Open Access Article
Open Access ArticlePhotocatalyzed generation of acyl radicals from aryl/alkyl acyl halides via nucleophilic substitution
Haowen
Zhan
,
Jiansheng
Chen
,
Yajing
Luo
,
Yang
Ke
and
Xinjun
Tang
 *
*
Faculty of Materials Science and Chemistry, China University of Geosciences, Wuhan 430074, China. E-mail: tangxinjun@cug.edu.cn
First published on 10th March 2026
Abstract
Herein, we introduce a nucleophilic catalysis strategy based on a 1-methylpyridine-2(1H)-thione (MPT) catalyst, which mediates the in situ generation of photoactive intermediates from various acyl halides. This strategy enables the efficient formation of both aryl and alkyl acyl radicals upon visible-light irradiation, including those derived from otherwise unreactive alkyl acyl fluorides/chlorides.
Acyl halides are inexpensive, abundant, highly reactive, and commercially available chemicals that have been extensively employed in organic synthesis,1,2 traditionally engaging in transformations via acyl cation intermediates. Concurrently, visible-light photoredox catalysis has emerged as a powerful, sustainable, and environmentally benign synthetic strategy.3–5 This strategy has unlocked diverse acyl radical precursors, including ketoacids, aldehydes, and notably, acyl chlorides, thereby significantly expanding the synthetic toolbox for radical acylation reactions with high synthetic potential.
Through photooxidative pathways, ketoacids have been established as versatile aryl and alkyl acyl sources, enabling efficient construction of C–C6–9 and C–X10 bonds. Aldehydes are also used as acyl radical sources via a photoinduced hydrogen atom transfer process, participating in radical addition reactions,11,12 coupling reactions13 and functional group conversion reactions.14,15 Through the photoreductive pathway, acid anhydride16 and acid chloride17–20 have been utilized for acyl radical generation, though the latter have been limited to aryl acid chlorides. Additional methods, such as the phosphorus-mediated deoxygenation of carboxylic acids,21–23 along with thioester24–26 and acyl silanes,27 further complement the toolbox for acyl radical generation.
However, the photocatalytic application of carboxylic acid derivatives, particularly alkyl acyl halides and acid anhydrides, remains constrained by redox potential limitations28–30 (Scheme 1a). For instance, acetyl chloride (Ered1/2 = −1.98 V vs. SCE29) and acetyl anhydride (Ered1/2 = −2.03 V vs. SCE29) exceed the typical reducing capacity of fac-Ir(ppy)3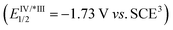 . As for acyl fluorides, the high C–F bond dissociation energy makes direct generation of acyl radicals through homolytic C–F bond cleavage highly challenging. Additionally, the low leaving-group tendency of F− renders acyl fluorides chemically inert among acyl halides. Although there have been a few recent reports describing acyl radical formation from aryl fluorides through charge-transfer complex (CTC) activation31 or transition-metal catalyzation,32 such transformations remain underexplored, especially for alkyl acyl fluorides, which currently constitute uncharted territory in photocatalytic radical generation.
. As for acyl fluorides, the high C–F bond dissociation energy makes direct generation of acyl radicals through homolytic C–F bond cleavage highly challenging. Additionally, the low leaving-group tendency of F− renders acyl fluorides chemically inert among acyl halides. Although there have been a few recent reports describing acyl radical formation from aryl fluorides through charge-transfer complex (CTC) activation31 or transition-metal catalyzation,32 such transformations remain underexplored, especially for alkyl acyl fluorides, which currently constitute uncharted territory in photocatalytic radical generation.
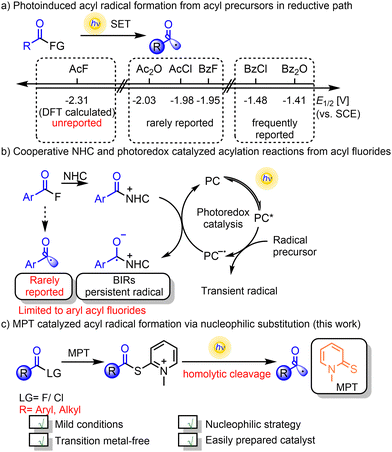 | ||
| Scheme 1 (a) Photoinduced acyl radical formation. (b) Cooperative NHC and photoredox catalyzed acylation reactions. (c) MPT-catalyzed acyl radical formation via nucleophilic substitution. | ||
Recently, Scheidt's group and Studer's group independently demonstrated the single-electron reduction of NHC–acyl adducts to generate Breslow intermediate-driven radicals (BIRs), using N-benzoylimidazole33 and acyl fluorides,34 respectively. Later, the cooperative NHC and photoredox catalyzed acylation reactions proceeding from acyl fluorides via BIRs were further developed by Studer,35–39 Zhang40–42 and Feng.43,44 In addition, other researchers have extended these co-catalytic systems to accommodate complex coupling reactions.45–51 In these reactions, NHC catalysis is vital for forming Breslow intermediates, which are more easily reduced to ketyl radicals than acyl fluorides are. However, these acylation reactions are limited to aryl acyl fluorides (Scheme 1b). Thus, it is important to develop a new, simple, and universal methodology using only one photocatalyst for the generation of acyl radicals from aryl/alkyl acyl fluorides.
Recently, Melchiorre's group52–55 and our group (MPT catalyst)56 reported the generation of carbon radicals through an SN2 pathway. This MPT catalyst exhibits good nucleophilicity and visible-light absorptivity.56,57 Herein, we report a photoinduced MPT-catalyzed nucleophilic substitution of acyl halides to generate acyl radicals. Both acyl fluorides and acyl chlorides can be applied in this reaction to form the nucleophilic substitution intermediate, which can be photoexcited and undergo homolytic cleavage of the C(sp2)–S bond to generate acyl radicals. In particular, the aryl and alkyl acyl fluorides/chlorides are all tolerated in this reaction (Scheme 1c).
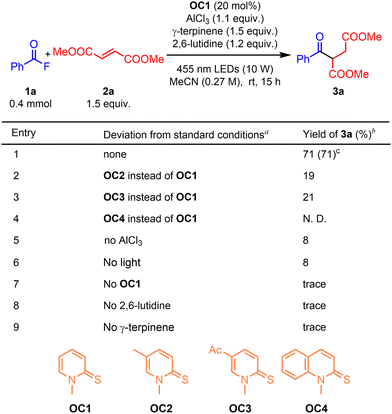
To test our hypothesis, we initially used benzoyl fluoride (1a) and dimethyl fumarate (2a) as model substrates. To increase the reactivity of the benzoyl fluoride, we added AlCl3 as the additive. To our delight, the desired product 3a could be obtained in 33% yield (see the SI, Table S1). After screening the reaction conditions (see the SI, Tables S1–S7), the highest yield could be obtained as 71% (Table 1, entry 1). Other MPT catalysts gave lower yields (Table 1, entries 2–4). The yields dropped a lot without AlCl3 or light (Table 1, entries 5 and 6). The reaction was shut down without OC1, 2,6-lutidine or γ-terpinene (Table 1, entries 7–9).
| a Reaction conditions: 1a (0.4 mmol), 2a (1.5 equiv.), OC1 (20 mol%), AlCl3 (1.1 equiv.), γ-terpinene (1.5 equiv.), 2,6-lutidine (1.2 equiv.), MeCN (1.5 mL) with irradiation by a 10 W 455 nm LED at room temperature for 15 h under a N2 atmosphere. b Yield determined by 1H NMR analysis of the crude mixture using 0.4 mmol of mesitylene as an internal standard. c Isolated yield. |
|---|
|
With the optimized conditions in hand, we then investigated the scope of the reaction (Scheme 2). We first tested the para-substituted benzoyl fluorides (3b–3m). para-Alkyl/phenyl benzoyl fluorides (3b–3e) gave moderate yields (56–65%). Halogens (3f–3h) were tolerated in the reactions and showed similar reactivity (52–63%). Benzoyl fluorides with electron-donating groups on the para position (3j–3m) achieved higher yields (62–68%). Meanwhile, the yields dropped when the electron-donating groups were on the meta and ortho positions (3n–3o). Electron-withdrawing groups could also be applied in this reaction in which 1.4 equiv. of AlCl3 was added to yield 3p in 51% yield. It should be pointed out that carbonyl-containing groups (1p) may deactivate AlCl3 and decrease the formation of the intermediate, while N-containing groups (1i) may cause AlCl3 to precipitate. To our delight, di- and tri-substituted benzoyl fluorides showed good reactivities in this reaction (3q–3u). 2-Naphthoyl fluoride also reacted smoothly and afforded the product in 41% yield (3v). Notably, the alkyl acyl fluoride 1w and 1x could be applied in this reaction and afford 3w and 3x in 40% and 41% yields, respectively. These alkyl acyl fluorides were not suitable for the cooperative NHC and photoredox catalyzed system.34–51
Aryl acyl chlorides17–20 could also be applied in this reaction. Acyl chlorides were more reactive than acyl fluorides. Both electron-rich and electron-poor acyl chlorides were good substrates for this reaction and gave the product in moderate to good yields (3y–3ad). These results suggested that electronic effects play a dominant role in this reaction (3ab–3ad, 3af–3ag). Interestingly, heteroaromatic acyl chlorides (1ae–1ag) and alkyl acyl chlorides (1w–1x, 1ah–1ai) were suitable for this reaction. Meanwhile, N-substituted aminoacyl chlorides and carbonochloridates were not good substrates in these reactions.
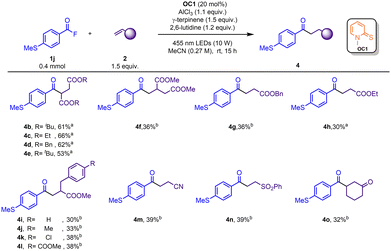
We also investigated several Michael acceptors in the Giese reactions with 4-MeS benzoyl fluoride as the model substrate (Scheme 3). Fumarate diesters were suitable for this reaction, yielding 4b–4e in 53–66% yields. Acrylic ester derivatives (4f–4h) were reactive with lower yields (30%–36%). Electron-poor 1,1-disubstituted alkenes could also be applied in this reaction and afforded the corresponding products in moderate yields (4i–4l). Alkenes bearing various electron-withdrawing groups such as nitrile, sulfone, and ketone could afford the products (4m–4o) in moderate yields.
To demonstrate the synthetic utility, we smoothly performed a gram-scale preparation of 3a and obtained 3a in 53% yield (see the SI, Scheme S3a). Subsequent derivatization of 3a was efficiently achieved (see the SI, Scheme S3b). Lactam58 and lactone59,60 could be synthesized from 3a in 60–72% yields (5–7). Interestingly, α-bromide derivative 861,62 could be formed in 74% yield, serving as an intermediate for the synthesis of hydantoins.61
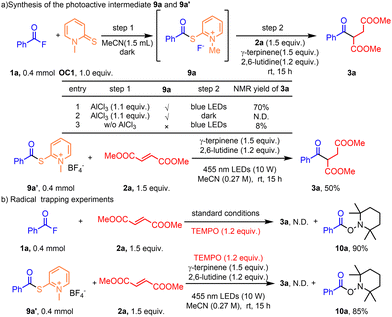
Control experiments were conducted to gain more insights into the reaction mechanism. In the presence of AlCl3, 1a and OC1 could form the photoactive intermediate 9a in the dark. 9a could be photoexcited to generate an acyl radical and afford the corresponding product 3a. The reaction could not work without AlCl3 or without light (Scheme 4a). We tried to isolate intermediate 9a, but failed. Therefore, we synthesized 9a′63 instead and subjected 9a′ to the standard reaction conditions affording product 3a in 50% yield. Radical trapping experiments with TEMPO showed that no desired product was found and adduct 10a was isolated in 90% and 85% yields, respectively (Scheme 4b). Additionally, no redshift phenomenon was observed with UV-Vis spectroscopic analysis (see the SI, Scheme S8). The possibility of an electron donor–acceptor (EDA) complex process could be excluded. The light on/off experiment showed that this reaction was not a chain reaction (see the SI, Scheme S10). The addition of triplet quenchers64–66 significantly suppressed the reaction (see the SI, Table S8), which indicated the involvement of a triplet state species. These experimental results confirm the generation of nucleophilic intermediates 9, which, upon absorption of blue light, undergo homolytic cleavage to produce acyl radicals.
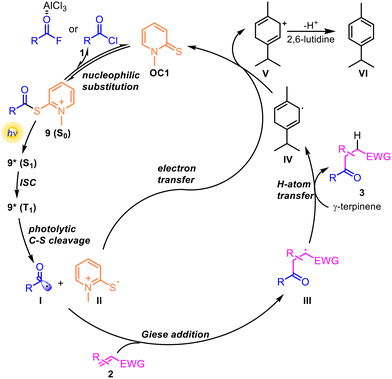
Based on the above results and related reports,52–56 a plausible catalytic mechanism is proposed (Scheme 5). In the presence of AlCl3, the nucleophilic catalyst OC1 reacted with acyl halides to generate the key intermediate 9 through a nucleophilic substitution pathway. Upon visible-light irradiation, 9 was photoexcited to the S1 state and underwent intersystem crossing (ISC) to the T1 state, from which homolytic C–S bond cleavage generated acyl (I) and thiyl (II) radicals. Then, the acyl radical was trapped by Michael acceptors, followed by HAT to form the corresponding product 3. Meanwhile, the thiyl radical II could be reduced by carbon radical IV through a SET process to reproduce the nucleophilic catalyst OC1, thereby closing the catalytic cycle. Carbocation V generated after single-electron transfer underwent aromatization with the base 2,6-lutidine to give VI.
In summary, we developed a photoinduced MPT-catalyzed nucleophilic substitution of acyl halides to generate acyl radicals. Aryl/alkyl acyl fluorides and chlorides were suitable in this reaction. Notably, alkyl acyl fluorides proved ineffective as substrates in previously reported photoinduced acylation reactions. The MPT catalyst and acyl halides could form the photoactive nucleophilic substitution intermediates, which then could be photoexcited to generate acyl radicals via C(sp2)–S bond homolytic cleavage. This nucleophilic substitution strategy does not rely on the redox properties of acyl halides, including those with low redox potentials such as alkyl acyl fluorides. It offers a powerful, modular, and practical strategy for constructing valuable aroyl/alkanoyl motifs. The easily prepared catalyst, mild reaction conditions, good yields, and broad scope show the potential synthetic utility of this method. Further studies including other acylation reactions are being conducted in our laboratory.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details and NMR spectra. See DOI: https://doi.org/10.1039/d6cc00687f.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22102160), the “CUG Scholar” Scientific Research Funds at China University of Geosciences (Wuhan) (Project No. 2022014), the Wuhan Talent Plan (Excellent Young Talents, No. 20230350051), and the Zhejiang Provincial Natural Science Foundation of China (No. LQ22B020004).References
- C. Song, C. Pang, Y. Deng, H. Cai, X. Gan and Y. R. Chi, ACS Catal., 2024, 14, 6926–6935 CrossRef CAS
.
- M. Tao, J. Qian, L. Deng, D. M. Wilson, X. Zhang and J. Liu, Nat. Chem., 2025, 17, 532–540 CrossRef CAS PubMed
.
- C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322–5363 CrossRef CAS PubMed
.
- K. L. Skubi, T. R. Blum and T. P. Yoon, Chem. Rev., 2016, 116, 10035–10074 CrossRef CAS
.
- N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed
.
- L. Chu, J. M. Lipshultz and D. W. C. MacMillan, Angew. Chem., Int. Ed., 2015, 54, 7929–7933 CrossRef CAS
.
- W.-M. Cheng, R. Shang, H.-Z. Yu and Y. Fu, Chem. – Eur. J., 2015, 21, 13191–13195 CrossRef CAS PubMed
.
- J. Ning, G. Jiang, H. Du and D. Kong, Org. Lett., 2025, 27, 5995–6000 CrossRef PubMed
.
- Y. Liu, Y.-Y. Cheng, J.-X. Yu, C. Wang, H.-L. Hu, G. Liang, F.-X. Li, H.-Y. Hou, X.-N. Guo, C.-H. Tung and L.-Z. Wu, Org. Lett., 2025, 27, 6777–6782 CrossRef CAS PubMed
.
- J. Liu, Q. Liu, H. Yi, C. Qin, R. Bai, X. Qi, Y. Lan and A. Lei, Angew. Chem., Int. Ed., 2014, 53, 502–506 CrossRef CAS PubMed
.
- P. Cheng, Z. Qing, S. Liu, W. Liu, H. Xie and J. Zeng, Tetrahedron Lett., 2014, 55, 6647–6651 CrossRef CAS
.
- D.-N. Chen, D.-J. Luo, L.-L. Qin, F.-Q. Zhang, Y. Wang, Y. Lei, Y.-X. Chen and P.-J. Xia, Chin. J. Chem., 2026, 44(7), 973–978 CrossRef CAS
.
- Z. Hu, W. Zhang and X. U. Tao, Chin. J. Chem., 2023, 41, 3593–3597 CrossRef CAS
.
- N. Iqbal, S. Choi, Y. You and E. J. Cho, Tetrahedron Lett., 2013, 54, 6222–6225 CrossRef CAS
.
- Y. Zhang, P. Ji, Y. Dong, Y. Wei and W. Wang, ACS Catal., 2020, 10, 2226–2230 CrossRef CAS PubMed
.
- G. Bergonzini, C. Cassani and C.-J. Wallentin, Angew. Chem., Int. Ed., 2015, 54, 14066–14069 CrossRef CAS PubMed
.
- S.-M. Xu, J.-Q. Chen, D. Liu, Y. Bao, Y.-M. Liang and P.-F. Xu, Org. Chem. Front., 2017, 4, 1331–1335 RSC
.
- L. Bao, Z.-X. Wang and X.-Y. Chen, Org. Lett., 2022, 24, 8223–8227 CrossRef CAS PubMed
.
- Y. Zhou, Q. Jiang, Y. Cheng, M. Hu, X.-H. Duan and L. Liu, Org. Lett., 2024, 26, 2656–2661 CrossRef CAS PubMed
.
- X. Yang, M. Yang, S. Qin, H. Li, W. Duan, J. Han and L. Yu, Org. Chem. Front., 2025, 12, 6132–6138 RSC
.
- E. E. Stache, A. B. Ertel, T. Rovis and A. G. Doyle, ACS Catal., 2018, 8, 11134–11139 CrossRef CAS PubMed
.
- M. Zhang, J. Xie and C. Zhu, Nat. Commun., 2018, 9, 3517 CrossRef
.
- W.-H. Tang, L.-Y. Wu, Q.-Q. Zhou and J.-P. Wan, Org. Chem. Front., 2025, 12, 2340–2345 RSC
.
- M. Ociepa, O. Baka, J. Narodowiec and D. Gryko, Adv. Synth. Catal., 2017, 359, 3560–3565 CrossRef CAS
.
- A. R. Norman, M. N. Yousif and C. S. P. McErlean, Org. Chem. Front., 2018, 5, 3267–3298 RSC
.
- H. Yang, Y. Pan, C. Zhang, C. Chen, H. Tang, H. Zhang, G. Deng and X. Yang, Org. Lett., 2025, 27, 4980–4985 CrossRef CAS PubMed
.
- L. Capaldo, R. Riccardi, D. Ravelli and M. Fagnoni, ACS Catal., 2018, 8, 304–309 CrossRef CAS
.
- G. T. Cheek and P. A. Horine, J. Electrochem. Soc., 1984, 131, 1796 Search PubMed
.
- D. Occhialini, K. Daasbjerg, H. Lund, K. Hult, T. R. Norin, P. Coppens and O. Buchardt, Acta Chem. Scand., 1993, 47, 1100–1106 Search PubMed
.
- H. Li, M. Peng, Z. Lai, L. Ning, X. Chen, X. Zhang, P. Wang, R. Szostak, M. Szostak and J. An, Chem. Commun., 2021, 57, 5195–5198 Search PubMed
.
- L. Bao, Z.-X. Wang and X.-Y. Chen, Org. Lett., 2023, 25, 565–568 CrossRef CAS PubMed
.
- N. Oku, R. Saeki, Y. Doi, K. Yamazaki and T. Miura, Org. Lett., 2025, 27, 3361–3367 Search PubMed
.
- A. V. Bay, K. P. Fitzpatrick, R. C. Betori and K. A. Scheidt, Angew. Chem., Int. Ed., 2020, 59, 9143–9148 CrossRef CAS PubMed
.
- Q.-Y. Meng, N. Döben and A. Studer, Angew. Chem., Int. Ed., 2020, 59, 19956–19960 CrossRef CAS
.
- K. Liu and A. Studer, J. Am. Chem. Soc., 2021, 143, 4903–4909 CrossRef CAS PubMed
.
- Q.-Y. Meng, L. Lezius and A. Studer, Nat. Commun., 2021, 12, 2068 CrossRef CAS PubMed
.
- X. Yu, Q.-Y. Meng, C. G. Daniliuc and A. Studer, J. Am. Chem. Soc., 2022, 144, 7072–7079 CrossRef CAS PubMed
.
- X. Yu, A. Maity and A. Studer, Angew. Chem., Int. Ed., 2023, 62, e202310288 Search PubMed
.
- S. K. Banjare, L. Lezius and A. Studer, Angew. Chem., Int. Ed., 2025, 64, e202511689 CrossRef CAS
.
- L. Wang, R. Ma, J. Sun, G. Zheng and Q. Zhang, Chem. Sci., 2022, 13, 3169–3175 RSC
.
- L. Wang, J. Sun, J. Xia, M. Li, L. Zhang, R. Ma, G. Zheng and Q. Zhang, Sci. China: Chem., 2022, 65, 1938–1944 CrossRef CAS
.
- J. Xia, R. Ma, L. Wang, J. Sun, G. Zheng and Q. Zhang, Org. Chem. Front., 2024, 11, 3089–3099 RSC
.
- S. Li, H. Shu, S. Wang, W. Yang, F. Tang, X.-X. Li, S. Fan and Y.-S. Feng, Org. Lett., 2022, 24, 5710–5714 CrossRef CAS PubMed
.
- S. Li, C. Zhang, S. Wang, W. Yang, X. Fang, S. Fan, Q. Zhang, X.-X. Li and Y.-S. Feng, Org. Lett., 2024, 26, 1728–1733 CrossRef CAS PubMed
.
- A. Mavroskoufis, K. Rajes, P. Golz, A. Agrawal, V. Ruß, J. P. Götze and M. N. Hopkinson, Angew. Chem., Int. Ed., 2020, 59, 3190–3194 CrossRef CAS PubMed
.
- H. Huang, Q.-S. Dai, H.-J. Leng, Q.-Z. Li, S.-L. Yang, Y.-M. Tao, X. Zhang, T. Qi and J.-L. Li, Chem. Sci., 2022, 13, 2584–2590 RSC
.
- X. Tao, Q. Wang, L. Kong, S. Ni, Y. Pan and Y. Wang, ACS Catal., 2022, 12, 15241–15248 CrossRef CAS
.
- Z. Wang, X. Li, W. Li, Y. Cao and H. Li, Org. Chem. Front., 2023, 10, 4250–4255 RSC
.
- S. Tian, N. Chen, K. Cheng and Q. Wang, Org. Lett., 2024, 26, 4351–4355 CrossRef CAS PubMed
.
- R. Yang, J. Liao, H. Xi, M.-M. Li and W. Ding, J. Org. Chem., 2025, 90, 8767–8783 Search PubMed
.
- C.-B. Li, X.-N. Li, Z.-C. Li, J. Li, Z.-X. Wang, Z.-H. Gao and S. Ye, Angew. Chem., Int. Ed., 2025, 64, e202421151 CrossRef CAS PubMed
.
- B. Schweitzer-Chaput, M. A. Horwitz, E. de Pedro Beato and P. Melchiorre, Nat. Chem., 2019, 11, 129–135 CrossRef CAS PubMed
.
- S. Cuadros, M. A. Horwitz, B. Schweitzer-Chaput and P. Melchiorre, Chem. Sci., 2019, 10, 5484–5488 Search PubMed
.
- D. Mazzarella, G. Magagnano, B. Schweitzer-Chaput and P. Melchiorre, ACS Catal., 2019, 9, 5876–5880 Search PubMed
.
- E. de Pedro Beato, D. Mazzarella, M. Balletti and P. Melchiorre, Chem. Sci., 2020, 11, 6312–6324 Search PubMed
.
- Y. Wang, S. Fan and X. Tang, Org. Lett., 2024, 26, 4002–4007 Search PubMed
.
- L. Li, Y. Wang, L. Yuan and X. Tang, Chin. J. Chem., 2025, 43, 911–915 Search PubMed
.
- C. J. H. Morton, R. Gilmour, D. M. Smith, P. Lightfoot, A. M. Z. Slawin and E. J. MacLean, Tetrahedron, 2002, 58, 5547–5565 CrossRef CAS
.
- C. Forzato, G. Furlan, P. Nitti, G. Pitacco, E. Valentin, E. Zangrando, P. Buzzini, M. Goretti and B. Turchetti, Tetrahedron: Asymmetry, 2008, 19, 2026–2036 CrossRef CAS
.
- K. Bica and P. Gaertner, Eur. J. Org. Chem., 2008, 3453–3456 Search PubMed
.
- Q. Yu, J. Yu, H. Bao, X. Hu, D. Ying, L. Wu, F. Liu, H. Jiang, Z. Jinxia and S. Zhang, Synth. Commun., 2018, 48, 1939–1944 CrossRef CAS
.
- Y. Zhao, X. Jiang and Y.-Y. Yeung, Angew. Chem., Int. Ed., 2013, 52, 8597–8601 CrossRef CAS PubMed
.
- T. Sakakibara, Y. Watabe, M. Yamada and R. Sudoh, Bull. Chem. Soc. Jpn., 1988, 61, 247–253 CrossRef CAS
.
- C. Kim, J. Jeong, W. Lee and S. Hong, J. Am. Chem. Soc., 2026, 148, 7635–7644 CrossRef CAS PubMed
.
- A. Farmilo and F. Wilkinson, Chem. Phys. Lett., 1975, 34, 575–580 CrossRef CAS
.
- Y.-D. Du, L.-B. Zeng and G.-Q. Chen, Org. Lett., 2026, 28, 1361–1367 Search PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |

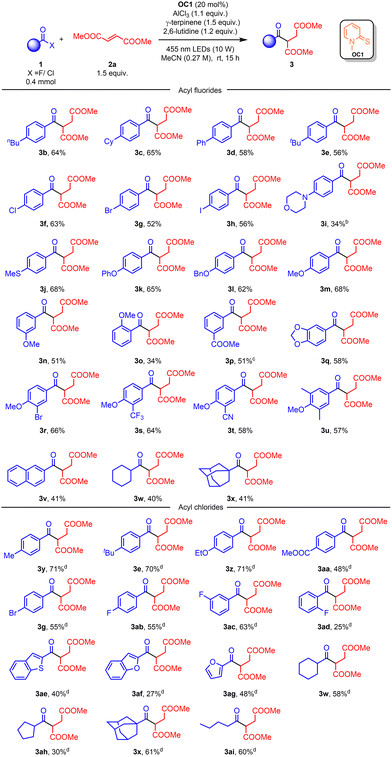
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Reaction conditions: 1 (0.4 mmol), 2a (1.5 equiv.), OC1 (20 mol%), AlCl3 (1.1 equiv.), γ-terpinene (1.5 equiv.), 2,6-lutidine (1.2 equiv.) in MeCN (1.5 mL) with irradiation by a 10 W 455 nm LED at room temperature for 15 h under a N2 atmosphere.
Reaction conditions: 1 (0.4 mmol), 2a (1.5 equiv.), OC1 (20 mol%), AlCl3 (1.1 equiv.), γ-terpinene (1.5 equiv.), 2,6-lutidine (1.2 equiv.) in MeCN (1.5 mL) with irradiation by a 10 W 455 nm LED at room temperature for 15 h under a N2 atmosphere.