Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quantifying the enamine-type nucleophilic reactivity of α-aryl vinyl azides
Prabaharan Thiruvengetam ,
Jan Brossette
,
Jan Brossette ,
Christoph Gross
,
Christoph Gross ,
Hendrik Zipse
,
Hendrik Zipse * and
Armin R. Ofial
* and
Armin R. Ofial *
*
Department Chemie, Ludwig-Maximilians-Universität München, Butenandtstr. 5-13, 81377 München, Germany. E-mail: zipse@cup.uni-muenchen.de; ofial@lmu.de
First published on 16th February 2026
Abstract
Analyzing the kinetics of the reactions of α-aryl-substituted vinyl azides (VA) with benzhydrylium ions by using the relationship lg k2(20 °C) = sN(N + E) quantified the nucleophilic reactivity of VA in dichloromethane and Cyrene™. The nucleophilicities N of VA and of further styrenes correlate linearly with DFT-calculated methyl cation affinities, which facilitates the design of styrenes with predictable reactivity.
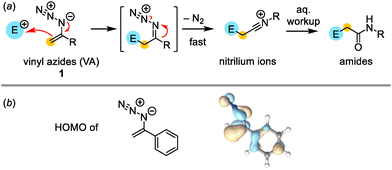
The nucleophilic reactivity of vinyl azides (VA)1 is often described to be ‘enamine-type’ owing to the fact that the α-nitrogen facilitates the attack of an electrophile at the terminal carbon of the π-system (Fig. 1a).2–5 Subsequent extrusion of molecular nitrogen is accompanied by rearrangement to nitrilium ions, which usually furnish amides upon aqueous workup.6
 | ||
| Fig. 1 (a) Reactions of vinyl azides (VA) with cationic electrophiles and subsequent aqueous workup furnishes amides. (b) Representation of the HOMO of (1-azidovinyl)benzene. | ||
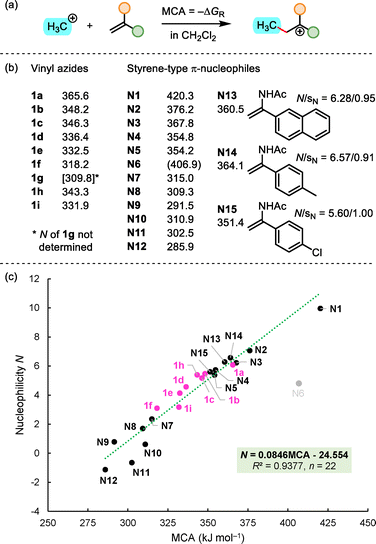
In this work, we carried out a systematic study on the nucleophilic reactivity of a series of nine α-aryl-substituted VA to embed them in the currently most comprehensive reactivity scale, that is, the benzhydrylium-based Mayr scale (BBMS).7 We show that the positioning of VA in BBMS makes it for the first time possible to compare the nucleophilic reactivity of VA with those of structurally related α-substituted ‘styrene-type’ π-systems including analogues with α-alkyl, α-(trialkylsiloxy)-, α-amido, or α-amino groups. Quantum-chemical calculations indicate that methyl cation affinities (MCA)8 are highly competent and substance-class encompassing descriptors for the nucleophilic reactivity of such π-nucleophiles.
We first analyzed the general electronic properties of VA by quantum-chemical calculations. MO energies of the parent 1-(azidovinyl)benzene were calculated at the r2SCAN-3c/SMD(DCM) level of theory using ORCA 6.0.1 and ChimeraX 1.9 for visualization (SI, Section 7.2).9 Fig. 1b illustrates that the HOMO of this VA is centered around the π-system of the C–C double bond with participation of the adjacent azido α-nitrogen in accord with the anticipated general reactivity as enamine-type nucleophile.
Encouraged by this insight, a series of nine VA (1a–1i, Fig. 2a) were synthesized from the respective alkenes by iodine azide addition and subsequent base-induced HI elimination.10 Subsequently, we set out to determine the VA nucleophilicity by using Mayr's benzhydrylium methodology and the Mayr-Patz equation (MPE, Fig. 2b), which is a linear free energy relationship.7,11,12 In the MPE, E represents the electrophilicity of the (reference) electrophile, while a nucleophile is characterized by two solvent-dependent parameters N and sN.7,11 The MPE has already been used to successfully quantify the nucleophilicities of a huge variety of π-nucleophiles, e.g., vinyl silanes, olefins, allyl silanes, silyl enol ethers, and enamides.11,12 This reliable framework makes it possible to straightforwardly compare VA reactivities with those of other nucleophilic species, also comprising enamines, which occur in many, more commonly used reactions with electrophiles.13,14
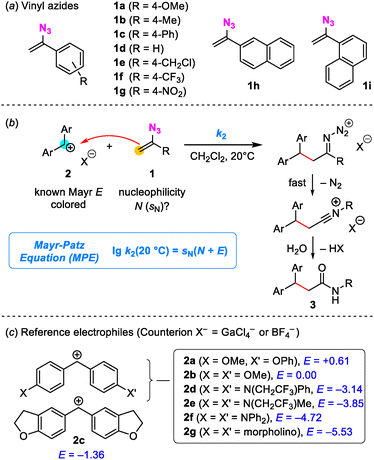 | ||
| Fig. 2 (a) Structures of investigated vinyl azides (VA). (b) Reactions of VA 1 with benzhydrylium ions 2 furnish nitrilium ions, which can be hydrolyzed to amides 3. (c) Structures of reference electrophiles 2a–2g and their electrophilicity parameters E (from ref. 11b; counterions: GaCl4− for 2a–2c and BF4− for 2d–2g). | ||
A series of electrophilic benzhydrylium ions (2) serve as the backbone of MPE-based nucleophilicity scales.7 Ions 2 (with BF4− or GaCl4− counterions) give deeply colored solutions when dissolved in dichloromethane. We planned, therefore, to follow the kinetics of the reactions of 2 with VA (1) by using photometric methods. Owing to the variable reactivity of the VA 1a–1i, a series of benzhydrylium ions 2 with electrophilicities in the range from E = +0.61 (2a) to −5.53 (2g) was used (Fig. 2c).11b Benzhydrylium ions 2c (E = −1.36) and 2e (E = −3.85) were selected to characterize typical reaction products of the kinetically investigated carbon–carbon bond forming reactions of VA 1 and benzhydrylium ions 2 (Fig. 2b). The reactions of 1a + 2c, 1d + 2e, and 1h + 2e led, after aqueous workup, to secondary amides 3 (SI, Section 4). Amide formation from the combination of 1 and 2 is rationalized by the initial formation of an iminodiazolium ion, followed by a Schmidt-type rearrangement to a nitrilium ion, which finally yields the isolated secondary amide by aqueous workup. Analogous amide formations were reported by Chiba and co-workers.6
Second-order rate constants k2 for the reactions of VA 1 with colored benzhydrylium ions 2a–2g were determined from the kinetics of their reactions in CH2Cl2 at 20 °C. The 1 + 2 reactions were monitored at or close to the absorbance maxima of the colored benzhydrylium ions by using stopped-flow or conventional UV-Vis spectroscopy. The VA nucleophiles were used in excess over the benzhydrylium ions (that is, [2]0 ≪ [1]0) which ensured pseudo-first order reaction conditions. Least squares-fitting of the mono-exponential decay functions A = A0 exp(−kobst) + C to the time-dependent experimental absorbances allowed us to determine the first-order rate constants kobs (s−1) at a given VA concentration. After carrying out analogous kinetic measurements with the same benzhydrylium ion 2 at four to five different VA concentrations, the second-order rate constant k2 (M−1 s−1) of the bimolecular 1 + 2 reaction was calculated as the slope of the linear correlation of kobs with [VA]0. It should be noted that the second-order rate constants k2 for the reactions of 1c and 1f with 2c were determined from only three kobs values. Furthermore, our attempts to determine the kinetics of the reactions of VA 1g (p-NO2) with 2 did not give rise to mono-exponential decay curves and were discarded therefore. The second-order rate constants k2 of all kinetically investigated 1 + 2 reactions in dichloromethane are listed in Table S1 (SI). DFT calculated Gibbs activation energies [ΔG‡ at the r2SCAN-3c/SMD(DCM) level of theory, SI, Section 7.5] for the adduct formation of the parent VA 1d with the electrophiles 2c–2g correlate linearly with the experimental ΔG‡ indicating that the determined second-order rate constants k2 reflect the C–C bond-formation process.
Next, the MPE-defined nucleophilic reactivity parameters N (and sN) of the individual VAs 1a–1i were calculated from linear correlations of lg k2 vs. the electrophilicity E of 2.11b Fig. 3 depicts such correlations for vinyl azides 1a (p-OMe), 1d (H), 1f (p-CF3), and 1i (naphth-1-yl). The slopes of the linear correlations reflect the nucleophiles’ sensitivity parameter sN, while the intercepts with the abscissa (that is, lg k2 = 0) correspond to the VA nucleophilicity parameters N (= −E). Individual correlations of VA 1a–1i are shown in the SI (Section 1), and the calculated N (and sN) are embedded in Fig. 4.
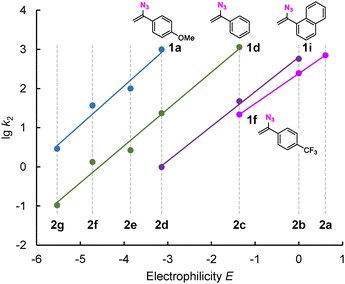 | ||
Fig. 3 Correlations of lg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k2 for reactions of vinyl azides 1 with benzhydrylium ions 2 with the electrophilicities E of 2 (all data from Table S1, SI). k2 for reactions of vinyl azides 1 with benzhydrylium ions 2 with the electrophilicities E of 2 (all data from Table S1, SI). | ||
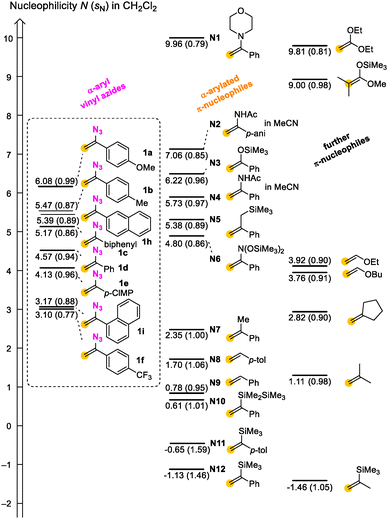 | ||
| Fig. 4 Comparison of nucleophilic reactivities of α-aryl VAs with those of structurally analogous π-nucleophiles. N and sN for all nucleophiles, except the VAs (this work), are from sources quoted in ref. 12 (p-ani = 4-MeO-C6H4-; biphenyl = 4-Ph-C6H4-; p-ClMP = 4-ClCH2-C6H4-; p-tol: 4-Me-C6H4-). | ||
The relative reactivities of VA 1 can roughly be expressed solely by their N parameters when the slight variability in the sensitivity factors sN (from 0.79 to 0.99) is neglected. The nucleophilicities N of the parent α-azidostyrene 1d and its p-X-substituted analogues correlate excellently with the Hammett substituent parameter σp of the X group.15 The correlation N = −3.738σp + 4.869 (R2 = 0.9474, n = 6, Fig. S10 in SI) can thus be used to predict the nucleophilic reactivities of further ring-substituted α-azido-styrene derivatives. The Hammett system, however, does not allow us to include both naphthyl compounds 1h and 1i in the correlation. Yet, the nucleophilicity scale in Fig. 4 shows that the β-naphthyl VA 1h is located considerably above the parent VA 1d, close to the p-Me VA 1b, in accord with the expected positioning for an electron-rich π-system. The α-naphthyl VA 1i is by two orders of magnitude less nucleophilic than 1h, which may be rationalized by unfavorable peri-interactions16 that cause a twist of the vinylic π-system out of the plane of the aromatic system (SI, Section 7.3).
Usually, second-order rate constants k2(20 °C) > 10−5 M−1 s−1 are required for efficient and productive bimolecular reactions between electrophiles and nucleophiles. It may be surprising, therefore, that nucleophilic attack of VA at relatively weak electrophiles, such as p-quinone methides (p-QM),17 dialkyl arylidenemalonates,18 or N-tosyl imines6a with E < −1019 have been reported. For example, the reaction of VA 1d (N = 4.57, sN = 0.94) with the p-QM 4-benzylidene-2,6-di-tert-butylcyclohexa-2,5-dien-1-one (E = −15.58)19a is predicted by the MPE to proceed unproductively slow (k2MPE = 4.5 × 10−11 M−1 s−1 at 20 °C). A significant enhancement of the electrophiles’ reactivity can be achieved by Lewis acid activation. Thus, complexation of p-QMs and N-tosyl imines with BF3·OEt2 and of arylidenemalonates with TiCl4 were the optimum strategies used in reported reactions of these electrophiles with VA.6a,17,18 Stronger electrophiles, such as Selectfluor (E = −5.20)20 undergo reactions with VA without the need of catalysts or promoters.10 Furthermore, acidic conditions were used by Chiba et al. to generate cationic electrophiles from alcohol precursors.6 Slowly added VA 1d attacked thus generated tritylium ions Ph3C+ (E = +0.51),21 1,3-diphenylallylium ions (E = +2.70),22 or parent benzhydrylium ions Ph2CH+ (E = +5.47)23 under mild reaction conditions (at −40 °C in CH2Cl2)6a in accord with predictions by using the MPE.
Fig. 4 gives a comprehensive overview of further π-systems with reported nucleophilic reactivities N. Obviously, α-azidostyrene 1d (N = 4.57) is by more than three orders of magnitude a stronger nucleophile than the parent styrene N9 (N = 0.78). Though the N3 group is classified as an electron-accepting group according to its Hammett parameters σp = +0.08 and σm = +0.37,15 VAs indeed benefit in reactivity from the assumed ‘enamine-type’ electron-pair donating effect of the nitrogen at the π-system. Classical enamines, such as the α-morpholino-styrene N1, are considerably stronger nucleophiles though. Yet, VA 1d reaches almost the reactivity level of the analogous enamide N4 (N = 5.73 in MeCN). A difference of ΔN ≈ +1 is consistently kept between VA and enamides when the ring-substituents at the styrene unit are varied (p-OMe: ΔN = +0.98 for 1a vs. N2, p-Me: ΔN = +1.10 for 1b vs. N14, naphth-2-yl: ΔN = +0.89 for 1h vs. N13, see below for structures of N13 and N14.
Refining organic synthesis protocols to use sustainable-by-design solvents based on renewable, plant-based raw materials requires reactivity studies in such solvents. Therefore, Table 1 lists second-order rate constants k2, which were determined for the reactions of 2d–2g with the VA 1h in Cyrene™ (= dihydrolevoglucosenon), a biorenewable solvent for which Reichardt's normalized solvent parameter ENT = 0.333 indicates slightly higher polarity than for the standard solvent dichloromethane (ENT = 0.309).24,25
| Electrophiles | 2d | 2e | 2f | 2g |
|---|---|---|---|---|
| k2 (M−1 s−1) | 583 | 28.3 | 42.6 | 1.81 |
The individual second-order rate constants k2 of comparable reactions of 1h with 2 were determined to be higher in Cyrene™ than in dichloromethane. Though the correlation of lg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k2 with E is only moderate (R2 = 0.8338), the nucleophile-specific parameters N = 6.00 and sN = 0.90 for 1h in Cyrene can be tentatively determined, highlighting that biorenewable solvents are compatible with conditions for carbocation-nucleophile reactions and enable reaction rates at the same or even higher level than fossil-sourced, classical solvents.
k2 with E is only moderate (R2 = 0.8338), the nucleophile-specific parameters N = 6.00 and sN = 0.90 for 1h in Cyrene can be tentatively determined, highlighting that biorenewable solvents are compatible with conditions for carbocation-nucleophile reactions and enable reaction rates at the same or even higher level than fossil-sourced, classical solvents.
Methyl cation affinities (MCA), calculated as the negative Gibbs free energies (ΔGR) of the addition of the methyl cation (+CH3) to the vinylic double bond (Fig. 5a), reflect the intrinsic Lewis basicities of styrene-type π-systems. Nucleophilicities of the differently α-activated styrene-type π-nucleophiles N1–N15 without ortho-substituents at the aryl ring were collected from the current version of Mayr's Reactivity Database.12 The quantum-chemically calculated MCA values of 1a–1i and N1–N15 are listed in Fig. 5b (see SI for details). The data of 22 nucleophiles in Fig. 5c show a strong linear correlation of their nucleophilicity parameter N with the thermodynamic driving force for the C–C bond formation as expressed by MCA. This linear free energy relationship is extensive, as it ranges over 12 units along the N scale and covers π-systems from various classes of compounds (vinyl silanes, alkenes, allyl silanes, silyl enol ethers, enamides, and enamines) whose MCA values differ by 134 kJ mol−1. The N,N-bis(silyloxy)enamine N6 is an outlier for unknown reasons, however.
In summary, this work provides for the first time a quantitative fundament for the understanding of the scope of organic reactions with nucleophilic vinyl azides (VA) by embedding them in Mayr's comprehensive reactivity scales, in which VA had previously not been considered. The reliable and far-reaching correlation of π-nucleophilicity with quantum-chemically calculated methyl cation affinity (MCA) facilitates the future design of styrene-like nucleophiles with foreseeable reactivity. This work also underscores that MCA is a molecular property that should not be missed as a fitting parameter in multi-variate regression analysis26 or automated quantum-chemical approaches to predict polar reactivity.27,28
Project conceptualization was done jointly by C. G. and A. R. O. Experimental methodology development and kinetic investigations were carried out by C. G. and P. T. Results of the kinetic measurements were formally analyzed and visualized by C. G., P. T., and A. R. O. Quantum-chemical investigations, supervised by H. Z., were performed, analyzed, and visualized by J. B. The manuscript was written jointly with input from all authors.
Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article, including raw and evaluated data of kinetic measurements and NMR data (FID files) are available at Open Data LMU at https://doi.org/10.5282/ubm/data.761. Further data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc00190d.Acknowledgements
We thank the Department Chemie, LMU München for financial support. This project has received funding from the European Union's Horizon Europe research and innovation programme under the Marie Skłodowska-Curie grant agreement No. 101106557 “ResolveByBio” (MSCA-2022-PF to P. T.). C. G. and A. R. O. thank for funding by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – project number 530072453 (OF 120/3-1). J. B. and H. Z. acknowledge financial support by the Deutsche Forschungsgemeinschaft (RTG 2620 Ion Pair Effects in Molecular Reactivity, project number 426795949). We gratefully acknowledge the computational and data resources provided by the Leibniz Supercomputing Centre (https://www.lrz.de).References
- K. Banert in Organic Azides: Syntheses and Applications, ed. K. Banert, S. Bräse, Wiley & Sons, Chichester, 2010, ch. 5, pp. 115–166 Search PubMed.
- A. N. Thakore, J. Buchshriber and A. C. Oehlschlager, Can. J. Chem., 1973, 51, 2406 CrossRef CAS.
- H. Hayashi, A. Kaga and S. Chiba, J. Org. Chem., 2017, 82, 11981 CrossRef CAS PubMed.
- J. Fu, G. Zanoni, E. A. Anderson and X. Bi, Chem. Soc. Rev., 2017, 46, 7208 Search PubMed.
- F. Gholami, F. Yousefnejad, B. Larijani and M. Mahdavi, RSC Adv., 2023, 13, 990 RSC.
- (a) F.-L. Zhang, Y.-F. Wang, G. H. Lonca, X. Zhu and S. Chiba, Angew. Chem., Int. Ed., 2014, 53, 4390 CrossRef CAS; (b) F.-L. Zhang, X. Zhu and S. Chiba, Org. Lett., 2015, 17, 3138 CrossRef CAS PubMed.
- H. Mayr, Tetrahedron, 2015, 71, 5095 CrossRef CAS.
- D. Kadish, A. D. Mood, M. Tavakoli, E. S. Gutman, P. Baldi and D. L. Van Vranken, J. Org. Chem., 2021, 86, 3721 Search PubMed.
- (a) F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 73 CAS; (b) F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2017, 8, e1327 Search PubMed; (c) A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378 CrossRef CAS; (d) S. Grimme, A. Hansen, S. Ehlert and J.-M. Mewes, J. Chem. Phys., 2021, 154, 064103 CrossRef CAS PubMed; (e) B. de Souza, Angew. Chem., Int. Ed., 2025, 64, e202500393 CrossRef CAS; (f) C. Bannwarth, S. Ehlert and S. Grimme, J. Chem. Theory Comput., 2019, 15, 1652 CrossRef CAS; (g) E. C. Meng, T. D. Goddard, E. F. Pettersen, G. S. Couch, Z. J. Pearson, J. H. Morris and T. E. Ferrin, Protein Sci., 2023, 32, e4792 CrossRef CAS.
- Y.-F. Wang, M. Hu, H. Hayashi, B. Xing and S. Chiba, Org. Lett., 2016, 18, 992 CrossRef CAS.
- (a) H. Mayr and M. Patz, Angew. Chem., Int. Ed. Engl., 1994, 33, 938 CrossRef; (b) H. Mayr, T. Bug, M. F. Gotta, N. Hering, B. Irrgang, B. Janker, B. Kempf, R. Loos, A. R. Ofial, G. Remennikov and H. Schimmel, J. Am. Chem. Soc., 2001, 123, 9500 CrossRef CAS; (c) H. Mayr, B. Kempf and A. R. Ofial, Acc. Chem. Res., 2003, 36, 66 CrossRef CAS; (d) R. J. Mayer, N. Hampel, P. Mayer, A. R. Ofial and H. Mayr, Eur. J. Org. Chem., 2019, 412 CrossRef CAS.
- For a freely accessible and frequently updated database for N, sN, and E parameters refer to: https://www.cup.lmu.de/oc/mayr/reaktionsdatenbank2/(accessed 10/01/2026).
- D. S. Timofeeva, R. J. Mayer, P. Mayer, A. R. Ofial and H. Mayr, Chem. – Eur. J., 2018, 24, 5901 CrossRef CAS.
- M. J. Hensinger, A. Eitzinger, O. Trapp and A. R. Ofial, Chem. – Eur. J., 2024, 30, e202302764 Search PubMed.
- C. Hansch, A. Leo and R. W. Taft, Chem. Rev., 1991, 91, 165 CrossRef CAS.
- V. Balasubramaniyan, Chem. Rev., 1966, 66, 567 Search PubMed.
- C. Lin, Y. Shen, B. Huang, Y. Liu and S. Cui, J. Org. Chem., 2017, 82, 3950 CrossRef CAS PubMed.
- X. Zhu and S. Chiba, Chem. Commun., 2016, 52, 2473 RSC.
- (a) C. Gross, A. Eitzinger, P. Mayer and A. R. Ofial, Chem. – Eur. J., 2025, 31, e202501224 CrossRef CAS PubMed; (b) O. Kaumanns, R. Lucius and H. Mayr, Chem. – Eur. J., 2008, 14, 9675 CrossRef CAS; (c) R. Appel and H. Mayr, J. Am. Chem. Soc., 2011, 133, 8240 CrossRef CAS PubMed.
- D. S. Timofeeva, A. R. Ofial and H. Mayr, J. Am. Chem. Soc., 2018, 140, 11474 CrossRef CAS PubMed.
- S. Minegishi and H. Mayr, J. Am. Chem. Soc., 2003, 125, 286 CrossRef CAS.
- K. Troshin, C. Schindele and H. Mayr, J. Org. Chem., 2011, 76, 9391 CrossRef CAS PubMed.
- J. Ammer, C. Nolte and H. Mayr, J. Am. Chem. Soc., 2012, 134, 13902 CrossRef CAS.
- J. Sherwood, M. De Bruyn, A. Constantinou, L. Moity, C. R. McElroy, T. J. Farmer, T. Duncan, W. Raverty, A. J. Hunt and J. H. Clark, Chem. Commun., 2014, 50, 9650 RSC.
- C. Reichardt and T. Welton, Solvents and Solvent Effects in Organic Chemistry, 4th edn, Wiley-VCH, Weinheim, 2010 Search PubMed.
- M. Orlandi, M. Escudero-Casao and G. Licini, J. Org. Chem., 2021, 86, 3555 CrossRef CAS PubMed.
- N. Ree, A. H. Göller and J. H. Jensen, Digital Discovery, 2024, 3, 347 RSC.
- N. Ree, J. M. Wollschläger, A. H. Göller and J. H. Jensen, Chem. Sci., 2025, 16, 5676 RSC.
| This journal is © The Royal Society of Chemistry 2026 |