Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Nickel(II) driven activity enhancement of a donor–acceptor porous organic polymer for selective photoreduction of carbon dioxide
A John David a,
Nandini M Gotgi
a,
Nandini M Gotgi b,
Gopika Udayakumar
b,
Gopika Udayakumar a,
A Hameed Ibrahim
a,
A Hameed Ibrahim a,
Abhilash Pullanchiyodan
a,
Abhilash Pullanchiyodan *a,
Debashis Ghosh
*a,
Debashis Ghosh *c and
Anjan Das
*c and
Anjan Das *a
*a
aDepartment of Chemistry, SRM Institute of Science and Technology, Kattankulathur, Chennai, Tamil Nadu, India 603203. E-mail: anjand@srmist.edu.in
bDepartment of Chemistry, St. Joseph's University, 36 Lalbagh Road, Bengaluru-560027, Karnataka, India
cDepartment of Chemistry, R.D. & D.J. College, Munger 811201, Bihar, India
First published on 20th February 2026
Abstract
We report the design and synthesis of porous organic polymers (Py-DPEN) as a photocatalyst for the photoreduction of CO2 to carbon monoxide (CO). Incorporation of Ni(II) drastically enhanced the photocatalytic activity under visible light irradiation (70 W m−2, 470 nm), resulting in a significantly higher CO yield (38 µmol) with excellent selectivity. This pronounced improvement demonstrates the crucial role of Ni(II) in driving CO2 conversion using TEOA as a sacrificial electron donor in an acetonitrile–water medium. Comprehensive characterization, photocatalytic performance evaluation, and product selectivity studies confirm the outstanding efficiency of the system and provide valuable insights for the rational design of sustainable energy conversion catalysts.
The increasing levels of atmospheric carbon dioxide (CO2) due to anthropogenic activities have led to severe environmental challenges, including global warming and climate change.1 To address this issue, the development of innovative and sustainable technologies for CO2 capture, utilization, and conversion is required. Among various strategies, photocatalytic reduction of CO2 into value-added chemicals using solar energy has garnered significant interest as a promising approach to mitigate CO2 emissions and produce useful carbon-based fuels.2
Photocatalytic CO2 reduction mimics natural photosynthesis by harnessing solar energy to convert CO2 into valuable chemical feedstocks. However, the high bond dissociation energy of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in CO2 makes this process energy-intensive, especially when aiming to produce energy-rich products such as CO, HCOOH, HCHO, CH3OH, and CH4.3 Moreover, CO2 reduction involves multiple proton-coupled electron transfer steps, which complicates the development of efficient photocatalysts.4 Among the various reduction pathways, the selective conversion of CO2 to CO is particularly appealing due to its relevance in syngas production – a two-electron reduction process. Additionally, CO serves as an essential intermediate for synthesizing higher-order products such as CH3OH, C2H5OH, and other valuable hydrocarbons using the well-established Fischer–Tropsch process.5
O bond in CO2 makes this process energy-intensive, especially when aiming to produce energy-rich products such as CO, HCOOH, HCHO, CH3OH, and CH4.3 Moreover, CO2 reduction involves multiple proton-coupled electron transfer steps, which complicates the development of efficient photocatalysts.4 Among the various reduction pathways, the selective conversion of CO2 to CO is particularly appealing due to its relevance in syngas production – a two-electron reduction process. Additionally, CO serves as an essential intermediate for synthesizing higher-order products such as CH3OH, C2H5OH, and other valuable hydrocarbons using the well-established Fischer–Tropsch process.5
Among the various heterogeneous photocatalytic materials,6 porous organic polymers (POPs)7 have emerged as a promising class of porous materials with tuneable structures, high surface areas, and excellent chemical stability. In the context of photocatalysis, POPs offer several advantages, such as robust chemical frameworks, extended π-conjugated networks, and permanent porosity, which are important for efficient heterogeneous photocatalytic CO2 reduction.8 The incorporation of transition metal atoms, such as Nickel, within the POP backbone can significantly enhance the catalytic activity by acting as a redox-active centre as well as CO2 binding sites, promoting selective CO2 reduction pathways.9
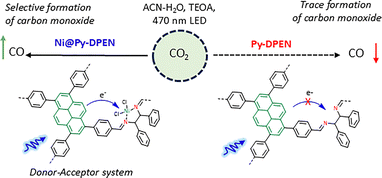
In this study, we report the synthesis and characterization of a pyrene-based π-conjugated POP (Py-DPEN) and investigated how the incorporation of Ni(II) in the organic framework enhances the photocatalytic activity for selective photoreduction of carbon dioxide (Fig. 1). We presumed that the synergistic electron transfer between the light-harvesting pyrene-based organic framework (donor) and the redox-active Ni centres (acceptor) effectively couples the catalytic cycles.10
The structural characteristics of Py-DPEN and Ni@Py-DPEN are explored using a combination of spectroscopic and microscopic techniques, while photocatalytic performance is assessed through controlled CO2 reduction experiments. 13C CP-MAS NMR spectra confirmed the formation of the imine bonds in the materials. The signal corresponding to the (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) is at 160.9 ppm (Fig. S2). The crystalline structure and ordered nature of Py-DPEN and Ni@Py-DPEN were confirmed by powder X-ray diffraction (PXRD) analysis (Fig. S3). The incorporation of Ni(II) centres into Py-DPEN was confirmed by comparing the IR spectra of Py-DPEN and Ni@Py-DPEN. A significant red shift in the C
N) is at 160.9 ppm (Fig. S2). The crystalline structure and ordered nature of Py-DPEN and Ni@Py-DPEN were confirmed by powder X-ray diffraction (PXRD) analysis (Fig. S3). The incorporation of Ni(II) centres into Py-DPEN was confirmed by comparing the IR spectra of Py-DPEN and Ni@Py-DPEN. A significant red shift in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching frequency was observed in the IR spectrum of Ni@Py-DPEN, appearing at 1574 cm−1, compared to 1614 cm−1 for Py-DPEN (Fig. 2a). Apart from this shift, the IR spectrum of Ni@Py-DPEN showed no major changes relative to Py-DPEN, indicating that the backbone structure was retained during the post-metalation process. The Brunauer–Emmett–Teller (BET) surface area calculated for the Py-DPEN is 328 m2 g−1 and that for Ni@Py-DPEN is 164 m2 g−1 (Fig. S5). Transmission electron microscopy (TEM) and scanning electron microscopy (SEM) images confirmed the presence of microrod and clustered structures in the Ni@Py-DPEN (Fig. 2c and d). Moreover, these images indicated that no metal oxides or nanoparticles formed during the incorporation of NiCl2 into the POP framework. Energy-dispersive X-ray (EDX) mapping, based on both TEM and SEM, further demonstrated a uniform distribution of Ni throughout the Ni@Py-DPEN POP matrix (Fig. S9 and 10). There is a large decrease in charge-transfer resistance (Rct) in Ni@Py-DPEN (28 kohm) compared to Py-DPEN (45 kohm), confirming efficient electron transfer in Ni@Py-DPEN, and upon light excitation the Rct decreases further due to facile electron transfer in the excited state (Fig. 2i). Additionally, we observed very high transient photocurrent for Ni@Py-DPEN (1.1 µA) compared to Py-DPEN (0.3 µA) (Fig. 2j).
N stretching frequency was observed in the IR spectrum of Ni@Py-DPEN, appearing at 1574 cm−1, compared to 1614 cm−1 for Py-DPEN (Fig. 2a). Apart from this shift, the IR spectrum of Ni@Py-DPEN showed no major changes relative to Py-DPEN, indicating that the backbone structure was retained during the post-metalation process. The Brunauer–Emmett–Teller (BET) surface area calculated for the Py-DPEN is 328 m2 g−1 and that for Ni@Py-DPEN is 164 m2 g−1 (Fig. S5). Transmission electron microscopy (TEM) and scanning electron microscopy (SEM) images confirmed the presence of microrod and clustered structures in the Ni@Py-DPEN (Fig. 2c and d). Moreover, these images indicated that no metal oxides or nanoparticles formed during the incorporation of NiCl2 into the POP framework. Energy-dispersive X-ray (EDX) mapping, based on both TEM and SEM, further demonstrated a uniform distribution of Ni throughout the Ni@Py-DPEN POP matrix (Fig. S9 and 10). There is a large decrease in charge-transfer resistance (Rct) in Ni@Py-DPEN (28 kohm) compared to Py-DPEN (45 kohm), confirming efficient electron transfer in Ni@Py-DPEN, and upon light excitation the Rct decreases further due to facile electron transfer in the excited state (Fig. 2i). Additionally, we observed very high transient photocurrent for Ni@Py-DPEN (1.1 µA) compared to Py-DPEN (0.3 µA) (Fig. 2j).
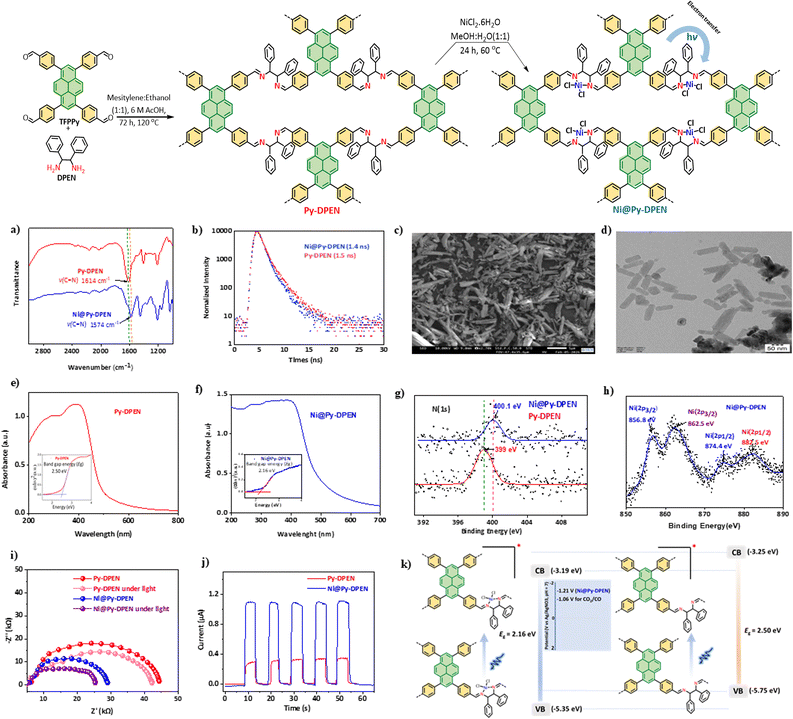 | ||
| Fig. 2 (a) IR spectra of Py-DPEN and Ni@Py-DPEN; (b) lifetime of Py-DPEN and Ni@Py-DPEN; (c) SEM image of Ni@Py-DPEN; (d) TEM image of Ni@Py-DPEN; (e) UV-Vis (Tauc plot) of Py-DPEN; (f) UV-Vis (Tauc plot) of Ni@Py-DPEN; (g) comparative N(1s) XPS analysis of Py-DPEN and Ni@Py-DPEN (390 eV to 414 eV region); (h) Ni(2p) XPS analysis of Ni@Py-DPEN (850 eV to 890 eV region); (i) EIS Nyquist plot of Py-DPEN and Ni@Py-DPEN in the presence and absence of light; (j) transient photocurrent responses of Py-DPEN and Ni@Py-DPEN; (k) the light absorbing ability of Py-DPEN and Ni@Py-DPEN and arrangements of the valence band (VB) and conduction band (CB); standard reduction potentials of Ni@Py-DPEN and CO2/CO vs Ag/AgNO3.2b | ||
X-ray photoelectron spectroscopy (XPS) analysis confirmed the successful incorporation of Ni(II) into the Py-DPEN framework. Ni@Py-DPEN exhibited characteristic Ni 2p3/2 and Ni 2p1/2 peaks at 856.8 and 874.4 eV, along with satellite peaks at 862.5 and 880.9 eV, consistent with the Ni(II) oxidation state and with no evidence of Ni(0) or NiO impurities (Fig. 2h). Comparison of the high-resolution N1s spectra showed a positive shift from 399.1 to 400.1 eV upon metalation (Fig. 2g), indicating coordination of Ni(II) via imine nitrogen atoms, while spectral deconvolution supported the expected nitrogen environments (Fig. S8).11 These results confirm selective Ni(II) coordination without structural alteration or extraneous metal species. Inductively coupled plasma-optical emission spectrometry (ICP-OES) analysis indicated that 2.64 wt% of Ni content was present in the Ni@Py-DPEN POP and only 0.08% leached after the photocatalytic run. TGA further demonstrated excellent thermal stability of both Py-DPEN and Ni@Py-DPEN up to ∼300 °C after NiCl2 incorporation (Fig. S4).
The UV-Vis spectrum of Ni@Py-DPEN POP showed a broad absorption from 370–500 nm, indicating strong visible-light harvesting (Fig. 2f). The red shift relative to the solid-state UV-vis spectra of TFPPy (donor) and Ni@DPEN (acceptor) (Fig. S7) confirms the formation of a donor–acceptor complex. The Tauc plot (Fig. 2f inset) revealed a reduced band gap of 2.16 eV for Ni@Py-DPEN compared to 2.50 eV for Py-DPEN (Fig. 2e), demonstrating that incorporating the Ni(II) centre enhances electron delocalization and lowers the band gap, improving photocatalytic efficiency. The photoluminescence spectrum reveals that, upon excitation at 390 nm, Ni@Py-DPEN POP exhibits an emission peak at 440 nm (Fig. S6). Time-resolved emission measurements indicate an average excited-state lifetime of 1.4 ns for Ni@Py-DPEN POP, compared to 1.5 ns for Py-DPEN POP (Fig. 2b). This slight reduction in lifetime from Py-DPEN POP to Ni@Py-DPEN POP provides clear evidence of efficient energy or electron transfer from the POP backbone to the Ni centre.
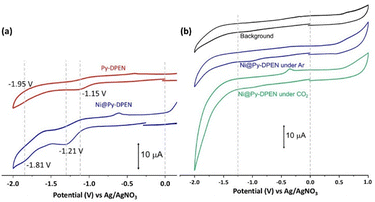
The electrochemical behaviour of Ni@Py-DPEN was examined by cyclic voltammetry (CV) in CH3CN using a glassy carbon working electrode, a platinum counter, and an Ag/AgNO3 reference electrode (Table 1). Two irreversible redox waves appeared at −1.21 V and −1.81 V vs Ag/AgNO3 under Ar (Fig. 3a). In comparison, the Py-DPEN-modified electrode showed similar irreversible waves at −1.15 V and −1.95 V, indicating that the cathodic redox processes originate from the ligand sites. In CH3CN/H2O (90/10, v/v), both electrodes exhibited broad irreversible waves near −1.05 V under Ar (Fig. S11). Upon CO2 saturation (20 min sparging), Ni@Py-DPEN displayed a strong catalytic current starting at −1.25 V (Fig. 3b), whereas Py-DPEN POP showed no catalytic response up to −2.0 V (Fig. S12). These results indicate that the Ni(II) centres in Ni@Py-DPEN POP are responsible for CO2 reduction.
| Catalyst | Reduction potential (Ered) | Conduction band energy [ECB = −e(Ered + 4.40)] (eV) | Band gap energy (Eg) (eV) | Valence band energy [EVB = ECB − Eg] (eV) |
|---|---|---|---|---|
| a E vs. Ag/AgNO3 (10 mM) measured in CH3CN containing 0.1 M TBAPF6. | ||||
| Ni@Py-DPEN | −1.21, −1.81 | −3.19 | 2.16 | −5.35 |
| Py-DPEN | −1.15, −1.95 | −3.25 | 2.50 | −5.75 |
As a typical run, a CH3CN–H2O–TEOA (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v) solution containing Ni@Py-DPEN (or Py-DPEN) (3 mg) was irradiated at λex = 470 nm (light power = 70 W m−2) under a CO2 atmosphere (Table 2). The main product was CO with CH4 formation to a lesser degree in either solution (the selectivity of CO was found to be 89.4% and 84.2%, respectively), and HCOOH and H2 were not detected (Table S1). The photochemical results clearly indicate the importance of the Ni centre in the POP material for CO2 reduction. Additionally, we have tested photochemical CO2 reduction using the Ni@Py-DPEN catalyst (3 mg), in CH3CN–TEOA (4
2 v/v) solution containing Ni@Py-DPEN (or Py-DPEN) (3 mg) was irradiated at λex = 470 nm (light power = 70 W m−2) under a CO2 atmosphere (Table 2). The main product was CO with CH4 formation to a lesser degree in either solution (the selectivity of CO was found to be 89.4% and 84.2%, respectively), and HCOOH and H2 were not detected (Table S1). The photochemical results clearly indicate the importance of the Ni centre in the POP material for CO2 reduction. Additionally, we have tested photochemical CO2 reduction using the Ni@Py-DPEN catalyst (3 mg), in CH3CN–TEOA (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) solution under similar conditions with CO (8.19 µmol) and CH4 (1.17 µmol) (Table 2; as well as Table S1). Interestingly, the yields of CO and CH4 were much less in CH3CN–TEOA (4
1 v/v) solution under similar conditions with CO (8.19 µmol) and CH4 (1.17 µmol) (Table 2; as well as Table S1). Interestingly, the yields of CO and CH4 were much less in CH3CN–TEOA (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) than that of CH3CN–H2O–TEOA (9
1 v/v) than that of CH3CN–H2O–TEOA (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v). The availability of the proton source in CH3CN–H2O–TEOA solution might be the main reason for the high product formation.9 The apparent quantum yield of CO was found to be ΦCO = 1.98% and 0.426% with Ni@Py-DPEN POP in CH3CN–TEOA–H2O and CH3CN–TEOA, respectively (see the SI for details).
2 v/v). The availability of the proton source in CH3CN–H2O–TEOA solution might be the main reason for the high product formation.9 The apparent quantum yield of CO was found to be ΦCO = 1.98% and 0.426% with Ni@Py-DPEN POP in CH3CN–TEOA–H2O and CH3CN–TEOA, respectively (see the SI for details).
| Entry | Product | Ni@Py-DPEN (µmol) | Py-DPEN (µmol) | Ni@Py-DPENb (µmol) |
|---|---|---|---|---|
a Irradiated with a blue LED at λex = 470 nm (light power = 70 W m−2). Formation of CO and CH4, during the photoirradiation of a CO2-saturated solution containing 3 mg of POP in CH3CN–H2O–TEOA (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v, 4.0 mL).b Ni@Py-DPEN (3 mg) in CH3CN–TEOA (4 2 v/v, 4.0 mL).b Ni@Py-DPEN (3 mg) in CH3CN–TEOA (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v, 4.0 mL) at 298 K. HCOOH and H2 were not detected for all reactions.c In an inert atmosphere (under N2). 1 v/v, 4.0 mL) at 298 K. HCOOH and H2 were not detected for all reactions.c In an inert atmosphere (under N2). |
||||
| 1 | CO | 38.0 | 8.90 | 8.19 |
| 2 | CH4 | 4.5 | 1.66 | 1.17 |
| 3c | CO | 0 | — | — |
Based on the above experimental evidence, a possible reaction mechanism has been proposed (Fig. S16). The reductive quenching of the excited state of the pyrene part of Ni@Py-DPEN POP by TEOA should (ETEOA/TEOA+˙ = +0.34 V)12 allow smooth transfer of an electron from Pyrene−˙ to the Ni(II) catalyst site, converting Ni(II) into Ni(I). The Ni(I) might bind with CO2 and further one electron addition followed by one proton addition should induce CO2 reduction via the proton coupled electron transfer pathway (PCET).13 After CO2 reduction occurs, the Ni(II) catalyst site returns to its Ni(II) state, completing the cycle. The binding and activation of CO2 by Ni has been well studied by DFT calculation.14
We report a donor–acceptor imine-based metallo-porous organic polymer (POP) for visible-light-driven CO2 reduction with good selectivity. The conjugated framework minimizes the band gap upon Ni(II) incorporation, enhancing charge separation and enabling efficient charge-transfer-driven catalysis. In this architecture, the pyrene derivative acts as the light-harvesting material, while Ni@DPEN functions as the redox-active acceptor; their spatial integration within the POP pores promotes rapid excited-state electron transfer to the Ni sites in the presence of triethanolamine as a sacrificial electron donor. This synergistic design drives multielectron CO2 to CO conversion with 89.4% selectivity, delivering 38 µmol of CO in 6 h using triethanolamine in an acetonitrile–water mixture as the electron donor. Electrochemical studies confirm the redox behaviour of Ni@Py-DPEN POP, as well as its effectiveness towards CO2 photoreduction.
AD and DG designed the project. AJD and NMG performed experiments. AD, AP, and DG wrote the manuscript. AD acquired the funding and directed the research.
Conflicts of interest
There are no conflicts to declare.Data availability
The supplementary information (SI) includes all experimental details, including the synthesis and characterization of the materials reported in this study. See DOI: https://doi.org/10.1039/d6cc00397d.Acknowledgements
A. J. D. and H. I. acknowledge the Department of Chemistry, SRM institute of Science and Technology, Kattankulathur, Tamil Nadu, India, for financial support through an Institute Doctoral Fellowship. A. D. sincerely acknowledges SRMIST for the seed grant (No. SRMIST/R/AR(A/SERI2024/174/14–342), the Anusandhan National Research Foundation (ANRF-ECRG, Grant No. ANRF/ECRG/2024/003299/CS), and the RSC-Newton International Alumni Fellowship (AL\24100051) for the financial support. D. G. sincerely thanks the ANRF (Grant No. SUR/2022/000359) for financial support. We also thank the SRM Central Instrumentation Facility (SRM-SCIF) and the Nano Research Centre (SRM-NRC) for providing access to instrumentation facilities.References
- (a) K. S. Song, P. W. Fritz and A. Coskun, Chem. Soc. Rev., 2022, 51, 9831–9852 RSC; (b) J. Low, B. Cheng and J. Yu, Appl. Surf. Sci., 2017, 392, 658–686 CrossRef CAS; (c) H. A. Patel and C. T. Yavuz, Faraday Discuss., 2015, 183, 401–412 RSC; (d) M. Zhou, Z. Wang, A. Mei, Z. Yang, W. Chen, S. Ou, S. Wang, K. Chen, P. Reiss, K. Qi, J. Ma and Y. Liu, Nat. Commun., 2023, 14, 2473 CrossRef CAS PubMed; (e) S. Zhu, X. Li, X. Jiao, W. Shao, L. Li, X. Zu, J. Hu, J. Zhu, W. Yan, C. Wang, Y. Sun and Y. Xie, Nano Lett., 2021, 21, 2324–2331 CrossRef CAS PubMed; (f) K. Sun, Y. Qian and H.-L. Jiang, Angew. Chem., Int. Ed., 2023, 62, e202217565 CrossRef CAS PubMed; (g) P. Verma, F. A. Rahimi, D. Samanta, A. Kundu, J. Dasgupta and T. K. Maji, Angew. Chem., Int. Ed., 2022, 61, e202116094 CrossRef CAS PubMed; (h) K. Khaletskaya, A. Pougin, R. Medishetty, C. Rösler, C. Wiktor, J. Strunk and R. A. Fischer, Chem. Mater., 2015, 27, 7248–7257 CrossRef CAS; (i) S. Karmakar, S. Barman, F. A. Rahimi, D. Rambabu, S. Nath and T. K. Maji, Nat. Commun., 2023, 14, 4508 CrossRef CAS PubMed.
- (a) X. Yu, Z. Yang, B. Qiu, S. Guo, P. Yang, B. Yu, H. Zhang, Y. Zhao, X. Yang, B. Han and Z. Liu, Angew. Chem., Int. Ed., 2019, 58, 632–636 CrossRef CAS PubMed; (b) A. Singh, P. Verma, D. Samanta, A. Dey, J. Dey and T. K. Maji, J. Mater. Chem. A, 2021, 9, 5780–5786 RSC.
- (a) X. Yao, K. Chen, L.-Q. Qiu, Z.-W. Yang and L.-N. He, Chem. Mater., 2021, 33, 8863–8872 CrossRef CAS; (b) P. Verma, A. Singh, F. A. Rahimi, P. Sarkar, S. Nath, S. K. Pati and T. K. Maji, Nat. Commun., 2021, 12, 7313 CrossRef CAS PubMed; (c) K.-G. Liu, F. Bigdeli, A. Panjehpour, A. Larimi, A. Morsali, A. Dhakshinamoorthy and H. Garcia, Coord. Chem. Rev., 2023, 493, 215257 CrossRef CAS.
- Z. Chen, Y. Hu, J. Wang, Q. Shen, Y. Zhang, C. Ding, Y. Bai, G. Jiang, Z. Li and N. Gaponik, Chem. Mater., 2020, 32, 1517–1525 CrossRef CAS.
- (a) D. K. Chauhan, N. Sharma and K. Kailasam, Mater. Adv., 2022, 3, 5274–5298 RSC; (b) S. Wang, B. Y. Guan and X. W. D. Lou, J. Am. Chem. Soc., 2018, 140, 5037–5040 CrossRef CAS PubMed.
-
(a) S. Sun, M. Watanabe, J. Wu, Q. An and T. Ishihara, J. Am. Chem. Soc., 2018, 140, 6474–6482 CrossRef CAS PubMed;
(b) S. Neat
![[u with combining cedilla]](https://www.rsc.org/images/entities/char_0075_0327.gif) , J. A. Macia-Agullo, P. Concepcion and H. Garcia, J. Am. Chem. Soc., 2014, 136, 15969–15976 CrossRef PubMed;
(c) A. Bathla, J. Lee, S. A. Younis and K.-H. Kim, Mater. Today Chem., 2022, 24, 100870 CrossRef CAS.
, J. A. Macia-Agullo, P. Concepcion and H. Garcia, J. Am. Chem. Soc., 2014, 136, 15969–15976 CrossRef PubMed;
(c) A. Bathla, J. Lee, S. A. Younis and K.-H. Kim, Mater. Today Chem., 2022, 24, 100870 CrossRef CAS. - (a) R. Das, R. Paul, A. Parui, A. Shrotri, C. Atzori, K. A. Lomachenko, A. K. Singh, J. Mondal and S. C. Peter, J. Am. Chem. Soc., 2023, 145, 422–435 CrossRef CAS PubMed; (b) T. Kajiwara, M. Fujii, M. Tsujimoto, K. Kobayashi, M. Higuchi, K. Tanaka and S. Kitagawa, Angew. Chem., Int. Ed., 2016, 55, 2697–2700 CrossRef CAS PubMed; (c) Z. Zhong, X. Wang and B. Tan, Chem. – Eur. J., 2025, 31, e202404089 CrossRef CAS PubMed; (d) W.-J. Wang and K. Chen, Chin. Chem. Lett., 2025, 36, 109998 CrossRef CAS; (e) H. Ali, Y. Orooji, A. Y. A. Alzahrani, M. H. A. Mughram, A. M. Abu-Dief, I. Omar, A. Hayat, D. Yue and Y. Xu, Coord. Chem. Rev., 2025, 543, 216884 CrossRef CAS.
- (a) G. M. Eder, D. A. Pyles, E. R. Wolfson and P. L. McGrier, Chem. Commun., 2019, 55, 7195–7198 RSC; (b) C. Sarkar, R. Paul, D. Q. Dao, S. Xu, R. Chatterjee, S. C. Shit, A. Bhaumik and J. Mondal, ACS Appl. Mater. Interfaces, 2022, 14, 37620–37636 CrossRef CAS PubMed; (c) G. Mukherjee, J. Thote, H. B. Aiyappa, S. Kandambeth, S. Banerjee, K. Vanka and R. Banerjee, Chem. Commun., 2017, 53, 4461–4464 RSC; (d) Y. Ma, X. Yi, S. Wang, T. Li, B. Tan, C. Chen, T. Majima, E. R. Waclawik, H. Zhu and J. Wang, Nat. Commun., 2022, 13, 1400 CrossRef CAS PubMed.
- (a) K. Niu, Y. Xu, H. Wang, R. Ye, H. L. Xin, F. Lin, C. Tian, Y. Lum, K. C. Bustillo, M. M. Doeff, M. T. M. Koper, J. Ager, R. Xu and H. Zheng, Sci. Adv., 2017, 3, e1700921 CrossRef PubMed; (b) N. Zahir, V. Rajangam, S. S. Kalanur, S. I. Nikitenko and B. G. Pollet, Energy Environ. Mater., 2025, 8, e70014 CrossRef CAS.
- N. Xu, Y. Diao, X. Qin, Z. Xu, H. Ke and X. Zhu, Dalton Trans., 2020, 49, 15587 RSC.
- A. Jati, S. Dam, S. Kumar, K. Kumar and B. Maji, Chem. Sci., 2023, 14, 8624–8634 RSC.
- B. Kurpil, Y. Markushyna and A. Savateev, ACS Catal., 2019, 9, 1531–1538 CrossRef CAS.
- D. Ghosh, H. Takeda, D. C. Fabry, Y. Tamaki and O. Ishitani, ACS Sustainable Chem. Eng., 2019, 7, 2648–2657 CrossRef CAS.
- (a) S. Liang, X. Zhong, Z. Zhong, H. Deng and W.-Y. Wong, Appl. Catal., B, 2023, 337, 122958 CrossRef CAS; (b) J. Song, E. L. Klein, F. Neese and S. Ye, Inorg. Chem., 2014, 53, 7500–7507 CrossRef CAS PubMed; (c) P. Yadav, S. Kumar, N. Velankanni, T. D. Kühne, S. Gosavi, R. K. M. Raghupathy, R. Bhosale, G. Held, M. Shelke and S. Ogale, J. Phys. Energy, 2024, 6, 025019 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |