Open Access Article
Open Access ArticleLight-driven silane functionalization using disulfide and dichloromethane
Jiin
Lee†
ab,
Hyunji
Kim†
a and
Sunggi
Lee
 *ab
*ab
aDepartment of Physics and Chemistry, DGIST, Daegu 42988, Republic of Korea. E-mail: sunggi.lee@dgist.ac.kr
bCenter for Basic Science, DGIST, Daegu 42988, Republic of Korea
First published on 27th February 2026
Abstract
Organosilicon compounds are of great importance in material and synthetic chemistry. Herein, we disclose a practical and general silane functionalization protocol that enables direct access to silanols and silyl ethers. The process features mild, environmentally benign conditions and enables the transformation of diverse silanes into valuable organosilicon compounds.
Organosilicon compounds have been broadly utilized as valuable building blocks in material science1 and organic synthesis (Scheme 1).2 Among them, silanols and silyl ethers find wide applications in silicon-based materials, metallasiloxane synthesis,3 C–H activation,4 organocatalysis,5 and medicinal chemistry.6 Motivated by the applications of organosilicon compounds, research has been actively conducted in the field of organic synthesis to develop methods for synthesizing these compounds. These are typically achieved through the hydrolysis of silyl chlorides7 or the oxidation of silanes with stoichiometric oxidants such as permanganate, osmium tetroxide, dioxirane, and ozone.8 However, the hydrolysis of silyl chlorides often suffers from low efficiency owing to the condensation of silanols to siloxanes, while oxidation of silanes with theses reagents is limited by narrow substrate scope, poor functional group tolerance, and the generation of substantial waste. To address issues, transition-metal-catalyzed9 or photocatalytic10 methods have emerged as an attractive approach. However, most reported methods rely on noble metal catalysts and often suffer from the undesired formation of disiloxanes, arising from competitive reactions of silanols with hydrosilanes (Fig. 1A). In 2019, Wu and co-workers disclosed a photocatalytic method employing eosin Y, where hydrosilanes undergo chlorination and subsequent reaction with alcohols to afford the corresponding silyl ethers (Fig. 1B).11 Although significant progress has been made, practical and robust methods for accessing silanols and silyl ethers still limited. We hypothesized that the in situ generation of silyl chlorides from silanes under mild conditions would provide a versatile precursor, enabling their subsequent conversion into silanols and silyl ethers through reaction with oxygen nucleophiles.
 | ||
| Fig. 1 Previous works and this work. (A) Transition-metal catalyzed oxidation of silanes. (B) Wu's Eosin Y-catalyzed access to silyl ether. (C) This work. | ||
Herein we report a practical, photocatalyst-free method for silane functionalization using disulfides and dichloromethanes under purple light irradiation (Fig. 1C). In our design, thiyl radicals were generated in situ from disulfides without the need for additional catalysts and efficiently engaged in hydrogen atom transfer (HAT) to form silyl radicals.12 The resulting silyl radicals then underwent halogen atom transfer (XAT) with dichloromethane, thereby furnishing silyl chlorides under mild conditions. Furthermore, the in situ generated silyl chlorides can be selectively converted into either silanols (with water) or silyl ethers (with alcohols), offering modular access to multiple classes of valuable organosilicon compounds from the same hydrosilane precursor. This method provides an environmentally benign alternative to existing photocatalytic approaches, relying exclusively on inexpensive commercial reagents and expanding the synthetic utility of hydrosilanes.
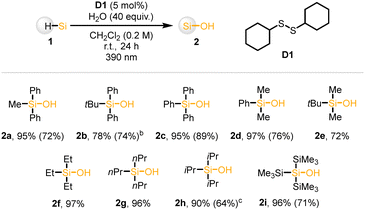
We started our investigation of silanol synthesis using diphenylmethyl silane (1a) as a model substrate. After the systemic screening, the optimal conditions were obtained as follows (see SI for further information): 1a (1.0 equiv.), dicyclohexyl disulfide (D1, 20 mol%), H2O (40 equiv.) in a solution of CH2Cl2 at room temperature under 390 nm irradiation for 24 hours (Table 1, entry 1). Under these conditions, the desired silanol 1b was obtained in 72% isolated yield. Based on this result, several control experiments were conducted. Reducing the disulfide loading from 5 mol% to 1 mol%, the reaction efficiency was significantly dropped (entry 2). Additionally, diphenyl disulfide (D2) was not efficient for product formation due to the low BDE of aryl thiols (ca. 79 kcal mol−1) (entry 3).13 When CH2Cl2 was replaced with other chlorine sources, including n-BuCl or 1,2-dichloroethane (DCE) or CH3Cl, the yield decreased significantly (entry 4–6). Replacing 390 nm with 440 nm did not afford the desired product (entry 7). Without water, only trace amounts of silanol were observed, while silyl chloride was obtained in 82% yield (entry 8).
| Entry | Deviation from standard conditionsa | Yieldb [%] |
|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), D1 (5 mol%), H2O (40 equiv.), CH2Cl2 (0.2 M), 390 nm irradiation at r.t. for 24 h. b Yield was determined by 1H NMR spectroscopy using CH2Br2 as an internal standard. c Isolated yield. d Silyl chloride was obtained. | ||
| 1 | None | 97 (72c) |
| 2 | 1 mol% of D1 | 3 |
| 3 | D2 instead of D1 | 8 |
| 4 | n-Bucl instead of CH2Cl2 | 10 |
| 5 | DCE instead of CH2Cl2 | 38 |
| 6 | CHCl3 instead of CH2Cl2 | 43 |
| 7 | 440 nm instead of 390 nm | Trace |
| 8 | Without H2O | Traced |
With the optimized conditions in hand, various silanes were investigated to access corresponding silanols (Table 2). In general, a broad range of silanes bearing aryl, alkyl, and silyl substituents were smoothly converted into the corresponding silanols. Mono-, di- and triaryl substituted silanes afforded the desired product (2a–2d) with high yield. In addition, trialkylsilanes including tBuMe2SiH, Et3SiH, and nBu3SiH, were well tolerated, giving the excellent yield (2e–2g). Plus, sterically demanding iPr3SiH furnished silnaol 2h in 87% yield. Furthermore, tris(trimethylsilyl)silane (supersilane) also underwent smooth conversion to the silanol in 96% yield (2i), highlighting generality of this protocol.
Encouraged by the success of silanol formation, we sought to extend the protocol to the synthesis of silyl ethers using alcohols as nucleophiles. Reaction optimization revealed that the presence of alcohols together with silanes (1.5 equiv.) and with 1.5 equiv. of imidazole afforded corresponding silyl ethers (see SI for further optimization, Table S5). As shown in Table 3, a wide range of silanes were compatible with this transformation, providing the desired silyl ethers. Several aryl-substituted silanes gave silyl ether with excellent yield (3a–3c). Also, trialkyl substituted silyl ether (3d–3g) were obtained in high yields. Additionally, supersilane underwent smooth transform to provide 3h in a moderate yield.
Mechanistic studies were carried out to elucidate reaction mechanism (Fig. 2). Radical trapping experiments using ethyl acrylate resulted in the formation of the corresponding hydrosilylation product (5a), providing direct evidence for the generation of a silyl radical intermediate under the photochemical conditions (Fig. 2A). A plausible mechanism for this reaction is proposed in Fig. 2B. Under 390 nm purple light irradiation, the S–S bond in dicyclohexyl disulfide undergoes homolytic cleavage,13 generating thiyl radicals to produce a silyl radical through HAT. The silyl radical can undergo XAT with dichloromethane to generate silyl chloride. In the presence of H2O or alcohol, the reaction furnishes the corresponding silanol or silyl ether. The resulting chloromethyl radical can abstract a hydrogen atom from thiol, thereby generating a thiyl radical and propagating the radical chain.
 | ||
Fig. 2 (A) Radical trapping experiments. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Tris(trimethylsilyl)silane (1.0 equiv.), D1 (5 mol%), ethyl acrylate (1.0 equiv.) in MeCN (0.2 M) at r.t. under 390 nm irradiation. (B) Plausible mechanism. Tris(trimethylsilyl)silane (1.0 equiv.), D1 (5 mol%), ethyl acrylate (1.0 equiv.) in MeCN (0.2 M) at r.t. under 390 nm irradiation. (B) Plausible mechanism. | ||
In conclusion, we have developed a simple and general method for functionalization of silanes using commercially available and environmentally benign reagents. This transformation proceeds under mild reaction conditions to produce various organosilicon compounds including silanols, silyl ether, and silyl chlorides. We believe this transformation offers a valuable platform for future applications in organosilicon chemistry and related fields.
H. K. and J. L. designed and carried out the experiments. J. L. and S. L. wrote the manuscript. S. L. supervised the project.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the Supplementary Information (SI). Supplementary information includes experimental procedures, characterization data, and NMR spectra. See DOI: https://doi.org/10.1039/d6cc00213g.Acknowledgements
This study was supported by the National Research Foundation (NRF) of Korea and funded by the Ministry of Science and ICT(RS-2024-00454580).References
-
(a) Y. Liu, I. Imae, A. Makishima and Y. Kawakami, Synthesis and characterization of poly(silphenylenesiloxane)s containing functional side groups, a study to high-temperature elastomer, RSC Appl. Interfaces, 2003, 4, 27–34 CAS;
(b) R. Murugavel, M. G. Walawalkar, M. Dan, H. W. Roesky and C. N. R. Rao, Transformations of Molecules and Secondary Building Units to Materials:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A Bottom-Up Approach, Acc. Chem. Res., 2004, 37, 763–774 CrossRef CAS PubMed.
A Bottom-Up Approach, Acc. Chem. Res., 2004, 37, 763–774 CrossRef CAS PubMed. - (a) E. W. Colvin, Silicon in organic synthesis, Chem. Soc. Rev., 1978, 7, 15–64 RSC; (b) M. Lalonde and T. Chan, Use of organosilicon reagents as protective groups in organic synthesis, Synthesis, 1985, 817–845 CrossRef CAS.
- (a) S. E. Denmark and C. S. Regens, Palladium-Catalyzed Cross-Coupling Reactions of Organosilanols and Their Salts: Practical Alternatives to Boron- and Tin-Based Methods, Acc. Chem. Res., 2008, 41, 1486–1499 CrossRef CAS PubMed; (b) R. Murugavel, A. Voigt, M. G. Walawalkar and H. W. Roesky, Hetero- and Metallasiloxanes Derived from Silanediols, Disilanols, Silanetriols, and Trisilanols, Chem. Rev., 1996, 96, 2205–2236 CrossRef CAS PubMed.
- (a) Q.-L. Hu, K.-Q. Hou, J. Li, Y. Ge, Z.-D. Song, A. S. C. Chan and X.-F. Xiong, Silanol: a bifunctional group for peptide synthesis and late-stage functionalization, Chem. Sci., 2020, 11, 6070–6074 RSC; (b) Y. Lin, K.-Z. Jiang, J. Cao, Z.-J. Zheng, Z. Xu, Y.-M. Cui and L.-W. Xu, Iridium-Catalyzed Intramolecular C–H Silylation of Siloxane-Tethered Arene and Hydrosilane: Facile and Catalytic Synthesis of Cyclic Siloxanes, Adv. Synth. Catal., 2017, 359, 2247–2252 CrossRef CAS; (c) M. Parasram and V. Gevorgyan, Silicon-Tethered Strategies for C–H Functionalization Reactions, Acc. Chem. Res., 2017, 50, 2038–2053 CrossRef CAS PubMed.
- (a) J. W. Attard, K. Osawa, Y. Guan, J. Hatt, S.-I. Kondo and A. Mattson, Silanediol Anion Binding and Enantioselective Catalysis, Synthesis, 2019, 2107–2115 CAS; (b) K. M. Diemoz, J. E. Hein, S. O. Wilson, J. C. Fettinger and A. K. Franz, Reaction Progress Kinetics Analysis of 1,3-Disiloxanediols as Hydrogen-Bonding Catalysts, J. Org. Chem., 2017, 82, 6738–6747 CrossRef CAS PubMed; (c) T. Min, J. C. Fettinger and A. K. Franz, Enantiocontrol with a Hydrogen-bond Directing Pyrrolidinylsilanol Catalyst, ACS Catal., 2012, 2, 1661–1666 CrossRef CAS; (d) A. G. Schafer, J. M. Wieting, T. J. Fisher and A. E. Mattson, Chiral silanediols in anion-binding catalysis, Angew. Chem., 2013, 52, 11321–11324 CrossRef CAS PubMed.
- (a) A. K. Franz and S. O. Wilson, Organosilicon Molecules with Medicinal Applications, J. Med. Chem., 2013, 56, 388–405 CrossRef CAS PubMed; (b) G. K. Min, D. Hernández and T. Skrydstrup, Efficient Routes to Carbon–Silicon Bond Formation for the Synthesis of Silicon-Containing Peptides and Azasilaheterocycles, Acc. Chem. Res., 2013, 46, 457–470 CrossRef CAS PubMed; (c) J.-L. Panayides, D. L. Riley, F. Hasenmaile and W. A. L. van Otterlo, The role of silicon in drug discovery: a review, RSC Med. Chem., 2024, 15, 3286–3344 RSC; (d) M. C. Parrott, M. Finniss, J. C. Luft, A. Pandya, A. Gullapalli, M. E. Napier and J. M. DeSimone, Incorporation and Controlled Release of Silyl Ether Prodrugs from PRINT Nanoparticles, J. Am. Chem. Soc., 2012, 134, 7978–7982 CrossRef CAS PubMed; (e) R. Ramesh and D. S. Reddy, Quest for Novel Chemical Entities through Incorporation of Silicon in Drug Scaffolds, J. Med. Chem., 2018, 61, 3779–3798 CrossRef CAS PubMed.
- (a) J. A. Cella and J. C. Carpenter, Procedures for the preparation of silanols, J. Organomet. Chem., 1994, 480, 23–26 CrossRef CAS; (b) E. J. Corey and A. Venkateswarlu, Protection of hydroxyl groups as tert-butyldimethylsilyl derivatives, J. Am. Chem. Soc., 1972, 94, 6190–6191 CrossRef CAS; (c) L. J. Tyler, Phenylsilanetriol, J. Am. Chem. Soc., 1955, 77, 770–771 CrossRef CAS.
- (a) W. Adam, R. Mello and R. Curci, O-Atom Insertion into Si−H Bonds by Dioxiranes: A Stereospecific and Direct Conversion of Silanes into Silanols, Angew. Chem., Int. Ed. Engl., 1990, 29, 890–891 CrossRef; (b) M. Cavicchioli, V. Montanari and G. Resnati, Oxyfunctionalization reactions by perfluoro cis-2, 3-dialkyloxaziridines. Enantioselective conversion of silanes into silanols, Tetrahedron Lett., 1994, 35, 6329–6330 CrossRef CAS; (c) P. D. Lickiss and R. Lucas, Oxidation of sterically hindered organosilicon hydrides using potassium permanganate, J. Organomet. Chem., 1996, 521, 229–234 CrossRef CAS; (d) L. Spialter, L. Pazdernik, S. Bernstein, W. A. Swansiger, G. R. Buell and M. E. Freeburger, Mechanism of the reaction of ozone with the silicon-hydrogen bond, J. Am. Chem. Soc., 1971, 93, 5682–5686 CrossRef CAS; (e) K. Valliant-Saunders, E. Gunn, G. R. Shelton, D. A. Hrovat, W. T. Borden and J. M. Mayer, Oxidation of tertiary silanes by osmium tetroxide, Inorg. Chem., 2007, 46, 5212–5219 CrossRef CAS PubMed.
- (a) S. Bähr, S. Brinkmann-Chen, M. Garcia-Borràs, J. M. Roberts, D. E. Katsoulis, K. N. Houk and F. H. Arnold, Selective enzymatic oxidation of silanes to silanols, Angew. Chem., Int. Ed., 2020, 59, 15507–15511 CrossRef PubMed; (b) J. Y. Corey and J. Braddock-Wilking, Reactions of hydrosilanes with transition-metal complexes: formation of stable transition-metal silyl compounds, Chem. Rev., 1999, 99, 175–292 CrossRef CAS PubMed; (c) W.-S. Huang, H. Xu, H. Yang and L.-W. Xu, Catalytic Synthesis of Silanols by Hydroxylation of Hydrosilanes: From Chemoselectivity to Enantioselectivity, Chem. – Eur. J., 2024, 30, e202302458 CrossRef CAS PubMed; (d) M. Jeon, J. Han and J. Park, Catalytic Synthesis of Silanols from Hydrosilanes and Applications, ACS Catal., 2012, 2, 1539–1549 CrossRef CAS; (e) Y. Kikukawa, Y. Kuroda, K. Yamaguchi and N. Mizuno, Diamond-Shaped [Ag4]4+ Cluster Encapsulated by Silicotungstate Ligands: Synthesis and Catalysis of Hydrolytic Oxidation of Silanes, Angew. Chem., Int. Ed., 2012, 51, 2434–2437 CrossRef CAS PubMed; (f) S. Li, H. Li, C.-H. Tung and L. Liu, Practical and selective bio-inspired iron-catalyzed oxidation of Si–H bonds to diversely functionalized organosilanols, ACS Catal., 2022, 12, 9143–9152 CrossRef CAS; (g) U. Prieto-Pascual, A. Rodríguez-Diéguez, Z. Freixa and M. A. Huertos, Tailor-made synthesis of hydrosilanols, hydrosiloxanes, and silanediols catalyzed by di-silyl rhodium(III) and iridium(III) complexes, Inorg. Chem., 2023, 62, 3095–3105 CrossRef CAS PubMed.
- (a) J. Li, D. Xu, G. Shi, X. Liu, J. Zhang and B. Fan, Oxidation of Silanes to Silanols with Oxygen via Photoredox Catalysis, ChemistrySelect, 2021, 6, 8345–8348 CrossRef CAS; (b) Y. Okada, M. Oba, A. Arai, K. Tanaka, K. Nishiyama and W. Ando, Diorganotelluride-Catalyzed Oxidation of Silanes to Silanols under Atmospheric Oxygen, Inorg. Chem., 2010, 49, 383–385 CrossRef CAS PubMed.
- (a) X. Fan, P. Xiao, Z. Jiao, T. Yang, X. Dai, W. Xu, J. D. Tan, G. Cui, H. Su, W. Fang and J. Wu, Neutral-Eosin-Y-Photocatalyzed Silane Chlorination Using Dichloromethane, Angew. Chem., Int. Ed., 2019, 58, 12580–12584 CrossRef CAS PubMed; (b) X. Fan, M. Zhang, Y. Gao, Q. Zhou, Y. Zhang, J. Yu, W. Xu, J. Yan, H. Liu, Z. Lei, Y. C. Ter, S. Chanmungkalakul, Y. Lum, X. Liu, G. Cui and J. Wu, Stepwise on-demand functionalization of multihydrosilanes enabled by a hydrogen-atom-transfer photocatalyst based on eosin Y, Nat. Chem., 2023, 15, 666–676 CrossRef CAS PubMed.
- J. Lee and S. Lee, Dehalogenative deuteration of alkyl and aryl bromides by thiyl radical catalysis under visible-light irradiation, Chem. Commun., 2024, 60, 5526–5529 RSC.
- F. Dénès, M. Pichowicz, G. Povie and P. Renaud, Thiyl Radicals in Organic Synthesis, Chem. Rev., 2014, 114, 2587–2693 CrossRef PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |