Open Access Article
Open Access ArticleBeyond apoptosis: platinum phototherapeutics overcome resistance by triggering diverse cell death pathways
Shu
Chen
ab,
Zhigang
Wang
 *c and
Guangyu
Zhu
*c and
Guangyu
Zhu
 *ab
*ab
aDepartment of Chemistry, City University of Hong Kong, 83 Tat Chee Avenue, Hong Kong SAR 999077, People's Republic of China. E-mail: guangzhu@cityu.edu.hk
bCity University of Hong Kong Shenzhen Research Institute, Shenzhen 518057, People's Republic of China
cNation-Regional Engineering Lab for Synthetic Biology of Medicine, International Cancer Center, School of Pharmacy, Shenzhen University Medical School, Shenzhen 518060, People's Republic of China. E-mail: wangzg@szu.edu.cn
First published on 17th March 2026
Abstract
Platinum (Pt)-based drugs remain the cornerstone of malignant tumor treatment, but are severely constrained by drug resistance. Conventional Pt agents primarily induce apoptosis, yet resistant cancer cells can bypass this pathway, leading to treatment failure. In contrast, phototherapeutic Pt complexes, leveraging photodynamic therapy and photoactivated chemotherapy, represent a novel approach to overcoming resistance by either enhancing apoptosis or activating alternative programmed cell death (PCD) pathways. This review provides a comprehensive overview of Pt-based photosensitizers and photoactivated agents that trigger diverse PCD pathways, including apoptosis, autophagy, ferroptosis, pyroptosis, and immunogenic cell death, to combat resistance. We elucidate the unique features and mechanistic underpinnings of each pathway, providing in-depth analyses of representative complexes to illustrate their PCD induction strategies and effectiveness in reducing resistance. Finally, we present critical insights into and forward-looking perspectives on the development of next-generation Pt phototherapeutics that leverage diverse PCD pathways. Overall, this review highlights the significance of multimodal PCD induction, proposing that targeting these pathways offers a novel strategic opportunity for Pt-based anticancer agents to overcome drug resistance.
1. Introduction
Platinum (Pt)-based chemotherapeutic agents, including cisplatin, carboplatin, and oxaliplatin (Fig. 1a), play a pivotal role in modern oncology. They are integral to nearly 50% of chemotherapy regimens and significantly enhance survival rates across various cancer types.1–3 Driven by this established therapeutic success, extensive research has focused on understanding their molecular mechanisms and developing innovative Pt-based complexes to further augment anticancer efficacy.4–6 It is widely acknowledged that Pt-based anticancer drugs enter cells primarily through passive diffusion or active transport via specific transporters, notably copper transporter 1 (Ctr1) for cisplatin and organic cation transporters (OCTs) for oxaliplatin.7–9 Once inside the cell, these drugs undergo aquation (Fig. 1b), forming intrastrand and interstrand cross-links with purine bases in nuclear DNA.9,10 These Pt–DNA adducts distort the DNA double helix, hindering vital processes such as replication and transcription. This disruption triggers cellular stress responses, often culminating in apoptosis.11–13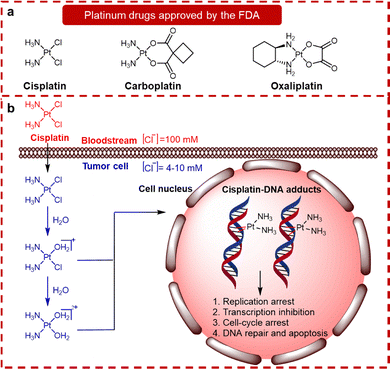 | ||
| Fig. 1 (a) The chemical structures of the FDA-approved Pt(II) drugs. (b) The mechanism of cisplatin and related Pt-based anticancer drugs involves a four-step process: cellular uptake, activation/aquation, DNA binding, and the cellular processing of the resulting DNA damage that ultimately triggers apoptosis.14 | ||
Pt complexes hold a central position among metal-based anticancer agents, underscored by three key advantages that remain unmatched by other metals at a similar scale.15 First, Pt drugs have an unparalleled record of regulatory approval and clinical use.16 Since the discovery of cisplatin's antitumor activity in the late 1960s, three generations of Pt drugs have received FDA approval and become first-line therapies for multiple solid tumors.1 Second, Pt's therapeutic efficacy stems from distinctive ligand-exchange kinetics paired with a well-defined mechanism of action. Pt(II) complexes display moderate ligand-exchange rates that balance stability in the extracellular, chloride-rich environment with sufficient reactivity for intracellular activation and formation of durable DNA adducts, which finally induce cell-cycle arrest and apoptosis.12,17 Comparable kinetics are rarely achieved by other metals, as decades of structure–activity relationship studies have delivered limited clinical success for non-Pt alternatives.18–20 Third, except arsenic trioxide, no non-Pt metal-based drug has yet gained FDA approval specifically for cancer therapy (Fig. 2). Most remain in preclinical or early-phase clinical development because of shortcomings compared to Pt drugs. For instance, Pd(II) analogs—despite structural similarity to Pt(II)—undergo ligand substitution 104–105 times faster, rendering them prone to thiol-mediated deactivation before reaching nuclear DNA and consequently resulting in poor antitumor efficacy.21,22 Likewise, Au(III) complexes explored as cisplatin alternatives have proven relatively unstable, readily reducing to metallic gold under physiological conditions.23–25 Although ruthenium compounds are the most promising non-Pt candidates, with lower toxicity and selective anticancer activity, they still lack large phase III trials and the decades of clinical validation that underpin Pt drugs.26 Overall, while non-Pt metals may offer benefits such as reduced toxicity and novel mechanisms, none has matched the proven efficacy, clinical versatility, and global therapeutic impact of Pt complexes in oncology so far.
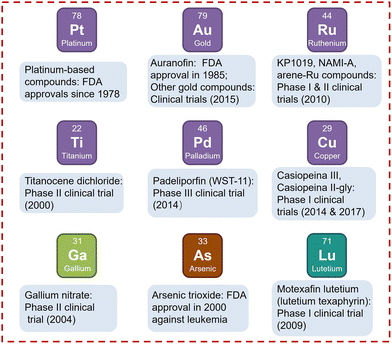 | ||
| Fig. 2 The metal-based drugs that have entered clinical trials.23,27 | ||
Despite its success, the efficacy of Pt-based chemotherapy is often severely compromised by intrinsic or acquired drug resistance, which contributes to treatment failure in over 90% of cases.28–31 Resistance mechanisms are diverse and include reduced drug uptake, enhanced efflux, increased intracellular detoxification, augmented DNA damage repair capacity, and the downregulation or blockade of apoptosis.1,32–35 Among these, defective apoptosis is particularly consequential, as it operates downstream of Pt–DNA adduct formation and directly counteracts cytotoxicity.32 In resistant cancers, apoptotic dysfunction commonly involves the overexpression of anti-apoptotic proteins (e.g., Bcl-2 and Bcl-xL), loss or inactivation of pro-apoptotic proteins (e.g., BAX), alterations in the p53 pathway, and activation of pro-survival signaling (e.g., PI3K/Akt).36–38 Accordingly, restoring apoptotic signaling by targeting these mechanisms represents a critical strategy for overcoming Pt resistance.
Beyond restoring apoptosis, engaging alternative cell-death programs offers a complementary route to overcome drug resistance. According to the Nomenclature Committee on Cell Death (NCCD), cell death is broadly classified into accidental cell death (ACD) and regulated cell death (RCD).39–41 ACD is an uncontrolled biological process triggered by overwhelming physical or chemical insults, whereas RCD, also known as programmed cell death (PCD), involves a highly orchestrated, genetically encoded cascade of molecular events.42 PCD encompasses apoptosis, autophagy, pyroptosis, ferroptosis, and immunogenic cell death (ICD), among others, and plays critical roles in cancer progression and therapeutic response.42 These non-apoptotic pathways provide novel therapeutic targets and offer significant opportunities to enhance cancer treatments and mitigate drug resistance.43
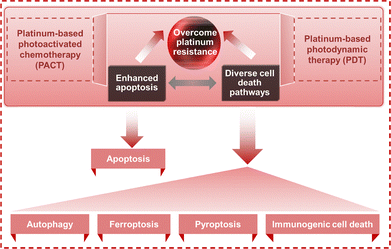
Overcoming Pt resistance requires complexes that either amplify suppressed apoptotic signaling or activate alternative PCD pathways that remain effective in resistant cells. Photoactivatable Pt complexes offer a means to achieve both goals while addressing key drawbacks of traditional Pt(II) chemotherapeutics, including systemic toxicity, severe side effects, and intrinsic or acquired resistance. The core concept is light-controlled drug action via photoactivatable platforms that offer high spatiotemporal precision, on-demand activation, and minimal invasiveness. These complexes are primarily developed based on two strategies: photoactivated chemotherapy (PACT) and photodynamic therapy (PDT). In PACT, inert Pt complexes remain non-toxic in the dark but are selectively activated within light-irradiated tumor tissues to release cytotoxic Pt(II) species, often accompanied by the generation of reactive oxygen species (ROS). The localized, on-demand activation confines therapeutic effects to the tumor site, thereby drastically reducing off-target toxicity relative to traditional Pt regimens.44–46 In PDT, Pt complexes act as photosensitizers (PSs) that, upon light irradiation, form a triplet excited state which generates cytotoxic ROS via two primary pathways: type I photoreaction, involving electron transfer to produce radical species such as superoxide (O2˙−) and hydroxyl (˙OH) radicals, and type II photoreaction, involving energy transfer to molecular oxygen to generate singlet oxygen (1O2).47–51 This ability to generate high levels of ROS independent of DNA binding provides a powerful, orthogonal mechanism to overcome classical Pt resistance.
PACT and PDT leverage precise spatiotemporal control to synergize Pt-based cytotoxicity with photoactivated species, amplifying apoptotic signaling. More importantly, they can directly trigger non-apoptotic PCD pathways, which bypass established resistance mechanisms. Specifically, intrinsic apoptotic signaling can be enhanced by generating a lethal flux of ROS that overwhelms mitochondrial defenses. Furthermore, photochemical disruption of lysosomal membranes can initiate autophagic cell death, a non-apoptotic pathway that effectively circumvents common resistance mechanisms. Beyond autophagy, the localized and intense ROS burst generated during the photodynamic process can cause catastrophic oxidative damage to lipids, thereby triggering ferroptosis—an iron-dependent cell death pathway that is impervious to classic apoptotic resistance factors. In parallel, photoactivation can promote ROS generation and activate caspase-1, leading to gasdermin D (GSDMD)-mediated pyroptosis. This highly inflammatory and lytic cell death mode results in plasma membrane rupture and the release of inflammatory contents, thereby effectively compensating for apoptotic defects. Moreover, the cytotoxic species generated during PDT or PACT can induce ICD, characterized by calreticulin (CRT) exposure and the release of damage-associated molecular patterns (DAMPs) such as adenosine triphosphate (ATP) and high-mobility group box 1 (HMGB1). These DAMPs stimulate a robust antitumor immune response, promoting the clearance of cells that would otherwise be resistant.
This review systematically explores the burgeoning field of Pt-based photoactivated agents and PSs engineered to initiate diverse PCD pathways—including apoptosis, autophagy, ferroptosis, pyroptosis, and ICD—as a targeted strategy to surmount drug resistance (Fig. 3). Following a detailed examination of the distinct characteristics and mechanisms governing each PCD pathway, we provide in-depth analyses of representative photoactivated Pt complexes to illustrate their specific mechanisms of PCD induction and their potential to circumvent resistance. Finally, we offer a critical summary and a forward-looking perspective on the trajectory for developing next-generation Pt phototherapeutics that leverage novel PCD paradigms to overcome drug resistance. To maintain a focused discussion, Pt-based nanoparticle systems loaded with Pt agents are deliberately excluded, with the focus on small-molecule complexes. This review highlights the induction of diverse cell death modalities as a key therapeutic strategy for circumventing resistance.
 | ||
| Fig. 3 Illustration of Pt phototherapeutics overcoming resistance by either intensifying apoptotic signals or triggering diverse cell death pathways. | ||
2. Pt phototherapeutics that induce apoptosis
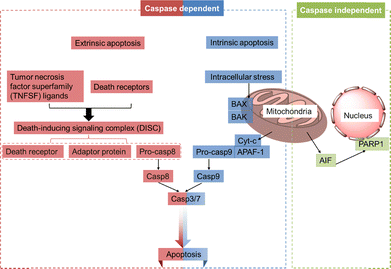
Apoptosis, the first described form of PCD, is characterized by distinct morphological changes, including cell shrinkage, membrane blebbing, chromatin condensation, and fragmentation into apoptotic bodies.52,53 It is classified into caspase-dependent and caspase-independent pathways (Fig. 4).54 Caspase-dependent apoptosis proceeds through two main pathways: the extrinsic pathway, activated by extracellular stimuli, and the intrinsic pathway, triggered by intracellular stress.55 The extrinsic pathway is initiated when tumor necrosis factor superfamily (TNFSF) ligands bind to death receptors, forming the death-inducing signaling complex (DISC).56–58 Within the DISC, procaspase-8 undergoes autoproteolytic cleavage, activating caspase-8, which then activates downstream executioner caspases, primarily caspase-3/7, culminating in extrinsic apoptosis.59 The intrinsic apoptotic pathway, activated by stressors such as DNA damage and oxidative stress, triggers the activation of pro-apoptotic BCL-2 family proteins (BAX and BAK), which subsequently mediate mitochondrial outer membrane permeabilization (MOMP). This process releases apoptogenic factors such as cytochrome c and apoptosis-inducing factor (AIF). Cytochrome c binds apoptotic protease-activating factor-1 (APAF-1), inducing its oligomerization and recruiting procaspase-9 to form the apoptosome.60,61 Within the apoptosome, procaspase-9 undergoes autocatalytic activation, which, in turn, activates executioner, primarily caspases-3/7, to complete the intrinsic apoptotic pathway.59 Concurrently, AIF translocates to the nucleus, causing DNA condensation and fragmentation, indicative of caspase-independent apoptosis.62 Pt-based anticancer drugs primarily activate the intrinsic pathway by inducing DNA crosslinks and mitochondrial dysfunction, leading to PCD. Furthermore, photoactivatable Pt complexes offer a promising strategy to overcome resistance by generating ROS capable of engaging both intrinsic and extrinsic apoptotic mechanisms.Pt-based PSs show promise for PDT, but their application is hindered by challenges such as drug resistance and the hypoxic tumor microenvironment.63,64 To address these limitations, Wang et al. developed a naphthalimide-modified cyclometalated Pt(II) complex, PtPAN (complex 1, Fig. 5).65 In cisplatin-resistant A549cisR lung cancer cells, complex 1 exhibited only moderate dark toxicity under both normoxia (half-maximum inhibitory concentration IC50: 9.43 ± 0.43 µM) and hypoxia (IC50: 7.92 ± 2.23 µM). Upon near-infrared (NIR) two-photon irradiation, however, photocytotoxicity increased significantly, with IC50 values of 1.25 ± 0.39 µM (normoxia) and 0.18 ± 0.08 µM (hypoxia). Notably, the phototherapeutic index (PI, defined as IC50 in the dark/IC50 under irradiation) reached 44 under hypoxia—approximately six times higher than that under normoxia (PI = 7.54)—demonstrating its efficacy in overcoming limited oxygen availability. Furthermore, complex 1 effectively reduced Pt resistance, as evidenced by low resistance factors (RFs, defined as IC50 in resistant cells/IC50 in sensitive cells) under irradiation: 0.03 in hypoxia (IC50: 5.92 ± 0.57 µM in A549 and 0.18 ± 0.08 µM in A549cisR) and 0.54 in normoxia (IC50: 2.18 ± 1.21 µM in A549 and 1.25 ± 0.39 µM in A549cisR). Mechanistically, complex 1 exhibits negligible covalent interactions with calf thymus DNA (CT-DNA) in the dark, but efficiently unwinds the DNA superhelix via photoinduced production of ROS through a combination of both type I and type II photodynamic reactions. This ROS-mediated damage is associated with mitochondrial membrane depolarization and a substantial increase in apoptosis, as evidenced by strong Annexin V-FITC (green) and propidium iodide (red) signals in irradiated cells and negligible signals in dark controls. Flow cytometry confirmed this shift, with the percentage of apoptotic cells rising to 34.0% post-irradiation compared to 4.70% in the dark. In vivo studies further underscore the potential of complex 1 as a two-photon-activated PS, demonstrating near-complete tumor eradication with minimal systemic toxicity. These findings position complex 1 as a Pt-based phototherapeutic that surmounts both hypoxic limitations and cisplatin resistance through ROS-driven cell death induced by type I and type II photodynamic pathways, thereby bypassing the primary resistance pathways associated with conventional Pt chemotherapeutics.
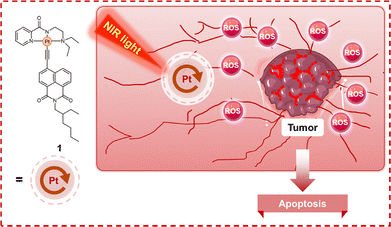 | ||
| Fig. 5 The structure of complex 1 and its mechanism of action. Its antitumor effect is driven by near-infrared light-induced ROS production, which triggers apoptotic cell death in the tumor cells. | ||
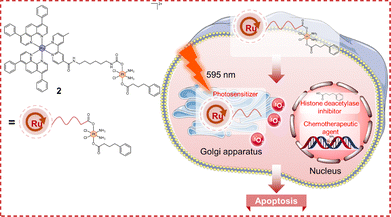
The clinical success of Pt(II)-based chemotherapeutics has spurred the development of Pt(IV) prodrugs, which are designed to reduce adverse effects and improve pharmacological performance by undergoing reduction specifically within cancer cells.66–69 Concurrently, Ru(II) polypyridyl complexes have emerged as promising PSs for PDT due to their superior tunability and photochemical properties.70–72 Heterobimetallic complexes incorporating Pt with a second metal represent a significant advancement in anticancer metallodrug design, as they integrate Pt's core cytotoxic mechanism with complementary actions from the secondary metal to broaden the therapeutic profile and overcome platinum resistance.73 Building on these advances, Gasser, Gibson, and colleagues developed a novel Pt(IV)–Ru(II) conjugate (2, Fig. 6) to combine cancer-activated chemotherapy with PDT.74 Complex 2 entered cells primarily via energy-dependent endocytosis. Following internalization and reduction of the Pt(IV) center, the constituent metals exhibited distinct subcellular localization: Pt accumulated primarily in the nucleus, while Ru localized predominantly to the Golgi apparatus. Upon irradiation, the Golgi-localized Ru species generated singlet oxygen, thereby exerting PDT effects. This combined action conferred potent photo-induced cytotoxicity in ovarian carcinoma A2780 cells and their resistant variants—A2780cisR (cisplatin-resistant) and A2780ADR (doxorubicin-resistant)—with IC50 values of 0.08–0.10 µM (480 nm) and 0.16–0.19 µM (595 nm). Notably, it significantly overcame resistance, showing RFs of 1.25 (480 nm) and 1.19 (595 nm) in A2780cisR/A2780 and 1.12 (480 nm) and 1.06 (595 nm) in A2780ADR/A2780. In contrast, cisplatin yielded substantially higher RF values of 4.30 and 1.98, respectively. Mechanistic studies revealed that apoptosis induced by cisplatin and complex 2 in the dark likely proceeded via the ERK/p53/PUMA pathway, as evidenced by the unaffected caspase-3/7 activity. In contrast, irradiated complex 2 significantly increased caspase-3/7 activity, suggesting a shift to a different photoactivated apoptotic mechanism. This shift may account for the potent ability of irradiated complex 2 to overcome resistance. Moreover, the marked rescue of cell viability by both apoptotic and paraptotic inhibitors upon irradiation implicates concurrent activation of these two death pathways. Complex 2 effectively penetrated multicellular tumor spheroids (MCTS), retaining dark cytotoxicity (IC50 = 7.32 µM) and achieving nanomolar phototoxicity (IC50 = 0.49–0.68 µM). This dual-action conjugate represents a promising strategy for overcoming resistance in cancer therapy and warrants further development.
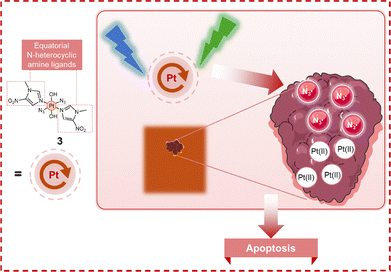
The prevailing approach for enhancing the anticancer activity of Pt(IV) complexes focuses on axial functionalization; however, the photocytotoxic efficacy of diazido Pt(IV) systems is profoundly affected by the choice of equatorial ligands, which govern their photoactivation dynamics.75–77 Nitroimidazole antibiotics, traditionally used to treat anaerobic infections, have recently gained recognition for their anticancer potential.78–80 In this context, Sadler, Shi, and Clarkson developed a diazido Pt(IV) prodrug (complex 3, Fig. 7) featuring two equatorial 1-methyl-5-nitroimidazole ligands.81 Complex 3 demonstrated potent and oxygen-independent photocytotoxicity under blue-light irradiation (IC50 < 5 µM) against bladder cancer cells, while exhibiting low dark toxicity and high selectivity over normal bladder cells (with IC50 values 3–20 times higher). Critically, it displayed a pronounced ability to circumvent cisplatin resistance, maintaining efficacy across varying oxygen concentrations. Its cytotoxicity upon blue light irradiation (465 nm) remained consistent across A2780 (IC50: 2.9 ± 0.9 µM under normoxia and 1.7 ± 0.6 µM under hypoxia) and cisplatin-resistant A2780cisR cells (IC50: 0.84 ± 0.06 µM under normoxia and 1.7 ± 0.3 µM under hypoxia), resulting in RFs of 0.29 and 1.0 in normoxia and hypoxia, respectively. By contrast, cisplatin showed an approximately 10-fold decrease in potency against A2780cisR cells, underscoring complex 3's superior resistance-bypassing profile. Mechanistically, photoactivated complex 3 generated significant ROS and subsequently induced apoptosis in SW780 bladder cancer cells under normoxic conditions, with early apoptosis increasing from 1.49% to 8.11% and late apoptosis from 6.67% to 45.82% relative to dark controls. This pronounced apoptotic response may account for the observed reversal of drug resistance under normoxia. In contrast, under hypoxia, complex 3 did not primarily induce apoptosis but instead promoted nuclear Pt accumulation and exacerbated mitochondrial membrane depolarization. These alternative mechanisms collectively contribute to overcoming both cisplatin- and hypoxia-mediated resistance. In addition to its cellular effects, complex 3 exhibited high liver microsomal stability and photo-enhanced Pt accumulation in rat bladder tissue, underscoring its potential for clinical translation as a phototherapeutic agent for bladder cancer. These findings establish equatorial nitroimidazole ligands as a viable strategy to enhance the photocytotoxicity of diazido Pt(IV) complexes, thereby overcoming dual resistance mechanisms and advancing the development of phototherapeutic Pt drug candidates.
3. Pt phototherapeutics that induce autophagy
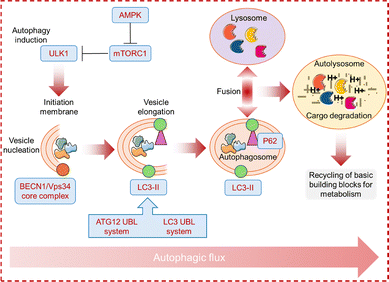
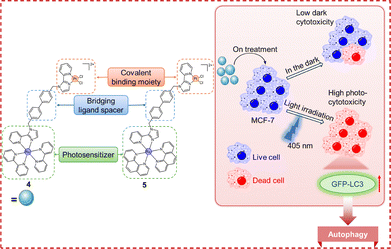
Autophagy is a conserved catabolic process vital for cellular homeostasis, acting as a double-edged sword: it promotes cell survival under stress but induces cell death when dysregulated.82,83 Autophagy is triggered by stimuli such as nutrient or growth factor deprivation, infection, and organelle damage, and it regulates cell development, differentiation, and tissue remodelling.84 Among its various forms, macroautophagy (hereafter referred to as autophagy) is the most prevalent and involves the formation of double-membraned autophagosomes that engulf cytoplasmic cargo for lysosomal degradation.54 The process proceeds through initiation, nucleation, elongation, autophagosome closure, and autolysosome formation (Fig. 8).85,86 Initiation is mediated by the ULK1 complex, followed by nucleation, which requires the BECN1/Vps34 lipid-kinase complex. Phagophore elongation employs ATG12 and light chain 3 (LC3) conjugation systems, in which LC3-I is lipidated to LC3-II—a canonical autophagosomal marker. The adaptor p62 binds ubiquitinated cargo and LC3-II,87 delivering substrates for degradation; accordingly, p62 accumulation signifies impaired autophagic flux.88 After lysosomal fusion, degraded materials are recycled to sustain metabolism and stress adaptation.89 Conversely, dysregulated flux disrupts proteostasis and homeostasis, promoting cell death.90 This functional duality underpins the therapeutic relevance of autophagy in cancer. Specifically, the induction of autophagy represents a promising avenue for countering Pt resistance in oncology. This mechanism can overcome resistance either by activating alternative, non-apoptotic cell death pathways or by selectively degrading pro-survival proteins that contribute to chemoresistance. Therefore, the rational design of novel Pt complexes that potently induce autophagy offers a strategic approach to re-sensitizing tumors to conventional chemotherapy and thereby improving chemotherapeutic efficacy against resistant cancers.The therapeutic potential of heterobimetallic complexes in PDT stems from their tunable photophysical properties.73,91 By leveraging synergistic intermetallic interactions, achieved through deliberate metal selection and ligand design, these complexes offer a superior alternative to conventional monometallic metallodrugs.92–94 A notable example is the work by Kumbhar et al., who developed heterobimetallic Ru(II)–Pt(II) polypyridyl complexes [Ru(bpy)2(BPIMBp)PtCl2]2 (4, Fig. 9) and [Ru(phen)2(BPIMBp)PtCl2]2 (5, Fig. 9).95 Cell-based studies revealed potent photocytotoxicity in MCF-7 cells (IC50 < 20 µM) upon irradiation at 450 nm, while maintaining low dark toxicity (IC50 > 300 µM). Because Pt-resistant cancers frequently exploit basal autophagy for survival,96 pharmacological modulation of autophagy – either by inhibiting cytoprotective flux or by inducing lethal autophagy – has emerged as a strategy to overcome chemoresistance.97 Motivated by this rationale, the authors investigated whether complexes 4 and 5 act as autophagy-modulating chemosensitizers to trigger autophagic cell death and thereby potentially overcome drug resistance. To monitor autophagy induction, the authors quantified microtubule-associated protein 1A/1B-LC3 fluorescence, using GFP-LC3 puncta formation as a marker. Treatment with complex 4 induced approximately a 3-fold increase in GFP-LC3 fluorescence in MCF-7 cells, while complex 5 induced approximately a 2-fold increase, both significantly exceeding the cisplatin-induced autophagy. These results suggest that the Ru-polypyridyl PS component of complexes 4 and 5 may partially overcome cisplatin resistance by inducing autophagy in cancer cells, although further detailed investigation is needed to confirm this.
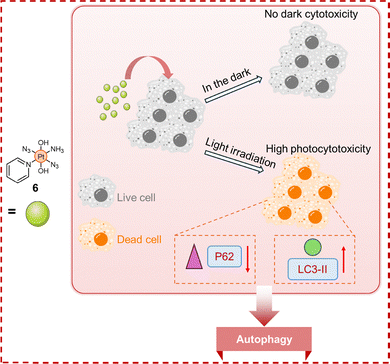
Pt(IV) diazido complexes, designed for photoactivation, exhibit distinctive photobiological properties.98 As reported by Bednarski and Sadler et al., trans,trans,trans-[Pt(N3)2(OH)2(py)(NH3)] (complex 6, Fig. 10) is a highly effective photoactivated anticancer agent, displaying significant cytotoxicity against a panel of human cancer cells when exposed to UVA irradiation, while remaining non-toxic in the dark.99 The mechanism underlying the light-induced cytotoxicity for complex 6 is distinct from that of cisplatin. First, complex 6 exhibited no cross-resistance in three cell lines resistant to oxoplatin (cis,trans,cis-[PtCl2(OH)2(NH3)2], a Pt(IV) prodrug derived from cisplatin), with RFs ranging from 0.88 to 1.20. In contrast, cisplatin exhibited complete cross-resistance in the same cell lines, with RFs of 2.07–2.89. Second, morphological changes in HL-60 cells treated with complex 6 differed significantly from those induced by cisplatin or etoposide,100,101 a topoisomerase II inhibitor used as a positive control for apoptosis. While cisplatin and etoposide induced characteristic apoptotic features, such as cell shrinkage, membrane blebbing, and caspase-3 activation, complex 6-treated cells exhibited slight swelling without apoptotic markers. Annexin V/propidium iodide assays further confirmed the absence of apoptosis in cells treated with complex 6 following UVA irradiation. Moreover, at IC90 concentrations, cisplatin and etoposide induced S–G2 and G2–M cell cycle arrests, respectively, whereas complex 6 exerted a minimal impact on cell cycle distribution. Notably, autophagy appeared to play a central role in the cytotoxicity of UVA-activated complex 6. HL-60 cells exposed to the activated complex for 6 hours showed a marked increase in LC3B-II levels and a corresponding decrease in p62 levels, consistent with autophagic activation. These findings provide novel insights, demonstrating that the photoinduced cytotoxicity of trans-diazido Pt(IV) complexes proceeds via an autophagy-mediated pathway distinct from cisplatin's mechanism, thereby presenting a promising strategy to circumvent Pt resistance.
4. Pt phototherapeutics that induce ferroptosis
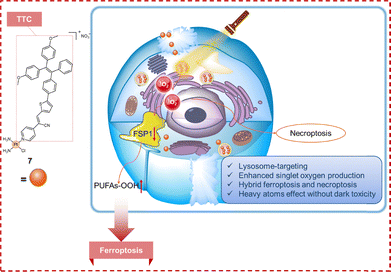
Ferroptosis is an iron-dependent form of regulated cell death driven by lethal accumulation of lipid peroxides (Fig. 11).102 Its core mechanism is intrinsically tied to cellular iron metabolism: extracellular Fe3+ bound to transferrin is imported via transferrin receptor 1 (TFR1), reduced to Fe2+ by STEAP3 in endosomes, and released into the labile iron pool by divalent metal transporter 1 (DMT1).103 The availability of this redox-active iron can be further modulated by phosphorylase kinase catalytic subunit gamma 2 (PHKG2).104 Excess Fe2+ promotes lipid peroxidation (LPO) through Fenton and Fenton-like chemistry, while enzymatic peroxidation of polyunsaturated fatty acids (PUFAs) by lipoxygenases (LOXs) also contributes.105 A primary defense against ferroptosis is mediated by glutathione peroxidase GPX4, which utilizes reduced glutathione (GSH) to reduce lipid hydroperoxides (LOOHs) to non-toxic lipid alcohols.106,107 GPX4 activity requires adequate GSH, whose synthesis depends on cysteine, primarily imported as cystine via the system Xc− antiporter.108,109 Inhibition of system Xc− (e.g., by erastin) or direct GPX4 inactivation (e.g., by RAS-selective lethal 3) causes lethal LOOH accumulation. GSH levels are further modulated by glutamine uptake through SLC1A5 and its conversion to glutamate by glutaminase (GLS).110 Beyond GPX4, ferroptosis suppressor protein 1 (FSP1) provides an independent protective axis:110 myristoylated FSP1 regenerates the radical-trapping antioxidant ubiquinol from ubiquinone using NAD(P)H, thereby suppressing LPO propagation.111 This underscores the protective roles of metabolites such as ubiquinol, squalene, and coenzyme Q10 (CoQ10), which are produced via the mevalonate pathway.112,113 Therapeutically, ferroptosis induction represents a promising strategy to mitigate Pt resistance. This iron-dependent form of cell death can bypass classical apoptotic defects by leveraging lipid peroxidation to eradicate resistant cancer cells. Consequently, the development of Pt-based agents designed to trigger ferroptosis provides a viable alternative to overcome intrinsic and acquired chemoresistance, thereby expanding the therapeutic utility of Pt-based chemotherapeutics.The rapid proliferation and resistance to apoptosis in cancer cells drive them into a state of heightened lipid and iron metabolism. This creates a metabolic vulnerability, paradoxically setting the stage for effective tumor eradication via ferroptosis.114 Therefore, PSs capable of inducing ferroptosis are of great interest for combating apoptosis-resistant tumors.115–117 Guo, He, and Chen et al. developed TTC-Pt (complex 7, Fig. 12), a monofunctional Pt(II) complex exhibiting aggregation-induced emission (AIE) activity. The incorporation of Pt(II) into the TTC framework enhanced intersystem crossing (ISC), substantially boosting singlet oxygen production upon photoactivation.118 TTC-Pt exhibited potent photoinduced growth inhibition against HeLa cells, with an IC50 of 0.34 ± 0.05 µM, significantly surpassing its precursor TTC (IC50 > 64.00 µM) and cisplatin (IC50 = 25.28 ± 1.32 µM), and showed negligible dark toxicity. Mechanistic investigations using specific inhibitors revealed that TTC-Pt-mediated PDT primarily induced ferroptosis and necroptosis, as evidenced by the inhibition of ferroptosis with ferrostatin-1 and the inhibition of necroptosis with necrostatin-1, rather than apoptosis or autophagy. Localized to lysosomes, TTC-Pt triggered ferroptosis via lipid peroxide accumulation and selectively downregulated FSP1 expression after irradiation, without altering GPX4 levels. This mechanism delineates a non-canonical pathway for ferroptosis induction driven by FSP1 suppression rather than GPX4 inhibition, likely resulting from TTC-Pt-induced disruption of cellular redox homeostasis. The concurrent induction of ferroptosis and necroptosis establishes a dual-mechanism strategy to effectively eliminate apoptosis-resistant tumor cells, offering a promising approach to circumventing chemotherapeutic resistance.
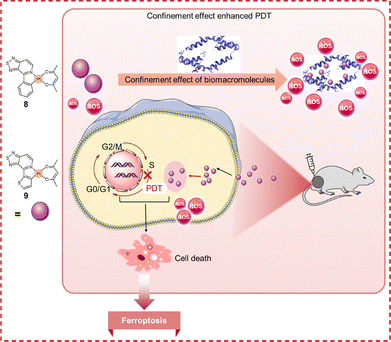
The aggregation of PSs often quenches ROS generation, thereby limiting their efficacy in PDT.119–121 To overcome this challenge and advance PDT applications, particularly in combating drug resistance, high-performance PSs operable under physiological conditions are essential. Yuan and Hu et al. developed two Pt-based PSs, complexes 8 and 9 (Fig. 13), which bind to biomacromolecules to mitigate aggregation-caused quenching and enhance photosensitizing efficacy in biologically relevant environments.122 Incorporating abundant heteroatoms into the Pt framework conferred these complexes a strong binding affinity for biological macromolecules. Upon confinement within bovine serum albumin (BSA), the molecular motion of the complexes became restricted, thereby promoting the involvement of the central metal in excited-state charge transfer. This significantly boosted ROS generation, yielding a 2–3-fold increase in ROS production for BSA-bound complexes compared to free agents. The complexes exhibited dark cytotoxicity (IC50 = 6 µM), likely due to biomacromolecule binding. Upon white light irradiation, cytotoxicity was markedly enhanced (IC50: complex 8 = 2.72 µM and complex 9 = 0.88 µM). This enhancement correlated with light-dependent BSA degradation (remaining percentages: 58.38% for complex 8 and 45.48% for complex 9) and elevated intracellular ROS and oxidative stress. Cell death pathway analysis using specific inhibitors—ferrostatin-1 (Fer-1) for ferroptosis, Z-VAD-FMK for apoptosis, and VX-765 for pyroptosis—revealed that ferroptosis inhibition substantially abrogated cytotoxicity, identifying ferroptosis as the primary PDT pathway. The induction of ferroptosis may circumvent conventional apoptotic signaling and its associated resistance mechanisms. This feature renders ferroptosis a promising therapeutic strategy for combating drug-resistant malignancies, though further investigation is warranted. Notably, complex 9 demonstrated superior biocompatibility and tumor eradication efficacy in vivo, achieving significant tumor growth inhibition post-irradiation compared to controls. This work establishes the approach of leveraging Pt(II)–biomacromolecule interactions to develop potent PSs for ferroptosis-mediated PDT, which show considerable potential in overcoming chemoresistance limitations.
Ferrocene, initially explored in conjugates with tamoxifen and chloroquine, has become a valuable moiety in drug design.123,124 Building on this concept, Sadler, Sicilia, and colleagues developed a ferrocene-conjugated Pt(IV) complex, Pt–Fe (complex 10, Fig. 14), by appending the photoactive trans,trans,trans-[Pt(N3)2(OH)2(py)2] to a ferrocene unit.125 The ferrocene antenna red shifts the absorption profile, enabling Fe → Pt charge transfer and photoactivation at longer wavelengths. Upon 465 nm irradiation, complex 10 exhibited potent photocytotoxicity in A2780 ovarian cancer cells under both normoxia (IC50 = 0.8 ± 0.2 µM) and hypoxia (IC50 = 1.3 ± 0.3 µM), with comparable activity in cisplatin-resistant A2780cisR cells (normoxia: 1.4 ± 0.5 µM; hypoxia: 1.0 ± 0.1 µM), yielding RFs of 1.8 (normoxia) and 0.8 (hypoxia). In contrast, cisplatin under identical conditions showed only modest activity in A2780 cells (normoxia: 0.9 ± 0.5 µM; hypoxia: 8.8 ± 1 µM) and markedly reduced efficacy in A2780cisR cells (normoxia: 7.67 ± 0.04 µM; hypoxia: 16.2 ± 1.0 µM), with RFs of 8.5 (normoxia) and 1.8 (hypoxia), underscoring complex 10's ability to overcome resistance to both cisplatin and hypoxia. Mechanistically, photoactivated complex 10 markedly elevated ROS levels via ferrocene-mediated Fenton-type catalysis and induced lipid peroxidation, along with approximately 50% GSH depletion and approximately 60% suppression of GPX4. These hallmarks indicate ferroptosis as the primary cell death pathway, distinguishing complex 10 from conventional Pt(II) drugs and typical PSs. Collectively, the intermetallic charge-transfer design, robust photocytotoxicity, resilience to resistance, and ferroptosis induction position complex 10 as a promising photochemotherapeutic prodrug and a blueprint for the next-generation phototherapeutic agents.
5. Pt phototherapeutics that induce pyroptosis
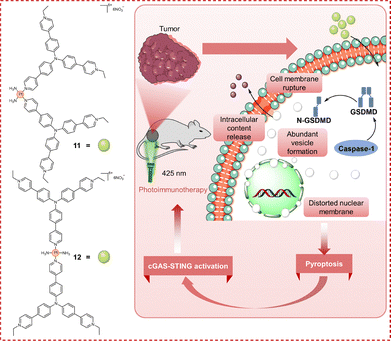
Pyroptosis is an immunogenic form of lytic, programmed cell death characterized by hallmark morphological alterations, such as cell swelling, plasma membrane pore formation, membrane rupture, and chromatin fragmentation.126,127 This process has garnered significant attention due to its critical role in immunity and its potential applications in cancer therapy. Pyroptosis is primarily executed through canonical and non-canonical pathways, both of which converge on GSDMD cleavage (Fig. 15).128–130 The canonical pathway is initiated by inflammasomes, which are multiprotein complexes (comprising pattern recognition receptors and adaptors such as ASC) that assemble in response to stimuli, such as pathogens, hypoxia, and damage-associated molecular patterns (DAMPs). Upon activation, inflammasomes catalyze the maturation of procaspase-1 into caspase-1; the latter subsequently cleaves both pro-interleukin-1β (pro-IL-1β) and pro-IL-18 into their bioactive forms while also cleaving GSDMD. The liberated GSDMD N-terminal domain oligomerizes in the plasma membrane, forming pores that induce lytic cell death and facilitate the extracellular release of potent inflammatory cytokines, particularly IL-1β and IL-18. This cytokine release can effectively reprogram the tumor microenvironment (TME), mitigating immunosuppression to convert immunologically “cold” tumors into “hot” phenotypes, thereby enhancing antitumor immunity.131,132 The non-canonical pathway is primarily activated by Gram-negative bacterial lipopolysaccharide (LPS), which directly activates caspase-4/5 in humans (or caspase-11 in mice) to cleave GSDMD, initiating pyroptosis independent of inflammasomes. However, the non-canonical pathway lacks the capacity to process pro-IL-1β and pro-IL-18. Notably, pyroptosis can propagate through a mechanism in which extracellular vesicles containing GSDMD pore-forming structures, released from pyroptotic donor cells, integrate into the membrane of bystander cells, thereby triggering a cascade of pyroptosis.133 This domino-like amplification of inflammatory signaling and immune activation holds significant therapeutic potential and could overcome limitations of traditional treatments, such as apoptosis-driven immune evasion and apoptosis-related drug resistance. Therefore, the development of Pt-based agents designed to elicit pyroptosis provides a viable therapeutic approach to overcoming chemoresistance and enhancing treatment efficacy in refractory malignancies.The activation of the cGAS-STING pathway, a promising anticancer immunotherapeutic strategy, can be effectively achieved through photo-induced pyroptosis.131,134–136 Previous work has established the biological applications of triphenylamine derivatives.137,138 On this basis, Mao, Tan, and colleagues developed two novel small-molecule photoactivators, complexes 11 and 12 (Fig. 16), to target the cGAS-STING pathway and induce pyroptosis.139 These Pt(II) complexes are structural analogues of cisplatin and transplatin, respectively, and incorporate an alkylated triphenylamine ligand. Complexes 11 and 12 exhibited negligible dark cytotoxicity (IC50 > 200 µM) across multiple cancer cell lines (HeLa, MDA-MB-231, A549, and U14). However, under 425 nm light irradiation, both complexes demonstrated significant photocytotoxicity, with the PI ranging from 17 to 106. Complex 11 displayed higher photocytotoxicity overall, with HeLa and U14 cells being particularly sensitive, as demonstrated by PIs of 79 and 106, respectively. In contrast, neither cisplatin nor transplatin showed any photocytotoxicity under identical conditions. PDT mediated by complexes 11 and 12 elicited pyroptosis, which was thoroughly confirmed through multiple lines of evidence. Transmission electron microscopy (TEM) of irradiated HeLa cells treated with complexes 11 or 12 revealed the characteristic pyroptotic morphology: cell membrane rupture, abundant vesicle formation, intracellular content release, and distorted nuclear membranes—observations further corroborated by confocal microscopy. In contrast, unirradiated controls displayed no such alterations. Inhibitor studies reinforced these findings; cell death was reduced by necrostatin-1 (a necroptosis inhibitor) and more significantly suppressed by necrosulfonamide (NSA; a necroptosis/pyroptosis inhibitor). Conversely, inhibitors of autophagy (3-methyladenine and chloroquine) or pan-caspase activity (Z-VAD-FMK) had minimal effects. Furthermore, western blotting detected a significant increase in GSDMD-N expression, the pyroptosis executioner protein responsible for plasma membrane pore formation, following photoactivation of the complexes. Thus, integrated morphological assessment, pharmacological inhibition, and biochemical detection unequivocally demonstrated that these complexes induce pyroptotic cell death. Moreover, light activation of these complexes sequentially caused damage to mitochondrial DNA (mtDNA), the nuclear envelope, and nuclear DNA—including G-quadruplex structures and double-stranded DNA. This cascade of genomic stress activated the cGAS-STING pathway. Along with the concomitant induction of pyroptosis, this dual activation elicited potent antitumor immune responses. Beyond their potent cellular effects, photoactivated complexes 11 and 12 significantly inhibited tumor growth in animal models. This was accompanied by the release of the 2′,3′-cyclic GMP-AMP (cGAMP) and cytokines, enhanced infiltration of CD8+/CD4+ T cells, and promotion of dendritic cell maturation. This study thereby pioneers small-molecule STING photoactivators and demonstrates that coupling their targeted activation with pyroptosis induction offers a strategic, dual-pronged approach to developing novel immunotherapeutics capable of overcoming resistance.
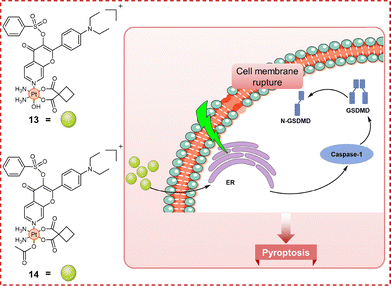
Photoactivated Pt(IV) prodrugs derived from clinical Pt(II) agents have drawn significant interest and typically employ O-donor axial ligands as the photosensitive components.140 In contrast, N-donor ligands, especially N-heteroaromatics, offer a compelling alternative by promoting ligand-to-metal charge transfer (LMCT) and thereby enhancing Pt(IV) photoreactivity.141,142 Very recently, our group developed two green-light-activatable carboplatin-based Pt(IV) prodrugs, flavoplatins 13 and 14 (Fig. 17), which incorporate flavonol derivatives as axial N-donor ligands.143 These prodrugs undergo rapid photoreduction to release the active Pt(II) species together with the axial ligands. Upon green-light irradiation, flavoplatin 13 exhibited IC50 values of 18.3 ± 4.8 µM and 14.2 ± 4.2 µM in A2780 and A2780cisR cells, respectively, with an RF of 0.8, while flavoplatin 14 achieved IC50 values of 15.1 ± 0.7 µM and 15.6 ± 4.5 µM in the same cell lines, corresponding to an RF of 1.0. By comparison, carboplatin's photocytotoxicity in both cell lines exceeded 500 µM, indicating at least a 27-fold potency advantage for both flavoplatins. Mechanistically, both complexes preferentially accumulated in the ER and promptly induced pyroptosis via the NLRP3/caspase-1/GSDMD pathway. This was evidenced by elevated NLRP3 expression, reduced pro-caspase-1 levels (by 39% for 13 and 28% for 14), and a >3-fold increase in GSDMD N-terminal fragment, accompanied by a 26% decrease in full-length GSDMD for 13 in treated A2780cisR cells. These results confirm that light-activated flavoplatins can trigger pyroptosis in A2780cisR cells, a mechanism distinct from the apoptosis typically induced by conventional Pt(II) drugs, thereby offering a potential strategy to circumvent platinum resistance. Collectively, these findings highlight the therapeutic potential of Pt(IV) prodrugs bearing axial N-donor ligands, particularly their ability to engage non-apoptotic cell death pathways to mitigate drug resistance.
6. Pt phototherapeutics that induce ICD
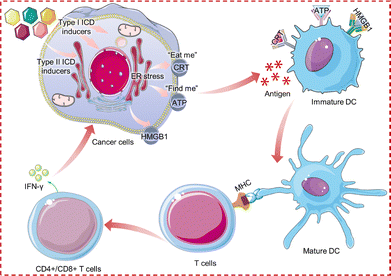
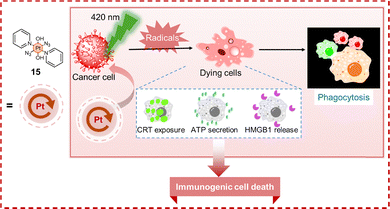
ICD, a form of PCD defined by the NCCD as capable of eliciting adaptive immunity in immunocompetent hosts, was first identified in 2005 by Kroemer et al. based on their observation that anthracycline-treated tumor cells can provoke adjuvant-free immune responses.41,144 This stress-induced process stimulates inflammatory responses that activate cytotoxic T lymphocyte (CTL)-mediated adaptive immunity and establishes long-lasting immunological memory, thereby gaining significant research interest in recent years.53,145,146 The induction of ICD is characterized by several biochemical hallmarks (Fig. 18): (i) during early apoptosis, endoplasmic reticulum (ER)-resident calreticulin (endo-CRT) translocates to the plasma membrane (ecto-CRT), where it functions as an “eat me” signal by binding to the CD91 receptor on immature dendritic cells (DCs) and macrophages, thereby promoting phagocytosis; (ii) dying cells actively secrete ATP, which serves as a “find me” signal to attract antigen-presenting cells via P2RX7 receptor binding; and (iii) during late apoptosis, membrane damage facilitates the release of the nuclear protein HMGB1 into the extracellular space, further enhancing immunogenicity.147–149 Crucially, the simultaneous occurrence of CRT exposure, ATP secretion, and HMGB1 release is essential for ICD efficacy. The absence or neutralization of any single hallmark abrogates immunogenicity, compromising the effectiveness of immunochemotherapy.150,151 ER stress is a pivotal trigger for ICD, with ICD inducers classified as type I (e.g., mitoxantrone, doxorubicin, and oxaliplatin) or type II (e.g., coxsackievirus B3, hypericin, and oncolytic viruses) according to their ER targeting specificity.149,152,153 Type I inducers act on diverse cellular compartments and indirectly provoke ER stress and DAMP release through secondary effects. In contrast, type II inducers primarily localize within the ER, directly disrupting its homeostasis via oxidative stress, an action that facilitates ICD.154 Type II inducers are thought to elicit a stronger ICD immune response due to their capacity for generating higher DAMP levels.147 The immunogenicity of ICD ultimately arises from the release of DAMPs, which signal through pattern recognition receptors (PRRs) on antigen-presenting cells (APCs). This interaction orchestrates the uptake and presentation of tumor antigens, culminating in APC activation and the priming of tumor-specific T cells. These effector T cells subsequently infiltrate and eliminate malignant cells. The strategic development of Pt-based ICD inducers exemplifies a promising approach to overcoming conventional drug resistance. Unlike cisplatin, which induces immunologically silent apoptosis, ICD inducers emit a suite of danger signals that activate dendritic cells and prime a cytotoxic T cell response. This mechanism effectively recruits the host's immune system to eradicate both primary and metastatic cancer cells, thus bypassing intrinsic resistance mechanisms and potentially conferring durable, systemic protection against relapse.Photoactivatable Pt(IV) complexes represent compelling candidates for overcoming Pt resistance through a mechanism of action distinct from that of cisplatin. A prime example is trans,trans,trans-[Pt(N3)2(OH)2(py)2] (complex 15, Fig. 19), a stable and non-toxic prodrug in the dark that becomes highly cytotoxic upon UVA/visible light irradiation.155 Upon photoactivation, complex 15 generates reactive Pt(II) species that target DNA, while simultaneously releasing ROS and reactive nitrogen species (RNS). These species collectively elevate intracellular oxidative stress and promote the induction of ICD. Photoactivated complex 15 (at 1 or 5 µM), unlike cisplatin (at 20 µM), induced significant ecto-CRT exposure in A2780 ovarian cancer cells upon irradiation, as confirmed by flow cytometry and confocal microscopy. This response was comparable to that of established ICD inducers such as doxorubicin (at 20 µM) and oxaliplatin (at 100 µM). Furthermore, photoactivated complex 15, together with doxorubicin and oxaliplatin, stimulated the release of the key ICD biomarkers HMGB1 and ATP from A2780 cells in a concentration-dependent manner. In contrast, untreated cells, cisplatin, or complex 15 in the dark failed to elicit these biomarkers. Crucially, photoactivated complex 15 uniquely promoted efficient phagocytosis of murine CT26 carcinoma cells by J774-A1 macrophages (35.1 ± 5.6%), significantly outperforming both complex 15 in the dark (2.8 ± 0.4%) and untreated controls (2.4 ± 0.2%). Remarkably, oxaliplatin (100 µM), despite inducing ecto-CRT and ICD biomarkers, did not elicit comparable phagocytosis, and cisplatin (20 µM) showed negligible activity. This dual ability to induce ICD biomarkers and potent phagocytosis reveals a new mechanism of action for complex 15, highlighting its potential to circumvent Pt resistance by eliciting robust antitumor immunity.
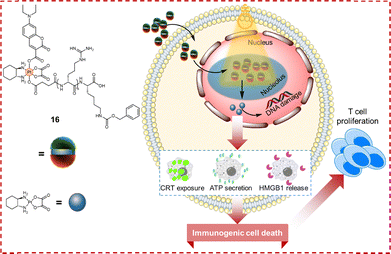
Light-triggered ICD inducers remain rare, positioning photoactivatable Pt complexes as compelling candidates. Rationally designed photocaged Pt(IV) prodrugs enable spatiotemporally controlled activation, minimizing off-target effects while potentially eliciting ICD-mediated antitumor immunity. Motivated by this potential, our group developed coumaplatin (complex 16, Fig. 20), an oxaliplatin-derived prodrug functionalized with a photocaging coumarin moiety and a nuclear localization sequence (R8K) for nucleolus targeting.156 Coumaplatin exhibited exceptional stability in the dark and rapid photoactivation within cells. Upon irradiation, it significantly outperformed oxaliplatin, demonstrating a more than 2-fold increase in DNA binding efficiency and up to 96-fold enhanced phototoxicity. Additionally, coumaplatin exhibited a superior ability to circumvent Pt resistance. In Pt-resistant A2780cisR and A549cisR cells, oxaliplatin displayed RFs of 5.1 and 2.8, respectively, while photoactivated coumaplatin markedly reduced these RFs to 1.3 and 0.6. Critically, photoactivated coumaplatin—unlike oxaliplatin or its inactive form of coumaplatin—induced ICD-associated DAMPs in resistant cells. Specifically, it significantly induced CRT exposure on A549cisR cell membranes, as confirmed by both microscopy and flow cytometry. Furthermore, photoactivated coumaplatin triggered HMGB1 translocation from the nucleus to the cytoplasm and significantly increased extracellular ATP secretion (56.7 nM versus 35.9 nM with oxaliplatin). Notably, low-dose photoactivated coumaplatin elicited ICD and robust T cell activation in resistant cells, effects that are unattainable even with high-dose oxaliplatin. By leveraging nucleolus-targeted photoactivation, coumaplatin uniquely induced potent ICD and profoundly overcame Pt resistance, heralding a paradigm shift for light-triggered antitumor immunity.
Cancer immunotherapy, while transformative, is often undermined by tumor immune evasion via downregulation of major histocompatibility complex class I (MHC-I).157,158 To address this challenge, Guo, Mao, and colleagues rationally designed a trinuclear Ru–Pt complex, TriRuPt (complex 17, Fig. 21), which leverages the photophysical properties of transition metals to modulate MHC-I expression.159 Complex 17 demonstrated superior light-triggered cytotoxicity compared to cisplatin, effectively overcoming drug resistance, as demonstrated by IC50 values of 0.144 µM in human lung adenocarcinoma A549 cells and 0.245 µM in cisplatin-resistant A549cisR cells, yielding a PI exceeding 100 and an RF of 1.7. In contrast, cisplatin exhibited IC50 values of 2.74 µM (A549) and 12.0 µM (A549cisR), with an RF of 4.4. Mechanistically, complex 17 functions as a PS to generate ROS, initiating a dual immunomodulatory cascade: (1) light-driven upregulation of MHC-I, which promotes T cell recognition and activation, and (2) induction of ICD, characterized by release of DAMPs such as HMGB1, cell-membrane exposure of CRT, and elevated ATP secretion, thereby enhancing dendritic cell-mediated antigen presentation and amplifying T cell responses. Through this dual mechanism, complex 17 converts immune-“cold” tumors to immune-“hot” phenotypes and acts synergistically with immune checkpoint inhibitors (ICIs) in vivo, resulting in suppressed tumor growth, reprogramming of the tumor microenvironment, and enhanced adaptive immunity. Overall, this metal-mediated PDT approach simultaneously elevates MHC-I levels and triggers ICD to enhance ICI therapy, uncovering a novel immunoregulatory function of transition metal complexes and paving the way for metal-based strategies to circumvent tumor immune evasion and resistance via integrated immunomodulation.
7. Conclusions and future perspectives
Conventional Pt-based chemotherapeutics induce cytotoxicity primarily by inhibiting proliferation and triggering apoptosis. However, their efficacy is often compromised by intrinsic or acquired resistance, frequently resulting from defects within apoptotic signaling cascades. In contrast, photoactivatable Pt complexes represent a transformative strategy, leveraging the light-triggered generation of reactive Pt species and/or radicals that target cellular components independent of classical Pt–DNA adduct formation, thereby effectively bypassing canonical resistance mechanisms. By either potentiating apoptosis or engaging alternative PCD modalities, including autophagy, ferroptosis, pyroptosis, and ICD (Fig. 22), these agents dismantle tumor defenses through the combined actions of photoinduced species. For example, autophagy can proceed despite defects in apoptosis, providing an alternative degradation route. Ferroptosis, driven by iron-dependent lipid peroxidation or inhibition of ferroptosis suppressor protein 1, eliminates cells refractory to apoptosis. Pyroptosis, initiated via inflammasome activation and subsequent gasdermin-mediated pore formation, induces direct lytic death while provoking robust inflammatory responses that synergize with immunotherapy. ICD converts immunologically “cold” tumors into “hot” ones by promoting dendritic cell maturation and cytotoxic T lymphocyte infiltration. Collectively, photoactivatable Pt complexes that trigger diverse cell death pathways provide a versatile, spatiotemporally precise approach to re-sensitizing resistant tumors and improving therapeutic outcomes (Table 1).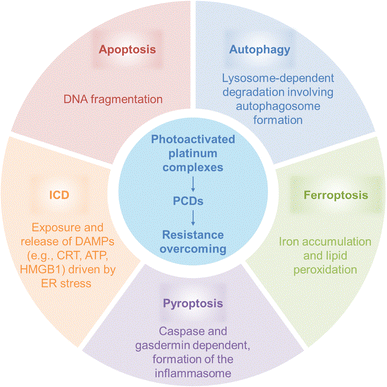 | ||
| Fig. 22 Expanding the cell death repertoire: a schematic of the biochemical features of diverse programmed cell death pathways induced by photoactivated Pt complexes to circumvent drug resistance. | ||
| PCD | Representative Pt complex | Mechanism | Key marker | Resistance-bypassing advantage | Ref. |
|---|---|---|---|---|---|
| a RF (resistance factor) = IC50 (platinum-resistant cell line)/IC50 (platinum-sensitive cell line). | |||||
| Apoptosis | Complex 2 (irradiated at 480/595 nm) | Photoactivated shift to caspase-mediated apoptosis | Increased caspase 3/7 activity under irradiation | Overcomes resistance; low RFsa (1.06–1.25 in A2780cisR/A2780 and A2780ADR/A2780) vs. cisplatin (RFs: 1.98–4.30) | 74 |
| Autophagy | Complex 6 (UVA-activated) | UVA-activated autophagic flux | Increased LC3B-II; decreased p62 | Overcome resistance; low RFs (0.88–1.20) vs. cisplatin (RFs: 2.07–2.89) in resistant cell lines | 99 |
| Ferroptosis | Complex 10 (465 nm-irradiated) | Photoactivated ROS elevation via Fenton catalysis, leading to lipid peroxidation and antioxidant depletion | Elevated ROS; lipid peroxidation; ∼50% GSH depletion; ∼60% GPX4 reduction | Bypasses cisplatin and hypoxia resistance; RFs 0.8–1.8 (normoxia/hypoxia) in A2780cisR/A2780 vs. cisplatin 1.8–8.5 | 125 |
| Pyroptosis | Flavoplatin 13 (green light-activated) | ER-targeted activation of the NLRP3/caspase-1/GSDMD pathway | Elevated NLRP3; reduced pro-caspase-1; >3-fold increased GSDMD-NT; 26% decreased full-length GSDMD | Circumvents cisplatin resistance with an RF of 0.in A2780cisR/A2780 cells; ≥27-fold more photocytotoxic than carboplatin | 143 |
| Immunogenic cell death (ICD) | Complex 16 (visible light 450 nm-irradiated) | Photoactivated DAMP exposure triggering the immune response | CRT membrane exposure; HMGB1 nuclear-to-cytoplasmic translocation; extracellular ATP secretion | Circumvents Pt resistance; RFs 0.6–1.3 in A2780cisR/A549cisR vs. oxaliplatin 2.8–5.1 | 156 |
Future advancements in phototherapeutic Pt complexes necessitate a strategic shift from primarily inducing apoptosis toward the deliberate engagement of non-apoptotic PCD pathways, particularly ICD and pyroptosis, which uniquely leverage antitumor immunity to counteract immunosuppressive microenvironments. A major bottleneck remains the lack of predictive design principles; the activation of these pathways in current photoactivatable Pt complexes is largely discovered through serendipity. Emerging studies have begun to identify actionable targets that trigger non-apoptotic PCD, such as mTOR inhibition for autophagy-dependent cell death, GPX4 inhibition for ferroptosis, ER stress for ICD, and GSDMD cleavage for pyroptosis, pointing to opportunities for rational design. Beyond these, additional forms of cell death—such as disulfidptosis, oncosis, paraptosis, necroptosis, necrosis, and parthanatos—represent further frontiers. The rational design of Pt phototherapeutics to modulate these targets may open new avenues to overcome resistance. Moreover, the strategic induction of multi-pathway cell death, potentially via combinatorial approaches, offers a powerful means to circumvent single-agent resistance and improve therapeutic outcomes. In parallel, deep learning (DL) can help predict multifaceted cytotoxic profiles in cases where structure–activity relationships (SARs) are unclear, but its success depends on large-scale, high-quality datasets; therefore, high-throughput synthesis, screening, and evaluation of photoactivatable Pt complexes will be essential to generate robust training data.
Equally critical is the transition to physiologically relevant preclinical models. Many resistance studies still rely on monolayer cultures that do not adequately recapitulate tumor architecture, hypoxia gradients, and stromal interactions. Patient-derived organoids and tumoroids preserve native 3D organization, genetic heterogeneity, and microenvironmental fidelity, providing more predictive platforms to evaluate whether phototherapeutic Pt agents can overcome clinically relevant resistance mechanisms. In addition, tissue penetration by light is strongly wavelength-dependent, with NIR (700–900 nm) light achieving substantially deeper penetration and reduced off-target phototoxicity, thereby enhancing clinical translatability. Accordingly, future efforts should prioritize the rational design and development of NIR-activatable Pt complexes.
In summary, photoactivatable Pt complexes represent a transformative leap forward in overcoming Pt resistance by redirecting cytotoxicity from conventional DNA damage-driven apoptosis toward multimodal, immunity-engaging regulated cell death modalities. By orchestrating diverse PCD pathways with spatiotemporal control, phototherapeutic Pt agents offer robust solutions to chemoresistance and lay the groundwork for next-generation precision oncology therapeutics.
Author contributions
The manuscript was written through contributions of S. C., Z. G., and G. Z. All authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
We thank the Hong Kong Research Grants Council (grant nos. CityU 11313222, CityU 11304923, CityU 11306724, and CityU C1018-23G), the National Natural Science Foundation of China (grant nos. 22277103 and 22525703), the Science Technology and Innovation Committee of Shenzhen Municipality (JCYJ20210324120004011), and the City University of Hong Kong (grant no. 7020014) for funding support. Parts of the scheme and figures were drawn using pictures from Servier Medical Art. Servier Medical Art by Servier is licensed under a Creative Commons Attribution 4.0 Unported License (https://creativecommons.org/licenses/by/4.0/).References
- L. Kelland, Nat. Rev. Cancer, 2007, 7, 573–584 Search PubMed.
- B. W. Harper, A. M. Krause-Heuer, M. P. Grant, M. Manohar, K. B. Garbutcheon-Singh and J. R. Aldrich-Wright, Chem. – Eur. J., 2010, 16, 7064–7077 CrossRef CAS PubMed.
- N. J. Wheate, S. Walker, G. E. Craig and R. Oun, Dalton Trans., 2010, 39, 8113–8127 Search PubMed.
- T. C. Johnstone, K. Suntharalingam and S. J. Lippard, Chem. Rev., 2016, 116, 3436–3486 CrossRef CAS PubMed.
- M. D. Hall, H. R. Mellor, R. Callaghan and T. W. Hambley, J. Med. Chem., 2007, 50, 3403–3411 Search PubMed.
- S. Dilruba and G. V. Kalayda, Cancer Chemother. Pharmacol., 2016, 77, 1103–1124 Search PubMed.
- J. J. Roberts and A. J. Thomson, Prog. Nucleic Acid Res. Mol. Biol., 1979, 22, 71–133 CAS.
- O. Pinato, C. Musetti and C. Sissi, Metallomics, 2014, 6, 380–395 CrossRef CAS PubMed.
- E. R. Jamieson and S. J. Lippard, Chem. Rev., 1999, 99, 2467–2498 Search PubMed.
- S. W. G. Tait and D. R. Green, Nat. Rev. Mol. Cell Biol., 2010, 11, 621–632 CrossRef CAS PubMed.
- T. Boulikas and M. Vougiouka, Oncol. Rep., 2003, 10, 1663–1682 Search PubMed.
- D. Wang and S. J. Lippard, Nat. Rev. Drug Discovery, 2005, 4, 307–320 CrossRef CAS PubMed.
- S. Dasari and P. B. Tchounwou, Eur. J. Pharmacol., 2014, 740, 364–378 CrossRef CAS PubMed.
- T. C. Johnstone, K. Suntharalingam and S. J. Lippard, Philos. Trans. R. Soc., A, 2015, 373, 20140185 Search PubMed.
- K. D. Mjos and C. Orvig, Chem. Rev., 2014, 114, 4540–4563 CrossRef CAS PubMed.
- S. Rottenberg, C. Disler and P. Perego, Nat. Rev. Cancer, 2021, 21, 37–50 CrossRef CAS PubMed.
- N. Nagai, R. Okuda, M. Kinoshita and H. Ogata, J. Pharm. Pharmacol., 1996, 48, 918–924 CrossRef CAS PubMed.
- E. J. Anthony, E. M. Bolitho, H. E. Bridgewater, O. W. L. Carter, J. M. Donnelly, C. Imberti, E. C. Lant, F. Lermyte, R. J. Needham, M. Palau, P. J. Sadler, H. Shi, F.-X. Wang, W.-Y. Zhang and Z. Zhang, Chem. Sci., 2020, 11, 12888–12917 RSC.
- A. Casini and A. Pöthig, ACS Cent. Sci., 2024, 10, 242–250 Search PubMed.
- Y. Wang, B. Cao, Q. Wang, S. Zhong, X. Fang, J. Wang, A. S. C. Chan, X. Xiong and T. Zou, Nat. Commun., 2025, 16, 7347 Search PubMed.
- M. D. Coskun, F. Ari, A. Y. Oral, M. Sarimahmut, H. M. Kutlu, V. T. Yilmaz and E. Ulukaya, Bioorg. Med. Chem., 2013, 21, 4698–4705 CrossRef CAS PubMed.
- Ž. D. Bugarčić, J. Bogojeski and R. van Eldik, Coord. Chem. Rev., 2015, 292, 91–106 CrossRef.
- T. Lazarević, A. Rilak and Ž. D. Bugarčić, Eur. J. Med. Chem., 2017, 142, 8–31 Search PubMed.
- I. Ott, Coord. Chem. Rev., 2009, 253, 1670–1681 Search PubMed.
- S. Nobili, E. Mini, I. Landini, C. Gabbiani, A. Casini and L. Messori, Med. Res. Rev., 2010, 30, 550–580 Search PubMed.
- C. S. Allardyce and P. J. Dyson, Platinum Met. Rev., 2001, 45, 62–69 Search PubMed.
- N. Muhammad and Z. Guo, Curr. Opin. Chem. Biol., 2014, 19, 144–153 CrossRef CAS PubMed.
- R. Oun, Y. E. Moussa and N. J. Wheate, Dalton Trans., 2018, 47, 6645–6653 Search PubMed.
- J. T. Hartmann and H.-P. Lipp, Expert Opin. Pharmacother., 2003, 4, 889–901 Search PubMed.
- Z. H. Siddik, Oncogene, 2003, 22, 7265–7279 Search PubMed.
- A.-M. Florea and D. Büsselberg, Cancers, 2011, 3, 1351–1371 CrossRef CAS PubMed.
- M. Ohmichi, J. Hayakawa, K. Tasaka, H. Kurachi and Y. Murata, Trends Pharmacol. Sci., 2005, 26, 113–116 Search PubMed.
- G. Giaccone, Drugs, 2000, 59, 9–17 CrossRef CAS PubMed.
- J. Zhou, Y. Kang, L. Chen, H. Wang, J. Liu, S. Zeng and L. Yu, Front. Pharmacol., 2020, 11, 343 CrossRef CAS PubMed.
- V. Brabec and J. Kasparkova, Drug Resistance Updates, 2002, 5, 147–161 CrossRef CAS PubMed.
- F. H. Igney and P. H. Krammer, Nat. Rev. Cancer, 2002, 2, 277–288 CrossRef CAS PubMed.
- S. Elmore, Toxicol. Pathol., 2007, 35, 495–516 CrossRef CAS PubMed.
- E. V. Sazonova, G. S. Kopeina, E. N. Imyanitov and B. Zhivotovsky, Cell Death Discovery, 2021, 7, 155 CrossRef CAS PubMed.
- U. Ziegler and P. Groscurth, Physiology, 2004, 19, 124–128 CrossRef CAS PubMed.
- L. Galluzzi, M. C. Maiuri, I. Vitale, H. Zischka, M. Castedo, L. Zitvogel and G. Kroemer, Cell Death Differ., 2007, 14, 1237–1243 CrossRef CAS PubMed.
- L. Galluzzi, I. Vitale, S. A. Aaronson, J. M. Abrams, D. Adam, P. Agostinis, E. S. Alnemri, L. Altucci, I. Amelio, D. W. Andrews, M. Annicchiarico-Petruzzelli, A. V. Antonov, E. Arama, E. H. Baehrecke, N. A. Barlev, N. G. Bazan, F. Bernassola, M. J. M. Bertrand, K. Bianchi, M. V. Blagosklonny, K. Blomgren, C. Borner, P. Boya, C. Brenner, M. Campanella, E. Candi, D. Carmona-Gutierrez, F. Cecconi, F. K.-M. Chan, N. S. Chandel, E. H. Cheng, J. E. Chipuk, J. A. Cidlowski, A. Ciechanover, G. M. Cohen, M. Conrad, J. R. Cubillos-Ruiz, P. E. Czabotar, V. D'Angiolella, T. M. Dawson, V. L. Dawson, V. De Laurenzi, R. De Maria, K.-M. Debatin, R. J. DeBerardinis, M. Deshmukh, N. Di Daniele, F. Di Virgilio, V. M. Dixit, S. J. Dixon, C. S. Duckett, B. D. Dynlacht, W. S. El-Deiry, J. W. Elrod, G. M. Fimia, S. Fulda, A. J. García-Sáez, A. D. Garg, C. Garrido, E. Gavathiotis, P. Golstein, E. Gottlieb, D. R. Green, L. A. Greene, H. Gronemeyer, A. Gross, G. Hajnoczky, J. M. Hardwick, I. S. Harris, M. O. Hengartner, C. Hetz, H. Ichijo, M. Jäättelä, B. Joseph, P. J. Jost, P. P. Juin, W. J. Kaiser, M. Karin, T. Kaufmann, O. Kepp, A. Kimchi, R. N. Kitsis, D. J. Klionsky, R. A. Knight, S. Kumar, S. W. Lee, J. J. Lemasters, B. Levine, A. Linkermann, S. A. Lipton, R. A. Lockshin, C. López-Otín, S. W. Lowe, T. Luedde, E. Lugli, M. MacFarlane, F. Madeo, M. Malewicz, W. Malorni, G. Manic, J.-C. Marine, S. J. Martin, J.-C. Martinou, J. P. Medema, P. Mehlen, P. Meier, S. Melino, E. A. Miao, J. D. Molkentin, U. M. Moll, C. Muñoz-Pinedo, S. Nagata, G. Nuñez, A. Oberst, M. Oren, M. Overholtzer, M. Pagano, T. Panaretakis, M. Pasparakis, J. M. Penninger, D. M. Pereira, S. Pervaiz, M. E. Peter, M. Piacentini, P. Pinton, J. H. M. Prehn, H. Puthalakath, G. A. Rabinovich, M. Rehm, R. Rizzuto, C. M. P. Rodrigues, D. C. Rubinsztein, T. Rudel, K. M. Ryan, E. Sayan, L. Scorrano, F. Shao, Y. Shi, J. Silke, H.-U. Simon, A. Sistigu, B. R. Stockwell, A. Strasser, G. Szabadkai, S. W. G. Tait, D. Tang, N. Tavernarakis, A. Thorburn, Y. Tsujimoto, B. Turk, T. Vanden Berghe, P. Vandenabeele, M. G. Vander Heiden, A. Villunger, H. W. Virgin, K. H. Vousden, D. Vucic, E. F. Wagner, H. Walczak, D. Wallach, Y. Wang, J. A. Wells, W. Wood, J. Yuan, Z. Zakeri, B. Zhivotovsky, L. Zitvogel, G. Melino and G. Kroemer, Cell Death Differ., 2018, 25, 486–541 CrossRef PubMed.
- D. Tang, R. Kang, T. V. Berghe, P. Vandenabeele and G. Kroemer, Cell Res., 2019, 29, 347–364 CrossRef CAS PubMed.
- F. Peng, M. Liao, R. Qin, S. Zhu, C. Peng, L. Fu, Y. Chen and B. Han, Signal Transduction Targeted Ther., 2022, 7, 286 CrossRef CAS PubMed.
- K. M. Kuznetsov, K. Cariou and G. Gasser, Chem. Sci., 2024, 15, 17760–17780 RSC.
- H. Shi, R. C. Marchi and P. J. Sadler, Angew. Chem., Int. Ed., 2025, 64, e202423335 CrossRef CAS PubMed.
- Y. Zhang, C. Xu, X. Yang and K. Pu, Adv. Mater., 2020, 32, 2002661 CrossRef CAS PubMed.
- T. C. Pham, V.-N. Nguyen, Y. Choi, S. Lee and J. Yoon, Chem. Rev., 2021, 121, 13454–13619 CrossRef CAS PubMed.
- J.-J. Hu, Q. Lei and X.-Z. Zhang, Prog. Mater. Sci., 2020, 114, 100685 CrossRef CAS.
- K. Xiong, F. Wei, Y. Chen, L. Ji and H. Chao, Small Methods, 2023, 7, 2201403 CrossRef CAS PubMed.
- M. Dichiara, O. Prezzavento, A. Marrazzo, V. Pittala, L. Salerno, A. Rescifina and E. Amata, Eur. J. Med. Chem., 2017, 142, 459–485 CrossRef CAS PubMed.
- A. P. Castano, T. N. Demidova and M. R. Hamblin, Photodiagn. Photodyn. Ther., 2004, 1, 279–293 CrossRef CAS PubMed.
- L. Duprez, E. Wirawan, T. Vanden Berghe and P. Vandenabeele, Microbes Infect., 2009, 11, 1050–1062 CrossRef CAS PubMed.
- K. Peng, Y. Zheng, W. Xia and Z.-W. Mao, Chem. Soc. Rev., 2023, 52, 2790–2832 RSC.
- G. E. Villalpando-Rodriguez and S. B. Gibson, Oxid. Med. Cell. Longevity, 2021, 2021, 9912436 CrossRef PubMed.
- G. V. Putcha, C. A. Harris, K. L. Moulder, R. M. Easton, C. B. Thompson and E. M. Johnson Jr, J. Cell Biol., 2002, 157, 441–453 CrossRef CAS PubMed.
- N. Yan and Y. Shi, Annu. Rev. Cell Dev. Biol., 2005, 21, 35–56 CrossRef CAS PubMed.
- J. P. Sheridan, S. A. Marsters, R. M. Pitti, A. Gurney, M. Skubatch, D. Baldwin, L. Ramakrishnan, C. L. Gray, K. Baker, W. I. Wood, A. D. Goddard, P. Godowski and A. Ashkenazi, Science, 1997, 277, 818–821 CrossRef CAS PubMed.
- M. Kist and D. Vucic, EMBO J., 2021, 40, e106700 CrossRef CAS PubMed.
- A. Ashkenazi and V. M. Dixit, Science, 1998, 281, 1305–1308 CrossRef CAS PubMed.
- Q. Hu, D. Wu, W. Chen, Z. Yan, C. Yan, T. He, Q. Liang and Y. Shi, Proc. Natl. Acad. Sci. U. S. A., 2014, 111, 16254–16261 CrossRef CAS PubMed.
- S. J. Riedl and G. S. Salvesen, Nat. Rev. Mol. Cell Biol., 2007, 8, 405–413 CrossRef CAS PubMed.
- J. M. Schriewer, C. B. Peek, J. Bass and P. T. Schumacker, J. Am. Heart Assoc., 2013, 2, e000159 CrossRef PubMed.
- T. Zhong, J. Yu, Y. Pan, N. Zhang, Y. Qi and Y. Huang, Adv. Healthcare Mater., 2023, 12, 2300253 CrossRef CAS PubMed.
- Y. Wu, S. Li, Y. Chen, W. He and Z. Guo, Chem. Sci., 2022, 13, 5085–5106 RSC.
- Y. Wang, X. Shi, H. Fang, Z. Han, H. Yuan, Z. Zhu, L. Dong, Z. Guo and X. Wang, J. Med. Chem., 2022, 65, 7786–7798 CrossRef CAS PubMed.
- M. D. Hall and T. W. Hambley, Coord. Chem. Rev., 2002, 232, 49–67 CrossRef CAS.
- K. Suntharalingam, Y. Song and S. J. Lippard, Chem. Commun., 2014, 50, 2465–2468 RSC.
- R. Ma, Y. Wang, L. Yan, L. Ma, Z. Wang, H. C. Chan, S.-K. Chiu, X. Chen and G. Zhu, Chem. Commun., 2015, 51, 7859–7862 RSC.
- X. Wang, X. Wang, S. Jin, N. Muhammad and Z. Guo, Chem. Rev., 2018, 119, 1138–1192 CrossRef PubMed.
- F. E. Poynton, S. A. Bright, S. Blasco, D. C. Williams, J. M. Kelly and T. Gunnlaugsson, Chem. Soc. Rev., 2017, 46, 7706–7756 RSC.
- J. Liu, C. Zhang, T. W. Rees, L. Ke, L. Ji and H. Chao, Coord. Chem. Rev., 2018, 363, 17–28 CrossRef CAS.
- J. D. Knoll and C. Turro, Coord. Chem. Rev., 2015, 282, 110–126 CrossRef PubMed.
- L. Ma, L. Li and G. Zhu, Inorg. Chem. Front., 2022, 9, 2424–2453 RSC.
- J. Karges, T. Yempala, M. Tharaud, D. Gibson and G. Gasser, Angew. Chem., Int. Ed., 2020, 59, 7069–7075 CrossRef CAS PubMed.
- H. Shi, C. Imberti and P. J. Sadler, Inorg. Chem. Front., 2019, 6, 1623–1638 RSC.
- J. Huang, W. Ding, X. Zhu, B. Li, F. Zeng, K. Wu, X. Wu and F. Wang, Front. Chem., 2022, 10, 876410 CrossRef CAS PubMed.
- E. Shaili, L. Salassa, J. A. Woods, G. Clarkson, P. J. Sadler and N. J. Farrer, Chem. Sci., 2019, 10, 8610–8617 RSC.
- R. Czarnomysy, D. Radomska, A. Muszyńska, J. M. Hermanowicz, I. Prokop, A. Bielawska and K. Bielawski, Molecules, 2020, 25, 2860 CrossRef CAS PubMed.
- R. Navarro-Peñaloza, B. Landeros-Rivera, H. López-Sandoval, R. Castro-Ramírez and N. Barba-Behrens, Coord. Chem. Rev., 2023, 494, 215360 CrossRef.
- P. J. Jenks, Infect. Dis., 2010, 1413–1414 Search PubMed.
- H. Shi, G. J. Clarkson and P. J. Sadler, Inorg. Chem. Front., 2024, 11, 7898–7909 RSC.
- W. W.-Y. Yim and N. Mizushima, Cell Discovery, 2020, 6, 6 CrossRef CAS PubMed.
- N. Mizushima, B. Levine, A. M. Cuervo and D. J. Klionsky, Nature, 2008, 451, 1069–1075 CrossRef CAS PubMed.
- S. Fulda, A. M. Gorman, O. Hori and A. Samali, Int. J. Cell Biol., 2010, 2010, 214074 Search PubMed.
- B. Linder and D. Kögel, Biology, 2019, 8, 82 CrossRef CAS PubMed.
- A. M. Cuervo, Nat. Cell Biol., 2010, 12, 735–737 CrossRef CAS PubMed.
- J. Chicote, V. J. Yuste, J. Boix and J. Ribas, Front. Pharmacol., 2020, 11, 580343 CrossRef CAS PubMed.
- T. Lamark, V. Kirkin, I. Dikic and T. Johansen, Cell Cycle, 2009, 8, 1986–1990 CrossRef CAS PubMed.
- V. Kirkin and V. V. Rogov, Mol. Cell, 2019, 76, 268–285 CrossRef CAS PubMed.
- Q. Chen, J. Kang and C. Fu, Signal Transduction Targeted Ther., 2018, 3, 18 CrossRef PubMed.
- P. S. Felder, S. Keller and G. Gasser, Adv. Ther., 2020, 3, 1900139 CrossRef.
- J. Zhu, J. Á. Rodríguez-Corrales, R. Prussin, Z. Zhao, A. Dominijanni, S. L. Hopkins, B. S. J. Winkel, J. L. Robertson and K. J. Brewer, Chem. Commun., 2017, 53, 145–148 RSC.
- K. Sakai, H. Ozawa, H. Yamada, T. Tsubomura, M. Hara, A. Higuchi and M.-A. Haga, Dalton Trans., 2006, 3300–3305 RSC.
- A. Jain, J. Wang, E. R. Mashack, B. S. J. Winkel and K. J. Brewer, Inorg. Chem., 2009, 48, 9077–9084 CrossRef CAS PubMed.
- S. Singh, M. Varma, B. Shravage, P. Kulkarni and A. Kumbhar, J. Chem. Sci., 2021, 133, 89 CrossRef CAS.
- G. Das, B. V. Shravage and E. H. Baehrecke, Cold Spring Harbor Perspect. Biol., 2012, 4, a008813 Search PubMed.
- B. M. Gąsiorkiewicz, P. Koczurkiewicz-Adamczyk, K. Piska and E. Pękala, Invest. New Drugs, 2021, 39, 538–563 Search PubMed.
- J. Šima, Coord. Chem. Rev., 2006, 250, 2325–2334 Search PubMed.
- A. F. Westendorf, J. A. Woods, K. Korpis, N. J. Farrer, L. Salassa, K. Robinson, V. Appleyard, K. Murray, R. Grünert, A. M. Thompson, P. J. Sadler and P. J. Bednarski, Mol. Cancer Ther., 2012, 11, 1894–1904 CrossRef CAS PubMed.
- W. Zhang, P. Gou, J.-M. Dupret, C. Chomienne and F. Rodrigues-Lima, Transl. Oncol., 2021, 14, 101169 CrossRef CAS PubMed.
- S. Jamil, I. Lam, M. Majd, S.-H. Tsai and V. Duronio, Cancer Cell Int., 2015, 15, 79 CrossRef PubMed.
- S. J. Dixon, K. M. Lemberg, M. R. Lamprecht, R. Skouta, E. M. Zaitsev, C. E. Gleason, D. N. Patel, A. J. Bauer, A. M. Cantley, W. S. Yang, B. Morrison and B. R. Stockwell, Cell, 2012, 149, 1060–1072 CrossRef CAS PubMed.
- Y. Mou, J. Wang, J. Wu, D. He, C. Zhang, C. Duan and B. Li, J. Hematol. Oncol., 2019, 12, 34 CrossRef PubMed.
- W. S. Yang, K. J. Kim, M. M. Gaschler, M. Patel, M. S. Shchepinov and B. R. Stockwell, Proc. Natl. Acad. Sci. U. S. A., 2016, 113, E4966–E4975 CAS.
- R. Shah, M. S. Shchepinov and D. A. Pratt, ACS Cent. Sci., 2018, 4, 387–396 CrossRef CAS PubMed.
- B. Lu, X. B. Chen, M. D. Ying, Q. J. He, J. Cao and B. Yang, Front. Pharmacol., 2018, 8, 992 CrossRef PubMed.
- P. Maher, Ageing Res. Rev., 2005, 4, 288–314 CrossRef CAS PubMed.
- S. Li, H. Yuan, Y. Chen and Z. Guo, Fundam. Res., 2023, 3, 525–528 CrossRef CAS PubMed.
- Y. Qiu, Y. Cao, W. Cao, Y. Jia and N. Lu, Pharmacol. Res., 2020, 159, 104919 CrossRef CAS PubMed.
- Y. Yu, Y. Yan, F. Niu, Y. Wang, X. Chen, G. Su, Y. Liu, X. Zhao, L. Qian, P. Liu and Y. Xiong, Cell Death Discovery, 2021, 7, 193 CrossRef CAS PubMed.
- S. Doll, F. P. Freitas, R. Shah, M. Aldrovandi, M. C. da Silva, I. Ingold, A. Goya Grocin, T. N. X. da Silva, E. Panzilius, C. H. Scheel, A. Mourão, K. Buday, M. Sato, J. Wanninger, T. Vignane, V. Mohana, M. Rehberg, A. Flatley, A. Schepers, A. Kurz, D. White, M. Sauer, M. Sattler, E. W. Tate, W. Schmitz, A. Schulze, V. O’Donnell, B. Proneth, G. M. Popowicz, D. A. Pratt, J. P. F. Angeli and M. Conrad, Nature, 2019, 575, 693–698 CrossRef CAS PubMed.
- J. Garcia-Bermudez, L. Baudrier, E. C. Bayraktar, Y. Shen, K. La, R. Guarecuco, B. Yucel, D. Fiore, B. Tavora, E. Freinkman, S. H. Chan, C. Lewis, W. Min, G. Inghirami, D. M. Sabatini and K. Birsoy, Nature, 2019, 567, 118–122 CrossRef CAS PubMed.
- K. Hadian, Biochemistry, 2020, 59, 637–638 CrossRef CAS PubMed.
- Y. Lv, M. Wu, Z. Wang and J. Wang, Cell Biol. Toxicol., 2023, 39, 827–851 CrossRef PubMed.
- X. Jiang, B. R. Stockwell and M. Conrad, Nat. Rev. Mol. Cell Biol., 2021, 22, 266–282 CrossRef PubMed.
- Z. Xie, H. Hou, D. Luo, R. An, Y. Zhao and C. Qiu, Inflammation, 2021, 44, 35–47 CrossRef CAS PubMed.
- J. Li, F. Cao, H.-L. Yin, Z.-J. Huang, Z.-T. Lin, N. Mao, B. Sun and G. Wang, Cell Death Discovery, 2020, 11, 88 CrossRef PubMed.
- X. Zheng, M. Liu, Y. Wu, Y. Chen, W. He and Z. Guo, RSC Chem. Biol., 2024, 5, 141–147 RSC.
- A. Turksoy, D. Yildiz and E. U. Akkaya, Coord. Chem. Rev., 2019, 379, 47–64 CrossRef CAS.
- C. Li, J. Li, Y. Pang, L. Mei, W. Xu, Z. Zhang, C. Han and Y. Sun, Chem. Eng. J., 2024, 498, 155471 CrossRef CAS.
- D. Cui, J. Huang, X. Zhen, J. Li, Y. Jiang and K. Pu, Angew. Chem., Int. Ed., 2019, 58, 5920–5924 CrossRef CAS PubMed.
- Y. Wu, Y. Yin, S. Wang, H. Yu, Y. Ai, Y. Yuan and R. Hu, Dyes Pigm., 2025, 240, 112862 CrossRef CAS.
- M. Patra and G. Gasser, Nat. Rev. Chem., 2017, 1, 0066 CrossRef CAS.
- S. Peter and B. A. Aderibigbe, Molecules, 2019, 24, 3604 CrossRef CAS PubMed.
- H. Shi, F. Ponte, J. S. Grewal, G. J. Clarkson, C. Imberti, I. Hands-Portman, R. Dallmann, E. Sicilia and P. J. Sadler, Chem. Sci., 2024, 15, 4121–4134 RSC.
- H. Li, T. Yang, J. Zhang, K. Xue, X. Ma, B. Yu and X. Jin, Cell Death Discovery, 2024, 10, 32 CrossRef PubMed.
- A. Zychlinsky, M. C. Prevost and P. J. Sansonetti, Nature, 1992, 358, 167–169 CrossRef CAS PubMed.
- R. Guo, H. Wang and N. Cui, Mediators Inflammation, 2021, 2021, 9925059 CrossRef PubMed.
- Y. Fang, S. Tian, Y. Pan, W. Li, Q. Wang, Y. Tang, T. Yu, X. Wu, Y. Shi, P. Ma and Y. Shu, Biomed. Pharmacother., 2020, 121, 109595 CrossRef CAS PubMed.
- P. Yu, X. Zhang, N. Liu, L. Tang, C. Peng and X. Chen, Signal Transduction Targeted Ther., 2021, 6, 128 CrossRef PubMed.
- Y. Zheng, K. Peng, Q. Cao and Z. W. Mao, Angew. Chem., Int. Ed., 2025, 64, e202507186 CrossRef CAS PubMed.
- Q. Wang, Y. Wang, J. Ding, C. Wang, X. Zhou, W. Gao, H. Huang, F. Shao and Z. Liu, Nature, 2020, 579, 421–426 CrossRef CAS PubMed.
- S. S. Wright, P. Kumari, V. Fraile-Ágreda, C. Wang, S. Shivcharan, S. Kappelhoff, E. G. Margheritis, A. Matz, S. O. Vasudevan, I. Rubio, M. Bauer, B. Zhou, S. K. Vanaja, K. Cosentino, J. Ruan and V. A. Rathinam, Cell, 2025, 188, 280–291 CrossRef CAS PubMed.
- J. Liu, J. Zhou, Y. Luan, X. Li, X. Meng, W. Liao, J. Tang and Z. Wang, Cell Commun. Signaling, 2024, 22, 22 CrossRef CAS PubMed.
- W. Zhang, G. Li, R. Luo, J. Lei, Y. Song, B. Wang, L. Ma, Z. Liao, W. Ke, H. Liu, W. Hua, K. Zhao, X. Feng, X. Wu, Y. Zhang, K. Wang and C. Yang, Exp. Mol. Med., 2022, 54, 129–142 CrossRef CAS PubMed.
- X. Hu, H. Zhang, Q. Zhang, X. Yao, W. Ni and K. Zhou, J. Neuroinflammation, 2022, 19, 242 CrossRef CAS PubMed.
- L. Y. Liu, H. Fang, Q. Chen, M. H.-Y. Chan, M. Ng, K.-N. Wang, W. Liu, Z. Tian, J. Diao, Z. W. Mao and V. W.-W. Yam, Angew. Chem., Int. Ed., 2020, 59, 19229–19236 CrossRef CAS PubMed.
- Y. F. Zhong, H. Zhang, W. T. Liu, X. H. Zheng, Y. W. Zhou, Q. Cao, Y. Shen, Y. Zhao, P. Z. Qin, L.-N. Ji and Z. W. Mao, Chem. – Eur. J., 2017, 23, 16442–16446 CrossRef CAS PubMed.
- Y. Y. Ling, X. Y. Xia, L. Hao, W. J. Wang, H. Zhang, L. Y. Liu, W. Liu, Z. Y. Li, C. P. Tan and Z. W. Mao, Angew. Chem., Int. Ed., 2022, 61, e202210988 CrossRef CAS PubMed.
- J. J. Wilson and S. J. Lippard, Chem. Rev., 2014, 114, 4470–4495 CrossRef CAS PubMed.
- H. C. Tai, Y. Zhao, N. J. Farrer, A. E. Anastasi, G. Clarkson, P. J. Sadler and R. J. Deeth, Chem. – Eur. J., 2012, 18, 10630–10642 CrossRef CAS PubMed.
- F. Juliá, ChemCatChem, 2022, 14, e202200916 CrossRef.
- Q. Zhou, N.-H. Chu, J. Xu, K.-C. Law and G. Zhu, Inorg. Chem. Front., 2026 10.1039/D6QI00078A.
- N. Casares, M. O. Pequignot, A. Tesniere, F. Ghiringhelli, S. Roux, N. Chaput, E. Schmitt, A. Hamai, S. Hervas-Stubbs, M. Obeid, F. Coutant, D. Métivier, E. Pichard, P. Aucouturier, G. Pierron, C. Garrido, L. Zitvogel and G. Kroemer, J. Exp. Med., 2005, 202, 1691–1701 CrossRef CAS PubMed.
- N. Yatim, S. Cullen and M. L. Albert, Nat. Rev. Immunol., 2017, 17, 262–275 CrossRef CAS PubMed.
- L. Galluzzi, I. Vitale, S. Warren, S. Adjemian, P. Agostinis, A. B. Martinez, T. A. Chan, G. Coukos, S. Demaria, E. Deutsch, D. Draganov, R. L. Edelson, S. C. Formenti, J. Fucikova, L. Gabriele, U. S. Gaipl, S. R. Gameiro, A. D. Garg, E. Golden, J. Han, K. J. Harrington, A. Hemminki, J. W. Hodge, D. M. S. Hossain, T. Illidge, M. Karin, H. L. Kaufman, O. Kepp, G. Kroemer, J. J. Lasarte, S. Loi, M. T. Lotze, G. Manic, T. Merghoub, A. A. Melcher, K. L. Mossman, F. Prosper, Ø. Rekdal, M. Rescigno, C. Riganti, A. Sistigu, M. J. Smyth, R. Spisek, J. Stagg, B. E. Strauss, D. Tang, K. Tatsuno, S. W. van Gool, P. Vandenabeele, T. Yamazaki, D. Zamarin, L. Zitvogel, A. Cesano and F. M. Marincola, J. Immunother. Cancer, 2020, 8, e000337 CrossRef PubMed.
- D. V. Krysko, A. D. Garg, A. Kaczmarek, O. Krysko, P. Agostinis and P. Vandenabeele, Nat. Rev. Cancer, 2012, 12, 860–875 CrossRef CAS PubMed.
- J. X. Zou, M. R. Chang, N. A. Kuznetsov, J. X. Kee, M. V. Babak and W. H. Ang, Chem. Sci., 2025, 16, 6160–6187 RSC.
- Y. Zhang, B.-T. Doan and G. Gasser, Chem. Rev., 2023, 123, 10135–10155 CrossRef CAS PubMed.
- I. Martins, Y. Wang, M. Michaud, Y. Ma, A. Q. Sukkurwala, S. Shen, O. Kepp, D. Métivier, L. Galluzzi, J.-L. Perfettini, L. Zitvogel and G. Kroemer, Cell Death Differ., 2014, 21, 79–91 CrossRef CAS PubMed.
- G. Kroemer, L. Galluzzi, O. Kepp and L. Zitvogel, Annu. Rev. Immunol., 2013, 31, 51–72 CrossRef CAS PubMed.
- L. Zhang, N. Montesdeoca, J. Karges and H. Xiao, Angew. Chem., Int. Ed., 2023, 62, e202300662 CrossRef CAS PubMed.
- A. Tesniere, F. Schlemmer, V. Boige, O. Kepp, I. Martins, F. Ghiringhelli, L. Aymeric, M. Michaud, L. Apetoh, L. Barault, J. Mendiboure, J.-P. Pignon, V. Jooste, P. van Endert, M. Ducreux, L. Zitvogel, F. Piard and G. Kroemer, Oncogene, 2010, 29, 482–491 CrossRef CAS PubMed.
- I. Martins, O. Kepp, F. Schlemmer, S. Adjemian, M. Tailler, S. Shen, M. Michaud, L. Menger, A. Gdoura, N. Tajeddine, A. Tesniere, L. Zitvogel and G. Kroemer, Oncogene, 2011, 30, 1147–1158 CrossRef CAS PubMed.
- V. Novohradsky, J. Pracharova, J. Kasparkova, C. Imberti, H. E. Bridgewater, P. J. Sadler and V. Brabec, Inorg. Chem. Front., 2020, 7, 4150–4159 RSC.
- Z. Deng, N. Wang, Y. Liu, Z. Xu, Z. Wang, T.-C. Lau and G. Zhu, J. Am. Chem. Soc., 2020, 142, 7803–7812 CrossRef CAS PubMed.
- D. Dersh, J. Hollý and J. W. Yewdell, Nat. Rev. Immunol., 2021, 21, 116–128 CrossRef CAS PubMed.
- X. Chen, Q. Lu, H. Zhou, J. Liu, B. Nadorp, A. Lasry, Z. Sun, B. Lai, G. Rona, J. Zhang, M. Cammer, K. Wang, W. Al-Santli, Z. Ciantra, Q. Guo, J. You, D. Sengupta, A. Boukhris, H. Zhang, C. Liu, P. Cresswell, P. L. M. Dahia, M. Pagano, I. Aifantis and J. Wang, Cell, 2023, 186, 3903–3920 CrossRef CAS PubMed.
- L.-Y. Liu, J. Li, P. Sun, W. Yu, T.-Z. Ma, Y.-L. Zeng, Z. Zhao, Q. Wei, Y. Liu, J. P. Li, Z.-W. Mao and Z. Guo, J. Med. Chem., 2026, 69, 4629–4641 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |