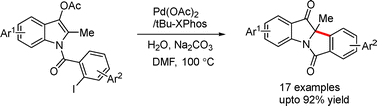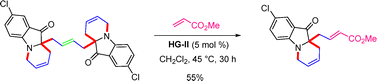Accessing the less traveled path: regioselective synthesis of 3-oxindoles
Tirtha
Mandal†
 ,
Gargi
Chakraborti†
,
Gargi
Chakraborti†
 ,
Nabin
Parui†
,
Nabin
Parui†
 and
Jyotirmayee
Dash
and
Jyotirmayee
Dash
 *
*
School of Chemical Sciences, Indian Association for the Cultivation of Science, Jadavpur, Kolkata, 700032, India. E-mail: ocjd@iacs.res.in
First published on 25th March 2026
Abstract
Indoxyls, commonly known as 3-oxindoles, are privileged heterocyclic scaffolds found in natural products, bioactive molecules, and functional materials. Despite their close structural relationship to 2-oxindoles, selective access to 3-oxindoles, particularly 2-functionalized variants, remains significantly more challenging, making regioselective control a central synthetic issue. While methods for constructing 3-functionalized 2-oxindoles are well established, the synthesis of indoxyl frameworks has emerged as a rapidly expanding and conceptually distinct area of research. This review traces the advances made in the twenty-first century in the construction of 3-oxindole frameworks, emphasizing regioselective and asymmetric strategies. Developments in metal catalysis, organocatalysis, photocatalysis, electrocatalysis, and hypervalent iodine chemistry are discussed, showcasing approaches to C2-functionalization, spirocyclization, and dimerization. Collectively, these advances demonstrate the growing potential of indoxyl chemistry to expand chemical space and inspire future developments in organic synthesis and total synthesis.
1. Introduction
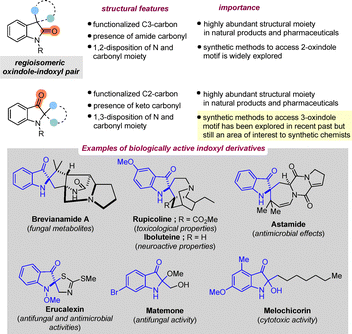
Regioselective transformations are fundamental to organic synthesis, enabling functionalization at specific positions in a molecule. However, controlling site-selectivity in chemical reactions remains challenging, particularly when the desired regioisomer is less accessible. The pursuit of such inaccessible regioisomers continues to inspire the development of new synthetic strategies to explore new chemical space.The oxindole–indoxyl pair represents a classic example of such regioisomeric systems (Scheme 1). While a wide array of methods have been established for the synthesis of functionalized 2-oxindole derivatives,1–3 the preparation of the regioisomeric 3-oxindoles has remained comparatively underexplored,4–6 presumably due to the inherent synthetic challenges associated with their synthesis. Nevertheless, the indoxyl or pseudooxindole core represents an important structural motif found in numerous natural products and pharmaceutically relevant compounds. Representative examples include brevianamides A and B (natural fungal metabolites),7 rupicoline (toxicological relevance),8 iboluteine (neuroactive properties),9 austamide (antimicrobial/antiparasitic activity),10 the phytoalexin erucalexin (antifungal and antimicrobial activities),11 matemone (antifungal activity),12 mitragynine pseudoindoxyl (analgesic activity),13 duocarmycin (anticancer activity),14 and melochicorin (cytotoxic activity).15 These examples highlight the significance of the indoxyl scaffold, making it an attractive target for synthetic method development (Scheme 1).
Despite this significance, enantioselective approaches to indoxyl derivatives bearing a chiral centre at the 2-position remain surprisingly rare and are less explored. Since 2010, considerable efforts have been devoted to the development of new synthetic protocols for accessing diverse classes of indoxyl derivatives. Recent breakthroughs in this field include (1) metal catalyzed transformations, (2) hypervalent iodine mediated reactions and (3) organocatalytic strategies. These methodologies have enabled access to structurally complex spirocyclic, fused, and acyclic unnatural 3-oxindoles bearing stereogenic centers. In addition, the indoxyl motif has been exploited as a versatile intermediate for the synthesis of other indole and oxindole derivatives. This vast build-up of molecular complexity establishes the indoxyl scaffold as a powerful platform for natural product synthesis, while also highlighting the feasibility of addressing highly congested C2 stereocenters through asymmetric synthesis. This review aims to summarize recent developments in the construction of the pseudooxindole core and to stimulate further applications of this versatile synthetic framework.
2. Synthesis of indoxyl derivatives
2.1 Synthesis of acyclic indoxyls
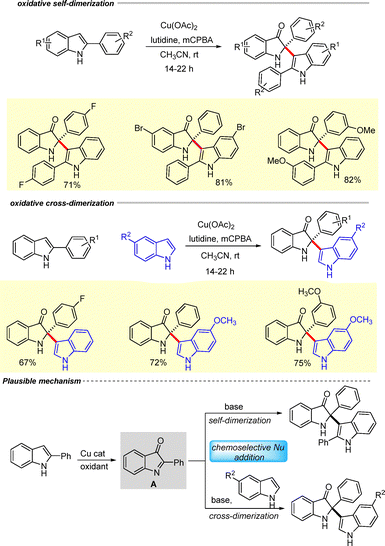
Kumar and co-workers developed an efficient protocol for the synthesis of 2,2-disubstituted indolin-3-ones under mild conditions.17 The reaction proceeds via copper-catalyzed in situ oxidative dearomatization of 2-arylindoles to indol-3-ones, which undergo chemoselective nucleophilic addition in the presence of a base, leading to either self-dimerization or cross-addition with indoles (Scheme 3). The cross-dimerization displays a broad substrate scope with respect to substituents on both aryl rings and affords the corresponding 2-indolyl-substituted 3-oxindoles in good yields under the optimized conditions [Cu(OAc)2 (30 mol%), 2,6-lutidine (2 equiv.), and m-CPBA (30 mol%)]. Furthermore, the methodology was extended to cross-addition of indoles at the C2-position of 2-arylindoles, affording the desired products in good yields.
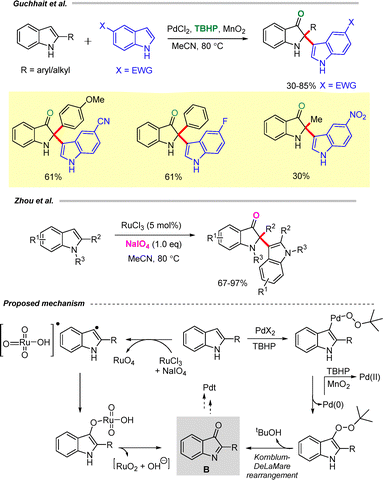
Guchhait et al. reported oxidative dearomatization of indoles via Pd-catalyzed C–H oxygenation to access indolin-3-ones (Scheme 4).18 This method enables the synthesis of indolin-3-ones bearing C2-quaternary centers, including chiral indoxyl motifs that are prevalent in indole alkaloids. A broad range of 2-arylindoles as well as C2-unsubstituted indoles were successfully employed, and the reaction was shown to tolerate both electron-withdrawing and electron-donating substituents on the indole ring.18
Later, Zhou and co-workers reported a Ru catalyzed synthesis of disubstituted indoline-3-ones via oxidative dearomatization of indoles using sodium metaperiodate (Scheme 4).19
Both transformations proceed through the formation of a common intermediate B, which undergoes chemoselective C3-alkylation of the indole to deliver the desired indoxyl derivatives. In the Pd-catalyzed pathway, the reaction is initiated by electrophilic palladation of the indole, followed by oxidation with TBHP and subsequent reductive elimination to generate an indole-3-tert-butyl peroxide intermediate. This intermediate undergoes a Kornblum–DeLaMare rearrangement to furnish intermediate B. The catalytic cycle is completed by reoxidation of Pd(0) to Pd(II) using the TBHP/MnO2 system (Scheme 4).18 In contrast, Zhou et al. proposed that the active RuO4 species, generated in situ from RuCl4 and NaIO4, reacts with indole to form a radical pair, which leads to the formation of an oxidized indole complex. A subsequent electron-transfer process generates [RuO2 + HO−] along with the indolinium intermediate B. The active RuO4 catalyst is regenerated in the presence of NaIO4, sustaining the catalytic cycle (Scheme 4).19
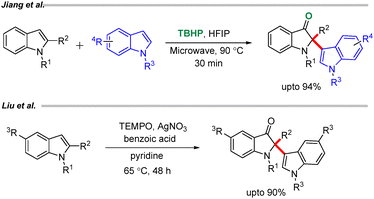
A metal free approach to access the C2-indolyl 2,2-disubstituted indoline 3-ones was reported by Jiang et al. using TBHP and HFIP under microwave irradiation (Scheme 5).20 The reaction exhibits a broad substrate scope, is scalable, and proceeds via a radical pathway. In a complementary approach, Lin et al. described a metal-free oxidative homodimerization of indole derivatives employing TEMPO and AgNO3, which afforded the corresponding products efficiently (Scheme 5).21 Both reactions proceed through the formation of a common intermediate B (Scheme 4) generated via oxidation of the indole nucleus, mediated by TBHP/HFIP in the former case and TEMPO/AgNO3 in the latter. This intermediate subsequently undergoes chemoselective nucleophilic addition with another indole molecule, leading to the formation of the desired C2-functionalized indolin-3-one derivatives.
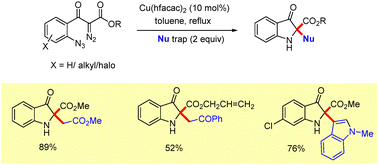
The West group reported a copper catalyzed domino azide coupling/nucleophilic trapping strategy for the synthesis of 2,2-disubstituted 3-oxindoles.22 Treatment of doubly stabilized diazo substrates with Cu(hfacac)2 in toluene under reflux, in the presence of silyl ketene acetals, furnished the desired oxindole derivatives in good yields (Scheme 6). Mechanistically, the reaction proceeds via the formation of an intermediate B (Scheme 4), which undergoes subsequent nucleophilic trapping to deliver the final products.
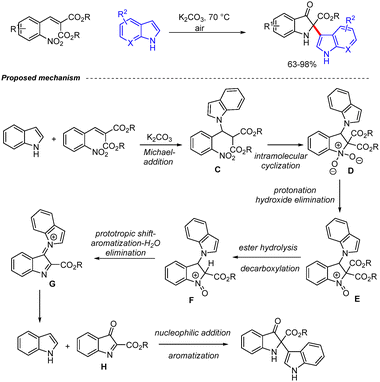
In 2025, Saikia and co-workers reported a base-promoted, mild synthetic route for the construction of pseudoindoxyl derivatives from 2-nitrobenzylidenemalonates and indoles (Scheme 7).23 The transformation is initiated by a Michael addition, furnishing the isolable intermediate C, which undergoes base-mediated intramolecular cyclization to generate intermediate D. Subsequent protonation of D, followed by elimination of hydroxide, affords intermediate E. Hydroxide-assisted ester hydrolysis and decarboxylation lead to intermediate F. A sequence involving prototropic shift, aromatization, and water elimination generates intermediate G, which upon hydrolysis releases the parent indole along with the key indoxyl intermediate H. Finally, chemoselective nucleophilic addition of indole to intermediate H provides the desired indoxyl derivatives (Scheme 7).23
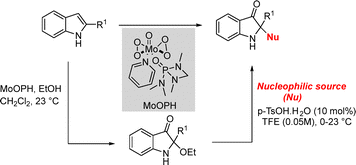
In 2021, the Smith group reported an oxodiperoxymolybdenum(pyridine)(hexamethylphosphoric triamide (MoOPH)-mediated general strategy for accessing 2,2-disubstituted indoxyls from 2-substituted indoles via nucleophile coupling with a 2-alkoxyindoxyl intermediate.24 The method exhibits broad functional-group tolerance with respect to both the nucleophile and indole substitution, tolerating 2-alkyl substituents as well as free indole N–H groups. The strategy was successfully extended to the total synthesis of the alkaloids brevianamide A and trigonoliimine C (Scheme 8).24
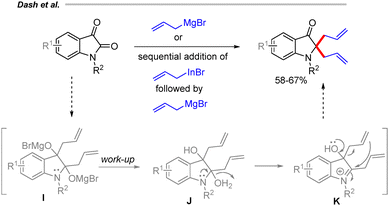
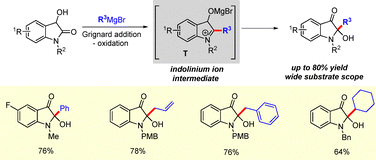
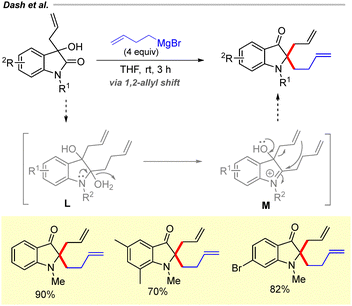
In 2015, our group developed a method for the synthesis of 2,2-diallyl oxindole derivatives via allyl Grignard addition to isatin derivatives (Scheme 9).25 The reaction is proposed to proceed through the formation of a diallylmagnesium bromide intermediate I. Upon completion, acidic work-up generates intermediate J, which facilitates the formation of an indolinium ion intermediate K. A subsequent 1,2-allylic shift leads to the formation of the desired diallyl oxindoles.25
Recently, in 2023, we demonstrated that this strategy could be extended to the synthesis of 2-allyl-2-(but-3-enyl)-3-oxindole derivatives following butenylmagnesium bromide addition to C3-allyl 2-oxindole derivatives (Scheme 10).26 Mechanistically, the reaction proceeds in a similar manner to that depicted in Scheme 9, involving the formation of an indolinium ion intermediate M.
 | ||
| Scheme 10 Synthesis of 2-allyl-2-(but-3-enyl)-3-oxindole derivatives using 3-butenylmagnesium bromide Grignard reaction. | ||
Moreover, we explored the proposed 1,2-allyl shift through the synthesis of C2-allyl-2,2-disubstituted 3-oxindoles.27 Addition of allylmagnesium bromide to C2-functionalized 2-hydroxyindoxyls generated the corresponding 2,2-disubstituted oxindoles in good yields via migration of the C3-allyl group to the C2 position (Scheme 11). The reaction follows the same pathway as described previously in Schemes 9 and 10via the formation of indolinium ion intermediate O.
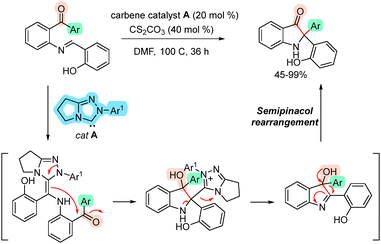
An elegant NHC-catalyzed method for the synthesis of biaryl indoxyls was reported in 2024 by the Biju group.28 Mechanistic investigations, supported by DFT studies, indicated that the reaction proceeds via a rearomative proton transfer coupled with aryl migration through an ortho-quinone methide intermediate, thereby facilitating a semipinacol rearrangement (Scheme 12).28
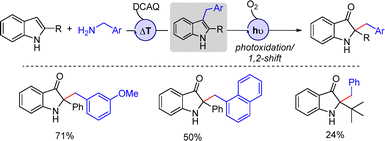
A sequential one-pot anthraquinone-catalyzed thermal C3-benzylation of indoles with benzylamines was reported in 2014 by Lerch and co-workers, furnishing 3-benzylindole intermediates that subsequently underwent visible-light-mediated photooxidation followed by a 1,2-shift to afford 2,2-disubstituted indolin-3-ones in good yields (Scheme 13).29
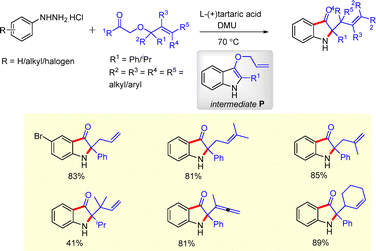
In 2017, Xie and co-workers developed a tandem Fischer cyclization–Claisen rearrangement strategy for the synthesis of 2,2-disubstituted indoline-3-ones (Scheme 14).30 Reaction of phenylhydrazine with allyloxyketones in the presence of L-(+)-tartaric acid and dimethylurea led to the in situ formation of intermediate P, which underwent a spontaneous Claisen rearrangement to furnish the corresponding oxindole derivatives.
 | ||
| Scheme 14 Synthesis of disubstituted oxindoles via a Fischer cyclization–Claisen rearrangement strategy. | ||
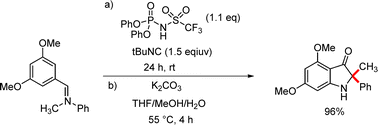
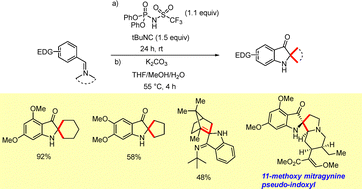
An interrupted Ugi reaction was utilized for the synthesis of disubstituted 3-indoxyls.31 The mild reaction conditions represent a notable advantage over the classical Houben–Hoesch reaction. Although the electron-rich, sterically hindered imine intermediates initially proved resistant to acidic hydrolysis, the key hydrolysis step was accomplished smoothly by treatment with K2CO3 in a warm THF/MeOH/H2O mixture (Scheme 15).31
Chiral indolin-3-ones constitute important structural motifs found in numerous natural products and pharmaceutical agents. However, in comparison to indoline-2-one derivatives, the enantioselective synthesis of indoxyls bearing a chiral center at the C2 position remains less developed.
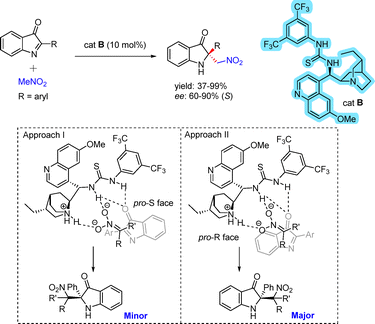
An aza-Henry reaction between nitroalkanes and 2-aryl-3H-indoline-3-ones under thiourea–cinchona catalysis afforded chiral 2,2-disubstituted indolin-3-ones in moderate to good yields and enantiomeric excess (Scheme 16).32 The stereochemical outcome was attributed to a dual hydrogen-bonding interaction between the thiourea moiety and the carbonyl group. Simultaneously, the nitronate species, generated by deprotonation of nitromethane at the quinuclidine core, remained proximal to its site of formation. This spatial arrangement favored approach of the nitronate toward the oxindole framework and the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond rather than the C
N bond rather than the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group, thereby providing a rationale for both the regio- and stereochemical outcomes. The observed enantioselectivity of the major isomer can be attributed to more favorable stereoelectronic interactions between the nitronate and the indoline, as depicted in approach II, where nucleophilic attack occurs on the pro-R face of the substrate.
O group, thereby providing a rationale for both the regio- and stereochemical outcomes. The observed enantioselectivity of the major isomer can be attributed to more favorable stereoelectronic interactions between the nitronate and the indoline, as depicted in approach II, where nucleophilic attack occurs on the pro-R face of the substrate.
 | ||
| Scheme 16 Asymmetric synthesis of disubstituted 3-oxindoles using a thiourea–cinchona catalyst system. | ||
A proline-catalyzed Mannich reaction of 3H-indol-3-ones with aldehydes to afford chiral indoline-3-ones in excellent enantiomeric excess was reported by Rueping and co-workers (Scheme 17).33 However, a considerable decrease in enantioselectivity was observed when branched-chain aldehydes were employed (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 56% ee).33
1 dr, 56% ee).33
Xu and co-workers reported an asymmetric Michael addition of activated indolin-3-ones to nitroalkenes for the synthesis of enantioenriched 2,2-disubstituted indolin-3-ones in good to excellent yields and high enantiomeric excess (Scheme 18).34 A ternary transition state was proposed, in which hydrogen bonding between the thiourea moiety and the nitro group of the nitroalkene operates in concert with proton abstraction at the C2 position by the tertiary quinuclidine nitrogen, generating a tight ion pair. In this arrangement, Si-face attack at the nitroalkene is sterically favored.
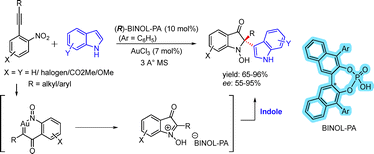
Dual catalysis employing gold and chiral phosphoric acids was demonstrated through an enantioselective redox annulation of nitroalkynes with indoles, enabling the synthesis of 2,2-disubstituted indolin-3-ones (Scheme 19).35 The reaction could be conducted in both cascade and stepwise modes without significant loss of yield or enantioselectivity.
 | ||
| Scheme 19 Enantioselective annulation of nitroalkynes with indoles for the synthesis of 2,2-disubsituted indolin-3-ones. | ||
Li and co-workers developed the first organocatalytic asymmetric α-sulfenylation of 2-substituted indolin-3-ones with N-(alkylthio or arylthio) succinimides using a Cinchona-derived squaramide catalyst.36 A variety of chiral 2,2-disubstituted indolin-3-ones bearing S- and N-containing heteroquaternary carbon stereocenters were obtained in excellent yields and enantioselectivities (Scheme 20). The bifunctional catalyst activates indolin-3-one to generate a nucleophilic enolate species, while simultaneously enhancing the electrophilicity of the succinimide through hydrogen-bonding interactions with the squaramide moiety. This cooperative activation enables enantioselective sulfenylation, proceeding via Re-face attack to furnish the desired product.
A chiral phosphoric acid-catalyzed asymmetric synthesis of arylindolyl indolin-3-ones bearing both axial and central chirality was achieved via the reaction of 3-arylindoles with 2-aryl-3H-indol-3-ones, providing the desired products in high yields with excellent enantioselectivity and diastereoselectivity (Scheme 21), as reported by Yuan et al.37 The (R)-CPA catalyst and the substrates form a well-organized three-component complex through multiple hydrogen-bonding interactions, which governs the stereochemical outcome of the reaction.
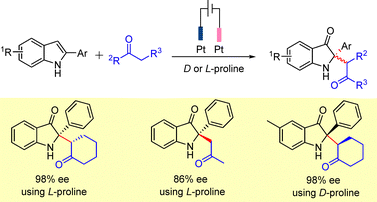
The synthesis of a wide variety of disubstituted chiral indoline-3-ones was recently achieved by merging electrocatalysis and organocatalysis.38 The method is oxidant- and metal-free and proceeds via anodic oxidation coupled with asymmetric proline-catalyzed alkylation in an undivided cell under constant-current conditions. Products with opposite absolute configurations were obtained with high enantioselectivity depending on whether D- or L-proline was employed as the chiral catalyst (Scheme 22).
Huang et al. reported a copper-catalyzed intramolecular oxidative amidation protocol for the synthesis of 2-hydroxyindoxyls bearing an isopropyl group at the C2 position (Scheme 23).44 Molecular oxygen served as the oxidant, and the reaction required elevated temperatures and proceeded through a radical pathway.
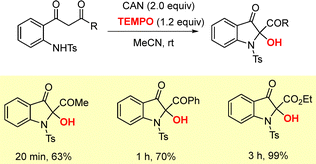
Yang and co-workers reported an oxidative cyclization of 2-aminophenyl-1,3-diones using CAN and TEMPO as oxidants for the synthesis of 2-hydroxyindolin-3-ones in moderate to excellent yields (up to 99%, Scheme 24).45 The protocol was further applied to synthetic studies toward the natural product matemone, starting from readily available 2-amino-4-bromobenzoic acid, enabling efficient installation of the key precursor (Scheme 24).
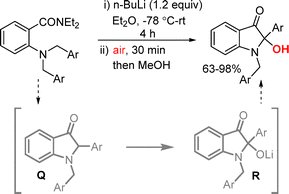
Coldham and co-workers reported a BuLi-promoted intramolecular cyclization protocol for the synthesis of 2-hydroxyindoxyls.46 Treatment of a 2-(benzylamino)benzamide substrate with BuLi led to the in situ generation of a 2-arylidoline-3-one intermediate Q, which underwent rapid autoxidation followed by hydrolysis to furnish the desired 2-hydroxyindoxyls in good yields (Scheme 25).
In 2025, the Szpilman group reported the synthesis of a broad range of 2-substituted-2-hydroxyindolin-3-ones using a hypervalent iodine reagent (PIFA) via a double umpolung reaction of 2-aminophenyl-3-oxopropanoates.47 Mechanistic investigations, supported by DFT calculations, indicated that two C-enolonium species served as key intermediates in this transformation (Scheme 26).
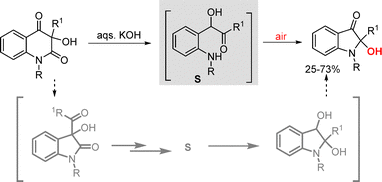
Ring contraction of 3-hydroxy-2,4(1H,3H)-quinolinediones in aqueous potassium hydroxide resulted in the formation of 2-hydroxyindoxyls through intermediate S (Scheme 27).48 The chemoselectivity of the reaction was governed by the nature of the N-substituent and the reaction conditions. N-Phenyl-substituted derivatives afforded 2-hydroxyindoxyls, whereas N-alkyl- and N-benzyl-substituted substrates afforded the corresponding dioxindoles. The reaction was proposed to proceed via formation of a 3-hydroxy-2-oxindole intermediate, which, upon ring opening and rearrangement, furnished the desired products following aerial oxidation.
In 2019, we developed a one-pot approach to pseudooxindole 2-hydroxyindoxyls via simple Grignard addition to 3-hydroxy oxindoles (Scheme 28).27 The reaction was proposed to proceed through a 1,2-hydride shift to generate 2-indoxyl intermediate T, which upon autoxidation furnished N-substituted 2-hydroxyindoxyls. Key features of this domino nucleophilic addition–oxidation protocol included the use of simple substrates, excellent yields, scalability, a broad substrate scope, and operational ease.27
2.2 Synthesis of cyclic indoxyls
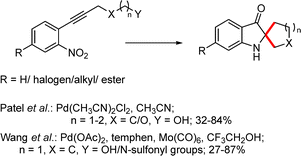
2.2.1.1 Metal catalyzed approaches. Patel et al. developed a palladium-catalyzed intramolecular addition of nitro and alcohol functionalities to alkynes, enabling the synthesis of C2-spiroindoxyl derivatives (Scheme 29).49,50 Hexynol- and propargyl glycol-derived substrates afforded the corresponding C2-spiroindoxyls in good yields (78–84%). Formation of the C2-spiropseudoindoxyl framework was proposed to proceed via a 5-exo-dig nitroalkyne cyclization through cycloisomerization, followed by nucleophilic addition and reduction.
Along similar lines, Wang and co-workers reported a simple and efficient method for the synthesis of a wide range of N,N′-ketal spiroindoxyls from o-alkynylnitrobenzenes in the presence of a palladium catalyst (Scheme 29).51
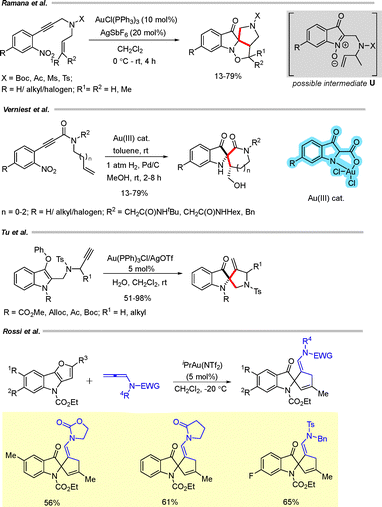
Ramana and co-workers described a cycloisomerization–cycloaddition cascade of o-alkynylnitrobenzenes to obtain C2-spiroindoxyl derivatives (Scheme 30).52 In the presence of 10 mol% AuCl(PPh3) and 20 mol% AgSbF6, the alkynylnitrobenzenes underwent cyclization via the formation of intermediate U, leading to the desired C2-spiroindoxyl products.
Verniest and co-workers investigated an Au(III)-catalyzed cycloisomerization of o-nitrophenylpropiolamides to obtain C2-spiroindoxyls in high yields (Scheme 30).53 A range of o-nitrophenylpropiolamides furnished the corresponding spiropyrryloxys as single diastereomers upon subsequent hydrogenation in the presence of 10% Pd/C.
Tu and coworkers reported a C2-selective cyclization of alkynyl oximes to obtain C2-spiroindoxyls (Scheme 30).54 Treatment of the alkynyl oximes with Au(PPh3)Cl/AgOTf (5 mol%) in CH2Cl2 led to the formation of the corresponding spiroindoxyl derivatives.
The reaction of allenamides with furano–indole derivatives was investigated under gold(I) catalysis for the synthesis of a new class of spirocyclic indoline-3-ones.55 The transformation proceeded through a cascade process involving addition of a gold-activated allene to the furan moiety of the furoindole, followed by sequential ring-opening and ring-closing events to afford 2-spirocyclopentane-1,2-dihydro-3H-indolin-3-ones in good yields. It was observed that the reaction required C2-substituted furano–indoles, and only C2-alkyl substituents were compatible under the developed conditions.56
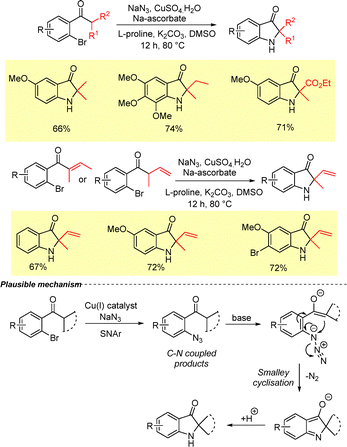
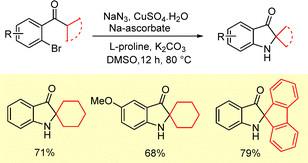
The intramolecular version of the sequential SNAr-Smalley cyclization protocol developed by Ramana et al. was employed for the synthesis of spirocyclic indoline-3-oes from α-bromophenyl cyclic sec-alkyl ketones in the presence of CuI and sodium azide (Scheme 31).16 The reaction follows the same mechanistic course as discussed previously in Scheme 2.
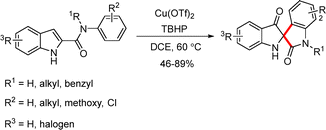
A copper-catalyzed spirocyclization of 1H-indole-2-carboxamides was described by Li et al. for the synthesis of spiroindoxyls (Scheme 32).56 In the presence of Cu(OTf)2 (5 mol%) and tert-butyl hydroperoxide (TBHP, 3.0 equiv.), a series of spiroindoxyl derivatives were obtained in high yields.
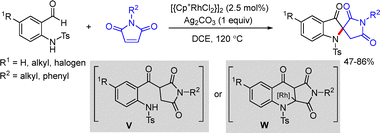
Li and co-workers reported a Rh(III)-catalyzed hydrogenative coupling of N-sulfonyl 2-aminobenzaldehydes with olefins to obtain spiroindoxyls (Scheme 33).57 The coupling reaction proceeded in the presence of [Cp*RhCl2]2 (2.5 mol%) and Ag2CO3 (1.0 equiv.), producing the desired spiroindoxyls via the formation of plausible reaction intermediates V and W.
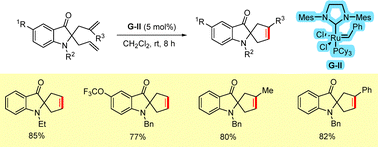
In 2015, we used ring-closing metathesis (RCM) for the synthesis of a novel class of spirocyclopentene 3-oxindole derivatives from the corresponding diallyl oxindoles.25 The RCM proceeded smoothly in the presence of the second-generation Grubbs catalyst (G-II) to afford the desired oxindoles in high yields (Scheme 34).
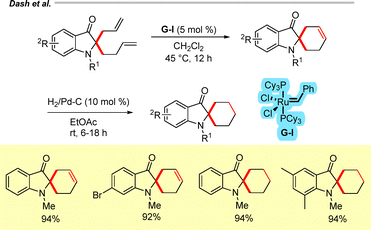
More recently, we extended this RCM-based strategy to the synthesis of spirocyclohexene-3-oxindole and spirocyclohexane-3-oxindole derivatives via sequential Ru-catalyzed olefin metathesis (G-I) followed by catalytic hydrogenation of 2-allyl-2-(but-3-enyl)-3-oxindoles (Scheme 35).26
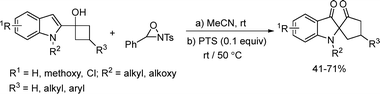
Zhu and co-workers developed a visible-light-mediated reaction of indole-tethered alcohols, followed by dearomatization of indoles, to synthesize C2-spiroindoxyls under mild conditions (Scheme 36).58 Visible light acted as a bifunctional catalyst to redox-catalyze the cascade providing an efficient route to obtain the corresponding nitrogen heterocycles.
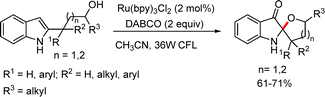
2.2.1.2 Metal free methods. Fan et al. synthesized C2-spiroindoxyls via an iodophenyltriene(III)-mediated tandem C–H oxidation using iodobenzene diacetate and tetrabutylammonium iodide (Scheme 37).59 The acyl precursors reacted smoothly in the presence of PhI(OAc)2 (2.0 equiv.) and Bu4NI (2.0 equiv.) as oxidants, affording the desired products in high yields.
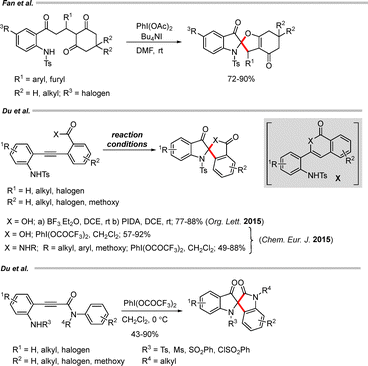
Du and co-workers described a hypervalent iodine (PIDA)-mediated cascade cyclization to obtain C2-spiroindoxyls via aminocarboxylation of diarylalkyl oxoaminocarboxylates.60 In a related study, another hypervalent iodine reagent, PhI(OCOCF3)2, was employed to enable a single-oxidant cascade cyclization of diarylacetylene derivatives, furnishing spiroindoxyls bearing N,O-ketal or N,N-ketal motifs in high yields (Scheme 37). Furthermore, Du and co-workers investigated an additional oxidative cascade annulation using the same hypervalent iodine reagent, in which 2-sulfonamido-N-phenylpropiolamide derivatives underwent oxidation in the presence of PhI(OCOCF3)2 (2.2 equiv.) to generate C2-spiroindoxyl compounds in moderate to good yields.61
An oxidative dearomatization/rearrangement of indole-2-ylcyclobutanols in the presence of 3-phenyl-2-tosyl-1,2-oxaziridine as the oxidant afforded C2-spiroindoxyl derivatives (Scheme 38), as reported by Zhang's group.62
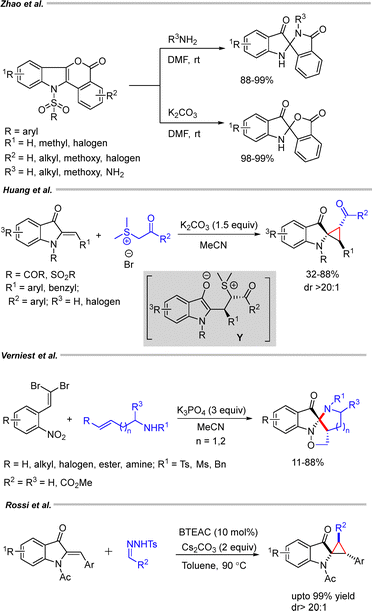
Zhao and coworkers investigated a one-pot ring-contraction rearrangement of isochromeno[4,3-b]indole-5(11H)-one, which afforded the corresponding N,N′-unsubstituted ketals in excellent yields (Scheme 39).63 The transformation was proposed to proceed via a hydroxide-ion-mediated saponification–tosyl removal step, followed by spirocyclization and protonation to generate the C2-spiroindoxyl products.
Huang et al. studied the reaction of sulfur ylides with (Z)-2-indenyl hydrazines, followed by a 1,4-addition/substitution cyclization to obtain cyclopropane-fused spiroindoxyls (Scheme 39).64 The transformation was proposed to proceed via the formation of intermediate Y.
A single-step, metal-free reaction of o-nitrodibromoalkenes with secondary amines was demonstrated by Verniest's group, delivering C2-spiroindoxyl derivatives (Scheme 39).65 The cascade transformation of sterically hindered dibromoalkenes with sulfonamides afforded the corresponding products in moderate yields (11–71%).
A diastereoselective cyclopropanation of aza-aurones with tosylhydrazones was reported for the synthesis of C2-spirocyclopropanated 3-oxindoles (Scheme 39).66 The reaction proceeded in the presence of a catalytic amount of benzyltriethylammonium chloride and Cs2CO3 as the base, affording 2-spirocyclopropyl indoline-3-ones with a broad substitution pattern on both the aza-aurones and tosylhydrazones.
An interrupted Ugi reaction developed by Sorensen et al. was employed for the synthesis of spirocyclic indoxyls.67 The electron-rich, sterically hindered cyclic imine intermediates furnished the desired products in good yields. Intriguingly, the highly hindered camphor-derived imine substrate provided the cyclic indoline-3-one in good yield (Scheme 40). This methodology was applied as a key step for the construction of the indoxyl core of 11-methoxy mitragynine indoxyl.
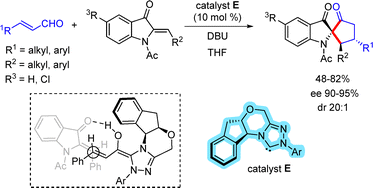
Glorius and co-workers reported the first N-heterocyclic carbene (NHC)-catalyzed reaction between α,β-unsaturated aldehydes and aza-aurones for the synthesis of spirocyclic 3-oxindoles (Scheme 41).68 A range of enantiomerically enriched C2-spiroindoxyl derivatives were obtained in high yields and with excellent stereoselectivities using an NHC catalyst (10 mol%) in the presence of DBU (150 mol%). The NHC catalyst reacts with the enal substrate to generate an NHC-bound homoenolate intermediate. This species forms hydrogen-bonding interactions with the aza-aurone, organizes the transition state, and directs the Michael addition from the back face, thereby providing a key intermediate that accounts for the observed stereoselectivity.
Xu et al. reported, for the first time, the synthesis of pyrrolidinyl spirooxoximes via an asymmetric [3+2] cyclization of o-hydroxy aromatic aldimines with (Z)-1-acetyl-2-benzylideneindoline-3-one, enabled by a hydrogen-bonding network (Scheme 42).69 A wide range of spirooxoximes were obtained in high to excellent yields with excellent enantioselectivities. The stereochemical outcome is governed by steric interactions, where the TS-II pathway is disfavored, thereby directing the formation of the observed enantiomer.
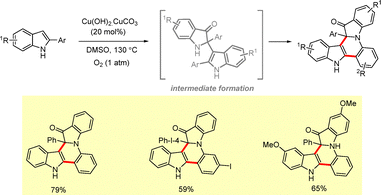
2.2.2.1 Metal catalyzed approaches. Sang et al. reported a copper catalyzed oxidative approach involving simultaneous C–H bond formation for the synthesis of a fused 3-oxindole framework.70 The use of 2-arylindoles as starting materials, an inexpensive copper catalyst [Cu(OH)2·CuCO3], and molecular oxygen as the oxidant rendered this method synthetically useful for the construction of six-membered fused indole and quinoline skeletons in good yields (Scheme 43).
A palladium-catalyzed intramolecular deacetylative dearomatization reaction of 3-acetoxyindoles was developed by Jia and co-workers. A range of fused tetracyclic indolin-3-ones bearing tetrasubstituted C2-stereocenters were achieved in good yields (Scheme 44).71
2.2.2.2 Organocatalytic approaches. 3H-Indol-3-ones have frequently been employed as dienophiles in enantioselective aza–Diels–Alder reactions for the construction of structurally complex fused indoxyl derivatives.
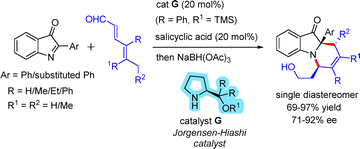
Rueping and co-workers reported a BINOL-derived phosphoric acid (PA)-catalyzed reaction of cyclopentadiene and 3H-indol-3-ones, resulting in aza-tetracyclic products with good yields and enantiomeric excess (Scheme 45).72
Chen and co-workers investigated the asymmetric cycloaddition of 3H-indol-3-ones with 2,4-dienals in the presence of the Jørgensen–Hayashi catalyst (catalyst G).73 This protocol enabled the synthesis of a wide range of tricyclic polyhydropyrido[1,2-a]indole frameworks with excellent enantioselectivity (Scheme 46).
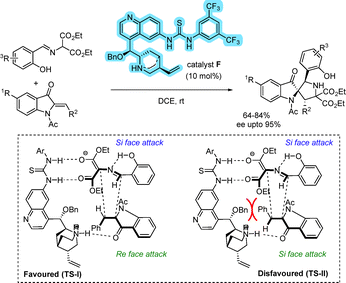
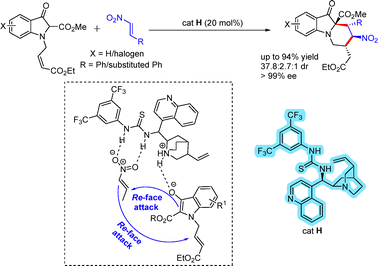
Functionalized piperidino[1,2-a]indoline derivatives were synthesized via an asymmetric Michael addition between nitrostyrenes and suitable 3-oxindole precursors (Scheme 47).74 The stereochemical outcome of the reaction was unambiguously established by X-ray crystallographic analysis of the product. A hydrogen-bonding interaction between the thiourea moiety of the catalyst enhances the electrophilicity of the nitroolefin. Simultaneously, the tertiary amine group generates an enolate from the indolin-3-one. The resulting enolate undergoes Michael addition via a Re-face attack on the nitroolefin. Subsequently, the carbanion intermediate performs a second Re-face attack on the double bond of the α,β-unsaturated ester, leading to the formation of the product with the desired stereochemical outcome.
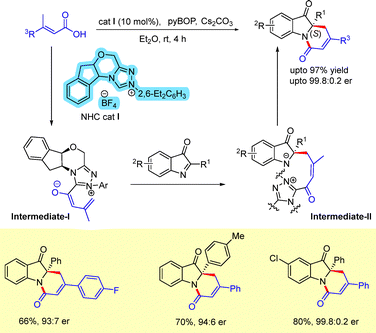
An N-heterocyclic carbene (NHC)-catalyzed formal [4+2] annulation of 2-aryl-3H-indol-3-ones with α,β-unsaturated carboxylic acids bearing a γ-hydrogen was reported for the construction of structurally important C2-quaternary indolin-3-ones (Scheme 48).75 A broad substrate scope was demonstrated, delivering the products in excellent yields and enantioselectivities. The reaction is proposed to involve in situ generation of vinyl enolates, followed by γ-addition to cyclic ketimines, affording chiral tricyclic fused indoxyls. Under the optimized conditions, the NHC catalyst reacts with the α,β-unsaturated carboxylic acid to generate a deprotonated intermediate I, which undergoes nucleophilic addition to the cyclic imine to afford intermediate II. Subsequent lactam formation of intermediate II furnishes the desired [4+2] annulation product.
An enantioselective multicomponent reaction of 2-aryl-3H-indol-3-ones, aldehydes, and diethyl 2-aminomalonate was studied under axially chiral cyclic phosphoric acid catalysis, leading to the formation of 2,3-dihydro-1H-imidazo[1,5-a]indol-9(9aH)-one derivatives, representing a new class of fused indoxyl frameworks (Scheme 49).76 The reaction proceeded with good yields and excellent ee values with a broad range of functional groups. Furthermore, oxidation of the 2,3-dihydro-1H-imidazo[1,5-a]indol-9(9aH)-one derivatives provided the corresponding 1H-imidazo[1,5-a]indol-9(9aH)-ones.76 An azomethine ylide, generated from the reaction of aldehyde and diethyl 2-aminomalonate, forms dual hydrogen bonding with both the axially chiral cyclic phosphoric acid catalyst and the indol-3-one. This organization promotes an enantioselective [3+2] cycloaddition, which governs the stereochemical outcome of the reaction.
 | ||
| Scheme 49 Synthesis of fused indoxyls via phosphoric acid catalyzed asymmetric multicomponent reaction. | ||
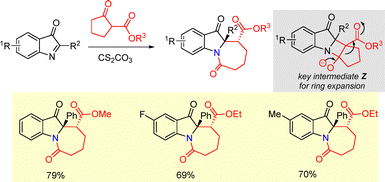
2.2.2.3 Other approaches. Zhang et al. reported a base-mediated method for the synthesis of fused seven-membered polycyclic indoline-3-one derivatives via insertion of cyclic C-acylimines into cyclic diketones (Scheme 50).77 The reaction proceeds via base mediated formation of intermediate Z, which facilitates the ring-expansion process. The cyclic C-acylimines were generated in situ from the corresponding 2-alkynyl arylazides via a Pd-catalyzed oxidative intramolecular cyclization.77
2.3 Synthesis of dimeric indoxyls
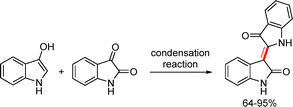
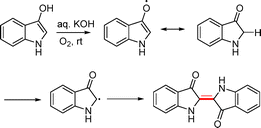
Two of the most well-known dimeric indoxyls are the organic dyes indirubin and indigo. The dimeric indoxyl derivative indirubin was readily prepared via a condensation reaction between 2,3-dihydro-1H-indol-3-one and isatin (Scheme 51).78The synthesis of indigo derivatives has been reported using several methods. Indoxyl readily underwent aerial oxidation in alkaline solution to generate indigo (Scheme 52).79 The initially formed indoxyl anion loses an electron to generate a radical species, which subsequently dimerizes via a radical coupling mechanism to afford indigo.79
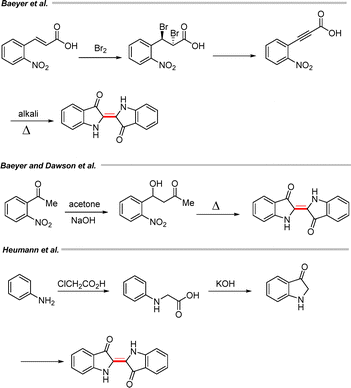
Baeyer developed a practical method in which o-nitrocinnamic acid was first converted into its dibromo derivative, which upon treatment with potassium hydroxide generated o-nitrophenylpropiolic acid (Scheme 53). This compound subsequently produced indigo under alkaline conditions. Later, Baeyer and Drewsen employed o-nitrobenzaldehyde as the starting material; treatment with acetone in the presence of NaOH afforded the corresponding Claisen–Schmidt condensation product, which upon heating with alkali produced indigo in good yield (Scheme 53).80 With the aim of developing a scalable synthesis of indigo, Heumann introduced a practical route starting from aniline.81 Aniline was first converted to phenylglycine by reaction with chloroacetic acid, which then furnished indigo under alkaline conditions via the intermediate formation of 3-oxindole (Scheme 53).81,82
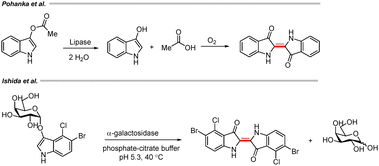
Reports on enzyme-mediated synthesis of indigo derivatives have also been highlighted in recent years. Pohanka et al. investigated the enzymatic cleavage of indoxyl acetate in the presence of lipase, wherein the generated 3-indoxyl underwent rapid aerial oxidation to afford indigo (Scheme 54).83 Similarly, Ishida and co-workers carried out enzymatic hydrolysis of an indoxyl galactoside using α-galactosidase. The substrate was hydrolyzed by the glycosidase, and the liberated indoxyl was subsequently oxidized and dimerized to form the indigo dye (Scheme 54).84
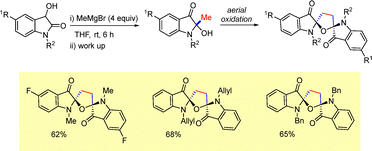
In 2019, we reported a unique method for the synthesis of a novel class of spiro-furan bisindoxyls.27 Treatment of 3-hydroxy-2-oxindoles with methylmagnesium bromide furnished the desired dimeric indoxyls via the formation of 2-methyl-2-hydroxyindoxyls (Scheme 55). The intermediate 2-hydroxyindoxyl derivatives underwent rapid aerial oxidation to afford the corresponding dimeric spiro-indoxyls.
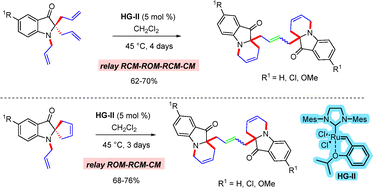
Recently, we have developed a domino metathesis approach for the synthesis of a new class of fused dimeric indoxyl ring systems.85 Treatment of diallyl oxindoles with the second-generation Hoveyda–Grubbs catalyst (HG-II) generated dimeric 3-oxindoles via a relay ring closing metathesis–ring opening metathesis–ring closing metathesis–cross metathesis (RCM–ROM–RCM–CM) sequence (Scheme 56). Under the same reaction conditions, the corresponding spirocyclic oxindoles also underwent a relay ring opening metathesis–ring closing metathesis–cross metathesis (ROM–RCM–CM) process to generate the dimeric oxindoles in improved yields (Scheme 56).
Intriguingly, the fused dimeric indoxyls underwent cross metathesis with methyl acrylate in the presence of HG-II, affording the corresponding monomeric fused indoxyl derivatives (Scheme 57).85
3. Summary and outlook
The 3-oxindole (indoxyl) motif represents a unique and versatile building block in organic synthesis, enabling a wide range of unusual reactions, rearrangements, and the rapid generation of molecular complexity. Substantial efforts have been devoted to this area, and remarkable advances have been achieved over the past decade. In this review, we have summarized key developments in the construction of diverse classes of indoxyl-based ring systems. Recent progress has been driven by the successful integration of complementary synthetic strategies and catalytic platforms, including metal catalysis, organocatalysis, and cascade processes, which have collectively enabled access to structurally diverse 3-oxindole architectures. Given the ubiquitous presence of the 3-oxindole motif in natural products and bioactive molecules, the development of new and efficient synthetic methods for this scaffold remains an active and continually evolving field. Looking ahead, several challenges and opportunities remain. These include the development of enantioselective variants, broader implementation of photoredox catalysis, electrocatalysis, and photoelectrocatalysis, and innovative strategies for the selective functionalization of the C2 position. In addition, inspiration from total synthesis is expected to play a crucial role, as access to less readily available regioisomers, such as 3-oxindoles over 2-oxindoles, can enable more sophisticated, atom-economical, and convergent construction of complex molecular frameworks. In view of the diverse synthetic applications and emerging reactivity profiles of the 3-oxindole scaffold, we anticipate that new and increasingly creative transformations will continue to enrich this field in the years to come.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
JD thanks ICMR [Grant Number: IIRPIG-2024-01-01128] and SERB-CRG for funding.References
- C. Marti and E. M. Carreira, Eur. J. Org. Chem., 2003, 2209 CrossRef CAS.
- C. V. Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46, 8748 CrossRef CAS PubMed.
- D. Cheng, Y. Yshihara, B. Tan and F. C. Barbas, ACS Catal., 2014, 4, 743 Search PubMed.
- P. S. Dhote, P. Patel, K. Vanka and C. V. Ramana, Org. Biomol. Chem., 2021, 19, 7970–7994 Search PubMed.
- Y. Ji, X. He, C. Peng and W. Huang, Org. Biomol. Chem., 2019, 17, 2850–2864 RSC.
- Deeksha, D. Chaudhary, I. K. Gurjar, A. P. Menon, A. R. Deorkar and R. Singh, Asian J. Org. Chem., 2025, 14, e202500102 CrossRef CAS.
- J. M. Finefield, J. C. Frisvad, D. H. Sherman and R. M. Williams, J. Nat. Prod., 2012, 75, 812 Search PubMed.
- C. Niemann and J. W. Kessel, J. Org. Chem., 1966, 31, 2265 Search PubMed.
- D. F. Dickel, C. L. Holden, R. C. Maxfield, L. E. Paszek and W. I. Taylor, J. Am. Chem. Soc., 1958, 80, 123 CrossRef CAS.
- P. S. Steyn, Tetrahedron Lett., 1971, 3331 CrossRef CAS.
- M. S. C. Pedras and D. P. O. Okinyo, Chem. Commun., 2006, 1848 Search PubMed.
- I. Carletti, B. Banaigs and P. Amade, J. Nat. Prod., 2000, 63, 981 CrossRef CAS PubMed.
- Reports. A. Váradi, G. F. Marrone, T. C. Palmer, A. Narayan, M. R. Szabó, V. Le Rouzic, S. G. Grinnell, J. J. Subrath, E. Warner, S. Kalra, A. Hunkele, J. Pagirsky, S. O. Eans, J. M. Medina, J. Xu, Y.-X. Pan, A. Borics, G. W. Pasternak, J. P. McLaughlin and S. Majumdar, J. Med. Chem., 2016, 59, 8381–8397 CrossRef.
- K. S. MacMillan and D. L. Boger, J. Med. Chem., 2009, 52, 5771–5780 Search PubMed.
- R. S. Bhakuni, Y. N. Shukla and R. S. Thakur, Phytochemistry, 1991, 30, 3159 Search PubMed.
- Y. Goriya and C. V. Ramana, Chem. Commun., 2013, 49, 6376–6378 Search PubMed.
- A. Singh, S. Vanaparthi, S. Choudhury, R. Krishnan and I. Kumar, RSC Adv., 2019, 9, 24050–24056 Search PubMed.
- S. K. Guchhait, V. Chaudhury, V. A. Rana, G. Priyadarshani, S. Kandekar and M. Kashyap, Org. Lett., 2016, 18, 1534–1537 Search PubMed.
- X.-Y. Zhou, X. Chen, L.-G. Wang, D. Yang and J.-H. Li, Synlett, 2018, 835–839 Search PubMed.
- X. Jiang, B. Zhu, K. Lin, G. Wang, W.-K. Su and C. Yu, Org. Biomol. Chem., 2019, 17, 2199–2203 Search PubMed.
- F. Lin, Y. Chen, B. Wang, W. Qin and L. Liu, RSC Adv., 2015, 5, 37018–37022 Search PubMed.
- T. M. Bolt, B. J. Atienza and F. G. West, RSC Adv., 2014, 4, 31955–31959 Search PubMed.
- P. J. Arandhara and A. K. Saikia, Org. Lett., 2025, 27, 6641–6647 Search PubMed.
- F. Xu and M. W. Smith, Chem. Sci., 2021, 12, 13756–13763 Search PubMed.
- K. Dhara, T. Mandal, J. Das and J. Dash, Angew. Chem., Int. Ed., 2015, 54, 15831–15835 CrossRef CAS PubMed.
- N. Parui, T. Mandal, S. Maiti and J. Dash, Chem. – Eur. J., 2024, 30, e202401059 Search PubMed.
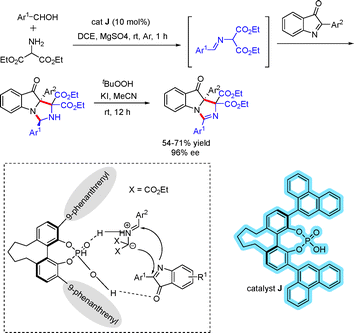
- T. Mandal, G. Chakraborti, S. Maiti and J. Dash, Org. Lett., 2019, 21, 8044–8048 CrossRef CAS PubMed.
- R. C. Das, S. Barik, A. A. Kunhiraman, A. Goswami, A. Mondal, M. De and A. Biju, ACS Catal., 2024, 14, 4202–4210 CrossRef CAS.
- S. Lerch, L.-N. Unkel and M. Brasholz, Angew. Chem., Int. Ed., 2014, 53, 6558–6562 CrossRef CAS PubMed.
- Z. Xia, J. Hu, Y.-Q. Gao, Q. Yao and W. Xie, Chem. Commun., 2017, 53, 7485–7488 Search PubMed.
- J. S. Schneekloth Jr., J. Kim and E. J. Sorensen, Tetrahedron, 2009, 65, 3096–3101 Search PubMed.
- A. Parra, R. Alfaro, L. Marzo, A. Moreno-Carrasco, J. L. Garcia Ruano and J. Alemán, Chem. Commun., 2012, 48, 9759–9761 Search PubMed.
- M. Rueping, R. Rasappan and S. Raja, Helv. Chim. Acta, 2012, 95, 2296–2303 Search PubMed.
- C.-Y. Jin, Y. Wang, Y.-Z. Liu, C. Shen and P.-F. Xu, J. Org. Chem., 2012, 77, 11307–11312 CrossRef CAS PubMed.
- R.-R. Liu, S.-C. Ye, C.-J. Lu, G.-L. Zhuang, J.-R. Gao and Y.-X. Jia, Angew. Chem., Int. Ed., 2015, 54, 11205–11208 CrossRef CAS.
- Y.-L. Zhao, X.-H. Fei, Y.-Q. Tang, P.-F. Xu, F.-F. Yang, B. He, X.-Z. Fu, Y.-Y. Yang, M. Zhou, Y.-H. Mao, Y.-X. Dong and C. Li, J. Org. Chem., 2019, 84, 8168–8176 Search PubMed.
- X. Yuan, X. Wu, F. Peng, H. Yang, C. Zhub and H. Fu, Chem. Commun., 2020, 56, 12648–12651 RSC.
- F.-Y. Lu, Y.-J. Chen, Y. Chen, X. Ding, Z. Guan and Y.-H. He, Chem. Commun., 2020, 56, 623–626 RSC.
- Z. Wang and L. S. Jimenez, J. Am. Chem. Soc., 1994, 116, 4977 CrossRef CAS.
- C.-S. Chien, T. Takanami, T. Kawasaki and M. Sakamoto, Chem. Pharm. Bull., 1985, 33, 1843 CrossRef CAS.
- C. I. Altinis Kiraz, T. J. Emge and L. S. Jimenez, J. Org. Chem., 2004, 69, 2200 CrossRef CAS PubMed.
- B. Staskun, J. Org. Chem., 1988, 53, 5287 CrossRef CAS.
- C. W. Rees and C. R. Sabet, J. Chem. Soc., 1965, 870 RSC.
- J. B. Huang, T. T. Mao and Q. Zhu, Eur. J. Org. Chem., 2014, 2878 CrossRef CAS.
- S.-S. Wen, Z.-F. Zhou, J.-A. Xiao, J. Li, H. Xiang and H. Yang, New J. Chem., 2017, 41, 11503 RSC.
- I. Coldham, H. Adams, N. J. Ashweek, T. A. Barker, A. T. Redder and M. C. Skilbeck, Tetrahedron Lett., 2010, 51, 2457 CrossRef CAS.
- B.-E. Oded, S. Maity, H. Kornweitz and A. M. Szpilman, J. Org. Chem., 2025, 90, 9649–9657 CrossRef CAS PubMed.
- S. Kafka, A. Klasek and J. Kosmrlj, J. Org. Chem., 2001, 66, 6394 CrossRef CAS PubMed.
- C. V. Ramana, P. Patel, K. Vanka, B. Miao and A. Degterev, Eur. J. Org. Chem., 2010, 5955 CrossRef CAS.
- P. Patel and C. V. Ramana, Org. Biomol. Chem., 2011, 9, 7327 RSC.
- L. W. Chen, J. L. Xie, H. J. Song, Y. X. Liu, Y. C. Gu and Q. M. Wang, Org. Chem. Front., 2017, 4, 1731 Search PubMed.
- C. V. Suneel Kumar and C. V. Ramana, Org. Lett., 2014, 16, 4766 CrossRef CAS PubMed.
- N. Marien, B. Brigou, B. Pinter, F. De Proft and G. Verniest, Org. Lett., 2015, 17, 270 CrossRef CAS PubMed.
- Y. Q. Zhang, D. Y. Zhu, Z. W. Jiao, B. S. Li, F. M. Zhang, Y. Q. Tu and Z. Bi, Org. Lett., 2011, 13, 3458 CrossRef CAS PubMed.
- V. Pirovano, E. Brambilla, S. Rizzato, G. Abbiati and E. Rossi, J. Org. Chem., 2019, 84, 5150 Search PubMed.
- L. Kong, M. Wang, F. Zhang, M. Xu and Y. Li, Org. Lett., 2016, 18, 6124 CrossRef CAS PubMed.
- T. Zhang, Z. Qi, X. Zhang, L. Wu and X. Li, Chem. – Eur. J., 2014, 20, 3283 Search PubMed.
- M. Zhang, Y. Duan, W. Li, Y. Cheng and C. Zhu, Chem. Commun., 2016, 52, 4761 Search PubMed.
- Y. Sun, J. Gan and R. Fan, Adv. Synth. Catal., 2011, 353, 1735 Search PubMed.
- (a) X. Zhang, W. Hou, D. Zhang-Negrerie, K. Zhao and Y. Du, Org. Lett., 2015, 17, 5252 CrossRef CAS PubMed; (b) X. Zhang, C. Yang, D. Zhang-Negrerie and Y. Du, Chem. – Eur. J., 2015, 21, 5193 Search PubMed.
- B. Zhang, X. Zhang, B. Hu, D. Sun, S. Wang, D. Zhang-Negrerie and Y. Du, Org. Lett., 2017, 19, 902 Search PubMed.
- J. B. Peng, Y. Qi, A. J. Ma, Y. Q. Tu, F. M. Zhang, S. H. Wang and S. Y. Zhang, Chem. – Asian J., 2013, 8, 883 Search PubMed.
- X. Zhang, G. Zhou, Y. Zhang, D. Zhang-Negrerie, Y. Du and K. Zhao, J. Org. Chem., 2016, 81, 11397 CrossRef CAS PubMed.
- X. Tang, H. P. Zhu, J. Zhou, Y. Chen, X. L. Pan, L. Guo, J. L. Li, C. Peng and W. Huang, Org. Biomol. Chem., 2018, 16, 8169 Search PubMed.
- N. Marien, B. N. Reddy, F. De Vleeschouwer, S. Goderis, K. Van Hecke and G. Verniest, Angew. Chem., Int. Ed., 2018, 57, 5660 CrossRef CAS PubMed.
- V. Pirovano, E. Brambilla, M. Riva, S. Leoni, S. Rizzato, D. Garanzini, G. Abbiati and E. Rossi, Org. Biomol. Chem., 2021, 19, 3925 RSC.
- J. Kim, J. S. Schneekloth, Jr. and E. J. Sorensen, Chem. Sci., 2012, 3, 2849 RSC.
- C. Guo, M. Schedler, C. G. Daniliuc and F. Glorius, Angew. Chem., Int. Ed., 2014, 53, 10232–10236 CrossRef CAS PubMed.
- L. Zhang, Y. Wang, X. Hu and P. Xu, Chem. – Asian J., 2016, 11, 834 CrossRef CAS.
- P. Sang, Y. Xie, J. Zou and Y. Zhang, Adv. Synth. Catal., 2012, 354, 1873–1878 CrossRef CAS.
- R.-X. Liang, K. Wang, L.-J. Song, W.-J. Sheng and Y.-X. Jia, RSC Adv., 2019, 9, 13959–13967 Search PubMed.
- M. Rueping and S. Raja, Beilstein J. Org. Chem., 2012, 8, 1819–1824 CrossRef CAS PubMed.
- J.-X. Liu, Q.-Q. Zhou, J.-G. Deng and Y.-C. Chen, Org. Biomol. Chem., 2013, 11, 8175–8178 RSC.
- Y.-L. Zhao, Y. Wang, J. Cao, Y.-M. Liang and P.-X. Xu, Org. Lett., 2014, 16, 2438–2441 Search PubMed.
- S. Fang, S. Jin, R. Ma, T. Lu and D. Du, Org. Lett., 2019, 21, 5211–5214 CrossRef CAS PubMed.
- X. Yuan, X. Wu, P. Zhang, F. Peng, C. Liu, H. Yang, C. Zhu and H. Fu, Org. Lett., 2019, 21, 2498–2503 CrossRef CAS PubMed.
- P. Li, R. Shang, Z. Zhou, G. Hu and X. Zhang, Eur. J. Org. Chem., 2020, 2146–2152 CrossRef CAS.
- H. M. Riepl and C. Urmann, Helv. Chim. Acta, 2012, 95, 1461–1477 CrossRef CAS.
- R. Vauquelin, L. J.-Condat, N. Joly, N. Jullian, E. Choque and P. Martin, Molecules, 2024, 20, 4804, DOI:10.3390/molecules29204804.
- N. R. Krishnaswamy and C. N. Sundaresan, Reson., 2003, 8, 42–48 Search PubMed.
- A. Baeyer and V. Drewsen, J. Chem. Soc., 2016, 6, 18–22 Search PubMed.
- (a) https://de.wikipedia.org/wiki/Heumann-Synthese ; (b) https://de.wikipedia.org/wiki/Karl_Heumann_(Chemiker) .
- T. Valek, A. Kostelnik, P. Valkova and M. Pohanka, Int. J. Anal. Chem., 2019, 8538340, DOI:10.1155/2019/8538340.
- S. Nagata, H. Tomida, H.-L. Hirose, H.-N. Tanaka, H. Ando, A. Imamura and H. Ishida, RSC Adv., 2019, 9, 28241–28247 RSC.
- T. Mandal, K. Dhara, N. Parui and J. Dash, ChemCatChem, 2020, 12, 4754–4759 CrossRef CAS.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |