Open Access Article
Open Access ArticleSynthesis of sulfur- and oxygen-bridged cationic [4]-helicenes mediated by Friedel–Crafts-SNAr tandem reactions for red-light-driven organophotoredox catalysis
Ryoga
Hasebe
a,
Rumi
Hanada
a,
Yuta
Tanaka
a,
Yuta
Goto
b,
Mio
Takeuchi
a,
Hiroyoshi
Takamura
 b,
Isao
Kadota
*b,
Kenta
Tanaka
b,
Isao
Kadota
*b,
Kenta
Tanaka
 *c and
Yujiro
Hoshino
*c and
Yujiro
Hoshino
 *a
*a
aGraduate School of Environment and Information Sciences, Yokohama National University, Tokiwadai, Hodogaya-ku, Yokohama 240-8501, Japan. E-mail: hoshino-yujiro-hy@ynu.ac.jp
bGraduate School of Environmental, Life, Natural Science and Technology, Okayama University, 3-1-1 Tsushima-Naka, Kitaku, Okayama 700-8530, Japan. E-mail: kadota-i@okayama-u.ac.jp
cResearch Institute for Interdisciplinary Science, Okayama University, 3-1-1 Tsushima-Naka, Kitaku, Okayama 700-8530, Japan. E-mail: ktanaka@okayama-u.ac.jp
First published on 30th March 2026
Abstract
The synthesis of sulfur- and oxygen-bridged cationic [4]-helicenes via a tandem Friedel–Crafts–SNAr reaction of a diaryl sulfide or a diaryl ether with a (thio)salicylic acid has been developed. The sulfur-bridged cationic [4]-helicenes are suitable as catalysts for photoredox reactions under low-energy light sources such as red LED light.
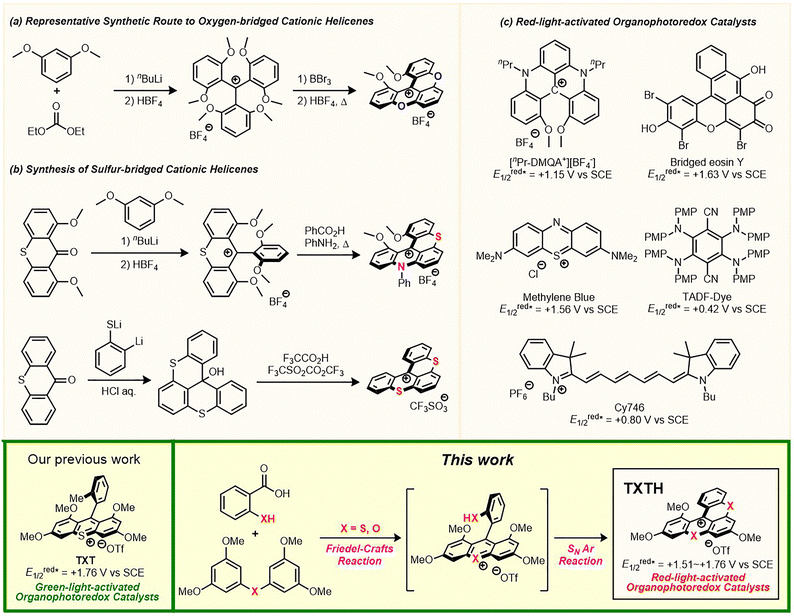
Cationic helicenes, originating from the pioneering studies on cationic triarylmethyl organic dyes by Hofmann, Verguin, and Lauth, have attracted considerable attention in organic chemistry owing to their unique structural and electronic properties.1 Oxygen- and nitrogen-bridged cationic helicenes are typically synthesized via SNAr reactions of triarylmethyl cations, which are prepared from 1,3-dimethoxybenzene and diethyl carbonate (Fig. 1(a)).2 In contrast to the numerous reports on oxygen- and nitrogen-bridged cationic helicenes, only two examples of sulfur-bridged cationic helicenes have been described, both synthesized via a two-step route starting from thioxanthone (Fig. 1(b)).3 Moreover, the physical properties of sulfur-bridged cationic helicenes remain underexplored; for example, to date, no reports have addressed their application as photoredox catalysts. Therefore, the development of efficient synthetic routes to sulfur-bridged cationic helicenes as well as the investigation of their physical properties and potential applications are highly desirable.
 | ||
| Fig. 1 Representative synthetic routes to cationic helicenes and red-light-activated organophotoredox catalysts. | ||
Visible-light-mediated photoredox catalysis has seen widespread development, particularly in the context of sustainable energy-conversion systems.4 Conventional photoredox reactions often rely on the use of high-energy light sources such as blue LED light; however, recent advances in energy-efficient systems have prompted a shift in attention toward lower-energy light sources such as red LED light. Although metal-based photoredox catalysts that are activated by red light have been extensively studied, red-light-activated organophotoredox catalysts remain relatively underexplored (Fig. 1 (c)).5 In particular, although Gianetti and co-workers reported a cationic helicene photocatalyst, [nPr-DMQA+][BF4−], that functions as a red-light-driven photoredox catalyst in 2020,5b only a few examples of cationic helicene photocatalysts have been reported to date.
Recently, we have reported the design and synthesis of thioxanthylium-based organophotoredox catalysts (Fig. 1, ‘Our previous work’) which were prepared from diaryl sulfides and aryl-acid chlorides in the presence of trifluoromethanesulfonic acid (TfOH) via a Friedel–Crafts reaction.6 These catalysts can be activated by green LED light to efficiently promote a variety of photoredox reactions.7 Accordingly, we hypothesized that using (thio)salicylaldehyde instead of aryl-acid chloride could allow performing a Friedel–Crafts–SNAr tandem reaction to yield cationic helicenes. Here, we report the synthesis of sulfur- and oxygen-bridged cationic helicenes via Friedel–Crafts–SNAr tandem reactions and their application as organophotoredox catalysts under red-light irradiation (Fig. 1, ‘This work’).
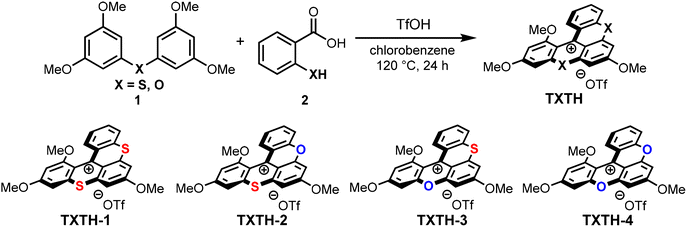
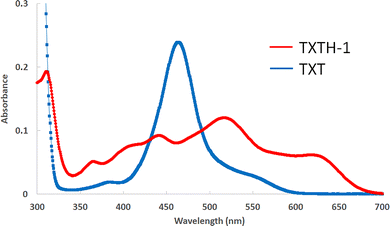
To evaluate the relationship between their structural and physical properties, various cationic [4]-helicenes (TXTH-1–4) were initially synthesized in moderate to good yield via Friedel–Crafts–SNAr tandem reactions between diaryl (thio)ether 1 and (thio)salicylic acid 2 in chlorobenzene at 120 °C (24 h) in the presence of TfOH (Table 1).6 The as-obtained cationic [4]-helicenes TXTH-1–4 exhibit moderate excited-state reduction potentials (E1/2(C*/C•−) = +1.51 to +1.76 V vs. SCE) and an absorption band in the visible spectrum. Compared with TXT (Fig. 1), the cyclic voltammograms of TXTH-1 revealed slightly negatively shifted reduction potentials (Fig. 2). Furthermore, the absorption spectrum is markedly red-shifted relative to that of TXT (Fig. 3), which can be attributed to the π-extension of the framework. In particular, TXTH-1 exhibits a broad absorption band extending to the red region of the visible spectrum (λabs < 700 nm), suggesting that it could be activated by lower-energy irradiation sources such as red light.
| Catalyst | Yielda (%) | E 1/2(C*/C˙−)b (V) | E 1/2(C/C˙−)c (V) | E 0,0 (eV) | Excitation λmax (nm) | Emission λmax (nm) |
|---|---|---|---|---|---|---|
| a All reactions were conducted using diaryl (thio)ether 1 (0.2 mmol), (thio)salicylaldehyde acid 2 (0.6 mmol), and TfOH (0.6 mmol) in chlorobenzene (120 °C; N2 atmosphere; 24 h). b Excited-state oxidation potentials were estimated on the basis of the ground-state redox potentials and the intersection of the absorption and emission bands. c Determined by cyclic voltammetry in CH3CN vs. SCE. d The values for TXT were extracted from previous reports.6a | ||||||
| TXTH-1 | 71 | +1.51 | −0.42 | 1.93 | 517 | 682 |
| TXTH-2 | 72 | +1.63 | −0.46 | 2.09 | 481 | 647 |
| TXTH-3 | 69 | +1.54 | −0.56 | 2.10 | 477 | 630 |
| TXTH-4 | 72 | +1.76 | −0.58 | 2.34 | 457 | 600 |
| TXT | — | +1.76 | −0.39 | 2.15 | 464 | 630 |
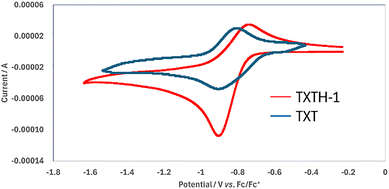 | ||
| Fig. 2 Cyclic voltammograms of TXTH-1 and TXT (10 mM); supporting electrolyte: tetra-n-butylammonium perchlorate (100 mM); in CH3CN. | ||
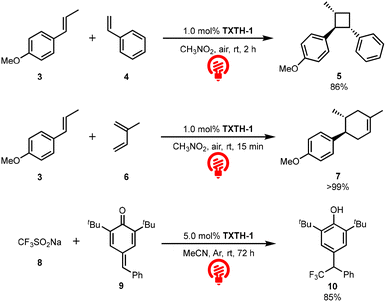
Encouraged by these promising results, we investigated TXTH-1 as a catalyst in photoredox reactions (Scheme 1). When the [2+2] cycloaddition reaction of trans-anethole (3) with styrene (4) was conducted in the presence of TXTH-1 under red-LED-light irradiation, the desired product (5) was obtained in high yield.7c Since the excited-state reduction potential of TXTH-1 is higher than that of trans-anethole (3), one-electron oxidation efficiently occurs (TXTH-1: E1/2(C*/C˙−) = +1.51 V vs. SCE; 3: Ep/2 = +1.24 V vs. SCE). Notably, TXTH-1 also catalyzed a radical-cation Diels–Alder reaction, affording the desired cyclohexene derivative (7) in high yield.6a Finally, p-quinone methide (9) underwent efficient trifluoromethylation to give the corresponding product (10) in 85% yield.8 This transformation proceeds via oxidation of the Langlois reagent (8; Ep/2 = +1.05 V vs. SCE) by TXTH-1 to generate a trifluoromethyl radical, which subsequently adds to p-quinone methide (9). These results demonstrate that TXTH-1 can serve as an effective catalyst for various photoredox reactions under red-LED-light irradiation.
In summary, we have developed a synthetic route to cationic sulfur and oxygen-bridged [4]-helicenes based on Friedel–Crafts–SNAr tandem reactions. The reactions of a diaryl (thio)ether with a (thio)salicylic acid in the presence of TfOH successfully yield the cationic [4]-helicenes in good yield. These catalysts exhibit moderate excited-state reduction potentials (E1/2(C*/C•−) = +1.51 to +1.76 V vs. SCE) and an absorption band in the visible spectrum. Especially TXTH-1 produces an absorption band that extends to the red region of the visible spectrum, thus enabling the efficient promotion of [2+2] cycloaddition, radical-cation Diels–Alder reaction, and trifluoromethylation under red-LED-light irradiation. The present reaction provides a promising platform for the synthesis of cationic helicenes and the development of red-light-driven organophotoredox catalysis.
This work was supported by Murata Science Foundation, Ichiju Industrial Science and Technology Promotion Foundation, Foundation for Interaction in Science & Technology, ENEOS Tonen General Research/Development Encouragement & Scholarship Foundation, The Yakumo Foundation for Environmental Science, Okayama Foundation for Science and Technology, JKA and its promotion funds from KEIRIN RACE, Amano Institute of Technology, Casio Science Promotion Foundation, Wesco Scientific Promotion Foundation, and JSPS Program for Forming Japan's Peak Research Universities (J-PEAKS) Grant Number JPJS00420230010.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5cc07398g.References
-
(a) J. Bosson, J. Gouin and J. Lacour, Chem. Soc. Rev., 2014, 43, 2824 RSC
; (b) D. F. Duxbury, Chem. Rev., 1993, 93, 381–433 CrossRef CAS
.
-
(a) J. C. Martin and R. G. Smith, J. Am. Chem. Soc., 1964, 86, 2252–2256 CrossRef CAS
; (b) F. Torricelli, J. Bosson, C. Besnard, M. Chekini, T. Bürgi and J. Lacour, Angew. Chem., Int. Ed., 2013, 52, 1796–1800 CrossRef CAS PubMed
; (c) B. W. Laursen and F. C. Krebs, Angew. Chem., Int. Ed., 2000, 39, 3432–3434 CrossRef CAS
; (d) G. M. Labrador, C. Besnard, T. Bürgi, A. I. Poblador-Bahamonde, J. Bosson and J. Lacour, Chem. Sci., 2019, 10, 7059–7067 RSC
; (e) J. Guin, C. Besnard and J. Lacour, Org. Lett., 2010, 12, 1748–1751 CrossRef CAS PubMed
; (f) T. J. Sørensen, A. Ø. Madsen and B. W. Laursen, Tetrahedron Lett., 2013, 54, 587–590 CrossRef
; (g) M. Marinova, S. Pascal, L. Guénée, C. Besnard, B. Shivachev, K. Kostova, C. Villani, R. Franzini, V. Dimitrov and J. Lacour, J. Org. Chem., 2020, 85, 11908–11923 CrossRef CAS PubMed
; (h) J. Gouin, T. Bürgi, L. Guénée and J. Lacour, Org. Lett., 2014, 16, 3800–3803 CrossRef CAS PubMed
; (i) M. Rosenberg, M. Santella, S. A. Bogh, A. V. Muñoz, H. O. B. Andersen, O. Hammerich, I. Bora, K. Lincke and B. W. Laursen, J. Org. Chem., 2019, 84, 2556–2567 CrossRef CAS PubMed
; (j) R. Kaur, J. Moutet, D. D. Mills and T. L. Gianetti, Chem. – Eur. J., 2025, 31, e202404135 CrossRef CAS PubMed
; (k) P. Huszthy, K. Lempert and G. Simig, J. Chem. Soc., Perkin Trans. 2, 1985, 1351–1354 RSC
.
-
(a) G. D. Figuly, C. K. Loop and J. C. Martin, J. Am. Chem. Soc., 1989, 111, 654–658 CrossRef CAS
; (b) C. Nicolas, G. Bernardinelli and J. Lacour, J. Phys. Org. Chem., 2010, 23, 1049–1056 CrossRef CAS
.
-
(a) D. A. Nicewicz and D. W. C. MacMillan, Science, 2008, 322, 77–80 CrossRef CAS PubMed
; (b) N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed
; (c) M. H. Shaw, J. Twilton and J. D. W. C. MacMillan, J. Org. Chem., 2016, 81, 6898–6926 CrossRef CAS PubMed
.
-
(a) M. M. Hossain, A. C. Shaikh, R. Kaur and T. L. Gianetti, J. Am. Chem. Soc., 2024, 146, 7922–7930 CrossRef CAS PubMed
; (b) L. Mei, J. M. Veleta and T. L. Gianetti, J. Am. Chem. Soc., 2020, 142, 12056–12061 CrossRef CAS PubMed
; (c) A. H. Schade and L. Mei, Org. Biomol. Chem., 2023, 21, 2472–2485 RSC
; (d) N. Sellet, J. Frey, M. Cormier and J.-P. Goddard, Chem. Sci., 2024, 15, 8639–8650 RSC
; (e) K. Rybicka-Jasińska, T. Wdowik, K. Łuczak, A. J. Wierzba, O. Drapała and D. Gryko, ACS Org. Inorg. Au, 2022, 2, 422–426 CrossRef PubMed
; (f) L. R. Beck, K. A. Xie, S. L. Goldschmid, S. K. Kariofillis, C. L. Joe, T. C. Sherwood, M. Sezen-Edmonds and T. Rovis, SynOpen, 2023, 07, 76–87 CrossRef CAS
; (g) Y. Miyamoto, K. Muraoka, S. Murakami, T. Matsudaira and H. Ohmiya, J. Am. Chem. Soc., 2025, 147, 41185–41192 CrossRef CAS PubMed
; (h) R. P. Pandian, T. K. Chandrashekar, G. S. S. Saini and A. L. Verma, Faraday Trans., 1993, 89, 677 RSC
; (i) A. Gizatullin, T. Yuan, S. Grotjahn, L. Cavallo, B. König, C. Zhu and M. Rueping, Angew. Chem., Int. Ed., 2026, e26086 CAS
; (j) M. Tanioka, A. Kuromiya, R. Ueda, T. Obata, A. Muranaka, M. Uchiyama and S. Kamino, Chem. Commun., 2022, 58, 7825–7828 RSC
.
-
(a) K. Tanaka, M. Kishimoto, Y. Tanaka, Y. Kamiyama, Y. Asada, M. Sukekawa, N. Ohtsuka, T. Suzuki, N. Momiyama, K. Honda and Y. Hoshino, J. Org. Chem., 2022, 87, 3319–3328 CrossRef CAS PubMed
; (b) K. Tanaka, Y. Tanaka, M. Kishimoto, Y. Hoshino and K. Honda, Beilstein J. Org. Chem., 2019, 15, 2105–2112 CrossRef CAS PubMed
; (c) K. Tanaka, K. D. Omata, Y. Asada, Y. Hoshino and K. Honda, J. Org. Chem., 2019, 84, 10669–10678 CrossRef CAS PubMed
; (d) K. Tanaka, M. Kishimoto, M. Sukekawa, Y. Hoshino and K. Honda, Tetrahedron Lett., 2018, 59, 3361–3364 CrossRef CAS
; (e) A. Mizutani, M. Kondo, S. Itakura, H. Takamura, Y. Hoshino, M. Nishikawa, I. Kadota, K. Kusamori and K. Tanaka, Bull. Chem. Soc. Jpn., 2025, 98, uoaf044 CrossRef CAS
.
-
(a) S. Nohara, S. Iwai, N. Yamaguchi, Y. Asada, Y. Kamiyama, Y. Tanaka, K. Tanaka and Y. Hoshino, Synlett, 2023, 2525–2529 CAS
; (b) K. Tanaka, Y. Asada and Y. Hoshino, Chem. Commun., 2022, 58, 2476–2479 RSC
; (c) K. Tanaka, Y. Iwama, M. Kishimoto, N. Ohtsuka, Y. Hoshino and K. Honda, Org. Lett., 2020, 22, 5207–5211 CrossRef CAS PubMed
; (d) K. Tanaka, Y. Asada, Y. Hoshino and K. Honda, Org. Biomol. Chem., 2020, 18, 8074–8078 RSC
; (e) H. Ando, H. Takamura, I. Kadota and K. Tanaka, Chem. Commun., 2024, 60, 4765–4768 RSC
; (f) M. R. El-kholany, T. Senoo, A. Mizutani, H. Takamura, T. Suzuki, I. Kadota and K. Tanaka, Org. Lett., 2025, 27, 4870–4874 CrossRef CAS PubMed
; (g) S. Kodaki, H. Ando, H. Takamura, I. Kadota and K. Tanaka, Precis. Chem., 2025, 3, 801–805 CrossRef CAS PubMed
; (h) Y. Goto, H. Ando, H. Takamura, I. Kadota and K. Tanaka, Org. Lett., 2026, 28, 147–151 CrossRef CAS PubMed
.
- K. G. Ghosh, P. Chandu, S. Mondal and D. Sureshkumar, Tetrahedron, 2019, 75, 4471–4478 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |