Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Copper-catalyzed allylic substitution of nitroallyl derivatives with Grignard reagents
Louis
Clavier
a,
Nicolas
Fincias
a,
Stellios
Arseniyadis
 *b and
Laurent
El Kaim
*a
*b and
Laurent
El Kaim
*a
aLaboratoire de Synthèse Organique (LSO-UMR76523) CNRS, Ecole Polytechnique, ENSTA, Institut Polytechnique de Paris, 828 Bd des Maréchaux, 91128 Palaiseau Cedex, France. E-mail: laurent.elkaim@ensta.fr
bQueen Mary University of London, Department of Chemistry, Mile End Road, E1 4NS, London, UK. E-mail: s.arseniyadis@qmul.ac.uk
First published on 12th March 2026
Abstract
Grignard reagents engage with nitroallyl derivatives in the presence of copper(I) salts to promote allylic substitution of the nitro group. The reaction is particularly effective with aryl Grignards and unsubstituted nitroallyl compounds but is also applicable to diversely substituted nitroallyl derivatives and both alkyl and vinyl Grignards. A mechanism involving a Cu(I)/Cu(III) catalytic cycle is proposed for this transformation.
Owing to the strong electron-withdrawing nature of the nitro group, aliphatic nitro compounds readily undergo α-deprotonation and subsequent C–C bond formation under mild conditions, making them versatile intermediates in synthesis.1 Subsequent reduction of the nitro group to the corresponding primary amine provides efficient access to five- and six-membered nitrogen heterocycles.2 Alternatively, depending on the substitution pattern at the α-position, the relatively weak C–NO2 bond can undergo fragmentation, resulting in the extrusion of a nitrite species.3
The ability of nitroallyl derivatives to serve as surrogates for allylic acetates or carbonates in Tsuji–Trost reactions with carbon-, nitrogen-, and sulfur-based nucleophiles was first recognized by Hegedus, Tamura, and Ono using palladium catalysts (Fig. 1A).4a–c Despite several applications building on these seminal studies,4d–f the use of nitroallyl compounds as electrophiles in transition-metal catalyzed allylations are rare. In this context, we recently demonstrated that copper(I) salts could catalyze the substitution of nitroallyl derivatives with B2pin2, providing efficient access to allyl boronic esters (Fig. 1B).5 Building on this reactivity, we sought to evaluate whether copper catalysts could mediate analogous substitutions with other nucleophiles, including Grignard reagents. While Grignards are well known to participate in copper-mediated allylic substitution of ethers and carboxylates,6 the extension to nitroallyl electrophiles is not trivial as competitive nucleophilic addition to the nitro group typically predominates, leading to hydroxylamine or amine byproducts (Fig. 1C).7 Herein, we report a copper-catalyzed allylic substitution of nitroallyl derivatives with Grignard reagents, enabling selective C–C bond formation with concomitant nitrite elimination (Fig. 1D).
Allylic nitro derivatives are readily accessible from simple nitroalkanes. Condensation with aldehydes followed by elimination furnishes nitroalkenes, which can be deprotonated and functionalized with a range of electrophiles to enable α-substitution at the nitro group. As a representative example, symmetrical nitro diester 1a was prepared in four steps from nitromethane, methyl acrylate, and acetaldehyde.5 Selection of 1a minimizes stereochemical complexity in the Grignard addition while simultaneously probing the compatibility of ester functionality under the reaction conditions.
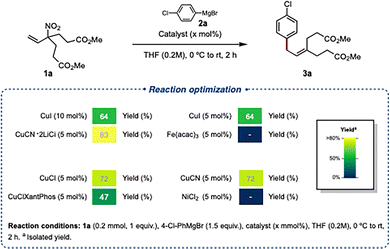
Treatment of 1a with 4-chlorophenylmagnesium bromide (2a, 2.0 equiv.) at 0 °C in the presence of CuI (10 mol%) afforded the desired diester 3a in 63% NMR yield. Reducing the amount of Grignard reagent to 1.5 equiv. or the CuI loading to 5 mol% had little effect on the reaction outcome (Fig. 2). In contrast, lowering the reaction temperature (CuI, −20 or −78 °C) completely suppressed coupling after 2 h. Modest improvements were observed upon replacement of CuI with CuCl or CuCN, although 4,4′-dichlorodiphenyl was consistently detected as a homocoupling byproduct. Optimal results were obtained using the more soluble CuCN·2LiCl complex, delivering 3a in 83% yield (82% isolated). These conditions were therefore adopted for subsequent studies.
The scope of the allylic substitution was first evaluated using a series of aromatic and aliphatic Grignard reagents with nitroallylic derivative 1a. As shown in Fig. 3, arylmagnesium bromides underwent efficient coupling to afford substituted diesters 3a–j in moderate to good yields. The reaction proved largely insensitive to the electronic properties of the aryl group with comparable efficiencies observed for both electron-rich and electron-poor substrates. Likewise, substitution at the 2-, 3-, or 4-position had minimal effect on reactivity, as illustrated by the similar outcomes obtained with mesityl and phenylmagnesium bromides. Heterocyclic Grignard reagents, such as 2-thienyl- and 3-pyridylmagnesium bromides (not shown), failed to deliver any substituted products. In contrast, vinyl- and styrylmagnesium bromides reacted efficiently with 1a to afford dienyl esters 3k (54%) and 3l (31%) in moderate yields. Reactions with alkylmagnesium reagents also proved challenging. For example, methylmagnesium bromide gave 3m in only 22% yield, likely due to competing direct addition onto the nitro group versus Cu-catalyzed allylic substitution. Consistent with this hypothesis, increasing the copper loading from 5 to 50 mol% improved the yield to 43%, highlighting the delicate balance between nucleophile reactivity and catalyst control.
The reaction scope was next evaluated with respect to the nitroallyl precursor using 4-methoxyphenyl magnesium bromide as the aryl Grignard, except for 3u for which the 4-chloro analogue was used (Fig. 4). The efficiency of the Grignard addition proved highly sensitive to the substitution pattern on the terminal alkene. While the reaction with 1a proceeded efficiently at 0 °C, introduction of a methyl group at the terminal position led to a more sluggish reaction, affording 3u in only 17% isolated yield and 50% conversion. Increasing the Grignard loading to 3 equiv. and the copper catalyst to 10 mol% slightly improved the yield (30%), however the product was formed along with a significant amount of 4,4′-dichloro-1,1′-biphenyl (4). Homocoupling of aryl Grignard reagents to biphenyl derivatives is a well-known transition-metal-catalysed process often promoted by an oxidant such as O2. In the present system, we attribute this side reaction to the nitrite anion released during the allylic substitution, consistent with observations made in our previous borylcupration of nitroallyl derivatives.5 The oxidizing potential of nitrite toward Grignard reagents under these conditions was confirmed by treatment of 4-chlorophenylmagnesium bromide with a stoichiometric amount of potassium nitrite in THF (Fig. 5A). The presence of terminal substituents on the alkene likely slows down the kinetics of the reaction with the intermediate arylcopper derivative, making it more susceptible to further reaction with the oxidant and arylmagnesium bromide. While terminal substituents are poorly tolerated, the alkene can be functionalized with an additional aromatic ring, as shown by the formation of 3r in 59% isolated yield. We also prepared several unsymmetrical nitroallyl compounds to investigate the diastereoselectivity of the elimination step. As illustrated by the various examples in Fig. 4, the diastereoselectivity is primarily influenced by the size of the substituents. Hence, while 3n was obtained with a poor E/Z selectivity, using a smaller methyl group compared to a cyanoethyl- (3o, 40%, E/Z = 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) or to a phenylsulfonylethyl group (3p, 40%, E/Z = 67
15) or to a phenylsulfonylethyl group (3p, 40%, E/Z = 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33) lead to higher selectivities, with preferential formation of the Z isomer. Fluorine-containing nitroallyl derivatives such as 1g and 1h were also prepared to evaluate whether the addition of the Grignard reagent, here 4-methoxyphenyl magnesium bromide, would promote a defluorination process rather than the elimination of the nitro group. Similar to what we observed in our previous borylation study,5 only nitro elimination was detected, resulting in the formation of 3s and 3t in 95 and 83% yield, respectively.
33) lead to higher selectivities, with preferential formation of the Z isomer. Fluorine-containing nitroallyl derivatives such as 1g and 1h were also prepared to evaluate whether the addition of the Grignard reagent, here 4-methoxyphenyl magnesium bromide, would promote a defluorination process rather than the elimination of the nitro group. Similar to what we observed in our previous borylation study,5 only nitro elimination was detected, resulting in the formation of 3s and 3t in 95 and 83% yield, respectively.
 | ||
| Fig. 4 Substrate scope. All reactions were carried out on a 0.2 mmol scale. aReaction run using 3 equiv. of Grignard reagent and 10 mol% of the copper catalyst. | ||
The strong influence of the substitution pattern on the alkene together with previous studies on the addition of Grignard reagents onto allylic esters6e suggest a reaction mechanism involving a Cu(I)/Cu(III) catalytical cycle. This mechanism is consistent with the formation of bis-aryl derivatives as side products. The latter may be formed either directly from the Cu(III) allyl intermediate B or by an oxidation of the Cu(I)Ar2MgBr intermediate A by the nitrite in solution leading to an Ar2Cu(III)X species which may lead to the bis-aryl by reductive elimination (Fig. 5B).
In conclusion, we have successfully demonstrated the first copper-catalysed substitution of nitroallyl derivatives by Grignard reagents. This work builds on our previous studies, including the cuproborylation of nitroallyl compounds5 and earlier research using alkyl lithium derivatives and stoichiometric amounts of copper(I) salts.8 The ability to achieve similar transformations using catalytic amounts of copper and Grignard reagents, rather than alkyl lithium, is particularly significant. Not only does this approach reduce the quantity of transition metal required, but it also remains compatible with sensitive functional groups, such as esters. Following these successful additions of B2pin2 and Grignards, we plan to explore the reactivity of other nucleophiles to gain a comprehensive understanding of the full scope of the copper-catalysed allylic substitution of nitroally derivatives.
The authors gratefully thank Ecole Polytechnique, ENSTA and Queen Mary University of London for financial support.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The datasets supporting this article have been uploaded as part of the supplementary information (SI). Supplementary information: full experimental procedures and characterization data for all compounds are provided. See DOI: https://doi.org/10.1039/d6cc00637j.References
- (a) A. Noble and J. C. Anderson, Chem. Rev., 2013, 113, 2887–2939 CrossRef CAS PubMed; (b) A. Z. Halimehjani, I. N. N. Namboothiri and S. E. Hooshmand, RSC Adv., 2014, 4, 48022–48084 RSC; (c) A. Z. Halimehjani, I. N. N. Namboothiri and S. E. Hooshmand, RSC Adv., 2014, 4, 51794–51829 RSC; (d) A. Y. Sukhorukov, A. A. Sukhanova and S. G. Zlotin, Tetrahedron, 2016, 72, 6191–6281 CrossRef CAS; (e) N. Nishiwaki, Molecules, 2020, 25, 3680 CrossRef CAS PubMed.
- (a) D. Formenti, F. Ferretti, F. K. Scharnagl and M. Beller, Chem. Rev., 2019, 119, 2611–2680 CrossRef CAS PubMed; (b) M. Orlandi, D. Brenna, R. Harms, S. Jost and M. Benaglia, Org. Process Res. Dev., 2018, 22, 430–445 CrossRef CAS.
- (a) N. Ono, T. Yanai, A. Kamimura and A. Kaji, J. Chem. Soc., Chem. Commun., 1986, 1285–1287 RSC; (b) M. P. Crozet, S. Lapouge, M. Kaafarani and P. Vanelle, Tetrahedron. Lett., 1994, 35, 3055–3058 CrossRef CAS; (c) Z. Li, C. Alameda-Angulo, B. Quiclet-Sire and S. Z. Zard, Tetrahedron, 2011, 67, 9844–9852 CrossRef CAS; (d) C.-L. Lo, P. S. Akula, B.-C. Hong, G.-H. Lee and S.-Y. Chien, Org. Lett., 2022, 24, 3353–3357 CrossRef CAS PubMed For cleavage under radical conditions: ; (e) D. D. Tanner, B. B. Blackburn and G. E. Diaz, J. Am. Chem. Soc., 1981, 103, 1557–1559 CrossRef CAS; (f) J. DuPuis, B. Giese, J. Hartung and M. Leising, J. Am. Chem. Soc., 1985, 107, 4332–4333 CrossRef CAS; (g) F. A. Luzzio, J. P. Ott and D. Y. Duveau, J. Org. Chem., 2006, 71, 5027–5030 CrossRef CAS PubMed.
- (a) R. Tamura and L. S. Hegedus, J. Am. Chem. Soc., 1982, 104, 3727–37292 CrossRef CAS; (b) R. Tamura, Y. Kai, M. Kakihana, K. Hayashi, M. Tsuji, T. Nakamura and D. Oda, J. Org. Chem, 1986, 51, 4375–4385 CrossRef CAS; (c) N. Ono, I. Hamamoto and A. Kaji, J. Chem. Soc. Perkin Trans. 1, 1986, 1439–1443 RSC For applications, see: ; (d) R. Padilla-Salinas, R. R. Walvoord, S. Tcyrulnikov and M. C. Kozlowski, Org. Lett, 2013, 15, 3966–3969 CrossRef CAS PubMed; (e) T. Nakano, K. Miyazaki and A. Kamimura, J. Org. Chem, 2014, 79, 8103–8109 CrossRef CAS PubMed; (f) M. Dolé Kerim, S. Jia, C. Theodorakidou, S. Prévost and L. El Kaïm, Chem. Commun., 2018, 54, 10917–10920 RSC.
- N. Fincias, L. Clavier, C. Escande de Messières, M. Guillard, M. Dole Kerim, N. Casaretto, J. Garrec, S. Arseniyadis and L. El Kaïm, JACS Au, 2025, 5, 99–110 CrossRef CAS PubMed.
- (a) Y. Gendreau and J. F. Normant, Tetrahedron, 1979, 35, 1517–1521 CrossRef CAS; (b) C. C. Tseng, S. D. Paisley and H. L. Goering, J. Org. Chem., 1986, 51, 2884–2891 CrossRef; (c) J. E. Bäckvall, M. Sellen and B. Grant, J. Am. Chem. Soc., 1990, 112, 6615–6621 CrossRef; (d) C. C. Tseng, S.-J. Yen and H. Goering, J. Org. Chem., 1986, 51, 2892–2895 CrossRef; (e) A. S. E. Karlström and J.-E. Bäckvall, Chem. – Eur. J., 2001, 7, 1981–1989 CrossRef.
- I. Sapountzis and P. Knochel, J. Am. Chem. Soc., 2002, 124, 9390–9391 CrossRef CAS PubMed; S.-S. Meng, F. Li, X. Tang and A. S. C. Chan, Org. Lett., 2023, 25, 3718–3722 CrossRef PubMed.
- N. Ono, I. Hamamoto and A. Kaji, J. Chem. Soc., Chem. Commun., 1984, 274–275 RSC.
| This journal is © The Royal Society of Chemistry 2026 |