Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Concise synthesis of triasteranones from barbaralones
Serena Yeungab,
Emily G. Allenab,
Kieran D. Jones ab,
Stephen P. Argent
ab,
Stephen P. Argent b and
Hon Wai Lam
b and
Hon Wai Lam *ab
*ab
aThe GSK Carbon Neutral Laboratories for Sustainable Chemistry, University of Nottingham, Jubilee Campus, Triumph Road, Nottingham, NG7 2TU, UK. E-mail: hon.lam@nottingham.ac.uk
bSchool of Chemistry, University of Nottingham, University Park, Nottingham, NG7 2RD, UK
First published on 25th March 2026
Abstract
Triasteranes are underexplored, topologically appealing polycyclic compounds with a star-like shape that have been challenging to synthesize. Herein, we report the concise syntheses of triasteranone derivatives from readily accessible barbaralones.
Bridged or caged polycyclic hydrocarbons are of significant interest because of their unique, often topologically appealing structures.1 Their rigid, often highly strained frameworks are useful for studying bonding and reactivity. Their challenging synthesis continues to inspire advances in synthetic methodology, and they have applications in medicinal chemistry,1b,c supramolecular chemistry and materials science,1c and as energetic materials.2
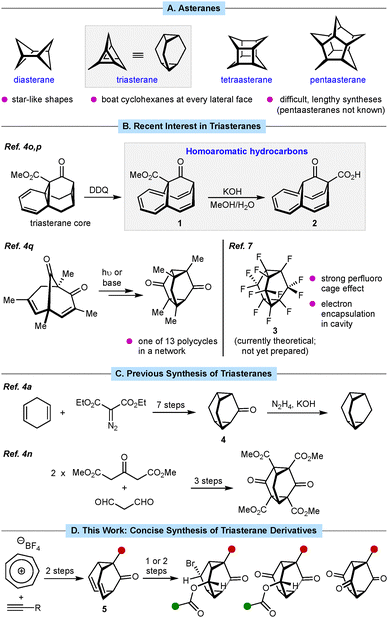
Asteranes are interesting polycyclic hydrocarbons consisting of two carbon–carbon bonds (in the case of diasterane) or two cycloalkanes linked by a carbon atom at every position (Fig. 1A).3–6 This arrangement results in a star-like shape with every lateral face being a cyclohexane in a boat conformation. First described by Musso and co-workers in 1965,4a asteranes have been studied by the Musso group and others.3–6 Recently, there has been interest in triasteranes as intermediates in the synthesis of stable, neutral, homoaromatic hydrocarbons such as 1 and 2,4o,p and as a member of a network of 13 distinct polycycles accessed by “shapeshifting” skeletal evolution from a common starting material (Fig. 1B).4q DFT calculations have also suggested that perfluorinated triasterane 3 (which has not yet been prepared) exhibits a strong perfluoro cage effect due the low-energy,7 overlapping, inward-pointing lobes of its C–F σ* orbitals, and should therefore be able to encapsulate an electron.7,8
Despite their intriguing structures, triasteranes are challenging to synthesize.4 For example, the synthesis of triasteranone (4) requires seven steps from cyclohexa-1,4-diene and diethyl diazomalonate, which was then converted into triasterane by a Wolff–Kishner reduction (Fig. 1C, top).4a Grohmann and co-workers reported the three-step synthesis of the triasterane skeleton from commercial compounds, but only one example was described and the product is too densely functionalized for wide synthetic applicability (Fig. 1C, bottom).4n The difficulty in preparing triasteranes has hindered the study of their properties, reactivities, and applications, and more efficient methods are therefore required. Herein, we report a concise synthesis of substituted triasteranone derivatives in just three or four steps from commercial compounds (Fig. 1D).
Our approach exploits barbaralones 5, which are fluxional molecules that undergo Cope rearrangement between constitutional isomers,9 as versatile triasteranone precursors (Fig. 1D). Although Vedejs demonstrated the viability of this concept through the dibromination of barbaralone to give diastereomeric triasteranone dibromides in 72% combined yield, only one example was reported and there were no follow-up studies.4d,f This limitation likely stemmed from the then-challenging synthesis of barbaralones. However, in 2016, the Echavarren group reported a practical two-step synthesis of barbaralones from commercially available tropylium tetrafluoroborate and terminal alkynes (Fig. 1D).10a We recognized that integrating these methods could provide a streamlined, general access to triasteranones, thus opening the door to greater investigation of these underexplored compounds.
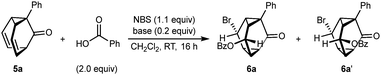
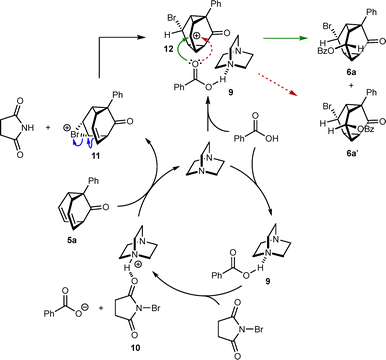
We first targeted triasteranones containing two different functional groups by the bromoesterification11 of barbaralones (Table 1). No reaction occurred when barbaralone 5a10a was stirred with NBS (1.1 equiv.) and benzoic acid (2.0 equiv.) in CH2Cl2 at room temperature for 16 h (entry 1). However, repeating the reaction with the addition of DABCO (20 mol%) gave a 6.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 inseparable mixture of diastereomeric triasteranones 6a and 6a′ in 92% NMR yield (entry 2).12 The relative configuration of the major isomer 6a was assigned by comparison of its NMR data with that of 6b, the stereochemistry of which was determined by X-ray crystallography (Scheme 1).12 For a structural analysis of 6b that covers substituent exit vector angles and distances between substituents, see the SI. The stereochemistry of 6a′ was assigned from NOESY NMR data (see the SI). Other bases such as quinuclidine, DBU, and pyridine gave inferior results (entries 3–5).
1 inseparable mixture of diastereomeric triasteranones 6a and 6a′ in 92% NMR yield (entry 2).12 The relative configuration of the major isomer 6a was assigned by comparison of its NMR data with that of 6b, the stereochemistry of which was determined by X-ray crystallography (Scheme 1).12 For a structural analysis of 6b that covers substituent exit vector angles and distances between substituents, see the SI. The stereochemistry of 6a′ was assigned from NOESY NMR data (see the SI). Other bases such as quinuclidine, DBU, and pyridine gave inferior results (entries 3–5).
| Entry | Base | Yieldb (%) | dr (6a:6a′)c |
|---|---|---|---|
| a Conducted with 5a (0.10 mmol) in CH2Cl2 (1 mL).b Combined yield of 6a and 6a′ as determined by 1H NMR analysis of the crude reaction mixtures using 1,3,5-trimethoxybenzene as an internal standard.c Determined by 1H NMR analysis of the crude reaction mixtures. | |||
| 1 | None | — | |
| 2 | DABCO | 92 | 6.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 3 | Quinuclidine | 69 | 4.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 4 | DBU | 66 | 5.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 5 | Pyridine | 61 | 5.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
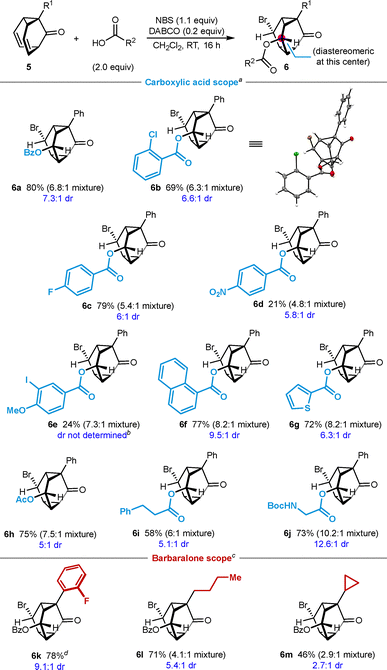
Barbaralone 5a reacted with various (hetero)aromatic carboxylic acids to give triasteranones containing 2-chlorophenyl (6b), 4-fluorophenyl (6c), 4-nitrophenyl (6d), 3-iodo-4-methoxphenyl (6e), 1-naphthyl (6f), or 2-thienyl groups (6g). The diastereoselectivities of the reactions as determined by 1H NMR analysis of the crude mixtures ranged from 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr (6c) to 10
1 dr (6c) to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr (6g). The products were isolated in up to 79% yield as inseparable mixtures of diastereomers in ratios between 4.8
1 dr (6g). The products were isolated in up to 79% yield as inseparable mixtures of diastereomers in ratios between 4.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (6d) and 8.2
1 (6d) and 8.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (6f). The yields were lower in the reactions producing 6d (21% yield) and 6e (24% yield) as significant starting materials remained. Aliphatic carboxylic acids such as acetic acid, 3-phenylpropionic acid, and N-Boc glycine also reacted with 5a to give triasteranones 6h–6j, respectively, with the latter giving the highest diastereoselectivity observed (12.6
1 (6f). The yields were lower in the reactions producing 6d (21% yield) and 6e (24% yield) as significant starting materials remained. Aliphatic carboxylic acids such as acetic acid, 3-phenylpropionic acid, and N-Boc glycine also reacted with 5a to give triasteranones 6h–6j, respectively, with the latter giving the highest diastereoselectivity observed (12.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr). Barbaralones containing 2-fluorophenyl, n-butyl, or cyclopropyl groups successfully reacted with benzoic acid to give triasteranones 6k–6m, respectively, in 46–78% yield. Comparison of the reactions producing 6a and 6k–6m shows that sterically more hindering barbaralone substituents result in higher diastereoselectivities.
1 dr). Barbaralones containing 2-fluorophenyl, n-butyl, or cyclopropyl groups successfully reacted with benzoic acid to give triasteranones 6k–6m, respectively, in 46–78% yield. Comparison of the reactions producing 6a and 6k–6m shows that sterically more hindering barbaralone substituents result in higher diastereoselectivities.
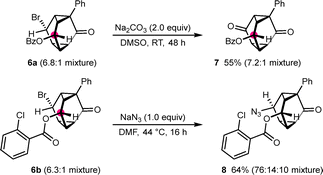
Further functionalization of the products was then investigated (Scheme 2). Reaction of triasteranone 6a (a 6.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomeric mixture) with DMSO and Na2CO3 (2.0 equiv.) at room temperature resulted in Kornblum oxidation to give triasteranedione 7 in 55% yield. Treatment of 6b (6.3
1 diastereomeric mixture) with DMSO and Na2CO3 (2.0 equiv.) at room temperature resulted in Kornblum oxidation to give triasteranedione 7 in 55% yield. Treatment of 6b (6.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of diastereomers) with NaN3 (1.0 equiv.) in DMF at 44 °C for 16 h gave azide 8 in 64% yield as a 76
1 mixture of diastereomers) with NaN3 (1.0 equiv.) in DMF at 44 °C for 16 h gave azide 8 in 64% yield as a 76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 mixture of diastereomers. The stereochemistry of the major diastereomer of 8 is consistent with nucleophilic substitution of the major diastereomer of 6b by an SN2 pathway. However, the formation of two other diastereomers suggests that other mechanisms of nucleophilic substitution could also be operative (see the SI).
10 mixture of diastereomers. The stereochemistry of the major diastereomer of 8 is consistent with nucleophilic substitution of the major diastereomer of 6b by an SN2 pathway. However, the formation of two other diastereomers suggests that other mechanisms of nucleophilic substitution could also be operative (see the SI).
A tentative mechanism for the bromoesterifications, using barbaralone 5a and benzoic acid as reactants, is shown in Scheme 3. First, hydrogen bonding of DABCO to benzoic acid gives 9, which reacts with NBS to give benzoate anion and the more electrophilic brominating agent 10, in which NBS is hydrogen-bonded to DABCO ammonium ion. This activation is essential, as evidenced by the lack of reaction of 5a with NBS and benzoic acid without DABCO (Table 1, entry 1), which is also consistent with the greater acidity of DABCO ammonium ion (pKa ∼ 8.9 in DMSO13) compared with benzoic acid (pKa ∼ 11.0 in DMSO14). Bromination of 5a with 10 liberates succinimide and gives cyclic bromonium ion 11, which is ring-opened by the remaining alkene to form triasteranyl carbocation 12. The diastereoselectivity depends on the base used (Table 1), which suggests the base is directly involved in the C–O bond-forming step. We propose that nucleophilic attack of carbocation 12 occurs from the carbonyl group of complex 9 to give diastereomeric triasteranones 6a and 6a′. However, we do not rule out other mechanisms.15
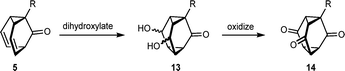
We next investigated whether barbaralones 5 could be dihydroxylated to give triasteranone diols 13, which could then be oxidized to give substituted triasteranetriones 14 (Scheme 4). The parent, unsubstituted triasteranetrione (14 with R = H) was first prepared by McDonald and Dreiding in the 1970s using lengthy routes,4g,h and it has been used to study the effect of π-accepting substituents on the average C–C bond length within cyclopropanes.4l,16 However, to our knowledge, substituted triasteranetriones have not been prepared previously.
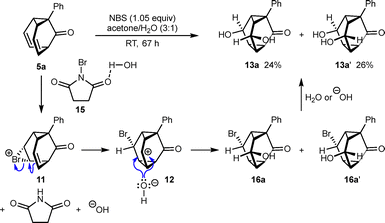
Our approach for the dihydroxylation of barbaralones employs NBS and water in the hope that the initial bromohydroxylation products 16 could react further with water to give diols 13 (Scheme 5). Indeed, reaction of barbaralone 5a with NBS (1.05 equiv.) in acetone/H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at room temperature for 67 h gave diastereomeric diols 13a (24% yield) and 13a′ (26% yield).12 In contrast to the bromoesterifications (Scheme 1), no base is required. Also, no reaction occurs when 5a is treated with NBS in acetone in the absence of water. A possible mechanism for this process follows a similar pathway to that shown in Scheme 3 to give triasteranyl carbocation 12, but here the brominating agent is likely to be 15, where NBS is activated by hydrogen bonding to water. Attack of 12 by hydroxide anion gives diastereomeric bromoalcohols 16a and 16a′. Finally, nucleophilic substitution of the bromides with water or hydroxide anion, most likely by an SN2 mechanism, gives diols 13a and 13a′.
1) at room temperature for 67 h gave diastereomeric diols 13a (24% yield) and 13a′ (26% yield).12 In contrast to the bromoesterifications (Scheme 1), no base is required. Also, no reaction occurs when 5a is treated with NBS in acetone in the absence of water. A possible mechanism for this process follows a similar pathway to that shown in Scheme 3 to give triasteranyl carbocation 12, but here the brominating agent is likely to be 15, where NBS is activated by hydrogen bonding to water. Attack of 12 by hydroxide anion gives diastereomeric bromoalcohols 16a and 16a′. Finally, nucleophilic substitution of the bromides with water or hydroxide anion, most likely by an SN2 mechanism, gives diols 13a and 13a′.
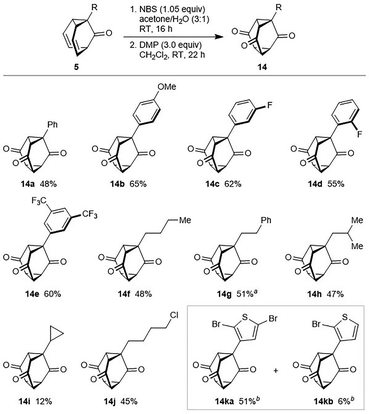
Subsequently, we found the reaction time could be reduced to 16 h, and oxidation of the resulting crude mixture of diols 13a and 13a′ with Dess–Martin periodinane (DMP, 3.0 equiv.) then gave triasteranetrione 14a in 48% yield over two steps (Scheme 6).12 The scope of this process was investigated using different barbaralones,10 which gave triasteranetriones in up to 65% yield over two steps. As well as 14a, the process can be used to prepare triasteranetriones with other aromatic substituents such as 4-methoxyphenyl (14b), 3-fluorophenyl (14c), 2-fluorophenyl (14d), and 3,5-bis(trifluoromethyl)phenyl groups (14e). Triasteranetriones with n-butyl (14f), 2-phenylethyl (14g), isobutyl (14h), cyclopropyl (14i), or 4-chlorobutyl groups (14j) can also be prepared, although the yield of 14i was only 12% because it was accompanied by a complex mixture of unidentified products and it was difficult to extract from the aqueous layer during workup. A 3-thienyl group in the barbaralone was found to undergo competitive bromination, but the use of 3.0 equivalents of NBS in the first step gave dibrominated thiophene-containing triasteranetrione 14ka12 in 51% yield and monobrominated product 14kb in 6% yield.
In summary, we have described the synthesis of substituted triasteranones, triasteranediones, and triasteranetriones from readily accessible barbaralones. At just three or four steps, this route provides access to triasteranones in a much more concise fashion compared with previous methods,4 which enables more extensive investigation of these fascinating compounds.
This work was supported by the Engineering and Physical Sciences Research Council (grant number EP/T517902/1) and the University of Nottingham.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article has been included as part of the supplementary information (SI) and the Nottingham Research Data Management Repository at: http://doi.org/10.17639/12985. Supplementary information: experimental procedures, characterization data for new compounds, and copies of NMR spectra. See DOI: https://doi.org/10.1039/d6cc00474a.CCDC 2522306 (6b), 2522307 (13a′), 2522308 (14a), 2522309 (14g) and 2522310 (14ka) contain the supplementary crystallographic data for this paper.17a–e
References
- (a) A. P. Marchand, Aldrichimica Acta, 1995, 28, 95–104 CAS; (b) T. P. Stockdale and C. M. Williams, Chem. Soc. Rev., 2015, 44, 7737–7763 RSC; (c) G. M. Locke, S. S. R. Bernhard and M. O. Senge, Chem. – Eur. J., 2019, 25, 4590–4647 CrossRef CAS PubMed; (d) N. Grover and M. O. Senge, Synthesis, 2020, 3295–3325 CAS.
- (a) J. P. Agrawal, Progr. Energy Combust. Sci., 1998, 24, 1–30 CrossRef CAS; (b) P. E. Eaton, R. L. Gilardi and M.-X. Zhang, Adv. Mater., 2000, 12, 1143–1148 CrossRef CAS; (c) M.-X. Zhang, P. E. Eaton and R. Gilardi, Angew. Chem., Int. Ed., 2000, 39, 401–404 CrossRef CAS; (d) S. Rajkumar, R. S. Choudhari, A. Chowdhury and I. N. N. Namboothiri, Thermochim. Acta, 2013, 563, 38–45 CrossRef CAS; (e) S. Lal, S. Rajkumar, A. Tare, S. Reshmi, A. Chowdhury and I. N. N. Namboothiri, Chem. – Asian J., 2014, 9, 3533–3541 CrossRef CAS; (f) S. Lal, L. Mallick, S. Rajkumar, O. P. Oommen, S. Reshmi, N. Kumbhakarna, A. Chowdhury and I. N. N. Namboothiri, J. Mater. Chem. A, 2015, 3, 22118–22128 RSC; (g) S. Lal, A. Chowdhury, N. Kumbhakarna, S. Nandagopal, A. Kumar and I. N. N. Namboothiri, Org. Chem. Front., 2021, 8, 531–548 RSC.
- For diasterane, see: A. Otterbach and H. Musso, Angew. Chem., Int. Ed. Engl., 1987, 26, 554–555 CrossRef.
- For studies on or involving triasteranes, see: (a) U. Biethan, U. v Gizycki and H. Musso, Tetrahedron Lett., 1965, 6, 1477–1482 CrossRef; (b) H. Musso and U. Biethan, Chem. Ber., 1967, 100, 119–131 CrossRef CAS; (c) U. Biethan, H. Klusacek and H. Musso, Angew. Chem., Int. Ed. Engl., 1967, 6, 176–177 CrossRef CAS; (d) E. Vedejs, Tetrahedron Lett., 1969, 10, 5045–5048 CrossRef; (e) H. Musso and H. Klusacek, Chem. Ber., 1970, 103, 3076–3086 CrossRef CAS; (f) E. Vedejs, R. A. Gabel and P. D. Weeks, J. Am. Chem. Soc., 1972, 94, 5842–5845 CrossRef CAS; (g) P. A. Knott and J. M. Mellor, J. Chem. Soc., Perkin Trans. 1, 1972, 1030–1033 RSC; (h) I. A. McDonald and A. S. Dreiding, Helv. Chim. Acta, 1973, 56, 2523–2534 CrossRef CAS; (i) C. B. Chapleo, I. A. McDonald and A. S. Dreiding, Helv. Chim. Acta, 1975, 58, 1813–1817 CrossRef CAS; (j) E. Herranz and F. Serratosa, Tetrahedron Lett., 1975, 16, 3335–3336 CrossRef; (k) E. Herranz and F. Serratosa, Tetrahedron, 1977, 33, 995–998 CrossRef CAS; (l) H. Irngartinger, J. Hauck, A. De Meijere, K. Michelsen and R. Machinek, Isr. J. Chem., 1989, 29, 147–151 CrossRef CAS; (m) S. M. F. Farnia, G. A. Olah and G. K. S. Prakash, J. Sci., Islamic Repub. Iran, 1993, 4, 268–272 CAS; (n) W. W. Win, K. G. Grohmann and L. Todaro, J. Org. Chem., 1994, 59, 2803–2808 CrossRef CAS; (o) T. Tran Ngoc, N. Grabicki, E. Irran, O. Dumele and J. F. Teichert, Nat. Chem., 2023, 15, 377–385 CrossRef CAS PubMed; (p) T. Tran Ngoc, J. van der Welle, T. Rüffer and J. F. Teichert, Synthesis, 2023, 2658–2669 CAS; (q) A. Sanchez, A. Gurajapu, W. Guo, W.-Y. Kong, C. J. Laconsay, N. S. Settineri, D. J. Tantillo and T. J. Maimone, J. Am. Chem. Soc., 2023, 145, 13452–13461 CrossRef CAS.
- For studies on tetraasteranes, see: (a) H.-M. Hutmacher, H.-G. Fritz and H. Musso, Angew. Chem., Int. Ed. Engl., 1975, 14, 180–181 CrossRef; (b) G. Kaiser and H. Musso, Chem. Ber., 1985, 118, 2266–2281 CrossRef CAS; (c) V. T. Hoffmann and H. Musso, Angew. Chem., Int. Ed. Engl., 1987, 26, 1006–1007 CrossRef; (d) V. T. Hoffmann and H. Musso, Chem. Ber., 1991, 124, 103–109 CrossRef CAS.
- For studies on pentaasteranes, see: (a) K. Ebel, H. Kruger and H. Musso, Chem. Ber., 1988, 121, 323–326 CrossRef CAS; (b) A. Bader, K. Ebel, H. Musso and N. Skuballa, Chem. Ber., 1988, 121, 327–338 CrossRef CAS.
- (a) A. Ghosh and J. Conradie, ACS Omega, 2023, 8, 4972–4975 CrossRef CAS PubMed; (b) X. Chen and Y. Bu, J. Phys. Chem. A, 2023, 127, 9672–9683 CrossRef CAS.
- M. Sugiyama, M. Akiyama, Y. Yonezawa, K. Komaguchi, M. Higashi, K. Nozaki and T. Okazoe, Science, 2022, 377, 756–759 CrossRef CAS PubMed.
- A. N. Bismillah, B. M. Chapin, B. A. Hussein and P. R. McGonigal, Chem. Sci., 2020, 11, 324–332 Search PubMed.
- (a) S. Ferrer and A. M. Echavarren, Angew. Chem., Int. Ed., 2016, 55, 11178–11182 CrossRef CAS PubMed See also: ; (b) P. R. McGonigal, C. de León, Y. Wang, A. Homs, C. R. Solorio-Alvarado and A. M. Echavarren, Angew. Chem., Int. Ed., 2012, 51, 13093–13096.s Search PubMed.
- For examples of intermolecular bromoesterification of alkenes, see: (a) G.-X. Li, Q.-Q. Fu, X.-M. Zhang, J. Jiang and Z. Tang, Tetrahedron: Asymmetry, 2012, 23, 245–251 Search PubMed; (b) W. Zhang, N. Liu, C. M. Schienebeck, X. Zhou, I. I. Izhar, I. A. Guzei and W. Tang, Chem. Sci., 2013, 4, 2652–2656 RSC; (c) L. Li, C. Su, X. Liu, H. Tian and Y. Shi, Org. Lett., 2014, 16, 3728–3731 CrossRef CAS PubMed; (d) C. Wan, R.-J. Song and J.-H. Li, Org. Lett., 2019, 21, 2800–2803 Search PubMed.
- The structures of products 6b, 13a′, 14a, 14g, and 14ka were confirmed by X-ray crystallography and the structures of the remaining products were assigned by analogy. See the SI for details.
- R. L. Benoit, D. Lefebvre and M. Fréchette, Can. J. Chem., 1987, 65, 996–1001 CrossRef CAS.
- W. N. Olmstead and F. G. Bordwell, J. Org. Chem., 1980, 45, 3299–3305 CrossRef CAS.
- Denmark and Burk have described the use of Lewis base catalysts for related bromolactonization reactions. For a seminal paper, see: S. E. Denmark and M. T. Burk, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 20655–20660 CrossRef CAS PubMed . However, the attempted reaction of barbaralone 5a with NBS (1.1 equiv.) and benzoic acid (2.0 equiv.) in the presence of (Me2N)2C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S (1.0 equiv.) in CH2Cl2 led to no conversion and starting materials were recovered unchanged.
S (1.0 equiv.) in CH2Cl2 led to no conversion and starting materials were recovered unchanged. - F. Allen, Acta Crystallogr. B, 1980, 36, 81–96 CrossRef CAS.
- (a) CCDC 2522306: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qnnqs; (b) CCDC 2522307: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qnnrt; (c) CCDC 2522308: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qnnsv; (d) CCDC 2522309: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qnntw; (e) CCDC 2522310: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qnnvx.
| This journal is © The Royal Society of Chemistry 2026 |