Open Access Article
Open Access ArticleSelective etching of binary nanoparticle superlattices via thermally induced asymmetric ligand evolution
Di Leia,
Hao Wanga,
Ting Wanga,
Fangyue Wua,
Ning Dinga,
Yajun Wang*b,
Tongtao Li*a and
Angang Dong *a
*a
aDepartment of Chemistry and Shanghai Key Laboratory of Molecular Catalysis and Innovative Materials, Fudan University, Shanghai 200433, China. E-mail: ttli@fudan.edu.cn; agdong@fudan.edu.cn
bCollege of Chemistry and Materials Engineering, Wenzhou University, Wenzhou 325027, China. E-mail: yajunwang@wzu.edu.cn
First published on 3rd March 2026
Abstract
We report a method to selectively etch either component in NaYF4/Fe3O4 binary nanoparticle superlattices using a single etchant. Thermally-induced evolution of oleate ligands on the NaYF4 nanoparticles reverses their relative etching susceptibility in oxalic acid, yielding non-close-packed nanoparticle arrays with tunable architectures.
The self-assembly of colloidal nanoparticles (NPs) provides a powerful bottom-up route to organize nanoscale building blocks into ordered superlattices with collective and emergent properties. Such superlattices have attracted intense interest for applications in electronic, optical, and mechanical properties, where long-range periodicity and structural coherence are essential.1–3 Importantly, the properties of these materials can be precisely tuned by the choice and number of NP building blocks, the length and functionality of the surface ligands, and the symmetry that governs their coupling within the lattice.4
Beyond conventional close-packed architectures, non-close-packed (NCP) ordered arrays—in which NPs remain periodically arranged but are separated by larger, well-defined distances—offer distinct advantages, including enhanced accessibility, porosity and electromagnetic field concentration. These features make NCP arrays particularly attractive as surface-enhanced Raman scattering (SERS) substrates5 and as templates for quantum dot or plasmonic device fabrication.6 Despite several elegant demonstrations based on highly specific interactions or finely balanced assembly conditions,7 a general and programmable strategy to access ordered NCP NP arrays is still lacking.
Selective removal of one component from binary NP superlattices (BNSLs) by chemical etching represents a particularly powerful route to transform dense arrays into ordered NCP architectures. In practice, however, most reported strategies rely on the intrinsic chemical differences between the two components and require the use of different etchants to achieve selectivity.5,8 A representative example is reported in Fe3O4/Au BNSLs, where Fe3O4 NPs are etched by acids while Au NPs require a cyanide solution such as KCN.5 Although effective, this etchant-dependent paradigm is inherently complex, limits programmability, and offers little insight into the molecular origin of etching selectivity. A simpler and mechanistically transparent strategy that enables selective etching using a single etchant—while retaining full structural order—therefore remains highly desirable for post-assembly structural editing.
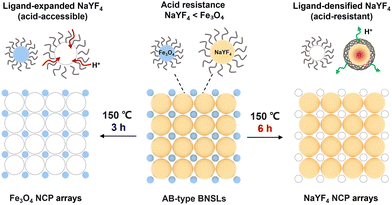
Here we introduce an alternative, ligand reorganization–governed strategy for selective etching of BNSLs using a single etchant. Using a NaYF4/Fe3O4 system, we exploit the thermally induced chemical transformation and crosslinking of oleate ligands on NaYF4 NPs, enabled by their low thermal conductivity and nanoscale heat accumulation under thermal treatment.9 As a result, the ligand shell on NaYF4 evolves from a loosely bound molecular layer into a rigid, interconnected network. When the degree of ligand crosslinking is low, NaYF4 remains susceptible to etching in oxalic acid; when crosslinking is sufficiently developed, acid penetration is effectively suppressed. In contrast, the ligand shell on the Fe3O4 NPs remains largely unchanged under the same conditions. This difference allows the relative etching susceptibility of the two components to be reversed simply by tuning the thermal treatment time, without changing the etching chemistry. Spectroscopic analyses reveal a molecular-level reconstruction of the ligand shell as the origin of this behavior, establishing a new conceptual basis for programmable post-assembly structural editing of nanocrystal superlattices.
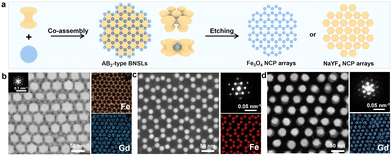
Monodisperse NaYF4 and Fe3O4 nanospheres (NSs) were synthesized following established protocols.10 For constructing AB-type BNSLs, Fe3O4 NSs (diameter: 13.1 ± 0.3 nm) and NaYF4 NSs (diameter: 33.7 ± 0.1 nm) were co-assembled at the liquid–air interface via slow evaporation of hexane (Fig. S1a and c).11 This process yields two-dimensional membranes with long-range order, which can be readily transferred onto carbon-coated transmission electron microscopy (TEM) grids for structural characterization and post-assembly processing (Fig. 1a). Low-magnification TEM images reveal long-range periodic ordering of the AB-type superlattice (Fig. 1b and Fig. S3), while the corresponding small-angle electron diffraction (SAED) pattern confirms crystalline coherence across the entire lattice. High-angle annular dark-field scanning TEM (HAADF-STEM) images combined with elemental mapping unambiguously assign Fe and Y to two interpenetrating sublattices, verifying the formation of a binary array (Fig. 1c).
By adjusting the thermal treatment time prior to etching, the target of oxalic acid etching can be reversibly switched. After a short duration of thermal treatment (3 h), NaYF4 is preferentially removed due to its intrinsically lower acid resistance compared with Fe3O4 (Fig. S4), yielding an NCP Fe3O4 array. Large-area TEM images show a periodic square array (Fig. 1d), while HAADF-STEM and Fe/O elemental mapping confirm the exclusive retention of Fe3O4 in the remaining lattice (Fig. 1e). The average diagonal center-to-center spacing is 36.6 nm (Fig. S5), consistent with the diameter of the removed NaYF4 NSs plus the oleate ligand shell. Both the particle size and lattice registry of Fe3O4 remain unchanged, indicating that oxalic acid selectively removes NaYF4 without disrupting the Fe3O4 sublattice.
Conversely, extending the thermal treatment to 6 h reverses the etching selectivity. Under identical oxalic acid conditions, Fe3O4 NSs are now preferentially removed, while NaYF4 NSs remain intact, producing a complementary NCP NaYF4 array. Low-magnification TEM images confirm the formation of an ordered NaYF4 array (Fig. 1f and Fig. S6), and HAADF-STEM with Y/F elemental mapping verifies the exclusive presence of NaYF4 NSs in the remaining lattice (Fig. 1g). Notably, the etching process is not instantaneous but evolves progressively with time: statistical TEM analysis after 3 h and 6 h of thermal treatment reveals a continuous, stepwise removal of the susceptible component until complete elimination is achieved (Fig. S7). This inversion of etching susceptibility establishes an “alternative etching” regime governed solely by the thermal treatment time of the superlattice, rather than by changing the chemical identity of the etchant.
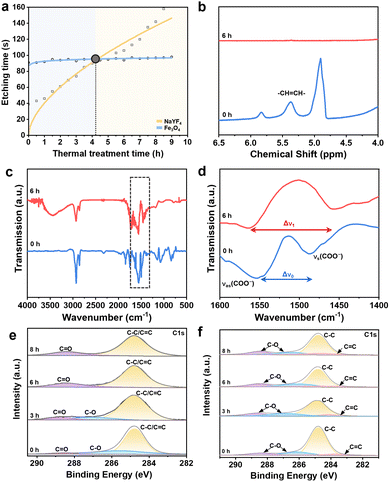
To rationalize the “alternative etching” behavior observed in Fig. 1, we quantified the time required for complete removal of NaYF4 and Fe3O4 NSs as a function of prior thermal treatment (Fig. 2a). The etching kinetics of Fe3O4 are largely insensitive to the thermal treatment time, whereas the etching kinetics of NaYF4 increase systematically with increasing thermal treatment duration. The plot reveals a crossover at 4 h, where the relative etching susceptibility of the two components is reversed. Without thermal treatment, NaYF4 is etched much faster than Fe3O4 and the superlattice collapses during etching (Fig. S4). A moderate treatment of 3 h stabilizes the lattice while maintaining preferential removal of NaYF4, yielding intact Fe3O4 NCP arrays. Extending the thermal treatment to 6 h drives the system beyond the crossover, where Fe3O4 is preferentially etched and the NaYF4 sublattice remains intact. This inversion originates from the intrinsic difference in thermal conductivity between the two cores, which leads to programmable modification of the surface states. We therefore probe the molecular origin of this behavior by examining how thermal treatment reconstructs the ligand shells of both components.
To elucidate the molecular origin of the thermally induced etching reversal, we first investigated the evolution of surface ligands on NaYF4 NSs by 1H nuclear magnetic resonance (NMR) spectroscopy. The ligand structures were analyzed using single-component NaYF4 samples subjected to different thermal treatments (0 h and 6 h), with spectra acquired in CDCl3 after redispersing the thermally treated NSs from silicon substrates (Fig. 2b and Fig. S8). The 1H NMR spectrum of the as-prepared NaYF4 NSs shows characteristic signals of oleate ligands. After 6 h thermal treatment, the alkene resonance at δ = 5.34 ppm, assigned to –HC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH– protons, disappears completely, indicating cleavage of the C
CH– protons, disappears completely, indicating cleavage of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in surface-bound oleate.12,13 This transformation enables ligand crosslinking and shell densification, forming a compact surface layer that suppresses acid penetration and underlies the enhanced etching resistance.
C bond in surface-bound oleate.12,13 This transformation enables ligand crosslinking and shell densification, forming a compact surface layer that suppresses acid penetration and underlies the enhanced etching resistance.
Fourier transform infrared (FTIR) spectroscopy reveals a pronounced reorganization of the ligand shell on NaYF4 NSs upon thermal treatment. As shown in Fig. 2c and d, pristine NaYF4 NSs exhibit characteristic oleate features, including CH2 stretching bands at 2924 and 2854 cm−1 and a weak ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H vibration at 3007 cm−1, consistent with surface-bound oleic acid ligands. After thermal treatment, the most pronounced changes occur in the carboxylate region. In the pristine sample, the bands at 1550 and 1485 cm−1 are associated with the asymmetric (νas) and symmetric (νs) stretching vibrations of the carboxylic group of the bound oleic acid. After thermal treatment, these bands shift to 1562 and 1457 cm−1. Accordingly, the separation Δν = νas − νs increases from 65 cm−1 to 105 cm−1.14–16 Fig. S9 shows the FTIR spectra of NaYF4 NSs following thermal treatment for different durations (0, 3, 6 and 8 h). With increasing annealing time, the νas band gradually shifts to higher wavenumbers, whereas the νs band moves to lower wavenumbers, leading to a progressive increase in Δν. Such an increase in Δν is commonly associated with a change in the carboxylate coordination environment, consistent with a transition from relatively flexible surface binding to a more compact and constrained coordination state. This evolution suggests that thermal treatment induces chemical reorganization and intermolecular coupling of oleate ligands on the NaYF4 surface.
C–H vibration at 3007 cm−1, consistent with surface-bound oleic acid ligands. After thermal treatment, the most pronounced changes occur in the carboxylate region. In the pristine sample, the bands at 1550 and 1485 cm−1 are associated with the asymmetric (νas) and symmetric (νs) stretching vibrations of the carboxylic group of the bound oleic acid. After thermal treatment, these bands shift to 1562 and 1457 cm−1. Accordingly, the separation Δν = νas − νs increases from 65 cm−1 to 105 cm−1.14–16 Fig. S9 shows the FTIR spectra of NaYF4 NSs following thermal treatment for different durations (0, 3, 6 and 8 h). With increasing annealing time, the νas band gradually shifts to higher wavenumbers, whereas the νs band moves to lower wavenumbers, leading to a progressive increase in Δν. Such an increase in Δν is commonly associated with a change in the carboxylate coordination environment, consistent with a transition from relatively flexible surface binding to a more compact and constrained coordination state. This evolution suggests that thermal treatment induces chemical reorganization and intermolecular coupling of oleate ligands on the NaYF4 surface.
X-ray photoelectron spectroscopy (XPS) provides direct evidence for thermally induced ligand reconstruction on NaYF4 NSs and highlights its contrast with Fe3O4 NSs. In the C 1s spectra of NaYF4 (Fig. 2e), thermal treatment induces a systematic decrease of the C–O component accompanied by an increase of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O contribution, which supports the formation of a more chemically constrained surface layer that correlates with the enhanced acid resistance of NaYF4 after thermal treatment. In contrast, the C 1s spectra of Fe3O4 NSs (Fig. 2f) show no signatures of ligand crosslinking under the same thermal treatment. Simultaneously, the Fe 2p3/2 and Fe 2p1/2 peaks (Fig. S10c) shift slightly toward higher binding energies after thermal treatment, indicating partial oxidation of Fe2+ to Fe3+.3,6 Thus, Fe3O4 primarily undergoes core-level oxidation rather than surface–ligand reconstruction. This stark difference demonstrates that only NaYF4 experiences thermally driven ligand reorganization and densification, providing the molecular basis for the programmable and reversible etching selectivity observed in the binary superlattices.
O contribution, which supports the formation of a more chemically constrained surface layer that correlates with the enhanced acid resistance of NaYF4 after thermal treatment. In contrast, the C 1s spectra of Fe3O4 NSs (Fig. 2f) show no signatures of ligand crosslinking under the same thermal treatment. Simultaneously, the Fe 2p3/2 and Fe 2p1/2 peaks (Fig. S10c) shift slightly toward higher binding energies after thermal treatment, indicating partial oxidation of Fe2+ to Fe3+.3,6 Thus, Fe3O4 primarily undergoes core-level oxidation rather than surface–ligand reconstruction. This stark difference demonstrates that only NaYF4 experiences thermally driven ligand reorganization and densification, providing the molecular basis for the programmable and reversible etching selectivity observed in the binary superlattices.
The spectroscopic evidence and etching trends in Fig. 2 converge on a unified mechanism: nanoscale heat accumulation in NaYF4 NSs triggers ligand intermolecular crosslinking and densification of the oleate shell, transforming a permeable organic corona into a compact barrier against acid attack.9 The surface ligands on the Fe3O4 NSs, in contrast, do not undergo comparable reconstruction under the same thermal conditions and remain relatively accessible to the etchant.3,6 The thermal conductivity of NaYF4 (0.5 W m−1 K−1), markedly lower than that of Fe3O4 (6 W m−1 K−1), results in preferential heat localization during thermal treatment.17,18 This divergence defines the molecular origin of the programmable and reversible etching selectivity in the binary superlattices.
Crucially, this asymmetric ligand evolution arises from the intrinsic difference in thermal conductivity and heat dissipation between the two inorganic cores, which dictates how efficiently thermal energy is delivered to the surface ligands. As schematically illustrated in Fig. 3, the superlattice therefore encodes a thermally programmable surface chemistry: at short thermal treatment times NaYF4 is preferentially etched, whereas prolonged thermal treatment renders NaYF4 acid-resistant and shifts the etching to Fe3O4. In this way, thermal treatment time is translated into controllable interfacial reactivity, enabling reversible switching of etching selectivity by engineering ligand-shell evolution rather than altering the etchant itself.
To evaluate the generality of the ligand-reorganization–governed etching mechanism, we extended this strategy to a second binary system composed of nanodumbbells (NDS) and NSs (Fig. 4a).10 Oleate-capped β-NaYF4:Yb/Er@NaGdF4@NaNdF4 NDs, defined by the diagonal head size (D) and waist diameter (d) (Fig. S2), were co-assembled with Fe3O4 NSs (21.1 ± 0.1 nm; Fig. S1b) using the same liquid–air interfacial method. The two components organize into a hexagonal AB2-type superlattice, in which each ND is surrounded by six NSs, while each nanosphere is coordinated by three NDs (Fig. 4a). The long-range order is confirmed by large grain sizes and the corresponding SAED pattern (Fig. 4b and Fig. S11). HAADF-STEM imaging and elemental mapping resolve that Fe3O4 NSs occupy the interstitial voids between adjacent NDs (Fig. 4b).
After thermal treatment, the same oxalic-acid etching protocol selectively removes one component from the dumbbell–sphere superlattice, yielding ordered NCP arrays. Preferential etching of the NDs produces a honeycomb-packed Fe3O4 lattice (Fig. 4c and Fig. S12),7 whose long-range order is preserved as evidenced by SAED, while Fe mapping confirms that only Fe3O4 NSs remain. The Fe3O4 NSs size is essentially unchanged, and the interparticle spacing matches the waist dimension of the removed NDs (Fig. S12).
Conversely, reversing the etching selectivity removes the Fe3O4 NSs while preserving the NDs, yielding an ordered hexagonal NCP ND array, as confirmed by SAED and Gd elemental mapping (Fig. 4d). After etching, the diagonal head size decreases from 43.0 to 37.3 nm, and the initially sharp hexagonal heads become visibly more rounded, suggesting facet-selective dissolution that progressively smooths the dumbbell geometry during etching, while the overall hexagonal registry is maintained (Fig. S13). Similarly, the etching process evolves progressively with time, as evidenced by the temporal statistics in Fig. S14. This establishes a general principle: etching selectivity is governed by particle composition rather than shape. By exploiting nanoscale heat accumulation in NaYF4 NPs, thermal energy is translated into ligand reconstruction and programmable interfacial chemistry, enabling reversible and morphology-independent control of etching sequences in binary superlattices.
We demonstrate a simple and controllable approach to selectively etch BNSLs. Thermally-induced reorganization and densification of the ligand shell on NaYF4 NPs inverts the relative etching preference of the two components without changing the etchant. The concept’s generality across different NaYF4 morphologies highlights its potential as a versatile platform for nanoscale design.
This work was supported by the NSFC (52333013, 52522317, 52503361, 22025501, and 22088101) and the Shanghai Municipal Science and Technology Project (24TS1410200, 23JC1400100, 2024ZDSYS02, 24JD1400700, and 24QA2700800).
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc00778c.References
- Y. Kang, X. Ye, J. Chen, Y. Cai, R. E. Diaz, R. R. Adzic, E. A. Stach and C. B. Murray, J. Am. Chem. Soc., 2013, 135, 42–45 CrossRef CAS.
- F. Schulz, O. Pavelka, F. Lehmkühler, F. Westermeier, Y. Okamura, N. S. Mueller, S. Reich and H. Lange, Nat. Commun., 2020, 11, 3821 CrossRef CAS PubMed.
- A. Dreyer, A. Feld, A. Kornowski, E. D. Yilmaz, H. Noei, A. Meyer, T. Krekeler, C. Jiao, A. Stierle, V. Abetz, H. Weller and G. A. Schneider, Nat. Mater., 2016, 15, 522–528 CrossRef CAS.
- Y. Liu, M. Klement, Y. Wang, Y. Zhong, B. Zhu, J. Chen, M. Engel and X. Ye, J. Am. Chem. Soc., 2021, 143, 16163–16172 CrossRef CAS.
- T. Udayabhaskararao, T. Altantzis, L. Houben, M. Coronado-Puchau, J. Langer, R. Popovitz-Biro, L. M. Liz-Marzán, L. Vuković, P. Král, S. Bals and R. Klajn, Science, 2017, 358, 514–518 CrossRef CAS PubMed.
- K. Santhosh, O. Bitton, L. Chuntonov and G. Haran, Nat. Commun., 2016, 7, 11823 CrossRef PubMed.
- M. P. Boneschanscher, W. H. Evers, J. J. Geuchies, T. Altantzis, B. Goris, F. T. Rabouw, S. A. P. van Rossum, H. S. J. van der Zant, L. D. A. Siebbeles, G. Van Tendeloo, I. Swart, J. Hilhorst, A. V. Petukhov, S. Bals and D. Vanmaekelbergh, Science, 2014, 344, 1377–1380 CrossRef CAS PubMed.
- M. Piotrowski, Z. Ge, X. Han, Y. Wang, A. K. Bandela and U. Thumu, Nanoscale, 2023, 15, 5188–5192 RSC.
- Y. Cai, M. Lu, X. Qin, D. Jin and J. Zhou, Nat. Commun., 2025, 16, 4927 CrossRef CAS PubMed.
- Z. Zheng, H. Wang, Y. Gao, S. Wan, L. Lv, J. Yang, D. Yang, Y. Ling, T. Li and A. Dong, ACS Nano, 2025, 19, 29788–29797 CrossRef CAS PubMed.
- A. Dong, J. Chen, P. M. Vora, J. M. Kikkawa and C. B. Murray, Nature, 2010, 466, 474–477 CrossRef CAS PubMed.
- H. Hu, M. Yu, F. Li, Z. Chen, X. Gao, L. Xiong and C. Huang, Chem. Mater., 2008, 20, 7003–7009 CrossRef CAS.
- Z. Chen, H. Chen, H. Hu, M. Yu, F. Li, Q. Zhang, Z. Zhou, T. Yi and C. Huang, J. Am. Chem. Soc., 2008, 130(10), 3023–3029 CrossRef CAS PubMed.
- K. I. Hadjiivanov, D. A. Panayotov, M. Y. Mihaylov, E. Z. Ivanova, K. K. Chakarova, S. M. Andonova and N. L. Drenchev, Chem. Rev., 2021, 121, 1286–1424 CrossRef CAS PubMed.
- L. Wang and Y. Li, Nano Lett., 2006, 6, 1645–1649 CrossRef CAS PubMed.
- Z. Wang, A. S. S. Singaravelu, R. Dai, Q. Nian, N. Chawla and R. Y. Wang, Angew. Chem., Int. Ed., 2020, 59, 9556–9563 CrossRef CAS PubMed.
- N. W. Park, W. Y. Lee, J. A. Kim, K. Song, H. Lim, W. D. Kim, S. G. Yoon and S. K. Lee, Nanoscale Res. Lett., 2014, 9, 96 CrossRef PubMed.
- J. M. Gonçalves, A. R. N. Bastos, S. J. L. Ribeiro, L. D. Carlos, R. L. L. Longo, J. M. A. Caiut and R. A. S. Ferreira, Nanoscale Adv., 2024, 6(5), 1486–1496 RSC.
| This journal is © The Royal Society of Chemistry 2026 |