Open Access Article
Open Access ArticleMechanochemical applications of magnesium nitride to fullerene chemistry: synthesis of pyrroline-fused tetra-functionalized [60]fullerene derivatives
Jun-Shen Chena,
Wen-Jie Qiu a,
Li-Feng Guoa and
Guan-Wu Wang
a,
Li-Feng Guoa and
Guan-Wu Wang *abc
*abc
aDepartment of Chemistry, University of Science and Technology of China, Hefei, Anhui 230026, P. R. China. E-mail: gwang@ustc.edu.cn
bKey Laboratory of Functional Molecular Solids, Ministry of Education, Anhui Laboratory of Molecule-Based Materials, and School of Chemistry and Materials Science, Anhui Normal University, Wuhu, Anhui 241002, P. R. China
cState Key Laboratory of Natural Product Chemistry, Lanzhou University, Lanzhou, Gansu 730000, P. R. China
First published on 4th March 2026
Abstract
A novel application of magnesium nitride (Mg3N2) for the synthesis of pyrroline-fused tetra-functionalized [60]fullerene (C60) derivatives is reported under solvent-free ball-milling conditions. This approach represents the first mechanochemical method to regioselectively achieve tetra-functionalization of C60. Notably, the present protocol enables efficient incorporation of nitrogen from Mg3N2 directly into fullerene products.
Mechanochemical organic transformations have become increasingly important over the past three decades.1 One of the charms of mechanochemistry is the ability to synthesize and functionalize compounds such as fullerenes that are insoluble or poorly soluble in common solvents. It is well known that fullerene derivatives have great potential applications in materials, biology and nanoscience.2 However, the functionalization of fullerenes is severely limited due to their poor solubility in common organic solvents. Thus, the emergence of mechanochemistry opens up more possibilities for constructing fullerene derivatives.3 The application of mechanochemistry to fullerene chemistry was first reported in 1996.4 To date, various syntheses of di- and multi-functionalized fullerenes have been reported, but methods for synthesizing multi-functionalized fullerenes in solutions often yield multiple regioisomers.3a,5 Therefore, it is of great significance to develop methods for the synthesis of single isomers of multi-adducts of [60]fullerene (C60) under solvent-free conditions.
Some inorganic salts, such as calcium carbide, have been used in organic synthesis as solid alternatives to gaseous reagents, despite their insolubility in organic solvents.6 Magnesium nitride (Mg3N2), a commercially available and stable solid, has significant potential to replace ammonia in organic synthesis by serving as a solid surrogate. However, up to now, there are only a few reports on both solution-based and solvent-free applications of Mg3N2.7 Furthermore, reactions performed in solution phases required high temperatures and long reaction times owing to poor solubility (Scheme 1a).7a–d Nevertheless, Mg3N2 conversion does not always require a solvent and may even be accelerated under solvent-free conditions. In 2021, González and Menéndez reported the first mechanochemical synthesis of primary amides, but still used a large quantity of ethanol as a proton source (Scheme 1b).7e
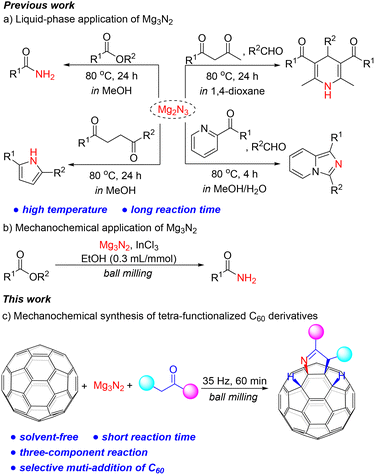 | ||
| Scheme 1 (a) Liquid-phase and (b, c) mechanochemical applications of Mg3N2 for the synthesis of N-containing compounds. | ||
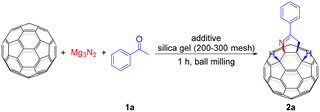
Motivated by the potential of fullerene mechanochemistry, we aim to bypass the poor solubility of both fullerenes and Mg3N2 by employing mechanical milling to synthesize C60 derivatives quickly and efficiently. Herein, we report the mechanosynthesis of tetra-functionalized C60 derivatives featuring a pyrroline heterocycle fused to a [6,6]-bond via one-pot three-component reaction. Notably, this is also the first regioselective synthesis of multi-functionalized derivatives of C60 under solvent-free conditions.
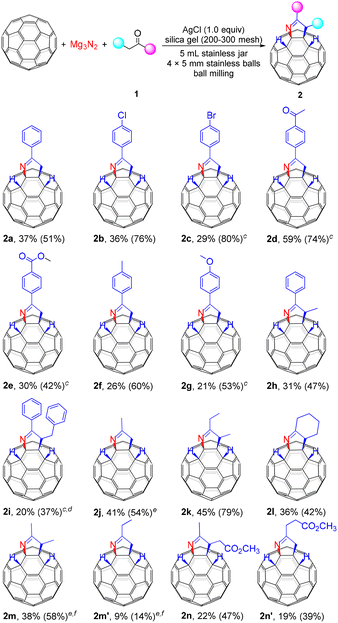
We began optimizing the conditions for the mechanochemical reaction of C60 with Mg3N2 and acetophenone. Initially, a mixture of C60 (0.04 mmol), 5.0 equiv. of Mg3N2 and 10.0 equiv. of acetophenone was milled in the presence of 1.0 equiv. of silver chloride (AgCl) and 36.0 mg of silica gel (200–300 mesh) as grinding auxiliary8,9 at a frequency of 35 Hz at room temperature for 1 h. Pleasingly, a tetra-functionalized product 2a was obtained regioselectively in 27% yield (Table 1, entry 1). Increasing the amount of acetophenone to 15.0 equiv. significantly improved the yield to 37%. However, further increasing the quantity of acetophenone to 20.0 equiv. gave a slightly lower yield (Table 1, entries 2 and 3). In addition, neither increasing nor decreasing the amount of Mg3N2 had any benefit to yields (Table 1, entries 4 and 5). We also investigated the amount of AgCl. When the reaction was performed without AgCl, the product yield was reduced to 14% (Table 1, entry 6). What's more, reactions with 0.5 and 1.5 equiv. of AgCl gave lower yields of 26% and 29%, respectively (Table 1, entries 7 and 8). Based on the above results, it is clear that the AgCl is not a determining factor. To exclude the effect of the chloride ion in AgCl, sodium chloride (NaCl) was used to replace AgCl, but it did not promote the reaction (Table 1, entry 9). Next, it was found that the use of zinc chloride (ZnCl2) and magnesium chloride (MgCl2) as Lewis acids provided a modest enhancement of the reaction compared to the non-additive conditions (Table 1, entries 10 and 11 vs. entry 6). Therefore, AgCl was considered to be a very weak Lewis acid interacting with the carbonyl group in the substrate. Some commonly used silver salts were also screened to improve the yield. Among them, silver bromide (AgBr) and silver nitrate (AgNO3) achieved yields of 22% and 23%, respectively (Table 1, entries 12 and 13). However, silver carbonate (Ag2CO3) and silver acetate (AgOAc), which possess weakly basic counteranions, only gave a trace yield and a 9% yield, respectively (Table 1, entries 14 and 15). Furthermore, without the use of silica gel as a grinding auxiliary, only a small amount of 2a could be obtained (Table 1, entry 16). In addition, we also tried to decrease the milling frequency to obtain a higher yield. However, this resulted in a slight reduction in yield (Table 1, entry 17). Replacing Mg3N2 with the more reactive calcium nitride (Ca3N2) led to the complete consumption of C60 but yielded no product (Table 1, entry 18).
| Entry | Molar ratio of C60/Mg3N2/1a | Additive (equiv.) | Yield of 2ab (%) |
|---|---|---|---|
| a Unless otherwise noted, all reactions were performed with C60 (0.04 mmol), Mg3N2, 1a, additive and silica gel (36.0 mg, 200–300 mesh) together with four stainless balls (5 mm in diameter) in a stainless jar (5 mL) and were milled vigorously (35 Hz) at room temperature for 1 h.b Isolated yields were based on C60. Values in parentheses were based on consumed C60.c No silica gel.d The milling frequency was 30 Hz.e Ca3N2 was used instead of Mg3N2. | |||
| 1 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
AgCl (1.0) | 27 (36) |
| 2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 15 15 |
AgCl (1.0) | 37 (51) |
| 3 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
AgCl (1.0) | 31 (46) |
| 4 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (1.0) | 28 (39) |
| 5 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (1.0) | 32 (39) |
| 6 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
0 | 14 (15) |
| 7 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (0.5) | 26 (30) |
| 8 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (1.5) | 29 (37) |
| 9 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
NaCl (1.0) | 12 (13) |
| 10 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
ZnCl2 (1.0) | 17 (71) |
| 11 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
MgCl2 (1.0) | 23 (74) |
| 12 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgBr (1.0) | 22 (25) |
| 13 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgNO3 (1.0) | 23 (36) |
| 14 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
Ag2CO3 (0.5) | Trace |
| 15 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgOAc (1.0) | 9 (9) |
| 16c | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (1.0) | Trace |
| 17d | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (1.0) | 29 (39) |
| 18e | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
AgCl (1.0) | 0 |
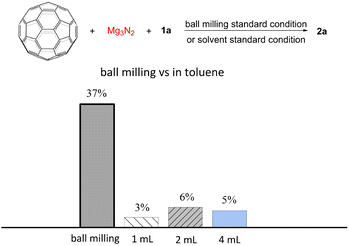
Having optimized the reaction under solvent-free conditions, we then investigated its performance in different volumes of toluene (Fig. 1). As illustrated in Fig. 1, the liquid-phase reaction in toluene for 1 h at room temperature gave the product 2a in only 3–6% yields. These results demonstrate the unique advantages of mechanochemical methods in the synthesis of fullerene derivatives. By eliminating solvents, mechanochemistry not only enables higher reactant concentrations but also overcomes the challenge of poor solubility of fullerenes, thereby significantly enhancing the reaction efficiency.
With the optimized reaction conditions in hand, a series of ketones were evaluated to investigate the substrate scope of this three-component reaction (Scheme 2). Firstly, acetophenones (1b and 1c) with electron-withdrawing substituents (Cl and Br) at the para-position were well tolerated in this transformation and afforded desired products 2b and 2c in yields of 36% and 29%, respectively. It is worth mentioning that the dosage was reduced to 10.0 equiv. when using the solid 1c. The same is true for other solid substrates. Satisfyingly, 1,4-diacetylbenzene (1d) possessing two acetyl groups afforded product 2d in 59% yield. When methyl 4-acetylbenzoate (1e) was used, the ester group remained intact, affording the desired product 2e in 30% yield. With the enhancement of the electron-donating effect, p-methyl- and p-methoxy-substituted acetophenones (1f and 1g) provided 2f and 2g in yields of 26% and 21%, respectively. Propiophenone (1h) and β-phenyl propiophenone (1i) could also participate in this reaction and gave the desired products 2h and 2i in 31% and 20% yields, respectively. Next, we examined the compatibility of aliphatic ketones. It was found that aliphatic ketones were well suitable for this reaction. Considering that acetone (1j) is highly volatile, 20.0 equiv. of acetone were used and provided the desired product 2j in 41% yield. 3-Pentanone (1k) and cyclohexanone (1l) were also amenable to this transformation, affording products 2k and 2l in 45% and 36% yields, respectively. When the unsymmetrical 2-butanone (1m) was tested, two isomers 2m and 2m′ were isolated in 38% and 9% yields, respectively. Satisfactorily, the ester group in methyl acetylacetonate was tolerated and did not participate in the reaction, resulting in a pair of isomers 2n and 2n′ with yields of 22% and 19%, respectively.
To determine the structures of products 2a–n′, they were characterized by matrix-assisted laser desorption/ionization time-of-flight high-resolution mass spectrometry (MALDI-TOF HRMS), 1H and 13C nuclear magnetic resonance (NMR), Fourier-transform infrared spectroscopy (FTIR), ultraviolet–visible (UV–vis), cyclic voltammetry (CV) and differential pulse voltammetry (DPV) spectra (see SI). Their 1H NMR spectra displayed the expected chemical shifts, and there was coupling between the two hydrogen atoms, with a coupling constant of 1.7–1.8 Hz, indicating that they were in the para-position on the six-membered ring. In the 13C NMR spectra, there were at least 50 peaks for the 56 sp2 carbons of the C60 cage and 4 peaks for the four sp3 carbons of the C60 moiety. Furthermore, the structure of 2c was unambiguously established by X-ray crystallography (Fig. 2).
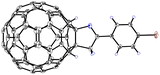 | ||
| Fig. 2 Oak ridge thermal ellipsoid plot (ORTEP) diagrams for compound 2c with 20% thermal ellipsoids. The solvent molecules are omitted for clarity. | ||
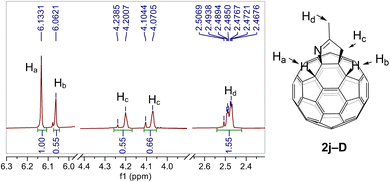
To investigate the potential role of Mg3N2 as an ammonia surrogate, when it was directly replaced with aqueous ammonia (NH3·H2O), the target product was obtained in only 3% yield. This result indicated that the direct condensation of ammonia, potentially generated from Mg3N2, with ketones was not the dominant pathway. Therefore, the nitride anion may directly condense with the used ketone or act as a base to remove the α-hydrogen of ketones or derivatives. Next, we further investigated the origin of the hydrogen atoms attached to the fullerene cage. Deuterated acetone (D, 99%) was selected as the starting material. The resulting product 2j-D was analyzed by 1H NMR spectroscopy. It was found that about equal amounts of deuterium atoms at the Hc and Hd positions underwent D/H exchange during the milling process, promoted by the strong base Mg3N2 (Fig. 3). In addition, the H/D ratio at Hb was comparable to that for each one at Hc and Hd positions, suggesting that an H/D scrambling equilibrium at these positions had been established during the reaction. This result indicates the presence of proton sources in the reaction system, likely originating from Si–OH groups of silica gel or residual water. Interestingly, the H/D ratio at Ha was found to be higher than that at Hb. The assignments of Ha and Hb were established based on the HMBC spectra of 2a and 2j (Fig. S54 and S83).
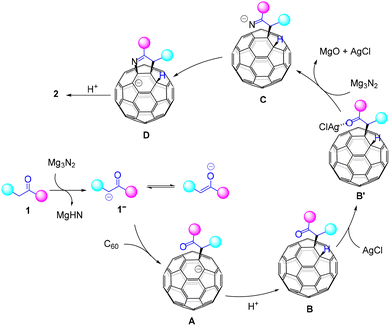
Based on the control experiments, a plausible reaction mechanism is proposed (Scheme 3). First, deprotonation at the α-carbon of ketone 1 by Mg3N2 generates carbanion 1−, which undergoes nucleophilic addition to C60 to form fullerenyl anion A, with Mg2+ serving as the countercation. Protonation of the anionic species A by the proton source present in the reaction system provides B. The nitride anion in Mg3N2 condenses with the carbonyl group in B with the assistance of AgCl, leading to the formation of the anionic ketoimine species C via the complex B′. This imine anion undergoes nucleophilic addition to the fullerene cage, providing an anionic intermediate D. The countercation in both C and D is likely to be a Mg2+ species. Finally, D is protonated by the proton source present in the reaction mixture and/or during workup process10 to afford the target product 2.
In summary, we have developed a new method for synthesizing pyrroline-fused tetra-functionalized C60 derivatives by employing Mg3N2 as a nitrogen source and base under mechanochemical conditions. This approach overcomes the poor solubility of both fullerenes and Mg3N2, enabling the efficient and regioselective synthesis of tetra-functionalized C60 derivatives in a short time of 1 h under ambient air and at room temperature. This mechanochemical strategy broadens the utility of Mg3N2 and expands the toolbox of incorporating nitrogen atoms into organic compounds.
We are grateful for financial support from the National Natural Science Foundation of China (22271271, 21372211).
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the finding of this study are available in this article and its supplementary information (SI). Supplementary information: detailed experimental procedures, characterisation data, NMR, UV-vis, CV and DPV spectra of compounds 2a–n′ and X-ray data of 2c. See DOI: https://doi.org/10.1039/d6cc00518g.CCDC 2525038 contains the supplementary crystallographic data for this paper.11
References
- (a) T. Friščić, Chem. Soc. Rev., 2012, 41, 3493–3510 RSC; (b) G.-W. Wang, Chem. Soc. Rev., 2013, 42, 7668–7700 RSC; (c) J. G. Hernández and C. Bolm, J. Org. Chem., 2017, 82, 4007–4019 CrossRef PubMed; (d) C. Bolm and J. G. Hernández, Angew. Chem., Int. Ed., 2019, 58, 3285–3299 CrossRef CAS PubMed; (e) H. Wang, P. Ying, J. Yu and W. Su, Chin. J. Org. Chem., 2021, 41, 1897–1924 CrossRef CAS; (f) P. Ying, J. Yu and W. Su, Adv. Synth. Catal., 2021, 363, 1246–1271 CrossRef CAS; (g) A. C. Jones, J. A. Leitch, S. E. Raby-Buck and D. L. Browne, Nat. Synth., 2022, 1, 763–775 CrossRef CAS; (h) V. Martinez, T. Stolar, B. Karadeniz, I. Brekalo and K. Uzarevic, Nat. Rev. Chem., 2023, 7, 51–65 CrossRef CAS PubMed; (i) R. Liu, X. He, T. Liu, X. Wang, Q. Wang, X. Chen and Z. Lian, Chem. – Eur. J., 2024, 30, e202401376 CrossRef CAS PubMed.
- (a) M. Chen, R. Guan and S. Yang, Adv. Sci., 2019, 6, 1800941 CrossRef PubMed; (b) K. Harano and E. Nakamura, Acc. Chem. Res., 2019, 52, 2090–2100 CrossRef CAS PubMed; (c) T. Umeyama and H. Imahori, Acc. Chem. Res., 2019, 52, 2046–2055 CrossRef CAS PubMed; (d) Z. Xing, S.-H. Li and S. Yang, Small Struct., 2022, 3, 2200012 CrossRef CAS.
- (a) S.-E. Zhu, F. Li and G.-W. Wang, Chem. Soc. Rev., 2013, 42, 7535–7570 RSC; (b) G.-W. Wang, Chin. J. Chem., 2021, 39, 1797–1803 CrossRef CAS.
- G.-W. Wang, Y. Murata, K. Komatsu and T. S. M. Wan, Chem. Commun., 1996, 2059–2060 RSC.
- (a) H. W. Liu, H. Xu, G. Shao and G.-W. Wang, Org. Lett., 2019, 21, 2625–2628 CrossRef CAS PubMed; (b) G. Shao, C. Niu, H. W. Liu, H. Yang, J. S. Chen, Y. R. Yao, S. Yang and G.-W. Wang, Org. Lett., 2023, 25, 1229–1234 CrossRef CAS PubMed; (c) H. Yang, S.-Q. Ye, G. Shao, J.-S. Chen and G.-W. Wang, Synlett, 2023, 34, 2047–2051 CrossRef CAS; (d) G. Shao, Y.-Y. Liu, C. Niu, Z.-C. Yin, S.-Q. Ye, Y.-R. Yao, M. Chen, J.-S. Chen, X.-L. Xia, S. Yang and G.-W. Wang, Chem. Sci., 2024, 15, 14899–14904 Search PubMed.
- K. S. Rodygin, M. S. Ledovskaya, V. V. Voronin, K. A. Lotsman and V. P. Ananikov, Eur. J. Org. Chem., 2021, 43–52 Search PubMed.
- (a) G. E. Veitch, K. L. Bridgwood and S. V. Ley, Org. Lett., 2008, 10, 3623–3625 CrossRef CAS PubMed; (b) K. L. Bridgwood, G. E. Veitch and S. V. Ley, Org. Lett., 2008, 10, 3627–3629 CrossRef CAS PubMed; (c) G. E. Veitch, K. L. Bridgwood, K. Rands-Trevor and S. V. Ley, Synlett, 2008, 2597–2600 CAS; (d) S. Long, M. Panunzio, A. Petroli, W. Qin and Z. Xia, Synthesis, 2011, 1071–1078 Search PubMed; (e) S. G. Patil, J. S. Jadhav and S. T. Sankpal, RSC Adv., 2020, 10, 11808–11815 Search PubMed; (f) J. Gómez-Carpintero, J. D. Sánchez, J. F. González and J. C. Menéndez, J. Org. Chem., 2021, 86, 14232–14237 CrossRef PubMed.
- (a) Z. Li, Z. Jiang and W. Su, Green Chem., 2015, 17, 2330–2334 RSC; (b) C. S. Yadav, R. Suhasini, V. Thiagarajan, D. Velmurugan and S. Kannadasan, ChemistrySelect, 2018, 3, 12576–12581 CrossRef CAS; (c) R. Aggarwal, M. Hooda, P. Kumar and G. Sumran, Lett. Org. Chem., 2021, 18, 532–537 CrossRef CAS.
- (a) R. Thorwirth, A. Stolle and B. Ondruschka, Green Chem., 2010, 12, 985–991 Search PubMed; (b) H.-G. Li and G.-W. Wang, J. Org. Chem., 2017, 82, 6341–6348 CrossRef CAS PubMed; (c) D. Kong, M. M. Amer and C. Bolm, Green Chem., 2022, 24, 3125–3129 RSC; (d) D. Kong and C. Bolm, Green Chem., 2022, 24, 6476–6480 RSC; (e) C. Grundke, J. Groß, N. Vierengel, J. Sirleaf, M. Schmitz, L. Krieger and T. Opatz, Org. Biomol. Chem., 2023, 21, 644–650 RSC; (f) C.-M. Chen, J.-X. Chen and C. T. To, Green Chem., 2023, 25, 2559–2562 RSC; (g) K. Xiang, P. Ying, T. Ying, W. Su and J. Yu, Green Chem., 2023, 25, 2853–2862 RSC; (h) S. Saha, A. A. Bhosle, A. Chatterjee and M. Banerjee, J. Org. Chem., 2023, 88, 10002–10013 CrossRef CAS PubMed; (i) S. Saha, A. B. Pinheiro, A. Chatterjee, Z. T. Bhutia and M. Banerjee, Green Chem., 2024, 26, 5879–5889 RSC.
- H.-S. Lin, Y. Matsuo, J.-J. Wang and G.-W. Wang, Org. Chem. Front., 2017, 4, 603–607 RSC.
- CCDC 2525038: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qrhvv.
| This journal is © The Royal Society of Chemistry 2026 |