigation, methodology (synthetic work), S. K.: investigation (TPD measurement), A. L.: methodology (catalysis testing), analysis of results, writing – review and editing, visualization, J. H.: investigation (DFT calculation), writing – review and editing, project administration, resources and funding acquisition.
Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information: additional catalytic experiments; synthetic procedures; product characterization (NMR including copies of relevant spectra); additional XPS; BET data and spectra; computational details; XYZ parameters. See DOI: https://doi.org/10.1039/d6cc00868b.Acknowledgements
This study was supported in part by the Uehara Memorial Foundation, the Scandinavia-Japan Sasakawa Foundation, and the Kondo Memorial Foundation. Financial support from the Academy of Finland [project no. 356338 (J. H.)] is acknowledged. The Finnish National Centre for Scientific Computing (CSC) is recognized for computational resources. XPS measurements were performed at the Core Facility Management and Technical Collaboration Center (CoMTeCC) of Gunma University.References
- (a) D. S. Su, G. Wen, S. Wu, F. Peng and R. Schlögl, Angew. Chem., Int. Ed., 2017, 56, 936–964 CrossRef CAS PubMed; (b) D. R. Dreyer and C. W. Bielawski, Chem. Sci., 2011, 2, 1233–1240 RSC; (c) D. R. Dreyer, A. D. Todd and C. W. Bielawski, Chem. Soc. Rev., 2014, 43, 5288–5301 RSC; (d) H. Jia, D. R. Dreyer and C. W. Bielawski, Tetrahedron, 2011, 67, 4431 CrossRef CAS; (e) D. R. Dreyer, H. Jia and C. W. Bielawski, Angew. Chem., Int. Ed., 2010, 49, 6813 CrossRef CAS; (f) P. Tang, G. Hu, M. Li and D. Ma, ACS Catal., 2016, 6, 6948–6958 CrossRef CAS; (g) M. S. Ahmadab and Y. Nishina, Nanoscale, 2020, 12, 12210–12227 RSC; (h) D. S. Su, J. Zhang, B. Frank, A. Thomas, X. Wang, J. Paraknowitsch and R. Schlögl, ChemSusChem, 2010, 3, 169–180 CrossRef CAS.
- (a) D. Yu, E. Nagelli, F. Du and L. Dai, J. Phys. Chem. Lett., 2010, 1, 2165–2173 CrossRef CAS; (b) J. Zhang, D. Su, A. Zhang, D. Wang, R. Schlögl and C. Hébert, Angew. Chem., Int. Ed., 2007, 46, 7319–7323 CrossRef CAS PubMed; (c) D. S. Su, S. Perathoner and G. Centi, Chem. Rev., 2013, 113, 5782–5816 CrossRef CAS PubMed; (d) R. P. Rocha and J. L. Figueiredo, Catalysts, 2025, 15, 443 CrossRef CAS; (e) S. Navalon, A. Dhakshinamoorthy, M. Alvaro, M. Antonietti and H. García, Chem. Rev., 2017, 46, 4501–4529 CAS; (f) J. Zhou, P. Yang, P. A. Kots, M. Cohen, Y. Chen, C. M. Quinn, M. D. de Mello, J. Anibal Boscoboinik, W. J. Shaw, S. Caratzoulas, W. Zheng and D. G. Vlachos, Nat. Commun., 2023, 14, 2293 CrossRef CAS PubMed.
- (a) S. Navalon, A. Dhakshinamoorthy, M. Alvaro and H. Garcia, Chem. Rev., 2014, 114, 6179–6212 CrossRef CAS; (b) S. C. Wu, L. H. Yu, G. D. Wen, Z. L. Xie and Y. M. Lin, J. Energy Chem., 2021, 58, 318–335 CrossRef CAS.
- J. Zhang, X. Liu, R. Blume, A. Zhang, R. Schlögl and D. S. Su, Science, 2008, 322, 73–77 CrossRef CAS PubMed.
- (a) D. S. Casadio, S. Aikonen, A. Lenarda, M. Nieger, T. Hu, S. Taubert, D. Sundholm, M. Muuronen, T. Wirtanen and J. Helaja, Chem. – Eur. J., 2021, 27, 5283–5291 CrossRef CAS; (b) T. Wirtanen, S. Aikonen, M. Muuronen, M. Melchionna, M. Kemell, F. Davodi, T. Kallio, T. Hu and J. Helaja, Chem. – Eur. J., 2019, 25, 12288–12293 CrossRef CAS PubMed; (c) T. Wirtanen, M. K. Mäkelä, J. Sarfraz, P. Ihalainen, S. Hietala, M. Melchionna and J. Helaja, Adv. Synth. Catal., 2015, 357, 3718–3726 CrossRef CAS; (d) A. Lenarda, T. Wirtanen and J. Helaja, Synthesis, 2023, 45–61 CrossRef CAS; (e) S. Wu, L. Yu, G. Wen, Z. Xie and Y. Lin, J. Energy Chem., 2021, 58, 318–335 CrossRef CAS; (f) H. Jung and C. W. Bielawski, RSC Adv., 2020, 10, 15598–15603 RSC; (g) H. Jia, D. R. Dreyer and C. W. Bielawski, Adv. Synth. Catal., 2011, 353, 528 CrossRef CAS; (h) W. Qi and D. Su, ACS Catal., 2014, 4, 3212–3218 CrossRef CAS.
- L. Enders, D. S. Casadio, S. Aikonen, A. Lenarda, T. Wirtanen, T. Hu, S. Hietala, L. S. Ribeiro, M. F. R. Pereira and J. Helaja, Catal. Sci. Technol., 2021, 11, 5962–5972 RSC.
- A. Lenarda, M. Melchionna, S. Aikonen, T. Montini, P. Fornasiero, T. Hu, M. Hummel and J. Helaja, ACS Catal., 2023, 13, 11362–11375 CrossRef CAS.
- M. Yang, A. Lenarda, S. Frindy, Y. Sang, V. Oksanen, A. Bolognani, L. Hendrickx, J. Helaja and Y. Li, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2303564120 CrossRef CAS.
- (a) O. I. Afanasyev, E. Kuchuk, D. L. Usanov and D. Chusov, Chem. Rev., 2019, 119, 11857–11911 CrossRef CAS PubMed; (b) S. Caron, R. W. Dugger, S. G. Ruggeri, J. A. Ragan and D. H. B. Ripin, Chem. Rev., 2006, 106, 2943–2989 CrossRef CAS PubMed; (c) J. Magano and J. R. Dunetz, Org. Process Res. Dev., 2012, 16, 1156–1184 CrossRef CAS.
- (a) J. Cossy and D. Belotti, Org. Lett., 2002, 4, 2557–2559 CrossRef CAS PubMed; (b) X. Jie, Y. Shang, Z.-N. Chen, X. Zhang, W. Zhuang and W. Su, Nat. Commun., 2018, 9, 5002 CrossRef PubMed; (c) S. Paganelli, R. Tassini and O. Piccolo, ChemistrySelect, 2022, 7, e202202393 CrossRef CAS.
- S. A. Girard, H. Huang, F. Zhou, G.-J. Deng and C.-J. Li, Org. Chem. Front., 2015, 2, 279–287 RSC.
- S. U. Dighe, F. Juliá, A. Luridiana, J. J. Douglas and D. Leonori, Nature, 2020, 584, 75–81 CrossRef PubMed.
- (a) S.-K. Tao, S.-Y. Chen, M.-L. Feng, J.-Q. Xu, M.-L. Yuan, H.-Y. Fu, R.-X. Li, H. Chen, X.-L. Zheng and X.-Q. Yu, Org. Lett., 2022, 24, 1011–1016 CrossRef CAS PubMed; (b) J. Hu, R. Ma, J. Hu, X. Liu, X. Liu, H. He, H. Yi and A. Lei, Green Chem., 2024, 26, 4684–4690 RSC.
- (a) J. Corpas, H. P. Caldora, E. M. Di Tommaso, A. C. Hernandez-Perez, O. Turner, L. M. Azofra, A. Ruffoni and D. Leonori, Nat. Catal., 2024, 7, 593–603 CrossRef CAS; (b) J. Kim, S. Kim, G. Choi, G. S. Lee, D. Kim, J. Choi, H. Ihee and S. H. Hong, Chem. Sci., 2020, 12, 1915–1923 RSC.
- (a) W.-C. Lin, T. Yatabe and K. Yamaguchi, Chem. Lett., 2025, 54, upaf043 CrossRef CAS; (b) F. Chen, H. Geng, C. Li, J. Wang, B. Guo, L. Tang and Y.-Y. Yang, J. Org. Chem., 2023, 88, 15589–15596 CrossRef CAS PubMed.
- A. Lenarda, I. Jain, A. Kaleva, V. Oksanen, S. Heikkinen, R. Koivula, T. Wirtanen, M. Melchionna, T. Hu and J. Helaja, Green Chem., 2026, 28, 199–212 RSC.
- H. Zhao, H. P. Caldora, O. Turner, J. J. Douglas and D. Leonori, Angew. Chem., Int. Ed., 2022, 61, e202201870 CrossRef CAS PubMed.
- J. Figueiredo, M. Pereira, M. Freitas and J. Órfão, Carbon, 1999, 37, 1379–1389 CrossRef CAS.
- W. Guo, L. Yu, L. Tang, Y. Wan and Y. Lin, Nanomicro Lett., 2024, 16, 125 Search PubMed.
- A. E. Wendlandt and S. S. Stahl, Angew. Chem., Int. Ed., 2015, 54, 14638–14658 CrossRef CAS PubMed.
- (a) G.-F. Han, F. Li, W. Zou, M. Karamad, J.-P. Jeon, S.-W. Kim, S.-J. Kim, Y. Bu, Z. Fu, Y. Lu, S. Siahrostami and J.-B. Baek, Nat. Commun., 2020, 11, 2209 CrossRef CAS PubMed; (b) G. Lv, H. Wang, Y. Yang, T. Deng, C. Chen, Y. Zhu and X. Hou, ACS Catal., 2015, 5, 5636–5646 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |