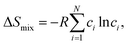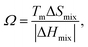Open Access Article
Open Access ArticleRise of high-entropy MXenes in electrocatalysis for sustainable energy conversion
Muhammad
Yousuf
 ,
Xiaopeng
Liu
,
Connor W.
Schroeder
,
Jonathan
Tjioe
,
Yurui
Liu
and
Yue
Wu
,
Xiaopeng
Liu
,
Connor W.
Schroeder
,
Jonathan
Tjioe
,
Yurui
Liu
and
Yue
Wu
 *
*
Department of Chemical and Biological Engineering, Iowa State University, 618 Bissell Road, Ames, IA 50011, USA. E-mail: yuewu@iastate.edu
First published on 29th April 2026
Abstract
High-entropy (HE) MXenes constitute an emerging class of two-dimensional carbides and carbonitrides that combine the tunable surface chemistry and metallic conductivity of MXenes with the configurational entropy, lattice distortion and multi-metal interactions of high-entropy materials (HEMs). This review presents the fundamental structural and thermodynamic features of MXenes, HEMs and HE-MXenes, establishing how multi-component M-sublattices and mixed terminations create ensembles of adsorption sites relevant to electrocatalysis. Progress in the synthesis and structural design of HE-MXenes is summarised, covering design of HE-MAX precursors, etching and delamination strategies, and advanced structural and chemical characterisation that enable control over composition, defects and surface terminations. The current understanding of electronic structure, active-site motifs and defect/termination chemistry in HE-MXenes is discussed, with emphasis on how lattice distortion and local coordination govern adsorption energetics. On this basis, recent advances in HE-MXenes and HE-MXene-based heterostructures for hydrogen evolution and oxidation, oxygen evolution and reduction, and other electrochemical conversion reactions, particularly CO2 and N2 reduction, are critically assessed against conventional MXenes, high-entropy catalysts and noble-metal systems. Furthermore, key challenges and opportunities are identified, including synthetic control of multi-metal distributions, coupled termination–defect engineering, operando interrogation of active phases and predictive modelling of high-dimensional composition space, with a view to guiding the rational deployment of HE-MXenes in sustainable electrocatalytic energy-conversion technologies.
1. Introduction
Electrochemical energy conversion technologies, including water electrolysis, fuel cells, metal–air batteries, and the electroreduction of CO2, are central to the transition towards low-carbon energy systems.1–5 Their practical deployment, however, is still constrained by sluggish multi-electron reaction kinetics and by the cost, scarcity, and sometimes limited durability of conventional noble-metal catalysts.6–9 Considerable effort has therefore been devoted to the discovery of earth-abundant electrocatalysts that combine high intrinsic activity with long-term stability and compatibility with device-level operating conditions. Two concepts have recently converged in this context. The first is the emergence of two-dimensional MXenes as a versatile family of electrically conductive carbides and nitrides with tunable surface chemistry. The second is the high-entropy materials (HEMs) paradigm, in which several principal elements are mixed in near-equimolar ratios to exploit configurational entropy, lattice distortion, and “cocktail” effects to stabilise single-phase solids with emergent properties.10–12 Their intersection, high-entropy (HE) MXenes, is now opening an unusually rich design space for electrocatalysts.Since the initial isolation of Ti3C2Tx in 2011 by selective etching of Ti3AlC2 MAX phases, the MXene family has expanded rapidly,13 as summarised in Fig. 1. Successive advances in MAX-phase chemistry, etching and delamination protocols, and post-synthetic modification have produced dozens of Mn+1XnTx compositions with diverse transition-metal sublattices, X = C and/or N, and mixed surface terminations (Tx = –O, –OH, –F, –Cl, –S, etc.). MXenes combine metal-like electronic conductivity with a layered architecture in which interlayer spacing, defect density, and heterostructure interfaces can be engineered almost independently. This has enabled applications ranging from supercapacitors and batteries to electromagnetic shielding, photothermal therapy, and heterogeneous catalysis.14–17 In parallel, high-entropy alloys, oxides, carbides, and chalcogenides have demonstrated that multi-component lattices can stabilise disordered yet single-phase structures, suppress phase segregation, and generate broad distributions of adsorption sites that are favourable for reactions such as the hydrogen evolution reaction (HER), oxygen evolution and reduction (OER/ORR), CO2 reduction (CO2RR), and nitrogen reduction (NRR).18–20
 | ||
| Fig. 1 Timeline of MXene synthesis and structural evolution (2011–2025), highlighting milestones in etching/delamination, assembly into architectures, and the emergence of HE-MAX phases and HE-MXenes.13–15,22,29,39,128–135 Reproduced with permission from ref. 13, copyright 2011, John Wiley and Sons. Reproduced with permission from ref. 14, copyright 2023, Elsevier. Reproduced with permission from ref. 15, copyright 2024, American Chemical Society. Reproduced with permission from ref. 22, copyright 2021, American Chemical Society. Reproduced with permission from ref. 29, copyright 2021, John Wiley & Sons. Reproduced with permission from ref. 39, copyright 2022, American Chemical Society. Reproduced with permission from ref. 128, copyright 2025, John Wiley & Sons. Reproduced with permission from ref. 129, copyright 2015, Royal Society of Chemistry. Reproduced with permission from ref. 130, copyright 2015, Springer Nature. Reproduced with permission from ref. 131, copyright 2014, Springer Nature. Reproduced with permission from ref. 132, copyright 2019, John Wiley & Sons. Reproduced with permission from ref. 133, copyright 2017, Springer Nature. Reproduced with permission from ref. 134, copyright 2020, Springer Nature. Reproduced with permission from ref. 135, copyright 2019, Springer Nature. | ||
Extending the high-entropy concept to MXenes creates HE-MXenes, in which four or more transition metals occupy the M-sublattice of the parent MAX phase in near-equimolar ratios. Beyond single-metal MXenes, the design space has expanded through M-site mixing (random solid solutions and ordered double-M phases) and ordered vacancy motifs, which demonstrated that local coordination and surface chemistry can tune adsorption energetics while preserving the layered scaffold (Fig. 2).21 HE-MXenes occupy the top tier of this hierarchy: they retain the Mn+1Xn scaffold but distribute four or more transition metals across the M-sublattice, often in near-equimolar ratios, such that adsorption sites are defined by statistically varying metal neighbourhoods and their associated electronic structures. In later sections, this diversity is treated explicitly in terms of distributions of adsorption free energies and their impact on activity and selectivity.7 Early examples such as TiVNbMoC3Tx, TiVCrMoC3Tx and related multi-metal M4C3 and M2C systems showed that configurational entropy can stabilise layered carbides against phase separation, and that the resulting MXenes preserve high electronic conductivity while exhibiting enhanced structural robustness and rich local electronic heterogeneity.22–24 In HE-MXenes, lattice distortion, multi-metal bonding and heterogeneous surface terminations collectively generate a continuum of local environments and adsorption energies rather than a single “average” active site. This multiplicity of sites can increase the probability of finding near-optimal binding energies for different reaction intermediates on the same surface, while entropy-stabilised structures may improve tolerance to oxidation, dissolution and mechanical stress under operation.11,12,21–24 To date, much of the HE-MXene literature has focused on electrochemical energy storage, especially Li-, Na- and K-ion batteries, supercapacitors and lithium–sulfur systems, where HE-MXenes act as high-surface-area hosts, conductive scaffolds or polysulfide mediators.25–29 In contrast, systematic studies of HE-MXenes as electrocatalysts for key energy-conversion reactions are only beginning to emerge, even though the broader high-entropy electrocatalyst community has already shown that multicomponent oxides, sulfides and alloys can achieve outstanding HER, OER, ORR, CO2RR and NRR performance by using entropy-driven stabilisation of active phases, suppression of competing pathways and deliberate tuning of adsorption-energy distributions.18–20,30,31
 | ||
| Fig. 2 Structural hierarchy of MXenes from single-metal compositions to HE-MXenes, highlighting the Mn+1Xn backbone, typical terminations (Tx) and representative adsorption motifs. Reprinted with permission from ref. 21, copyright 2018, Springer Nature. | ||
Building on broader progress in high-entropy electrocatalysts, HE-MXenes can be viewed as an emerging platform for sustainable electrochemical energy conversion. Fig. 3A connects these materials concepts to a broader sustainability landscape in which electrolysers, fuel cells and other HE-MXene–based devices operate within low-carbon infrastructure. Fig. 3B extends this perspective to an integrated renewable-hydrogen energy cycle, in which HE-MXene electrocatalysts facilitate the conversion of intermittent solar, wind and hydro resources into chemical fuels and back to electricity via water splitting and fuel cells. A bibliometric survey of the literature reveals that publications explicitly focused on HE-MXenes have risen sharply since 2021 (Fig. 3C), underscoring both the youth of the field and the rapidly growing interest in exploiting high-entropy design within the MXene family for energy-relevant electrocatalysis.
The aim of this review is therefore to provide a coherent picture of how HE-MXenes can be designed, understood and implemented as electrocatalysts for sustainable energy conversion, rather than as a simple extension of either MXenes or HEMs alone. Entire chain from synthesis of HE-MAX precursors and etching/delamination into HE-MXenes, through control of vacancies and terminations, to structural design, mechanistic understanding and device-level integration for HER/HOR, OER/ORR, CO2RR, NRR, and related reactions is explicitly considered (Fig. 3A). Section 2 revisits the fundamentals of HEMs and HE-MXenes, setting the thermodynamic and structural background. Section 3 discusses synthesis and structural design of HE-MXenes, emphasising routes that enable control over composition, defects and surface terminations. Section 4 examines electronic structure, active sites and theoretical modelling, with a focus on how multi-metal and termination-induced heterogeneity can be captured in descriptor-based frameworks. Sections 5 and 6 survey the current state of HE-MXenes in hydrogen- and oxygen-related electrocatalysis, while Section 7 extends the discussion to other electrochemical conversion reactions such as CO2RR and NRR. Finally, Section 8 summarises the main challenges and opportunities for HE-MXenes to evolve from a promising materials concept into a robust platform for electrocatalysis in future sustainable energy technologies.
2. Fundamentals of MXenes, high-entropy materials and high-entropy MXenes
2.1 High-entropy materials and their electrocatalytic features
HEMs are multicomponent solid solutions in which four or more elements share a crystallographically equivalent sublattice in (approximately) equimolar ratios. Their design is usually framed in terms of the Gibbs free energy of mixing,| ΔGmix = ΔGmix − TΔSmix, |
where ΔGmix, is the Gibbs free energy of mixing, ΔHmix is the enthalpy of mixing, ΔSmix is the entropy of mixing, T is the absolute temperature (K), R is the gas constant, ci is the atomic (molar) fraction of component i, i indexes the components, N is the total number of components, and ln denotes the natural logarithm. The “entropy parameter.”
uses the average melting point Tm of the elements (K) to gauge whether entropy can compensate the enthalpy of mixing; |ΔHmix| denotes the absolute value of the mixing enthalpy. Empirical analysis of high-entropy alloys (HEAs) shows that disordered solid solutions are favoured when the mixing enthalpy lies roughly in −15 < ΔHmix < 5 kJ mol−1, the atomic size mismatch δ (typically reported as a percentage measure of the dispersion in atomic radii among constituents) is below ∼5%, and Ω > 1; systems with ΔSmix > 1.5R are commonly classified as “high-entropy”. These criteria, originally formulated for bulk HEAs, have since been extended to ceramics and low-dimensional systems and provide a quantitative starting point for designing high-entropy carbides, nitrides, oxides and, by extension, HE-MXenes.
Yeh and co-workers rationalised HEM behaviour in terms of four “canonical” high-entropy effects: a thermodynamic high-entropy effect, lattice distortion, sluggish diffusion and the “cocktail” effect.23 In brief, the thermodynamic effect stabilises single-phase solid solutions that would otherwise decompose; lattice distortion introduces local strain and modifies bond lengths/angles; sluggish diffusion reflects the complex energy landscape for migrating atoms in a disordered lattice; and the cocktail effect refers to emergent properties arising from multi-element synergy rather than from any individual component. For electrocatalysis, these effects are not merely conceptual: they control how many distinct local environments exist at the surface, how stable those environments are under potential cycling, and how activity and durability can be combined in a single phase. Their practical impact has been most clearly demonstrated so far in metallic HEAs in Fig. 4. Oxidative corrosion of MXenes typically initiated at edges, vacancies, and termination defects, where O/OH species adsorbed and subsequently enabled oxygen ingress together with metal migration/leaching that nucleated oxide/(oxy)hydroxide domains. In HE-MXenes, configurational-entropy stabilisation, lattice distortion, and sluggish diffusion could collectively retard this process by (i) reducing the driving force for element segregation into distinct oxide phases and (ii) increasing migration barriers for both metal and oxygen species in the disordered lattice. As a result, oxide growth and preferential leaching of individual metals could be kinetically suppressed, and compositionally complex surface (oxy)carbide/(oxy)hydroxide layers may form that further impede oxidant transport. Although systematic atomic-scale verification for HE-MXenes remained limited, this framework provided a mechanistic basis for the corrosion/oxidation tolerance often associated with high-entropy compositions.
 | ||
| Fig. 4 HEAs as a model platform for data-driven electrocatalyst discovery and CO2 reduction. (A) Concept of complex solid-solution HEAs with millions of distinct surface sites, highlighting the need to integrate theory, synthesis and characterisation in a high-throughput iterative loop. (B) Schematic of the computational–experimental discovery cycle and comparison between predicted and measured ORR activity maps on a composition-spread thin-film library. Reprinted with permission from ref. 32, copyright 2020, John Wiley & Sons. (C) Schematic representation of CO2 reduction to CO on a multimetal HEA surface via *CO2, *COOH and *CO intermediates. (D) Potential-dependent CO yield rates for AuAgCuPdPt HEAs compared with AuAg and Ag catalysts. (E) Wet-chemical synthesis route to AuAgCuPdPt HEA nanoparticles via rapid nucleation followed by freeze–thaw growth. Reprinted with permission from ref. 33, copyright 2025, Springer Nature. | ||
Among the different high-entropy electrocatalyst families, metallic HEAs provide the clearest precedent for how complex solid solutions can be rationally exploited in energy conversion. Batchelor et al. combined density functional theory (DFT) based adsorption–energy models with composition-spread Ag–Ir–Pd–Pt–Ru thin-film libraries and scanning droplet-cell measurements to build a closed, data-driven discovery loop, in which predictions, combinatorial synthesis and high-throughput characterisation iteratively refine one another and identify activity maxima for the ORR in a vast composition space (Fig. 4A and B).32 This work shows that the statistical distribution of adsorption sites on high-entropy surfaces can be treated explicitly and optimised, rather than sampled empirically, and it provides a methodological template that is directly transferable to high-entropy design on MXene basal planes. A complementary example is provided by Pt-optimised AuAgCuPdPt HEAs for CO2-to-CO conversion (Fig. 4C–E): Li and co-workers achieved faradaic efficiencies (FE) for CO above 90% over a wide potential window and significantly higher CO yield rates than monometallic Ag or bimetallic AuAg, while maintaining a single fcc phase and excellent stability in both flow-cell CO2RR and Zn–CO2 battery operation.33In situ Raman and ATR-FTIR spectroscopy revealed that the multimetal ensemble stabilises *CO2− and *COOH intermediates, weakens *CO binding and promotes water dissociation, thereby suppressing HER and enabling selective CO formation, an archetypal demonstration of how lattice distortion and cocktail effects in high-entropy systems translate into practical gains in electrocatalytic activity and selectivity.
Taken together, these studies yield several general lessons that are particularly relevant to the emerging role of HE-MXenes in electrocatalysis. First, successful high-entropy design requires balancing configurational entropy, mixing enthalpy and size mismatch so that genuine single-phase solid solutions are obtained rather than multiphase mixtures, exactly the challenge faced when selecting element sets for HE-MAX and HE-MXene systems. Second, cation and anion disorder, including vacancies and mixed-valence states, are central to both activity and stability; in perovskite and spinel HEMs, oxygen defects and multi-metal synergy correlate with enhanced NRR and OER performance, and an analogous role can be anticipated for vacancy and termination disorder in HE-MXenes. Third, the complexity of HEMs demands rigorous structural and electronic characterisation (XRD/Rietveld, electron microscopy, XPS, XANES/EXAFS) to confirm true single-phase behaviour and to resolve local coordination environments. Finally, high-entropy electrocatalysis has shown that composition–structure–function relationships must be established mechanistically rather than by empirical screening alone. This perspective directly underpins later sections, where high-entropy design principles are transplanted to the two-dimensional MXene platform for sustainable energy-conversion reactions.
2.2 High-entropy MXenes as 2D high-entropy materials
HE-MXenes belong to the emerging class of 2D-HEMs, which also includes oxides, chalcogenides, hydroxides and other carbides.34,35 These systems represent the latest step in the broader development of entropy-stabilised multi-principal-element materials, from 3D HEAs and ceramics to layered structures in which configurational entropy is implemented within an atomically thin framework (Fig. 5).24,36 In this context, HE-MXenes can be viewed as 2D carbide/carbonitride analogues in which the M-sublattice of the Mn+1Xn slab hosts a near-equimolar mixture of early transition metals (e.g. Ti, V, Nb, Mo, Cr, Zr, Ta), while the X (C/N) sublattice and surface terminations can also display compositional complexity.29 Compared with binary or ternary MXenes, this multicomponent character introduces additional configurational degrees of freedom that can be used to adjust local bonding, electronic structure and, ultimately, electrocatalytic response.37 | ||
| Fig. 5 Evolution of entropy-stabilised multi-principal-element materials from 3D HEAs/ceramics to 2D HEMs, providing the conceptual basis for HE-MXenes. (A–D) Representative structural models of multi-principal-element materials with increasing dimensional and compositional complexity. (E) Schematic thermodynamic basis of entropy stabilisation. (F) Representative disorder motifs, including interface formation, substitutional alloying, short-range ordering, and entropy-stabilised solid solutions. Reproduced with permission from ref. 36, copyright 2021, AAAS. | ||
From a thermodynamic perspective, HE-MXenes follow the same principle as bulk high-entropy ceramics: near-equimolar mixing of several cations on a crystallographically equivalent sublattice increases the molar configurational entropy and can stabilise a single-phase solid solution against decomposition.37 In the 2D limit, this entropy originates primarily from mixing on the metallic M-sublattice, but it may also arise from disorder on the non-metal sublattice (C/N) and from statistical distributions of surface terminations (O, OH, F, Cl, S, etc.).38 HE-MXenes therefore combine several levels of disorder: chemical disorder within each M layer, possible carbonitride disorder on the X sublattice, and termination disorder on both faces of the sheet, producing a spectrum of local coordination environments, bond lengths, and electronic states.29,35 This hierarchical disorder gives rise to lattice distortions, electronic inhomogeneity and modified orbital hybridisation, features that are central to the emerging electrocatalytic behaviour of HE-MXenes. These effects have been directly observed in representative HE-MXenes. Du et al. synthesised high-entropy M2CTx MXenes by etching a (Ti1/5V1/5Zr1/5Nb1/5Ta1/5)2AlC HE-MAX phase and showed that the transition metals are homogeneously dispersed within the MX slab at the atomic scale, consistent with a solid-solution character.29 The size mismatch among the five cations induced significant lattice distortions and internal strain, which strongly influenced Li nucleation and growth and enabled dendrite-free plating/stripping at the HE-MXene interface.29 Similar local distortions and expanded in-plane lattice parameters have been reported for other HE-MXenes when larger cations such as Nb, Zr and Ta are introduced into Ti-based frameworks, leading to measurable changes in the (100)/(010) spacings and selected-area electron diffraction patterns.38
Building on this, Nemani and co-workers realised high-entropy 413 MXenes TiVNbMoC3Tx and TiVCrMoC3Tx by etching the corresponding HE-MAX precursors. High-resolution XRD confirmed that the M4AlC3 MAX phases were single-phase with lattice parameters comparable to non-high-entropy analogues, while atomic-resolution STEM/EDS mapping revealed uniform distributions of Ti, V, Nb and Mo (or Cr) across the MXene flakes without detectable clustering.22 XPS depth profiling showed near-equimolar metal ratios through the sheet thickness and a complex mixture of metal–C/O/F bonds and oxidation states, reflecting the multi-metal surface chemistry. Du et al. first extended this concept to carbonitride MAX phases by synthesising a high-entropy carbonitride MAX, (Ti1/3V1/6Zr1/6Nb1/6Ta1/6)2AlCxN1−x, and demonstrating that the five metals form a solid solution on the M sublattice; etching then yielded high-entropy carbonitride MXenes with homogeneous multi-metal distributions.29 Subsequently, Zhou et al. showed that careful control of the M/Al/C ratios and sintering conditions yields single-phase high-entropy laminate MAX precursors and their two-dimensional derivatives without competing carbides or intermetallics.39 Etman and co-workers further reported Ti1.1V0.7CrxNb1.0Ta0.6C3Tz freestanding films that combine multi-metal mixing with good mechanical integrity and promising charge-storage performance.40 Together, these studies establish that HE-MXenes can be synthesised reproducibly in both M2X- and M4X3-type structures across a range of carbide and carbonitride chemistries.
Electronic–structure calculations complement these experimental observations. High-throughput DFT and Monte Carlo simulations for TiVNbMoC3 and TiVCrMoC3 indicate that, although the overall compositions are equimolar, energetically favoured configurations can exhibit partial interlayer chemical ordering, with certain metals enriched at the outer layers and others stabilised in the inner ones.41 This tendency becomes more pronounced at lower temperatures, but entropy-stabilised disordered configurations remain accessible at realistic synthesis temperatures, in agreement with experimental evidence for multi-metal occupation and selective etching behaviour.38 From an electrocatalytic viewpoint, HE-MXenes thus provide a dense ensemble of inequivalent adsorption sites, each with slightly different local coordination and electronic structure. DFT studies on oxygen-terminated TiVNbMoC3 have shown that the hydrogen adsorption free energy can be tuned within a narrow window around thermoneutrality by sampling different local metal environments on the same basal plane,42 while spectroscopic analyses of high-entropy carbides and carbonitrides reveal a rich variety of M–C–Tx and M–N–Tx bonds with subtly shifted binding energies, indicating variations in metal–ligand covalency and charge density.38,43 These ensemble and surface-chemistry effects, combined with the high in-plane conductivity of MXenes, underlie the enhanced and tunable electrocatalytic behaviour observed in HE-MXenes.38,43
Ultimately, HE-MXenes are best regarded as a subclass of functional two-dimensional HEMs in which configurational entropy is embedded within a layered carbide/carbonitride scaffold endowed with accessible, chemically active surfaces. Their development has established the synthetic feasibility, structural stability and homogeneous multi-metal distributions required for rational property optimisation, while theory and experiment agree that high-entropy mixing generates local strain, electronic heterogeneity and a spectrum of adsorption environments that can be exploited to engineer electrocatalytic performance beyond that of simpler MXene chemistries.18,44Table 1 compiles the experimentally realised HE-MXenes reported so far and highlights how this material family has already moved beyond purely structural demonstrations into functional electrocatalysis and energy storage. TiVNbMoC3 and TiVCrMoC3 derived from quinary HE-MAX phases deliver high and stable capacities as Li- and Na-ion battery anodes,27 while Ti1.1V1.2Cr0.8Nb1.0Mo0.9C4Tx and Ti1.1V0.7CrxNb1.0Ta0.6C3Tz exhibit competitive capacitances and cycling stability in alkaline and acidic supercapacitors.14,39,40 In parallel, (Ti1/5V1/5Zr1/5Nb1/5Ta1/5)2CTx promotes uniform Li plating with millivolt-scale nucleation overpotentials,29 and TiVNbMoC3-based architectures act as efficient sulfur hosts and separator mediators in Li–S batteries, enabling long-term cycling at practical loadings.15,45 More recently, HE-MXenes have begun to serve as conductive, corrosion-resistant supports for multicomponent electrocatalysts, for example, in V–Co2P@HE heterostructures for overall water splitting and in CuFeMnCoNi-NC/MXene anodes for anion-exchange membrane water electrolysers.31,46 Together with HE-MXenes designed for electromagnetic-wave absorption and other functionalities,43,47 these examples demonstrate that the HE-MXene concept is already technologically versatile, while also revealing that truly electrocatalysis-focused compositions remain relatively scarce.
| HE-MAX | HE-MXene (support/host) | Purpose/role | MAX synthesis temp. (°C) | Reaction/device | Key performance (selected) | Ref. |
|---|---|---|---|---|---|---|
| Ti1.1V1.2Cr0.8Nb1.0Mo0.9AlC4 | Ti1.1V1.2Cr0.8Nb1.0Mo0.9C4Tx (M5C4) | High-capacitance HE-MXene for alkaline supercapacitors | 1500 | Supercapacitor (1 M KOH, three-electrode) | 284.6 F g−1 at 1 A g−1; 170 F g−1 at 10 A g−1; reduced Rs and Rctvs. Nb2CTx, V2CTx, Ti3C2Tx | 14 |
| (Ti0.2V0.2Nb0.2Mo0.2W0.2)3AlC2 | (Ti0.2V0.2Nb0.2Mo0.2W0.2)3C2Tx | High-stability HE-MXene electrode for supercapacitors | 1550 | Supercapacitor (1 M H2SO4, three-electrode) and MXene-HF 72 h//AC asymmetric supercapacitor | 158.43 F g−1 at 5 mV s−1; 128.98 F g−1 at 0.5 A g−1; electrode: 100% retention after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 10 A g−1; device: 50.18 F g−1 at 1 A g−1, 13.98 Wh kg−1/163.35 W kg−1, 98.7% retention after 10 000 cycles at 10 A g−1; device: 50.18 F g−1 at 1 A g−1, 13.98 Wh kg−1/163.35 W kg−1, 98.7% retention after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 5 A g−1 000 cycles at 5 A g−1 |
136 |
| TiVCrMoAlC3 | TiVCrMoC3Tx/PVA composite film | High-entropy piezoelectric MXene host in a flexible PVA matrix for self-powered nanogenerators | — | Flexible piezoelectric nanogenerator (PENG); self-powered motion sensor | 500 mV and 790 pA at 3.47 N; stable over 1500 cycles; maximum output power = 110.76 pW; finger-motion sensing: ∼85 pA and ∼10 mV | 137 |
| TiVCrMoAlC3 | TiVCrMoC3Tx | Active HE-MXene for PEC-type photodetectors and saturable absorbers/mode lockers | — | Photodetector (1.0 M KOH) and mode-locked fiber lasers | Response time ≈ 100 fs; β = −0.134 cm GW−1; modulation depth = 66%; photodetector at 0.4 V: Pph = 1650 nA cm−2, Rph = 35.55 µA W−1, up to 33× Ti3C2Tx; mode-locked lasers at 1072.5/1553.3/1909.4 nm with SNR 57.4/55.8/54.1 dB | 138 |
| (Ti0.21V0.21Mo0.17Nb0.22Ta0.19)4Al0.93C3 | (Ti0.21V0.21Mo0.17Nb0.22Ta0.19)4C3Tx | Conductive HE-MXene substrate for anchoring MoSe2/Ni3Se4 nanosheets | 1350 | HER in 0.5 M H2SO4 and 1.0 M KOH | Acid: 67 mV @ 10 mA cm−2, 68.7 mV dec−1, Cdl = 115.7 mF cm−2. Alkaline: 73 mV @ 10 mA cm−2, 77.8 mV dec−1, Cdl = 87.5 mF cm−2. Stable for 24 h in both media | 139 |
| TiVNbMoAlC3 | TiVNbMoC3Tx/Fe3O4 | Oxidation-resistant HE-MXene platform for balanced dielectric-magnetic EM-wave absorption | — | EM-wave absorber | HE-MXene/Fe3O4-3: RLmin = −57.59 dB at 1.46 mm; EAB = 4.72 GHz at 1.53 mm; RCS = 1.76 dB m2; after 30 days, EAB = 4.92 GHz; magnetic saturation ≈ 46 emu g−1 | 140 |
| Ti2V0.9Cr0.1AlC2 | Ti2V0.9Cr0.1C2Tx | High-capacitance multimetal MXene electrode for supercapacitors | 1500 | Supercapacitor (1 M KOH, three-electrode) and symmetric supercapacitor | 553.27 F g−1 at 2 mV s−1; 165.51 F g−1 at 200 mV s−1; Rct = 8.12 Ω; 90.21% retention after 2000 cycles; device: 20.99 Wh kg−1 at 800 W kg−1, 85.18% retention after 2000 cycles | 141 |
| TiVNbMoAlC3 | TiVNbMoC3 (HE-MXene/G@PP) | Separator mediator and polysulfide regulator | 1600 | Li–S battery (LiPS capture + Li plating/stripping) | Capacity 654.8/509.4 mAh g−1 after 1200 cycles at 1C/2C (0.026/0.031% fade per cycle); Li‖Li symmetric cell stable 6000 h at 40 mA cm−2/40 mAh cm−2 | 15 |
| (Ti1/5V1/5Zr1/5Nb1/5Ta1/5)2AlC | (Ti1/5V1/5Zr1/5Nb1/5Ta1/5)2CTx | Uniform Li deposition and dendrite-free Li metal anodes | 1500 | Li plating/stripping (symmetric + full cells) | Li nucleation overpotential ≈ 6 mV; symmetric cell 1200 h at 1 mA cm−2, 1 mAh cm−2; deep plating up to 20 mAh cm−2; full cell ≈100 mAh g−1 at 10C | 29 |
| Ti1.0V0.7Cr0.05Nb1.0Ta1.0AlC3 | Ti1.1V0.7CrxNb1.0Ta0.6C3Tz | Expand MXene composition space for high-rate supercapacitors | 1500 | Supercapacitor (1 M H2SO4) | 490 F g−1 and 1688 F cm−3 at 2 mV s−1; 89% capacitance retention after 9000 cycles with 99.4% coulombic efficiency at 13.3 A g−1 | 39 |
| (TiVCrNbMo)5AlC4 | (TiVCrNbMo)5C4Tx | 2D HE-MXene support to build V–Co2P heterostructure for bifunctional water splitting | — | HER + OER (overall water splitting) | H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O2 ≈ 2 O2 ≈ 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, overall FE ≈ 94.8% 1, overall FE ≈ 94.8% |
31 |
| Mo2Ti2AlC3 | CuFeMnCoNi-NC/MXene (Mo2Ti2C3Tx) | AEMWE anode catalyst to overcome sluggish OER | 900 | OER (AEM water electrolyzer, Pt/C cathode) |
H
10 = 267 mV; Tafel 50.9 mV dec−1; 1.91 A cm−2 at 2.0 V (70 °C); Δη ≈ 3 mV after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles 000 cycles |
46 |
| TiVNbMoAlC3 | TiVNbMoC3 (HE-MXene host + interlayer) | Sulfur host and separator-modification layer in Li–S cells | - | Li–S battery sulfur redox + Li plating/stripping regulation | 1102.5 mAh g−1 at 1C; ≈0.057% decay per cycle over 400 cycles; 4.92 mAh cm−2 at 0.2C (5.4 mg cm−2, E/S 8.3 mL mg−1) | 45 |
| Ti1.0V0.7Cr0.05Nb1.00Ta1.0AlC3 | Ti1.1V0.7CrxNb1.0Ta0.6C3Tz | Freestanding HE-MXene films for Zn-ion hybrid capacitors and Li-ion batteries | 1500 | ZHSC and LIB anodes | ZHSC: 77 mAh g−1 at 0.5 A g−1, 43 mAh g−1 at 10 A g−1; 87% retention after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. LIB: 126 mAh g−1 at 0.01 A g−1; 48 mAh g−1 at 2 A g−1; stable 1000 cycles at 1 A g−1 000 cycles. LIB: 126 mAh g−1 at 0.01 A g−1; 48 mAh g−1 at 2 A g−1; stable 1000 cycles at 1 A g−1 |
40 |
| (Mo0.25Cr0.25Ti0.25V0.25)3AlC2 | f-(Mo0.25Cr0.25Ti0.25V0.25)3C2Tx | HE-MXene for electromagnetic (EM) wave absorption | 1400–1600 | EM absorber (35 wt% composite) | RLmin = −45.0 dB at 1.52 mm; effective absorption bandwidth (EAB) 5.6 GHz at 1.65 mm | 43 |
| ((Mo0.2Cr0.2Nb0.2Ti0.2V0.2)4AlC3) | f-(Mo0.2Cr0.2Nb0.2Ti0.2V0.2)4C3Tx | RLmin = −52.8 dB at 1.58 mm; optimum EAB 3.6 GHz at 1.50 mm | ||||
| TiVNbMoAlC3 | TiVNbMoC3 | To produce conductive, composition-flexible 2D carbides for battery electrodes | 1400 | LIB anode | Initial 556/1078 mAh g−1 (discharge/charge), ICE 51.6%; 725 mAh g−1 after 1000 cycles at 1 A g−1 | 27 |
| TiVCrMoAlC3 | TiVCrMoC3 | SIB anode | 127 mAh g−1 after 100 cycles at 100 mA g−1 (ICE 44.9%) | |||
| TiVCrMoC3 | LIB anode | 127 mAh g−1 after 100 cycles at 100 mA g−1 | ||||
| (Ti1/3V1/3Nb1/3)2GaC | (Ti1/3V1/3Nb1/3)2CTx | Fluorine-free synthesis of medium/HE-MXenes for Li-ion storage | 1300 | LIB anode | ≈200 mAh g−1 after 300 cycles at 200 mA g−1 | 47 |
| (Ti1/4V1/4Nb1/4Ta1/4)2GaC | (Ti1/4V1/4Nb1/4Ta1/4)2CTx | Li+ capacity ≈400 mAh g−1; 289 mAh g−1 after 300 cycles at 200 mA g−1 | ||||
| (Ti1/5V1/5Nb1/5Ta1/5Mo1/5)2GaC | (Ti1/5V1/5Nb1/5Ta1/5Mo1/5)2CTx | 302 mAh g−1 after 300 cycles at 200 mA g−1; best high-rate stability among the three |
3. Synthesis and structural design of high-entropy MXenes
3.1 Design principles for high-entropy MAX precursors
HE-MAX phases are the indispensable precursors for HE-MXenes and must be obtained as single-phase layered carbides or carbonitrides (most commonly M4AlC3 and M3AlC2 types) in which three to five early transition metals occupy the M sublattice in near-equimolar ratios, while the A and X sites remain relatively simple so that configurational entropy is concentrated on the metal lattice.48 The key design requirement is that this multicomponent M sublattice forms a genuine solid solution instead of decomposing into mixtures of binary MAX phases, simple carbides or intermetallics. This demands compatibility in atomic radius, electronegativity and valence state, and an overall combination that lies within acceptable ranges of mixing enthalpy and atomic size mismatch while providing sufficient configurational entropy at the synthesis temperature.48 DFT calculations for TiVNbMoAlC3 and TiVCrMoAlC3 showed that the corresponding four-metal M4AlC3 solid solutions have negative formation enthalpies and lie close to the convex hull when competing carbides and intermetallics are considered, whereas analogous three-metal compositions tend to split into multiple MAX phases.22 Only when four equimolar metals are included does the −TΔS term become large enough to stabilise a single-phase HE-MAX, a prerequisite for obtaining phase-pure HE-MXenes.22,35Empirical criteria derived from bulk high-entropy carbides and oxides provide a practical starting point for such design.27,35 Robust single-phase solid solutions are favoured when the atomic size mismatch on the disordered sublattice is modest and the configurational entropy exceeds the usual high-entropy threshold; translated to MAX phases, this means selecting M elements with similar metallic radii and comparable electronegativities, each of which is known to form conventional MAX phases with Al and C/N, and avoiding components that strongly prefer very stable competing carbides or intermetallics.48 This strategy was explicitly followed in the design of (Ti,V,Cr,Nb,Ta)4AlC3 and TiVNbMoAlC3, where each individual metal forms an M4AlC3-type phase, and the average M/Al/C stoichiometry was tuned to suppress binary carbides.22,39,48 X-ray diffraction with Rietveld refinement and electron microscopy confirmed that these HE-MAX precursors could be prepared as nearly single-phase materials with only minor secondary phases.27,39
Beyond such heuristic selection, thermodynamic screening based on phase-diagram calculations and DFT/CALPHAD approaches is increasingly used to identify promising HE-MAX compositions.49 Systematic surveys of MAX stability map the formation enthalpy relative to competing phases (ΔHcp) and show that many experimentally realised MAX compounds lie within a narrow window of metastability.48,50 For multicomponent M sublattices, this analysis is extended by constructing supercell models with random or quasi-random metal distributions and comparing their energies with mixtures of binary carbides, nitrides and intermetallics; cluster-expansion or Monte Carlo methods then capture the impact of short-range order and configurational entropy.35,48 Such calculations have guided the synthesis of new HE-MAX phases by identifying element sets and stoichiometries for which the high-entropy solid solution is thermodynamically competitive at realistic synthesis temperatures, and by flagging combinations that are likely to decompose.22,35
Kinetic factors are equally important because even thermodynamically viable compositions require sufficient diffusion and mixing to form homogeneous solid solutions. Most reported HE-MAX phases have therefore been synthesised by high-temperature reactive sintering of elemental powders or binary carbides, followed by extended dwell times to promote solid-state diffusion. Nemani et al., for example, prepared TiVNbMoAlC3 and TiVCrMoAlC3 by mixing equimolar transition-metal powders with Al and C and sintering at 1600 °C for 4 h under flowing Ar, then converting the resulting HE-MAX phases into multilayer and delaminated HE-MXenes (Fig. 6A).22 Zhou and co-workers synthesised (Ti,V,Cr,Nb,Ta)4AlC3 by solid-state reaction at 1500 °C for 10 h and showed that deviations from the optimal M/Al/C ratio led to substantial fractions of binary carbides, highlighting the narrow kinetic and compositional window for forming single-phase HE-MAX (Fig. 6B).39 Mechanical alloying followed by spark plasma sintering (SPS) offers a complementary approach: high-energy ball milling pre-mixes the constituents at the nanoscale, and a subsequent SPS step with rapid heating and applied pressure accelerates densification and diffusion while limiting grain growth, enabling HE-MAX formation from multicomponent powder mixtures.51 Recently, SPS has been pushed into an “ultra-rapid” regime, where multicomponent M4AlC3-type HE-MAX phases such as TiVNbMoAlC3, TiVCrMoAlC3 and (Ti0.2V0.2Cr0.2Nb0.2Mo0.2)4AlC3 were obtained with >99% phase purity in only 15 min by using a slight Al overfeed to generate a transient liquid phase that enhances mass transport among the refractory metals (Fig. 6C).27 While SPS clearly improves kinetic control and densification, it still requires careful optimisation of heating profiles, die materials and atmosphere to avoid contamination, undesired reactions and non-uniform microstructures.
 | ||
| Fig. 6 (A) Reactive sintering route to high-entropy M4AlC3 MAX phases and their subsequent conversion into multilayer and delaminated TiVNbMoC3Tx and TiVCrMoC3Tx HE-MXenes. Reprinted with permission from ref. 22, copyright 2021, American Chemical Society. (B) Representative microstructural characterisation of a 3D HE-MAX phase and its transformation into a 2D HE-MXene, illustrating the importance of composition control for avoiding binary carbide impurities. Reprinted with permission from ref. 39, copyright 2022, American Chemical Society. (C) Comparison between conventional furnace synthesis and an ultra-rapid SPS route, in which transient liquid Al and fast heating enable high-purity HE-MAX formation within minutes. Reprinted with permission from ref. 27, copyright 2024, John Wiley & Sons. | ||
From a practical standpoint, therefore, conventional tube-furnace solid-state reaction, SPS, and (less frequently) hot pressing have emerged as the main platforms for HE-MAX synthesis.22,27,39 Across all methods, precise control of the M/Al/C (and, where relevant, N) ratios and of the inert atmosphere is critical: small Al excess can assist dissolution of otherwise stable carbides, whereas too much Al leads to persistent intermetallics; similarly, deviations in C content shift the equilibrium towards competing carbides or graphite.39,48 At the same time, the HE-MAX composition must be chosen with subsequent etching and delamination in mind. Early-transition-metal M4AlC3 and M3AlC2 phases are generally amenable to selective Al removal in fluoride-containing media, but adding elements that form very stable oxides or non-volatile fluorides can impede etching or cause preferential dissolution. Representative examples of how specific MAX chemistries transform into MXenes under different MAX precursors and etching conditions are summarised in Table 2, which highlights the diversity of precursor structures and etchants in conventional (non-high-entropy) systems.52–61 Systematic studies on TiVNbMoAlC3, TiVCrMoAlC3 and (Ti,V,Cr,Nb,Ta)4AlC3 demonstrated that, when the MAX precursor is truly single-phase and the M elements are reasonably etchable, the multicomponent metal distribution is largely retained in the resulting HE-MXenes, with only modest deviations for more reactive species such as Cr.22,35,39 Thus, robust design of HE-MAX precursors demands an integrated strategy that combines thermodynamic screening (DFT/CALPHAD),49 elemental selection based on radius and valence compatibility, and kinetic control of synthesis (choice of solid-state reaction, SPS or hot pressing, and precise stoichiometry and atmosphere) to deliver single-phase M4AlC3 and M3AlC2 structures suitable for conversion into HE-MXenes.
| MAX precursors | MXenes | Etchants | Etching temp. | Ref. |
|---|---|---|---|---|
| Cr2TiAlC2 | Cr2TiC2Tx | Molten NH4HF2 | 180 °C for 40 min | 142 |
| Ti3AlC2 | Ti3C2Tx | NH4F | 150 °C for 24 h | 53 |
| Ti3AlC2 | Ti3C2Tx | 1 M NH4HF2 | Room temperature | 54 |
| (Nb0.8Zr0.2)4AlC3 | (Nb0.8Zr0.2)4C3Tx | LiF + 12 M HCl | 50 °C for 168 h | 55 |
| Ti4AlN3 | Ti4N3Tx | 59% KF + 2 9% LiF + 12% NaF | 550 °C for 30 min | 143 |
| V2AlC | V2CTx | 2 g LiF + 40 M HCl | 90 °C for 72 h | 57 |
| Ti2AlC | Ti2CTx | 0.9 M LiF + 6 M HCl | 40 °C for 15 h | 58 |
| Ti3AlCN | Ti3CNTx | 0.66 g LiF + 6 M HCl | 30 °C for 12 h | 59 |
| Mo2Ga2C | Mo2CTx | 3 M LiF + 12 M HCl | 50 °C for 3 h | 144 |
| Hf3[Al(Si)]4C6 | Hf3C2Tx | 35% HF | Room temperature | 61 |
3.2 Etching and delamination of HE-MAX into HE-MXenes
The transformation of HE-MAX phases into their corresponding HE-MXenes follows the same basic principle as for conventional MAX phases: selective removal of the A-layer (typically Al) to generate a layered carbide or nitride that can subsequently be delaminated into few-layer or single-layer flakes. However, the presence of multiple transition metals in HE-MAX phases introduces important additional constraints on etching chemistry, because etching conditions now control not only A-layer removal but also elemental homogeneity within the M-sublattice, defect density in the basal planes and the distribution of surface terminations. Careful selection and optimization of etchants and delamination protocols is therefore central to obtaining phase-pure HE-MXenes with preserved equimolar metal ratios and well-defined surface chemistry for electrocatalysis.56,62,63 | ||
| Fig. 7 Conventional and in situ HF-based etching routes relevant to HE-MXenes. (A) Direct HF etching of a HE-MAX phase to yield layered HE-MXene flakes that are subsequently incorporated into a HE-MXene/G@PP separator architecture. Reproduced with permission from ref. 15, copyright 2024, American Chemical Society. (B) Structural evolution of Ti3AlC2 during etching with in situ HF-forming agents to produce Ti3C2Tx: removal of Al and installation of mixed terminations increase the interlayer distance, while the bottom panel sketches a typical workflow involving etching, washing/centrifugation and recovery of MXene powder. Reproduced with permission from ref. 64, copyright 2022, John Wiley & Sons. | ||
Given the safety and scalability limitations of concentrated HF, there is growing interest in replacing direct HF with in situ HF-forming systems such as NH4HF2, NaF/HCl, HBF4 and NaBF4/HCl. Gentile et al. systematically compared Ti3C2Tx produced by these agents with material obtained by conventional 5 wt% HF, showing that in situ routes can achieve complete A-layer removal, comparable crystallinity and electrochemical performance while introducing distinct interlayer spacings and intercalated cations (Na+, NH4+, Cl−) that modulate the MXene structure and charge-storage behaviour.64 The schematic in Fig. 7B, illustrates how in situ HF etching not only replaces Al with mixed terminations but also expands the (002) spacing and yields stacked MXene platelets suitable for further delamination. Translating these insights to HE-MAX systems implies that etchant choice must be optimised not only for complete and composition-preserving A-layer removal, but also for controlled introduction of terminations and interlayer species that favour the targeted electrocatalytic reaction while reducing the reliance on highly hazardous HF.64 These conventional and in situ HF approaches provide the baseline chemistry from which more sustainable, termination-engineered etching protocols for HE-MXenes will be developed in later sections.
 | ||
| Fig. 8 Emerging etching and exfoliation strategies relevant to HE-MXenes. (A) Molten-salt synthesis of Pt–M@Ti3C2Tx. (B) SEM and EDS maps of Pt–Co@Ti3C2Tx. Reprinted with permission from ref. 66, copyright 2023, John Wiley & Sons. (C) Halogen-ion-mediated hydrothermal synthesis of Mo2CTx. (D) SEM images showing morphology retention on scale-up. Reprinted with permission from ref. 67, copyright 2025, John Wiley & Sons. (E) scCO2-assisted exfoliation scheme. (F) SEM images of pristine and exfoliated samples. Reprinted with permission from ref. 68, copyright 2024, John Wiley & Sons. | ||
Halide-mediated hydrothermal etching represents a complementary fluoride-free strategy. In this case, concentrated halide solutions (e.g. Cl−, Br−, I−) at elevated temperature promote A-layer dissolution and introduce halogen-rich terminations without using HF.7 Xu et al. recently demonstrated a halogen-ion-mediated hydrothermal route that enables gram- to tens-of-gram-scale synthesis of diverse MXenes, including Mo2CTx, with controllable morphology and termination chemistry (Fig. 8C and D).67 Compared with classical alkali-hydrothermal and electrochemical protocols,7 this approach combines safer processing with explicit control over the halide content of the surface, features that are particularly attractive for HE-MXenes where subtle changes in terminations can strongly modulate multi-metal active sites.
Supercritical CO2 (scCO2)-assisted etching and exfoliation is an emerging technique designed to overcome diffusion limitations and throughput constraints of purely liquid-phase methods. Kim et al. showed that scCO2, combined with suitable etchants, can intercalate and delaminate Ti3AlC2, producing few-layer MXene nanosheets without harsh acids or bases and with improved control over layer spacing (Fig. 8E and F).68 In this process, scCO2 enhances transport of etchant species into the MAX structure, facilitates removal of by-products and enables continuous, scalable processing.7 Extending scCO2-assisted strategies to HE-MAX phases could substantially increase the practical availability of HE-MXenes and allow simultaneous tuning of termination composition and interlayer structure, provided that the phase stability of multicomponent carbides under supercritical conditions is confirmed experimentally. Overall, molten-salt, halide-hydrothermal and scCO2-assisted routes expand the toolbox for tailoring defect density, termination chemistry and oxidation resistance beyond what is accessible with HF-based methods. For HE-MXenes, in which the interplay of multiple metals and terminations underpins their emerging electrocatalytic potential, rational adaptation and combination of these etching strategies will be essential.
A complementary direction is purely mechanical or hydrodynamic delamination without chemical intercalants, which directly targets scalable production and preservation of flake integrity. Inman et al. showed that multilayer Ti3C2Tx can be efficiently delaminated by passing concentrated slurries through a three-roll mill, where high shear at the roll gaps peels off single- and few-layer flakes with capacitances comparable to LiCl-delaminated MXene (Fig. 9A).74 High-pressure homogenisation (HPH) extends this concept by combining shear, cavitation and impact forces in a flow-through geometry: ml-MXene is driven through a small orifice into a larger reactor volume, generating cavitation bubbles that nucleate and collapse along the basal planes, leading to delaminated MXene with high yield and throughput and without Li+ or bulky organic intercalants (Fig. 9B).74 In a related shear-stress-induced delamination (SSID) strategy, Zhou et al. used a pan-mill-type wet grinder to subject m-Ti3C2Tx to intense hydrodynamic shear between rotating and stationary discs.75 Compared with manual handshaking, ultrasonication and ball milling (Fig. 9C), the SSID process (Fig. 9D and E) generated few-layer flakes (G-MXene) with larger lateral size, fewer defects and lower oxidation, which translated into higher film conductivity and better mechanical integrity.75
 | ||
| Fig. 9 Delamination strategies and their influence on MXene flake morphology. (A) Shear-delamination of multilayer MXene using a three-roll mill. Reprinted with permission from ref. 76, copyright 2022, Springer Nature. (B) High-pressure homogeniser processing of multilayer MXene, where flow constriction and cavitation drive intercalant-free delamination. Reprinted with permission from ref. 74, copyright 2023, Elsevier. (C) Comparison of mechanical delamination routes: hand shaking, ultrasonication and ball milling. (D) Shear-stress-induced delamination in a pan-mill-type wet grinder. (E) Molecular-dynamics depiction of G-MXene delamination and dispersion during SSID. Reprinted with permission from ref. 75, copyright 2023, Elsevier. | ||
For HE-MXenes, these observations highlight several design principles. First, delamination routes that minimise flake fragmentation and edge oxidation, such as soft organic-base intercalation, HPH, or SSID-type shear grinding, are preferable because catalytic activity and electronic transport depend strongly on lateral size and structural integrity.65,74–76 Second, any method employing strong bases, cavitation or impact must be examined for its influence on multimetal stoichiometry and local terminations, since preferential oxidation or dissolution of one component could erode the intended high-entropy chemistry. Finally, because practical electrocatalytic devices will require gram- to kilogram-scale quantities of HE-MXenes, scalable mechanical schemes such as HPH and SSID, potentially combined with mild intercalation, are especially attractive for translating the emerging promise of HE-MXenes in electrocatalysis from laboratory studies to larger-scale applications.
 | ||
| Fig. 10 Defect and termination control during etching and its implications for MXene-based HER electrocatalysts. (A) and (B) Atomic-resolution HAADF-STEM images and statistics of Ti vacancies and vacancy clusters in monolayer Ti3C2Tx prepared with different HF concentrations, showing increased defect clustering with stronger etching. Reprinted with permission from ref. 77, copyright 2016, American Chemical Society. (C) DFT models of H adsorption on pristine Ti3C2Tx, Ti-vacancy-rich Ti3C2Tx, and Pt anchored at Ti-vacancy sites. (D) Schematic HER pathway on Pt@Ti3C2Tx in acidic media. (E) Calculated HER free-energy diagrams showing that Pt–VTi sites provide the most favourable ΔGH*. Reprinted with permission from ref. 78, copyright 2025, Elsevier. | ||
Building on this, Yang et al. engineered Ti-vacancy-rich Ti3C2Tx as a support for ultra-low-loading Pt, using HF etching time to tune VTi concentration and then anchoring Pt by photodeposition.78 DFT models show that H adsorption on pristine Ti3C2Tx, on vacancy-rich Ti3C2Tx with O terminations, and on Pt anchored at VTi sites leads to distinct charge distributions and hydrogen adsorption free energies (Fig. 10C and E); Pt occupying Ti-vacancy sites gives ΔGH* ≈ −0.38 eV, much closer to thermoneutral than on vacancy-free or vacancy-only surfaces, and is associated with enhanced electronic density of states near the Fermi level and improved charge transport. The corresponding schematic HER pathway for PMX-24 in acidic media (Fig. 10D) highlights how Ti vacancies act as specific anchoring sites that stabilise atomically dispersed Pt and maintain high mass activity at ultralow loading.
For HE-MXenes, these case studies imply that an “appropriate” etching and delamination protocol must do more than yield exfoliated flakes. It should (i) retain the near-equimolar metal distribution of the parent HE-MAX while avoiding preferential dissolution of more labile elements, (ii) generate a controlled, moderate density of metal vacancies and vacancy clusters that can act as active sites or anchoring sites without compromising conductivity, and (iii) produce termination ensembles (O/OH, F, Cl/Br, etc.) that are compatible with the targeted electrocatalytic reaction. Because different metals in a HE-MXene are likely to have different vacancy formation energies and affinities for specific terminations, small changes in HF concentration, etching time or post-treatment can translate into non-uniform local chemistries that alter adsorption energetics. At present, etching and post-treatments mainly controlled termination chemistry at the ensemble level (overall –O/–OH/–F/–Cl ratios) rather than enabling site-specific placement of a given termination above a selected metal atom. Consequently, differences in metal–termination affinities could translate into batch-to-batch variability unless processing histories and termination compositions are carefully standardised and quantified. Systematic comparison of HF and in situ HF etching, molten-salt and alkali-assisted routes, together with carefully chosen delamination strategies, therefore needs to be coupled with atomic-scale characterisation (AC-HAADF-STEM, XPS, EPR, ICP) to map how processing histories reshape defect and termination landscapes.77,78 In the context of high-entropy electrocatalysis, controlling these landscapes is central to preserving compositional integrity while deliberately creating the ensembles of active sites that underpin the performance gains discussed in later sections.
3.3 Structural and chemical characterization
Establishing that a material is a genuine HE-MXene, rather than a mixture of phases or a partially etched laminate, requires complementary diffraction, microscopy and spectroscopy (Fig. 11). At the average-structure level, powder XRD with Rietveld refinement is used to confirm that HE-MAX precursors are single-phase and to track their transformation into HE-MXenes. For TiVNbMoAlC3 and TiVCrMoAlC3, Nemani et al. showed that both the MAX and MXene stages display single sets of reflections consistent with M4AlC3 and M4C3 stacking, with progressive broadening and low-angle shifts of the (002) peak on going from MAX to multilayer and then delaminated films (Fig. 11A).22 Similar XRD signatures, single-phase M2AX patterns and expanded (000ℓ) reflections after etching have been reported for Ga-containing medium/high-entropy (Ti,V,Nb,Ta,Mo)2CTx and related systems, underlining that high-entropy mixing can be realised in both M4X3- and M2X-type MXenes.16,47 | ||
| Fig. 11 Structural and chemical characterisation of representative HE-MXenes. (A) XRD patterns of TiVNbMoAlC3 and TiVCrMoAlC3 HE-MAX phases and their corresponding HE-MXenes, showing phase evolution and the low-angle shift/broadening of the (002) peak after etching/delamination. Reprinted with permission from ref. 22, copyright 2021, American Chemical Society. (B) Atomic-resolution HAADF-STEM image of TiVNbMoC3Tx showing multi-metal occupation and lattice distortion. Reprinted with permission from ref. 29, copyright 2021, John Wiley & Sons. (C) Aberration-corrected HAADF-STEM image of (Ti,V,Zr,Nb,Ta)2CTx illustrating the layered structure and local distortion. Reprinted with permission from ref. 47, copyright 2023, John Wiley & Sons. (D) TEM, AFM, SAED, HAADF-STEM/EDS, XPS and XRD analyses of monolayer TVNMC HE-MXenes confirming single-layer thickness and uniform elemental distribution. Reprinted with permission from ref. 16, copyright 2025, Elsevier. (E) XRD, morphology and elemental mapping of Ga-containing (Ti,V,Nb,Ta,Mo)2CTx MXenes prepared by Lewis-acid molten-salt etching, showing phase purity, accordion-like morphology and homogeneous multi-metal distribution. Reprinted with permission from ref. 47, copyright 2023, John Wiley & Sons. (F) Representative EDS spectrum of (Ti,V,Zr,Nb,Ta)2CTx confirming near-equimolar metal mixing on the M-sublattice. Reprinted with permission from ref. 29, copyright 2021, John Wiley & Sons. | ||
Local chemical disorder and lattice distortion, which are central to high-entropy behaviour, must be resolved by high-resolution electron microscopy. STEM-HAADF imaging of TiVNbMoC3Tx reveals atomic columns with varying intensities arranged in a distorted hexagonal lattice, consistent with multi-metal occupation of the M-sublattice (Fig. 11B).29 Du et al. observed analogous contrast fluctuations and non-ideal M–X–M angles in (Ti,V,Zr,Nb,Ta)2CTx, directly visualising the strain field associated with multi-principal-element mixing (Fig. 11B and F).29 More recently, monolayer TVNMC HE-MXenes have been characterised by a combination of TEM, SAED, AFM and atomic-resolution HAADF-STEM/EDS, demonstrating single-layer morphology, uniform Ti/V/Nb/Mo/C distributions and Vegard-like shifts in diffraction peaks relative to binary analogues (Fig. 11D).16 Together, these studies show that genuine HE-MXenes exhibit both long-range layered order and nanoscale chemical disorder without detectable metal clustering.
Spectroscopic probes then establish oxidation states, termination chemistry and element-specific local environments. XPS survey and high-resolution spectra provide the relative amounts of C, N, O, F, Cl and other anions, deconvolve M–C, M–N, M–O and M–Cl/F contributions and verify near-equimolar metal ratios through depth-profiling.22,42,46,77,78 In HE-MXenes such as (Ti,V,Zr,Nb,Ta)2CTx and Ga-derived (Ti,V,Nb,Ta,Mo)2CTx, the coexistence of multiple oxidation states and mixed –O/–Cl (or –F/–O) terminations has been correlated with the high density of distinct adsorption sites inferred from electrochemical measurements.29,47 Element-selective X-ray absorption spectroscopy (XANES/EXAFS) further refines this picture by resolving coordination numbers, bond lengths and disorder parameters separately for each metal, allowing short-range order and local electronic structure to be quantified in truly multicomponent lattices.35,79
For electrocatalysis, static ex situ characterisation is insufficient: structural and chemical evolution under potential must also be monitored. Operando Raman and XAS have begun to reveal termination rearrangements, oxidation-state changes and possible reconstruction of HE-MXene surfaces during HER, OER and related reactions, while electrochemical impedance spectroscopy links flake restacking, defect density and termination composition to charge-transfer resistance and ion transport.80,81 Careful integration of these operando tools with the ex situ methods summarised above is therefore essential to build reliable composition–structure–function relationships and to substantiate the claimed high-entropy advantages of HE-MXenes in sustainable electrocatalysis.22,38
4. Electronic structure, active sites and theoretical modelling
4.1 Electronic structure and lattice distortion in high-entropy MXenes
Substituting several transition metals on the M-sublattice profoundly modifies the electronic structure of MXenes relative to mono-metal systems such as Ti3C2Tx or Mo2CTx. In single-metal MXenes, states near the Fermi level originate from a narrow set of M-d orbitals, giving a relatively well-defined d-band centre and a limited spread of adsorption energies for surface intermediates. In contrast, HE-MXenes such as TiVNbMoC3Tx or (Ti,V,Zr,Nb,Ta)2CTx feature multiple d-manifolds with slightly different energies and occupancies, producing broadened projected densities of states and locally varying metal–C/T bond strengths (Fig. 12A). High-throughput DFT calculations on TiVNbMoC3 and TiVCrMoC3 have shown that distinct local metal ensembles give rise to a wide distribution of local d-band centres and crystal-orbital Hamilton populations for M–C and M–T bonds, in sharp contrast to the electronically more uniform Ti3C2Tx.82 Charge-transfer analyses indicate partial electron donation from more electropositive metals (e.g. Zr, Nb) to more electronegative neighbours (e.g. V, Mo, Ta), which adjusts the occupation of antibonding d states and further diversifies the local electronic environments. | ||
| Fig. 12 Electronic structure and lattice distortion in HE-MXenes and related HEMs. (A) Evolution of MXene lattices from ordered to high-entropy configurations as the number of metals on the M-sublattice increases. Reprinted with permission from ref. 82, copyright 2025, Springer Nature. (B) Schematic of lattice distortion in high-entropy MAX/MX phases caused by random multi-metal occupancy. Reprinted with permission from ref. 29, copyright 2021, John Wiley & Sons. (C) Phonon-transport engineering in HE-MXenes, where cation disorder enhances phonon scattering and lowers lattice thermal conductivity. Reproduced with permission from ref. 86, copyright 2024, Elsevier. (D) Local strain, lattice expansion/contraction, and defect formation in heavily distorted high-entropy lattices. Reproduced with permission from ref. 87, copyright 2025, John Wiley & Sons. (E) Lattice-distortion engineering in high-entropy diborides, showing the role of vacancies and nanoclusters in tuning electrical and interfacial properties. Reproduced with permission from ref. 88, copyright 2025, Elsevier. | ||
This electronic heterogeneity translates directly into continuous distributions of adsorption energies for electrocatalytic intermediates. On MXene basal planes, intermediates may adsorb on on-top M sites or on multi-metal bridge/hollow motifs coordinated by two or three neighbouring surface metal atoms; in HE-MXenes these can be viewed as bi-/tri-metal ensemble sites whose atomic identities vary statistically across the surface. Therefore, the “continuous” adsorption-energy distribution arose from the local ensemble geometry/composition of the coordinating metals (e.g., a given bridge/hollow motif) and its corresponding site-specific electronic structure, which can be captured by a local d-band centre (or related bond-strength descriptor) rather than a single global d-band centre for the entire surface. In practice, intermediates did not converge on one universal “tri-metal bridge site”; instead, the preferred motif depended on the adsorbate and termination environment, while the binding strength within a given motif scaled with the local electronic descriptor. For HE-MXenes, DFT studies on O-terminated TiVNbMoC3 found that the hydrogen adsorption free energy, ΔGH*, spans a broad range across different surface sites, with a significant fraction of configurations having near-optimal ΔGH* values close to zero, thereby rationalising improved HER activity compared with simpler carbides.82 A systematic survey of HE M2C(T)2 MXenes (M = Ti, V, Nb, Mo, Ta; T = O, OH, F) further demonstrated quasi-continuous ΔGH* distributions arising from random metal and termination arrangements; importantly, each composition statistically contains a subset of sites with ΔGH* in the optimum ±0.1–0.2 eV window even when the average binding is suboptimal.18 Similar ensemble effects have been predicted and, in related HEMs, observed for *O, *CO and *N2Hx intermediates, suggesting that HE-MXenes can host dense landscapes of near-optimal sites for multi-step reactions such as OER/ORR, CO2RR and NRR.83–85
Electronic complexity is intimately linked to lattice distortion. Random occupancy of metals with different radii and bonding preferences generates local compressive and tensile regions, deviations from ideal hexagonal symmetry and non-uniform interatomic distances in both HE-MAX precursors and their MXene derivatives (Fig. 12B).29 In (Ti,V,Zr,Nb,Ta)2CTx, atomic-resolution STEM has directly visualised such distortions, while phonon-transport calculations on model HE-MXenes show that cation disorder introduces strong static strain fields and intensive phonon scattering, driving the lattice thermal conductivity to values far below those of single-metal analogues (Fig. 12C).86 More generally, entropy-engineered 2D materials and high-entropy ceramics exhibit strain-induced lattice expansion/contraction, grain-boundary formation and vacancy generation (Fig. 12D), which modulate bond lengths, local symmetry and charge density around active sites.29,87 Recent work on high-entropy diborides further illustrates how lattice distortion, metal vacancies and chemical nanoclusters cooperate to enhance charge transport and interfacial polarization (Fig. 12E), providing a broader framework for understanding how distortion-controlled local fields and defect landscapes can be exploited in high-entropy systems.88 Taken together, HE-MXenes should be viewed as electronically and mechanically heterogeneous 2D carbides/carbonitrides in which broadened, compositionally tunable d-bands coexist with disorder-induced strain and defect fields. This combination naturally generates statistical ensembles of adsorption sites with a continuum of binding energies and dynamic response to external fields, which underpins their potential as platform electrocatalysts for complex, multi-electron energy-conversion reactions.
4.2 Surface terminations and defect chemistry
In MXenes, and even more so in HE-MXenes, surface terminations are part of the crystal structure rather than incidental functional groups. The composition and arrangement of –O, –OH, –F, –Cl and other terminations are set by the etching route and subsequent treatments,7,42,89 but their thermodynamic role is now better quantified. High-throughput DFT screening across 29 transition metals, four layer thicknesses (n = 1–4) and eleven terminations showed that the energy above the convex hull, ΔEhull, is governed at least as strongly by the terminating species as by the underlying metal carbide or nitride (Fig. 13A–C).90 Large regions of the Mn+1XnT2 design space are predicted to be thermodynamically stable or metastable only for specific terminations (in particular O- and F-based groups), while other terminations stabilise only narrow subsets of compositions. This map is directly relevant to HE-MXenes: any attempt to combine multi-principal M-sublattices with unconventional terminations must respect these stability windows or rely on electrochemical environments that can kinetically stabilise metastable surfaces. | ||
| Fig. 13 Termination- and defect-dependent stability and mechanical behaviour of MXenes and implications for HE-MXenes. (A) High-throughput DFT heat map of the thermodynamic stability of Mn+1XnT2 compositions across transition metals and terminations. (B) Computational screening workflow and the number of stable/metastable compositions for different terminations. (C) Statistics of stable and metastable MXenes as a function of metal, layer number, termination, and carbide/nitride chemistry. Reprinted with permission from ref. 90, copyright 2024, American Chemical Society. (D) Pressure-dependent interlayer distances in OH-terminated MXenes and MXene heterostructures, illustrating the role of hydrogen-bonded terminations in structural response. Reprinted with permission from ref. 98, copyright 2023, Springer Nature. (E) Schematic of tunable MXene chemistry and defects enabled by different etching strategies. (F) Calculated 2D stiffness of M2CTx monolayers for different terminations, highlighting the stiffening effect of O-based terminations. Reprinted with permission from ref. 99, copyright 2021, John Wiley & Sons. | ||
For electrocatalysis, these terminations regulate not only stability but also the distribution of adsorption energies and interfacial properties. Existing experimental and computational studies on mono- and double-transition-metal MXenes have already shown that O-rich surfaces generally strengthen binding of H*, O* and OOH*, often pushing HER or OER activity towards the top of volcano plots, whereas F-rich surfaces tend to weaken adsorption and can suppress CO2 activation.89–93 In HE-MXenes, this termination dependence is superimposed on the compositional heterogeneity of the M-sublattice: the same –O or –OH group can experience very different local electronics depending on whether it is coordinated to, for example, Ti–V–Nb triplets or Mo–Ta–Zr ensembles. For sustainable energy conversion, a key design target is to control terminations and local active-site environments. Recent DFT work on multi-metal M2C(T)2 MXenes indicates that the distribution of hydrogen adsorption free energies ΔGH* depends jointly on termination identity and the underlying metal cluster.18 Importantly, current top-down synthesis routes (etching followed by washing/intercalation and optional post-treatments) did not enable deterministic, site-specific regulation of terminations on a metal-by-metal basis; instead, terminations populated surface sites statistically, with possible local preferences dictated by the metal identity and local coordination environment. This implied that HE-MXene surfaces should be described by joint distributions of metal-neighbourhood motifs and termination species, and it also raised the possibility of batch-to-batch differences in activity/stability if termination ratios and post-treatment histories varied between preparations. Accordingly, we emphasised the need to report etching/post-treatment conditions and to quantify termination composition (e.g., by XPS depth profiling and complementary surface-sensitive analyses) when comparing HE-MXenes across studies. Implying that mixed terminations on HE-MXenes generate a dense spectrum of adsorption sites, some of which naturally fall near optimal binding energies for HER and for the suppression of competing reactions. For sustainable energy conversion, a key design target is therefore not a single ideal termination, but a statistically favourable mixture of terminations and metal environments that provides both active and corrosion-resistant sites. Here, “corrosion-resistant” referred specifically to retarded surface oxidation and reduced metal dissolution/leaching under electrochemical bias, consistent with diffusion-limited degradation pathways expected for chemically disordered (high-entropy) lattices.
Defects introduce a second, closely coupled level of control. Point defects on the M, X or termination sublattices, antisite defects and extended defects such as grain boundaries all alter local coordination and charge distribution, thereby modifying both catalytic activity and stability.89,94–97 Atomistic simulations of OH-terminated MXenes under load illustrate how terminations and defects together govern mechanical response: in a model tribological heterostructure, the interlayer H–H and O–H distances remained nearly constant under increasing pressure, indicating that hydrogen bonding between opposing terminations plays a dominant role in controlling layer spacing and shear behaviour (Fig. 13D).98 Such mechanically robust, hydrogen-bonded interfaces are particularly attractive for HE-MXene electrodes that must tolerate repeated gas evolution, volume changes and bubble detachment during HER or OER without delamination.
The available defect-termination design space is further expanded by recent synthesis developments. Moving from concentrated HF to mixed-acid, non-aqueous or molten-salt etching allows one to reduce vacancy densities, access non-Al A-site chemistries and introduce less conventional terminations such as –N, –S, –Se or –Te, all of which are promising for tailoring adsorption and corrosion resistance in HE-Mxenes (Fig. 13E).99 The same perspective shows that surface terminations also control mechanical properties: calculated 2D stiffness values for M2CTx monolayers vary strongly with both metal choice and termination, with O-terminated MXenes generally stiffer than their F-, OH- or Cl-terminated counterparts across the 3d–5d series (Fig. 13F).47,61–64,99 Strong M–O bonding increases M–X bond strength and suppresses detrimental M–M interactions, whereas more ionic M–F or M–(OH) bonding has a much weaker stiffening effect.87–89 For HE-MXenes, these trends imply that appropriately chosen O-rich termination ensembles can simultaneously enhance electrocatalytic activity and mechanical robustness, while controlled introduction of vacancies can be used to create additional active sites at the cost of some stiffness. Overall, surface terminations and defects in HE-MXenes should be treated as coupled, designable variables rather than unavoidable by-products of etching. High-throughput thermodynamic maps (Fig. 13A–C), tribological simulations (Fig. 13D) and mechanical modelling (Fig. 13E and F) together suggest a practical strategy: first select metal combinations and terminations that fall within the thermodynamic and mechanical stability windows, then use etching, post-treatment and electrochemical conditioning to engineer a controlled population of vacancies and mixed terminations on those multi-metal lattices. Systematically linking such termination–defect motifs to HER, OER or CO2RR performance in HE-MXenes remains a major opportunity for future work in electrocatalysis for sustainable energy conversion.
5. High-entropy MXenes for hydrogen-related electrocatalysis
5.1 Hydrogen evolution activity and high-entropy MXene-based heterostructures
A central advantage of HE-MXenes for HER is that configurational disorder produces not a single hydrogen adsorption energy but a distribution of ΔGH* values associated with different local metal–termination ensembles. High-throughput DFT calculations on high-entropy M2C(T)2 compositions (M = Ti, V, Nb, Mo, Ta; T = O, OH, F) showed that multi-metal mixing broadens the ΔGH* spectrum on OH-terminated surfaces and increases the fraction of sites with near-optimal binding relative to monometallic Ti2C(T)2.18 HE-MXenes can therefore act as multi-site HER platforms in which activity is governed by a statistically significant subset of favourable local motifs rather than by the average adsorption energy of the surface (Fig. 14A and B). | ||
| Fig. 14 HE-MXenes as multi-site HER platforms and conductive scaffolds. (A) Schematic of distributed H adsorption and diffusion on HE-MXene surfaces. (B) H adsorption energies on OH-terminated HE-MXenes, showing many near-optimal sites. Reprinted with permission from ref. 18, copyright 2025, Elsevier. (C) Synthesis of V–Co2P@HE on Ni foam. (D) and (E) HER polarization and Tafel analysis of V–Co2P@HE and reference catalysts. (F) HER comparison with representative non-noble catalysts. Reprinted with permission from ref. 31, copyright 2023, Elsevier. | ||
The clearest experimental HER demonstration so far is the V–Co2P@HE heterostructure reported by Ma and co-workers.31 In this design, single-layer (TiVCrNbMo)5C4Tx HE-MXene was electrodeposited on Ni foam, followed by growth of V–Co layered hydroxide precursors and phosphidation to form a Co2P nanonetwork strongly coupled to the HE-MXene scaffold (Fig. 14C). The HE-MXene served as a conductive and hydrophilic support, while interfacial coupling and V doping tuned the electronic structure of Co2P and facilitated charge redistribution. XPS/XAS and DFT results indicated optimised water adsorption, reduced H2O dissociation barriers, and more favourable H adsorption at the heterointerface, consistent with accelerated Volmer–Heyrovsky kinetics.
In 1 M KOH, V–Co2P@HE required only ∼33 mV to reach 10 mA cm−2 and showed a Tafel slope of ∼47 mV dec−1, outperforming Co2P, Co2P@HE, V–Co2P and even Pt/C under the same conditions (Fig. 14D and E).31 It also displayed good durability at high current density and enabled efficient overall water splitting when used as part of a bifunctional electrode pair (Fig. 14F). These results suggest that, for HER, HE-MXenes are most compelling when used as electronically active, multicomponent scaffolds that provide both distributed adsorption sites and strong interfacial coupling to catalytically active phosphide-, sulfide- or hydroxide-type phases. At present, this HE-MXene-based heterostructure strategy is better supported experimentally than claims of stand-alone HE-MXene HER catalysis.
5.2 Hydrogen oxidation reaction and reversible hydrogen electrodes
Direct studies of HE-MXenes for HOR have not yet been reported. Nevertheless, a concise design blueprint can be drawn from MXene-supported alkaline HOR catalysts, where conductive carbide surfaces and strong metal–support interactions help balance H* and OH* adsorption. A representative example is Ru–CuO/MXene supported on Mo2TiC2Tx (Fig. 15A), which delivered higher alkaline HOR activity and better CO tolerance than commercial Pt/C (Fig. 15B and C) through interfacial charge redistribution at the Ru–O–Cu/MXene junction. 107 DFT and spectroscopic analyses showed that the heterointerface weakened H binding on Ru, strengthened OH binding on CuO, and lowered the overall HOR barrier (Fig. 15D–H). | ||
| Fig. 15 Ru–CuO/MXene heterojunctions for alkaline HOR. (A) Synthesis and structure of Ru–CuO/MXene. (B) and (C) HOR polarization and Tafel analyses. (D) Proposed HOR mechanism with Ru and CuO sites for H* and OH* adsorption. (E)–(H) Electronic-structure, adsorption-energy, charge-density and free-energy analyses of Ru–CuO/MXene. Reprinted with permission from ref. 100, copyright 2024, American Chemical Society. | ||
For HE-MXenes, the relevance of this example is conceptual rather than demonstrative. Multi-metal M-sublattices and mixed terminations could provide additional handles to tune electronic metal–support interactions, stabilise Ru- or Pt-lean heterojunctions, and balance H/OH adsorption under alkaline HOR conditions. Future studies should therefore test whether HE-MXene supports can outperform conventional MXenes in reversible hydrogen electrodes by improving heterointerface stability, lowering noble-metal loading, and sustaining activity under oxidising anode conditions. Until such experiments are available, HOR should be regarded as a promising design direction for HE-MXenes rather than an experimentally established application.
6. High-entropy MXenes for oxygen electrocatalysis
OER and ORR remain key kinetic bottlenecks in water electrolysers, fuel cells and rechargeable metal–air batteries.101 HE-MXenes are relevant to these reactions primarily as multicomponent conductive scaffolds and interfacial modulators, rather than as already established stand-alone oxygen electrocatalysts. Their multi-metal M-sublattices, mixed terminations and high conductivity can tune interfacial charge transfer, stabilise oxygen-active phases and broaden local adsorption environments, while direct experimental demonstrations of HE-MXenes in oxygen electrocatalysis are still limited.18,30,102–104 Accordingly, this section centres on emerging HE-MXene-based oxygen-electrocatalytic architectures, with conventional MXene examples included only where they provide concise transferable design rules.30,105,114,1156.1 Oxygen evolution reaction in water splitting and metal–air batteries
 | ||
| Fig. 16 Representative MXene-based OER architectures. (A) MXene platforms for OER, including 2D–2D stacks, single-atom sites and supported clusters. (B) and (C) NiFe-LDH/Ti3C2 synthesis and OER performance. Reprinted with permission from ref. 106, copyright 2024, Elsevier. (D) and (E) LDH/MXNF electrode fabrication and OER activity. Reprinted with permission from ref. 102, copyright 2024, MDPI. (F)–(I) H2PO2−/FeNi-LDH–V2C interface model, Zn–air battery concept, cycling stability and synthesis. Reprinted with permission from ref. 103, copyright 2021, Elsevier. | ||
For the present, these systems are best regarded as design guidance rather than direct HE-MXene evidence. Their main message is that HE-MXenes need not be the sole oxygen-active phase; instead, they can function as electronically tunable, multicomponent supports for LDH- or oxyhydroxide-based catalysts, where high-entropy mixing on the MXene side may further diversify interfacial adsorption energetics and improve durability.
 | ||
| Fig. 17 MXene–phosphide heterostructures for alkaline OER. (A) Schematic of NiFe-P/Ti3C2Tx, where bimetallic phosphide nanostructures are anchored on conductive Ti3C2Tx MXene. (B) Tafel plots showing faster OER kinetics for NiFe-P/Ti3C2Tx than for reference catalysts. Reprinted with permission from ref. 107, copyright 2025, Elsevier. (C) Synthesis of CoFe–P/Ti3C2Txvia CoFe-LDH growth on Ti3C2Tx followed by phosphidation. (D) LSV curves showing improved OER performance of CoFe–P/Ti3C2Tx relative to CoFe–P, CoFe-LDH, Ti3C2Tx and RuO2. Reprinted with permission from ref. 108, copyright 2022, John Wiley & Sons. | ||
 | ||
| Fig. 18 Representative OER/ORR-related high-entropy oxide–MXene systems. (A) HEBOx/Ti3C2Tx for alkaline OER. Reprinted with permission from ref. 19, copyright 2024, Elsvier. (B) Microwave synthesis of spinel HEO/Ti3C2Tx hybrids. Reprinted with permission from ref. 30, copyright 2023, John Wiley & Sons. (C) and (D) OER activity and kinetics of HEBOx, HEBOx/MXene and RuO2. Reprinted with permission from ref. 19, copyright 2024, Elsvier. (E) and (F) LSV and Tafel analysis of Ti3C2Tx, HEO/Ti3C2Tx hybrids and IrO2. Reprinted with permission from ref. 30, copyright 2023, John Wiley & Sons (G) MXene-supported double-atom catalysts in a rechargeable metal–air battery. Reprinted with permission from ref. 145, copyright 2021, John Wiley & Sons. (H) High-entropy spinel oxide with diverse local catalytic environments. Reprinted with permission from ref. 146, copyright 2024, American Chemical Society. | ||
6.2 Oxygen evolution reaction in fuel cells and metal–air systems
Direct ORR studies using HE-MXenes themselves remain scarce, so current evidence is strongest for MXene-supported or MXene-derived architectures. A representative non-precious example is Co–S–N–C@MXene, where the Ti3C2Tx substrate improves conductivity, active-site utilisation and durability, enabling ORR performance competitive with Pt/C in alkaline media and strong anion-exchange membrane fuel-cell output (Fig. 19A).109 Although this is not an HE-MXene system, it shows how a conductive 2D carbide scaffold can stabilise dense populations of catalytic sites. A more relevant high-entropy example is the MXene-derived M–TiO2/HEA heterojunction, in which laser treatment of metal-salt-loaded Ti3C2Tx simultaneously generated TiO2 and FeCoNiCrTi high-entropy alloy nanoparticles (Fig. 19B–D).110 Strong coupling at the HEA/TiO2/MXene interface improved oxygen-electrocatalytic behaviour and enabled durable photo-assisted Zn–air battery operation. This system is still not a direct demonstration of pristine HE-MXene ORR catalysis, but it illustrates how MXene-derived supports and high-entropy active phases can be integrated into bifunctional oxygen electrodes. The evidence in oxygen electrocatalysis presently supports a support- and interface-centred role for HE-MXenes and HE-MXene-inspired architectures. The most credible near-term direction is therefore not to claim established stand-alone HE-MXene OER/ORR catalysis, but to develop HE-MXenes as multicomponent conductive frameworks that stabilise high-entropy oxides, phosphides, alloys or related oxygen-active phases while tuning interfacial charge transfer, defect chemistry and structural durability. | ||
| Fig. 19 MXene-based high-entropy architectures for advanced cathodic electrocatalysis. (A) ORR comparison of Co–S–N–C@MXene with reference catalysts in alkaline media. Reprinted with permission from ref. 109, copyright 2025, American Chemical Society. (B) Laser-pulse synthesis of TiO2/high-entropy alloy heterostructures on Ti3C2Tx. (C) Tafel slope extracted from the OER polarization curves. (D) Corresponding Tafel plots of M-TiO2/HEAs and reference catalysts, highlighting the improved oxygen electrocatalysis and light-assisted Zn–air battery performance. Reprinted with permission from ref. 110, copyright 2025, John Wiley & Sons. (E) Schematic illustration of HE-MXene/G@PP and its role in suppressing lithium polysulfide dissolution while promoting electron transfer in Li–S batteries. Reprinted with permission from ref. 15, copyright 2024, American Chemical Society. | ||
7. High-entropy MXenes for other electrochemical conversion reactions
Beyond hydrogen and oxygen electrocatalysis, HE-MXenes are promising for selectivity-sensitive electrochemical transformations such as CO2 reduction and nitrogen-related reactions, where the key challenge is to favour target intermediates while suppressing competing HER.93,111,112 In principle, HE-MXenes are attractive because multi-metal mixing, mixed terminations and lattice distortion can generate a broad distribution of local adsorption environments and decouple the binding of *H from that of *CO2, *COOH, *N2 or *NNH.18,22,35,37,38 However, direct experimental demonstrations of HE-MXenes in these reactions remain limited. Accordingly, this section focuses on the most relevant HE-MXene-specific insights and HE-MXene-based design opportunities, while conventional MXene and other high-entropy systems are discussed only briefly where they provide transferable mechanistic guidance.93,111,113–1177.1 CO2 reduction reaction
 | ||
| Fig. 20 MXene-based active-site concepts for HE-MXene CO2RR. (A) CO2RR cell and DACs on Ti2CO2 MXene. (B) Adsorption and reaction energetics identifying DACs/Ti2CO2 compositions selective for CO2RR over HER. Reprinted with permission from ref. 118, copyright 2025, Royal Society of Chemistry. (C) TM–Ti–O descriptor for MXene-based SAC screening. (D) Electronic-structure analyses of Ti-, V- and Mn-modified Ti2CO2 surfaces. Reprinted with permission from ref. 119, copyright 2023, Elsevier. | ||
 | ||
| Fig. 21 Design concepts for MXene and HE-MXene CO2RR catalysts. (A) CO2RR/HER competition on terminated MXene surfaces. (B) Potential–pH phase diagrams of Ti3C2, Mo2C and V2C under CO2RR conditions. Reprinted with permission from ref. 120, copyright 2024, American Chemical Society. (C) Faradaic efficiencies of H2 and CO for Ag-intercalated Ti3C2Tx and Ag/Ti3C2Tx catalysts, showing selectivity tuning by interlayer engineering. (D) Illustration of in situ oxidation of nanostructured Ti3C2Tx to form TiOx/Ti3C2 heterostructures that modulate the electronic structure and enhance CO production. Reprinted with permission from ref. 121, copyright 2024, Elsevier. (E) MXene-based single-atom catalysts, where isolated transition-metal atoms anchored on O-terminated Ti2CO2 provide well-defined CO2 activation sites. (F) Calculated free-energy profile for CO2 reduction to CH4 on Ti@Ti2CO2, highlighting the sequence of proton–electron transfer steps and the low limiting potential. Reprinted with permission from ref. 122, copyright 2023, American Chemical Society. | ||
7.2 Nitrogen reduction reaction and ammonia-related chemistry
 | ||
| Fig. 22 (A) High-entropy and MXene-based NRR catalyst concepts. (A) Mixing-entropy-dependent NH3 production in high-entropy sulfides. (B) Comparison of key NRR and electrochemical metrics for high-entropy sulfide catalysts. Reprinted with permission from ref. 125, copyright 2025, Elsevier. (C) Ti3C2Tx nanosheets on a vertical metal host for ambient NRR. (D) Adsorption-site analysis and NRR performance of MXene/FeOOH hybrids. Reprinted with permission from ref. 123, copyright 2019, Elsevier. (E) Surface-tuned MXene/TiFeOx nanosheets. (F) NH3 yield and FE of TiFeOx-modified catalysts. Reprinted with permission from ref. 124, copyright 2020, American Chemical Society. | ||
 | ||
| Fig. 23 High-entropy sulfide and perovskite catalysts for nitrate/nitrogen reduction. (A) Formation of hollow NiCoFeV–S high-entropy sulfide. (B) NH3 yield showing improved activity of NiCoFeV–S over lower-entropy sulfides. Reprinted with permission from ref. 126, copyright 2025, Royal Society of Chemistry. (C) Synthesis of porous Bax(FeCoNiZrY)0.2O3−δ high-entropy perovskites. (D) NH3 yield of stoichiometric and vacancy-rich perovskites. (E) d-Band-centre analysis of candidate active sites. (F) Charge–density difference near oxygen-vacancy-associated Ni sites. (G) Free-energy profiles for eNRR on stoichiometric and vacancy-rich surfaces. Reprinted with permission from ref. 127, copyright 2022, Springer Nature. | ||
10. Perspectives and concluding remarks
HE-MXenes have emerged as a distinct branch of MXene chemistry in which multi-metal lattices, rich defect structures and heterogeneous terminations are deliberately combined to modulate adsorption energies and reaction pathways for key electrochemical conversions. This review indicate that convincing proof-of-concept has already been obtained across HER/HOR, OER/ORR and small-molecule transformations such as CO2RR and NRR, with activity and stability metrics that increasingly rival or complement conventional MXenes and selected noble-metal systems. At the same time, the field remains synthesis- and mechanism-limited: phase-pure HE-MAX precursors are difficult to prepare reproducibly, multi-step etching and delamination routes often compromise compositional integrity and termination control, and ensemble-averaged characterisation struggles to resolve the true distribution of local environments that govern catalytic behaviour. Most studies still rely on half-cell testing under modest current densities, leaving considerable uncertainty about long-term durability, degradation mechanisms and scalability in realistic devices.Looking ahead, HE-MXenes can play a central role in sustainable electrocatalysis only if synthetic, mechanistic and device-level challenges are addressed in a coordinated way. More predictive approaches to composition selection and phase stability, greener and better-controlled etching strategies, and multi-scale operando characterisation will be essential to transform high-entropy design from an empirical concept into a rigorous framework for tailoring active sites and stabilising them under bias. Equally important is systematic benchmarking in electrolyzers, fuel cells, metal–air batteries and CO2/NRR electrolysers under industrially relevant conditions, together with explicit consideration of element criticality, recyclability and life-cycle impacts. When combined with data-driven exploration of high-dimensional composition space and rational integration with other high-entropy and single-atom components, HE-MXenes have the potential to evolve from an emerging material class into a versatile platform for low-carbon energy technologies. In this context, several focused directions appear particularly important (Fig. 24):
 | ||
| Fig. 24 Schematic depiction of the core effects, properties and future development directions of HE-MXenes in electrocatalysis. | ||
(a) Phase stability calculations, high-throughput electronic-structure screening and ML-assisted evaluation of configurational-entropy descriptors (ΔSmix, Ω, δ, ΔHmix) should be coupled with combinatorial synthesis to identify composition windows that reliably yield single-phase HE-MAX precursors and robust HE-MXenes without extensive trial-and-error optimisation.
(b) Fluoride-free molten-salt, halogen, or electrochemical routes tailored to multi-metal carbides are needed to minimize preferential leaching and allow deliberate tuning of –O/–OH/–F/–Cl/–S terminations, while reducing the environmental burden and improving scalability relative to conventional HF or in situ HF protocols.
(c) Standard pipelines for HE-MXenes should integrate atomic-resolution STEM/EDS, pair-distribution-function analysis and operando XAS, XPS and vibrational spectroscopy under HER/HOR, OER/ORR, CO2RR and NRR conditions, combined with microkinetic and Monte-Carlo models that explicitly treat distributions of adsorption energies rather than single descriptor values.
(d) Future studies should increasingly report performance in realistic membrane–electrode assemblies, metal–air cells and flow-cell reactors, including overpotentials at ≥500–1000 mA cm−2, accelerated stress tests, gas-evolution and mass-transport effects, as well as post-mortem analysis of oxidation, metal dissolution, delamination and termination loss.
(e) Combining HE-MXenes with other high-entropy subsystems such as HEA nanoparticles, HEO or sulfides, and MXene-anchored single-atom centres offers a route to catalysts where activity, selectivity and stability emerge from cooperative interactions across multiple length scales, including possible self-healing or benign-oxidation behaviour under operation.
(f) Curated databases that capture full compositional statistics, synthesis conditions, structural and termination characterisation, and electrochemical protocols for HE-MXenes will enable interpretable ML models for property prediction and inverse design, provided that reporting standards are harmonised across laboratories.
(g) Many current HE-MXene formulations rely on elements such as Nb, Mo, Ta or noble metals. Future work should evaluate alternative, more abundant chemistries, develop recycling and regeneration strategies for spent electrodes, and integrate techno-economic and life-cycle assessments into materials selection and device design.
Alignment of these efforts is expected to convert HE-MXenes from an emerging scientific curiosity into a mature, versatile platform for electrocatalytic hydrogen, oxygen and small-molecule conversion within sustainable energy systems.
Author contributions
Muhammad Yousuf: writing – original draft, resources. Xiaopeng Liu: review and editing. Connor W. Schroeder: review and editing. Jonathan Tjioe: review and editing. Yurui Liu: review and editing. Yue Wu: writing – review & editing, project administration, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research findings, software, or code were included, and no new data were generated or analysed in this review.Acknowledgements
Prof. Yue Wu thanks the support of the Herbert L. Stiles Professorship from Iowa State University and NSF CBET 241425.Notes and references
- L. Gao, Y. Wei, J. Zhang, Y. Ou and L. Chen, Coord. Chem. Rev., 2026, 553, 217569 CrossRef CAS.
- L. Dadsena, T. Maharana and S. E. Jujjavarapu, Environ. Res., 2026, 123764 CrossRef CAS PubMed.
- W. Wang, X. X. Zhang, T. Yu, R. Guo and X. Liu, J. Energy Storage, 2026, 145, 119807 CrossRef CAS.
- X. Liang, T. Zhou, L. Liu, H. Shui, S. Zhang, Y. Zheng and L. Li, Int. J. Hydrogen Energy, 2026, 207, 153449 CrossRef CAS.
- R. Yang, J. Liu, M. Lin and P. Zeng, Global Energy Interconnection, 2026, 9, 159–185 CrossRef.
- Z.-Y. Tian, M. Yousuf, Z.-M. Wang, R. Jabeen, A. E. Mansy, S. Daniel and R. Kumar, in Environmental Chemistry: Advanced Concepts and Applications, ed. Z.-Y. Tian, Springer Nature, Singapore, 2025, pp. 15–41 Search PubMed.
- Y. Zhao, J. Zhang, X. Guo, X. Cao, S. Wang, H. Liu and G. Wang, Chem. Soc. Rev., 2023, 52, 3215–3264 RSC.
- M. Yousuf, Z. Y. Tian, Z. M. Wang, R. Jabeen, A. E. Mansy, S. Daniel and R. Kumar, in Environmental Chemistry: Advanced Concepts and Applications, ed. Z. Y. Tian, Springer Nature, Singapore, 2025, pp. 43–66 Search PubMed.
- Z. Y. Tian, M. Yousuf, Z. M. Wang, R. Jabeen, A. E. Mansy, S. Daniel and R. Kumar, in Environmental Chemistry: Advanced Concepts and Applications, ed. Z. Y. Tian, Springer Nature, Singapore, 2025, pp. 1–14 Search PubMed.
- D. Li, C. Liu, S. Tao, J. Cai, B. Zhong, J. Li, W. Deng, H. Hou, G. Zou and X. Ji, Nanomicro Lett., 2024, 17, 22 Search PubMed.
- X. Yan, Y. Zhou and S. Wang, Adv. Funct. Mater., 2025, 35, 2413115 CrossRef CAS.
- J. T. Ren, L. Chen, H. Y. Wang and Z. Y. Yuan, Chem. Soc. Rev., 2023, 52, 8319–8373 RSC.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Adv. Mater., 2011, 23, 4248–4253 CrossRef CAS PubMed.
- W. Ma, Z. Qiu, M. Wang, C. Tan, L. Hu, X. Lv, Q. Li, J. Li and J. Dang, Scr. Mater., 2023, 235, 115596 CrossRef CAS.
- Q. Liang, S. Wang, X. Lu, X. Jia, J. Yang, F. Liang, Q. Xie, C. Yang, J. Qian, H. Song and R. Chen, ACS Nano, 2024, 18, 2395–2408 CrossRef CAS PubMed.
- W. Song, D. Wang, S. Xiao, X. He, W. Xiong and J. Shen, Mater. Des., 2024, 240, 112857 CrossRef CAS.
- Z. Li, Y. Xiao, P. R. Chowdhury, Z. Wu, T. Ma, J. Z. Chen, G. Wan, T. H. Kim, D. Jing, P. He, P. J. Potdar, L. Zhou, Z. Zeng, X. Ruan, J. T. Miller, J. P. Greeley, Y. Wu and A. Varma, Nat. Catal., 2021, 4, 882–891 CrossRef CAS.
- M. H. Ghoncheh, Z. W. Chen, P. G. Demingos and C. V. Singh, iScience, 2025, 28, 113436 CrossRef CAS PubMed.
- X. Li, H. He, Y. Yu, Z. Wang, R. Zheng, H. Sun, Y. Liu and D. Wang, Appl. Surf. Sci., 2024, 645, 158838 CrossRef CAS.
- D. Singh, S. Razzaq, E. Tayyebi and K. S. Exner, ACS Catal., 2025, 15, 5589–5600 CrossRef CAS.
- B. Anasori and Y. Gogotsi, Graphene 2D Mater., 2022, 7, 75–79 CrossRef.
- S. K. Nemani, B. Zhang, B. C. Wyatt, Z. D. Hood, S. Manna, R. Khaledialidusti, W. Hong, M. G. Sternberg, S. K. R. S. Sankaranarayanan and B. Anasori, ACS Nano, 2021, 15, 12815–12825 CrossRef CAS PubMed.
- J. W. Yeh, S. K. Chen, S. J. Lin, J. Y. Gan, T. S. Chin, T. T. Shun, C. H. Tsau and S. Y. Chang, Adv. Eng. Mater., 2004, 6, 299–303 CrossRef CAS.
- C. M. Rost, E. Sachet, T. Borman, A. Moballegh, E. C. Dickey, D. Hou, J. L. Jones, S. Curtarolo and J.-P. Maria, Nat. Commun., 2015, 6, 8485 CrossRef CAS PubMed.
- L. Zhang, J. Shi, K. Niu, P. Jia, Y. Gao and G. Gao, ACS Appl. Nano Mater., 2023, 6, 20812–20822 CrossRef CAS.
- K. Li, P. Hao, Q. Zhang, Y. Wang, J. Zhang, S. Dmytro and Y. Zhou, Mater. Today Commun., 2024, 38, 108255 CrossRef CAS.
- L. Zhang, H. Li, X. Zhang, C. Liu, Y. Sun, Y. Zhang, Z. Fang, J. He, R. Wang, K. Jiang and D. Chen, Angew. Chem., Int. Ed., 2025, 64, e202418538 CrossRef CAS PubMed.
- W. Xu, H. Chen, K. Jie, Z. Yang, T. Li and S. Dai, Angew. Chem., Int. Ed., 2019, 58, 5018–5022 CrossRef CAS.
- Z. Du, C. Wu, Y. Chen, Z. Cao, R. Hu, Y. Zhang, J. Gu, Y. Cui, H. Chen, Y. Shi, J. Shang, B. Li and S. Yang, Adv. Mater., 2021, 33, 2101473 CrossRef CAS PubMed.
- C. E. Park, R. A. Senthil, G. H. Jeong and M. Y. Choi, Small, 2023, 19, 2207820 CrossRef CAS PubMed.
- W. Ma, Z. Qiu, J. Li, L. Hu, Q. Li, X. Lv and J. Dang, J. Energy Chem., 2023, 85, 301–309 CrossRef CAS.
- T. A. A. Batchelor, T. Löffler, B. Xiao, O. A. Krysiak, V. Strotkötter, J. K. Pedersen, C. M. Clausen, A. Savan, Y. Li, W. Schuhmann, J. Rossmeisl and A. Ludwig, Angew. Chem., Int. Ed., 2021, 60, 6932–6937 CrossRef CAS PubMed.
- L. Li, Z. Gao, W. Kang, Y. Feng, M. Hou, P. Yin, H. Liu and Z. Zhang, Sci. China Mater., 2025, 1–9 Search PubMed.
- Y. Cui, Y. Zhang, Z. Cao, J. Gu, Z. Du, B. Li and S. Yang, SusMat, 2022, 2, 65–75 CrossRef CAS.
- S. K. Nemani, M. Torkamanzadeh, B. C. Wyatt, V. Presser and B. Anasori, Commun. Mater., 2023, 4, 16 CrossRef CAS.
- Y. Sun and S. Dai, Sci. Adv., 2021, 7, eabg1600 CrossRef CAS PubMed.
- X. Zhao, Y. Chen, M. Feng, C. Xu, J. Du, X. Wang and Z. Liu, J. Alloys Compd., 2024, 992, 174586 CrossRef CAS.
- F. Yu, Y. Wang, K. Zhang, H. Shen, Y. Zhang, Z. Yang, G. Zeng, D. Cui, J. Xia, J. Liu, J. Li, M. Yang, H. Liu, C. Guo, W. Bao, K. Sun and J. Li, J. Mater. Chem. A, 2025, 13, 28699–28717 RSC.
- J. Zhou, Q. Tao, B. Ahmed, J. Palisaitis, I. Persson, J. Halim, M. W. Barsoum, P. O. Å. Persson and J. Rosen, Chem. Mater., 2022, 34, 2098–2106 CrossRef CAS.
- A. S. Etman, J. Zhou and J. Rosen, Electrochem. Commun., 2022, 137, 107264 CrossRef CAS.
- L. Gao, C. Li, W. Huang, S. Mei, H. Lin, Q. Ou, Y. Zhang, J. Guo, F. Zhang, S. Xu and H. Zhang, Chem. Mater., 2020, 32, 1703–1747 CrossRef CAS.
- C. Tsounis, P. V. Kumar, H. Masood, R. P. Kulkarni, G. S. Gautam, C. R. Müller, R. Amal and D. A. Kuznetsov, Angew. Chem., Int. Ed., 2023, 62, e202210828 CrossRef CAS PubMed.
- L. Qiao, J. Bi, G. Liang, Y. Yang, H. Wang and S. Wang, J. Adv. Ceram., 2023, 12, 1902–1918 CrossRef CAS.
- Z. Leong, H. Jin, Z. M. Wong, K. Nemani, B. Anasori and T. L. Tan, Chem. Mater., 2022, 34, 9062–9071 CrossRef CAS.
- M. Xu, Q. Zhu, Y. Li, Y. Gao, N. Sun and B. Xu, Energy Environ. Sci., 2024, 17, 7735–7748 RSC.
- W. Huang, K. Shen, C. Liu, C. Xu and J. Cheng, ACS Appl. Nano Mater., 2025, 9, 704–711 CrossRef.
- L. Chen, Y. Li, K. Liang, K. Chen, M. Li, S. Du, Z. Chai, M. Naguib and Q. Huang, Small Methods, 2023, 7, 2300054 CrossRef CAS PubMed.
- J. Zhou, M. Dahlqvist, J. Björk and J. Rosen, Chem. Rev., 2023, 123, 13291–13322 CrossRef CAS PubMed.
- M. Yousuf, M. F. Arshad and Z. Tian, Int. J. Chem. Kinet., 2024, 56, 310–322 CrossRef CAS.
- A. Zhou, Y. Liu, S. Li, X. Wang, G. Ying, Q. Xia and P. Zhang, J. Adv. Ceram., 2021, 10, 1194–1242 CrossRef CAS.
- A. H. Lashkari, A. Ostovari Moghaddam, M. Naseri and A. Shokuhfar, J. Mater. Res. Technol., 2023, 24, 5024–5031 CrossRef CAS.
- Q. Tao, J. Lu, M. Dahlqvist, A. Mockute, S. Calder, A. Petruhins, R. Meshkian, O. Rivin, D. Potashnikov, E. N. Caspi, H. Shaked, A. Hoser, C. Opagiste, R. M. Galera, R. Salikhov, U. Wiedwald, C. Ritter, A. R. Wildes, B. Johansson, L. Hultman, M. Farle, M. W. Barsoum and J. Rosen, Chem. Mater., 2019, 31, 2476–2485 CrossRef CAS.
- L. Wang, H. Zhang, B. Wang, C. Shen, C. Zhang, Q. Hu, A. Zhou and B. Liu, Electron. Mater. Lett., 2016, 12, 702–710 CrossRef CAS.
- J. Halim, M. R. Lukatskaya, K. M. Cook, J. Lu, C. R. Smith, L. Å. Näslund, S. J. May, L. Hultman, Y. Gogotsi, P. Eklund and M. W. Barsoum, Chem. Mater., 2014, 26, 2374–2381 CrossRef CAS PubMed.
- J. Yang, M. Naguib, M. Ghidiu, L. M. Pan, J. Gu, J. Nanda, J. Halim, Y. Gogotsi and M. W. Barsoum, J. Am. Ceram. Soc., 2016, 99, 660–666 Search PubMed.
- M. Alhabeb, K. Maleski, T. S. Mathis, A. Sarycheva, C. B. Hatter, S. Uzun, A. Levitt and Y. Gogotsi, Angew. Chem., Int. Ed., 2018, 57, 5444–5448 CrossRef CAS PubMed.
- F. Liu, J. Zhou, S. Wang, B. Wang, C. Shen, L. Wang, Q. Hu, Q. Huang and A. Zhou, J. Electrochem. Soc., 2017, 164, A709 Search PubMed.
- S. Kajiyama, L. Szabova, H. Iinuma, A. Sugahara, K. Gotoh, K. Sodeyama, Y. Tateyama, M. Okubo and A. Yamada, Adv. Energy Mater., 2017, 7, 1601873 CrossRef.
- F. Du, H. Tang, L. Pan, T. Zhang, H. Lu, J. Xiong, J. Yang and C. (John) Zhang, Electrochim. Acta, 2017, 235, 690–699 Search PubMed.
- A. Vahidmohammadi, A. Hadjikhani, S. Shahbazmohamadi and M. Beidaghi, ACS Nano, 2017, 11, 11135–11144 Search PubMed.
- J. Zhou, X. Zha, X. Zhou, F. Chen, G. Gao, S. Wang, C. Shen, T. Chen, C. Zhi, P. Eklund, S. Du, J. Xue, W. Shi, Z. Chai and Q. Huang, ACS Nano, 2017, 11, 3841–3850 CrossRef CAS PubMed.
- N. Chen, Z. Duan, W. Cai, Y. Wang, B. Pu, H. Huang, Y. Xie, Q. Tang, H. Zhang and W. Yang, Nano Energy, 2023, 107, 108147 CrossRef CAS.
- L. Liu, M. Orbay, S. Luo, S. Duluard, H. Shao, J. Harmel, P. Rozier, P. L. Taberna and P. Simon, ACS Nano, 2021, 16, 111–118 Search PubMed.
- A. Gentile, S. Marchionna, M. Balordi, G. Pagot, C. Ferrara, V. Di Noto and R. Ruffo, ChemElectroChem, 2022, 9, e202200891 Search PubMed.
- K. Arole, J. W. Blivin, A. M. Bruce, S. Athavale, I. J. Echols, H. Cao, Z. Tan, M. Radovic, J. L. Lutkenhaus and M. J. Green, Chem. Commun., 2022, 58, 10202–10205 Search PubMed.
- Y. Wang, L. Li, M. Shen, R. Tang, J. Zhou, L. Han, X. Zhang, L. Zhang, G. Kim and J. Q. Wang, Adv. Sci., 2023, 10, 2303693 Search PubMed.
- H. Xu, H. Shou, Z. Yan, K. Zhu, C. Wu, W. Jiang, Z. Liu, S. Wei, J. Shi, H. Akhtar, C. Wang and L. Song, Adv. Mater., 2025, 37, 2504586 Search PubMed.
- H. Kim, D. Gwon, D. Kim, J. Kee and J. Koo, Adv. Mater. Technol., 2024, 9, 2301437 Search PubMed.
- K. Montazeri, H. Badr, K. Ngo, K. Sudhakar, T. Elmelegy, J. Uzarski, V. Natu and M. W. Barsoum, J. Phys. Chem. C, 2023, 127, 10391–10397 CrossRef CAS.
- A. Thakur, N. Chandran B.S., K. Davidson, A. Bedford, H. Fang, Y. Im, V. Kanduri, B. C. Wyatt, S. K. Nemani, V. Poliukhova, R. Kumar, Z. Fakhraai and B. Anasori, Small Methods, 2023, 7, 2300030 Search PubMed.
- Y. N. Athreya, Effect of CTAB intercalation on electrochemical properties of Ti3C2Tx MXene, MS thesis, Drexel University, 2025 DOI:10.17918/00010916.
- S. Nouseen and M. Pumera, J. Mater. Chem. A, 2025, 13, 34055–34084 Search PubMed.
- Q. Fan, M. Chen, L. Li, M. Li, C. Xiao, T. Zhao, L. Pan, N. Liang, Q. Huang, L. Yu, L. Zhu, M. Naguib and K. Liang, Nat. Commun., 2025, 16, 5051 Search PubMed.
- A. Inman, K. Shevchuk, M. Anayee, W. Hammill, J. Lee, M. Saraf, C. E. Shuck, C. M. Armstrong, Y. He, T. Jin, M. Shekhirev, J. Capobianco and Y. Gogotsi, Chem. Eng. J., 2023, 475, 146089 Search PubMed.
- Z. Zhou, L. Wei, Y. Yi, S. Feng, Z. Zhan, D. Tian and C. Lu, J. Materiomics, 2023, 9, 1151–1159 Search PubMed.
- A. Inman, V. Šedajová, K. Matthews, J. Gravlin, J. Busa, C. E. Shuck, A. VahidMohammadi, A. Bakandritsos, M. Shekhirev, M. Otyepka and Y. Gogotsi, J. Mater. Res., 2022, 37, 4006–4016 CrossRef CAS.
- X. Sang, Y. Xie, M. W. Lin, M. Alhabeb, K. L. Van Aken, Y. Gogotsi, P. R. C. Kent, K. Xiao and R. R. Unocic, ACS Nano, 2016, 10, 9193–9200 Search PubMed.
- D. Yang, Y. Wang, X. Liu, Y. Gao, Y. Chen, G. Tian, S. Wang, L. Zhang and N. Tang, Electrochim. Acta, 2025, 536, 146759 Search PubMed.
- A. Kuzmin, in High-Entropy Alloys: Design, Manufacturing, and Emerging Applications, ed. G. Yasin, M. Abubaker Khan, M. Ahmed Afifi, T. A. Nguyen and Y. Zhang, Elsevier, 2024, pp. 121–155 Search PubMed.
- K. Li, P. Hao, K. Yuan, T. Yao, J. Zhang and Y. Zhou, Comput. Mater. Sci., 2023, 227, 112271 CrossRef CAS.
- Samia, M. H. Jameel, M. Arain, I. Hussain, M. B. Hanif, S. Atri, M. Z. H. Mayzan and H. Dai, Nano Mater. Sci., 2025, 7, 444–481 CrossRef CAS.
- M. Liu, L. Yang, Z. Wu, G. Chen, X. Wang, X. Yang, G. Liang and R. Che, Nat. Commun., 2025, 16, 5633 Search PubMed.
- R. B. Araujo and T. Edvinsson, J. Mater. Chem. A, 2023, 11, 12973–12983 Search PubMed.
- H.-B. Cui, Y. Wang, Y.-C. Wang, C. Chen, G. Guan and M.-Y. Han, ACS Nano, 2025, 19, 40641–40661 Search PubMed.
- B. Guo, Z. Zhou, W. Sun and X. Hu, iScience, 2025, 28, 113577 CrossRef CAS PubMed.
- P. Sharma, P. Singh and G. Balasubramanian, Carbon, 2024, 223, 119015 CrossRef CAS.
- H. Mei, Y. Zhang, P. Zhang, A. G. Ricciardulli, P. Samorì and S. Yang, Adv. Sci., 2024, 11, 2409404 Search PubMed.
- F. Gu, W. Wang, H. Meng, Y. Liu, L. Zhuang, H. Yu and Y. Chu, Matter, 2025, 8, 102004 Search PubMed.
- M. Tang, J. Li, Y. Wang, W. Han, S. Xu, M. Lu, W. Zhang and H. Li, Symmetry, 2022, 14, 2232 Search PubMed.
- E. Rems, Y. J. Hu, Y. Gogotsi and R. Dominko, Chem. Mater., 2024, 36, 10295–10306 Search PubMed.
- E. M. Ahmed, A. S. Ali, E. M. Hieba, Z. S. Shaban, M. S. Fathy, A. M. Amer, A. M. Ishmael, A. Bakr, H. R. M. Rashdan and A. Elzwawy, RSC Adv., 2025, 15, 44812–44863 Search PubMed.
- H. Li, Y. Chen and Q. Tang, ChemPhysChem, 2024, 25, e202400255 Search PubMed.
- L. Meng, E. Tayyebi, K. S. Exner, F. Viñes and F. Illas, ChemElectroChem, 2024, 11, e202300598 Search PubMed.
- Q. Wang, S. Qiao, C. Huang, X. Wang, C. Cai, G. He and F. Zhang, ACS Appl. Mater. Interfaces, 2024, 16, 24502–24513 Search PubMed.
- N. Li, S. Wei, Y. Xu, J. Liu, J. Wu, G. Jia and X. Cui, Electrochim. Acta, 2018, 290, 364–368 Search PubMed.
- B. Zhang, J. Shan, X. Wang, Y. Hu and Y. Li, Small, 2022, 18, 2200173 Search PubMed.
- M. Jussambayev, K. Shakenov, S. Sultakhan, U. Zhantikeyev, K. Askaruly, K. Toshtay and S. Azat, Carbon Trends, 2025, 19, 100471 Search PubMed.
- Q. Yang, S. J. Eder, A. Martini and P. G. Grützmacher, npj Mater. Degrad., 2023, 7, 6 Search PubMed.
- B. C. Wyatt, A. Rosenkranz and B. Anasori, Adv. Mater., 2021, 33, 2007973 CrossRef CAS PubMed.
- J. Liu, Y. Xiang, S. Fang, Z. Du, Z. Li, L. Gao, F. Fu, L. Lv, X. Gao, J. Zhou, D. Wu and X. Jian, ACS Appl. Mater. Interfaces, 2024, 16, 70508–70519 Search PubMed.
- Y. Zhao, D. P. Adiyeri Saseendran, C. Huang, C. A. Triana, W. R. Marks, H. Chen, H. Zhao and G. R. Patzke, Chem. Rev., 2023, 123, 6257–6358 CrossRef CAS PubMed.
- L. He, Q. Tang, Q. Fan, H. Zhuang, S. Wang, Y. Pang and K. Liang, Catalysts, 2024, 14, 708 Search PubMed.
- Y. Chen, H. Yao, F. Kong, H. Tian, G. Meng, S. Wang, X. Mao, X. Cui, X. Hou and J. Shi, Appl. Catal., B, 2021, 297, 120474 CrossRef CAS.
- A. Liu, X. Liang, X. Ren, W. Guan and T. Ma, Electrochem. Energy Rev., 2021, 5, 112–144 Search PubMed.
- P. V. Hlophe, H. Zhao, G. Li, K. N. Nigussa, S. Song and P. F. Msomi, Carbon Trends, 2025, 21, 100594 CrossRef CAS.
- Y. Sun, Z. Wang, Q. Zhou, X. Li, D. Zhao, B. Ding and S. Wang, Heliyon, 2024, 10, e30966 Search PubMed.
- L. Tan, J. Wang, S. Zhou, H. Zhu, J. Guo, Y. Chen, X. Li, Z. Dong, Q. Zhang and Y. Cong, J. Colloid Interface Sci., 2025, 689, 137263 Search PubMed.
- M. Xia, M. Yang, Y. Guo, X. Sun, S. Wang, Y. Feng and H. Ding, ChemNanoMat, 2022, 8, e202200113 Search PubMed.
- W. Huang, J. Zhang, G. Deng, G. Zhu, Y. Chen, C. Xu and J. Cheng, ACS Appl. Energy Mater., 2025, 8, 2612–2619 Search PubMed.
- S. Cui, X. Zhong, Z. Li, P. Zhu, J. Hu, X. Zhou, P. Kidkhunthod, X. Wang, B. Mei and B. Xu, Adv. Mater., 2025, 37, 2504099 Search PubMed.
- C. Y. J. Lim, A. D. Handoko and Z. W. Seh, Diamond Relat. Mater., 2022, 130, 109461 Search PubMed.
- S. Y. Hsu, C. C. Chi, M. Y. Lu, S. Y. Chang, Y. T. Lai, S. Y. Tsai and J. G. Duh, J. Alloys Compd., 2023, 947, 169645 Search PubMed.
- S. A. Talas, P. D. Kolubah, R. Khairova, M. Alqahtani, S. I. El-Hout, F. M. Alissa, J. K. El-Demellawi, P. Castaño and H. O. Mohamed, Mater. Horiz., 2025, 12, 7648–7682 RSC.
- T. Amrillah, A. R. Supandi, V. Puspasari, A. Hermawan and Z. W. Seh, Trans. Tianjin Univ., 2022, 28, 307–322 CrossRef CAS.
- M. Ozkan, K. A. M. Quiros, J. M. Watkins, T. M. Nelson, N. D. Singh, M. Chowdhury, T. Namboodiri, K. R. Talluri and E. Yuan, Chem, 2024, 10, 443–483 CAS.
- R. Morales-Salvador, J. D. Gouveia, Á. Morales-García, F. Viñes, J. R. B. Gomes and F. Illas, ACS Catal., 2021, 11, 11248–11255 CrossRef CAS.
- J. K. Pedersen, T. A. A. Batchelor, A. Bagger and J. Rossmeisl, ACS Catal., 2020, 10, 2169–2176 Search PubMed.
- Y. Sun, R. Yu, J. Sun, D. Legut, J. S. Francisco and R. Zhang, J. Mater. Chem. A, 2025, 13, 11703–11716 Search PubMed.
- S. Cao, Y. Liu, Y. Hu, J. Li, C. Yang, Z. Chen, Z. Wang, S. Wei, S. Liu and X. Lu, J. Colloid Interface Sci., 2023, 642, 273–282 Search PubMed.
- X. Liu, L. Yao, S. Zhang, C. Huang and W. Yang, Inorg. Chem., 2024, 63, 6305–6314 CrossRef CAS PubMed.
- G. Sun, Y. Zhang, H. Xie, Y. He, C. Zhang, Y. Hu, E. Liu, C. He and A. B. Wong, Nano Energy, 2024, 132, 110385 Search PubMed.
- A. Athawale, B. M. Abraham, M. V. Jyothirmai and J. K. Singh, J. Phys. Chem. C, 2023, 127, 24542–24551 Search PubMed.
- Y. Luo, G. F. Chen, L. Ding, X. Chen, L. X. Ding and H. Wang, Joule, 2019, 3, 279–289 Search PubMed.
- Y. Guo, T. Wang, Q. Yang, X. Li, H. Li, Y. Wang, T. Jiao, Z. Huang, B. Dong, W. Zhang, J. Fan and C. Zhi, ACS Nano, 2020, 14, 9089–9097 Search PubMed.
- L. Zhou, D. Feng, Z. Li, Z. Lv, H. Li, C. Ge, X. Zhang and T. Ma, ACS Appl. Mater. Interfaces, 2025, 17, 38050–38061 Search PubMed.
- Y. Lei, L. Zhang, X. Wang, D. Wang, Y. Zhao, B. Zhang, N. Zhang and H. Shang, Chem. Sci., 2025, 16, 18298–18308 RSC.
- K. Chu, J. Qin, H. Zhu, M. De Ras, C. Wang, L. Xiong, L. Zhang, N. Zhang, J. A. Martens, J. Hofkens, F. Lai and T. Liu, Sci. China Mater., 2022, 65, 2711–2720 Search PubMed.
- Q. Zou, A. Qiu, Y. He, E. Y. Xue, L. Wang, G. Yang, Y. Shen, D. Luo, Q. Liu and D. K. P. Ng, Adv. Sci., 2025, 12, 2410537 Search PubMed.
- M. Naguib, R. R. Unocic, B. L. Armstrong and J. Nanda, Dalton Trans., 2015, 44, 9353–9358 Search PubMed.
- C. Xu, L. Wang, Z. Liu, L. Chen, J. Guo, N. Kang, X. L. Ma, H. M. Cheng and W. Ren, Nat. Mater., 2015, 14, 1135–1141 Search PubMed.
- M. Ghidiu, M. R. Lukatskaya, M. Q. Zhao, Y. Gogotsi and M. W. Barsoum, Nature, 2014, 516, 78–81 Search PubMed.
- D. B. Velusamy, J. K. El-Demellawi, A. M. El-Zohry, A. Giugni, S. Lopatin, M. N. Hedhili, A. E. Mansour, E. Di Fabrizio, O. F. Mohammed and H. N. Alshareef, Adv. Mater., 2019, 31, 1807658 Search PubMed.
- M. R. Lukatskaya, S. Kota, Z. Lin, M. Q. Zhao, N. Shpigel, M. D. Levi, J. Halim, P. L. Taberna, M. W. Barsoum, P. Simon and Y. Gogotsi, Nat. Energy, 2017, 2, 17105 Search PubMed.
- Y. Li, H. Shao, Z. Lin, J. Lu, L. Liu, B. Duployer, P. O. Å. Persson, P. Eklund, L. Hultman, M. Li, K. Chen, X. H. Zha, S. Du, P. Rozier, Z. Chai, E. Raymundo-Piñero, P. L. Taberna, P. Simon and Q. Huang, Nat. Mater., 2020, 19, 894–899 Search PubMed.
- C. Xu, L. Chen, Z. Liu, H. M. Cheng and W. Ren, in 2D Metal Carbides and Nitrides (MXenes), ed. B. Anasori and Y. Gogotsi, Springer, Cham, 2019, pp. 89–109 Search PubMed.
- Y. Yan, X. Huang, X. Yan, F. Zhang, J. Pan, J. Wu and J. M. Moradian, Adv. Funct. Mater., 2025, 35, 2420578 Search PubMed.
- F. Yang, K. L. Chan, Z. Wu, F. Zhao, M. C. Wong, S. Y. Pang and J. Hao, Sci. China Mater., 2025, 68, 2860–2867 Search PubMed.
- X. Gao, S. Zhang, J. Zhang, S. Wei, Y. Chen, Y. Zhang, W. Zheng, Z. Huang, B. Du, Z. Xie, B. Wang, J. Lin, J. Liu and Y. Ge, Laser Photonics Rev., 2024, 18, 2400082 Search PubMed.
- H. Zou, W. Zhang, F. Chen, C. Zhang, J. Zhang, L. Ren, W. Wang, F. Zhang and Z. Fu, J. Adv. Ceram., 2026, 15, 9221235 Search PubMed.
- Y. Wu, C. Chen, F. Pan, X. Li and W. Lu, Chem. Eng. J., 2024, 499, 156024 Search PubMed.
- W. Ma, M. Wang, Q. Yi, D. Huang, J. Dang, Z. Lv, X. Lv and S. Zhang, Nano Energy, 2022, 96, 107129 Search PubMed.
- R. Iftikhar, Md. I. Kholil, S. Luo, M. A. Khan, M. Akter and A. Sinitskii, Mater. Today Commun., 2026, 51, 114598 Search PubMed.
- P. Urbankowski, B. Anasori, T. Makaryan, D. Er, S. Kota, P. L. Walsh, M. Zhao, V. B. Shenoy, M. W. Barsoum and Y. Gogotsi, Nanoscale, 2016, 8, 11385–11391 Search PubMed.
- R. Meshkian, L.-Å. Näslund, J. Halim, J. Lu, M. W. Barsoum and J. Rosen, Scr. Mater., 2015, 108, 147–150 Search PubMed.
- B. Wei, Z. Fu, D. Legut, T. C. Germann, S. Du, H. Zhang, J. S. Francisco and R. Zhang, Adv. Mater., 2021, 33, 2102595 Search PubMed.
- P. Dadvari, W. H. Hung and K. W. Wang, ACS Omega, 2024, 9, 27692–27698 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |