Open Access Article
Open Access ArticleCO2-based biodegradable plastics and their precursor production using photo-biocatalytic processes
Yu
Kita
a,
Mika
Takeuchi
b and
Yutaka
Amao
 *abc
*abc
aGraduate School of Science, Osaka City University, Sugimoto 3-3-138, Sumiyoshi-ku, Osaka 558-8585, Japan
bGraduate School of Science, Osaka Metropolitan University, Sugimoto 3-3-138, Sumiyoshi-ku, Osaka 558-8585, Japan
cResearch Centre for Artificial Photosynthesis, Osaka Metropolitan University, Sugimoto 3-3-138, Sumiyoshi-ku, Osaka 558-8585, Japan. E-mail: amao@omu.ac.jp
First published on 10th April 2026
Abstract
Biodegradable plastics are materials that can be decomposed by the action of microbes into water, CO2, and biomass. They are attractive materials to solve the issue of serious environmental pollution due to plastic disposal. Some biodegradable plastics are produced entirely from petroleum-based precursors. The production of new biodegradable plastics and their precursors from CO2 and persistent organic or bio-based compounds using visible-light driven redox with biocatalytic processes is one of the effective resolutions for the environmental issues of plastic pollution and global warming. In this review article, recent research on the visible-light driven production of CO2-based biodegradable plastics and their precursors using a system of light-driven redox and biocatalytic processes is introduced. As a first example, an overview of research into the production of biodegradable plastics, poly(hydroxybutyrate) (PHB) and their precursors such as acetate, shikimic acid, acetoin and so on from CO2 using a semiconductor photocatalyst-based photoredox system with a microbial cell as a biocatalytic process is provided. As a second example, an overview is provided of studies on the production of biodegradable plastic precursors, 3-hydroxybutyrate, L-malate and fumarate from CO2 and small organic molecules using an organic dye-based photoredox system with an electron donor, an electron mediator and enzyme-based biocatalytic processes.
Introduction
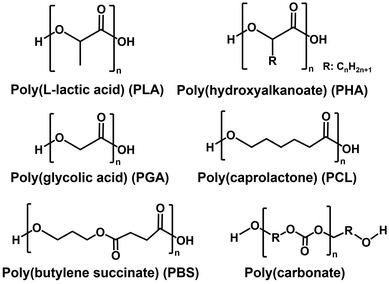
Plastic is a versatile, lightweight, and relatively inexpensive material that has been an indispensable part of modern life.1 Most plastics are made from fossil resources, primarily petroleum, and their entire manufacturing process lead to increasing greenhouse gas emissions.2 Plastics pose environmental pollution concerns throughout their life cycle, from production to use and ultimately disposal.3,4 However, plastics have a wide range of functions and are useful materials that support modern society, so it is necessary to establish methods for their production, use, and disposal without causing environmental pollution.5 Accordingly, biodegradable plastics are attracting attention as a material to replace conventional plastics.6–12 Biodegradable plastics and their derivative materials are broken down by microbes into water and CO2 within a period of time.13–17 Among the various biodegradable plastics, aliphatic polyester-based biodegradable plastics include poly(lactic acid) (PLA),18–21 poly(hydroxyalkanoate) (PHA),22–31 poly(glycolic acid) (PGA),32–35 poly(caprolactone) (PCL),36–40 poly(butylene succinate) (PBS),41–44 poly(carbonate)45–49 and so on, as shown in Fig. 1.The precursors of these biodegradable plastics are synthesised from various molecules, such as biological and petroleum-derived molecules. Therefore, it is required to establish precursor synthesis methods from renewable raw materials such as CO2 and biomass-derived molecules in the future. In natural photosynthesis, visible light energy is used as the driving force to produce glucose, starch, and oxygen using CO2 and water as raw materials.50–55 Thus, natural photosynthetic processes can serve as models for the CO2-based synthesis of polymers and their precursors. Natural photosynthesis consists of light-dependent reactions and dark reactions. The main components involved in the light-dependent reactions are the photosystem, the complexes that capture light energy, chlorophyll, the main light-absorbing pigments, the electron transport chain that transports electrons and contributes to the production of adenosine triphosphate (ATP) and the reduced form of nicotinamide adenine dinucleotide phosphate (NADPH), ATP synthase and ferredoxin NADP+ reductase. In summary, a light-dependent reaction is a process that converts absorbed visible-light energy into ATP and NADPH.
The dark reactions, collectively known as the Calvin cycle,56,57 do not directly require visible-light energy but utilize the ATP and NADPH produced during the light-dependent reactions to fix CO2 into organic compounds, such as ribulose-1,5-bisphosphate, and convert them into glucose through enzymatic processes. Biodegradable plastics and their precursors are synthesized starting from compounds synthesized from CO2 produced in the Calvin cycle using microalgae. It has been reported that D-lactate, a precursor for poly(lactic acid), was produced using malic enzyme (ME)-overexpressing strains of cyanobacterium Synechocystis sp. PCC6803, as shown in Fig. 2.58
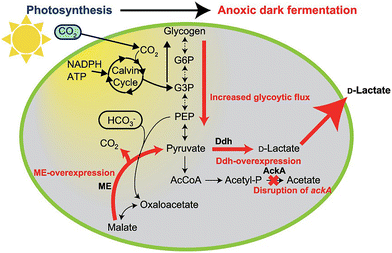 | ||
| Fig. 2 Light-driven D-lactate production from CO2 using Synechocystis sp. PCC6803. G6P: glucose-6-phosphate, G3P: glyceraldehyde 3-phosphate, PEP: phosphoenolpyruvic acid, ME: malic enzyme, Ddh: D-lactate dehydrogenase, AckA: acetate kinase A. Reproduced with permission from ref. 58. Copyright 2020, the American Chemical Society. | ||
A metabolic map of the TCA cycle in Synechocystis sp. PCC 6803 is shown in Fig. 3.59 As shown in Fig. 3, not only D-lactate but also various biodegradable plastic precursors, such as succinate, 3-hydroxybutyrate, and 3-hydroxypropionate, can be produced from CO2 under visible-light irradiation using Synechocystis sp. PCC 6803. Additionally, poly(hydroxybutyrate) (PHB) is produced from CO2 using Synechocystis sp. PCC 6803. The metabolic map in Fig. 3 shows that the production of acetyl Co-A from CO2 leads to the production of various biodegradable plastic precursors. It will also be possible to produce biodegradable plastic precursors, such as fumarate and malate.
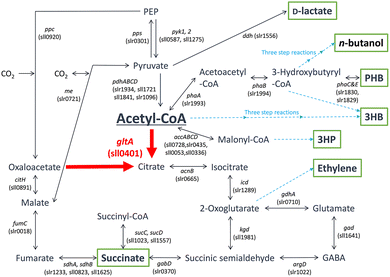 | ||
| Fig. 3 Metabolic map of the TCA cycle in Synechocystis sp. PCC 6803. accABCD: acetyl-CoA carboxylase, acnB: aconitase, argD: D-arginine dehydrogenase, citH, malate dehydrogenase, ddh: D-lactate dehydrogenase, fumC: fumarase, gabD: succinate-semialdehyde dehydrogenase, gad: glutamate decarboxylase, gdhA: glutamate dehydrogenase, gltA: citrate synthase, icd: isocitrate dehydrogenase, kgd: 2-oxoglutarate decarboxylase, me: malic enzyme, pdhABCD: pyruvate dehydrogenase, phaA: β-ketothiolase, phaB: acetoacetyl-CoA reductase, phaC&E: polyhydroxyalkane synthase, ppc: phosphoenolpyruvate carboxylase, pps: phosphoenolpyruvate synthase, pyk1, 2: pyruvate kinase, sdhA, sdhB: succinate dehydrogenase, sucC, sucD: succinate dehydrogenase. A. Reproduced with permission from ref. 59. Copyright 2019, The authors Creative Commons CC-BY-NC-ND license. | ||
In this way, by linking the TCA cycle and glycolysis with the photosynthetic process, it is possible to synthesize biodegradable plastics and their precursors from CO2 driven by light energy.
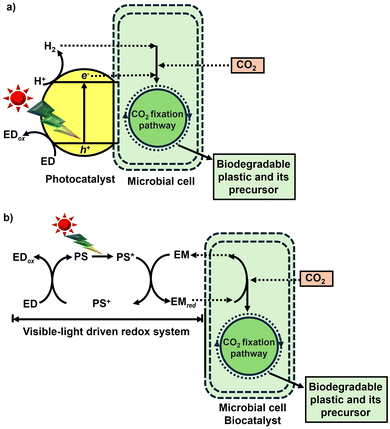
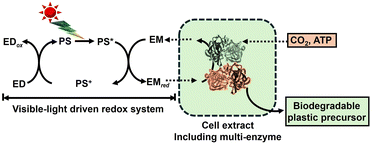
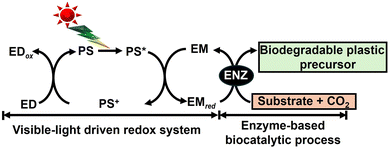
To develop a visible-light driven process for the production of CO2-based biodegradable plastics and their precursors, based on the mechanisms of natural photosynthesis and microalgae, a method of linking light-driven redox (light-dependent reaction) with reactions mediated by biocatalytic processes such as microbials or enzymes (dark reaction) has been envisaged, as shown in Fig. 4.
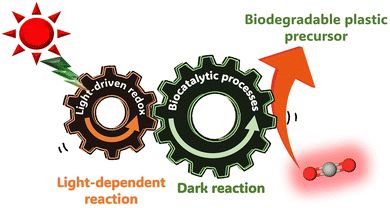 | ||
| Fig. 4 CO2-based biodegradable plastics and their precursor production using systems comprising visible-light-driven redox reactions and biocatalytic processes. | ||
Using semiconductor photocatalysts and organic dyes in a light-driven redox system, light-harvesting and sensitizing functions can be enhanced compared to natural photosynthesis, such as that in microalgae. Biocatalytic processes use microorganisms directly or through cascades of enzymes to produce biodegradable plastics and their precursors from CO2. By linking these two processes, it is expected that biodegradable plastics and their precursors can be produced by fixing CO2 to organic molecules using visible-light energy as a driving force.
In this review article, recent research on the visible-light-driven production of CO2-based biodegradable plastics and their precursors with a system comprising light-driven redox and biocatalytic processes is introduced. First, research on the production of biodegradable plastics and their precursors using semiconductor photocatalysts as photoredox and microbials as biocatalytic processes is introduced. Second, research on the production of biodegradable plastic precursors using organic dye-based photoredox and enzyme-based biocatalytic processes is introduced.
Strategy for the visible-light-driven production of CO2-based biodegradable plastics and their precursors with a photocatalyst and microbial cell hybrid system
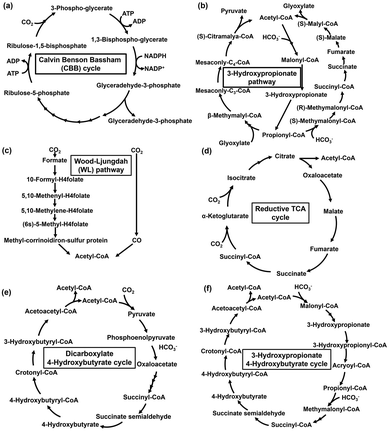
Semiconductor-based photocatalysts are highly efficient at capturing and converting solar energy compared to the light-harvesting function of natural photosynthesis. Meanwhile, natural CO2 fixation, primarily by plants and microorganisms, is the basis of life and biological processes. By directly contacting semiconductor-based photocatalysts with microbial cells expressing a CO2 fixation pathway, as shown in Fig. 5(a), light-driven biodegradable plastics and their precursors can be produced. Additionally, a system for biodegradable plastics and their precursor production from CO2 using microbial cells and extract containing enzymes as the biocatalyst in a visible light-driven redox system, consisting of an electron donor (ED), a visible light sensitizer (PS), and an electron mediator (EM), has been proposed, as shown in Fig. 5(b).In the system shown in Fig. 5(a) and (b), a visible light-responsive photocatalyst capable of decomposing water into oxygen and hydrogen is used.60–65 However, the CO2 fixation pathway within microbial cells for the production of biodegradable plastics and their precursors is shown in Fig. 6. As shown in Fig. 6, natural CO2 fixation pathways are classified into the following six categories: Calvin Benson Bassham (CBB) cycle,66 3-hydroxypropionate pathway,67 reductive acetyl-CoA (Wood-Ljungdahl (WL) pathway) pathway,68 reductive TCA cycle,69 dicarboxylate/4-hydroxybutyrate cycle,70 and 3-hydroxypropionate/4-hydroxybutyrate cycle.71 In particular, the reductive acetyl-CoA pathway, known as the Wood-Ljungdahl (WL) pathway, is expressed in Moorella thermoacetica (M. thermoacetica) and is used to produce acetate from CO272–75 combined with a photocatalyst.
Visible-light-driven acetate production from CO2 with a photocatalyst and microbial cell hybrid system
First, a production system for acetate, a precursor of biodegradable plastics, from CO2 using a combination of photocatalysts is introduced.Visible-light-driven acetate production from CO2 using the hybridisation of CdS nanoparticles and M. thermoacetica has been accomplished. Fig. 7(A) shows a schematic representation of acetate production from CO2 using a hybrid system comprising CdS nanoparticles and M. thermoacetica (ATCC 39073) under visible-light irradiation.76
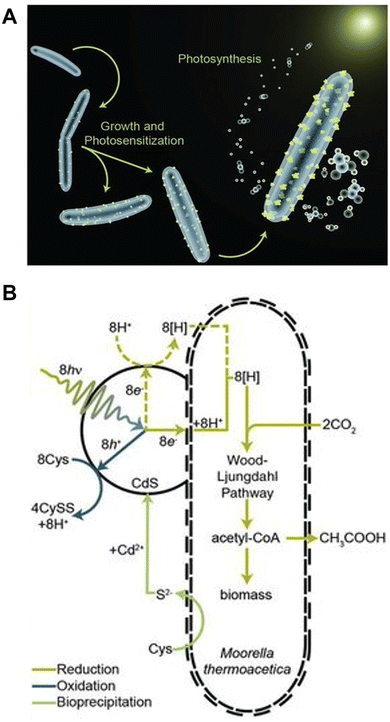 | ||
| Fig. 7 Image of the visible-light-driven acetate production from CO2 using a hybrid system comprising CdS nanoparticles and M. thermoacetica (A). Mechanism of visible-light-driven acetate production from CO2 using CdS nanoparticles and M. thermoacetica in the presence of Cys (B). Reproduced with permission from ref. 76. Copyright 2016, the American Association for the Advancement of Science. | ||
In this system, acetate can be produced from CO2 using the photosensitizing function of CdS and the WL pathway in M. thermoacetica. In this system, CdS is constantly regenerated by incorporating cysteine (Cys) into the reaction. Visible-light-driven acetate production by M. thermoacetica and CdS is a two-step, one-pot synthesis, as shown in Fig. 7(B). First, CdS precipitation by M. thermoacetica is induced by adding Cd2+ and cysteine as sulfur sources.77M. thermoacetica performs acetate production using electrons generated from irradiated CdS nanoparticles, as shown in Fig. 7(B).
The Cys quenches h+ of CdS, producing the oxidized disulfide form of cystine (CySS). Overall, this process is shown in Fig. 8.
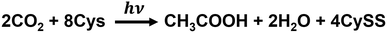 | ||
| Fig. 8 Overall process for the visible-light-driven acetate production from CO2 using the hybrid system of CdS–M. thermoacetica in the presence of Cys. | ||
Fig. 9 shows the time dependence of acetate production using a hybrid system of CdS–M. thermoacetica in the presence of Cys. The irradiation source is an in-house fabricated circular LED array composed of 405 ± 5 nm violet LEDs with a measured photon flux of 5.5 × 1018 cm−2 s−1. As shown in Fig. 9, ca. 1.3 mM of acetate production is observed using a hybrid system of CdS–M. thermoacetica under visible-light irradiation. However, no acetate production is observed using M. thermoacetica under only visible-light irradiation. Moreover, no acetate production is observed using a hybrid system of CdS-sterilised M. thermoacetica under visible-light irradiation. Additionally, a decrease in the acetate concentration is observed under dark conditions. A maximum yield of ca. 90% acetate based on the initial Cys concentration is accomplished using a hybrid system of CdS–M. thermoacetica under visible-light irradiation. After 12 h of irradiation, 1.43 mM of acetate is produced.
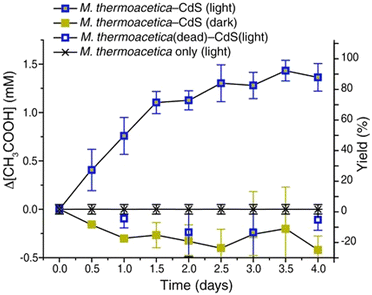 | ||
| Fig. 9 Time dependence of acetate production from CO2 using the hybrid system of CdS–M. thermoacetica in the presence of Cys under visible-light irradiation and dark conditions. Reproduced with permission from ref. 76. Copyright 2016, the American Association for the Advancement of Science. | ||
Visible-light-driven acetate production using CO2 as a raw material has also been reported using a hybrid system comprising gold nanoparticles (AuNPs) and M. thermoacetica.78 Using this system, acetate production is estimated to be 6.01 mmol g−1 per week.
A tandem inorganic–biological hybrid system consisting of a TiO2 nanoparticle loaded with a manganese(II) phthalocyanine (MnPc) cocatalyst, CdS and M. thermoacetica capable of oxygenic photosynthesis of acetate from CO2 has been reported.79Fig. 10(A) shows a schematic representation of acetate production from CO2 using a hybridisation system comprising CdS nanoparticles and M. thermoacetica under visible-light irradiation.
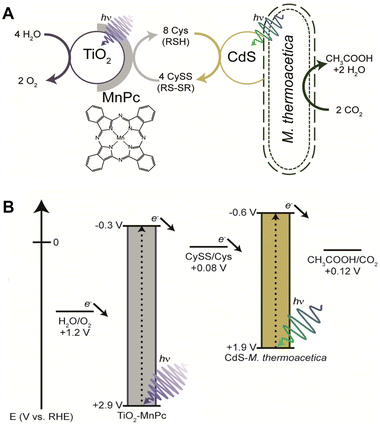 | ||
| Fig. 10 Visible-light-driven acetate production from CO2 using a hybrid system comprising TiO2 nanoparticles loaded with MnPc, CdS nanoparticles and M. thermoacetica (A). Energy level diagram depicting the relative alignment of the energies of TiO2 and CdS with the relevant redox processes (B). Reproduced with permission from ref. 79. Copyright 2016, American Chemical Society. | ||
A feature of this system is that the photooxidation catalyst TiO2 is loaded with the cocatalyst MnPc to regenerate the CySS/Cys redox shuttle, thereby coupling water oxidation with CySS reduction. Fig. 10(B) shows the time dependence of acetate production using the tandem system of TiO2 nanoparticles loaded with MnPc, CdS and M. thermoacetica in the presence of Cys. The irradiation source is a filtered 75 W Xenon lamp (AM1.5G, 5% Sun).
As shown in Fig. 11, comparing CdS–M. thermoacetica and MnPc-loaded TiO2 to the control, the combined tandem system shows increased acetate production. Both CdS–M. thermoacetica alone and the MnPc-loaded TiO2–M. thermoacetica control shows reduced acetate production below the stoichiometric limit imposed by Cys. Without MnPc, the TiO2-only tandem system performed poorly. A comparison of the cumulative electron equivalents of acetate (8e−) and O2 (4e−) measured over 3.5 days shows a strong correlation between the reduced product, acetate and the oxidized product, O2. Using this system, 1.2 mM of acetate is produced after 1 day.
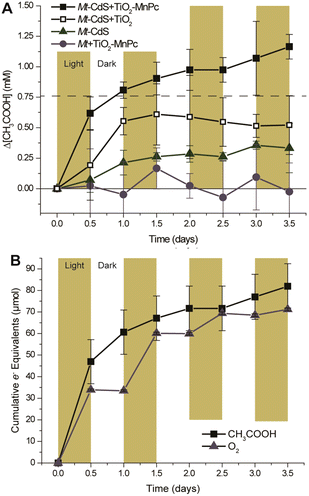 | ||
| Fig. 11 Time dependence of acetate production from CO2 with a tandem system comprising TiO2 nanoparticles loaded with MnPc, CdS and M. thermoacetica in the presence of Cys under visible-light irradiation (■) (A). □: Tandem system comprising TiO2 nanoparticles, CdS and M. thermoacetica, ▲: hybrid system comprising CdS and M. thermoacetica, ●: hybrid system of TiO2 nanoparticles loaded with MnPc and M. thermoacetica. Time dependence of the amount of acetate and oxygen produced using a tandem system of TiO2 nanoparticles loaded with MnPc, CdS and M. thermoacetica in the presence of Cys under visible-light irradiation (B). Reproduced with permission from ref. 79. Copyright 2016, the American Chemical Society. | ||
Visible-light-driven acetate production from CO2 has been reported in which perylene diimide derivatives (PDI) and poly(fluorene-co-phenylene) (PFP) are applied to the M. thermoacetica surface as photosensitizers to form a p–n heterojunction (PFP/PDI) layer, as shown in Fig. 12(A).80
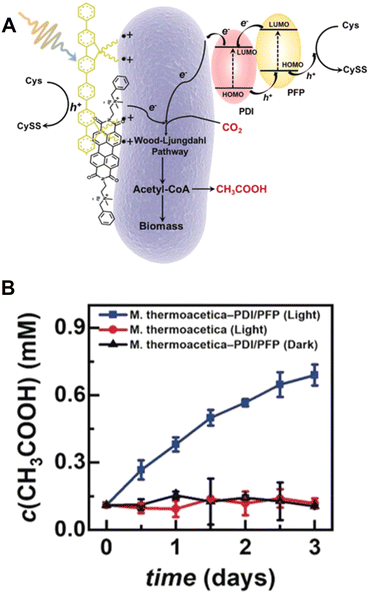 | ||
| Fig. 12 Visible-light-driven acetate production from CO2 using the PDI/PFP/M. thermoacetica hybrid system (A). Time-dependent acetate production from CO2 with the PDI/PFP/M. thermoacetica hybrid system in the presence of Cys under visible-light irradiation (■). ■: Only M. thermoacetica, ▲: dark condition (B). Reproduced with permission from ref. 80. Copyright 2020 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
The accumulated acetate yield of PDI/PFP/M. thermoacetica (ca. 0.63 mM) is significantly higher than that of PDI/M. thermoacetica (ca. 0.25 mM) or PFP/M. thermoacetica (ca. 0.4 mM) under irradiation using a Xenon fiber optic lamp with filters larger than 420 nm. Fig. 12(B) shows the time dependence of the acetate produced using PDI/PFP/M. thermoacetica in an alternating light–dark cycle of 12 h each.
After 3 days of incubation with irradiation, the accumulation amount of the acetate is estimated to be 0.63 mM. The quantum yield of 1.6% is calculated based on the initial Cys concentration. Here, the quantum yield is calculated using eqn (1) (n: number of electrons, C: total acetate concentration, V: total suspension volume, ϕ (cm−2 s−1): photo flux, A: area of illumination, t: reaction time, and NA: Avogadro's number). Acetate is produced by the 8-electron reduction of CO2; therefore, the n value is 8.
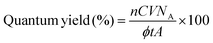 | (1) |
Sporomusa ovata (S. ovata) is a type of bacterium characterized by banana-shaped cells. S. ovata cells are strictly anaerobic, Gram-negative, spore-forming, straight or slightly curved rods that are motile by lateral flagella.81 There has been much research on the electrosynthesis of energy-containing carbon chains using S. ovata.82,83S. ovata is a candidate biological catalyst that converts sunlight, water, and CO2 into oxygen and liquid fuel or variable chemicals.
Visible-light-driven acetate production from CO2 using the hybridisation of CdS nanoparticles and S. ovata (CdS–S. ovata) has also been accomplished. Fig. 13 (A) shows a schematic representation of acetate production from CO2 using CdS–S. ovata under visible-light irradiation.84
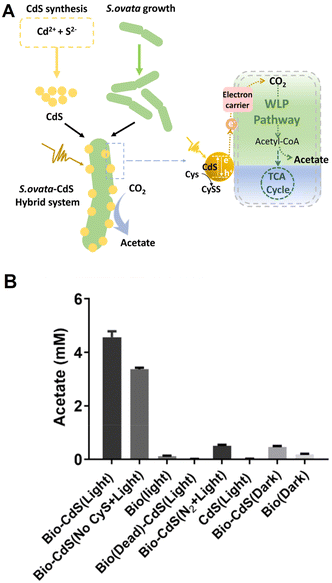 | ||
| Fig. 13 Schematic of the visible-light-driven acetate production from CO2 using CdS–S. ovata (A). Acetate production using CdS–S. ovata after 1 day of light irradiation in the presence of Cys. Bio-CdS (No CyS + Light): in the absence of Cys, Bio (light): only S. ovata, Bio(Dead)-CdS (Light): sterilized S. ovata and CdS hybrid system, Bio-CdS (N2 + Light): without CO2 gas, CdS (Light): only CdS. Dark: under dark conditions (B). Reproduced with permission from ref. 84. Copyright 2020, the American Chemical Society. | ||
In this system, photoelectrons are generated by CdS under light irradiation and transferred to S. ovata to achieve acetate production using CO2 as a raw material via the WL pathway. In this system, it is found that formate is produced as an intermediate and then is rapidly converted to the final product, acetate. Fig. 13(B) shows the visible-light-driven acetate production using CdS–S. ovata after 1 day of light irradiation and the controls in the presence of Cys. The intensity of light (400 ± 5 nm) is estimated to be 0.20 mW cm−2. Each experiment is pressurized with 2.0 atm, 80% N2 and 20% CO2. In this figure, Bio-CdS means CdS–S. ovata. As shown in Fig. 13(B), a high concentration of acetate is observed using CdS–S. ovata during visible-light irradiation in contrast to the other systems. Using CdS–S. ovata, the quantum yield reaches 16.8% ± 9%, and the active time of the system can last for 5 days. The quantum yield is also estimated using eqn (1).
Visible-light-driven acetate production from CO2 by the hybridization of a Z-scheme semiconductor photocatalytic system with S. ovata has been reported, as shown in Fig. 14(A).85 In this system, the Z-scheme consists of particulate semiconductors La and Rh co-doped SrTiO3 (SrTiO3: La, Rh) and Mo-doped BiVO4 (BiVO4: Mo). In this system, photocatalytic particles are immobilised onto a sheet.
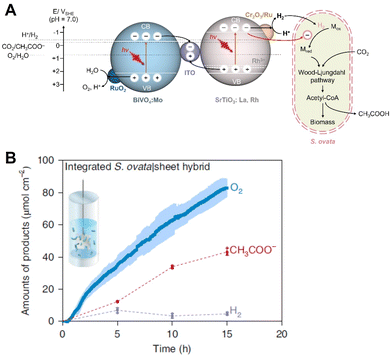 | ||
| Fig. 14 Schematic of the visible light-driven acetate production from CO2 by the hybridization of a Z-scheme semiconductor photocatalytic system consisting of SrTiO3: La, Rh and BiVO4: Mo with S. ovata (A). Time course of photosynthetic acetate, O2 and H2 production over S. ovata|Cr2O3/Ru-SrTiO3:La,Rh|ITO|RuO2-BiVO4:Mo hybrids (B). Reproduced with permission from ref. 85. Copyright 2022, The Authors, under exclusive license to Springer Nature Limited. | ||
Using the hybridization of a Z-scheme semiconductor photocatalytic system consisting of SrTiO3: La, Rh and BiVO4: Mo with S. ovata, ca. 40 and 80 µmol cm−2 of acetate and oxygen production are observed after 15 h with 1 sun (AM 1.5G, 100 mW cm−2) irradiation, as shown in Fig. 14(B). A gas mixture of 80% N2 and 20% CO2 is used in this system. The solar energy to acetate efficiency is estimated to be 0.70% ± 0.04%, and the apparent quantum yield is calculated to be 21.3% at 420 ± 15 nm. The quantum yield is also estimated using eqn (1) in this experiment.
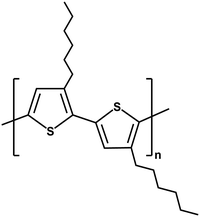
Visible-light-driven acetate production from CO2 using a system that hybridises a semiconducting polymer with S. ovata is also studied.86 The polymer semiconductor poly(3-hexylthiophene) (P3HT; chemical structure is shown in Fig. 15), widely used in the organic photovoltaic field,87,88 is applied in this system.
Fig. 16 shows the schematic representation of visible-light-driven acetate production from CO2 using a system of P3HT immobilised onto an indium tin oxide (ITO) coated glass substrate and S. ovata in the presence of Cys and an electron mediator (M).
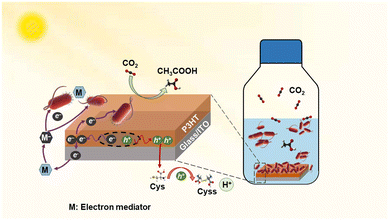 | ||
| Fig. 16 Schematic of the visible light-driven acetate production from CO2 using a system of PSHT immobilised ITO substrate with S. ovata in the presence of Cys and M. Reproduced with permission from ref. 86. Copyright 2024, the American Association for the Advancement of Science. | ||
Fig. 17 shows a schematic diagram of the exciton generation, separation, and transport processes in P3HT, [6,6]-phenyl-C61-butyric acid methyl ester (P3HT: PCBM) heterojunction (bulk heterojunction (BHJ)), and a film with an ITO/ZnO/P3HT:PCBM/MoO3 multilayer structure.
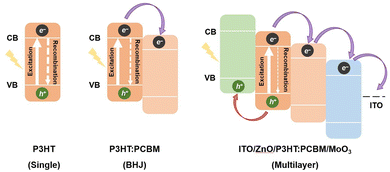 | ||
| Fig. 17 Schematic of the exciton separation and recombination of polymer semiconductor films with P3HT, P3HT:PCBM bulk-heterojunction (BHJ), and multilayer film structures. CB, conduction band; VB, valence band. Reproduced with permission from ref. 86. Copyright 2024, the American Association for the Advancement of Science. | ||
Here, visible-light-driven acetate production from CO2 is investigated using a hybrid system comprising each P3HT-immobilised device and S. ovata. Cys and potassium ferricyanide are added as a hole-trapping agent and an electron mediator, respectively. The LED white light is used as an irradiation source with adjustable light intensity. A gas mixture of 80% N2 and 20% CO2 is used as a carbon source in this system. Fig. 18 shows the total amount of acetate production from CO2 using a hybrid system comprising each P3HT-immobilised device and S. ovata.
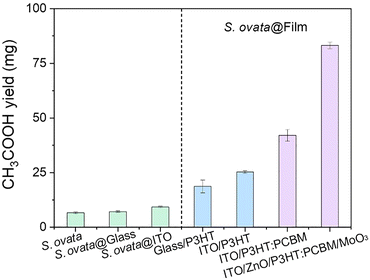 | ||
| Fig. 18 Total acetate production using a hybrid system comprising a P3HT-immobilised device and S. ovata after 1 week of light irradiation in the presence of Cys and potassium ferricyanide. Reproduced with permission from ref. 86. Copyright 2024, the American Association for the Advancement of Science. | ||
It is found that acetate production is improved using the hybrid system comprising ITO/ZnO/P3HT:PCBM/MoO3 multilayer and S. ovata compared to other devices, as shown in Fig. 18. The quantum yield of the hybrid system comprising ITO/ZnO/P3HT:PCBM/MoO3 multilayer and S. ovata reaches about 10%, which is obviously higher than 0.5% of the control system without a polymer semiconductor device. Additionally, in this experiment, the quantum yield is estimated using eqn (1).
Visible-light-driven acetate production from CO2 using a system hybridising InP/ZnSe/ZnS quantum dots (QDs) with S. ovata in the presence of Cys and potassium ferricyanide is studied.89 InP with a bandgap of 1.35 eV and a Bohr radius of approximately 10 nm can effectively absorb visible light.90Fig. 19(A) shows the schematic representation of visible-light-driven acetate production from CO2 using a hybridising system of InP/ZnSe/ZnS QDs and S. ovata.
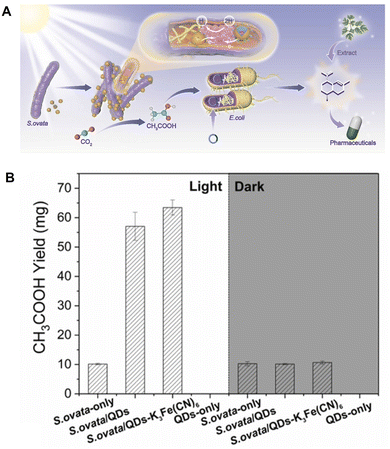 | ||
| Fig. 19 Schematic of the visible light-driven acetate production from CO2 using a hybrid system comprising InP/ZnSe/ZnS QDs and S. ovata (A). Total acetate production using the hybrid system comprising InP/ZnSe/ZnS QDs and S. ovata after 1 week of light irradiation in the presence of Cys and potassium ferricyanide (B). Reproduced with permission from ref. 89. Copyright 2022, Elsevier Ltd. | ||
By growing a multilayer shell of ZnSe and ZnS on InP QDs synthesized using the hot injection method, the surface of the QDs can be passivated, and the stability of the InP QDs can be improved.91,92 Furthermore, the low biotoxicity of InP QDs makes them potentially promising biological hybrid materials.93
In this system, Cys and potassium ferricyanide are also added as a hole-trapping agent and an electron mediator, respectively. The Xenon lamp with an AM 1.5 G filter is used as the irradiation source with adjusted light intensity. A gas mixture of 80% N2 and 20% CO2 is used as the carbon source in this system. Fig. 20(B) shows the total amount of acetate production from CO2 using a hybridising system comprising InP/ZnSe/ZnS QDs and S. ovata. It is found that acetate production is improved using the hybridising system comprising InP/ZnSe/ZnS QDs and S. ovata in the presence of ferricyanide compared to other conditions, as shown in Fig. 20(B). The quantum yield of the hybridising system comprising InP/ZnSe/ZnS QDs and S. ovata in the presence of ferricyanide reaches about 7.0% estimated using eqn (1).
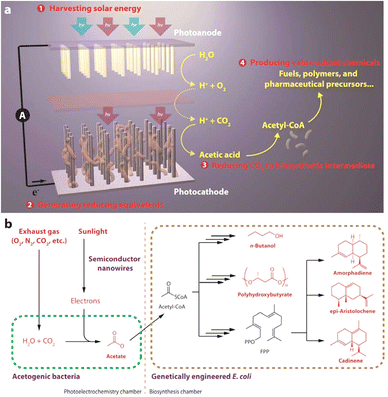 | ||
| Fig. 20 Schematic of a photoelectrochemical cell consisting of silicon nanowire-integrated S. ovata: (a) Proposed approach for solar-powered CO2 fixation includes four general steps: (1) harvesting solar energy, (2) generating reducing equivalents, (3) reducing CO2 to biosynthetic intermediates, and (4) producing value-added chemicals. Through the integration of materials science and biology, such an approach combines the advantages of solid-state devices with living organisms. (b) As a proof of concept, under mild conditions, sunlight can provide the energy to directly treat exhaust gas and generate acetate as the biosynthetic intermediate, which can be upgraded into liquid fuels, biopolymers, and pharmaceutical precursors. FPP: farnesyl pyrophosphate. Reproduced with permission from ref. 94. Copyright 2015, American Chemical Society. | ||
Visible-light-driven acetate production from CO2 using a photoelectrochemical cell consisting of silicon nanowire integrated S. ovata as a photocathode has been reported, as shown in Fig. 20.94
Using a photoelectrochemical cell consisting of silicon nanowire integrated with S. ovata, visible-light acetate production is accomplished. Moreover, variable organic molecules are produced using a hybridisation system comprising engineered E. coli with this photoelectrochemical cell. In addition, it has been found that acetate production is improved using methanol-adapted S. ovata in this system.95
Visible-light-driven production of other plastic precursors from CO2 using a photocatalyst and microbial cell hybrid system
Other plastic precursors that can be produced from CO2 driven by visible light using a photocatalyst and microbial cell hybrid system are introduced.Shikimic acid is a precursor for lignin production and can be a raw material for bioplastics.96
Visible-light-driven shikimic acid production using a system of polyphenol-functionalised indium phosphide (InP) with common heterotrophs, Saccharomyces cerevisiae (S. cerevisiae), has been reported, as shown in Fig. 21.97 In this system, yeast strain S. cerevisiae Δzwf1 is selected. S. cerevisiae Δzwf1 is genetically engineered to overexpress four genes to enhance carbon flux through the shikimic acid pathway.98
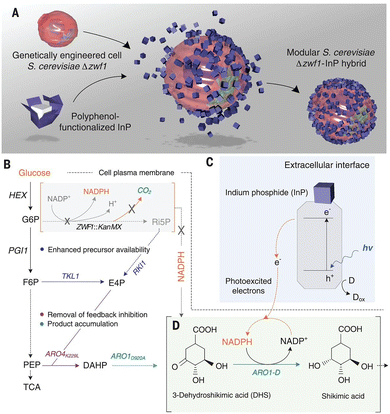 | ||
| Fig. 21 Schematic of the preparation of InP–S. cerevisiae Δzwf1 (A). Metabolic engineering diagram for the overproduction of shikimic acid. S. cerevisiae Δzwf1 has the oxidative pentose phosphate pathway disrupted (ZWF1), leading to low cytosolic NADPH pools, which directly affect the shikimic acid pathway and reduce carbon loss in the production of CO2 (B). Schematic of the cellular NADPH regeneration and shikimic acid biosynthesis assisted by photogenerated electrons from InP nanoparticles (C and D). G6P, glucose-6-phosphate; F6P, fructose-6-phosphate; Ri5P, ribulose-5-phosphate; E4P, erythrose-4-phosphate; PEP, phosphoenolpyruvate; DAHP, 3-deoxy-d-arabinoheptulosonate-7-phosphate; HEX, hexokinase; ZWF1, glucose-6-phosphate 1-dehydrogenase; PGI1, phosphoglucose isomerase; RKI1, ribose-5-phosphate ketol-isomerase; TKL1, transketolase; ARO4K229L, feedback-insensitive DAHP synthase; ARO1D920A, mutant pentafunctional aromatic enzyme; TCA, tricarboxylic acid cycle; h, Planck's constant; ν, frequency; h+, electron hole; e−, electron; D, putative electron donors in the cell culture medium; Dox, oxidized electron donor species. Reproduced with permission from ref. 97. Copyright 2018, the American Association for the Advancement of Science. | ||
Fig. 22 shows the specific shikimic acid production with the system using InP–S. cerevisiae Δzwf1 under visible-light irradiation (5.6 mW cm−2). The specific shikimic acid production with S. cerevisiae Δzwf1 or wild-type S. cerevisiae under the conditions of dark incubation is shown in Fig. 22. As shown in Fig. 22, the specific shikimic acid production increases using InP–S. cerevisiae Δzwf1 under visible-light irradiation compared with that of the system using S. cerevisiae Δzwf1 or wild-type S. cerevisiae under dark conditions.
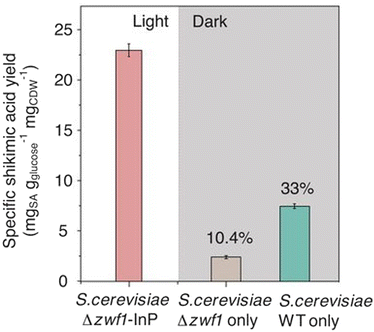 | ||
| Fig. 22 Total shikimic acid production with InP–S. cerevisiae Δzwf1 under visible-light irradiation (5.6 mW cm−2), with S. cerevisiae Δzwf1 or wild-type S. cerevisiae under the condition of dark incubation. Reproduced with permission from ref. 97. Copyright 2018, the American Association for the Advancement of Science. | ||
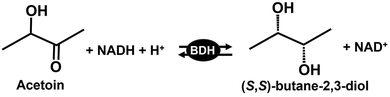
3-Hydroxybutanone (acetoin) and 2,3-butanediol are two important four-carbon platform compounds with pharmaceutical and chemical synthesis applications.99–102 (S,S)-Butane-2,3-diol is produced from acetoin in the presence of NADH with (S,S)-butanediol dehydrogenase (BDH; EC EC 1.1.1.76), as shown in Fig. 23.103–105
Visible-light-driven acetoin production from CO2 with a hybrid system comprising eosin Y and Ralstonia eutropha (R. eutropha) has been reported.106R. eutropha has attracted significant research attention due to its excellent CO2 fixation capacity and potential for expanding production diversity.107Fig. 24 shows the schematic representation of a hybrid system comprising eosin Y and R. eutropha for visible-light acetoin production from CO2. In this system, eosin Y specifically binds to the membrane-bound hydrogenase (MBH) of R. eutropha and achieves targeted electron transfer to the hydrogenase-mediated electron transport and transformation pathway of R. eutropha (pathway I). To enhance ATP synthesis, an oxygen-independent proton pump (Gloeobacter rhodopsin) is introduced into R. eutropha (pathway II). To promote carbon metabolic flux towards acetoin production, the by-product biosynthetic pathway is blocked (pathway III). The genetically engineered R. eutropha strain named REH01 is used in this system.
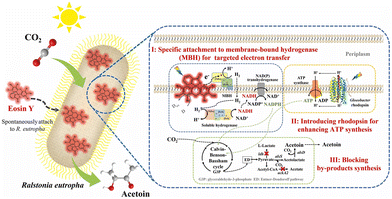 | ||
| Fig. 24 Visible-light-driven acetoin production from CO2 using a hybrid system comprising eosin Y and R. eutropha. Reproduced with permission from ref. 106. Copyright 2025, The Authors. Published by Elsevier Ltd. Creative Commons CC-BY license. | ||
Fig. 25(A) shows the time dependence of acetoin production using a hybrid system comprising eosin Y and REH01 during visible-light irradiation using LED lamps (λ = 520![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nm) with a certain light intensity (2, 5, or 10
nm) with a certain light intensity (2, 5, or 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mW cm−2). Throughout the reaction, a gas mixture of CO2/O2 (molar ratio: 99/1) is continuously sparged into the reaction system. In this figure, biohybrid means a hybrid system comprising eosin Y and REH01.
mW cm−2). Throughout the reaction, a gas mixture of CO2/O2 (molar ratio: 99/1) is continuously sparged into the reaction system. In this figure, biohybrid means a hybrid system comprising eosin Y and REH01.
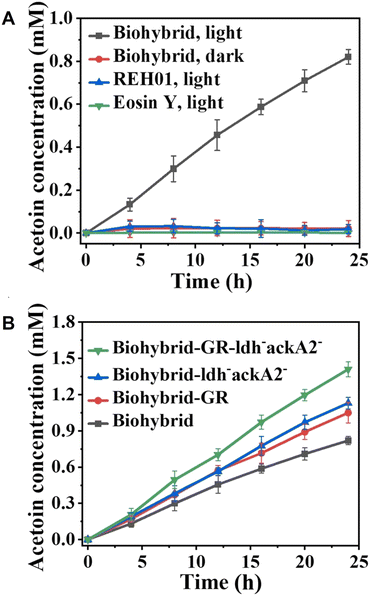 | ||
| Fig. 25 Time-dependent acetoin production from CO2 using a hybrid system comprising eosin Y and REH01 during visible-light irradiation. Red: dark condition, Blue: only REH01, Green: only eosin Y (A). Time dependence of acetoin production from CO2 using a hybrid system comprising eosin Y and REH01 during visible-light irradiation. Green: eosin Y and GR expressing REH01 without ldh and ackA2. Bule: eosin Y and REH01 without ldh and ackA2. Red: eosin Y and GR expressing REH01. Black: eosin Y and REH01 (B). Reproduced with permission from ref. 106. Copyright 2025, The Authors. Published by Elsevier Ltd. Creative Commons CC-BY license. | ||
As shown in Fig. 25(A), ca. 0.8 mM of acetoin production is observed after 24 h of irradiation. However, little acetoin production has been observed under other conditions. Furthermore, metabolic engineering strategies have been employed to enhance the acetoin-producing capability of REH01. REH01 strains lacking the GR and/or ldh and ackA2 genes have been prepared. Fig. 25(B) shows the time dependence of acetoin production with a hybrid system comprising eosin Y and genetically modified REH01 during visible-light irradiation. As shown in Fig. 25(B), the engineered eosin Y-GR expressing REH01 without the ldh and ackA2 biohybrid system achieves an acetoin yield of 1.41 ± 0.06 mM, which is 2.07 times larger than that of H2-supplied autotrophic fermentation. Therefore, carbon flux can be directed towards acetoin production by inhibiting the L-lactate and acetate biosynthetic pathways.
Visible-light-driven polyhydroxybutyrate (PHB) production from CO2 using a photocatalyst and microbial cell hybrid system
Among biodegradable plastics based on aliphatic polyesters, plastics derived from poly-3-hydroxybutyrate (PHB) are attractive because they are compostable, made from renewable feedstocks, and biodegradable.108–115 The PHB can be produced from CO2 driven by visible light using a photocatalyst and a microbial cell hybrid system. A hydrogen-oxidizing bacterium, Ralstonia eutropha, is used as a microbial cell for PHB production.116The PHB production using an electrochemical system comprising cobalt-phosphorus (Co-P) alloy cathode and cobalt phosphate (CoPi) anode with Ralstonia eutropha is reported for the first time.117
Visible-light-driven PHB production with a system comprising photocatalyst graphitic carbon nitride (g-C3N4) and Ralstonia eutropha H16 in the presence of triethanol amine (TEOA) is reported, as shown in Fig. 26(A).118 As shown in Fig. 26(A), PHB is produced in Ralstonia eutropha H16 by utilizing the TCA cycle, Calvin–Benson–Bassham (CBB) cycle, NADH regeneration, and hydrogen.
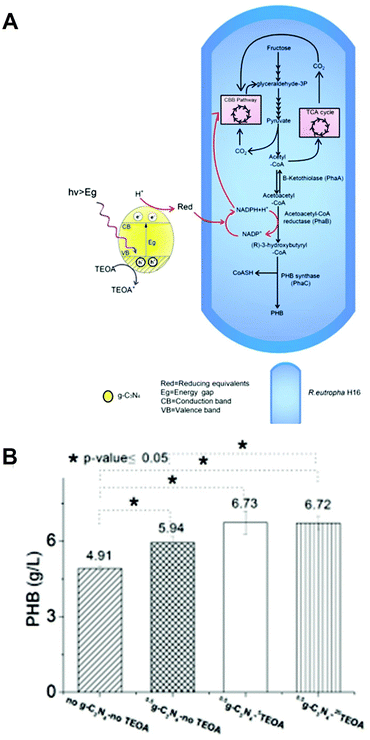 | ||
| Fig. 26 Schematic of the visible-light-driven PHB production from CO2 or fructose using a system comprising g-C3N4 and Ralstonia eutropha H16 in the presence of TEOA (A). Total PHB production using the system comprising g-C3N4 and Ralstonia eutropha H16 in the presence of TEOA after 96 h of irradiation with an LED light set at 4200 lux (B). Reproduced with permission from ref. 118. Copyright 2019, The Royal Society of Chemistry. | ||
Fig. 26(B) shows the total amount of PHB production using g-C3N4 and Ralstonia eutropha H16 in the presence of TEOA after 96 h of irradiation using an LED light set at 4200 lux. This figure also shows the amount of PHB produced in the absence of TEOA or g-C3N4. After 96 h irradiation, PHB production is improved 1.4 times to 6.73 ± 0.45 g l−1 with the system using g-C3N4 and Ralstonia eutropha H16 in the presence of TEOA. However, decreasing PHB production is observed using only Ralstonia eutropha H16 or without g-C3N4.
The development of a water-splitting enzymatic photocatalyst made of g-C3N4 coupled with H2O2-degrading catalase and its utilisation for a hybrid system with Ralstonia eutropha for visible-light-driven PHB production, as shown in Fig. 27(A).119 Using catalase, it is possible to decompose hydrogen peroxide produced due to the two-electron oxidation of water into oxygen120 by g-C3N4.121
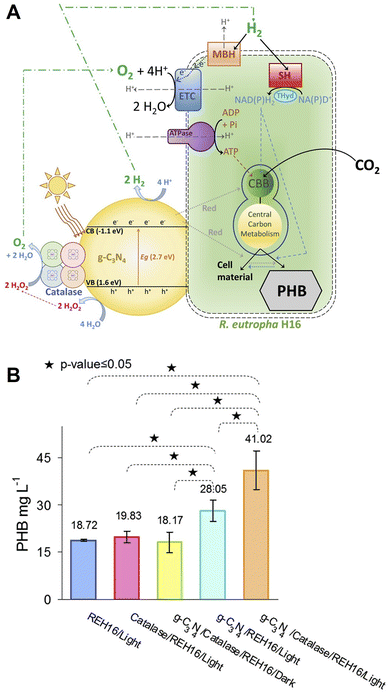 | ||
| Fig. 27 Schematic of the visible-light-driven PHB production from CO2 using a system comprising catalase-modified g-C3N4 and Ralstonia eutropha H16 (A). Total PHB production using the system comprising catalase-modified g-C3N4 and Ralstonia eutropha H16 after 48 h of irradiation using a 500 W Xe lamp with an AM1.5G filter (B). Reproduced with permission from ref. 119. Copyright 2020, The authors Creative Commons CC-BY-NC-ND license. | ||
Fig. 27(B) shows the total amount of PHB production using catalase-modified g-C3N4 and Ralstonia eutropha H16 after 48 h of irradiation using a 500 W Xe lamp with an AM1.5G filter. This figure also shows the amount of PHB produced using the system only Ralstonia eutropha H16, without g-C3N4, without catalase and under dark conditions.
The hybrid system built using the water-splitting g-C3N4-catalase photocatalyst doubles the production of the PHB by Ralstonia eutropha H16 from CO2 and increases it by 1.84-fold from fructose. After 48 h of irradiation, ca.41 mg of PHB production is observed using catalase-modified g-C3N4 and Ralstonia eutropha H16 without any sacrificial electron donor reagent, such as TEOA.
Visible-light-driven PHB production from CO2 using a system of conjugated polymer dots (Pdots) consisting of poly[N-9′-heptadecanyl-2,7-carbazole-alt-5,5-(4′,7′-di-2-thienyl-2′,1′,3′-benzo-thiadiazole)] (PFODTBT) and triblock copolymer ABA, poly(N,N-dimethylamino ethyl methacrylate)-B-poly(9,9-N-dihyxyl-2,7-fluorene)-B-poly(N,N-dimethylamino ethyl methacrylate) and PS-PEG-COOH (copolymer polystyrene grafted with carboxy-terminated polyethylene oxide) (PFODTBT Pdots)122 and Ralstonia eutropha H16 (RH16/NR/Pdots) has been reported, as shown in Fig. 28(A) and (B).123 In this system, Cys and neutral red (NR) are used as an electron donor and an electron mediator, as the example of Fig. 5(b). A Xenon fiber optic lamp with filters is employed using an AM 1.5G filter. The optimal optical power density is determined to be 2.5 mW cm−2. A gas mixture of 80% N2 and 20% CO2 is used as a carbon source in this system. Under light irradiation, the PHB production of RH16 is estimated to be only 7.3 ± 2.91 mg L−1 due to accumulation in the initial stage of autotrophic, as shown in Fig. 28(C).
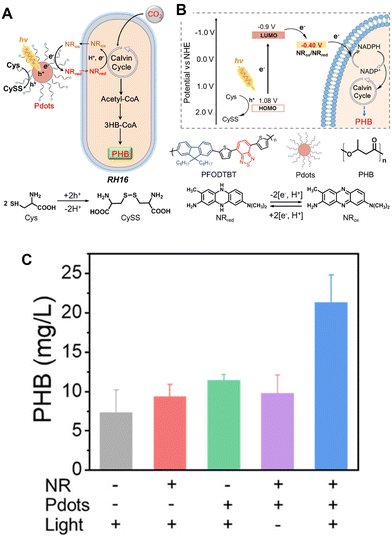 | ||
| Fig. 28 Schematic of the visible-light-driven PHB production from CO2 with RH16/NR/Pdots (A). Metabolic pathway of PHB production from CO2 in RH16. 3HB-CoA: 3-hydroxybutyryl-CoA; PHB: polyhydroxybutyrate; h+: electron hole; e−: electron; Cys: cysteine; CySS: cystine; NRox: the oxidized state of NR; NRred: the reduced state of NR (B). Schematic of Pdots promoting PHB production with RH16. Total PHB production using a system of RH16/NR/Pdots in the presence of Cys after 48 h of irradiation. “−” and “+” mean non-additive and added, respectively (C). Reproduced with permission from ref. 123. Copyright 2022, the American Chemical Society. | ||
The PHB productions of RH16/Pdots, RH16/NR, and RH16/NR/Pdots (under dark conditions) are estimated to be 9.3 ± 1.56, 11.4 ± 0.76, and 9.8 ± 2.35 mg L−1, respectively.
As shown in Fig. 28(C), the production of PHB from the biohybrid system is slightly higher than that of RH16, suggesting that the addition of Pdots and NR is responsible for the increased amount of PHB in the solution. Under visible-light irradiation, the cumulative PHB production of RH16/NR/Pdots (21.3 ± 3.78 mg L−1) increases significantly, which is an increase of 192% compared with only RH16. This is mainly because the visible-light-induced electrons of Pdots could be effectively transferred to the inside of RH16 by NR.
Visible-light-driven PHB production using a system comprising photocatalyst g-C3N4 and Rhodopseudomonas palustris (R. palustris)124 in the presence of sulfur nanoparticles (SNPs) (g-C3N4−SNPs−R. palustris) is reported, as shown in Fig. 29(A).125 In this system, SNPs act as dual electron donors, as shown in Fig. 29(A).
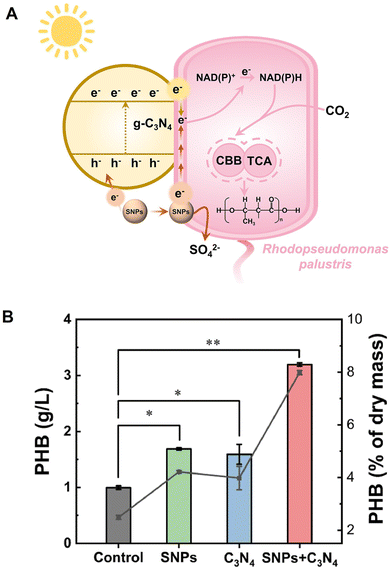 | ||
| Fig. 29 Schematic of visible-light-driven PHB production from CO2 using a system of g-C3N4−SNPs−R. palustris (A). Total PHB production from CO2 using a system of g-C3N4−SNPs−R. palustris after 8 days of irradiation (B). Reproduced with permission from ref. 125. Copyright 2025, The Research Center for Eco-Environmental Sciences, the Chinese Academy of Sciences. Published by Elsevier B.V. | ||
Fig. 29(B) shows the total PHB production using a system of g-C3N4−SNPs−R. palustris under irradiation with simulated daylight (∼7000 lux) after 8 days. A gas mixture of 80% N2 and 20% CO2 is used as a carbon source in this system. Fig. 29(B) shows the PHB concentrations and percentage of cell dry weight occupied by PHB on day 8 (grey line).
The optimized g-C3N4−SNPs−R. palustris achieves a remarkable PHB production titer of 3.19 ± 0.03 g L−1 with 11.79% ± 0.93% quantum efficiency using solely CO2 and SNPs.
A homogeneously autotrophic K/O co-doped g-C3N4 (K/O-CN) and R. eutropha biohybrid system is reported for visible-light-driven PHB production from CO2, as shown in Fig. 30(A).126 This system introduces nano-sized, scattered rod-like K/O co-doped g-C3N4 (K/O-CN) as a replacement for bulk g-C3N4, therefore increasing the interface contact and mass transfer at the abiotic–biotic interface R. eutropha.
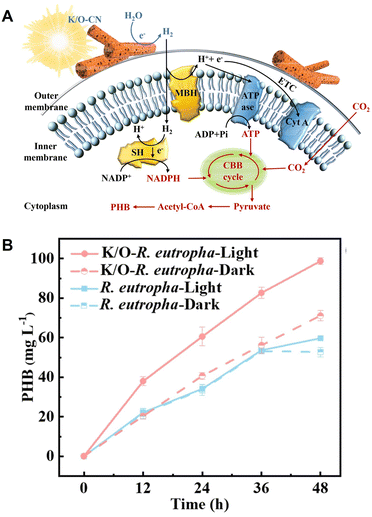 | ||
| Fig. 30 Schematic of the visible-light-driven PHB production from CO2 using a system of K/O-CN–R. eutropha (A). Time dependence of PHB production from CO2 using the hybrid system of K/O-CN–R. eutropha under visible-light irradiation (B). Reproduced with permission from ref. 126. Copyright 2026, The Royal Society of Chemistry. | ||
Fig. 30(B) shows the time dependence of PHB production using a system of K/O-CN–R. eutropha under visible-light irradiation with light at an intensity of 4000 lux (0.75 mW cm−2). A gas mixture (N2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2
H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2
CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O2 = 76
O2 = 76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) is used as a carbon source in this system. As shown in Fig. 30(B), the K/O-CN–R. eutropha biohybrid achieves a PHB yield of 49.35 ± 0.85 mg L−1 day−1, surpassing non-metal-based biohybrids by adding co-factors and reaching a quantum efficiency of 5.88% ± 0.16%, which exceeds most metal-based biohybrid systems. Using K/O-CN–R. eutropha, the PHB production is improved compared to the dark condition or the system excluding K/O-CN.
4) is used as a carbon source in this system. As shown in Fig. 30(B), the K/O-CN–R. eutropha biohybrid achieves a PHB yield of 49.35 ± 0.85 mg L−1 day−1, surpassing non-metal-based biohybrids by adding co-factors and reaching a quantum efficiency of 5.88% ± 0.16%, which exceeds most metal-based biohybrid systems. Using K/O-CN–R. eutropha, the PHB production is improved compared to the dark condition or the system excluding K/O-CN.
A nanosheet composed of g-C3N4 is one of the promising visible light-responsive photocatalysts.127 The g-C3N4 nanosheet (CNNS)–R. eutropha is reported for visible-light-driven PHB production from CO2, as shown in Fig. 31(A).128
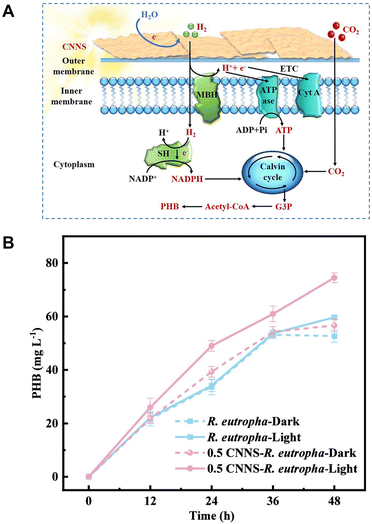 | ||
| Fig. 31 Schematic of the visible-light-driven PHB production from CO2 using a system of CNNS–R. eutropha (A). Time-dependent PHB production from CO2 using the hybrid system of CNNS–R. eutropha under visible-light irradiation (B). Reproduced with permission from ref. 128. Copyright 2025, American Chemical Society. | ||
Fig. 31(B) shows the time dependence of PHB production using the CNNS–R. eutropha system under continuous irradiation at an intensity of 4000 lx. A gas mixture (N2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2
H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2
CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O2 = 76
O2 = 76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) is used as a carbon source in this system. Comparative experiments are conducted under both light and dark conditions, as shown in Fig. 31(B). The CNNS–R. eutropha system exhibits the highest PHB yield of 37.25 ± 0.9 mg L−1 day−1 under irradiation with 0.5 g L−1 CNNS, demonstrating a 41.47% increase compared to the only R. eutropha under dark conditions (26.33 mg L−1 day−1). Thus, the PHB production yield also surpasses the yields observed under both light and dark conditions without CNNS. Under dark conditions, the PHB yield is similar to whether CNNS is added. This result indicates that CNNS does not exert toxic effects on R. eutropha and that the enhancement in PHB production within this system is light-dependent.
4) is used as a carbon source in this system. Comparative experiments are conducted under both light and dark conditions, as shown in Fig. 31(B). The CNNS–R. eutropha system exhibits the highest PHB yield of 37.25 ± 0.9 mg L−1 day−1 under irradiation with 0.5 g L−1 CNNS, demonstrating a 41.47% increase compared to the only R. eutropha under dark conditions (26.33 mg L−1 day−1). Thus, the PHB production yield also surpasses the yields observed under both light and dark conditions without CNNS. Under dark conditions, the PHB yield is similar to whether CNNS is added. This result indicates that CNNS does not exert toxic effects on R. eutropha and that the enhancement in PHB production within this system is light-dependent.
We focus on the photocatalyst in a hybrid system comprising photocatalysts and microbial cells, as introduced in this section. The photocatalysts used in hybrid systems are visible-light-responsive semiconductors, and most of them have a proven track record in hydrogen production based on the photolysis of water. In hybrid systems, efficient electron and proton transfer from the photocatalyst to the microbial cell governs the CO2 conversion rate. Therefore, developing catalysts that bring semiconductor photocatalysts into contact with the surface of microbial cells is crucial to achieving efficient CO2 conversion.
Next, the separation of products based on CO2 conversion using hybrid systems is discussed. Currently, the products from the hybrid systems described in this section are separated only by high-performance liquid chromatography (HPLC). In addition, since microbial cells are used, suppressing by-products is another challenge for scaling up this system.
Visible-light-driven 3-hydoroxybutylate production from CO2 and acetone with the photocatalytic dye and cell extract, including a multi-enzyme hybrid system
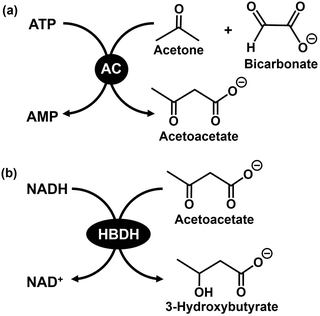
Another visible-light-driven production of biodegradable polymer precursor from CO2 method is to use a cell extract containing crude enzymes, as shown in Fig. 32, instead of the microbial cell shown in Fig. 5(b).This section describes the production of 3-hydroxybutyrate, a precursor of PHB, and a promising biodegradable plastic. The research example on visible-light-driven 3-hydroxybutyrate production with acetone carboxylase (AC; EC 6.4.1.6) and 3-hydroxybutyrate dehydrogenase (HBDH; EC 1.1.1.30) added as a catalyst in Fig. 32 is introduced. Fig. 33 shows the AC-catalysed acetoacetate production from acetone and bicarbonate in the presence of ATP (a) and HBDH-catalysed 3-hydroxybutyrate production based on the acetoacetate reduction in the presence of NADH (b). Using a dual-enzyme consisting of AC and HBDH, 3-hydroxybutyrate can be produced from acetone and bicarbonate via acetoacetate as an intermediate.129,130
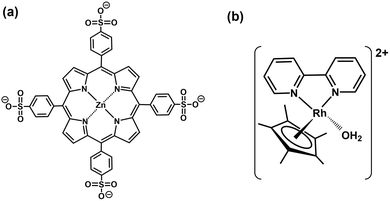
Visible-light-driven 3-hydroxybutylate production from bicarbonate or CO2 and acetone with a system comprising TEOA, zinc tetraphenylporphyrin tetrasulfonate (ZnTPPS4−; chemical structure is shown in Fig. 34(a)), rhodium coordination complex ([RhCp*(bpy)(H2O)]2+; Cp* = pentamethylcyclo-pentadienyl, bpy = 2,2′-bipyridyl; chemical structure is shown in Fig. 34(b)), NAD+, ATP, AC and HBDH is reported.131,132
AC133,134 and HBDH130 are expressed from photosynthetic bacteria Rhodobacter capsulatus SB1003 (Rb. capsulatus SB1003) cultured in acetone-bicarbonate medium. The cell extract containing AC and HBDH is obtained from Rb. capsulatus SB1003. First, visible-light-driven 3-hydroxybutyrate production from acetone and bicarbonate instead of CO2 gas using a system comprising TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+, ATP, and cell extract containing AC and HBDH is introduced.131 After 5 h of irradiation using a 250 W halogen lamp (λ > 390 nm), 379 µM of 3-hydroxybutyrate is produced in the sample solution of acetone (0.5 mM), TEOA (0.2 M), ZnTPPS4− (50 µM), [RhCp*(bpy)(H2O)]2+ (5.0 µM), NAD+ (2.0 mM), sodium bicarbonate (50 mM), sodium ATP (2.0 mM), magnesium chloride (5.0 mM) and cell extract (AC 0.051 U, HBDH 0.47 U) in 5.0 mL of 500 mM HEPES–NaOH buffer (pH 7.0). Using this system, the conversion yield of acetone to 3-hydroxybutyrate is approximately 76%.
Visible-light-driven 3-hydroxybutyrate production from acetone and directly captured CO2 from a mixture of N2 and CO2 gases using the system comprising TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+, ATP and enzyme extract containing AC and HBDH is introduced.132Fig. 35 shows the time dependence of acetoacetate (a) and 3-hydroxybutyrate (b) concentration in the sample solution of acetone (0.5 mM), TEOA (0.2 M), ZnTPPS4− (50 µM), [RhCp*(bpy)(H2O)]2+ (5.0 µM), NAD+ (2.0 mM), sodium ATP (2.0 mM), magnesium chloride (5.0 mM) and cell extract (AC 0.062 U, HBDH 0.7 U) in 5.0 mL of 500 mM HEPES–NaOH buffer (pH 7.0) under conditions with varying ratios of CO2 and N2 in the gas phase with irradiation using a 250 W Halogen lamp (λ > 390 nm).
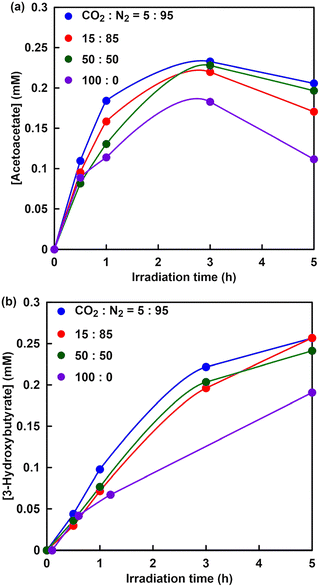 | ||
| Fig. 35 Time-dependent concentration of acetoacetate (a) and 3-hydroxybutyrate (b) in the sample solution of acetone, TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+, ATP, magnesium chloride and cell extract (AC, HBDH) in an HEPES–NaOH buffer (pH 7.0) under varying ratios of CO2 and N2 in the gas phase and irradiation. Reproduced with permission from ref. 132. Copyright 2023, The Royal Society of Chemistry. | ||
The results using this system are summarized in Table 1. The data in Table 1 are calculated from the figures published in ref. 132.
| [CO2]/[N2] | [3-Hydroxybutyrate] (µM) after 5 h of irradiation | Conversion yield for acetone to 3-hydoxybutyrate after 5 h of irradiation % |
|---|---|---|
| 0 | 0 | 0 |
| 0.05 | 257 | 51.4 |
| 0.15 | 257 | 51.4 |
| 0.5 | 241 | 48.2 |
| 1.0 | 190 | 38.0 |
As shown in Table 1, no significant difference in 3-hydroxybutyrate production is observed in the mixed gas with the ratio of CO2 to N2 not exceeding 0.5. In contrast, 3-hydroxybutyrate production decreases under conditions with 100% CO2 gas compared to other conditions. This is because the reaction system is isochoric and CO2 in the gas phase dissolves into the sample solution, leading to a decrease in pressure, as shown in Table 1. Under the condition of 100% CO2, especially, it is predicted that the catalytic activity of cell extract involving AC and HBDH is inhibited based on the decrease in pressure in the reaction system during irradiation. Therefore, gaseous CO2 can be directly used as a raw material without using bicarbonate because of the gas-phase low concentration gaseous CO2 trapping function of the HEPES–NaOH buffer solution used in the reaction. In addition, the captured gaseous CO2 is converted to bicarbonate in the HEPES–NaOH buffer and acts as a carboxylating agent for acetone with an enzyme extract, including AC. This suggests that several to 20% of the gaseous CO2 contained in gases emitted from thermal power stations, steel mills and chemical plants142 could be used as feedstock in this system.
Finally, waste acetone and low-concentration CO2 are used as raw materials in the visible-light-driven 3-hydroxybutyrate production with the system comprising TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+, ATP and cell extract, including AC and HBDH.132 Acetone is an abundant and useful chemical in the laboratory because it serves as a non-halogenated organic solvent that is miscible with water.143 Acetone is relatively cheap, reasonably harmless and is often the solvent of choice in chemical laboratories for cleaning glassware contaminated with organic deposits. Although acetone is useful as a solvent, it also requires an efficient method for recycling after use. Because black permanent marker ink contains various organic solvents, such as m-xylene, iso-butanol, resin and oil soluble dyestuff, it can be used as a model for insoluble stains. The commercial black permanent marker ink used in this experiment contains 58% m-xylene, 14% iso-butanol, 16% resin and 12% oil soluble dyestuff. Here, black permanent marker ink dissolved in acetone is used as a waste solvent model. After adding the waste acetone to the HEPES–NaOH buffer, the supernatant is separated from the precipitate, and the supernatant, including acetone, m-xylene and iso-butanol, is used for the visible-light-driven 3-hydroxybutyrate production. The reaction mixture consists of supernatant, including acetone (0.5 mM), sodium ATP (2.0 mM), magnesium chloride (5.0 mM), TEOA (0.2 M), ZnTPPS4− (50 µM), [RhCp*(bpy)(H2O)]2+ (5.0 µM), NAD+ (2.0 mM) and cell extract (AC: 0.062 U and HBDH: 0.7 U), with a mixed gas of a CO2 to N2 ratio of 0.15 in 5 mL of 500 mM HEPES–NaOH buffer. Fig. 36 shows the time dependence of acetoacetate and 3-hydroxybutyrate concentrations in the sample solution during irradiation using a 250 W halogen lamp (λ > 390 nm).
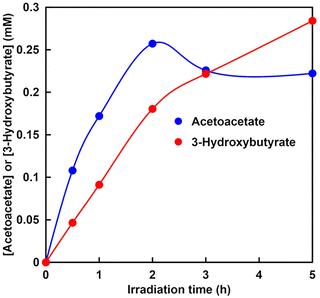 | ||
| Fig. 36 Time-dependent concentration of acetoacetate or 3-hydroxybutyrate in the solution of the supernatant containing acetone, ATP, magnesium chloride, TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+ and the cell extract (AC and HBDH:) in an HEPES–NaOH buffer in the presence of a gas mixture of a CO2 to N2 ratio of 0.15 in the gas phase under irradiation. Reproduced with permission from ref. 132. Copyright 2023, The Royal Society of Chemistry. | ||
After 5 h of irradiation, 0.28 mM of 3-hydroxybutyrate is produced. No significant difference is observed between pure and waste acetone in the visible-light-driven 3-hydroxybutyrate synthesis using low concentrations of gaseous CO2.
Strategy for the visible-light-driven production of CO2-based biodegradable plastic precursors using a photocatalytic dye and multi-enzyme hybrid system
The method of directly using microbial cells as a biocatalyst has the advantages of being simple and high in product yield, but it also has the drawback of difficulty in controlling the selectivity of the target product. Therefore, it has been proposed to produce biodegradable plastic precursors by incorporating an enzyme (ENZ) with the function of CO2 fixation into a photoredox system consisting of an electron donor (ED), a visible light sensitizer (PS), and an electron mediator (EM), as shown in Fig. 37.135–137By incorporating hydrogenase (H2ase; EC 1.12.2.1; cytochrome-c3 hydrogenase,138–144 EC 1.12.7.2; ferredoxin hydrogenase145–150) and formate dehydrogenase151–155 (FDH; EC 1.17.1.9; NAD+-dependent, EC 1.17.1.10; NADP+-dependent, and EC 1.17.98.4; selenium-containing formate dehydrogenase H) into a photoredox system consisting of ED, PS, and EM, visible-light-driven hydrogen production and formate production based on CO2 reduction are achieved.
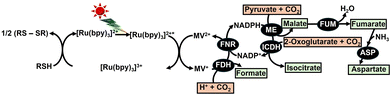
The first example of applying this system to the fixation of CO2 to a substrate is shown in Fig. 38.156
In this system, [Ru(bpy)3]2+ (chemical structure is shown in Fig. 39) is used as the PS.
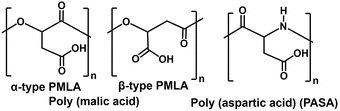
As shown in Fig. 38, FDH (EC 1.12.2.1), ICDH (EC 1.1.1.42), and ME (EC 1.1.1.40) can be used as CO2 conversion enzymes in the photoredox system. When FDH is used as the enzyme in a photoredox system, the MV+ can be used directly as a coenzyme instead of NAD(P)H. However, when ICDH or ME is used as an enzyme in a photoredox system, it is necessary to incorporate NADPH regeneration. As depicted in Fig. 38, biodegradable plastic precursors malate, fumarate, and aspartate are produced from CO2 as a raw material using a photoredox system incorporating enzymes. Polymers derived from malate or aspartate precursors are biodegradable, as shown in Fig. 40.
Fumarate can also be used as a precursor to biodegradable plastic PBS. For the first time, a system incorporating ME, FUM (EC 4.2.1.2), and ASP (EC 4.3.1.1) as catalysts in a photoredox system comprising RSH, [Ru(bpy)3]2+, MV2+, FNR (EC 1.18.1.2) and NADP+ is successfully used to produce fumarate and aspartate from CO2 and pyruvate under visible-light irradiation. Fig. 41 shows the time dependence of fumarate (a) and aspartate (b) production with a system comprising RSH (16 mM), [Ru(bpy)3]2+ (23 µM), MV2+ (0.16 mM), NADP+(0.32 mM), pyruvic acid (40 mM), sodium bicarbonate (0.16 M), ammonium ion (80 mM), manganese chloride (80 µM), FNR (0.5 U), ME (0.5 U), FUM (156 U) and ASP (3.0 U) in 4.0 mL of 0.16 M Tris-buffer solution (pH 7.9) during visible-light irradiation (1000 W halogen quartz lamp with 400 nm cut-off filter).
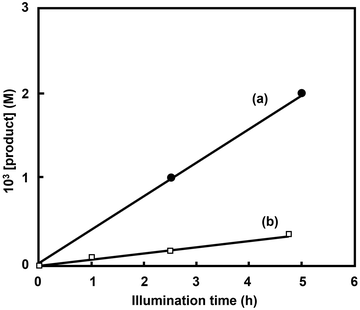 | ||
| Fig. 41 Time-dependent concentration of aspartate (a) and fumarate (b) in a system comprising RSH, [Ru(bpy)3]2+, MV2, NADP+, pyruvic acid, sodium bicarbonate, ammonium ion, manganese chloride, FNR, ME, FUM and ASP in Tris-buffer solution during visible-light irradiation. Reproduced with permission from ref. 156. Copyright 1988, The Royal Society of Chemistry. | ||
As shown in Fig. 41, fumarate and aspartate production increase during visible-light irradiation. After 5 h of irradiation, ca. 1.3 and 2.0 mM of fumarate and aspartate are produced. In the absence of ASP, no aspartate is produced and malate and fumarate are produced during visible-light irradiation. Additionally, visible-light-driven malate production with the system comprising RSH (19 mM), [Ru(bpy)3]2+ (21 µM), MV2+ (0.19 mM), NADP+(0.18 mM), pyruvic acid (47 mM), sodium bicarbonate (0.20 M), manganese chloride (95 µM), FNR (0.2 U) and ME (1.33 U) in 4.2 mL of 0.20 M Tris-buffer solution (pH 7.9) under a gaseous atmosphere of CO2 during visible-light irradiation (1000 W halogen quartz lamp with 400 nm cut-off filter). After 2 h of irradiation, 3.0 mM of malate production is observed.156
Thus, in order to produce biodegradable plastic precursors, such as malate, fumarate, and aspartate, it is essential to develop a visible-light-driven NAD(P)H regeneration system consisting of ED, PS, EM, and a catalyst that can be linked to an enzyme-based biocatalytic process. Visible-light-driven NAD(P)H regeneration, consisting of ED, PS, EM, and a catalyst, has been extensively studied in two main systems (Fig. 42).135–137 The first is a visible-light-driven NADPH regeneration system using FNR as a catalyst (Fig. 42(a)). The other is a visible-light-driven NAD(P)H regeneration system using [RhCp*(bpy)(H2O)]2+ as a catalyst (Fig. 42(b)). In the system shown in Fig. 42(a), visible-light-driven NADPH regeneration systems using [Ru(bpy)3]2+,156 5-deazariboflavin,162 riboflavin,162 proflavin,161,162 oligothiophene derivatives,164 CdS,165 CdSe quantum dots166 and polyethylene glycol-modified chlorophyll-a (PEG-Chl-a)167 as PSs are reported. In addition to 4,4′-bipyridinium salts, such as MV2+, 2,2′-bipyridinium salts and ferredoxin are used as EMs in this system.163 As shown in the system of Fig. 42(b), however, visible-light-driven NAD(P)H regeneration systems using carbon-doped TiO2,168 phosphate-doped TiO2,169 eosin-Y,170 porphyrin derivatives171,172 and proflavine173 as PSs are reported. Research examples on visible light-driven NAD(P)H regeneration systems are summarized and reported in several reviews.135–137,174,175
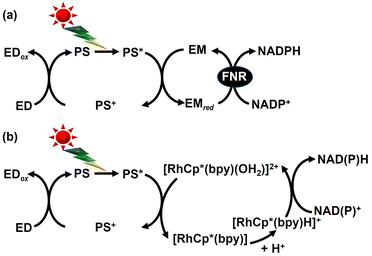 | ||
| Fig. 42 Visible-light-driven NAD(P)H regeneration using a system comprising ED, PS, EM and FNR (a) and ED, PS, and [RhCp*(bpy)(H2O)]2+ (b). | ||
The strategy for visible-light-driven CO2-based biodegradable plastic precursor production using a photocatalytic dye and multi-enzyme hybrid system involves the integrated coupling of an NAD(P)H regeneration system and an enzyme-based biocatalytic process.
Visible-light-driven malate production using a photocatalytic dye and enzyme hybrid system
As mentioned above, visible-light-driven malate production from CO2 and pyruvate using a system comprising RSH, [Ru(bpy)3]2+, MV2+, NADP+, FNR and ME has been developed for the first time. Some studies have been reported on the production of malate using visible light-driven NADPH regeneration catalysed by FNR in the presence of ME.Visible-light-driven malate production from CO2 and pyruvic acid (2.0 mM) using the system comprising RSH (27 mM), CdS or TiO2 microcrystals fixed in the interlayer spacings of sodium montmorillonite (TiO2/clay) (1.25 mM) as a PS, MV2+ (1.0 mM), NADP+ (0.1 mM), FNR (0.2 U) and ME (1.0 U) in CO2-saturated Tris-buffer (10 mL) has been reported.175 For the system using CdS or TiO2/clay, ca. 1.0 or 0.3 mM of malate is produced after 5 h of irradiation using a 500 W high-pressure Hg arc lamp, respectively.
Visible-light-driven malate production using chlorophyll derivative, zinc chlorin e6 (ZnCe6; chemical structure is shown in Fig. 43) as PS in the system shown in Fig. 52(a) has also been reported.176
After 3 h of irradiation using a 250 W tungsten lamp (λ > 365 nm), 0.65 mM of malate production is observed using the system comprising NADH (3.0 mM), ZnCe6 (50 µM), MV2+ (1.0 mM), FNR (4.0 U), pyruvic acid (10 mM), sodium bicarbonate (10 mM), NADP+ (10 mM) and ME (4.5 U) in 50 mM of Bis–Tris buffer (3.0 mL; pH 8.0).
It has been reported that NADPH can be regenerated even in the absence of MV2+ using PEG-Chl-a (the chemical structure is shown in Fig. 44) as PS, as depicted in Fig. 42(a).167
Using PEG-Chl-a (15 µM) as a PS, malate production in the system comprising sodium ascorbate (6.0 mM), sodium bicarbonate (180 mM), magnesium chloride (15 mM), sodium pyruvate (0.8 mM), NADP+ (3.2 mM), FNR (2.5 U) and ME (5.0 U) in 50 mM of phosphate buffer (10 mL; pH 7.4) during irradiation using a 60 W incandescent lamp from a distance of 10 cm (light intensity, 200 J m−2 s−1) is observed, and 0.15 mM of malate is produced after 4 h irradiation.
In addition, the optical activity of the malate produced in the reaction system introduced in this section, D- or L-, has not been identified.
Next, a research example on visible-light-driven malate production with ME added as a catalyst, as depicted in Fig. 42(b), is introduced. Visible-light-driven L-malate production from sodium bicarbonate and pyruvate using the system comprising triethanol amine (TEOA) as ED, water-soluble zinc porphyrin, ZnTPPS4− as PS, [RhCp*(bpy)(H2O)]2+, NAD+, and ME is introduced.177
After 5 h of irradiation using a 250 W Halogen lamp (λ > 390 nm), 0.16 mM of L-malate production is observed using the system comprising sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM) and ME (0.7 U) in CO2-saturated 500 mM HEPES–NaOH buffer (5.0 mL; pH 7.3). However, 40 µM of L-malate production is observed under Ar-saturated 500 mM HEPES–NaOH buffer (pH 7.3). This result suggests that the equilibrium between gaseous CO2 in the gas phase and bicarbonate ions in the sample solution is important for ME-catalysed L-malate production. In particular, this sealed isochoric system has the problem of a decrease in pressure within the system because gaseous CO2 in the gas phase of the reaction vessel is captured by the weak basic buffer solution. Therefore, visible-light-driven L-malate production has also been reported using an isobaric reaction system attached to a balloon filled with 1 L of CO2 gas.178 After 5 h of irradiation using a 250 W Halogen lamp (λ > 390 nm), ca. 0.16 mM of L-malate production is observed using the system comprising sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM) and ME (0.7 U) in 500 mM HEPES–NaOH buffer (5.0 mL; pH 7.3). A balloon filled with 1 L of CO2 gas is attached to the reaction vessel, and the pressure inside the reaction system is maintained at 1.0 atm. Furthermore, no sodium bicarbonate is added to the reaction sample. The increase in L-malate over the irradiation time indicates that bicarbonate is continuously captured from CO2 gas and fixed to pyruvate by ME together with NADH regenerated by visible light irradiation. Using this isobaric system, L-malate can be produced directly using gaseous CO2 as a raw material.
In addition, visible-light-driven L-malate production using a water-soluble cationic zinc porphyrin, zinc tetrakis(4-N,N,N-trimethylaminophenyl)porphyrin (ZnTMAP4+; chemical structure is shown in Fig. 45), as a PS has also been reported.179
After 5 h of irradiation using a 250 W Halogen lamp (λ > 390 nm), ca. 0.25 mM of L-malate production is observed with the system comprising sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM) and ME (0.7 U) in 500 mM HEPES–NaOH buffer (5.0 mL; pH 7.3). A balloon filled with 1 L of CO2 gas is attached to the reaction vessel, and the pressure inside the reaction system is maintained at 1.0 atm. Using ZnTMAP4+ as a photosensitiser, approximately 1.5 times more L-malate is produced than in the system with ZnTPPS4−. As a stable NAD+ reduction, NADH in a system comprising TEOA and [Cp*Rh(bpy)(H2O)]2+ with ZnTMAP4+ as a PS compared to a system using ZnTPPS4−; thus, the efficiency of visible-light-driven L-malate production is improved using ZnTMAP4+.
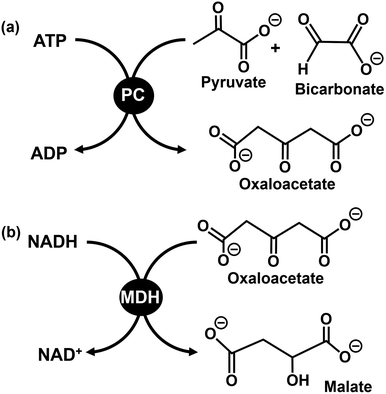
As described above, L-malate production from CO2 and pyruvate using ME as a photoredox catalyst is achieved. However, in the production of L-malate using ME as a catalyst, L-lactate is produced through the reduction of pyruvate under a low concentration of CO2 or bicarbonate. Therefore, a system mimicking the mitochondrial pyruvate malate shuttle,180,181 consisting of pyruvate carboxylase (PC; EC 6.4.1.1) and malate dehydrogenase (MDH; EC 1.1.1.37) in the presence of ATP and NADH, is proposed to produce L-malate using low-concentration CO2 as a feedstock.
Fig. 46 shows the PC-catalysed oxaloacetate production from pyruvate and bicarbonate in the presence of ATP (a) and MDH-catalysed L-malate production based on oxaloacetate reduction in the presence of NADH (b). Using a dual-enzyme consisting of PC and MDH, L-malate can be produced from pyruvate and bicarbonate via oxaloacetate as an intermediate. In addition, using PC as a catalyst, no reduction process of pyruvate is involved to suppress L-lactate production and enable the use of low-concentration CO2 as a raw material.182,183
Visible-light-driven L-malate production from pyruvate and CO2 using a system comprising TEOA, ZnTMAP4+, [RhCp*(bpy)(H2O)]2+, NAD+, ATP, acetyl-CoA, PC and MDH has been reported.184Fig. 47 shows the time dependence of L-malate production using a system comprising sodium pyruvate (5.0 mM), acetyl-CoA (1.0 mM), magnesium chloride (5.0 mM), sodium ATP (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), NAD+ (5.0 mM), [Rh Cp*(bpy)(H2O)]2+ (0.1 mM), PC (1.0 U) and MDH (20 U) in 5.0 mL of 500 mM HEPES–NaOH buffer solution (pH 7.8) under visible-light irradiation using a 250 W halogen lamp (λ > 390 nm). A balloon filled with 1 L of a gas mixture of N2 and CO2 is attached to the reaction vessel, and the pressure inside the reaction system is maintained at 1.0 atm. As shown in Fig. 47, more L-malate production is observed under 15.0% CO2 conditions compared with under 100% CO2 conditions. The change in the pH of the sample solution during the reaction is measured, and the pH decreases from 7.8 to 7.0 under 100% CO2 conditions. However, no change is observed in the pH of the sample solution during reaction under 15.0% CO2 conditions. It has been reported that the rate of visible-light-driven NADH regeneration decreases as the pH of the sample solution decreases. Therefore, the low L-malate production under 100% CO2 conditions is due to a decrease in NADH regeneration induced by a drop in pH during irradiation. By complementing the catalytic function of ME with PC and MDH, visible-light-driven L-malate production using low-concentration CO2 as a direct feedstock is accomplished.
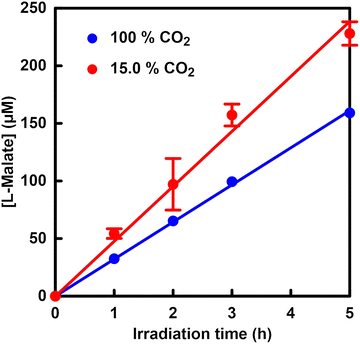 | ||
| Fig. 47 Time-dependent concentration of L-malate in a system comprising sodium pyruvate, acetyl-CoA, magnesium chloride, sodium ATP, TEOA, ZnTMAP4+, NAD+, [Rh Cp*(bpy)(H2O)]2, PC and MDH in an HEPES–NaOH buffer solution (pH 7.8) under visible-light irradiation. The gas phase of the reaction vessel was introduced with 15.0 (red) or 100% CO2 (blue) gas. Reproduced with permission from ref. 184. Copyright 2025, the American Chemical Society. | ||
Finally, an overview of the visible-light-driven L-malate production from CO2 and pyruvate using a photocatalytic dye and enzyme hybrid system, as reviewed in this section, is summarized in Table 2.
| System | Malate production (mM) | Ref. |
|---|---|---|
| RSH (19 mM), [Ru(bpy)3]2+ (21 µM), MV2+ (0.19 mM), NADP+(0.18 mM), pyruvic acid (47 mM), sodium bicarbonate (0.20 M), manganese chloride (95 µM), FNR (0.2 U) and ME (1.33 U) | 1.5 | 156 |
| RSH (27 mM), CdS or TiO2/clay (1.25 mM), MV2+ (1.0 mM), NADP+ (0.1 mM), pyruvic acid (2.0 mM), FNR (0.2 U) and ME (1.0 U) in a CO2-saturated buffer | 0.2 for CdS 0.06 for TiO2/clay | 175 |
| NADH (3.0 mM), ZnCe6 (50 µM), MV2+ (1.0 mM), FNR (4.0 U), pyruvic acid (10 mM), sodium bicarbonate (10 mM), NADP+ (10 mM) and ME (4.5 U) | 0.21 | 162 |
| Sodium ascorbate (6.0 mM), PEG-Chl-a (15 µM), sodium bicarbonate (180 mM), magnesium chloride (15 mM), sodium pyruvate (0.8 mM), NADP+ (3.2 mM), FNR (2.5 U) and ME (5.0 U) | 0.0375 | 167 |
| Sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM) and ME (0.7 U) in CO2-saturated buffer | 0.032 | 177 |
| Sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM) and ME (0.7 U) in CO2-saturated buffer | 0.032 | 178 |
| Sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM) and ME (0.7 U) in CO2-saturated buffer | 0.050 | 179 |
| sodium pyruvate (5.0 mM), acetyl-CoA (1.0 mM), magnesium chloride (5.0 mM), sodium ATP (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), NAD+ (5.0 mM), [RhCp*(bpy)(H2O)]2+ (0.1 mM), PC (1.0 U) and MDH (20 U) | 0.23 (15% CO2) | 184 |
| 0.15 (100% CO2) | ||
Visible-light-driven fumarate production from CO2 and pyruvate with the photocatalytic dye and enzyme hybrid system
Malate is a precursor to PMLA but can be converted by dehydration to fumarate, a precursor to a more versatile biodegradable plastic. In other words, by incorporating FUM in addition to ME, fumarate can be produced from CO2. The production of fumarate from CO2 as a raw material by hybridising a visible-light-driven NADH regeneration system consisting of a photocatalytic dye and a catalyst with a dual enzyme consisting of ME and FUM is introduced in this section.As mentioned above, visible-light-driven fumarate production from CO2 and pyruvate is achieved using a system comprising RSH, [Ru(bpy)3]2+, MV2+, NADP+, FNR, ME and FUM. No other studies have been reported on the production of fumarate using visible-light-driven NADPH regeneration catalysed by FNR in the presence of ME and FUM.
First, visible-light-driven fumarate production from pyruvate and bicarbonate instead of CO2 gas using a system comprising TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+, ME and FUM is introduced.172 The sample solution consists of TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), sodium pyruvate (5.0 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM), ME (0.7 U) and FUM (0.5 U) in 5.0 mL of CO2-saturated 500 mM HEPES–NaOH buffer (5.0 mL; pH 7.8). The sample solution is irradiated with visible light using a 250 W halogen lamp (λ > 390 nm). Using this system, the concentrations of L-malate and fumarate are estimated to be 194 and 48 µM, respectively, after 5 h of irradiation. Although the production of L-malate tends to reach a constant concentration, the fumarate concentration linearly increases during irradiation. Visible-light-driven fumarate production has also been reported using an isobaric reaction system attached to a balloon filled with 1 L of CO2 gas.178 After 5 h of irradiation using a 250 W halogen lamp (λ > 390 nm), ca. 190 and 49 µM of L-malate and fumarate production are observed using a system comprising sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM), ME (0.7 U) and FUM (0.5 U) in 500 mM HEPES–NaOH buffer (5.0 mL; pH 7.8). A balloon filled with 1 L of CO2 gas is attached to the reaction vessel, and the pressure inside the reaction system is maintained at 1.0 atm. Furthermore, no sodium bicarbonate is added to the reaction sample. No significant difference in fumarate production is observed under 100% CO2 conditions in the gas phase and the presence of bicarbonate conditions. However, it has been reported that as the CO2 ratio in the mixed gas in the balloon decreases, the production of L-malate and fumarate also decreases.
Using a system comprising TEOA, ZnTPPS4−, [RhCp*(bpy)(H2O)]2+, NAD+, ME and FUM, the concentration of fumarate produced remains significantly lower than that of L-malate, whether CO2 gas or bicarbonate is used as the feedstock. For example, it has been reported that the addition of phosphate improves the catalytic activity of FUM for the production of fumarate based on the dehydration of L-malate.185 In other words, anionic or cationic substances may control the catalytic activity of FUM. Thus, the effect of water-soluble zinc porphyrin on the FUM-catalysed dehydration of L-malate to produce fumarate has been investigated. The effects of water-soluble zinc porphyrins, ZnTPPS4− and ZnTMAP4+, as well as zinc tetra(4-carboxyphenyl)porphyrin (ZnTCPP4−) and zinc tetrakis(4-methylpyridyl)porphyrin (ZnTMPyP4+) on the enzyme activity of FUM for fumarate production have been investigated. Fig. 48 shows the chemical structures of ZnTCPP4− and ZnTMPyP4+.
As shown in Fig. 49, the addition of cationic zinc porphyrins, ZnTMPyP4+ or ZnTMAP4+, did not alter fumarate production from L-malate by FUM compared to the control experiment. By adding anionic zinc porphyrins, ZnTCPP4− or ZnTPPS4−, in contrast, fumarate production from L-malate with FUM is suppressed. In particular, fumarate production with FUM is strongly inhibited in the presence of ZnTPPS4−.
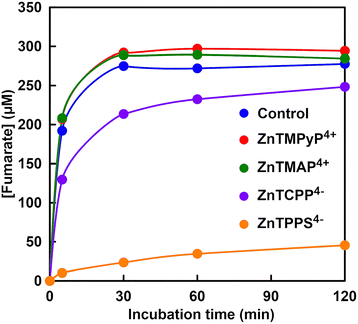 | ||
| Fig. 49 Time-dependent fumarate production from L-malate with FUM in the presence of zinc porphyrin during incubation. Reproduced with permission from ref. 186. Copyright 2023, The Royal Society of Chemistry. | ||
It is suggested that the sulfo-group bonded to zinc porphyrin has a stronger effect on the catalytic activity of FUM than that of the carboxy-group.186
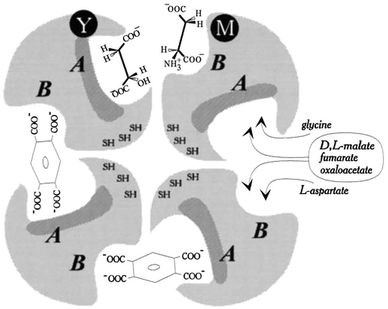
Here, we focus on the structural interactions between FUM and water-soluble zinc porphyrins. As presented in this section, FUM is an enzyme derived from porcine heart. The three-dimensional structures of porcine heart-derived and related FUMs have been reported.187–189 It has been reported that porcine heart-derived FUM is composed of four subunits. FUM has four active sites (site A) with high affinity for substrates, as well as an additional binding site (site B) with low affinity for substrates.189Fig. 50 is a schematic diagram showing the active high-affinity substrate binding sites (A) and low-affinity binding sites (B) in the FUM.
 | ||
| Fig. 50 Schematic showing the active high-affinity substrate binding sites (A) and low-affinity binding sites (B) in FUM. Reproduced with permission from ref. 189. Copyright 1998 ASBMB. Currently published by Elsevier Inc.; originally published by the American Society for Biochemistry and Molecular Biology. Creative Commons CC-BY-NC license. | ||
For example, tetracarboxylic acid, benzene-1,2,4,5-tetracarboxylate and sulfate have been reported as strong inhibitors for FUM.189 In particular, it is found that one molecule of benzene-1,2,4,5-tetracarboxylic acid can bind to both sites A and B simultaneously, and that four carboxyl groups of benzene-1,2,4,5-tetracarboxylic acid per FUM molecule bind with high affinity, acting as bridges between the two subunits. These results suggest that the four benzenesulfonate groups of ZnTPPS4− also bind with high affinity to the substrate-binding sites (sites A and B) of FUM, inhibiting the dehydration of L-malate and reducing the production of fumarate.
Therefore, a visible-light-driven fumarate production system has been reported that uses ZnTMAP4+ as PS instead of ZnTPPS4−, which does not affect FUM enzyme activity.179 Visible-light-driven fumarate production has also been reported using an isobaric reaction system attached to a balloon filled with 1 L of CO2 gas.179,190 After 5 h of irradiation using a 250 W halogen lamp (λ > 390 nm), ca. 97 µM of fumarate production is observed using a system comprising sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM), ME (0.7 U) and FUM (0.5 U) in 500 mM HEPES–NaOH buffer (5.0 mL; pH 7.8). A balloon filled with 1 L of CO2 gas is attached to the reaction vessel, and the pressure inside the reaction system is maintained at 1.0 atm. Fig. 51 shows the fumarate concentration using ZnTMAP4+ or ZnTPPS4− after 5 h of irradiation.
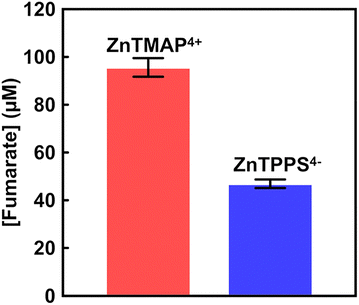 | ||
| Fig. 51 Concentration of fumarate production in a system comprising sodium pyruvate, magnesium chloride, TEOA, [RhCp*(bpy)(H2O)]2+, NAD+, ME, FUM and CO2 gas in the presence of ZnTMAP4+ (red) or ZnTPPS4− (blue) after 5 h of irradiation. Reproduced with permission from ref. 179. Copyright 2023, The Royal Society of Chemistry. | ||
The efficiency of visible-light-driven fumarate production from pyruvate and gaseous CO2 using ZnTMAP4+ as the PS in a system combining NAD+ reduction with [RhCp*(bpy)(H2O)]2+ and a dual-biocatalyst consisting of ME and FUM in the presence of TEOA is improved by approximately 2.0 times that produced in the system using ZnTPPS4−, a conventional PS.
As a relevant study of this system, fumarate production from pyruvate and bicarbonate using a core–shell type of benzodiimidazole oligomer-BaSO4-immobilised [RhCp*(bpy)(H2O)]2+ (BaPP@BDO-Rh) instead of water-soluble zinc porphyrin as a visible-light sensitising material with dual-biocatalyst consisting of ME and FUM has been reported, as shown in Fig. 52(A).191 In this study, ME and FUM are expressed in E. coli strain DE3 using the pET28a vector; then, the enzymes are purified for use in this reaction. The ME and FUM are designated MaeB and FumC, respectively. The sample solution consists of TEOA (1.0 M), 1 g L−1 BaPP@BDO-Rh, NAD+ (1.0 mM), sodium pyruvate (5.0 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM), MaeB (1.0 U) and FumC (1.0 U) in 3.0 mL of 100mM phosphate buffer. The sample solution is irradiated using a 100 W xenon lamp at room temperature as a visible light source. Using this system, the concentrations of L-malate and fumarate are estimated to be 114 and 63 µM, respectively, after 1 h of irradiation, as shown in Fig. 52(B).
 | ||
| Fig. 52 Schematic of the visible-light-driven fumarate production from pyruvate and CO2 using a system comprising TEOA, BaPP@BDO-Rh, NAD+, MaeB and FumC (A). Time-dependent malate and fumarate production from pyruvate and CO2 using the system comprising TEOA, BaPP@BDO-Rh, NAD+, MaeB and FumC (B). Reproduced with permission from ref. 191. Copyright 2023, The Royal Society of Chemistry. | ||
In this study, ME and FUM are expressed in E. coli strain DE3 using the pET28a vector; then, the enzymes are purified for use in this reaction. The ME and FUM are designated MaeB and FumC, respectively. The sample solution consists of TEOA (1.0 M), 1 g L−1 BaPP@BDO-Rh, NAD+ (1.0 mM), sodium pyruvate (5.0 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM), MaeB (1.0 U) and FumC (1.0 U) in 3.0 mL of 100 mM phosphate buffer. The sample solution is irradiated using a 100 W xenon lamp at room temperature as a visible light source. Using this system, the concentrations of L-malate and fumarate are estimated to be 114 and 63 µM, respectively, after 1 h of irradiation, as shown in Fig. 52(B).
However, no L-malate and fumarate production are observed under dark conditions. It is believed that malate production under irradiation and dark conditions in this reference is reversed. Moreover, 79 µM of fumarate is produced using this system after 3 h of irradiation. The production of L-malate and fumarate tends to reach a constant concentration during irradiation in this system.
Visible-light-driven fumarate production from pyruvate and CO2 using a system comprising TEOA, ZnTMAP4+, [RhCp*(bpy)(H2O)]2+, NAD+, ATP, acetyl-CoA, PC, MDH and FUM has been reported.184L-Malate and fumarate production are observed using a system comprising sodium pyruvate (5.0 mM), acetyl-CoA (1.0 mM), magnesium chloride (5.0 mM), sodium ATP (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), NAD+ (5.0 mM), [RhCp*(bpy)(H2O)]2+ (0.1 mM), PC (1.0 U), MDH (20 U) and FUM (0.5 U) in 5.0 mL of 500 mM HEPES–NaOH buffer solution (pH 7.8) under visible-light irradiation with a 250 W halogen lamp (λ > 390 nm). A balloon filled with 1 L of a gas mixture of N2 and CO2 is attached to the reaction vessel, and the pressure inside the reaction system is maintained at 1.0 atm. Under conditions using a gas mixture containing 15% CO2, ca. 213 and 57.6 µM of L-malate and fumarate are produced after 5 h of irradiation, respectively. Furthermore, no significant difference is observed between L-malate and fumarate production under mixed gas conditions containing 50% CO2.
However, it has also been shown that L-malate and fumarate production is reduced by approximately half under mixed gas conditions containing 100% CO2 or 2% CO2. By complementing the catalytic function of ME with PC and MDH and using it together with FUM, visible-light-driven fumarate production can be achieved using low-concentration CO2 (approximately 15%) as a direct feedstock.
Finally, an overview of the visible-light-driven fumarate production from CO2 and pyruvate using a photocatalytic dye and enzyme hybrid system, as reviewed in this section, is summarized in Table 3.
| System | Fumarate production (µM) | Ref. |
|---|---|---|
| TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), sodium pyruvate (5.0 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM), ME (0.7 U) and FUM (0.5 U) | 48 (5 h) | 177 |
| sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTPPS4− (10 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM), ME (0.7 U) and FUM (0.5 U) in a CO2-saturated buffer | 49 (5 h) | 178 |
| sodium pyruvate (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), [RhCp*(bpy)(H2O)]2+ (10 µM), NAD+ (0.5 mM), magnesium chloride (5.0 mM), ME (0.7 U) and FUM (0.5 U) in a CO2-saturated buffer | 97 (5 h) | 179 |
| TEOA (1.0 M), 1 g L−1 BaPP@BDO-Rh, NAD+ (1.0 mM), sodium pyruvate (5.0 mM), sodium bicarbonate (100 mM), magnesium chloride (5.0 mM), MaeB (1.0 U) and FumC (1.0 U) | 63 (1 h) | 191 |
| sodium pyruvate (5.0 mM), acetyl-CoA (1.0 mM), magnesium chloride (5.0 mM), sodium ATP (5.0 mM), TEOA (0.2 M), ZnTMAP4+ (50 µM), NAD+ (5.0 mM), [RhCp*(bpy)(H2O)]2+ (0.1 mM), PC (1.0 U), MDH (20 U) and FUM (0.5 U) in a 15% CO2-saturated buffer | 58 (5 h) | 184 |
Major challenges and future trends
Here, the problems and prospects for future demonstrations of the systems introduced in this study will be discussed. First, we discuss the challenges to the practical application of visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system. The reaction scale of visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system will be discussed. As described in the paper, the reaction scale of the hybrid system is approximately 1.0 L at the maximum laboratory level. Scaling up this system for material production involves several significant challenges, including maintaining product quality stability, ensuring process efficiency, and, in some cases, complying with regulatory requirements.In addition, although Xe and halogen lamps are currently used as visible light sources, it will be necessary to directly utilize solar-light in the future, and the design of the reaction vessel for this purpose will be an important factor. Visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system is still under development, and future developments in chemical engineering will likely pave the way for its practical application through the design of the reaction apparatus. However, a hybrid system has the advantage of efficiently promoting CO2 conversion within microbial cells through induced electrons from solar energy efficiently absorbed using semiconductor photocatalysts and organic dyes. This suggests the potential for more efficient utilization of solar energy compared to CO2 conversion by photosynthetic microorganisms such as microalgae. In other words, it would be possible to apply a CO2 conversion apparatus that utilizes the photosynthetic function of microalgae and other organisms, which is already in practical use, to a hybrid system.
Visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system is attracting attention as an innovative technology that can achieve both the resolution of global challenges and economic growth, but currently, high costs are a major challenge. In particular, this system relies on the growth of microorganisms, resulting in low productivity and increased equipment and purification costs. To overcome these challenges, it is necessary to work on improving large-scale culture technology, advanced separation and purification technology, and the development of bioreactors. Furthermore, the production of high-value-added biodegradable plastics and their precursors from CO2 will further enhance the value of this system.
Next, we focus on the direct use of CO2 gas in hybrid systems. In the visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system, a mixed gas with an approximate N2 to CO2 ratio of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 is used as the raw material. This is a mixed gas composition optimized for substance production in microbial cells. In some cases, adding hydrogen to a gas mixture can improve the efficiency of producing the desired substance. In particular, in the case of visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system, the use of mixed gases, such as exhaust gases emitted from coal-fired power plants, has not yet been considered.
20 is used as the raw material. This is a mixed gas composition optimized for substance production in microbial cells. In some cases, adding hydrogen to a gas mixture can improve the efficiency of producing the desired substance. In particular, in the case of visible-light-driven plastic precursor production from CO2 using a photocatalyst and microbial cell hybrid system, the use of mixed gases, such as exhaust gases emitted from coal-fired power plants, has not yet been considered.
In a visible-light-driven biodegradable plastic precursor production system using photocatalytic dyes and multi-enzymes, however, it is possible to change the composition of the CO2 gas used as a raw material. In other words, this system has the potential to use CO2 diluted with N2, etc., emitted from coal-fired power plants, as a raw material. It has been reported that exhaust gases contain approximately 0.02–0.3% NOx and SO2.192 Here, a significant advantage of the visible-light-driven biodegradable plastic precursor production system using photocatalytic dyes and multi-enzymes is that it allows for the capture of CO2 gas in the reaction solution using a weakly basic buffer. In addition, because a buffer solution is used, even if NOx or SO2 is captured, the pH of the reaction solution does not change, and it is expected that this does not significantly affect the total process.
Finally, we discuss the hybrid systems introduced in this study from the perspective of life cycle analysis (LCA). There are currently no reported cases of LCA estimation for photocatalysts, biocatalysts, or microbial cell hybrid systems. Therefore, this approach depending on reports on life cycle assessments (LCAs) for hydrogen production based on water splitting using semiconductor photocatalysts is discussed.193 According to this report, in the production of 1 kg of hydrogen using semiconductor photocatalysts, greenhouse gases are mainly released during the preparation of the photocatalyst (95.87%). However, we discuss LCA for biocatalytic CO2 conversion based on bioelectrochemical methods using microbial cells.194 LCA evaluations of this system indicate that its carbon footprint is comparable to that of fermentation or plant-based manufacturing methods. In other words, it goes without saying that a hybrid system comprising semiconductor photocatalysts and microbial cells also has the potential to contribute to CO2 reduction. These conclusions suggest that at this stage, there are many hurdles to overcome before hybrid systems can achieve significant CO2 reduction. However, the fact that value-added substances can be produced from CO2 and biomass-derived compounds using light energy as a driving force suggests that it would be better to shift the focus from CO2 reduction to its effective use as a raw material.
Conclusion and outlook
In this review article, focusing on biodegradable plastics and their precursor production from gaseous CO2 using systems comprising visible-light-driven redox reactions with biocatalytic processes, the following types are outlined.(1) The visible-light-driven production of biodegradable plastic precursors such as acetate, shikimic acid and acetoin from CO2 using a hybrid system comprising the inorganic- or organic-based semiconductor photocatalyst and a microbial cell.
(2) The visible-light-driven production of biodegradable plastic PHB from CO2 using a hybrid system comprising inorganic- or organic-based semiconductor photocatalysts and a microbial cell.
(3) The visible-light-driven production of biodegradable plastic precursor 3-hydroxybutyrate from acetone and CO2 using a combination of NAD+ reduction system of triethanolamine, water-soluble zinc porphyrin, [Cp*Rh(bpy)(H2O)]2+ and a cell extract including dual-enzymes.
(4) The visible-light-driven production of biodegradable plastic precursors L-malate fumarate from bio-based pyruvate and CO2 using a photoredox system consisting of an electron donor, a photosensitizer, an electron mediator and an enzyme-based biocatalyst.
In the future, it is expected that innovative technologies with a photo/biocatalytic hybrid system will be developed for the effective and practical use of the visible-light-driven production of biodegradable plastics and their precursors from gaseous CO2 and small organic molecules. Therefore, the research introduced in this article contributes to the CO2 fixation, and to alternative plastic or its precursor production for a sustainable society. These systems can fix gaseous CO2 in small organic molecules and convert it into high-value-added materials, resulting in the long-term storage of CO2 in molecules.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results have been included, and no new data were generated or analysed as part of this review article.Acknowledgements
Our work introduced in this review is partially supported by Grant-in-Aid for Specially Promoted Research (23H05404), Scientific Research (B) (25K01584), (23K23140), (22H01872), and (22H01871).Notes and references
- G. Q. Chen and M. K. Patel, Chem. Rev., 2012, 112, 2082 CrossRef CAS PubMed
.
- R. Kajaste and P. Oinas, AIMS Environ. Sci., 2021, 8, 371 CAS
.
- J. Hammer, M. H. Kraak and J. R. Parsons, Rev. Environ. Contam. Toxicol., 2012, 220, 1 CAS
.
- T. R. Walker and D. Xanthos, Resour., Conserv. Recycl., 2018, 133, 99 CrossRef
.
- P. Stoett, V. M. Scrich, C. I. Elliff, M. M. Andrade, N. de, M. Grilli and A. Turra, World Dev., 2024, 184, 106756 CrossRef
.
- R. Ciriminna and M. Pagliaro, ChemistryOpen, 2020, 9, 8 CrossRef CAS PubMed
.
-
Handbook of Bioplastics and Biocomposites Engineering Applications, ed S. Pilla, Wiley-Scrivener, Salem (MA), 2011 Search PubMed
.
- P. Skoczinski, M. Carus, G. Tweddle, P. Ruiz, N. Hark, A. Zhang, D. de Guzman, J. Ravenstijn, H. Käb and A. Raschka, Bio-based Building Blocks and Polymers – Global Capacities, Production and Trends, 2023–2028, GmbH, Hürth, Germany, 2024, 03 Search PubMed.
- C. Gioia, G. Giacobazzi, M. Vannini, G. Totaro, L. Sisti, M. Colonna, P. Marchese and A. Celli, ChemSusChem, 2021, 14, 4167 CrossRef CAS PubMed
.
- L. Filiciotto and G. Rothenberg, ChemSusChem, 2021, 14, 56 CrossRef CAS PubMed
.
- T. P. Haider, C. Völker, J. Kramm, K. Landfester and F. R. Wurm, Angew. Chem., Int. Ed., 2019, 58, 50 CrossRef CAS PubMed
.
- S. Agarwal, Macromol. Chem. Phys., 2020, 221, 2000017 CrossRef CAS
.
- A. Ammala, Prog. Polym. Sci., 2011, 36, 1015 CrossRef CAS
.
- A. S. Al Hosni, J. K. Pittman and G. D. Robso, Waste Manage., 2019, 97, 105 CrossRef CAS PubMed
.
- T. K. Chua, M. Tseng and M. K. Yang, AMB Express, 2013, 3, 8 CrossRef PubMed
.
- L. Cosgrove, P. L. McGeechan, G. D. Robson and P. S. Handley, Appl. Environ. Microbiol., 2007, 73, 5817 CrossRef CAS PubMed
.
- M. Karamanlioglu, A. Houlden and G. D. Robson, Int. Biodeter. Biodegr., 2014, 95, 301 CrossRef CAS
.
- V. Nagarajan, A. K. Mohanty and M. Misra, ACS Sustainable Chem. Eng., 2016, 4, 2899 CrossRef CAS
.
- O. Martin and L. Avérous, Polymer, 2001, 42, 6209 CrossRef CAS
.
- H. R. Kricheldorf and J. M. Jonté, Polym. Bull., 1983, 9, 276 CAS
.
- Y. K. Jung, T. Y. Kim and T. Yong, Biotechnol. Bioeng., 2009, 105, 161 CrossRef PubMed
.
- S. Bengtsson, A. Werker, M. Christensson and T. Welander, Bioresour. Technol., 2008, 99, 509 CrossRef CAS PubMed
.
- S. Mohapatra, B. Sarkar, D. P. Samantaray, A. Daware, S. Maity, S. Pattnaik and S. Bhattacharjee, Environ. Technol., 2017, 38, 1 CrossRef
.
- S. Bengtsson, A. Werker and T. Welander, Water Sci. Technol., 2008, 58, 323 CrossRef CAS PubMed
.
- F. Morgan-Sagastume, A. Karlsson, P. Johansson, S. Pratt, N. Boon, P. Lant and A. Werker, Water Res., 2010, 44, 5196 CrossRef CAS PubMed
.
- F. Morgan-Sagastume, M. Hjort, D. Cirne, F. Gérardin, S. Lacroix, G. Gaval, L. Karabegovic, T. Alexandersson, P. Johansson, A. Karlsson, S. Bengtsson, M. V. Arcos-Hernández, P. Magnusson and A. Werker, Bioresour. Technol., 2015, 181, 78 CrossRef CAS PubMed
.
- P. Chakravarty, V. Mhaisalkar and T. Chakrabarti, Bioresour. Technol., 2010, 101, 2896 CrossRef CAS PubMed
.
- G. Q. Chen, Chem. Soc. Rev., 2009, 38, 2434 RSC
.
- K. Sudesh, H. Abe and Y. Doi, Prog. Polym. Sci., 2000, 25, 1503 CrossRef CAS
.
- S. Chanprateep, J. Biosci. Bioeng., 2010, 110, 621 CrossRef CAS PubMed
.
- R. Rai, T. Keshavarz, J. A. Roether, A. R. Boccaccini and I. Roy, Mater. Sci. Eng. R, 2011, 72, 29 CrossRef
.
- P. K. Samantaray, A. Little, D. M. Haddleton, T. McNally, B. Tan, Z. Sun, W. Huang, Y. Ji and C. Wan, Green Chem., 2020, 22, 4055 RSC
.
- S. Mohapatra, S. Maity, H. R. Dash, S. Das, S. Pattnaik, C. C. Rath and D. Samantaray, Biochem. Biophys. Rep., 2017, 12, 206 Search PubMed
.
- K. Yamane, H. Sato, Y. Ichikawa, K. Sunagawa and Y. Shigaki, Polym. J., 2014, 46, 76 CrossRef
.
- E. Göktürk, A. G. Pemba and S. A. Miller, Polym. Chem., 2015, 6, 3918 RSC
.
- M. Labet and W. Thielemans, Chem. Soc. Rev., 2009, 38, 3484 RSC
.
-
J. O. Iroh, Polymer Data Handbook, ed J. E. Mark, Oxford University Press, New York, 1999, pp. 361–362 Search PubMed
.
- H. Bittiger, R. H. Marchessault and W. D. Niegisch, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1970, 26, 1923 CrossRef CAS
.
- R. A. Gross and B. Kalra, Science, 2002, 297, 803 CrossRef CAS PubMed
.
- Y. Ikada and H. Tsuji, Macromol. Rapid Commun., 2000, 21, 117 CrossRef CAS
.
- T. F. Nelson, R. Baumgartner, M. Jaggi, S. M. Bernasconi, G. Battagliarin, C. Sinkel, A. Künkel, H. P. E. Kohler, K. McNeill1 and M. Sander, Nat. Commun., 2022, 13, 5691 CrossRef CAS PubMed
.
- K. S. Savitha, B. R. Paghadar, M. S. Kumar and R. L. Jagadish, Polym. Chem., 2022, 13, 3562 RSC
.
- Y. Doi, K. Kasuya, H. Abe, N. Koyama, I. Shin-ichi, T. Koichi and Y. Yoshida, Polym. Degrad. Stab., 1996, 51, 281 CrossRef CAS
.
- K. Kasuya, K. Takagi, S. Ishiwatari, Y. Yoshida and Y. Doi, Polym. Degrad. Stab., 1998, 59, 327 CrossRef CAS
.
- A. Kilic, E. Yasar, H. İ. Önal, F. Koyuncu, M. Aydemir and F. Durap, Energy Fuels, 2025, 39, 23241 CrossRef CAS
.
- F. Tufano, C. Napolitano, M. Mazzeo, F. Grisi and M. Lamberti, Biomacromolecules, 2024, 25, 4523 CrossRef CAS PubMed
.
- S. Ye, S. Wang, L. Lin, M. Xiao and Y. Meng, Adv. Ind. Eng. Polym. Res., 2019, 2, 143 Search PubMed
.
- Y. Y. Wang, J. W. Fan and D. J. Darensbourg, Angew. Chem., Int. Ed., 2015, 54, 10206 CrossRef CAS PubMed
.
- D. Zhang, W. Wang, Z. Wang, D. Song, S. Liu, Y. Chen, X. Ma and L. Xia, J. CO2 Util., 2025, 102, 103236 CrossRef CAS
.
- A. Stirbet, D. Lazár, Y. Guo and G. Govindjee, Ann. Bot., 2019, 126, 511 CrossRef PubMed
.
- A. N. Tikhonov and W. K. Subczynski, Cell Biochem. Biophys., 2018, 77, 47 CrossRef PubMed
.
- K. Asada, Annu. Rev. Plant Physiol. Plant Mol. Biol., 1999, 50, 601 CrossRef CAS PubMed
.
- T. Shikanai, Annu. Rev. Plant Biol., 2007, 58, 199 CrossRef CAS PubMed
.
- Y. Okegawa, Y. Kagawa, Y. Kobayashi and T. Shikanai, Plant Cell Physiol., 2008, 49, 825 CrossRef CAS PubMed
.
- B. V. Trubitsin, A. V. Vershubskii, A. I. Priklonskii and A. N. Tikhonov, J. Photochem. Photobiol., B, 2015, 152, 400 CrossRef CAS PubMed
.
- J. Bassham, A. Benson and M. Calvin, J. Biol. Chem., 1950, 185, 781 CrossRef CAS PubMed
.
- J. K. Sainis, D. N. Dani and G. K. Dey, J. Plant Physiol., 2003, 160, 23 CrossRef CAS PubMed
.
- R. Hidese, M. Matsuda, T. Osanai, T. Hasunuma and A. Kondo, ACS Synth. Biol., 2020, 9, 260 CrossRef CAS PubMed
.
- S. Ito, N. Koyama and T. Osanai, Sci. Rep., 2019, 9, 6038 CrossRef PubMed
.
- K. Maeda and K. Domen, J. Phys. Chem. Lett., 2010, 1, 2655 CrossRef CAS
.
- S. J. A. Moniz, S. A. Shevlin, D. J. Martin, Z. X. Guoand and J. Tang, Energy Environ. Sci., 2015, 8, 731 RSC
.
- K. Takanabe, ACS Catal., 2017, 7, 8006 CrossRef CAS
.
- Y. Wang, H. Suzuki, J. Xie, O. Tomita, D. J. Martin, M. Higashi, D. Kong, R. Abe and J. Tang, Chem. Rev., 2018, 118, 5201 CrossRef CAS PubMed
.
- Q. Wang and K. Domen, Chem. Rev., 2020, 120, 919 CrossRef CAS PubMed
.
- K. Kaiya, Y. Ueki, H. Kawamoto, K. Watanabe, S. Yoshino, Y. Yamaguchi and A. Kudo, Chem. Sci., 2024, 15, 16025 RSC
.
- R. N. Ivanovsky, Y. I. Fal, I. A. Berg, N. V. Ugolkova, E. N. Krasilnikova, O. I. Keppen, L. M. Zakharchuc and A. M. Zyakun, Microbiology, 1999, 145, 1743 CrossRef CAS PubMed
.
- H. G. Wood, FASEB J., 1990, 5, 156 CrossRef PubMed
.
- M. C. Evans, B. B. Buchanan and D. I. Arnon, Proc. Natl. Acad. Sci. U. S. A., 1966, 55, 928 CrossRef CAS PubMed
.
- H. Berberoğlu, N. Barra, L. Pilon and J. Jay, J. Appl. Microbiol., 2007, 104, 105 Search PubMed
.
- W. Eisenreich, G. Strauss, U. Werz, G. Fuchs and A. Bacher, Eur. J. Biochem., 1993, 215, 619 CrossRef CAS PubMed
.
- S. W. Ragsdale, J. E. Clark, L. G. Ljungdahl and H. L. Drake, J. Biol. Chem., 1983, 258, 2364 CrossRef CAS PubMed
.
- S. W. Ragsdale and E. Pierce, Biochim. Biophys. Acta, 2008, 1784, 1873 CrossRef CAS PubMed
.
- H. H. Cheng, J. C. Syu, S. Y. Tien and L. M. Whang, Bioresour. Technol., 2018, 226, 229 CrossRef PubMed
.
- Y. Xiea, S. Erşanb, X. Guana, J. Wanga, J. Shac, S. Xua, J. A. Wohlschlegelc, J. O. Park and C. Liu, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2308373120 CrossRef PubMed
.
- M. Hermann, A. Teleki, S. Weitz, A. Niess, A. Freund, F. R. Bengelsdorf and R. Takors, Microb. Biotechnol., 2020, 13, 1831 CrossRef CAS PubMed
.
- K. K. Sakimoto, A. B. Wong and P. Yang, Science, 2016, 351, 74 CrossRef CAS PubMed
.
- D. P. Cunningham and L. L. Lundie Jr., Appl. Environ. Microbiol., 1993, 59, 7 CrossRef CAS PubMed
.
- H. Zhang, H. Liu, Z. Tian, D. Lu, Y. Yu, S. Cestellos-Blanco, K. K. Sakimoto and P. Yang, Nat. Nanotechnol., 2018, 13, 900 CrossRef CAS PubMed
.
- K. K. Sakimoto, S. J. Zhang and P. Yang, Nano Lett., 2016, 16, 5883 CrossRef CAS PubMed
.
- P. Gai, W. Yu, H. Zhao, R. Qi, F. Li, L. Liu, F. Lv and S. Wang, Angew. Chem., Int. Ed., 2020, 59, 7224 CrossRef CAS PubMed
.
- B. Möller, R. Oßmer, B. H. Howard, G. Gottschalk and H. Hippe, Arch. Microbiol., 1984, 139, 388 CrossRef
.
- K. Nevin, T. L. Woodard, A. E. Franks, Z. M. Summers and D. R. Lovley, mBio, 2010, 1, e00103 CrossRef PubMed
.
- D. R. Lovley and K. P. Nevin, Curr. Opin. Biotechnol, 2013, 24, 385 CrossRef CAS PubMed
.
- Y. He, S. Wang, X. Han, J. Shen, Y. Lu, J. Zhao, C. Shen and L. Qiao, ACS Appl. Mater. Interfaces, 2022, 14, 23364 CrossRef CAS PubMed
.
- Q. Wang, S. Kalathil, C. Pornrungroj, C. D. Sahm and E. Reisner, Nat. Catal., 2022, 5, 633 CrossRef CAS
.
- N. Wen, Q. Jiang and D. Liu, Sci. Adv., 2024, 10, eadp8567 CrossRef CAS PubMed
.
- S. Günes, H. Neugebauer and N. S. Sariciftci, Chem. Rev., 2007, 107, 1324 CrossRef PubMed
.
- G. Li, V. Shrotriya, J. Huang, Y. Yao, T. Moriarty, K. Emery and Y. Yang, Nat. Mater., 2005, 4, 864 CrossRef CAS
.
- N. Wen, Q. Jiang, J. Cui, H. Zh, B. Ji and D. Liu, Nano Today, 2022, 47, 101681 CrossRef CAS
.
- S. Tamang, C. Lincheneau, Y. Hermans, S. Jeong and P. Reiss, Chem. Mater., 2016, 28, 2491 CrossRef CAS
.
- M. Rafipoor, H. Tornatzky, D. Dupont, J. Maultzsch, M. D. Tessier, Z. Hens and H. Lange, J. Chem. Phys., 2019, 151, 154704 CrossRef PubMed
.
- M. D. Tessier, D. Dupont, K. De Nolf, J. De Roo and Z. Hens, Chem. Mater., 2015, 27, 4893 CrossRef CAS
.
- Y. Ding, J. R. Bertram, C. Eckert, R. R. Bommareddy, R. Patel, A. Conradie, S. Bryan and P. Nagpal, Energy Environ. Sci., 2019, 141, 10272 CAS
.
- C. Liu, J. J. Gallagher, K. K. Sakimoto, E. M. Nichols, C. J. Chang, M. C. Chang and P. Yang, Nano Lett., 2015, 15, 3634 CrossRef CAS PubMed
.
- J. Kim, S. Cestellos-Blanco, Y. X. Shen, R. Cai and P. Yang, Nano Lett., 2022, 22, 5503 CrossRef CAS PubMed
.
- S. Cheemanapalli, R. Mopuri, R. Golla, C. M. Anuradha and S. K. Chitta, Biomed. Pharmacother., 2018, 108, 547 CrossRef PubMed
.
- J. Guo, M. Suástegui, K. K. Sakimoto, V. M. Moody, G. Xiao, D. G. Nocera and N. S. Joshi, Science, 2018, 362, 813 CrossRef CAS PubMed
.
- M. Suástegui, C. Yu Ng, A. Chowdhury, W. Sun, M. Cao, E. House, C. D. Maranas and Z. Shao, Metab. Eng., 2017, 42, 134 CrossRef PubMed
.
- R. Hatti-Kaul, U. Törnvall, L. Gustafsson and P. Börjesson, Trends Biotechnol., 2007, 25, 119 CrossRef CAS PubMed
.
- Z. Xiao and J. R. Lu, Biotechnol. Adv., 2014, 32, 492 CrossRef CAS PubMed
.
- S. Maina, A. A. Prabhu, N. Vivek, A. Vlysidis, A. Koutinas and V. Kumar, Biotechnol. Adv., 2022, 54, 107783 CrossRef CAS PubMed
.
- W. Meng, C. Ma, P. Xu and C. Gao, Trends Biotechnol., 2022, 40, 958 CrossRef CAS PubMed
.
- Z. Guo, X. Zhao, Y. He, T. Yang, H. Gao, G. Li, F. Chen, M. Sun, J. K. Lee and L. Zhang, J. Microbiol. Biotechnol., 2016, 27, 92 CrossRef PubMed
.
- M. Otagiri, S. Ui, Y. Takusagawa, T. Ohtsuki, G. Kurisu and M. Kusunoki, FEBS Lett., 2010, 584, 219 CrossRef CAS PubMed
.
- Z. Wang, Q. Song, M. Yu, Y. Wang, B. Xiong, Y. Zhang, J. Zheng and X. Ying, Appl. Microbiol. Biotechnol., 2014, 98, 641 CrossRef CAS PubMed
.
- Y. Tian, Z. Guo, J. He, D. Xu, W. W. Li, S. Cheng and H. Song, J. CO2 Util., 2025, 93, 103051 CrossRef CAS
.
- B. Fu, X. Mao, Y. Park, Z. Zhao, T. Yan, W. Jung, D. H. Francis, W. Li, B. Pian, F. Salimijazi, M. Suri, T. Hanrath, B. Barstow and P. Chen, Nat. Chem., 2023, 15, 1400 CrossRef CAS PubMed
.
- J. Ackermann, S. Müller, A. Lösche, T. Bley and W. Babel, J. Biotechnol., 1995, 39, 9 CrossRef CAS
.
- C. R. Hankermeyerand and R. S. Tjeerdema, Rev. Environ. Contam. Toxicol., 1991, 159, 1 Search PubMed
.
- R. T. Chan, R. A. Russell, H. Marçal, T. H. Lee, P. J. Holden and L. J. R. Foster, Biomacromolecules, 2013, 15, 339 CrossRef PubMed
.
- E. Markl, H. Grünbichler and M. Lackner, Novel Tech. Nut. Food Sci., 2018, 2, 1 Search PubMed
.
- S. P. Mohandas, L. Balan, N. Lekshmi, S. S. Cubelio, R. Philip and I. S. Bright Singh, J. Appl. Microbio., 2017, 122, 698 CrossRef CAS PubMed
.
- A. A. Alarfaj, M. Arshad, E. N. Sholkamy and M. A. Munusamy, Braz. Arch. Bio. Tech., 2015, 58, 781 CrossRef CAS
.
- F. A. P. Tassia, L. J. Marcos, D. B. Watanabe, A. Bonomi, L. K. Quines, W. Schmidell and G. M. F. de Aragao, Bioch. Eng. J., 2019, 146, 97 CrossRef
.
- R. S. Sasidharan, S. G. Bhat and M. Chandrasekaran, Ann. Microbiol., 2015, 65, 455 CrossRef CAS
.
- A. Pohlmann, W. F. Fricke, F. Reinecke, B. Kusian, H. Liesegang, R. Cramm, T. Eitinger, C. Ewering, M. Pötter, E. Schwartz, A. Strittmatter, I. Voss, G. Gottschalk, A. Steinbüchel, B. Friedrich and B. Bowien, Nat. Biotechnol., 2006, 24, 1257 CrossRef PubMed
.
- C. Liu, B. C. Colón, M. Ziesack, P. A. Silver and D. G. Nocera, Science, 2016, 352, 1210 CrossRef CAS PubMed
.
- M. Xu, P. L. Tremblay, L. Jiang and T. Zhang, Green Chem., 2019, 21, 2392 RSC
.
- P. L. Tremblay, M. Xu, Y. Chen and T. Zhang, iScience, 2020, 23, 100784 CrossRef CAS PubMed
.
- J. Liu, Y. Zhang, L. Lu, G. Wu and W. Chen, Chem. Commun., 2012, 48, 8826 RSC
.
- J. Liu, Y. Liu, N. Liu, Y. Han, X. Zhang, H. Huang, Y. Lifshitz, S. T. Lee, J. Zhong and Z. Kang, Science, 2015, 347, 970 CrossRef CAS PubMed
.
- P. B. Pati, G. Damas, L. Tian, D. L. A. Fernandes, L. Zhang, I. B. Pehlivan, T. Edvinsson, C. M. Araujo and H. Tian, Energy Environ. Sci., 2017, 10, 1372 RSC
.
- W. Yu, M. V. Pavliuk, A. Liu, Y. Zeng, S. Xia, Y. Huang, H. Bai, F. Lv, H. Tian and S. Wang, ACS Appl. Mater. Interfaces, 2023, 15, 2183 CrossRef CAS PubMed
.
- Z. Sun, J. A. Ramsay, M. Guay and B. A. Ramsay, Appl. Microbiol. Biotechnol., 2007, 74, 69 CrossRef CAS PubMed
.
- Y. Li, S. Li, K. Qu, J. Yang, S. Wang and Z. Yan, J. Environ. Sci., 2025, 55, DOI:10.1016/j.jes.2025.07.037
.
- W. Wang, M. Zhang, M. Guo, J. Wang, X. Wang, J. Yin, L. Chen and Y. Li, Green Chem., 2026, 28, 213 RSC
.
- Y. Zheng, L. Lin, B. Wang and X. Wang, Angew. Chem., Int. Ed., 2015, 54, 12868 CrossRef CAS PubMed
.
- W. Wang, H. Zhang, M. Guo, H. Dong, M. Zhang and L. Chen, ACS Sustainable Chem. Eng., 2025, 13, 7043 CrossRef CAS
.
- Y. Kita, R. Fujii and Y. Amao, Sustainable Energy Fuels, 2023, 7, 360 RSC
.
- Y. Kita and Y. Amao, Catal. Surv. Asia, 2023, 27, 67 CrossRef CAS
.
- Y. Kita and Y. Amao, Chem. Commun., 2022, 58, 11131 RSC
.
- Y. Kita and Y. Amao, Green Chem., 2023, 25, 2699 RSC
.
- C. Rosier, N. Leys, C. Henoumont, M. Mergeay and R. Wattiez, Appl. Microbiol. Biotechnol., 2012, 78, 4516 CAS
.
- M. K. Sluis, R. A. Larsen, J. G. Krum, R. Anderson, W. W. Metcalf and S. A. Ensign, J. Bacteriol., 2002, 184, 2969 CrossRef CAS PubMed
.
- T. Katagiri and Y. Amao, Green Chem., 2020, 22, 6682 RSC
.
- Y. Amao, Sustainable Energy Fuels, 2018, 2, 1928 RSC
.
- Y. Amao, Chem. Rev., 2026, 126, 1635 CrossRef CAS PubMed
.
- R. Gupta and A. K. Pal, Sustainable Energy Fuels, 2024, 8, 4709 RSC
.
- W. Lubitz, H. Ogata, O. Rüdiger and E. Reijerse, Chem. Rev., 2014, 114, 4081 CrossRef CAS PubMed
.
- H. Jia, L. Wanb, Y. Gaoc, P. Duc, W. Lic, H. Luoc, J. Ningc, Y. Zhaoc, H. Wangd, L. Zhangd and L. Zhang, J. Energy Chem., 2023, 85, 348 CrossRef
.
- S. I. Allakhverdiev, V. D. Kreslavski, V. Thavasi, S. K. Zharmukhamedov, V. V. Klimov, T. Nagata, H. Nishihara and S. Ramakrishna, Photochem. Photobiol. Sci., 2009, 8, 148 CrossRef CAS PubMed
.
- Y. Liu, C. Pulignani, S. Webb, S. J. Cobb, S. Rodríguez-Jiménez, D. Kim, R. D. Milton and E. Reisner, Chem. Sci., 2024, 15, 6088 RSC
.
- K. P. Sokol, W. E. Robinson, J. Warnan, N. Kornienko, M. M. Nowaczyk, A. Ruff, J. Z. Zhang and E. Reisner, Nat. Energy, 2018, 3, 944 CrossRef CAS
.
- C. A. Caputo, L. Wang, R. Beranek and E. Reisner, Chem. Sci., 2015, 6, 5690 RSC
.
- K. A. Brown, S. Dayal, X. Ai, G. Rumbles and P. W. King, J. Am. Chem. Soc., 2010, 13, 9672 CrossRef PubMed
.
- K. W. Brown, M. B. Wilker, M. Boehm, G. Dukovic and P. W. King, J. Am. Chem. Soc., 2012, 134, 5627 CrossRef CAS PubMed
.
- M. B. Wilker, K. E. Shinopoulos, K. A. Brown, D. W. Mulder, P. W. King and G. Dukovic, J. Am. Chem. Soc., 2014, 136, 4316 CrossRef CAS PubMed
.
- Y. Honda, H. Hagiwara, S. Ida and T. Ishihara, Angew. Chem., Int. Ed., 2016, 55, 8045 CrossRef CAS PubMed
.
- Y. Honda, Y. Shinohara and H. Fujii, Catal. Sci. Technol., 2020, 10, 6006 RSC
.
- Y. Honda, M. Yamamoto, Y. Shinohara, Y. Hatanaka, M. Watanabe, T. Ishihara and H. Fujii, ACS Appl. Nano Mater., 2025, 8, 21294 CrossRef CAS
.
- Y. Amao, J. CO2 Util., 2018, 26, 623 CrossRef CAS
.
- M. Moon, G. W. Park, J. Lee, J. S. Lee and K. Min, J. CO2 Util., 2020, 42, 101353 CrossRef CAS
.
- W. Ma, Q. Geng, C. Chen, Y. C. Zheng, H. L. Yu and J. H. Xu, ChemBioChem, 2023, 24, e202300390 CrossRef CAS PubMed
.
- A. Maier, L. M. Mguni, A. C. R. Ngo and D. Tischler, ChemCatChem, 2024, 16, e202401021 CrossRef CAS
.
- X. Guo, X. Wang, Y. Liu, Q. Li, J. Wang, W. Liu and Z. K. Zhao, Chem. – Eur. J., 2020, 26, 16611 CrossRef CAS PubMed
.
- D. Mandler and I. Willner, J. Chem. Soc., Perkin Trans. 2, 1998, 997 Search PubMed
.
- X. Huang, L. Xu, H. Qian, X. Wang and Z. Tao, J. Nanobiotechnol., 2022, 20, 295 CrossRef CAS PubMed
.
- Z. Chi, G. L. Liu, C. G. Liu and Z. M. Chi, Appl. Microbiol. Biotechnol., 2016, 10, 841 Search PubMed
.
- C. M. Krell and D. Seebach, Eur. J. Org. Chem., 2000, 1207 CrossRef CAS
.
- K. Tabata, K. Kasuya, H. Abe, K. Masuda and Y. Doi, Appl. Environ. Microbiol., 1999, 65, 4268 CrossRef CAS PubMed
.
- H. Adelnia, H. D. N. Tran, P. J. Little, I. Blakey and H. T. Ta, ACS Biomater. Sci. Eng., 2021, 7, 2083 CrossRef CAS PubMed
.
- J. J. Pueyo and C. Gömez-Moreno, Enzyme Microb. Technol., 1992, 14, 8 CrossRef CAS
.
- M. T. Bes, A. L. de Lacey, V. M. Fernandez and C. Gömez-Moreno, Bioelectrochem. Bioenerg., 1995, 38, 179 CrossRef CAS
.
- Y. Kim, K. Ikebukuro, H. Muguruma and I. Karube, J. Biotechnol., 1998, 59, 213 CrossRef CAS
.
- H. Inoue, M. Yamachika and H. Yoneyama, J. Chem. Soc., Faraday Trans., 1992, 88, 2215 RSC
.
- K. A. Brown, M. B. Wilker, M. Boehm, H. Hamby, G. Dukovic and P. W. King, ACS Catal., 2016, 6, 2201 CrossRef CAS
.
- T. Itoh, H. Asada, K. Tobioka, Y. Kodera, A. Matsushima, M. Hiroto, H. Nishimura, T. Kamachi, I. Okura and Y. Inada, Bioconjugate Chem., 2000, 11, 8 CrossRef CAS PubMed
.
- Z. Jiang, C. Lüa and H. Wu, Ind. Eng. Chem. Res., 2005, 44, 4165 CrossRef CAS
.
- Q. Shi, D. Yang, Z. Jiang and J. Li, J. Mol. Catal. B, 2006, 43, 44 CrossRef CAS
.
- S. H. Lee, D. H. Nam, J. H. Kim, J. O. Baeg and C. B. Park, ChemBioChem, 2009, 10, 1621 CrossRef CAS PubMed
.
- J. H. Kim, S. H. Lee, J. S. Lee, M. Lee and C. B. Park, Chem. Commun., 2011, 47, 10227 RSC
.
- Y. Katagiri and Y. Amao, Sustainable Energy Fuels, 2022, 6, 2581 RSC
.
- D. H. Nam and C. B. Park, ChemBioChem, 2012, 13, 1278 CrossRef CAS PubMed
.
- G. Zhao, C. Yang, W. Meng and X. Huang, J. Mater. Chem. A, 2024, 12, 3209 RSC
.
- H. Wu, C. Tian, X. Song, C. Liu, D. Yang and Z. Jiang, Green Chem., 2013, 15, 1773 RSC
.
- Y. Amao and M. Ishikawa, Catal. Commun., 2007, 8, 523 CrossRef CAS
.
- M. Takeuchi and Y. Amao, Sustainable Energy Fuels, 2023, 7, 355 RSC
.
- M. Takeuchi and Y. Amao, RSC Sustainability, 2023, 1, 1874 RSC
.
- M. Takeuchi and Y. Amao, Dalton Trans., 2024, 53, 418 RSC
.
- P. H. Wang, K. Correia, H. C. Ho, N. Venayak, K. Nemr, R. Flick, R. Mahadevan and E. A. Edwards, ISME J., 2019, 13, 1042 CrossRef CAS PubMed
.
- M. J. MacDonald, J. Biol. Chem., 1995, 270, 20051 CrossRef CAS PubMed
.
- M. Takeuchi and Y. Amao, RSC Sustainability, 2024, 2, 2491 RSC
.
- M. Takeuchi and Y. Amao, New J. Chem., 2024, 48, 18055 RSC
.
- A. Horikawa and Y. Amao, Energy Fuels, 2025, 39, 19537 CrossRef CAS
.
- M. Takeuchi and Y. Amao, RSC Sustainability, 2023, 1, 90 RSC
.
- M. Takeuchi and Y. Amao, New J. Chem., 2023, 47, 17679 RSC
.
- I. A. Rose, J. V. B. Warms and D. J. Kuo, Biochemistry, 1992, 31, 9993 CrossRef CAS PubMed
.
- I. A. Rose, J. V. B. Warms and R. G. Yuan, Biochemistry, 1993, 32, 8504 CrossRef CAS PubMed
.
- S. Beeckmans and E. van Driessche, J. Biol. Chem., 1998, 273, 31661 CrossRef CAS PubMed
.
- M. Takeuchi and Y. Amao, J. Jpn. Petrol. Inst., 2024, 67, 167 CrossRef CAS
.
- G. Li, Z. Lin, X. Li, Y. Zhang, W. Zhu, Y. Shao, Q. Xue, Q. Fan, T. Tan and H. Cao, J. Mater. Chem. A, 2024, 12, 192 RSC
.
- S. K. Guttikunda and P. Jawahar, Atmos. Environ., 2014, 92, 449 CrossRef CAS
.
- V. B. Y. Oh, S. F. Ng and W. J. Ong, J. Cleaner Prod., 2022, 379, 134673 CrossRef CAS
.
- S. Das, S. Das, M. M. Ghangrekar and B. Min, J. Environ. Chem. Eng., 2025, 13, 119221 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |