Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Nickel-catalyzed cross-electrophile-coupling of thioesters with alkyl bromides to form ketones under mechanochemical conditions
Ivo H. Lindenmaier†
a,
Robert C. Richter† a,
Liesa Renza,
Mattis T. Vochezera,
Marius Schiera,
Pascal Faßnachta,
Markus Ströbele
a,
Liesa Renza,
Mattis T. Vochezera,
Marius Schiera,
Pascal Faßnachta,
Markus Ströbele b and
Ivana Fleischer
b and
Ivana Fleischer *a
*a
aInstitute of Organic Chemistry, Faculty of Science, Eberhard Karls Universität Tübingen, Auf der Morgenstelle 18, 72076, Tübingen, Germany. E-mail: ivana.fleischer@uni-tuebingen.de
bInstitute of Inorganic Chemistry, Faculty of Science, Eberhard Karls Universität Tübingen, Auf der Morgenstelle 18, 72076, Tübingen, Germany
First published on 13th February 2026
Abstract
A solvent-minimized nickel-catalyzed synthesis of ketones, enabled by mechanochemistry, using thioesters as an acyl source is demonstrated. Mechanistic studies suggest a radical-chain-type mechanism.
Ketones represent a fundamental compound class in organic chemistry, ubiquitous in pharmaceuticals and natural products.1 Classical syntheses typically rely on nucleophilic 1,2-additions to carboxylic acid derivatives. However, due to the electrophilicity of the resulting ketone, undesired over-addition can occur, necessitating pre-functionalization or metal catalysis (Scheme 1A).2–5 These approaches also depend on nucleophilic reagents, often organometallic species, which limit the functional group tolerance and are frequently toxic, air-sensitive and less readily available than electrophiles.6 Another approach towards ketones is the Wacker-type oxidation of alkenes.7–10
 | ||
| Scheme 1 Brief overview of traditional ketone syntheses alongside solution phase and mechanochemical XEC alternatives. | ||
In addition, cross-electrophile couplings (XECs) have emerged as a powerful alternative, enabling the union of two electrophiles under reductive conditions.6 Due to the abundance of electrophiles and high functional group tolerance, this strategy offers a broad scope. The first ketone synthesis via nickel-catalyzed XEC was reported by Mukaiyama in 1981.11 Subsequent developments, independently pioneered by the Weix and Gong groups, expanded the scope of acyl–alkyl XECs through the use of bidentate nitrogen ligands and acyl chlorides.12,13 Since then, various XEC variants have been developed offering a diverse toolkit towards the synthesis of ketones (Scheme 1B).14–19 Nonetheless, XECs still face challenges including extended reaction times, elevated temperatures, and reproducibility issues associated with heterogeneous metal reductants.6,20 Mechanochemistry offers a promising solution by enabling rapid, solvent-minimized reactions under ambient conditions.21 Vigorous mixing conditions improve the reactivity of solid reductants, enhancing the reaction efficiency and reproducibility.21–26
Moreover, mechanochemical methods align with green chemistry principles, offering a more sustainable alternative to conventional solution-phase synthesis.27–30 Despite progress in mechanochemical XECs,21,31–33 application to ketone synthesis remains underexplored, with to the best of our knowledge only a single report by Browne and co-workers describing the coupling of twisted amides with alkyl bromides (Scheme 1C).31 In this work, N-acyl glutarimides were used as the only acyl source. While they have several advantages,34 fine-tuning the reactivity is rather difficult. Our ongoing studies on thioesters demonstrated that they are attractive electrophiles in coupling reactions as they are readily accessible, bench-stable and their reactivity is tunable by the modulation of the S-substituent.35–37 While thioesters represent established electrophiles in solution-phase chemistry,2,38,39 they have never been applied to mechanochemistry. Motivated by this potential, we envisioned a mechanochemical XEC of thioesters with alkyl halides to access ketones under ambient conditions (Scheme 1D).
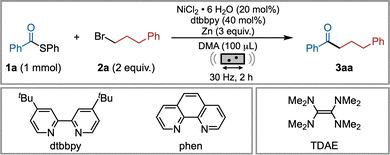
Preliminary experiments revealed that S-ethyl thioesters remained unreactive, while S-phenyl thioesters furnished the desired ketone. Ligand screening identified bidentate nitrogen ligands, particularly 1,10-phenanthroline (phen) and 4,4′-di-tert-butyl-2,2′-dipyridine (dtbbpy), as most effective (Table 1, entries 1 and 2), while other ligand classes resulted in diminished reactivity (see the SI, Section S3). Subsequently, liquid-assisted grinding (LAG) media were explored. Polar amide solvents such as DMA, commonly used in XECs, proved effective, while protic MeOH presented an interesting alternative (entry 3). Even water could serve as an LAG medium, leading to substantial but not complete loss of yield. This highlights moisture tolerance, which is consistent with ambient conditions and utilization of non-dried LAG agents (see the SI, Section S3).
| Entry | Variation | Yielda (%) |
|---|---|---|
| Reaction conditions: performed in a stainless steel (SS) milling jar (10 ml) with 2 × 4 g SS milling balls for 2 h at 30 Hz.a GC-FID yields.b N,N-Dimethylpropyleneurea (DMPU) instead of DMA.c NiCl2·6H2O (10 mol%), dtbbpy (20 mol%), Zn (2 equiv.), pyridine (2 equiv.) mixer mill (30 Hz, 1h). | ||
| 1 | None | 51 |
| 2b | Phen as ligand | 44 |
| 3 | MeOH as LAG | 47 |
| 4 | + Pyridine (1 equiv.) | 64 |
| 5 | + TPPO (1 equiv.) | 60 |
| 6 | + ZnBr2 (1 equiv.) | 6 |
| 7 | Mn instead of Zn | 21 |
| 8 | TDAE instead of Zn | 27 |
| 9c | Pyridine as LAG and 10 mol% catalyst loading | 78 |
| 10c | + NaCl (2 equiv.) | >99 |
| 11c | No Ni/no ligand | 2/0 |
Additive screening revealed that both pyridine and triphenylphosphine oxide (TPPO) enhanced the reaction efficiency, whereas ZnBr2 had a detrimental effect (entries 4–6). These results suggest that Lewis-basic pyridine and TPPO may reduce the inhibitory effect of in situ generated Zn(II), an interpretation supported by a previous report by Weix,.40 and the observed solid-state structure of a [(pyridine)ZnBr3]− complex obtained from the crude reaction mixture via single-crystal X-ray diffraction (see the SI, Section S4). Alternative reductants such as manganese and tetrakis(dimethylamino)ethylene (TDAE) were both tolerated but provided lower yields compared to zinc (entries 7 and 8). The use of other nickel salts decreased the efficiency (see the SI, Section S3). A key discovery was that pyridine could act as both an additive and LAG agent, eliminating the need for DMA. This change increased the yield, reduced the catalyst loading (10 mol%), and shortened the reaction time to 1 h (entry 10). Finally, adding NaCl, potentially acting as a grinding auxiliary or enabling more complex interactions,31,41 further improved the outcome, defining the optimized conditions for subsequent studies. Control experiments confirmed the necessity of both NiCl2·6H2O and the ligand (entry 11).
With the optimized conditions at hand, the substrate scope was investigated (Scheme 2). Aside from simple benzoic thioester (3aa), sterically hindered thioesters could be coupled in good yields (3ba, 3ca). An upscaled reaction gave 3aa in 89% yield (3.99 g). Both electron-withdrawing and -donating groups were accommodated, including aryl halides (3da, 3ea), a trifluoromethyl group (3fa), an ester (3ga), or a methoxy substituent (3ha) in good yields. Meanwhile, more electron-rich benzodioxol decreased the yield (3ia). It should be noted that electron-withdrawing substituents led to increased amounts of the decarbonylated coupling product. To our delight, aliphatic thioesters could also be used, efficiently yielding dialkylketones in excellent yields (3ja, 3la). However, homobenzylic thioesters suffered from decarbonylation and homocoupling, producing higher amounts of side-products (3ka). Unsaturated alkyl chains were also viable (3ma). Gratifyingly, ketones with secondary aliphatic cycles on the acyl moiety could be obtained in good to high yields (3na, 3qa), and heterocyclic substrates were well tolerated (3oa, 3pa). Notably, even the coupling of a sterically encumbered tertiary thioester was successful, albeit in a reduced yield (3ra). Next, the alkyl bromide scope was studied. Primary alkyl bromides afforded products in good yields (3ja, 3jb). The reaction was compatible with alkyl and aryl chlorides (3jc, 3jd), a trifluoromethyl group (3jf) and even aryl bromides (3je), representing a handle for further functionalization. Ketones containing silyl- or methyl-protected alcohols (3jg, 3jh) as well as esters (3ai, 3ji) could be produced in moderate to good yields. Secondary alkyl bromides were also feasible despite their higher steric demand, delivering ketones in good to high yields (3jj–3jl). Notable limitations included the use of tertiary (low conversions) and benzylic bromides (homocoupling). Finally, thioesters derived from pharmaceutically active compounds Ibuprofen, Oxaprozin or Probenecid as well as an intermediate in the Roflumilast synthesis could be coupled in moderate to good yields (3sa–3va).
Next, we turned our attention to the reaction mechanism (Scheme 3), as both a Negishi-type as well as a radical-chain mechanism seemed possible.21,42 We were especially interested in the activation of thioesters, as there is no clear mechanistic evidence on their oxidative addition (OA) to nickel. Consequently, we sought to synthesize the OA complex by adding thioester 1a to a dtbbpy-preligated nickel(0) complex, affording Ni1 (Scheme 3A). Interestingly, a mixture of the acyl complex Ni1a and its CO-deinserted isomer Ni1b was obtained with the latter as the major product (ca. 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 65). This was revealed by 1H13C-HMBC NMR spectroscopy, showing the presence or absence of 3J coupling of the carbonyl carbon with a proton in the ortho-position of the aromatic ring. Varying the addition temperature (−80 °C to rt) or measurement conditions (−40 °C to rt) did not significantly influence this ratio, suggesting that a fast equilibrium between these two species is present in solution. While attempts to crystallize Ni1 remained unsuccessful, a 4-fluoro-substituted derivative Ni2a could be characterized by XRD, displaying a square-planar coordination of nickel(II) with an intact acyl moiety, although the acyl form was also the minor form in solution for this derivative. The solid-state structure is in line with the literature and the observed diamagnetism.43
65). This was revealed by 1H13C-HMBC NMR spectroscopy, showing the presence or absence of 3J coupling of the carbonyl carbon with a proton in the ortho-position of the aromatic ring. Varying the addition temperature (−80 °C to rt) or measurement conditions (−40 °C to rt) did not significantly influence this ratio, suggesting that a fast equilibrium between these two species is present in solution. While attempts to crystallize Ni1 remained unsuccessful, a 4-fluoro-substituted derivative Ni2a could be characterized by XRD, displaying a square-planar coordination of nickel(II) with an intact acyl moiety, although the acyl form was also the minor form in solution for this derivative. The solid-state structure is in line with the literature and the observed diamagnetism.43
Because the isomer ratio Ni1a/Ni1b in THF solution may not be directly transferable to mechanochemical conditions, we performed a control experiment: Upon subjecting Ni1 to the ball mill, several scrambling products were obtained. Interestingly, the major products resembled decarbonylated species, indicating that under these conditions, Ni1b was also the major species or reacted faster. However, upon subjecting Ni1 to a stoichiometric coupling with alkyl bromide 2a, mainly the ketone product 3aa was obtained, along with a minor amount of decarbonylated product 4. This suggests that Ni1a reacts faster with 2a than Ni1b and is (re-)generated from Ni1b during the reaction in a Curtin-Hammett-like scenario. A reason could be that Ni1b requires extrusion of CO before radical capture is possible. Notably, for more electron-poor aryl thioesters, considerable amounts of the decarbonylated product were observed in catalytic reactions, suggesting that this reaction may be competitive in cases where extrusion of CO is more favorable. Evidence for a radical-chain mechanism was obtained by suppression of the coupling reaction when adding a radical inhibitor (TEMPO) and the formation of the ring-opened coupling product (3an) upon using a radical reporter (Scheme 3B). The radical activation of alkyl bromides by SET from nickel is consistent with numerous literature reports.6 While Browne and co-workers also reported the formation of organozinc species under similar conditions,24,31 the absence of 3am contradicts a Negishi-type mechanism, which is also supported by the finding that organic reductants can be used (Table 1, entry 8). Moreover, adding a Michael-acceptor to the standard reaction resulted in the formation of the formal Giese-adduct 4, pointing towards the formation of an acyl radical,35,44 although a competitive migratory insertion mediated by nickel cannot be excluded. Thus, we propose a radical-chain mechanism, consistent with recent literature,31,45 involving oxidative addition of the thioester to afford the respective oxidative-addition complex (Scheme 3C). Capture of the alkyl radical, likely produced by SET from a Ni(I) species,46 followed by reductive elimination yields the coupling product, while the catalytic cycle is closed by reduction of the resulting Ni(II) complex with zinc. The detailed mechanism of oxidative addition may be a subject of future investigations.
In conclusion, an efficient procedure for XEC of aliphatic and aromatic thioesters with alkyl bromides was developed. Applicability was demonstrated on numerous examples, including biologically relevant compounds. The mechanochemical set-up enables rapid, simple reactions without the need for Schlenk-techniques or a Glovebox. Compared to recent mechanochemical methods for the synthesis of ketones, our work featured comparable yields at shorter reaction times. Moreover, preliminary mechanistic studies advance the current understanding on mechanochemical transformation of acyl electrophiles.
Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting this article have been included as part of the supplementary information (SI). Supplementary information: general procedures, further condition screening and compound characterization. See DOI: https://doi.org/10.1039/d6cc00297h.CCDC 2491069 (Ni2a) contains the supplementary crystallographic data for this paper.47
Acknowledgements
We thank the analytics department of the University of Tübingen for their excellent work. The financial support from the University of Tübingen, FCI (scholarship I. H. L.) and Studienstiftung des Deutschen Volkes (scholarship R. C. R.) is gratefully acknowledged.References
- P. Ertl and T. Schuhmann, J. Nat. Prod., 2019, 82, 1258–1263 CrossRef CAS PubMed.
- H. Tokuyama, S. Yokoshima, T. Yamashita and T. Fukuyama, Tetrahedron Lett., 1998, 39, 3189–3192 Search PubMed.
- S. Nahm and S. M. Weinreb, Tetrahedron Lett., 1981, 22, 3815–3818 CrossRef CAS.
- M. Haddach and J. R. McCarthy, Tetrahedron Lett., 1999, 40, 3109–3112 CrossRef CAS.
- R. Lerebours, A. Camacho-Soto and C. Wolf, J. Org. Chem., 2005, 70, 8601–8604 CrossRef CAS PubMed.
- L. E. Ehehalt, O. M. Beleh, I. C. Priest, J. M. Mouat, A. K. Olszewski, B. N. Ahern, A. R. Cruz, B. K. Chi, A. J. Castro, K. Kang, J. Wang and D. J. Weix, Chem. Rev., 2024, 124, 13397–13569 CrossRef CAS PubMed.
- J. Smidt, W. Hafner, R. Jira, R. Sieber, J. Sedlmeier and A. Sabel, Angew. Chem., Int. Ed. Engl., 1962, 1, 80–88 Search PubMed.
- C. Jiang, Y. Wu, Y. Zhang, J. Zong, N. Wang, G. Liu, R. Liu and H. Yu, Angew. Chem., Int. Ed., 2025, 64, e202413901 CrossRef CAS PubMed.
- I. Yadav and R. Gramage-Doria, Chem. Commun., 2025, 61, 8471–8481 RSC.
- J. J. Dong, W. R. Browne and B. L. Feringa, Angew. Chem., Int. Ed., 2015, 54, 734–744 CrossRef CAS PubMed.
- M. Onaka, Y. Matsuoka and T. Mukaiyama, Chem. Lett., 1981, 531–534 CrossRef CAS.
- A. C. Wotal and D. J. Weix, Org. Lett., 2012, 14, 1476–1479 CrossRef CAS PubMed.
- F. Wu, W. Lu, Q. Qian, Q. Ren and H. Gong, Org. Lett., 2012, 14, 3044–3047 Search PubMed.
- H. Yin, C. Zhao, H. You, K. Lin and H. Gong, Chem. Commun., 2012, 48, 7034–7036 Search PubMed.
- J. Wang, B. P. Cary, P. D. Beyer, S. H. Gellman and D. J. Weix, Angew. Chem., Int. Ed., 2019, 58, 12081–12085 Search PubMed.
- F. T. Pulikottil, R. Pilli, R. V. Suku and R. Rasappan, Org. Lett., 2020, 22, 2902–2907 CrossRef CAS PubMed.
- F. Yang, D. Ding and C. Wang, Org. Lett., 2020, 22, 9203–9209 CrossRef CAS PubMed.
- C.-G. Yu and Y. Matsuo, Org. Lett., 2020, 22, 950–955 CrossRef CAS PubMed.
- J. Zhuo, Y. Zhang, Z. Li and C. Li, ACS Catal., 2020, 10, 3895–3903 CrossRef CAS.
- S. K. Nimmagadda, S. Korapati, D. Dasgupta, N. A. Malik, A. Vinodini, A. S. Gangu, S. Kalidindi, P. Maity, S. S. Bondigela, A. Venu, W. P. Gallagher, S. Aytar, F. González-Bobes and R. Vaidyanathan, Org. Process Res. Dev., 2020, 24, 1141–1148 CrossRef CAS.
- A. C. Jones, W. I. Nicholson, J. A. Leitch and D. L. Browne, Org. Lett., 2021, 23, 6337–6341 CrossRef CAS PubMed.
- S. Wu, W. Shi and G. Zou, New J. Chem., 2021, 45, 11269–11274 RSC.
- M. T. J. Williams, L. C. Morrill and D. L. Browne, Adv. Synth. Catal., 2023, 365, 1477–1484 CrossRef CAS.
- Q. Cao, J. L. Howard, E. Wheatley and D. L. Browne, Angew. Chem., Int. Ed., 2018, 57, 11339–11343 CrossRef CAS PubMed.
- Q. Cao, R. T. Stark, I. A. Fallis and D. L. Browne, ChemSusChem, 2019, 12, 2554–2557 Search PubMed.
- J. Yin, R. T. Stark, I. A. Fallis and D. L. Browne, J. Org. Chem., 2020, 85, 2347–2354 Search PubMed.
- J. F. Reynes, F. Leon and F. García, ACS Org. Inorg. Au, 2024, 4, 432–470 CrossRef CAS PubMed.
- F. Effaty, X. Ottenwaelder and T. Friščić, Curr. Opin. Green Sustainable Chem., 2021, 32, 100524 CrossRef CAS.
- J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13–19 CrossRef CAS PubMed.
- K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145–2162 CrossRef CAS PubMed.
- A. C. Jones, M. T. J. Williams, L. C. Morrill and D. L. Browne, ACS Catal., 2022, 12, 13681–13689 CrossRef CAS PubMed.
- C. Fan, B. Wang, T. Wu, Q. Kang, H. Wang, J. Sun and X. Wei, Cell Rep. Phys. Sci., 2024, 5, 101831 CrossRef CAS.
- H. Wang, W. Ding and G. Zou, J. Org. Chem., 2023, 88, 12891–12901 CrossRef CAS PubMed.
- C. Liu and M. Szostak, Chem. – Eur. J., 2017, 23, 7157–7173 Search PubMed.
- P. H. Gehrtz, P. Kathe and I. Fleischer, Chem. – Eur. J., 2018, 24, 8774–8778 CrossRef CAS PubMed.
- V. J. Geiger, G. Lefèvre and I. Fleischer, Chem. – Eur. J., 2022, 28, e202202212 CrossRef CAS PubMed.
- A. Haupt and I. Fleischer, ChemCatChem, 2025, 17, e202500221 CrossRef CAS.
- S.-Q. Tang, J. Bricard, M. Schmitt and F. Bihel, Org. Lett., 2019, 21, 844–848 CrossRef CAS PubMed.
- H.-G. Cheng, H. Chen, Y. Liu and Q. Zhou, Asian J. Org. Chem., 2018, 7, 490–508 CrossRef CAS.
- D. C. Batesky, M. J. Goldfogel and D. J. Weix, J. Org. Chem., 2017, 82, 9931–9936 CrossRef CAS PubMed.
- B. Zhang, J. Song, H. Liu, J. Shi, J. Ma, H. Fan, W. Wang, P. Zhang and B. Han, Green Chem., 2014, 16, 1198–1201 RSC.
- A. K. Pandey, ChemCatChem, 2022, 14, e202101982 CrossRef CAS.
- G. C. Tucci and R. H. Holm, J. Am. Chem. Soc., 1995, 117, 6489–6496 CrossRef CAS.
- P. Blakskjær, B. Høj, D. Riber and T. Skrydstrup, J. Am. Chem. Soc., 2003, 125, 4030–4031 CrossRef PubMed.
- D. A. Everson, B. A. Jones and D. J. Weix, J. Am. Chem. Soc., 2012, 134, 6146–6159 CrossRef CAS PubMed.
- S. Biswas and D. J. Weix, J. Am. Chem. Soc., 2013, 135, 16192–16197 CrossRef CAS PubMed.
- CCDC 2491069: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2pm52l.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |