Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Bis(N-heterocyclic carbene)-borylene-mediated heteroallene activation
An-Ping
Koh†
a,
Gaël
Vernezoul†
 a,
Jun
Fan†
a,
Zheng-Feng
Zhang
b,
Ming-Der
Su
a,
Jun
Fan†
a,
Zheng-Feng
Zhang
b,
Ming-Der
Su
 *bc and
Cheuk-Wai
So
*bc and
Cheuk-Wai
So
 *a
*a
aSchool of Chemistry, Chemical Engineering and Biotechnology, Nanyang Technological University, Singapore 637371, Singapore. E-mail: CWSo@ntu.edu.sg
bDepartment of Applied Chemistry, National Chiayi University, Chiayi 60004, Taiwan. E-mail: midesu@mail.ncyu.edu.tw
cDepartment of Medicinal and Applied Chemistry, Kaohsiung Medical University, Kaohsiung 80708, Taiwan
First published on 17th February 2026
Abstract
This study describes a bis(N-heterocyclic carbene)–borylene that activates heteroallenes. The borylene undergoes a bora-Staudinger reaction with azide to afford an NHC–iminoborane, and also catalyzes the cyclotrimerization of isocyanates to give isocyanurates. In the presence of excess pinacolborane, it catalyzes the chemoselective hydroboration of isocyanates to form N-boryl formamides.
Low-oxidation-state boron derivatives with pronounced electrophilic and/or nucleophilic character effectively mimic transition metals in the activation of enthalpically strong bonds and small molecules.1 In this context, borylenes R–B: (R = substituent), which contain a lone pair of electrons and two empty orbitals, have captured significant attention.2 Free mono-coordinate borylenes, such as H2N-B:, F-B: and Ph-B: are transient and observed spectroscopically in inert gas matrices at very low temperatures.3 The coordination of a Lewis base to this Lewis-ambiphilic B(I) center can significantly enhance borylene stability. In 2011, Bertrand and co-workers demonstrated that the parent borylene H–B: can be stabilized by two cyclic (alkyl)(amino)carbenes (cAACs), affording a borylene complex featuring a nucleophilic B(I) center.4 Since then, numerous three-coordinate borylenes supported by various Lewis bases have been isolated, many of which exhibit remarkable nucleophilic and reducing ability.5–11 By decreasing the number of supporting Lewis bases, Bertrand, Stephan, and co-workers subsequently showed that a stable two-coordinate cAAC-aminoborylene complex displays transition-metal-like activation of dihydrogen and carbon monoxide.12 Braunschweig and co-workers further discovered that transient two-coordinate cAAC-arylborylenes are capable of activating and functionalizing dinitrogen.13 Driess et al. showed that a transient bis(two-coordinate NHC-borylene) mediated the deoxygenation of a –C–O–C– skeleton.14 More recently, alternative strategies for the in situ generation of two-coordinate base-stabilized borylenes through ligand dissociation of a bis-base-stabilized three-coordinate borylene have emerged.15,16 Cummins, Gilliard, and co-workers demonstrated that the thermal extrusion of dinitrogen from a diazoborane produces a reactive N-heterocyclic carbene (NHC)–mesitylborylene that can be transferred to suitable substrates.17 They also showed that transient cAAC-haloborylenes can be released from boranorbornadienes.18 In addition, Braunschweig and co-workers reported the in situ formation of two-coordinate cAAC-arylborylene, cAAC-borylborylene and cAAC-cyclohexylborylene via the dissociation of CO and phosphine ligands from their respective three-coordinate borylene complexes.19–21
However, catalytic organic transformations mediated by low-oxidation-state boron compounds remain surprisingly underexplored. Two examples have been reported. First, Wang, Mo, and co-workers illustrated that a bis(silylene)-amidoborylene complex can serve as a precatalyst, reacting with CO2 to generate an oxo-bridged boryl-anion–silylium-cation species that catalyzes the N-formylation of amines using CO2 and HBpin.22 Second, we showed that an N-phosphinoamidinato NHC-diborene complex catalyzes the hydroboration of CO2 with HBpin.23
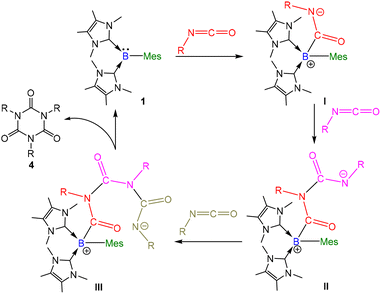
Recently, we reported the bis(NHC)-arylborylene complex [(IMe)2BMes] (1, IMe =:C{N(Me)C(Me)}2, Mes = mesityl) that exhibits strong nucleophilic character and is capable of capturing and stoichiometrically functionalizing carbon dioxide.24 Building on this reactivity, we hypothesized that compound 1 should also be competent for the functionalization and/or catalytic transformation of other heteroallenes, namely, azide and isocyanate.
To begin our study, diisopropylphenyl azide (DippN3) was examined. Compound 1 underwent the bora-Staudinger reaction with DippN3 in toluene at room temperature for 4 h to afford the NHC-iminoborane [(IMe)MesB![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NDipp] (2, yield: 76%, Scheme 1), along with the displacement of N2 and IMe. Compound 2 was isolated as a yellow crystalline solid from the concentrated reaction mixture. Such Staudinger-type reactivity of a three-coordinate bis(Lewis base)-stabilized borylene complex to form a Lewis base-stabilized iminoborane through ligand dissociation has not been reported before. Moreover, the dissociation of an NHC ligand from a borylene center is rare. In addition to our method, carbene-iminoboranes are commonly synthesized by 1,2-elimination of suitable boranamine precursors.25 The 11B{1H} NMR signal of compound 2 (22.0 ppm) is in the range of reported 11B NMR signals of three-coordinate boron centers in carbene-iminoborane complexes (12.4–24 ppm).26 The molecular structure obtained via X-ray crystallography shows that the B–N bond (1.355(5) Å, Fig. 1) is typical of a double bond.
NDipp] (2, yield: 76%, Scheme 1), along with the displacement of N2 and IMe. Compound 2 was isolated as a yellow crystalline solid from the concentrated reaction mixture. Such Staudinger-type reactivity of a three-coordinate bis(Lewis base)-stabilized borylene complex to form a Lewis base-stabilized iminoborane through ligand dissociation has not been reported before. Moreover, the dissociation of an NHC ligand from a borylene center is rare. In addition to our method, carbene-iminoboranes are commonly synthesized by 1,2-elimination of suitable boranamine precursors.25 The 11B{1H} NMR signal of compound 2 (22.0 ppm) is in the range of reported 11B NMR signals of three-coordinate boron centers in carbene-iminoborane complexes (12.4–24 ppm).26 The molecular structure obtained via X-ray crystallography shows that the B–N bond (1.355(5) Å, Fig. 1) is typical of a double bond.
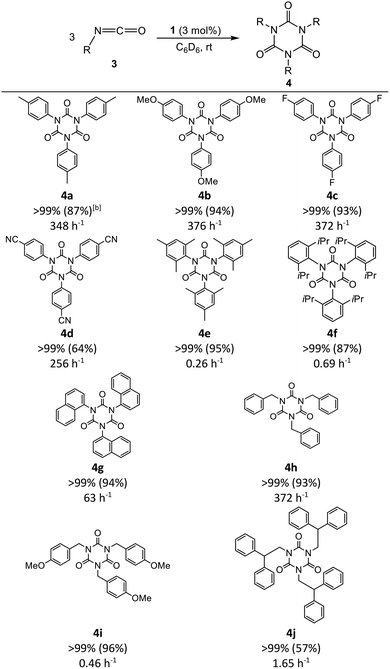
The reactivity of compound 1 toward isocyanates was further examined. Cyclotrimerization of the isocyanates was observed using both stoichiometric and catalytic amounts of 1. These results indicate that compound 1 exhibits catalytic activity. In this context, 3 mol% of compound 1 was used to catalyze the cyclotrimerization of the aryl isocyanate ArNCO (Ar = 4-CH3C6H4 (3a), Table 1), as well as its derivatives with electron-donating (Ar = 4-MeOC6H4 (3b)) and electron-withdrawing substituents (Ar = 4-FC6H4 (3c), 4-NCC6H4 (3d)) in C6D6 at room temperature for 5 min, which afforded aryl isocyanurates 4a–4d in moderate-to-high isolated yields (64–94%). As expected, a longer reaction time was required for the catalytic cyclotrimerization of sterically hindered mesityl isocyanate (3e), diisopropylphenyl isocyanate (3f), and 1-naphthyl isocyanate (3g) to afford the corresponding products 4e–4g in good isolated yields (57–95%, TOF: 0.26–66 h−1). Second, the cyclotrimerization of non-aromatic isocyanates, namely, benzylisocyanate (4h), 4-methoxybenzylisocyanate (4i), and 2,2-diphenylethyl isocyanate (4j), was also achieved in moderate-to-high isolated yields (57–96%, TOF: 0.45–370 h−1). Third, after the full conversion of p-tolyl isocyanate 4a, the reaction mixture was analyzed using 11B{1H} NMR spectroscopy. A major signal at −6.9 ppm was observed in the spectrum, indicating the regeneration of compound 1 (Fig. S4). In the catalysis, the borylene center in compound 1 attacks an isocyanate to form [(IMe)2(Mes)BC(NAr)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)] (I, Scheme 2). The negatively charged nitrogen center subsequently attacks a second isocyanate molecule to form [(IMe)2(Mes)B{C(
O)] (I, Scheme 2). The negatively charged nitrogen center subsequently attacks a second isocyanate molecule to form [(IMe)2(Mes)B{C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)(NAr)}2] (II). This reacts with a third isocyanate molecule to form [(IMe)2(Mes)B{C(
O)(NAr)}2] (II). This reacts with a third isocyanate molecule to form [(IMe)2(Mes)B{C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)(NAr)}3] (III), the terminal two-coordinated nitrogen atom of which undergoes an intramolecular nucleophilic attack on the boron-bonded carbonyl center to afford the six-membered isocyanurate 4 and regenerate compound 1. Similar mechanisms have been found in the catalytic cyclotrimerization of isocyanates mediated by an NHC,27N-heterocyclic olefin,28 proazaphosphatrane,29 2-phosphaethynolate anion30 and NHC-silyliumylidene cation.31
O)(NAr)}3] (III), the terminal two-coordinated nitrogen atom of which undergoes an intramolecular nucleophilic attack on the boron-bonded carbonyl center to afford the six-membered isocyanurate 4 and regenerate compound 1. Similar mechanisms have been found in the catalytic cyclotrimerization of isocyanates mediated by an NHC,27N-heterocyclic olefin,28 proazaphosphatrane,29 2-phosphaethynolate anion30 and NHC-silyliumylidene cation.31
| a Reaction conditions: isocyanate substrates (0.30 mmol), C6D6 (0.40 mL), catalyst 1 (3 mol%). b NMR yields were determined via1H NMR spectroscopy based on the resonances of the products with reference to the internal standards cyclohexane or 1,3,5-trimethoxybenzene. Isolated yields are reported in parentheses. All the catalytic trials were repeated in triplicate. |
|---|
|
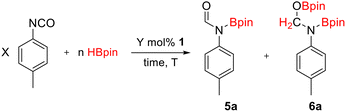
Due to the ability of compound 1 to demonstrate catalytic cyclotrimerization, we attempted to explore the catalytic hydroboration of isocyanates with HBpin. 1 mol% of compound 1 could not catalyze the hydroboration of p-tolyl isocyanate with 1 equivalent of HBpin, as catalytic cyclotrimerization occurred (entry 1, Table 2). Upon increasing the amount of HBpin to 5 equivalents, the catalytic hydroboration of p-tolyl isocyanate proceeded smoothly at room temperature, giving the corresponding N-borylated formamide with 19% conversion (entry 2, Table 2). This indicates that the presence of excess HBpin prevents intermediate I (Scheme 2) from reacting with the second molecule of isocyanate. Optimizing the catalyst loading of compound 1 to 5 mol% further improved the reaction outcome, increasing the conversion of the N-borylated formamide to 96%.
| Entry | Y (mol%) | n (eq.) | T (°C) | Time (h) | Yield (%) | |
|---|---|---|---|---|---|---|
| 5a | 6a | |||||
Reaction conditions: required amount of isocyanate substrates (0.30 mmol), required amount of 1 (1, 3 or 5 mol%), required amount of HBpin (0.30 or 1.50 mmol) and C6D6 (0.40 mL) in a J-Young NMR tube.a NMR yield of N-borylated formamide 5a was determined via1H NMR spectroscopy based on isocyanate consumption and the appearance of the NC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H signal.b NMR yield of N-,O-bis(boryl)hemiaminal 6a was determined via1H NMR spectroscopy based on N-borylated formamide consumption and the appearance of the NCH2OBpin signal. The catalytic trials were repeated in triplicate.c Optimized conditions. O)H signal.b NMR yield of N-,O-bis(boryl)hemiaminal 6a was determined via1H NMR spectroscopy based on N-borylated formamide consumption and the appearance of the NCH2OBpin signal. The catalytic trials were repeated in triplicate.c Optimized conditions. |
||||||
| 1 | 1 | 1 | 25 | 8 | <1 | <1 |
| 2 | 1 | 5 | 25 | 5 | 19 | 6 |
| 3 | 3 | 5 | 25 | 6 | 93 | 7 |
| 4c | 5 | 5 | 25 | 6 | 96 | 6 |
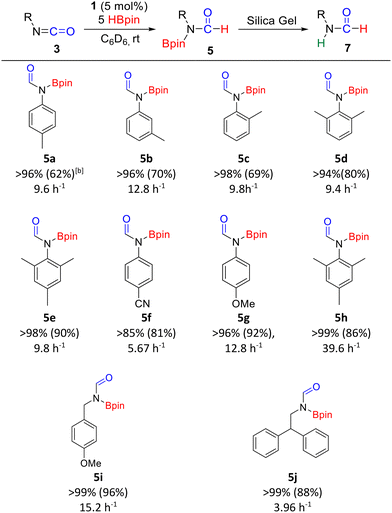
With the optimized parameters in hand, the scope and selectivity were then investigated (Table 3). The hydroboration of aromatic isocyanates containing electron-donating and electron-withdrawing substituents converted them to the respective N-borylated formamides (5a–5h, isolated yields of 7a–7h: 62–92%). Non-aromatic isocyanates were also hydroborated to their respective N-borylated formamides (5i–5j, isolated yields of 7i–7j: 88–96%). Interestingly, although the hydroboration of aromatic isocyanates afforded small quantities of the N-,O-bis(boryl) hemiaminal species, the hydroboration of non-aromatic isocyanates yielded the N-borylated formamide exclusively.
a Reaction conditions: Isocyanate substrate (0.30 mmol), C6D6 (0.40 mL), catalyst 1 (5 mol%).
b NMR yields of 5 were determined via1H NMR spectroscopy based on isocyanate consumption and the appearance of the NC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H signal. Isolated yields of 7 are reported in parentheses. All the catalytic trials were repeated in triplicate. O)H signal. Isolated yields of 7 are reported in parentheses. All the catalytic trials were repeated in triplicate.
|
|---|
|
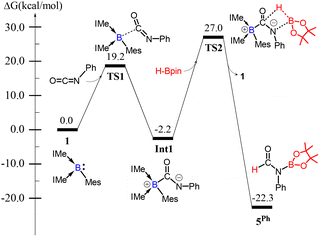
The catalytic mechanism for the 1-catalyzed hydroboration of phenyl isocyanate was studied using DFT calculations (Scheme 3). The borylene center in 1 first attacks the carbon atom of phenyl isocyanate through TS1 (ΔG‡ = 19.2 kcal mol−1) to form [(IMe)2(Mes)BC(NPh)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)] (Int 1, ΔG = −2.2 kcal mol−1). The negatively charged nitrogen center subsequently attacks the boron center of HBpin, while the hydrogen atom of HBpin attacks the carbonyl group via a 4-membered-ring transition state (TS2, ΔG‡ = 27.0 kcal mol−1), which leads to the formation of product 5Ph and regeneration of catalyst 1 (ΔG = −22.3 kcal mol−1).
O)] (Int 1, ΔG = −2.2 kcal mol−1). The negatively charged nitrogen center subsequently attacks the boron center of HBpin, while the hydrogen atom of HBpin attacks the carbonyl group via a 4-membered-ring transition state (TS2, ΔG‡ = 27.0 kcal mol−1), which leads to the formation of product 5Ph and regeneration of catalyst 1 (ΔG = −22.3 kcal mol−1).
In conclusion, bis(N-heterocyclic carbene)-mesitylborylene 1 mediated the bora-Staudinger reaction with azide to form the NHC-iminoborane, as well as the catalytic cyclotrimerization and chemoselective hydroboration of isocyanates to form isocyanurates and N-borylated formamides, respectively.
A.-P. Koh and G. Vernezoul designed and conducted the catalytic studies. J. Fan carried out the azide activation. Z.-F. Zhang and M.-D. Su designed and performed the DFT calculations. All authors have given approval to the final version of the manuscript.
Conflicts of interest
There are no conflicts to declare.Data availability
The supporting data has been provided as part of the supplementary information (SI). Supplementary information: experimental details, NMR spectra and DFT calculations. See DOI: https://doi.org/10.1039/d6cc00146g.CCDC 2270478 (2), 2257721 (4a), 2257722 (4b), 2257723 (4c), 2257724 (4d), 2257725 (4e), 2257726 (4h), 2517514 (4j) contain the supplementary crystallographic data for this paper.32a–h
Acknowledgements
This work is supported by the Ministry of Education Singapore, AcRF Tier 1 (RG8/25) and A*STAR MTC Individual Research Grants (M21K2c0117). M.-D. Su acknowledges the National Center for High-Performance Computing of Taiwan for generous amounts of computing time, and the Ministry of Science and Technology of Taiwan for the financial supportNotes and references
- M.-A. Légaré, C. Pranckevicius and H. Braunschweig, Chem. Rev., 2019, 119, 8231–8261 Search PubMed.
- M. Soleilhavoup and G. Bertrand, Angew. Chem., Int. Ed., 2017, 56, 10282–10292 CrossRef CAS PubMed and reference therein.
- (a) P. L. Timms, J. Am. Chem. Soc., 1967, 89, 1629–1632 CrossRef CAS; (b) C. A. Thompson, L. Andrews, J. M. L. Martin and J. El-Yazal, J. Phys. Chem., 1995, 99, 13839–13849 CrossRef CAS; (c) K. Edel, M. Krieg, D. Grote and H. F. Bettinger, J. Am. Chem. Soc., 2017, 139, 15151–15159 Search PubMed.
- R. Kinjo, B. Donnadieu, M. A. Celik, G. Frenking and G. Bertrand, Science, 2011, 333, 610–613 Search PubMed.
- H. Braunschweig, R. D. Dewhurst, F. Hupp, M. Nutz, K. Radacki, C. W. Tate, A. Vargas and Q. Ye, Nature, 2015, 522, 327–330 CrossRef CAS PubMed.
- L. Kong, Y. Li, R. Ganguly, D. Vidovic and R. Kinjo, Angew. Chem., Int. Ed., 2014, 53, 9280–9283 CrossRef CAS PubMed.
- A. Gärtner, L. Meier, M. Arrowsmith, M. Dietz, I. Krummenacher, R. Bertermann, F. Fantuzzi and H. Braunschweig, J. Am. Chem. Soc., 2022, 144, 21363–21370 CrossRef.
- C. Pranckevicius, C. Herok, F. Fantuzzi, B. Engels and H. Braunschweig, Angew. Chem., Int. Ed., 2019, 58, 12893–12897 CrossRef CAS.
- D. A. Ruiz, M. Melaimi and G. Bertrand, Chem. Commun., 2014, 50, 7837–7839 RSC.
- M. Arrowsmith, S. Endres, M. Heinz, V. Nestler, M. C. Holthausen and H. Braunschweig, Chem. – Eur. J., 2021, 27, 17660–17668 CrossRef CAS PubMed.
- (a) H. Wang, L. Wu, Z. Lin and Z. Xie, J. Am. Chem. Soc., 2017, 139, 13680–13683 CrossRef CAS; (b) H. Wang, L. Wu, Z. Lin and Z. Xie, Angew. Chem., Int. Ed., 2018, 57, 8708–8713 CrossRef CAS; (c) H. Wang, J. Zhang, H. K. Lee and Z. Xie, J. Am. Chem. Soc., 2018, 140, 3888–3891 CrossRef CAS.
- F. Dahcheh, D. Martin, D. W. Stephan and G. Bertrand, Angew. Chem., Int. Ed., 2014, 53, 13159–13163 CrossRef CAS PubMed.
- (a) M. A. Légaré, G. Bélanger-Chabot, R. D. Dewhurst, E. Welz, I. Krummenacher, B. Engels and H. Braunschweig, Science, 2018, 359, 896–900 CrossRef; (b) M.-A. Légaré, M. Rang, G. Bélanger-Chabot, J. I. Schweizer, I. Krummenacher, R. Bertermann, M. Arrowsmith, M. C. Holthausen and H. Braunschweig, Science, 2019, 363, 1329–1332 CrossRef; (c) M.-A. Légaré, G. Bélanger-Chabot, M. Rang, R. D. Dewhurst, I. Krummenacher, R. Bertermann and H. Braunschweig, Nat. Chem., 2020, 12, 1076–1080 CrossRef; (d) A. Gärtner, U. S. Karaca, M. Rang, M. Heinz, P. D. Engel, I. Krummenacher, M. Arrowsmith, A. Hermann, A. Matler, A. Rempel, R. Witte, H. Braunschweig, M. C. Holthausen and M.-A. Légaré, J. Am. Chem. Soc., 2023, 145, 8231–8241 CrossRef PubMed; (e) M. Rang, M. Heinz, A. Halkić, M. Weber, R. D. Dewhurst, A. Rempel, M. Härterich, M. C. Holthausen and H. Braunschweig, J. Am. Chem. Soc., 2024, 146, 11048–11053 CAS.
- J. Fan, S. Pan, S. Yao, C. Ding, G. Frenking and M. Driess, J. Am. Chem. Soc., 2025, 147, 6925–6933 CrossRef CAS.
- M. Arrowsmith, D. Auerhammer, R. Bertermann, H. Braunschweig, G. Bringmann, M. A. Celik, R. D. Dewhurst, M. Finze, M. Grüne, M. Hailmann, T. Hertle and I. Krummenacher, Angew. Chem., Int. Ed., 2016, 55, 14464–14468 Search PubMed.
- H. Braunschweig, I. Krummenacher, M.-A. Légaré, A. Matler, K. Radacki and Q. Ye, J. Am. Chem. Soc., 2017, 139, 1802–1805 CrossRef CAS.
- C. Zhang, C. C. Cummins and R. J. Gilliard, Science, 2024, 385, 327–331 CrossRef CAS.
- C. Zhang, R. J. Gilliard and C. C. Cummins, Chem. Sci., 2024, 15, 17873–17880 Search PubMed.
- C. Pranckevicius, M. Weber, I. Krummenacher, A. K. Phukan and H. Braunschweig, Chem. Sci., 2020, 11, 11055–11059 RSC.
- Y. Konrad, A. Jayaraman, I. Krummenacher and H. Braunschweig, Angew. Chem., Int. Ed., 2025, 64, e202423669 CrossRef CAS.
- M. Michel, L. Endres, F. Fantuzzi, I. Krummenacher and H. Braunschweig, Chem. Sci., 2025, 16, 5632–5639 RSC.
- X. Chen, Y. Yang, H. Wang and Z. Mo, J. Am. Chem. Soc., 2023, 145, 7011–7020 CrossRef CAS PubMed.
- J. Fan, J.-Q. Mah, M.-C. Yang, M.-D. Su and C.-W. So, J. Am. Chem. Soc., 2021, 143, 4993–5002 CrossRef PubMed.
- J. Fan, A.-P. Koh, C.-S. Wu, M.-D. Su and C.-W. So, Nat. Commun., 2024, 15, 3052 CrossRef CAS.
- Y. Fan, J. Cui and L. Kong, Eur. J. Org. Chem., 2022, e202201086 CrossRef.
- P. Cui, R. Guo, L. Kong and C. Cui, Inorg. Chem., 2020, 59, 5261–5265 CrossRef CAS.
- H. A. Duong, M. J. Cross and J. Louie, Org. Lett., 2004, 6, 4679–4681 CrossRef CAS.
- C. Li, W. Zhao, J. He and Y. Zhang, Chem. Commun., 2019, 55, 12563–12566 RSC.
- S. M. Raders and J. G. Verkade, J. Org. Chem., 2010, 75, 5308–5311 CrossRef CAS PubMed.
- D. Heift, Z. Benkő, H. Grützmacher, A. R. Jupp and J. M. Goicoechea, Chem. Sci., 2015, 6, 4017–4024 RSC.
- Y.-C. Teo, D. Loh, B.-X. Leong, Z.-F. Zhang, M.-D. Su and C.-W. So, Inorg. Chem., 2023, 62, 16867–16873 CrossRef CAS PubMed.
- (a) CCDC 2270478: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2g6m7k; (b) CCDC 2257721: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2fsbq9; (c) CCDC 2257722: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2fsbrb; (d) CCDC 2257723: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2fsbsc; (e) CCDC 2257724: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2fsbtd; (f) CCDC 2257725: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2fsbvf; (g) CCDC 2257726: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2fsbwg; (h) CCDC 2517514: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qhp42.
Footnote |
| † Equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |