Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Electrochemical nitrogen atom insertion enabled by a manganese complex†
Minki Jeon,
Pooja Kumari Jat,
Juyoung Baek,
Geon Kang ,
Jaejeong Kim and
Isaac Choi
,
Jaejeong Kim and
Isaac Choi *
*
Department of Chemistry, Chungbuk National University, Chungcheongbuk-do 28644, Republic of Korea. E-mail: isaac.choi@chungbuk.ac.kr
First published on 26th March 2026
Abstract
Herein, we report a manganaelectrooxidative nitrogen atom insertion, supported by mechanistic studies, exhibiting broad functional group tolerance.
In recent years, skeletal editing has emerged as a groundbreaking platform in molecular engineering,1 and significant progress has been clearly witnessed.2 Indeed, this innovative synthetic toolbox enables chemists to achieve a highly streamlined synthetic process when late-stage reconstruction of molecular frameworks is required.3 Within the realm of skeletal editing, nitrogen insertion has been generally realized by the oxidation-induced C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond cleavage. Following key early examples in the 20th century,4 recent contributions by Morandi,5 Levin,6 and Leonori,7 among others,8 have established the modular assembly of nitrogen-containing heterocycles (Fig. 1A). Transition metal catalysts have also shown promise for nitrogen insertion, though reports on such reactions remain sparse.9 However, these reactions typically rely on suprastoichiometric amounts of terminal oxidants or pre-oxidized precursors under relatively harsh conditions.
C bond cleavage. Following key early examples in the 20th century,4 recent contributions by Morandi,5 Levin,6 and Leonori,7 among others,8 have established the modular assembly of nitrogen-containing heterocycles (Fig. 1A). Transition metal catalysts have also shown promise for nitrogen insertion, though reports on such reactions remain sparse.9 However, these reactions typically rely on suprastoichiometric amounts of terminal oxidants or pre-oxidized precursors under relatively harsh conditions.
Recently, there has been growing interest in electrochemical organic synthesis, which provides chemists with an environmentally benign method for constructing molecules.10 In this synthetic strategy, an applied electrical potential drives electron flow, specifically enabling oxidation processes at electrode surfaces. Thus, electricity facilitates oxidative chemical transformations through anodic events, thereby avoiding the need for expensive or toxic chemical oxidants. In this context, Cheng,11 Ackermann,12 and Koh13 independently reported nitrogen atom insertion reactions under electrochemical settings (Fig. 1B). Indeed, the strategic selection of readily accessible nitrogen sources, such as ammonia or ammonium carbamate, significantly enhances the efficiency and practicality of the reaction manifold, while the use of external chemical oxidants is rendered unnecessary.
Based on these precedent studies, we envisaged that manganese could be an ideal candidate for a sustainable electrochemical nitrogen insertion reaction with its low toxicity and abundance. Importantly, in recent reports of the Lin group, manganese(II) catalysts play a pivotal role in electrochemical diazidation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds.14 Inspired by these studies, we hypothesized that manganese could serve as an effective platform to control reactivity through ligand modification, thereby enabling nitrogen-atom insertion in place of the well-established diazidation process in the presence of azides under electrochemically oxidative conditions (Fig. 1C).
C bonds.14 Inspired by these studies, we hypothesized that manganese could serve as an effective platform to control reactivity through ligand modification, thereby enabling nitrogen-atom insertion in place of the well-established diazidation process in the presence of azides under electrochemically oxidative conditions (Fig. 1C).
As a result, we have established a novel electrochemical nitrogen-atom insertion reaction using a manganese complex. Salient features of our strategy include (a) detailed screening of well-defined manganese complexes, (b) preliminary mechanistic investigations to understand the modus operandi of present work, and (c) a wide range of functional group tolerance.
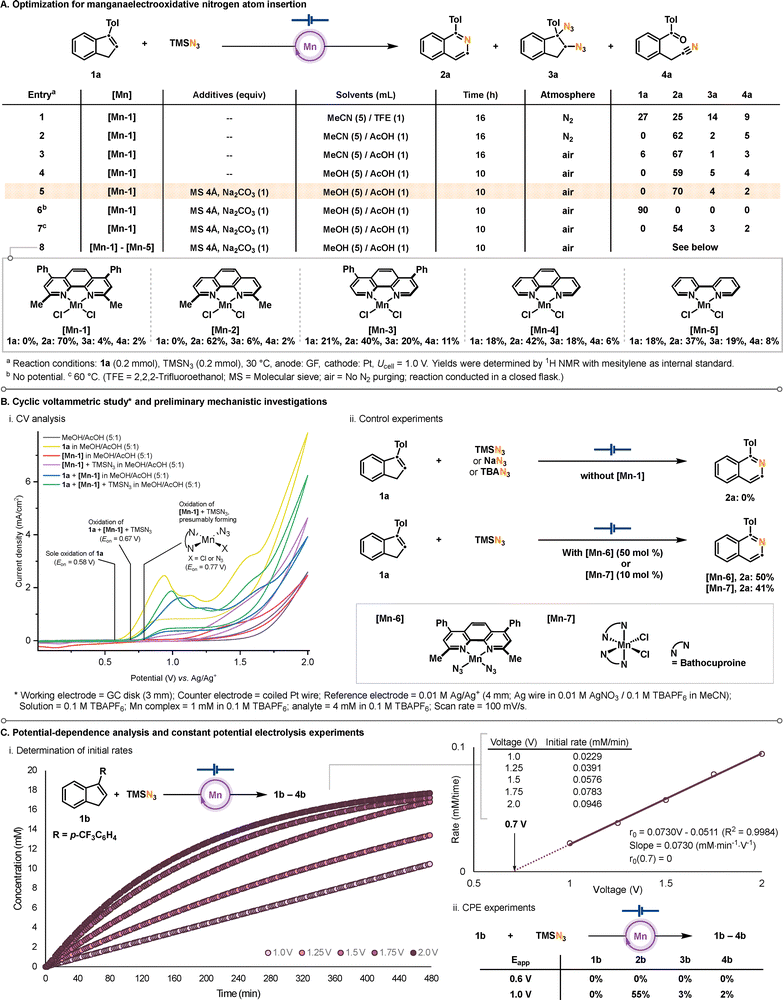
We set out to explore optimal reaction conditions for the electrochemical nitrogen insertion into indene 1a with TMSN3 as a nitrogen source in the presence of a manganese complex in an undivided cell setup (Scheme 1A and Table S1–S5). Initial experiments (entries 1 and 2) indicated that while diazidation indeed occurred,14b isoquinoline 2a was afforded as a major product. To enhance operational simplicity, the reaction was conducted under air, which resulted in a slight improvement in the product yield (entry 3). However, under MeCN/AcOH solvent conditions, the cell resistance was measured to be high. Although LiClO4 was employed as the electrolyte to alleviate this issue, severe passivation of the electrode surface was observed (Table S1). Solvent screening revealed that replacing MeCN with MeOH led to predominant formation of 2a with low resistance, although diazidation byproduct 3a and nitrile derivative 4a slightly increased (entry 4). Interestingly, the addition of molecular sieves and a base improved the yield while suppressing byproducts (entry 5).
Control experiments confirmed the essential role of electrooxidation process and the need for milder conditions (entries 6–7). These results indirectly suggest that under air atmosphere, molecular oxygen exerts a greater influence on downstream chemical processes in the reaction manifold rather than serving as a terminal oxidant for Mn(II) species.15 Finally, a range of manganese complexes ([Mn-1]–[Mn-5]) was evaluated (entry 8, Scheme 1A and Scheme S2), and the bathocuproine ligand exhibited the highest catalytic efficiency, affording 2a in 70% yield (entry 5, Scheme 1).
Based on the observed reactivity for nitrogen-atom insertion, we sought to elucidate its mechanistic features. To this end, a potentiodynamic electrochemical approach was employed (Scheme 1Bi and Scheme S3–S13). In cyclic voltammetry, the anodic wave of 1a near 1.0 V shifts to more positive potentials in the presence of manganese with a change in peak shape, indicating that manganese influences the electrochemical oxidation of 1a. Furthermore, in the potential region above approximately 1.25 V, the anodic current of green trace decreases relative to that observed for 1a alone, suggesting that manganese species may alter the oxidation pathway of 1a. Additional CV studies with increasing the concentration of TMSN3 do not lead to a meaningful change in current density (Scheme S14–S17), indicating that direct capture of the electrochemically generated radical cation of 1a by azide anion is unlikely to be operative. Consistently, no product formation is observed in the absence of [Mn-1] (Scheme 1Bii). Furthermore, when [Mn-6] and [Mn-7] were subjected to the electrochemical reaction conditions (Scheme 1Bii), product formation was observed, albeit with slightly reduced efficiency. Additionally, CV studies of the [Mn-6] clearly demonstrate that the in situ generated Mn(II)–N3 species is involved in the catalytic system under the electrooxidative conditions (Scheme S18).
To gain more detailed insight, the reaction with 1b was profiled under a series of applied cell voltages ranging from 1.0 to 2.0 V (Scheme 1Ci). The initial rate shows a linear dependence on the applied potential, implying that catalytic turnover is sensitive to the electrochemical driving force. Also, extrapolation of the rate–potential relationship toward lower potentials suggests productive electrocatalysis ceases at approximately 0.7 V. Consistent with this analysis, constant-potential electrolysis (CPE) at 0.6 V produced no detectable product while the starting material was also fully consumed, likely due to unproductive direct oxidation (Scheme 1Cii). In contrast, electrolysis at 1.0 V afforded the desired products. These results are consistent with CV studies shown in Scheme 1Bi (green trace), in which onset potential of the reaction mixture is observed at approximately 0.67 V, highlighting the crucial role of manganese catalyst in enabling the electrochemical nitrogen atom insertion.
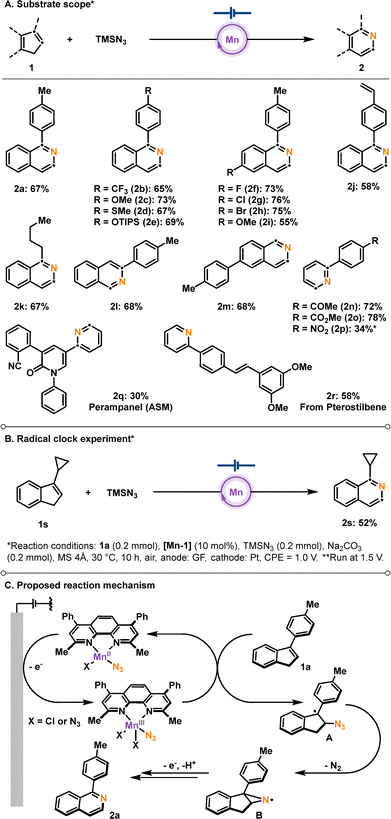
With a mechanistic understanding in hand, the scope of the manganese-catalyzed electrochemical nitrogen-atom insertion was explored (Scheme 2A). A series of differently substituted 1-arylisoquinolines 2 was afforded, revealing that not only electron-withdrawing substituents but also electron donating groups were fully tolerated under the optimized conditions. Importantly, in the case of a substrate bearing a styrene moiety, a desired product 2j was solely formed. The C3-substituents on indene is not required (2l–2m), and the method was also successfully applied to the synthesis of pyridines (2n–2p). Furthermore, the developed protocol could be extended to the synthesis of a pharmaceutical compound, Perampanel (2q), and a derivative of a natural product, Pterostilbene (2r). Interestingly, a reaction with a radical probe 1s resulted in the formation of nitrogen-inserted product 2s, while a ring opened product was not detected (Scheme 2B).15,16
On the basis of our mechanistic insights, a plausible pathway for manganaelectrooxidative nitrogen atom insertion is proposed (Scheme 2C). Initially, the in situ generated Mn(II)–N3 complex undergoes anodic oxidation, thereby affording an azide radical. The resulting azide radical reacts with substrate 1a to generate intermediate A, which subsequently undergoes annulation to form the aziridine radical intermediate B.9c This species then undergoes intramolecular rearrangement followed by oxidative aromatization to furnish the final product 2a.
In conclusion, we have established a manganese-catalyzed electrocatalytic strategy for nitrogen incorporation into unsaturated frameworks that proceeds under mild conditions without the use of external oxidants. Mechanistic experiments indicate the participation of manganese–azide intermediates which is competent to generate azide radical. The reaction displays broad substrate compatibility and accommodates diverse functional groups, thereby offering a practical and environmentally benign approach for constructing nitrogen-containing molecular architectures.
MJ: investigation, formal analysis, data curation, visualization, writing – original draft; PKJ: investigation; JB: investigation; GK: investigation; JK: investigation; IC: conceptualization, supervision, project administration, funding acquisition, writing – review & editing.
I. C. and the Choi group thank MSIT, MOE, NRF, and CBNU. This work was supported by the National Research Foundation of Korea (NRF) grant, funded by the Korea government (MSIT) (No. RS-2023-00210101) and the Global Learning & Academic Research Institution for Master's·PhD Students, and Postdocs (G-LAMP) Program of the NRF grant funded by the Ministry of Education (No. RS-2024-00445180). This work was supported by a funding for the academic research program of Chungbuk National University in 2025.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information includes experimental procedures, details of kinetic studies, and analytical data. See DOI: https://doi.org/10.1039/d6cc00221h.References
- (a) M. D. Levin, Synlett, 2024, 35, 1471–1474 CrossRef CAS PubMed; (b) J. Jurczyk, J. Woo, S. F. Kim, B. D. Dherange, R. Sarpong and M. D. Levin, Nat. Synth., 2022, 1, 352–364 CrossRef CAS PubMed; (c) B. Iddon, O. Meth-Cohn, E. F. V. Scriven, H. Suschitzky and P. T. Gallagher, Angew. Chem., Int. Ed. Engl., 1979, 18, 900–917 Search PubMed.
- (a) F.-P. Wu, C. C. Chintawar, R. Lalisse, P. Mukherjee, S. Dutta, J. Tyler, C. G. Daniliuc, O. Gutierrez and F. Glorius, Nat. Catal., 2024, 7, 242–251 CrossRef CAS PubMed; (b) J. Masson-Makdissi, R. F. Lalisse, M. Yuan, B. D. Dherange, O. Gutierrez and M. D. Levin, J. Am. Chem. Soc., 2024, 146, 17719–17727 CrossRef CAS PubMed; (c) S. F. Kim, H. Schwarz, J. Jurczyk, B. R. Nebgen, H. Hendricks, H. Park, A. Radosevich, M. W. Zuerch, K. Harper, M. C. Lux, C. S. Yeung and R. Sarpong, J. Am. Chem. Soc., 2024, 146, 5580–5596 CrossRef CAS PubMed; (d) G. L. Bartholomew, S. L. Kraus, L. J. Karas, F. Carpaneto, R. Bennett, M. S. Sigman, C. S. Yeung and R. Sarpong, J. Am. Chem. Soc., 2024, 146, 2950–2958 CrossRef CAS PubMed; (e) B. A. Wright, A. Matviitsuk, M. J. Black, P. García-Reynaga, L. E. Hanna, A. T. Herrmann, M. K. Ameriks, R. Sarpong and T. P. Lebold, J. Am. Chem. Soc., 2023, 145, 10960–10966 CrossRef CAS PubMed; (f) J. Woo, C. Stein, A. H. Christian and M. D. Levin, Nature, 2023, 623, 77–82 CrossRef CAS PubMed; (g) B. D. Dherange, M. Yuan, C. B. Kelly, C. A. Reiher, C. Grosanu, K. J. Berger, O. Gutierrez and M. D. Levin, J. Am. Chem. Soc., 2023, 145, 17–24 Search PubMed; (h) J. Woo, A. H. Christian, S. A. Burgess, Y. Jiang, U. F. Mansoor and M. D. Levin, Science, 2022, 376, 527–532 CrossRef CAS PubMed; (i) E. E. Hyland, P. Q. Kelly, A. M. McKillop, B. D. Dherange and M. D. Levin, J. Am. Chem. Soc., 2022, 144, 19258–19264 CrossRef CAS PubMed; (j) G. L. Bartholomew, F. Carpaneto and R. Sarpong, J. Am. Chem. Soc., 2022, 144, 22309–22315 CrossRef CAS PubMed; (k) S. H. Kennedy, B. D. Dherange, K. J. Berger and M. D. Levin, Nature, 2021, 593, 223–227 CrossRef CAS PubMed; (l) J. Jurczyk, M. C. Lux, D. Adpressa, S. F. Kim, Y.-H. Lam, C. S. Yeung and R. Sarpong, Science, 2021, 373, 1004–1012 CrossRef CAS PubMed; (m) B. D. Dherange, P. Q. Kelly, J. P. Liles, M. S. Sigman and M. D. Levin, J. Am. Chem. Soc., 2021, 143, 11337–11344 Search PubMed; (n) K. J. Berger, J. L. Driscoll, M. Yuan, B. D. Dherange, O. Gutierrez and M. D. Levin, J. Am. Chem. Soc., 2021, 143, 17366–17373 CrossRef CAS PubMed.
- (a) S. Wiesler, G. Sennari, M. V. Popescu, K. E. Gardner, K. Aida, R. S. Paton and R. Sarpong, Nat. Commun., 2024, 15, 4125 CrossRef CAS PubMed; (b) S. Holovach, K. P. Melnykov, I. Poroshyn, R. T. Iminov, D. Dudenko, I. Kondratov, M. Levin and O. O. Grygorenko, Chem. – Eur. J., 2023, 29, e202203470 CrossRef CAS PubMed; (c) T. Hosoya, T. Hiramatsu, T. Ikemoto, M. Nakanishi, H. Aoyama, A. Hosoya, T. Iwata, K. Maruyama, M. Endo and M. Suzuki, Org. Biomol. Chem., 2004, 2, 637–641 Search PubMed.
- (a) A. Albini, G. Bettinetti and G. Minoli, J. Am. Chem. Soc., 1997, 119, 7308–7315 CrossRef CAS; (b) K. Narasimhan and P. R. Kumar, Heterocycles, 1984, 22, 1369–1375 CrossRef CAS; (c) D. S. Dime and S. McLean, J. Org. Chem., 1981, 46, 4999–5000 CrossRef CAS; (d) R. B. Miller and J. M. Frincke, J. Org. Chem., 1980, 45, 5312–5315 Search PubMed; (e) M. Ikeda, F. Tabusa, Y. Nishimura, S. Kwon and Y. Tamura, Tetrahedron Lett., 1976, 17, 2347–2350 CrossRef; (f) M. I. Fremery and E. K. Fields, J. Org. Chem., 1964, 29, 2240–2243 CrossRef CAS.
- (a) A.-S. K. Paschke, E. J. Meeus, M. A. Masota, F. Hoffmann, N. M. Grob and B. Morandi, J. Am. Chem. Soc., 2026, 148, 55–60 CrossRef CAS PubMed; (b) J. C. Reisenbauer, A.-S. K. Paschke, J. Krizic, B. B. Botlik, P. Finkelstein and B. Morandi, Org. Lett., 2023, 25, 8419–8423 CrossRef CAS PubMed; (c) P. Finkelstein, J. C. Reisenbauer, B. B. Botlik, O. Green, A. Florin and B. Morandi, Chem. Sci., 2023, 14, 2954–2959 RSC; (d) J. C. Reisenbauer, O. Green, A. Franchino, P. Finkelstein and B. Morandi, Science, 2022, 377, 1104–1109 CrossRef CAS PubMed.
- (a) T. J. Pearson, R. Shimazumi, J. L. Driscoll, B. D. Dherange, D.-I. Park and M. D. Levin, Science, 2023, 381, 1474–1479 CrossRef CAS PubMed; (b) P. Q. Kelly, A. S. Filatov and M. D. Levin, Angew. Chem., Int. Ed., 2022, 61, e202213041 CrossRef CAS PubMed.
- (a) R. Mykura, R. Sánchez-Bento, E. Matador, V. K. Duong, A. Varela, L. Angelini, R. J. Carbajo, J. Llaveria, A. Ruffoni and D. Leonori, Nat. Chem., 2024, 16, 771–779 Search PubMed; (b) R. Sánchez-Bento, B. Roure, J. Llaveria, A. Ruffoni and D. Leonori, Chemistry, 2023, 9, 3685–3695 CrossRef; (c) E. Matador, M. J. Tilby, I. Saridakis, M. Pedrón, D. Tomczak, J. Llaveria, I. Atodiresei, P. Merino, A. Ruffoni and D. Leonori, J. Am. Chem. Soc., 2023, 145, 27810–27820 CrossRef CAS PubMed; (d) A. Ruffoni, C. Hampton, M. Simonetti and D. Leonori, Nature, 2022, 610, 81–86 CrossRef CAS PubMed.
- (a) X.-X. Zhang, S.-T. Xu, X.-T. Li, T.-T. Song, D.-W. Ji and Q.-A. Chen, J. Am. Chem. Soc., 2025, 147, 11533–11542 CrossRef CAS PubMed; (b) E. Z. Schroeder, C. Lin, Y. Hu, Z.-Y. Dai, A. F. Griffin, T. S. Hotvedt, I. A. Guzei and J. M. Schomaker, J. Am. Chem. Soc., 2024, 146, 22085–22092 CrossRef PubMed; (c) A. Lin, A. Ghosh, S. Yellen, Z. T. Ball and L. Kürti, J. Am. Chem. Soc., 2024, 146, 21129–21136 CrossRef CAS PubMed; (d) C. Amber, L. T. Göttemann, R. T. Steele, T. M. Petitjean and R. Sarpong, J. Org. Chem., 2024, 89, 17655–17663 CrossRef CAS PubMed; (e) G. Li, M. N. Lavagnino, S. Z. Ali, S. Hu and A. T. Radosevich, J. Am. Chem. Soc., 2023, 145, 41–46 CrossRef CAS PubMed; (f) S. C. Patel and N. Z. Burns, J. Am. Chem. Soc., 2022, 144, 17797–17802 CrossRef CAS PubMed; (g) F. R. Bou-Hamdan, F. Lévesque, A. G. O'Brien and P. H. Seeberger, Beilstein J. Org. Chem., 2011, 7, 1124–1129 CrossRef CAS PubMed.
- (a) Z. Wang, H. Xu, X. Han, S. Fan and J. Zhu, Org. Lett., 2024, 26, 8559–8564 CrossRef CAS PubMed; (b) T. Heilmann, J. M. Lopez-Soria, J. Ulbrich, J. Kircher, Z. Li, B. Worbs, C. Golz, R. A. Mata and M. Alcarazo, Angew. Chem., Int. Ed., 2024, 63, e202403826 Search PubMed; (c) J. Wang, H. Lu, Y. He, C. Jing and H. Wei, J. Am. Chem. Soc., 2022, 144, 22433–22439 CrossRef CAS PubMed; (d) M.-M. Xu, W.-B. Cao, X.-P. Xu and S.-J. Ji, Chem. Commun., 2018, 54, 12602–12605 RSC.
- (a) N. E. S. Tay, D. Lehnherr and T. Rovis, Chem. Rev., 2022, 122, 2487–2649 CrossRef CAS PubMed; (b) K.-J. Jiao, Z.-H. Wang, C. Ma, H.-L. Liu, B. Cheng and T.-S. Mei, Chem Catal, 2022, 2, 3019–3047 CAS; (c) H. Huang, K. A. Steiniger and T. H. Lambert, J. Am. Chem. Soc., 2022, 144, 12567–12583 CrossRef CAS PubMed; (d) L. F. T. Novaes, J. Liu, Y. Shen, L. Lu, J. M. Meinhardt and S. Lin, Chem. Soc. Rev., 2021, 50, 7941–8002 RSC; (e) K. Yamamoto, M. Kuriyama and O. Onomura, Acc. Chem. Res., 2020, 53, 105–120 CrossRef CAS PubMed; (f) C. Schotten, T. P. Nicholls, R. A. Bourne, N. Kapur, B. N. Nguyen and C. E. Willans, Green Chem., 2020, 22, 3358–3375 RSC; (g) C. Kingston, M. D. Palkowitz, Y. Takahira, J. C. Vantourout, B. K. Peters, Y. Kawamata and P. S. Baran, Acc. Chem. Res., 2020, 53, 72–83 CrossRef CAS PubMed; (h) J. E. Kim, S. Choi, M. Balamurugan, J. H. Jang and K. T. Nam, Trends Chem., 2020, 2, 1004–1019 Search PubMed; (i) Q. Jing and K. D. Moeller, Acc. Chem. Res., 2020, 53, 135–143 CrossRef CAS PubMed; (j) G. Hilt, ChemElectroChem, 2020, 7, 395–405 CrossRef CAS; (k) T. Fuchigami and S. Inagi, Acc. Chem. Res., 2020, 53, 322–334 CrossRef CAS PubMed; (l) M. Yan, Y. Kawamata and P. S. Baran, Angew. Chem., Int. Ed., 2018, 57, 4149–4155 CrossRef CAS PubMed; (m) N. Sauermann, T. H. Meyer, Y. Qiu and L. Ackermann, ACS Catal., 2018, 8, 7086–7103 CrossRef CAS; (n) J.-i Yoshida, K. Kataoka, R. Horcajada and A. Nagaki, Chem. Rev., 2008, 108, 2265–2299 CrossRef CAS PubMed.
- S. Liu and X. Cheng, Nat. Commun., 2022, 13, 425 CrossRef CAS PubMed.
- B. S. Zhang, S. L. Homolle, T. Bauch, J. C. A. Oliveira, S. Warratz, B. Yuan, X. Y. Gou and L. Ackermann, Angew. Chem., Int. Ed., 2024, 63, e202407384 CrossRef CAS PubMed.
- G.-Q. Sun, X. Wang, R. Hu, W. Rao, Y. Zhao and M. J. Koh, Nat. Synth., 2026, 5, 466–475 Search PubMed.
- (a) L. F. T. Novaes, Y. Wang, J. Liu, X. Riart-Ferrer, W.-C. Cindy Lee, N. Fu, J. S. K. Ho, X. P. Zhang and S. Lin, ACS Catal., 2022, 12, 14106–14112 CrossRef CAS; (b) N. Fu, G. S. Sauer, A. Saha, A. Loo and S. Lin, Science, 2017, 357, 575–579 CrossRef CAS PubMed.
- X. Sun, X. Li, S. Song, Y. Zhu, Y.-F. Liang and N. Jiao, J. Am. Chem. Soc., 2015, 137, 6059–6066 Search PubMed.
- W. Yang, Y. Lan, Y. Bai, Z. Zhao, Y. Song, R. Chang, X. Shang, S. Li, S. Jia, S. Liu, S.-J. Li and L. Niu, Chemistry, 2025, 12, 102702 Search PubMed.
Footnote |
| † Dedicated to Professor Chulbom Lee on the occasion of his 60th birthday. |
| This journal is © The Royal Society of Chemistry 2026 |