Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Amphiphilic poly(aminoester) dendrimers: click reaction–enabled synthesis, structure-governed self-assembly and degradation
Yonghui
Ma†
a,
Junyue
Zheng†
a,
Dandan
Zhu†
a,
Xianhui
Lin
a,
Dan
Yang
a,
Aoxue
Mei
a,
Chi
Ma
a,
Haijuan
Dong
c,
Ling
Peng
 *b and
Xiaoxuan
Liu
*b and
Xiaoxuan
Liu
 *a
*a
aState Key Laboratory of Natural Medicines, Joint International Research Laboratory of Target Discovery and New Drug Innovation (Ministry of Education), Jiangsu Key Laboratory of Drug Discovery for Metabolic Diseases, Center of Advanced Pharmaceuticals and Biomaterials, China Pharmaceutical University, Nanjing 210009, P. R. China. E-mail: xiaoxuanliu@cpu.edu.cn
bAix Marseille University, CNRS, Centre Interdisciplinaire de Nanoscience de Marseille, UMR 7325, «Equipe Labellisée Ligue Contre le Cancer», 163 avenue de Luminy, 13288 Marseille, France. E-mail: ling.peng@univ-amu.fr
cThe Public Laboratory Platform, China Pharmaceutical University, Nanjing 211198, China
First published on 26th March 2026
Abstract
Poly(aminoester) (PAE) dendrimers are attractive materials for biomedical applications owing to their biodegradability and precise structure, effective synthesis and favourable safety profile. We report here a click chemistry–enabled divergent–convergent strategy for the efficient synthesis of amphiphilic PAE dendrimers. These dendrimers exhibit negligible cytotoxicity, generation-dependent degradation behaviour, and tunable self-assembly controlled by the hydrophobic chain length. This study offers an accessible synthesis of PAE dendrimers, advancing their use in future biomedical applications.
Biodegradable biomaterials, particularly poly(aminoesters) (PAEs), have gained considerable attention for biomedical applications, owing to their intrinsic biodegradability.1 PAEs combine hydrolysable ester bonds with ionizable amine functionalities, facilitating efficient drug and nucleic acid delivery.2,3 However, their inherent polydispersity poses a significant challenge to their clinical translation.2 Dendrimers with well-defined structures and highly branched architectures4 are unique precision materials with biomedical advantages.5 Yet, synthesizing high-generation dendrimers, in particular PAE dendrimers,6 is challenging due to structural defects and purification difficulties, hindering their clinical progress.7
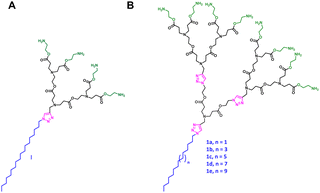
To address these challenges, we have pioneered a novel synthetic approach for constructing supramolecular dendrimers via the self-assembly of small amphiphilic dendrimers.8 Expanding on this concept, a series of amphiphilic dendrimers with varied hydrophilic–hydrophobic structures have been designed for diverse biomedical applications.9–12 These amphiphilic dendrimers were synthesized by coupling hydrophilic dendrons to hydrophobic entities via click chemistry—a method known for efficiency, simplicity, and mild reaction conditions.13 Leveraging this approach, we synthesized a small amphiphilic PAE dendrimer Ivia a click reaction between an azido-functionalized hydrophobic C18 alkyl chain and an alkyne-bearing hydrophilic PAE dendron with four terminal groups (Fig. 1A).14 This dendrimer seamlessly integrates the structural features of lipid and dendrimer vectors. It incorporates hydrolysable ester bonds and ionizable amine functionalities (e.g., tertiary and primary amines). The presence of tertiary amines in the dendrimer interior imparts pH-responsive behavior, enabling endosomal escape via the “proton sponge effect” and facilitating drug release under acidic conditions. Moreover, the ester bonds confer greater hydrolytic lability than the amide linkages in polyamidoamine (PAMAM) dendrimers, enhancing degradability and promoting carrier clearance and cargo release under physiological conditions. All these features enable this amphiphilic PAE dendrimer to achieve effective delivery of small interfering RNA (siRNA) with excellent biodegradability.
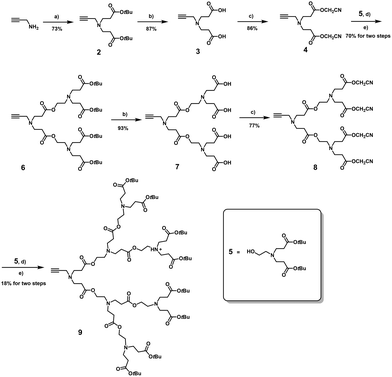
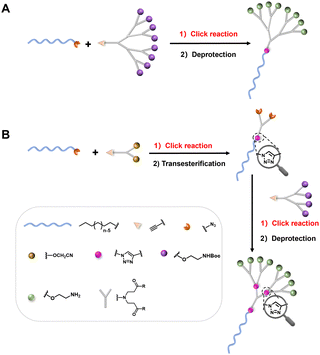
Inspired by the promising feature of this PAE dendrimer, we aimed to construct higher-generation amphiphilic PAE dendrimers (1a–1e) with varied hydrophobic chain lengths (Fig. 1B and Fig. S1) for potential application in drug delivery. To this end, we sought to extend our previous strategy14 by coupling a higher-generation hydrophilic PAE dendron bearing eight terminal groups to hydrophobic alkyl chains of varying lengths via click chemistry (Fig. 2A). Although the previously reported method employing reactive cyanomethyl ester intermediates successfully constructed a four-terminal PAE-based hydrophilic dendron, extending this approach to eight-terminal analogs was not satisfactory, giving higher-generation dendrimers in very low yields. Specifically, a transesterification reaction between the four-terminal cyanomethyl ester intermediate 8 and the branched alcohol 5 containing two tert-butyl ester termini afforded the target dendron 9 with eight tert-butyl ester termini in an extremely low yield of only 18% (Scheme 1). This can be ascribed to the steric hindrance and incomplete coupling efficiency during the assembly of the higher-generation dendron. Also, the increased number of ester bonds made higher-generation PAE dendrimers more susceptible to nucleophilic attack or degradation during prolonged synthesis conditions and purification steps, leading to further structural defects, hence low yields.
 | ||
| Fig. 2 Synthesis strategies for amphiphilic PAE dendrimers: (A) previously reported approach; (B) approach developed in this study. | ||
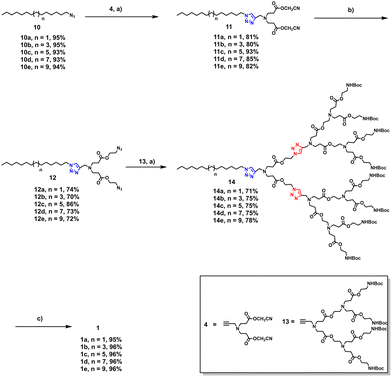
To overcome these limitations, we were committed to developing novel approaches for robust and efficient construction of amphiphilic PAE dendrimers, particularly those of higher generation. We envisioned a divergent-convergent strategy combined with click chemistry for synthesizing the PAE dendrimers (1a–1e) (Fig. 2B). Specifically, an azide-functionalized hydrophobic alkyl chain was first conjugated to a smaller alkynyl-bearing PAE dendron with two terminal groups via a Cu(I)-catalyzed Huisgen cycloaddition. The resulting intermediate was then subjected to transesterification to yield an amphiphilic dendrimer bearing two terminal azide groups. This intermediate was subsequently coupled with, via a second click reaction, an alkynyl-terminated PAE dendron containing four terminal groups, followed by deprotection to afford the amphiphilic PAE dendrimer with eight terminal groups. Using this approach, amphiphilic PAE dendrimers (1a–1e) with various chain lengths were successfully synthesized. We present below their synthesis and the evaluation of their degradation and self-assembly behaviour.
Scheme 2 illustrates a detailed step-by-step synthesis of amphiphilic PAE dendrimers (1a–1e). Azide-functionalized hydrophobic alkyl chains (10a–10e) (Scheme S1) and the alkynyl-containing hydrophilic PAE dendrons 4 (Scheme 1) and 13 (Scheme S2) were prepared according to previously reported procedures.14 Then, 10a–10e were respectively coupled with 4via a CuSO4·5H2O/sodium ascorbate-mediated copper-catalyzed azide–alkyne cycloaddition (CuAAC), affording the corresponding 11a–11e. Subsequent transesterification of 11a–11e with 2-azidoethanol under DBU-promoted alkaline conditions yielded the azide-functionalized intermediates (12a–12e). A second click reaction between 12a–12e and dendron 13 under identical CuAAC conditions produced dendrimers (14a–14e). Further deprotection of Boc groups with trifluoroacetic acid (TFA) furnished the target PAE dendrimers (1a–1e) in yields exceeding 95%. The structures and purities of the final dendrimers (1a–1e) were confirmed using 1H NMR and 13C NMR spectroscopy, high-resolution mass spectrometry (HRMS), and high-performance liquid chromatography (HPLC) (Fig. S2–S6).
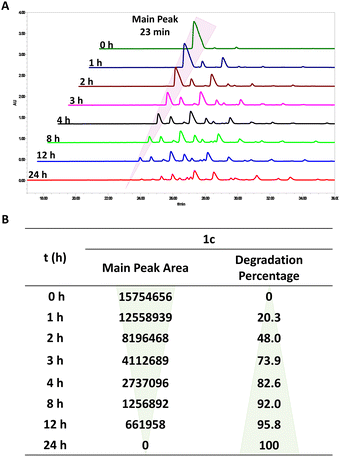
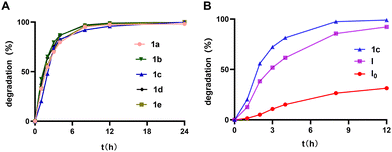
The PAE dendrimers (1a–1e), featuring ester functionalities within their backbone, are expected to be susceptible to hydrolytic degradation. We therefore assessed their degradation behaviour in aqueous solution at 25 °C using HPLC. Taking 1c as a representative example, the peak with retention time of 23 min in HPLC featuring the intact dendrimer 1c steadily decreased and vanished completely after 24 h, while numerous peaks with longer retention times appeared, corresponding to the degradation products (Fig. 3A, Fig. S7). Integration of the peak at 23 min revealed a sharp decline in area within 12 h, with full disappearance by 24 h (Fig. 3B), highlighting a rapid degradation.
Similarly, the other dendrimers exhibited comparable degradation profiles to 1c, with complete degradation observed within 24 h at room temperature (Fig. S8–S11). Further quantitative analysis confirmed that all dendrimers share a similar degradation pattern (Fig. 4A), despite variations in hydrophobic alkyl chain length ranging from C14 to C22. This consistent degradation behaviour can be attributed to their identical hydrophilic dendrons, indicating that hydrolytic degradation is governed primarily by dendritic architecture rather than alkyl chain length. These results highlight the pivotal role of the dendron structure in dictating the stability and degradation of amphiphilic PAE dendrimers.
Dendrimers often exhibit a “dendritic effect,” wherein their properties change with generation and size.15,16 This effect encompasses multivalency—absent in monomeric analogues but present across all generations—and generation-dependent property evolution, both of which modulate dendrimer characteristics. To probe the influence of this dendritic effect on degradation, we compared 1c with the previously reported lower-generation amphiphilic PAE dendrimers with four terminals (I) and two terminals (I0), which share the same hydrophobic alkyl chain length but has a lower ester bond density (Fig. 4B, Fig. S7, S12 and S13). Notably, 1c degraded faster than its lower-generation analogues I and I0, consistent with generation-dependent degradation. This may be attributed to the increasing dendrimer generation, which creates steric hindrance, making the ester linkage unstable and hence ready for hydrolysis. This “dendritic effect” thus accelerates degradation in the higher-generation PAE dendrimers, which can be explored for generation-dependent drug release.
Owing to their amphiphilic nature, 1a–1e are expected to self-assemble in an aqueous environment. We therefore evaluated their critical aggregation concentration (CAC) utilizing a fluorescence assay with Nile red as a probe at room temperature. As summarized in Table 1 and shown in Fig. S14, all dendrimers exhibited pronounced self-assembly. Notably, the CAC decreased from 53.7 µM for 1a to 5.01 µM for 1e with increasing alkyl chain length, revealing a clear chain length–dependent aggregation behaviour. This trend can be attributed to strengthened hydrophobic interactions within the increasing alkyl chain length among 1a–1e. These results align with our previous observation17 and demonstrate that tuning the balance between hydrophilic and hydrophobic segments modulates self-assembly, providing a foundation for the rational design of modular PAE dendrimers for the desired application.
| Dendrimer | CAC (µM) |
|---|---|
| 1a | 53.7 |
| 1b | 20.3 |
| 1c | 11.5 |
| 1d | 6.16 |
| 1e | 5.01 |
We further assessed the cytotoxicity of dendrimers (1a–1e) across multiple cell lines, including mouse fibroblast L929, human liver cell L02, and Madin-Darby canine kidney cell MDCK. All dendrimers exhibited half maximal inhibitory concentration (IC50) exceeding 100 µM, indicating negligible cytotoxicity (Table 2 and Fig. S15). Given that the dendrimers underwent complete degradation within 24 h at room temperature (25 °C) (Fig. 3 and S7) and within 8 h at 37 °C (Fig. S16), these findings demonstrate that both the intact dendrimers and their degradation products showed no cytotoxic effects. This favourable safety profile supports the potential of these dendrimers for further biomedical applications.
| Dendrimer | IC50 (µM) | ||
|---|---|---|---|
| L929 | L02 | MDCK | |
| 1a | >100 | >100 | >100 |
| 1b | >100 | >100 | >100 |
| 1c | >100 | >100 | >100 |
| 1d | >100 | >100 | >100 |
| 1e | >100 | >100 | >100 |
In summary, we developed a divergent-convergent strategy combined with click chemistry for the rapid and efficient synthesis of higher-generation amphiphilic PAE dendrimers. This method streamlines the synthesis of higher-generation PAE dendrimers by reducing synthetic steps and reaction times, with improved overall yields. The dendrimers exhibit excellent and fast degradability along with good safety profiles, with degradation performance governed by a generation-dependent “dendritic effect”. Additionally, their self-assembly behavior is tunable through variations in hydrophobic chain length, highlighting their potential as a modular platform for future biomedical applications. Nevertheless, the rapid degradation of PAE dendrimers must be carefully assessed to ensure their effective use in biomedical applications without compromising their advantageous properties. We are actively working in this direction.
L. P., X. L.: project conceptualization and supervision; Y. M., J. Z., D. Z., A. M., D. Y., C. M., X. L.: methodology, investigation, data analysis; H. D.: data analysis; Y. M., D. Z., L. P., X. L.: wrote the paper; all approved the manuscript.
Conflicts of interest
There are no conflicts to declare.Data availability
All the data supporting this study have been included in this manuscript and in the supplementary information (SI). The SI includes experimental details, degradation and CAC data, copies of the 1H NMR, 13C NMR, HPLC, and HRMS spectra, as well as safety data. See DOI: https://doi.org/10.1039/d6cc00300a.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81701815, 51773227), the Natural Science Foundation of Jiangsu Province (BK20170735), the Youth Thousand-Talents Program of China, the Jiangsu Specially-Appointed Professors Program, the Jiangsu Mass Entrepreneurship and Innovation Talent Program, and the Key Program for International S&T Cooperation Projects of China (2018YFE0117800).References
- A. Kirillova, T. R. Yeazel, D. Asheghali, S. R. Petersen, S. Dort, K. Gall and M. L. Becker, Chem. Rev., 2021, 121, 11238–11304 CrossRef CAS PubMed.
- X. Wang, Z. Zhang and N. Hadjichristidis, Prog. Polym. Sci., 2023, 136, 101634 CrossRef CAS.
- Y. Liu, Y. Li, D. Keskin and L. Shi, Adv. Healthcare Mater., 2019, 8, 1801359 CrossRef PubMed.
- D. A. Tomalia, Pharmaceutics, 2024, 16, 1530 CrossRef CAS PubMed.
- Q. Ni, J. Wu, C. Galanakou, C. Zhang, D. A. Tomalia and L. Peng, Nat. Rev. Bioeng., 2025, 3, 806–807 CrossRef CAS.
- P. Moreno, G. Quelever and L. Peng, Tetrahedron Lett., 2015, 56, 4043 CrossRef CAS.
- S. Svenson, Chem. Soc. Rev., 2015, 44, 4131 RSC.
- Z. Lyu, L. Ding, A. Y.-T. Huang, C.-L. Kao and L. Peng, Mater. Today Chem., 2019, 13, 34–48 CrossRef CAS.
- Z. Lyu, L. Ding, A. Tintaru and L. Peng, Acc. Chem. Res., 2020, 53, 2936–2949 CrossRef CAS PubMed.
- Y. Jiang, Z. Lyu, B. Ralahy, J. Liu, T. Roussel, L. Ding, J. Tang, A. Kosta, S. Giorgio, R. Tomasini, X.-J. Liang, N. Dusetti, J. Iovanna and L. Peng, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2215308120 CrossRef CAS PubMed.
- Z. Lyu, B. Ralahy, T.-A. Perles-Barbacaru, L. Ding, Y. Jiang, B. Lian, T. Roussel, X. Liu, C. Galanakou, E. Laurini, A. Tintaru, S. Giorgio, S. Pricl, X. Liu, M. Bernard, J. Iovanna, A. Viola and L. Peng, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2322403121 CrossRef CAS PubMed.
- X. Liu, D. Dhumal, P. Santofimia-Castaño, J. Liu, M. Casanova, A. C. Garcia-Muñoz, T.-A. Perles-Barbacaru, A. Elkihel, W. Zhang, T. Roussel, C. Galanakou, J. Wu, E. Zerva, N. Dusetti, Y. Xia, X.-J. Liang, A. Viola, J. L. Iovanna and L. Peng, Sci. Adv., 2025, 11, eadu9948 CrossRef CAS PubMed.
- N. K. Devaraj and M. G. Finn, Chem. Rev., 2021, 121, 6697–6698 CrossRef CAS.
- C. Ma, D. Zhu, W. Lin, Y. Li, Y. Huang, H. Zhu, M. Ye, Y. Wang, L. Peng and X. Liu, Chem. Commun., 2022, 58, 4168 RSC.
- D. A. Tomalia, New J. Chem., 2012, 36, 264–281 RSC.
- A.-M. Caminade, A. Ouali, R. Laurent, C.-O. Turrin and J.-P. Majoral, Chem. Soc. Rev., 2015, 44, 3890 RSC.
- C. Chen, P. Posocco, X. Liu, Q. Cheng, E. Laurini, J. Zhou, C. Liu, Y. Wang, J. Tang, V. D. Col, T. Yu, S. Giorgio, M. Fermeglia, F. Qu, Z. Liang, J. J. Rossi, M. Liu, P. Rocchi, S. Pricl and L. Peng, Small, 2016, 12, 3667–3676 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |