Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Merging platinahelicene and nanographene: a strategy for circularly polarized phosphorescence in the near-infrared (NIR)
Wenying
Cui
 ab,
Huimin
Zhou
b,
Thierry
Roisnel
a,
Nicolas
Vanthuyne
ab,
Huimin
Zhou
b,
Thierry
Roisnel
a,
Nicolas
Vanthuyne
 c,
Chengshuo
Shen
b,
Joachim
Grzybowski
c,
Chengshuo
Shen
b,
Joachim
Grzybowski
 d,
J. A. Gareth
Williams
d,
J. A. Gareth
Williams
 e,
Monika
Srebro-Hooper
e,
Monika
Srebro-Hooper
 *d,
Yohan
Gisbert
*d,
Yohan
Gisbert
 *a and
Jeanne
Crassous
*a and
Jeanne
Crassous
 *a
*a
aUniv Rennes, CNRS, ISCR (Institut des Sciences Chimiques de Rennes) – UMR 6226, F-35000, Rennes, France. E-mail: yohan.gisbert@univ-rennes.fr; jeanne.crassous@univ-rennes.fr
bSchool of Chemistry and Chemical Engineering, Zhejiang Sci-Tech University, Hangzhou 310018, China
cAix Marseille Univ, CNRS, Centrale Med, FSCM, Marseille 13013, France
dFaculty of Chemistry, Jagiellonian University, Gronostajowa 2, 30-387, Krakow, Poland. E-mail: srebro@chemia.uj.edu.pl
eDepartment of Chemistry, University of Durham, Durham, DH1 3LE, UK
First published on 30th March 2026
Abstract
We present the first integration of a platinahelicene within a nanographene (NG), giving access to near-infrared circularly polarized phosphorescence with dissymmetry factors glum reaching 4 × 10−3. For comparison, a novel NG featuring a borahelicene displayed a blue-green circularly polarized fluorescence with glum = 6 × 10−4.
Over the past two decades, the development of nanographene (NG)-type molecules has experienced tremendous growth thanks to the introduction of highly efficient synthetic methodologies leading to diverse molecular architectures, including the incorporation of heteroatoms and chirality within the NG scaffold.1–6 This rapidly evolving research field now provides access to sophisticated chiral molecular materials with specific chiroptical activities in both ground and excited states (electronic circular dichroism, ECD; and circularly polarized luminescence, CPL respectively).7–12 However, achieving CPL in the near-infrared (NIR) remains a significant challenge with such materials: very few chiral NG-type molecules emit CPL above 750 nm.13–16 Such bathochromically shifted emission typically requires an extended π-conjugated system, involving sophisticated molecular architectures embedding multiple NG units, whose synthesis remains highly specific and challenging (Fig. 1a). We envisaged that the introduction of a metal17,18 (e.g., Fig. 1b) could offer a straightforward alternative approach to achieving NIR CPL emission in NGs. Platinum has a high spin–orbit coupling (SOC) constant that, in cyclometalated complexes in particular, can promote intersystem crossing and subsequent phosphorescence from triplet excited states.19,20 The association of a helically chiral nanographene with a platinum center may thus enable the chiroptical properties associated with the organic structure to be combined with room-temperature phosphorescence in the visible or NIR spectral regions.21 In line with this concept, platinum(II) helicene complexes – or “platinahelicenes” – have been developed that display circularly polarized, red phosphorescence at room temperature.22–27 In this communication, we describe the first preparation of helically chiral NGs that incorporate a heavy metal, namely platinum (NG1, Fig. 1c, left), and show how the straightforward combination of NG and platinahelicene within the same scaffold enables direct access to novel, helically chiral architectures that display CPL activity in the NIR. For comparison, the incorporation of boron28–32 in place of Pt(II) was carried out with the same NG scaffold (NG2, Fig. 1c, right), yielding a blue-green, fluorescent CPL emitter.
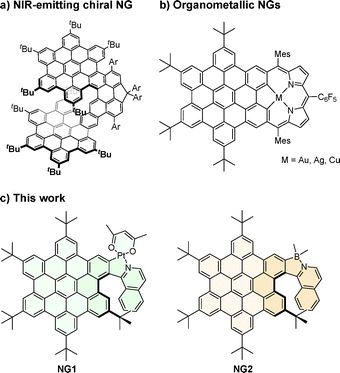 | ||
| Fig. 1 (a) A fully organic, NIR-emitting, chiral NG featuring extended π conjugation,13 (b) achiral organometallic nanographenes,17 and (c) the first helically chiral organometallic nanographene (left) and its analogue featuring a boron atom instead of the platinum center (right) presented in this work. | ||
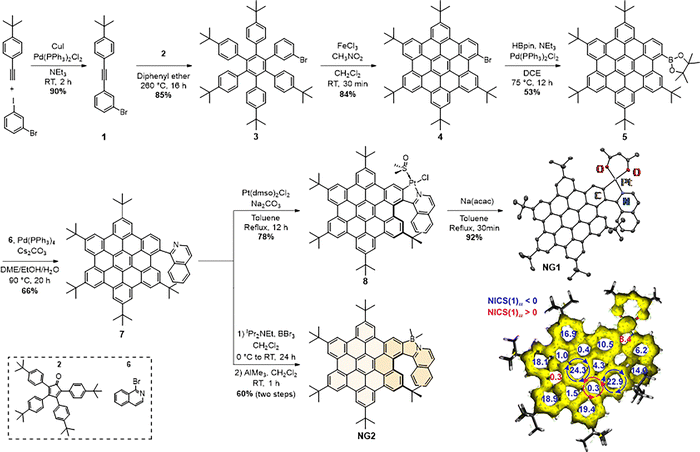
The platinum-containing helical nanographene NG1 was prepared following a 7-step synthetic sequence with 16% overall yield (Scheme 1). Sonogashira cross-coupling of 4-tert-butylphenylacetylene33 and 3-bromoiodobenzene gave diarylacetylene 1, and subsequent Diels–Alder cycloaddition with 2,3,4,5-tetrakis(4-tert-butylphenyl)cyclopentadienone342, followed by an in-situ chelotropic decarbonylation, yielded the hexaarylbenzene 3. The latter was subjected to a Scholl cyclodehydrogenation with FeCl3/CH3NO2, affording the brominated hexabenzocoronene derivative 4 in 84% yield, corresponding to the concomitant formation of five C–C bonds. Subsequent Miyaura borylation yielded the corresponding pinacol boronic ester 5, which was then subjected to a Suzuki–Miyaura cross-coupling with 1-bromoisoquinoline 6 to afford the key N^CH proligand 7, featuring both hexabenzocoronene (NG) and isoquinoline fragments. The platinum-containing helical nanographene NG1 was prepared from 7 by cycloplatination with Pt(dmso)2Cl2 followed by a ligand exchange with sodium acetylacetonate to afford NG1 as a deep-red solid. It was formed as a racemic mixture, from which the (P) and (M) enantiomers were isolated with an ee > 99% by preparative chiral HPLC (see Section S7 of the SI for details).
The proligand 7 was also used to prepare the metal-free analogue NG2, featuring a boron atom instead of the platinum center, by reaction with boron tribromide under basic conditions, followed by methylation at the boron center with trimethylaluminium to afford NG2 as a bright orange solid in 60% overall yield. Subsequent HPLC separation afforded both enantiomers of NG2 with ee > 99%.
The structures of NG1 and NG2 were unambiguously confirmed by high-resolution mass spectrometry (HRMS) and 1H/13C NMR spectroscopy. Furthermore, the 195Pt NMR of NG1 displayed a single signal at −2790 ppm, which falls within the expected range for such (N^C)Pt(II) complexes. Single crystals of NG1 suitable for X-ray diffraction were obtained by slow evaporation of a CH2Cl2/pentane solution of NG1, which crystallized in the C2/c space group (Scheme 1 and SI Section S5). The helicity of NG1 (angle between the planes of the terminal helicenic rings) is 39.35°, significantly lower than those of carbo[6]helicene (58.5°)35 and the one observed for a simpler (N^C)platina-[6]helicene (52.3°).23 This reduction is likely due to stabilizing, attractive, van der Waals/CH-π interactions between the quinoline moiety and the neighboring bulky tert-butyl group (Fig. S17 and S29). The NG1 molecules pack in separated pairs in the studied crystal, with a short Pt⋯Pt distance of 3.297 Å indicative of metallophilic interactions (Fig. S31). In line with findings for related systems,20,36 the nucleus-independent chemical shift (NICS) and anisotropy of the induced current density (AICD) analyses (Scheme 1 and SI Section S4), demonstrated extended π-delocalization and aromatic stabilization across the peripheral rings of the NG core. It also showed localized aromaticity at the center of the NG and on the quinoline moiety (evidenced by diatropic π-electron ring currents and large negative NICS values), as well as non-aromatic character of the platina- and boracycles (positive NICSs); however, a visible involvement of Pt(II) electrons in the induced ring current was observed for NG1.
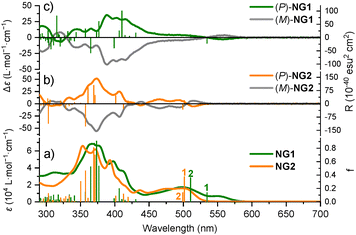
The UV-Visible absorption spectra of NG1 and NG2 in toluene (Fig. 2a) exhibit strong, well-defined absorption bands at λ < 400 nm, closely resembling the high-energy features of the proligand 7 (Fig. S2). In contrast to 7, NG1 and NG2 display additional lower-energy bands in the visible region (Table S2). NG1 shows bands at λ = 498 (ε = 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100) and 550 nm (6600 M−1 cm−1), the latter tailing to around 600 nm, whereas those of NG2 are hypsochromically shifted to λ = 476 (ε = 15
100) and 550 nm (6600 M−1 cm−1), the latter tailing to around 600 nm, whereas those of NG2 are hypsochromically shifted to λ = 476 (ε = 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300) and 499 nm (15
300) and 499 nm (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 M−1 cm−1).
800 M−1 cm−1).
TDDFT calculations revealed that, for both the complexes and the ligand, the higher-energy absorption arises predominantly from nanographene-centered ππ* transitions. The additional low-energy bands observed for NG1 and NG2 originate from the two lowest-energy excitations (no. 1 and 2, Fig. 2) of mixed ππ* and intraligand charge-transfer (ILCT) character, resulting from conjugation between the isoquinoline and nanographene fragments and helicenic moiety formed upon cyclization combined with the introduction of the platinum (NG1) or boron (NG2). In NG1, these excitations also include metal-to-ligand CT (MLCT) contributions, resulting in the appearance of the low-energy band at 550 nm, for which there is no apparent counterpart in NG2 or 7. More details of the assignments and computational methodology can be found in the SI Section S4.
The ECD of NG1 and NG2 were measured in the same conditions (Fig. 2c and 2b respectively). In each case, the enantiopure (P) and (M) forms display mirror-image spectra, which were assigned by TDDFT calculations (see SI Section S4). NG1 exhibits its strongest Cotton effect at 388 nm and its lowest energy band at 560 nm, whereas the corresponding features for NG2 occur at 373 nm and 515 nm, respectively, with the lowest-energy ECD intensity governed by the aforementioned excitations no. 1 and 2 (see SI Section S4). Consistent with the absorption data (vide supra), the ECD spectrum of NG1 is significantly bathochromically shifted relative to that of NG2. Similar absorption dissymmetry factors were obtained for both NGs, with gabs = 0.0015 (435 nm) and gabs = 0.0010 (408 nm) for NG1 and NG2, respectively (Fig. S8 and S12).
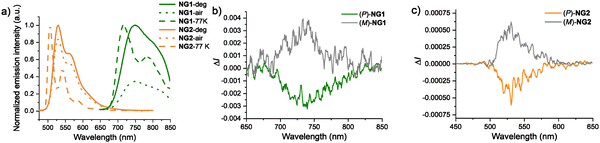
Both nanographenes are luminescent in toluene at room temperature (Fig. 3a and Table S2). NG1 emits at very low energy: almost all of its integrated emission lies at λ > 700 nm, in the NIR region, with only the onset falling into the deep red, showing the advantage of integrating a platinum center to red-shift the emission of the nanographene. There is a hint of vibrational structure, which becomes better resolved in the slightly blue-shifted spectrum recorded at 77 K. The emission lifetimes of 700 and 1200 ns recorded at 295 and 77 K respectively are consistent with phosphorescence from a triplet state, as is the significant quenching by oxygen observed in air-equilibrated solution (Fig. 3a). The emission is, however, weak, with a quantum yield Φlum in deoxygenated solution at 295 K of 0.33%. Efficient non-radiative decay through vibrational quenching is typical for low-energy emitters, suppressing quantum yields. Nevertheless, the fact that the lifetime is only modestly increased at 77 K suggests that a particularly low radiative rate constant kr may also be a limiting factor. Indeed, if it assumed that the emissive state is formed with unit efficiency such that kr = Φlum/τ, we can estimate kr to be about 4700 s−1, which is much lower than the values ≥105 s−1 that are found for efficient green-emitting Pt(II) and Ir(III)-based phosphors. Low kr values for phosphorescent organometallics are typically indicative of relatively inefficient SOC pathways, and often arise in complexes with extended conjugation, due to poor energy matching of the highest filled ligand orbitals with those of the metal.37,38
In contrast, NG2 displays bright green emission in solution, with λmax of 529 nm in deoxygenated toluene at 295 K, and a high quantum yield of 53% that is only marginally reduced upon aeration. The emission lifetime of 9.3 ns is indicative of fluorescence, and the estimated kr of around 6 × 107 s−1 supports a spin-allowed origin for the emission, in contrast to the phosphorescent Pt(II) analogue. As observed for the phosphorescence of NG1, the fluorescence of NG2 is a little blue-shifted at 77 K (as is normal for aromatics in the more rigid environment of a frozen glass), and vibrational structure becomes well resolved. The vibrational progression of around 1100 cm−1 is similar for NG1 and NG2, and evidently associated with the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of the highly conjugated helicene and nanographene systems. Finally, TDDFT calculations reasonably reproduce the measured fluorescence and phosphorescence energies of NG1 (and its precursor 8), NG2, and their parent ligand 7, linking the experimental trends to increased ILCT character (and MLCT for NG1 and 8) of the corresponding excited states in the complexes (see SI Section S4).
C bonds of the highly conjugated helicene and nanographene systems. Finally, TDDFT calculations reasonably reproduce the measured fluorescence and phosphorescence energies of NG1 (and its precursor 8), NG2, and their parent ligand 7, linking the experimental trends to increased ILCT character (and MLCT for NG1 and 8) of the corresponding excited states in the complexes (see SI Section S4).
The circularly polarized emission of the novel NGs was subsequently examined (Fig. 3b and c). Both enantiomers of each NG derivative were studied in ca. 10−5 M toluene solutions (degassed for NG1, under air for NG2) and, as expected, each pair displayed mirror-image spectra. The CPL signals for NG1 (Fig. 3b) span the range 690–810 nm, with a maximum at 740 nm and glum = 4 × 10−3 (Fig. S10), which is in the glum range of many efficient chiral phosphors (Table S1). The CPL of NG2 (Fig. 3c) ranges from 500 to 600 nm, peaking at 530 nm, with a glum = 6 × 10−4 (Fig. S14). This order-of-magnitude difference in glum may arise from the distinctly different natures of the emitting states. NG1 emits from a formally spin-forbidden T1 state localized mainly on the newly formed helicenic system and of predominantly 3ππ*/3MLCT character (with some 3ILCT), whereas NG2 emits from lowest-energy singlet state(s) dominated by (quinoline → NG) 1ILCT. Note that the magnitude (and sign) of CPL might also be shaped by vibronic effects.39,40 No CPL signal could be recorded for the NG-Pt(dmso)(Cl) 8, possibly due to its emission being very weak (Table S2).
In conclusion, the first metal-containing chiral nanographene, NG1, has been synthesized and characterized, thereby providing a new class of helically chiral, heavy-atom-containing polyaromatic systems. The platinum(II) center (compared to boron) establishes more effective π-conjugation within the newly formed helix and promotes efficient intersystem crossing to populate triplet states manifold, which results in NIR phosphorescence (λmax = 749 nm, emitting to >850 nm). Most significantly, NG1 also functions as an efficient chiral phosphor. It exhibits a high glum of 4 × 10−3, nearly an order of magnitude greater than that of its boron-containing fluorescent analogue NG2 (glum = 6 × 10−4). This pronounced chiroptical activity might be connected to localization of the emissive 3ππ*/3MLCT state at the helical scaffold. This work validates a powerful molecular design strategy, with direct integration of heavy atoms into a chiral nanographene framework, and it demonstrates the potential for accessing high-performance CPL materials operating in the biologically relevant NIR window.
Conflicts of interest
The authors declare no conflict of interest.Data availability
The data that support the findings of this study are available in the supplementary information (SI) section. Supplementary information: experimental procedures, photophysical and X-ray data, NMR spectra, HPLC analysis, computational details and calculated data. See DOI: https://doi.org/10.1039/d6cc00576d.CCDC 2502439 (NG1) contains the supplementary crystallographic data for this paper.41
Acknowledgements
This work was supported by the Ministère de la Recherche et de l’Enseignement Supérieur, the Centre National de la recherche Scientifique (CNRS), the China-French associated international program in ‘Functional Organophosphorus Materials’, the Chinese Scholarship Council (CSC) and the French National Agency (ANR projects LumoMat-E, 18-EURE-0012). Y. G. acknowledges financial support from the Région Bretagne (SAD 2023_CNRS_PITCH). Elsa Caytan is thanked for her assistance with NMR measurements. Part of this work has been performed using the RennesMR core facility (ScanMAT, UAR 2025 Univ Rennes-CNRS, France). M. S.-H. gratefully acknowledges the Polish high-performance computing infrastructure PLGrid (HPC Centers: ACK Cyfronet AGH, CI TASK) for providing computer facilities and support within computational grants no. PLG/2024/017254 and PLG/2025/018408. A part of the computational study was carried out using research infrastructure funded by the European Union in the framework of the Smart Growth Operational Programme, Measure 4.2; Grant No. POIR.04.02.00-00-D001/20, “ATOMIN 2.0 – Center for materials research on ATOMic scale for the INnovative economy”.References
- P. Izquierdo-García, J. M. Fernández-García, I. Fernández, J. Perles and N. Martín, J. Am. Chem. Soc., 2021, 143, 11864–11870 CrossRef PubMed.
- Y. Gu, Z. Qiu and K. Müllen, J. Am. Chem. Soc., 2022, 144, 11499–11524 CrossRef CAS PubMed.
- S. Míguez-Lago, J. P. Mora-Fuentes, C. M. Cruz and A. G. Campaña, Helicenes, John Wiley & Sons, Ltd, 2022, pp. 283–328 Search PubMed.
- V. Kumar, J. L. Páez, S. Míguez-Lago, J. M. Cuerva, C. M. Cruz and A. G. Campaña, Chem. Soc. Rev., 2025, 54, 4922–4947 RSC.
- W. Yang, Z. Ren, J. Feng, Z. Lv, X. Cheng, J. Zhang, D. Du, C. Chi and J. Shen, Angew. Chem., Int. Ed., 2024, 63, e202412681 CrossRef CAS PubMed.
- P. J. Evans, J. Ouyang, L. Favereau, J. Crassous, I. Fernández, J. Perles and N. Martín, Angew. Chem., Int. Ed., 2018, 57, 6774–6779 CrossRef CAS PubMed.
- V. Kumar, G. Venugopal, A. B. Jadhav, S. D. Dongre, R. Gonnade, J. Kumar, P. C. Ruer, B. Hupp, A. Steffen and S. S. Babu, Angew. Chem., Int. Ed., 2025, 64, e202422125 CrossRef CAS PubMed.
- G. Venugopal, V. Kumar, A. Badrinarayan Jadhav, S. D. Dongre, A. Khan, R. Gonnade, J. Kumar and S. Santhosh Babu, Chem. – Eur. J., 2024, 30, e202304169 CrossRef CAS PubMed.
- F. Gan, C. Shen, W. Cui and H. Qiu, J. Am. Chem. Soc., 2023, 145, 5952–5959 CrossRef CAS PubMed.
- P. Izquierdo-García, J. M. Fernández-García, S. Medina Rivero, M. Šámal, J. Rybáček, L. Bednárová, S. Ramírez-Barroso, F. J. Ramírez, R. Rodríguez, J. Perles, D. García-Fresnadillo, J. Crassous, J. Casado, I. G. Stará and N. Martín, J. Am. Chem. Soc., 2023, 145, 11599–11610 CrossRef PubMed.
- Molecular nanographenes: Synthesis, properties, and applications, ed. N. Martin and C. P. Nuckolls, Blackwell Verlag, Berlin, Germany, 2025 Search PubMed.
- P. Izquierdo-García, J. Lión-Villar, J. M. Fernández-García and N. Martín, Chem. Soc. Rev., 2025, 54, 11089–11104 RSC.
- G.-F. Huo, W.-T. Xu, J. Hu, Y. Han, W. Fan, W. Wang, Z. Sun, H.-B. Yang and J. Wu, Angew. Chem., Int. Ed., 2025, 64, e202416707 CrossRef CAS PubMed.
- Y. Hu, G. M. Paternò, X.-Y. Wang, X.-C. Wang, M. Guizzardi, Q. Chen, D. Schollmeyer, X.-Y. Cao, G. Cerullo, F. Scotognella, K. Müllen and A. Narita, J. Am. Chem. Soc., 2019, 141, 12797–12803 CrossRef CAS PubMed.
- J. Tan, X. Xu, J. Liu, S. Vasylevskyi, Z. Lin, R. Kabe, Y. Zou, K. Müllen, A. Narita and Y. Hu, Angew. Chem., Int. Ed., 2023, 62, e202218494 CrossRef CAS PubMed.
- Y.-Y. Ju, J.-F. Xing, Y.-M. Xie, X.-F. Li, X.-J. Zhao, G.-H. Nie, B. Zhang and Y.-Z. Tan, J. Am. Chem. Soc., 2025, 147, 45514–45522 CrossRef CAS PubMed.
- H. He, J. Lee, Z. Zong, N. Liu, Y. Noh, V. M. Lynch, J. Oh, J. Kim, J. L. Sessler and X.-S. Ke, Nat. Commun., 2025, 16, 1534 CrossRef CAS PubMed.
- R. N. Schaugaard, K. Raghavachari and L. Li, Inorg. Chem., 2018, 57, 10548–10556 CrossRef CAS PubMed.
- H. Yersin, Highly efficient OLEDs with phosphorescent materials, Wiley-VCH, Weinheim, 2008, ch 1 and 7 Search PubMed.
- Y. Zhang, W. Fu, X. Feng, Y. Ma, W. Yu, N. Yang and C. Shen, J. Mol. Struct., 2026, 1356, 145112 CrossRef CAS.
- X. Qiao, Q. Li, R. N. Schaugaard, B. W. Noffke, Y. Liu, D. Li, L. Liu, K. Raghavachari and L. Li, J. Am. Chem. Soc., 2017, 139, 3934–3937 CrossRef CAS PubMed.
- P. Vázquez-Domínguez, M. Horojat, E. Suits, F. J. F. de Córdova, N. Vanthuyne, D. Jacquemin, A. Ros and L. Favereau, Mater. Chem. Front., 2024, 8, 3799–3806 RSC.
- L. Norel, M. Rudolph, N. Vanthuyne, J. A. G. Williams, C. Lescop, C. Roussel, J. Autschbach, J. Crassous and R. Réau, Angew. Chem., Int. Ed., 2010, 49, 99–102 CrossRef CAS PubMed.
- E. Anger, M. Rudolph, C. Shen, N. Vanthuyne, L. Toupet, C. Roussel, J. Autschbach, J. Crassous and R. Réau, J. Am. Chem. Soc., 2011, 133, 3800–3803 CrossRef CAS PubMed.
- C. Shen, E. Anger, M. Srebro, N. Vanthuyne, K. K. Deol, T. D. Jefferson, G. Muller, J. A. G. Williams, L. Toupet, C. Roussel, J. Autschbach, R. Réau and J. Crassous, Chem. Sci., 2014, 5, 1915–1927 RSC.
- J. R. Brandt, X. Wang, Y. Yang, A. J. Campbell and M. J. Fuchter, J. Am. Chem. Soc., 2016, 138, 9743–9746 CrossRef CAS PubMed.
- Z.-P. Yan, X.-F. Luo, W.-Q. Liu, Z.-G. Wu, X. Liang, K. Liao, Y. Wang, Y.-X. Zheng, L. Zhou, J.-L. Zuo, Y. Pan and H. Zhang, Chem. – Eur. J., 2019, 25, 5672–5676 CrossRef CAS PubMed.
- A. Nowak-Król, P. T. Geppert and K. R. Naveen, Chem. Sci., 2024, 15, 7408–7440 RSC.
- M. Schnitzlein, K. Shoyama and F. Würthner, Chem. Sci., 2024, 15, 2984–2989 RSC.
- C. Shen, M. Srebro-Hooper, M. Jean, N. Vanthuyne, L. Toupet, J. A. G. Williams, A. R. Torres, A. J. Riives, G. Muller, J. Autschbach and J. Crassous, Chem. – Eur. J., 2017, 23, 407–418 CrossRef CAS PubMed.
- F. Full, Q. Wölflick, K. Radacki, H. Braunschweig and A. Nowak-Król, Chem. – Eur. J., 2022, 28, e202202280 CrossRef CAS PubMed.
- F. Full, A. Artigas, K. Wiegand, D. Volland, K. Szkodzińska, Y. Coquerel and A. Nowak-Król, J. Am. Chem. Soc., 2024, 146, 29245–29254 CrossRef CAS PubMed.
- A. F. Palermo, B. S. Y. Chiu, P. Patel and S. A. L. Rousseaux, J. Am. Chem. Soc., 2023, 145, 24981–24989 CAS.
- R. Nagpal, S. Arora, P. Ponnan, K. Nisa, A. Dandia and S. M. Singh Chauhan, ChemistrySelect, 2019, 4, 8444–8449 CrossRef CAS.
- R. H. Martin, Angew. Chem., Int. Ed. Engl., 1974, 13, 649–660 CrossRef.
- A. R. Krappe, J. C. Mayer, W. Zhang, L. M. Filla, G. Ligorio, F. Hermerschmidt, L. S. Eitelhuber, A. Güttler, M. Weber, B. Paulus, U. Resch-Genger, E. J. W. List-Kratochvil and S. Eigler, Chem. – Eur. J., 2025, 31, e202500742 CrossRef CAS PubMed.
- D. N. Kozhevnikov, V. N. Kozhevnikov, M. Z. Shafikov, A. M. Prokhorov, D. W. Bruce and J. A. Gareth Williams, Inorg. Chem., 2011, 50, 3804–3815 CrossRef CAS PubMed.
- P. Pander, R. Daniels, A. V. Zaytsev, A. Horn, A. Sil, T. J. Penfold, J. A. G. Williams, V. N. Kozhevnikov and F. B. Dias, Chem. Sci., 2021, 12, 6172–6180 RSC.
- Y. Liu, J. Cerezo, G. Mazzeo, N. Lin, X. Zhao, G. Longhi, S. Abbate and F. Santoro, J. Chem. Theory Comput., 2016, 12, 2799–2819 CrossRef CAS PubMed.
- T. Mori, Angew. Chem., Int. Ed., 2024, 63, e202319702 CrossRef CAS PubMed.
- CCDC 2502439: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2pzzvj.
| This journal is © The Royal Society of Chemistry 2026 |