Open Access Article
Open Access ArticleFrom borane to metal-bound boraindene: a cascade of bond breaking and making at rhodium
Emily J. Addisona,
Joseph A. Zurakowskia,
Paul D. Boylea,
David J. Nelson *b and
Marcus W. Drover
*b and
Marcus W. Drover *a
*a
aDepartment of Chemistry, Western University, 1151 Richmond Street, London, ON N8K 3G6, Canada. E-mail: marcus.drover@uwo.ca
bDepartment of Pure and Applied Chemistry, University of Strathclyde, 295 Cathedral Street, Glasgow, G1 1XL, UK. E-mail: david.nelson@strath.ac.uk
First published on 4th February 2026
Abstract
Few monobenzannulated 1-boraindene compounds have been reported. Here we report the synthesis of two 1-boraindenes, accessed by a bond-breaking and -making cascade from a secondary borane and diolefin, mediated by a simple pentamethylcyclopentadienyl-ligated rhodium(I) complex, [(Cp*)Rh(η2,η2-NBD)] (Cp* = C5Me5−, NBD = norbornadiene).
Main group heterocycles find diverse applications in medicine, catalysis, and materials sciences.1 The introduction of heteroatoms into a cyclic core allows for compound tunability.2 Heterocycles that include a group 13 element, for instance, provide a vacant p-orbital that extends π-conjugation and alters molecular properties.3 Lewis acid incorporation additionally offers a site for Lewis base binding, providing an axis for tuned physical properties; this is notably absent for more canonical carbon- and nitrogen-based rings.
Resembling indole and its carbon analogue, indene, 1-boraindenes are fused six-five heterocycles featuring boron within the smaller C4E ring (E = element). The electronic and structural properties of these compounds can be tuned by peripheral substitution. The chemistry of related six-five-six borafluorene analogues has been extensively explored,4 but considerably less attention has been given to 1-boraindenes (e.g., I, Fig. 1).5,6 1-Boraindenes display metallomimetic7 reactivity, such as reversible addition of H2 and cyclohexene hydrogenation.6 They remain relatively neglected, as noted by Braunschweig, who in 2024 described only the second example (II, Fig. 1).8,9 Developing creative and efficient synthetic routes to 1-boraindene cores is paramount to unlocking new applications. Related work on borafluorenes, for example,4 showcases their broad utility as organic light-emitting diodes,10 medical imaging agents,11 sensors,12 co-catalysts,13 and bifunctional ligands (III, Fig. 1).14,15
Herein we share an unusual route towards the 1-boraindene core from a secondary borane and diolefin through a series of Rh-mediated C(sp2)–F and C(sp3)–H activations, C–C reductive elimination, and alkene dehydrogenation and hydrogenation. This complex transformation merges borane and olefin groups to give a rhodium-bound 1-boraindene heterocycle, highlighting the resourcefulness of [(Cp*)RhI], resulting in an unprecedented approach to 1-boraindene cores. This report serves as a rare opportunity to dissect and repurpose well-defined bond activation pathways such as olefin functionalization and reductive elimination processes at Rh to rationally prepare new main group heterocycles.
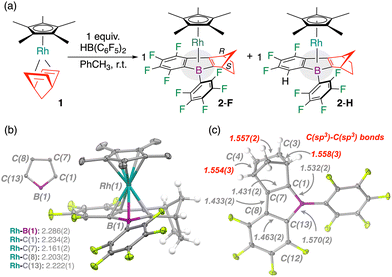
A yellow-orange C6D6 (or toluene) solution of [(Cp*)RhI(η2,η2-NBD)] (1) (Cp* = C5Me5−, NBD = norbornadiene), treated with 1 equiv. HB(C6F5)2 (Piers's borane) at 25 °C, changed color to dark red (Fig. 1). By 19F NMR spectroscopy, HB(C6F5)2 was consumed to produce several –C6Fx (x = 3–5) fragments. Following workup and extraction into pentane, this mixture consistently deposited red blocks that were suitable for single crystal X-ray diffraction analysis (25–30% isolated yield). The product, [Cp*Rh(1-boraindene-R)] (2-R: R = H or F), is an 18e− complex, comprising a core η5-BC4 ligand system that links norbornadiene and HB(C6F5)2 units (Scheme 1). In one resonance contributor, these ligands can be considered dianionic 6π-electronic systems analogous to Cp containing a Rh(III) center.16,17 By positive-mode APCI-MS, molecular ions ([M]+) for 2-H and 2-F were observed at m/z = 638.089 (calc’d 638.090 for 2-H) and m/z = 656.079 (calc’d 656.080 for 2-F) of the appropriate isotope patterns. The transformation requires 1 – no reaction is observed using (e.g.) [RhI(NBD)(μ-Cl)]2. 2-R are the first examples of 1-boraindene coordination complexes; a related phosphine-tethered borafluorene-Au analogue (III, Fig. 1) is known.14,15 The formation of 2-F formally involves the loss of one hydrogen and one fluorine atom, whereas the formation of 2-H involves the loss of two fluorine atoms; 2-H and 2-F differ by H/F exchange at C(12) (vide infra). Although this cascade to give 1-boraindene products is unprecedented, the overall transformation of coupling a diene and triarylborane to give a borole has some precedent using 1,3-butadiene and B(C6F5)3 at zirconium to give [Cp′Zr(η5-C4H4BC6F5)], from C–H activation.18
Structural analysis confirmed selective formation of the exo-product where [Cp*RhI] and the –CH2– bridgehead occupy the same face (Rh…H(C){NBD} = ca. 3.01 Å) (Scheme 1(b) and (c)). 2-H and 2-F contain point chirality at both NBD methines and planar chirality arising from the linkage of the 1-boraindene and NBD rings. In principle, 2-R could form multiple diastereomers but only one is observed experimentally. 2-H and 2-F contain a saturated external NBD fragment with d[C(3)-C(4)] = 1.557(2) Å c.f., d[C(1)–C(7)] = 1.431(2) Å, evidencing formal transfer hydrogenation. With the B-(C6F5) group front-facing, the Cp*Rh fragment is rear-tilted with closer contacts to C(7) and C(8), when compared to C(1) and C(13). A Rh–B(1) distance of 2.286(2) Å is well within the sum of van der Waals radii (3.80 Å)19 and close to the sum of covalent radii (2.26 Å).20 These metrics can be compared to Rh–B(1) for related borole compounds [CpRh(η5-C4H4BPh)]21,22 (2.304(4) Å) and [Rh(PPh3)2(Cl)(η5-C4H4BPh)]23 (2.400(5) Å). The structures of 2-R contain substitutional disorder, best modelled as a 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 split between C–H and C–F substituents at C(12), consistent with some C(12)-H/F exchange.24,25 This points to activation of both ortho C–F bonds of the pentafluorophenyl ring: one undergoes C(sp2)–F activation and C–C ring closure and another undergoes partial C(sp2)–F/C(sp2)–H exchange, presumably due to C–H reductive elimination.
40 split between C–H and C–F substituents at C(12), consistent with some C(12)-H/F exchange.24,25 This points to activation of both ortho C–F bonds of the pentafluorophenyl ring: one undergoes C(sp2)–F activation and C–C ring closure and another undergoes partial C(sp2)–F/C(sp2)–H exchange, presumably due to C–H reductive elimination.
Crystalline 2 was characterized in solution (see the SI). By 19F NMR spectroscopy, the 17 fluorine atoms observed in the crude mixture were clearly identified. [19F, 19F] COSY attributed ten of these to two different B-C6F5 rings (ca. 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio; multiplets at δF = −127.3/−127.4 (Fo), −156.8/−157.1 (Fp), and −163.7/−164.2 ppm (Fm)). The difference in peak separation between Fp and Fm (ΔδF ≈ 6 ppm) suggests a four-coordinate borane,26 also supported by 11B NMR spectroscopy (δB = 4.6 ppm, Δ1/2 = 250 Hz). Based on scalar coupling, the remaining seven fluorines comprise –C6F4 and –C6F3H rings. 19F{1H} NMR spectroscopy shows the latter coupling to an adjacent C(sp2)–H, as supported by the solid-state structure of 2-H. The presence of two 1-boraindene molecules is corroborated by 1H NMR spectroscopy, which shows the expected norbornyl-derived aliphatic signals and two sets of Cp*(CH3) signals (δH = 1.25/1.21 ppm) in a 1
1 ratio; multiplets at δF = −127.3/−127.4 (Fo), −156.8/−157.1 (Fp), and −163.7/−164.2 ppm (Fm)). The difference in peak separation between Fp and Fm (ΔδF ≈ 6 ppm) suggests a four-coordinate borane,26 also supported by 11B NMR spectroscopy (δB = 4.6 ppm, Δ1/2 = 250 Hz). Based on scalar coupling, the remaining seven fluorines comprise –C6F4 and –C6F3H rings. 19F{1H} NMR spectroscopy shows the latter coupling to an adjacent C(sp2)–H, as supported by the solid-state structure of 2-H. The presence of two 1-boraindene molecules is corroborated by 1H NMR spectroscopy, which shows the expected norbornyl-derived aliphatic signals and two sets of Cp*(CH3) signals (δH = 1.25/1.21 ppm) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. A characteristic resonance at δH = 6.88 ppm shows coupling to three fluorine atoms (3JH,F = 11.2, 4JH,F = 7.0, 5JH,F = 1.5 Hz) that is removed during 19F{1H} NMR analysis of the –C6F3H ring.
1 ratio. A characteristic resonance at δH = 6.88 ppm shows coupling to three fluorine atoms (3JH,F = 11.2, 4JH,F = 7.0, 5JH,F = 1.5 Hz) that is removed during 19F{1H} NMR analysis of the –C6F3H ring.
The reaction between 1 and HB(C6F5)2 was repeated at −78 °C (for 30 min) and then warmed to room temperature. Analysis by 19F{1H} NMR spectroscopy revealed that 2-H was not formed (Fig. S16), suggesting that 2-F is the kinetic product, while 2-H arises from a distinct, higher-energy pathway, possibly after HF elimination. This demonstrates that formation of 2-F is viable under these conditions, with the reaction likely proceeding upon warming to room temperature.
To gain further insight into this reaction we used DFT calculations at the r2SCAN0-D427,28/MA-Def2-QZVP29/SMD (toluene)30//r2SCAN-3c31 level of theory (with ORCA).32,33 Full details are in the Supporting Information. Free energies are quoted in kcal mol−1, relative to 1 and HB(C6F5)2 and include a 1.89 kcal mol−1 per molecule correction to reflect a 1 mol L−1 concentration.34 Transition state (TS) images were prepared using CYLView.35
A natural localized molecular orbital (NLMO) analysis of 2-F (NBO7)36 reveals the nature of the bonding between rhodium and the 1-boraindene fragment (Fig. 2). Visualization of the NLMOs with ChemCraft37 identified occupied NLMOs 077 and 083 that each result from mixing of a Rh dxz or dyz orbital with a p-orbital on a C atom of the alkene. Unoccupied NLMOs 154 and 160 are consistent with an antibonding interaction between the Rh d-orbitals and the C p-orbitals. Finally, unoccupied NLMOs 227 and 228 indicate antibonding interactions between the B p-orbital and C p-orbitals, with antibonding interactions with Rh in each case.
 | ||
| Fig. 2 Visualization of selected NLMOs for 2-F: NLMO 077 (occupied), NLMO 083 (occupied), NLMO 154 (unoccupied), NLMO 160 (unoccupied), NLMO 227 (unoccupied), and NLMO 228 (unoccupied). | ||
A proposed mechanism accounting for the formation of 2-F from 1 and [Cp*Rh(NBD)] was explored by DFT. While several pathways were identified, high computed barriers preclude definitive conclusions regarding the operative mechanism at this stage (Fig. S33 and S34, see SI). We therefore provide only a summary of the processes considered here, with a full discussion in the SI. Our investigation first considered hydroboration between NBD and HB(C6F5)2, which undergo a known exo-selective reaction38–40 at room temperature in the absence of a catalyst to give 3 (see the SI). Subsequent steps considered include ortho-C–F bond activation, a series of C–H bond activations, and scaffold rearrangement leading to formation of a new C–C bond via reductive elimination. Finally, a hydrogenation/dehydrogenation sequence was examined to account for formation of the observed product, 2-F. The key issues at this stage are around the timing and mechanism of HF elimination, which is difficult to track experimentally; the two emerging pathways involve either: (i) HF elimination at an early stage, therefore requiring a challenging C-C reductive elimination from rhodium(III), or (ii) a more facile reductive elimination from rhodium(V) but which then leads to highly stable intermediates that will not readily eliminate HF.
Additional control experiments were next conducted. Consistent with the calculated free energy difference between 1 + HB(C6F5)2 and its exo-hydroboration product (7.8 kcal mol−1), no stoichiometric exchange is observed between 1 and exo-3 (prepared via NBD hydroboration with 1 equiv. HB(C6F5)2)41,42 (Fig. 3a), consistent with the calculated equilibrium constant (1.9 × 10−6).
It is likely that during the formation of 2-F, HF is expelled where the ‘H’ is HB(C6F5)2-derived. To experimentally probe this, [DB(C6F5)2]2 was used, giving a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio of 2-H to 2-F (c.f. 1
3 ratio of 2-H to 2-F (c.f. 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 from [HB(C6F5)2]2). APCI-HRMS afforded an isotopic distribution envelope for 2-H and 2-F (Fig. S29 and S30). Unlike the spectrum of 2-H and 2-F generated from [HB(C6F5)2]2 (single isotopologues), the spectrum from the [DB(C6F5)2]2 reaction was best modeled as a 60
1 from [HB(C6F5)2]2). APCI-HRMS afforded an isotopic distribution envelope for 2-H and 2-F (Fig. S29 and S30). Unlike the spectrum of 2-H and 2-F generated from [HB(C6F5)2]2 (single isotopologues), the spectrum from the [DB(C6F5)2]2 reaction was best modeled as a 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 ratio of 2-H-d0 to 2-H-d1, and a 50
40 ratio of 2-H-d0 to 2-H-d1, and a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio of 2-F-d0 to 2-F-d1 (Fig. 3(b)). 1H NMR spectroscopy indicated no evidence of C(sp2)–D or methine C–D, but 2H NMR spectroscopy showed deuterium in the –CH2-CH2– fragment (Fig. S24 and S25). This suggests that both β-syn and β-anti C–H activation pathways operate, and that HF elimination is not product-determining. TSs for syn (TS-EF) and anti C-H activation (anti-TS-EF) were examined for the d0 and d1 isotopologues, as outlined in the Supporting Information. For the d0 substrate, TS-EF is 1.9 kcal mol−1 lower in energy than anti-TS-EF; in the case of the d1 substrate, anti-TS-EF is 0.3 kcal mol−1 lower in energy. These free energy differences are around the limits of DFT accuracy, but the trend suggests that selective deuteration of the hydroborated norbornadiene changes subsequent C–H activation selectivity.
50 ratio of 2-F-d0 to 2-F-d1 (Fig. 3(b)). 1H NMR spectroscopy indicated no evidence of C(sp2)–D or methine C–D, but 2H NMR spectroscopy showed deuterium in the –CH2-CH2– fragment (Fig. S24 and S25). This suggests that both β-syn and β-anti C–H activation pathways operate, and that HF elimination is not product-determining. TSs for syn (TS-EF) and anti C-H activation (anti-TS-EF) were examined for the d0 and d1 isotopologues, as outlined in the Supporting Information. For the d0 substrate, TS-EF is 1.9 kcal mol−1 lower in energy than anti-TS-EF; in the case of the d1 substrate, anti-TS-EF is 0.3 kcal mol−1 lower in energy. These free energy differences are around the limits of DFT accuracy, but the trend suggests that selective deuteration of the hydroborated norbornadiene changes subsequent C–H activation selectivity.
2-F does not convert into 2-H in solution, even in the presence of excess HB(C6F5)2 or B(C6F5)3, but trace B(C6F5)3 - used in the preparation of HB(C6F5)2 – alters the product distribution in favour of 2-F in the reaction between 1 and HB(C6F5)2; a reaction in the presence of 1 equiv. B(C6F5)3 in C6D6 at 25 °C afforded only 2-F (Fig. S24 and Fig. 3(c)). A new broad resonance at δF = −188 ppm was assigned to the formation of a species such as [FB(C6F5)3]−![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 43 which might arise from B(C6F5)3 trapping HF to give (HF)nB(C6F5)3,44 which decomposes to yield fluoroborate. Use of excess HB(C6F5)2 (5 equiv.) also supresses 2-H formation, giving 2-H and 2-F in a 1
43 which might arise from B(C6F5)3 trapping HF to give (HF)nB(C6F5)3,44 which decomposes to yield fluoroborate. Use of excess HB(C6F5)2 (5 equiv.) also supresses 2-H formation, giving 2-H and 2-F in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio. These observations support a mechanism in which HF elimination precedes 2-H formation.
4 ratio. These observations support a mechanism in which HF elimination precedes 2-H formation.
In sum, we have presented a unique approach towards an understudied class of borane-based heterocycle by taking advantage of a diolefin/secondary borane coupling strategy at Rh(I). A new complex 2-F and its hydrodefluorinated analogue 2-H have been synthesized and characterized using multinuclear NMR spectroscopy and single crystal X-ray diffraction. This transformation illustrates a rare and conceptually rich example of multi-step C–F and C–H bond making and breaking mediated by Rh/B cooperativity. These results not only elucidate an unusual reaction manifold of fluorinated organoboranes and a ubiquitous diolefin precursor but also highlight the broader potential of B–Rh cooperation in enabling multi-step synthesis of an emergent main-group heterocycle class.
The authors are grateful to Western University, the Council of Ontario Universities for a John C. Polanyi award to M. W. D., the Canadian Foundation for Innovation (LOF-212442), the Natural Sciences and Engineering Research Council of Canada (Discovery Grant, RGPIN-2020-04480 (M. W. D.), Discovery Launch Supplement, DGECR-2020-00183), and graduate award (CGS-D/NSERC Vanier to J. A. Z.), and the Ontario Government (OGS award to E. J. A.) for funding. The calculations reported in this manuscript were performed using the ARCHIE-WeSt High-Performance Computer, hosted at the University of Strathclyde (archie-west.ac.uk); D. J. N. thanks J. Buzzard, Dr K. Karina-Ossowska, and Dr R. Martin for their assistance with this facility.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, spectroscopic data, computational details, crystallographic details, and additional references. See DOI: https://doi.org/10.1039/d6cc00158k. In addition, computational data underpinning this work can be downloaded from the ioChem-BD instance hosted at the Barcelona Supercomputing Centre: https://doi.org/10.19061/iochem-bd-6-620.CCDC 2494566 (2-F/2-H) contains the supplementary crystallographic data for this paper.45
References
- C. Gao and S. A. Blum, Trends Chem., 2021, 3, 645–659 CrossRef CAS.
- A. Amin, T. Qadir, P. K. Sharma, I. Jeelani and H. Abe, Open Med. Chem. J., 2022, e187410452209010 CrossRef CAS.
- N. Matsumi, K. Naka and Y. Chujo, J. Am. Chem. Soc., 1998, 120, 5112–5113 CrossRef CAS.
- X. Su, T. A. Bartholome, J. R. Tidwell, A. Pujol, S. Yruegas, J. J. Martinez and C. D. Martin, Chem. Rev., 2021, 121, 4147–4192 CrossRef CAS PubMed.
- W. Schacht and D. Kaufmann, Angew. Chem., Int. Ed. Engl., 1987, 26, 665–666 CrossRef.
- (a) A. Y. Houghton, V. A. Karttunen, W. E. Piers and H. M. Tuononen, Chem. Commun., 2014, 50, 1295–1298 RSC; (b) A. Y. Houghton, J. Hurmalainen, A. Mansikkamäki, W. E. Piers and H. M. Tuononen, Nat. Chem., 2014, 6, 983–988 CrossRef CAS PubMed.
- M.-A. Légaré, C. Pranckevicius and H. Braunschweig, Chem. Rev., 2019, 119, 8231–8261 CrossRef PubMed.
- N. Wieprecht, I. Krummenacher, L. Wüst, M. Michel, S. Fuchs, S. Nees, M. Härterich and H. Braunschweig, Chem. Sci., 2024, 15, 12496–12501 RSC.
- N. Wieprecht, I. Krummenacher, L. Wüst, M. Michel, M. Neder, A. Häfner, J. Karg and H. Braunschweig, Chem. Sci., 2026, 17, 2772–2780 RSC.
- C. D. Entwistle and T. B. Marder, Angew. Chem., Int. Ed., 2002, 41, 2927–2931 CrossRef CAS PubMed.
- F. Jäkle, Chem. Rev., 2010, 110, 3985–4022 CrossRef PubMed.
- L. Ji, S. Griesbeck and T. B. Marder, Chem. Sci., 2017, 8, 846–863 RSC.
- P. A. Chase, W. E. Piers and B. O. Patrick, J. Am. Chem. Soc., 2000, 122, 12911–12912 CrossRef CAS.
- S. Bontemps, G. Bouhadir, K. Miqueu and D. Bourissou, J. Am. Chem. Soc., 2006, 128, 12056–12057 CrossRef CAS PubMed.
- L. A. Essex, J. W. Taylor and W. H. Harman, Tetrahedron, 2019, 75, 2255–2260 CrossRef CAS.
- H. Braunschweig, I. Krummenacher and J. Wahler, Advances in Organometallic Chemistry, Elsevier, 2013, vol. 61, pp. 1–53 Search PubMed.
- G.-E. Herberich, B. Buller, B. Hessner and W. Os, J. Organomet. Chem., 1980, 195, 253–259 CrossRef CAS.
- G. J. Pindado, S. J. Lancaster, M. Thornton-Pett and M. Bochmann, J. Am. Chem. Soc., 1998, 120, 6816–6817 CrossRef CAS.
- S. Alvarez, Dalton Trans., 2013, 42, 8617 RSC.
- B. Cordero, V. Gómez, A. E. Platero-Prats, M. Revés, J. Echeverría, E. Cremades, F. Barragán and S. Alvarez, Dalton Trans., 2008, 2832 RSC.
- D. A. Loginov, D. V. Muratov, P. V. Petrovskii, Z. A. Starikova, M. Corsini, F. Laschi, F. De Biani Fabrizi, P. Zanello and A. R. Kudinov, Eur. J. Inorg. Chem., 2005, 1737–1746 CrossRef CAS.
- D. A. Loginov, D. V. Muratov, D. S. Perekalin, Z. A. Starikova, E. A. Petrovskaya, E. I. Gutsul and A. R. Kudinov, Inorg. Chim. Acta, 2008, 361, 1715–1721 CrossRef CAS.
- G. E. Herberich, W. Boveleth, B. Hessner, M. Hostalek, D. P. J. Kijffer and M. Negele, J. Organomet. Chem., 1987, 319, 311–326 CrossRef CAS.
- M. K. Whittlesey and E. Peris, ACS Catal., 2014, 4, 3152–3159 CrossRef CAS.
- M. F. Kuehnel, D. Lentz and T. Braun, Angew. Chem., Int. Ed., 2013, 52, 3328–3348 CrossRef PubMed.
- A. G. Massey and A. J. Park, J. Organomet. Chem., 1966, 5, 218–225 CrossRef CAS.
- M. Bursch, H. Neugebauer, S. Ehlert and S. Grimme, J. Chem. Phys., 2022, 156, 134105 CrossRef CAS PubMed.
- E. Caldeweyher, S. Ehlert, A. Hansen, H. Neugebauer, S. Spicher, C. Bannwarth and S. Grimme, J. Chem. Phys., 2019, 150, 154122 CrossRef PubMed.
- J. Zheng, X. Xu and D. G. Truhlar, Theor. Chem. Acc., 2011, 128, 295–305 Search PubMed.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378–6396 CrossRef CAS PubMed.
- S. Grimme, A. Hansen, S. Ehlert and J.-M. Mewes, J. Chem. Phys., 2021, 154, 064103 CrossRef CAS PubMed.
- F. Neese, WIREs Comput. Mol. Sci., 2012, 2, 73–78 CrossRef CAS.
- F. Neese, WIREs Comput. Mol. Sci., 2022, 12, e1606 CrossRef.
- J. N. Harvey, F. Himo, F. Maseras and L. Perrin, ACS Catal., 2019, 9, 6803–6813 CrossRef CAS.
- C. Y. Legault, CYLview20. Université de Sherbrooke, 2020, Http://Www.Cylview.Org (https://Http://Www.Cylview.Org) Search PubMed.
- E. D. Glendening, J. K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, P. Karafiloglou, C. R. Landis and F. Weinhold, NBO 7.0., Theoretical Chemistry Institute, University of Wisconsin, Madison, WI, 2018 Search PubMed.
- Chemcraft - graphical software for visualization of quantum chemistry computations. Version 1.8, build 682. https://www.chemcraftprog.com Search PubMed.
- A. Iannetelli, G. Tizzard, S. J. Coles and G. R. Owen, Inorg. Chem., 2018, 57, 446–456 CrossRef CAS PubMed.
- M. Sajid, A. Lawzer, W. Dong, C. Rosorius, W. Sander, B. Schirmer, S. Grimme, C. G. Daniliuc, G. Kehr and G. Erker, J. Am. Chem. Soc., 2013, 135, 18567–18574 CrossRef CAS PubMed.
- M. Sajid, G. Kehr, T. Wiegand, H. Eckert, C. Schwickert, R. Pöttgen, A. J. P. Cardenas, T. H. Warren, R. Fröhlich, C. G. Daniliuc and G. Erker, J. Am. Chem. Soc., 2013, 135, 8882–8895 CrossRef CAS PubMed.
- H. C. Brown and S. K. Gupta, J. Am. Chem. Soc., 1975, 97, 5249–5255 CrossRef CAS.
- I. Peuser, R. C. Neu, X. Zhao, M. Ulrich, B. Schirmer, J. A. Tannert, G. Kehr, R. Fröhlich, S. Grimme, G. Erker and D. W. Stephan, Chem. – Eur. J., 2011, 17, 9640–9650 CrossRef CAS PubMed.
- R. Taube, S. Wache and J. Sieler, J. Organomet. Chem., 1993, 459, 335–347 CrossRef CAS.
- Q. Long, J. Gao, N. Yan, P. Wang and M. Li, Org. Chem. Front., 2021, 8, 3332–3341 RSC.
- CCDC 2494566: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2pqsw3.
| This journal is © The Royal Society of Chemistry 2026 |