Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Transition metal dichalcogenide catalysts incorporating hollow carbon spheres toward water splitting and supercapacitors
Xiao
Zhang
 *
*
Advanced Institute for Materials Research (WPI-AIMR), Tohoku University, Sendai, 980-8577, Japan. E-mail: zhang.xiao.d8@tohoku.ac.jp
First published on 10th February 2026
Abstract
Transition metal dichalcogenides (TMDs), e.g., MoS2 and MoSe2, are widely used as catalysts for electrochemical water splitting and green energy conversion because of their high stability and favorable hydrogen adsorption energies. However, their intrinsically low electrical conductivity and limited surface area often lead to poor hydrogen evolution reaction (HER) performance. The high conductivity and layered structure of hollow carbon spheres (HCSs) compensate for these shortcomings, thus combining TMDs with HCSs has been extensively investigated. In addition, non-metal dopants can be introduced into carbon frameworks to control the growth of hybrid TMD nanostructures and to increase electrochemical activity by creating additional active sites. This review first focuses on the development of N-, O-, and P-doped HCSs, reviewing synthesis routes, layer numbers, and performance metrics. It then highlights recent advances in integrating layered molybdenum sulfide and selenide with HCSs, emphasizing synthesis methods, composition tuning, heterojunction formation, as well as electrochemical applications such as water splitting and supercapacitors, while systematically discussing growth kinetics and the influence of Ni and Co based components. Finally, the article addresses the remaining challenges and outlines future prospects for TMD–HCS composite catalysts in advanced applications.
Introduction
The development of green energy conversion technologies has attracted significant attention as a promising alternative to traditional fossil fuels.1–3 Because of its high energy density and carbon-free nature, hydrogen (H2) is widely regarded as a leading clean energy carrier.4,5 Green hydrogen produced via electrochemical water splitting has been widely developed as a leading technology in emerging energy sectors.6–8 During water splitting, the hydrogen evolution reaction (HER) occurs at the cathode while the oxygen evolution reaction (OER) occurs at the anode, driving the operation of water electrolyzers.9,10 The efficiency of catalysts plays an important role in this case, and environmentally friendly energy storage technologies are urgently needed to support the advancement of artificial intelligence (AI) and big data.11–13 Supercapacitors, promising for AI applications, are attracting significant attention because of their high power density and rapid charge–discharge capability.14–17 These studies have focused on improving energy density and cyclic stability, both of which depend strongly on catalyst performance.Layered two-dimensional (2D) transition metal dichalcogenides (TMDs) are investigated as bifunctional electrocatalysts for overall water splitting because their stacked structures provide efficient charge-transport pathways and large specific surface areas.18–26 The catalytic performance of TMDs is highly sensitive to chemical and phase composition, size distribution, and microstructure. For example, molybdenum disulfide (MoS2) has been extensively studied for the HER due to its high stability, a near-optimal hydrogen adsorption free energy ΔGH* = 0.08 eV, and comparable d-band electronic and chemical properties.27,28 However, the HER activity of MoS2 is generally poorer in alkaline media than in acidic solvents. The OER performance of MoS2 is also strongly influenced by its edge sites. Moreover, the impact of phase composition on TMD electrochemical behaviour, such as the 1T and 2H phases of MoS2, has attracted considerable attention. Because a single type of TMDs could rarely serve as an effective bifunctional catalyst, current research emphasizes controlling synthesis kinetics to increase active site density, suppress aggregation, tune composition, construct heterostructures for favourable band alignment, and enhance conductivity. To overcome intrinsic limitations, these 2D TMDs are often integrated with other layered materials, including hollow carbon, graphene, and MXene.29–32
Carbon-based nanomaterials, such as graphene, carbon nanotubes, and hollow carbon spheres (HCSs), provide effective support for TMD electrocatalysts by enhancing conductivity, increasing the number of active sites, and preventing nanosheet aggregation.13–16 HCSs are particularly advantageous because their hollow structures relieve mechanical stress and prevent structural collapse of active materials during electrode operation, thereby improving cyclic stability. A variety of hollow carbon structures have been synthesized using different strategies. For example, Wei and co-workers reported a yolk–shell porous carbon sphere as an electrode material for high-performance capacitive deionization.33 Liao et al. prepared novel flower-like hierarchical carbon spheres featuring multi-scale pores,34 while Huang et al. reported a kinetics-controlled, ultrafast co-assembly strategy to construct hollow mesoporous carbon spheres for high-performance supercapacitors.11 Single and multilayer HCSs have been created through various chemical approaches, most notably the hard template method. In contrast to soft template approaches, hard template methods using materials such as metal oxides, polymer spheres, or inorganic salts, are widely employed for the fabrication of hollow carbon structures due to their superior controllability. The preparation process including the template removal step critically influence the microstructures, wall thicknesses, and overall activities of the HCSs. Among the template choices, silica (SiO2) spheres of different sizes have attracted the greatest attention due to their easy availability and controllability via the classical Stöber method.35 During synthesis, a polymerization reaction between soluble phenolic resin (PR) and formaldehyde occurs on the surface of the SiO2 spheres, yielding hollow PR spheres. These can then be converted into HCSs by removing the SiO2 with a NaOH solution. By carefully controlling the deposition of SiO2 gel and organic layers, the number of HCS layers has been tuned from one up to seven, as reported in the literature.36 Single and double layered HCSs have been extensively investigated for energy conversion applications. Single-shelled HCSs exhibit thin carbon shells and a high density of accessible active sites, which facilitates controlled growth of TMDs on their surfaces. However, their structural stability is often insufficient, particularly in supercapacitor systems, due to limited mechanical robustness and suboptimal space utilization. In contrast, double-shelled HCSs offer significantly enhanced structural stability and improved space utilization, making them more suitable for applications requiring long-term cyclic durability.
Hollow carbon electrodes have been extensively investigated as supercapacitor materials due to their chemical inertness, excellent electrical conductivity, long-term cyclic stability, and large internal void space. To enhance the energy density of double-layer supercapacitors, porous carbon nanomaterials doped with non-metal elements (such as N, O, B, and P) have been developed.15,37,38 These heteroatoms are incorporated into the carbon framework to increase the specific surface area, generate hierarchical porous structures that facilitate electrolyte transport and ion exchange, and introduce redox active sites that contribute pseudocapacitance through faradaic reactions. For instance, Yang's group reported N- and O-doped HCSs with mesoporous structures, achieving a specific surface area as high as 935 m2 g−1.38 The well-defined porous structure significantly improved ion and electron transport, while the high heteroatom content (∼30%) provided substantial pseudocapacitance via redox reactions of functional groups. In addition to non-metal doping, noble metals and transition metals have also been incorporated into hollow carbons to further enhance electrochemical performance.39–43 Such metal-loaded hollow carbon nanomaterials have been developed for diverse applications in green energy conversion.
The integration of 2D TMDs with HCSs has been extensively investigated in the HER, OER, oxygen reduction reaction (ORR), batteries, supercapacitors, and other energy related applications. Among the TMDs, transition metal sulfides are widely studied.36,44–46 The hexagonal arrangement of MoS2 nanosheets provides abundant active sites and orbitals favorable for oxidative intermediates, thus enhancing the catalytic performance of the material. Furthermore, cobalt-doped 1T-MoS2 heterogeneous interfaces optimize the Gibbs free energy of hydrogen adsorption (ΔGH*) and reduce intermediate adsorption, resulting in improved HER activity.46 Iron- and nickel-doped MoS2 have also been reported as bifunctional electrocatalysts, simultaneously enhancing HER and OER performance.45 In addition, hybrid Co9S8–MoS2 alloys incorporated with HCSs effectively mitigate the capacity fading during cycling by stabilizing the MoS2 nanosheet structure.7 Layered selenides such as MoSe2 and NiSe2 have likewise been integrated with hollow carbon materials, due to the great potential of TMD-HCS composites as efficient bifunctional catalysts for diverse energy conversion and storage applications.10,48–51 For example, Luan and co-workers demonstrated the growth of MoS2−2xSe2x nanosheets on HCSs, achieving enhanced electrochemical performance.50
The synthesis kinetics of HCSs and their incorporation with TMDs present important challenges that warrant detailed mechanistic and application-oriented studies. Layered TMDs can grow both vertically and horizontally onto hollow carbon substrates.13,52,53 For instance, MoS2 nanosheets have been deposited either on the surfaces or within HCSs, exhibiting distinct electrochemical activities.37 Kinetic control of MoS2 growth on HCSs has revealed the complex interaction processes between MoS2 and carbon frameworks. Moreover, the strong interfacial bonding and uniform distribution of TMDs on HCSs play a critical role in determining the catalytic performance of the resulting composites. HCSs synthesized using SiO2 templates are frequently employed for loading layered TMDs, as the reaction between silica and metal ions facilitates tight integration with the carbon matrix and ensures homogeneous distribution.54 For example, Liu et al. fabricated shape-controlled cobalt–molybdenum silicate nanosheet arrays on HCSs, which were subsequently converted into Co9S8–MoS2@HCS composites via in situ sulfurization using S ions in place of silicate ions.7 These composites demonstrated superior electrochemical activity, enhanced specific capacity, and improved cyclic stability of the electrode material.
A large number of TMD-incorporated hollow carbon composites have been reported, demonstrating promising electrochemical activities. These composites have shown significant potential in green energy conversion, including applications in electrochemical water splitting for the HER and OER, electrochemical redox processes for chemical fuel production, and as electrode materials for batteries and supercapacitors. When layered TMD nanomaterials are integrated with HCSs, the charge carrier transport pathways are enhanced, increasing the density of active sites available for redox reactions. Despite these advances, relatively few reviews have addressed the synthesis control and energy conversion applications of TMD–HCS composites. Some of the published review articles have summarized the challenges associated with HCSs in advanced electrocatalytic applications; however, comprehensive coverage of their roles in energy conversion remains limited. Owing to their unique morphology and outstanding catalytic activity, HCSs have emerged as important materials in the field of energy chemistry.
This review emphasizes recent progress in TMD–HCS composite catalysts, focusing on key aspects such as synthesis control of single and multilayer HCSs, the influence of non-metal doping, the growth and compositional tuning of TMDs on HCSs, bandgap modulation, and overall catalytic performance. Our recent work has centred on the design and synthesis of hollow carbon nanostructures.10,12,18,29,33 Through hard template strategies, we established synthetic routes for single- and multi-layered HCS-based nanoskeletons. Their performance was further enhanced through non-metal heteroatom doping. Moreover, a range of TMDs was incorporated either within the cavities or on the outer surfaces of these HCSs to probe their electrochemical properties. Recognizing the central role of HCSs in electrochemical systems, this review highlights recent progress in the synthesis and application of TMD/HCS-based catalysts. Fig. 1 shows an overview of the key aspects reviewed for TMD–HCS composite catalysts in green energy conversion. The discussion begins with synthesis control, which is fundamental to tailoring composite properties. Topics include synthetic regulation, non-metal doping (e.g., N, O, and P), and the resulting electrochemical performance of HCSs. The growth of MoS2 nanosheets is then examined, highlighting growth orientations (vertical or horizontal) and deposition positions (inside or outside of HCSs). This review also covers the development of MoSe2, MoS2/MoSe2 alloys, and other TMDs such as NiS2, NiS/Ni3S4, and NiCo2S4. Practical applications of these composites represent another major focus, with emphasis on their roles in electrochemical processes, HER/OER performance, and supercapacitor applications. Finally, the review addresses the major challenges and future prospects of TMD-HCS composite catalysts in energy related technologies.
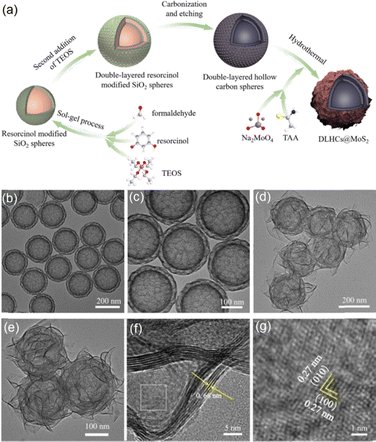
Synthesis and non-metal doping of HCSs
Carbon electrodes, particularly hollow carbon-based materials such as 1D nanotubes, 2D graphene, and 3D HCSs, have played pivotal roles in green energy conversion systems, including batteries and supercapacitors, due to their excellent conductivity and long-term cyclic stability.55,56 Among these, HCSs are especially advantageous due to their large internal voids, micrometre scale dimensions, and robust physicochemical stability.57,58 Considerable progress has been made in both the synthesis and application of HCSs. The hard template method, one of the most widely adopted strategies, has been extensively developed for fabricating HCSs for energy storage and conversion.59–62 This approach relies on templates that can be readily inserted and removed. Carbon precursors are often formed through the co-condensation of polymers and resins on the template surfaces. Subsequent high-temperature removal of the template yields the target carbon shells. Careful assembly and deposition of the precursors on the template were key to ensuring uniform cavity diameters and precise shell thicknesses.Among the various hard templates, sol–gel SiO2 spheres are frequently employed due to their inert nature, low cost, and ease of preparation and control. In early studies, the polymerization of soluble PR with formaldehyde was carried out on the surface of SiO2 spheres, forming a carbon precursor layer. HCSs could be fabricated through subsequent high-temperature carbonization followed by removal of the SiO2 cores with NaOH solution.63,64 A quite common preparation process is illustrated in Fig. 2a.65 In this approach, the sol–gel SiO2 spheres synthesized via the classical Stöber method were dispersed in water, where resorcinol and formaldehyde polymerized to form a resorcinol-formaldehyde (RF) layer on the template surface. Single-layer HCSs were then obtained by carbonization at 700 °C followed by template removal using a NaOH solution, and uniform hollow spheres were successfully fabricated as shown in the SEM image in Fig. 2b. Moreover, the wall thicknesses of the HCSs can be tuned by adjusting the deposition time of the RF layers.
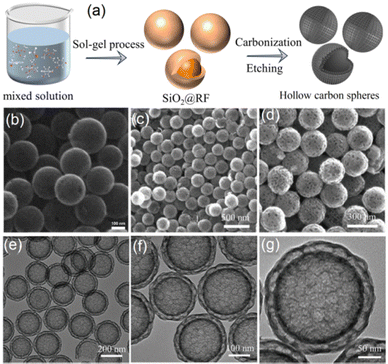 | ||
| Fig. 2 (a) Schematic illustration of the typical preparation procedure of HCSs using SiO2 templates. (b) SEM images obtained for an example of single-layer HCSs. (c) and (d) SEM images of double-layer HCSs. (e)–(g) TEM images of double-layer HCSs. (a) and (b) were reproduced with permission from ref. 64. Copyright 2025 Elsevier. (c)–(g) Were reproduced with permission from ref. 65. Copyright 2022 Wiley-VCH GmbH. | ||
In the case of the SiO2 template method developed for fabricating HCSs, the diameters and shell thicknesses of HCSs can be precisely tuned by adjusting the size of the SiO2 spheres and the polymerization time. The intrinsic porous structure of sol–gel SiO2 spheres imparts distinctive porosity to the resulting HCSs. For instance, HCSs with uniform mesoporous shells have been synthesized by controlling the hydrolysis of tetraethyl orthosilicate and the polymerization of resorcinol and formaldehyde, followed by a pyrolysis-etching process.66 The presence of mesopores within HCSs facilitates electromagnetic wave penetration and multiple reflections.66 Moreover, multilayer HCSs can be synthesized by repeating sol–gel SiO2 deposition and RF layer formation, as shown in the SEM and TEM images of double-layer HCSs in Fig. 2c–g.65 A homogeneous porous carbon shell was successfully coated onto single-layer HCSs, and these multilayer structures exhibit increased void space, enlarged surface area, and higher density of active sites.
Despite the many advantages of HCSs, the energy density of hollow carbon-based electrochemical capacitors remains limited due to their reliance on electric double-layer capacitance. Introducing non-metal heteroatoms such as N, O, P, and B into carbon frameworks has proven to be an effective strategy to optimize the porous structure, increase the density of active sites, enlarge the surface area, and enhance the pseudocapacitive contribution.67–69 Among these, N- and O-doped hollow carbon materials have been most extensively investigated, due to their facile preparation and ability to generate hierarchical porous structures.16,28,37 As electrode materials, these materials often exhibit significantly improved capacitance. In general, N-doping can be achieved by incorporating N-rich precursors during the polymerization process, while O-doping can be introduced through chemical etching. In addition, B atoms can be incorporated into the porous carbon frameworks via high-temperature treatment, whereas P-doping is often accomplished through subsequent phosphating.29 Collectively, these non-metal doped hollow carbon materials have been successfully developed and studied for diverse energy conversion applications.
The incorporation of heteroatoms into a carbon framework often significantly modifies the local electronic environment and disrupts the inherent electroneutrality of carbon materials. In the course of the oxygen reduction reaction, sp2-hybridized carbon structures can generate sp3-hybridized or defect sites, while simultaneously enhancing the effective participation of π electrons. These combined effects effectively boost the catalytic efficiency of the carbon-based electrocatalysts.70 However, the phenomenon of “self-purification” often drives heteroatoms to redistribute across the material surface, leading to poor stoichiometric control of dopant concentration.71 Instead of using conventional doping strategies (that includes sequential steps of pre-carbonization, activation, and heteroatom incorporation) which are inherently labour-intensive and time-consuming, it is recommended to utilize more straightforward facile synthetic approaches for producing the target sustainable and high-performance HCS electrocatalysts. For instance, Chen and colleagues developed a streamlined one-step strategy to synthesize HCSs through the pyrolysis of polypyrrole-coated polystyrene (PPy@PS) core–shell nanoparticles, followed by chemical activation of the HCSs utilizing KOH to yield an activated hollow carbon sphere (A-HCS) material which fits for electrode applications, delivering outstanding supercapacitor performance with a specific capacitance of 535 F g−1 (Fig. 3).38 According to Chen et al., this A-HCS material having a high specific surface area coupled with hollow interiors exhibited well-defined porosity confined within an ultrathin curved carbon shell of less than 12 nm, as well as abundant N- and O-containing functional groups.38 A comparative overview of the capacitance performance of some reported hollow carbon-based materials is shown in Table 1.
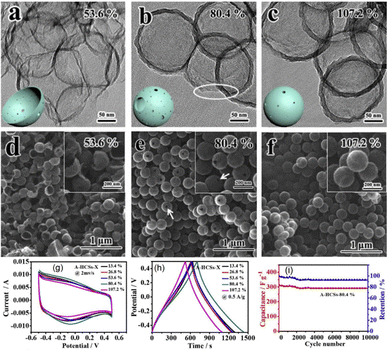 | ||
| Fig. 3 TEM and SEM images of the reported (a) and (d) A-HCSs-53.6%; (b) and (e) A-HCSs-80.4%, and (c) and (f) A-HCSs-107.2%. The green-colored spherical insets in (a), (b), and (c) show the schematic illustration of HCSs. (g) CV curves of activated composites at 2 mV s−1. (h) Galvanostatic charge–discharge curves obtained for activated composites in 6 M KOH electrolytes with a constant current density of 0.5 A g−1. (i) Specific capacitance of A-HCSs-80.4% during charge–discharge cycles at current densities of 0.5 A g−1 for the total of 104 cycles. Reproduced from ref. 38 with permission. Copyright 2017 Elsevier. | ||
| Sample | Specific capacitance (F g−1) | Ref. |
|---|---|---|
| Microporous HCSs | 240 (2 mV s−1) | 72 |
| N and O doped HCSs | 210 (0.5 A g−1) | 73 |
| N doped porous CSs | 388 (1 A g−1) | 74 |
| Mesoporous HCSs | 253 (1 A g−1) | 75 |
| Porous CSs | 260 (0.5 A g−1) | 76 |
| Graphite HCSs | 273 (0.5 A g−1) | 77 |
| Porous N-doped HCSs | 213 (0.5 A g−1) | 78 |
| Hierarchical porous CSs | 401 (0.5 A g−1) | 38 |
To further enhance device capacitance in the case of utilizing HCS materials in energy storage systems, the development of well-defined pore structures and the incorporation of heteroatoms into HCSs are often highly desirable. In this case, the relatively large specific surface area (of around 935 m2 g−1) and hierarchical porosity of the N- and O-doped HCSs fabricated by Chen et al. featuring macro-, micro-, and mesoporous structures collectively promoted the efficient ion and electron transport of the composite system.38 Since the high heteroatom content contributes significant pseudocapacitance through redox faradaic reactions of the electrochemically active functional groups, the activated HCSs delivered excellent specific capacitance of 535 F g−1 at a current density of 0.2 A g−1, while still retaining 55% of their maximum capacitance at 10 A g−1.38 The activated heteroatom-doped HCSs combine high surface area, hierarchical porosity, and abundant functional groups to deliver excellent electrochemical performance.
MoS2 and HCS composite catalysts
MoS2, a representative TMD, is often regarded as an inorganic analogue of graphene. The edge sites of MoS2 (composed of exposed Mo and S atoms) often enable strong covalent bonding, whereas the interlayer S atoms are bound only via weak van der Waals interactions. Due to its stacking symmetry, MoS2 can crystallize into three distinct polymorphs, tetragonal (1T-MoS2), hexagonal (2H-MoS2), and rhombohedral (3R-MoS2).64 In its monolayer form, MoS2 often consists of a molybdenum layer sandwiched between two S layers, with each Mo atom coordinated to six S atoms in a trigonal prismatic geometry.79 In general, the electronic structure and catalytic behaviour of MoS2 are highly dependent on its phase composition, with the 2H phase exhibiting semiconducting properties, while the 1T phase is considered to be metallic.80 The electrochemical activity is likewise phase sensitive.14 Specifically, 1T-MoS2 adopts an octahedral coordination derived from a trigonal prism rotated by 60° around the c-axis.81 The partially occupied Mo 4d and S 3p orbitals near the Fermi level activate the basal planes of 1T-MoS2, generating abundant catalytic sites for hydrogen generation. As a result, 1T-MoS2 often demonstrates superior catalytic performance compared to 2H-MoS2 and is widely considered a promising bifunctional electrocatalyst. Notably, prior studies have shown that the basal-plane activity of 1T-MoS2 yields a conductivity approximately 107 times higher than its 2H counterpart.82,83MoS2 nanosheets have emerged as promising alternatives to platinum for HER application. Numerous strategies have been investigated to further enhance the catalytic efficiency of MoS2 nanosheets, aiming to increase the density of their active sites, improve structural stability, and optimize hydrogen adsorption. Nevertheless, the OER activity of 1T-MoS2 nanosheets remains relatively weak.16,27 Considerable efforts have therefore been devoted to boosting their overall electrochemical performance.50 For instance, interface engineering and the incorporation of metal atoms into otherwise inert planes have been explored as effective approaches for MoS2-based catalysts.64,65 Hybrid composites of MoS2 with conductive metals or alloys have also been shown to accelerate charge transport, generate supplementary active sites, and enhance durability. Carbonaceous materials can also serve as excellent support for the MoS2 nanosheet components, enabling synergistic improvements in both HER and OER activity. Key design principles have thus centered on maximizing active site exposure, facilitating electron transport, and inducing favorable phase transitions in MoS2.
To overcome these above-mentioned issues, MoS2 nanosheets have been integrated with conductive substrates to form diverse heterostructures aimed at boosting their electrochemical performance. Carbon-based materials including graphene, reduced graphene oxide, carbon nanotubes, and HCSs have been extensively investigated in this context.16,35 Among these options, HCSs have attracted particular interest due to their high porosity, well-defined internal cavities, excellent thermal stability, and superior electrical conductivity.64 In comparison with carbon nanotubes, HCSs exhibit a distinctive pore structure that could provide abundant anchoring sites for MoS2 nanosheets while accommodating the volume fluctuations during prolonged cycling processes. Consequently, the combination of MoS2 nanosheets with the high surface area and low density of HCSs enhances the ion/electron transport, energy storage capability, and catalytic activity of the material, making them highly promising for advanced electrochemical applications.
Considering the phase structure of MoS2, the hydrothermal synthesis route offers distinct advantages, including high crystallinity and uniform particle size distribution, due to its mild operating conditions compared to chemical vapor deposition, electrochemical, or other chemical methods.84,85 Preparation parameters e.g., reaction time, solvent choice, and the incorporation of ionic liquids strongly influence the morphology and phase purity of MoS2.64 For instance, Tian and co-workers demonstrated phase purity control within a pH range of 5–8; Lee et al. synthesized composite catalysts across a broad pH spectrum for property comparison; and Li's group systematically investigated the combined effects of pH and solvent environment.86–88 Recently, Liu and co-workers synthesized a composite material having enhanced HER and supercapacitor performance with homogeneous growth of MoS2 nanosheets on HCSs.64 In this approach, hybrid MoS2/HCSs composites (CMS-x-y in Fig. 4, where x represents the weight ratio of Na2MoO4·2H2O and y corresponds to the pH value) were synthesized via a solvothermal route to uniformly anchor MoS2 nanosheets onto HCSs.64 Notably, the CMS-20-3.5 composites exhibited a mixed 1T/2H phase structure, delivering high energy density when assembled with activated carbon into an asymmetric supercapacitor device.
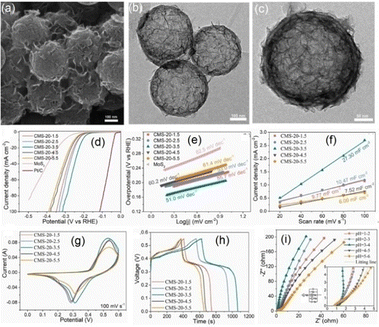 | ||
| Fig. 4 (a)–(c) TEM images of MOS2 modified HCSs (denoted as CMS-20-3.5). (d)–(i) Electrochemical performance of CMS-x-y composites in 0.5 M H2SO4 media: (d) LSV curves, (e) Tafel plots, (f) Cdl values, (g) CV curves of the composite catalysts at a scan rate of 100 mV s−1, (h) GCD curves of the composites at a current density of 1 A g−1, and (i) Nyquist plots of the composites with the inset demonstrating the corresponding equivalent circuit diagram. Reproduced from ref. 64 with permission. Copyright 2025 Elsevier. | ||
Based on the TEM images (at varying magnifications) of the CMS-20-3.5 composite catalyst presented in Fig. 4a–c, the thin MoS2 nanosheets were vertically and uniformly grown on HCS substrate without noticeable aggregation.64 This homogeneous distribution was attributed to the co-presence of ethanol and water within a pH range of 3–4, where the increased concentration of H+ ions promoted hydrolysis of the S precursor and enhanced solvent acidity.64 Such uniform growth maximized the utilization of MoS2 active sites, while the thin carbon layer of HCSs facilitated charge transport within the composite system, thereby improving catalytic performance. The LSV curves in Fig. 4d demonstrated that the CMS-20-3.5 composite exhibited the highest HER activity using optimized parameters, and the fact that the electrode prepared from CMS-20-3.5 showed the lowest Tafel slope (of 51.0 mV dec−1) indicated the superior reaction kinetics of the material.64 In addition, the Cdl value derived from CV measurements at different sweep rates (Fig. 4c) confirmed that CMS-20-3.5 also exhibited the largest capacitance of 27.30 mF cm−2 in this case.64
The Tafel slopes of the HCSs@MoS2 composite catalysts showed a clear correlation with the LSV curves, with values around 62 mV dec−1, indicating that all the corresponding composites followed a similar reaction pathway.64 This HCS composite fabricated by Liu et al. demonstrated much higher HER activity and a lower Tafel slope compared to its pure MoS2 nanosheets counterpart, because of the improved conductivity that was due to the presence of HCSs and the prevention of MoS2 stacking, which exposed a greater number of active sites. During H2 generation in acidic media, the Volmer step (eqn (1)) involving proton adsorption on the MoS2 surface occurred first, followed by the rate-determining Heyrovsky step (eqn (2)) for H2 desorption.64 The hollow carbon shells were able to increase the number of accessible active sites while promoting efficient electron transport from the carbon framework to MoS2 through strong coupling interactions, thereby facilitating the overall H2 evolution process.
| H3O+ + e− → Hads + H2O | (1) |
| Hads + H3O+ + e− → H2 + H2O | (2) |
The electrochemical energy storage behaviour of this HCSs@MoS2 composite material was further examined by Liu et al. through CV, GCD, and EIS analyses.64 As shown in Fig. 4d and g, the CV and GCD profiles of the electrodes at varying scan rates demonstrated that increasing the scan rate led to a progressive rise in peak current, indicative of excellent kinetic reversibility and robust rate capability. The observed broadening of the oxidation peak toward higher potentials and the shift of the reduction peak toward lower potentials were because of the electrode polarization effects.64,89,90 In this case, the CMS-20-3.5 composites also exhibited the largest current response and the most pronounced CV enclosed area, underscoring its superior specific capacitance. Complementary EIS measurements (Fig. 4f) further corroborated these findings. All composites fabricated displayed characteristic pseudosemicircles in the high-frequency region and distinct slopes in the low-frequency domain, in which CMS-20-3.5 showed the smallest semicircle diameter and steepest slope, signifying reduced charge transfer resistance, enhanced ion diffusion, and accelerated transport kinetics.64 Collectively, these features highlighted the pivotal role of hollow carbon shells in optimizing the capacitive performance of MoS2 modified composites. In addition, an asymmetric supercapacitor was assembled by Liu and co-workers using CMS-20-3.5 and active carbon (AC) in 3 M KOH electrolyte.64 The estimated Cdl value of CMS-20-3.5||AC was 102.5 F g−1 at 1 A g−1 with reasonably high stability maintained even after 5000 cycles. This device also achieved an excellent maximum energy density of 36.4 Wh kg−1 at a corresponding power density of 800 W kg−1, attributed to the intrinsic high stability of the HCSs@MoS2 composites.64 The hollow carbon shells provided mechanical resilience against deformation arising from repeated expansion and contraction during long-term charge–discharge cycling. Moreover, serving as a robust substrate, the HCSs were able to effectively mitigate the aggregation of MoS2 with its lamellar structure, preserving accessible active sites and enhancing the electrochemical efficiency of the composite material.64
Precise control over synthesis has been critical in the case of HCS-based MoS2 modified composite catalysts to achieving satisfied performance. The porous structure of HCSs provides abundant nucleation sites for MoS2 deposition while the incorporation of non-metal dopants into the carbon framework further introduces active centres for MoS2 growth.91–94 Wei and co-workers demonstrated a controllable strategy for constructing P-doped MoS2 nanopetals on N-doped HCSs (as shown in the TEM images in Fig. 5a–i), yielding enhanced H2 generation efficiency.37 By tuning relevant synthetic parameters, layered MoS2 nanopetals were able to be directed to grow either on the exterior or within the hollow carbon shells.37 The hierarchical N-C@P-MoS2 core–shell structures (fabricated by Wei et al. via calcination and hydrothermal treatment) exhibited superior conductivity and much higher density of active sites.37 These composites delivered outstanding HER performance, characterized by a low onset overpotential (117 mV), a small Tafel slope (68 mV dec−1), and excellent stability, because of the synergistic effects of the MoS2 and HCS components in a rationally engineered hierarchical structure.
 | ||
| Fig. 5 TEM images collected for (a) N-doped HCSs, (c) C@MoS2-2, (d) N-C@MoS2-2, (e) N-C@P-MoS2-2, and (h) and (i) N-C/P-MoS2-2 (inside decoration) composite systems. High-resolution TEM images of (b) N-doped HCSs, and (f) and (g) N-C@P-MoS2-2 composites. Reproduced from ref. 37 with permission. Copyright 2019 Elsevier. | ||
The porous structure and thin carbon shells of HCSs again were advantageous for enhancing the conductivity of the composites and facilitating free diffusion of reactants during electrochemical processes. The TEM analysis in Fig. 5f revealed that the P-MoS2 nanosheets exhibited fewer layers with increased interplanar spacing (0.67–0.72 nm, Fig. 5g). This enlargement of the (002) plane in 2H-MoS2 offered additional active sites for H2 adsorption and desorption.7,37,94,95 In addition, Wei et al. also successfully encapsulated P-doped MoS2 nanosheets inside the N-doped HCSs (N-C/P-MoS2) by adjusting precursor loading (Fig. 5h–i). However, in this case, the confinement of the active sites within the carbon shells hindered accessibility, resulting in diminished HER activity of the composites.37 These further confirmed the high dependency of electrocatalytic activity of MoS2 modified HCS composites on structural configuration, particularly the exposure of active sites and overall conductivity, with non-metal doping emerging as a powerful strategy to optimize the electrochemical performance of this hybrid system.
Detailed HER and relevant data provided by Wei and co-workers are presented in Fig. 6, with the HER polarization curves of C@MoS2, N-C@MoS2, and N-C@P-MoS2 (Fig. 6a) highlighting the pronounced influence of heteroatom doping.37 Compared to the undoped system (C@MoS2), N-C@MoS2 exhibited significantly enhanced activity. This was because the introduction of N heteroatoms modified the electronic configuration by inducing irregular charge distribution around adjacent carbon atoms, improving the intrinsic conductivity of the material. Moreover, substitution of P atoms for S atoms in the basal plane altered the electronic environment of the neighbouring S atoms, effectively tuning the conductivity of the material. In this case, the P atoms acted as the new catalytic centres, expanding the interlayer spacing and increasing the density of accessible active sites.37 The synergistic contribution of N and P dopants in facilitating H2 production is illustrated in the proposed reaction pathways for N-C@P-MoS2 composites in Fig. 6e. Since the P-doped MoS2 nanosheet modified N-doped HCSs (N-C@P-MoS2-2) delivered the most outstanding performance (with a low onset overpotential of 117 mV, an overpotential of 188 mV at 10 mA cm−2 and higher current densities under identical potentials), it was clear that dual N and P doping played a pivotal role in optimizing the conductivity and increasing the active site density of the MoS2 modified HCS system.37
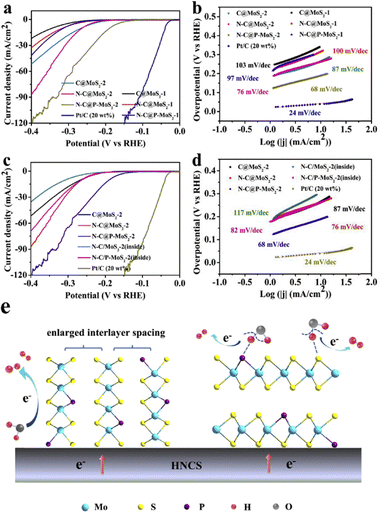 | ||
| Fig. 6 (a) Detailed HER polarization curves of the composite catalysts at a scan rate of 5 mV s−1. (b) Corresponding Tafel plots derived from the HER polarization curves. (c) HER polarization curves of the composite systems, and (d) corresponding Tafel plots. (e) Proposed reaction mechanism for H2 generation using the N-C@P-MoS2 system. Reproduced from ref. 37 with permission. Copyright 2019 Elsevier. | ||
Among the various HCSs, double-layer HCSs (DLHCs) can be fabricated via a SiO2-templated approach by tuning the deposition and polymerization steps. Compared with single-layer DCSs, DLHCs often exhibit a more uniform morphology, and both their inner and outer carbon shells exhibit rough and porous surfaces that offer abundant nucleation sites for loading TMDs.96 DLHCs thus are considered promising hosts for growing TMDs to enhance energy storage performance and rate capability. Luan et al. developed a core–shell MoS2 decorated DLHC composite material (DLHCs@MoS2 in Fig. 7) having synergistic enhancement in electrochemical activity; exhibiting low Tafel slope, small onset potential, and outstanding supercapacitor performance (delivering a high specific capacitance of 399.4 F g−1 at a current density of 0.5 A g−1).35 In this case, the DLHC framework provided excellent structural stability, inhibited the aggregation of MoS2 nanosheets, increased the exposure of active sites, and enhanced the electrical conductivity of the composite system.35 As illustrated in Fig. 7a, a RF polymer layer was sequentially deposited twice onto sol–gel-derived SiO2 spheres, followed by subsequent removal of the silica template using NaOH to yield a well-defined highly porous DLHC substrate (Fig. 7b and c, with an interlayer spacing of approximately 18 nm between the two carbon shells).35 The MoS2 nanosheet components were then uniformly coated onto the DLHC surfaces via hydrothermal treatment, producing a distinct core–shell structure (Fig. 7d and e), with MoS2 nanosheets adhered tightly to the DLHCs surface, forming a robust and continuous shell.35 According to Luan et al., the MoS2 nanosheets were determined to consist of 8–12 layers, and the measured interlayer distance of 0.66 nm for the (002) planes confirmed the formation of the 2H-phase MoS2.35,47,97,98 The magnified region highlighted in Fig. 7f and shown in Fig. 7g displays the characteristic honeycomb lattice corresponding to the (010) and (100) planes, further verifying the crystalline structure.
 | ||
| Fig. 7 (a) Detailed synthetic processes for MoS2 nanosheet modified DLHCs. (b) and (c) TEM images of the DLHCs substrate. (d)–(g) TEM images of the DLHCs@MoS2-1 composites at different magnifications, with (g) showing the region highlighted by the white box in (f). Reproduced with permission from ref. 35. Copyright 2022 Elsevier. | ||
The analysis of the X-ray absorption near-edge structure (XANES) and X-ray absorption fine structure (XAFS) spectra (Fig. 8a and b) shows that the Mo K-edge of the DLHCs@MoS2-1 composites shifts to higher energy relative to pristine MoS2, demonstrating the stronger electronic interaction between MoS2 and the DLHC base, arising from the electron transport at the interfaces.13,35,99 In addition, differences between the spectra of DLHCs@MoS2-1 and metallic Mo confirmed the presence of a Mo–O–C heterointerface which originated from carbonyl groups formed during NaOH etching. The HER data of the DLHCs@MoS2 composites with varying MoS2 loading presented in Fig. 8c and d demonstrated that as the amount of assembled MoS2 nanosheets increased, the onset potential became progressively more negative, indicating a decline in catalytic activity. This trend was particularly evident for the DLHCs@MoS2-1.5 and DLHCs@MoS2-2 composites, both of which exhibited larger onset potentials, reflecting reduced HER performance.
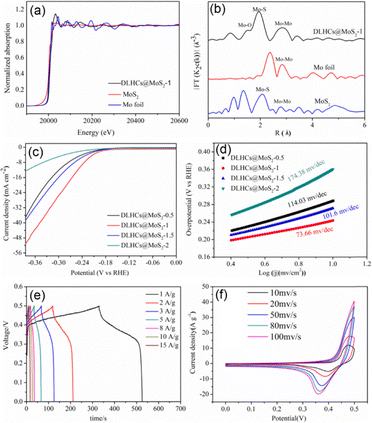 | ||
| Fig. 8 Data on the chemical environment of the DLHCs@MoS2-1 composites as probed via X-ray absorption spectroscopy: (a) Mo K-edge spectra of the EXAFS spectra; (b) Fourier transform of EXAFS spectra. (c) LSV curves and (d) Tafel slopes of the DLHCs@MoS2 composite systems. (e) and (f) Data collected using the DLHCs@MoS2-1 electrode: (e) GCD curves collected at different current densities and (f) CV curves at varying scan rates. Reproduced with permission from ref. 35. Copyright 2022 Elsevier. | ||
According to Luan et al., the behaviour described above was due to the intrinsic characteristics of MoS2. Since MoS2 exposes active sites primarily along its S edges, its ultrathin nanosheet structure tends to agglomerate easily and suffers from limited stability.27 When MoS2 is integrated with DLHCs, however, a greater number of S edge sites become accessible.37 Both insufficient and excessive loading of MoS2 nanosheets reduced the number of effectively exposed active sites, diminishing the overall catalytic performance.100 Moreover, the Tafel relationship describing the correlation between H2 generation overpotential and current density can be expressed as shown in eqn (3).
η = blog![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j + a j + a | (3) |
The GCD and CV curves in Fig. 8e and f show an increased current density and extended charge–discharge times of the composite system.35 In this case, the CV profiles exhibit distinct redox peaks corresponding to the reversible changes in the valence state of Mo (Mo(IV) ⇔ Mo(V) ⇔ Mo(VI)), which significantly enhances the capacitance performance of the composites. The presence of DLHC components facilitates charge transport to the active sites on the MoS2 components with promoted electrochemical reactions, and the hierarchical core–shell structure enhances the structural stability of the material. When utilized as supercapacitor electrodes, MoS2 nanosheets anchored on DLHCs expand the electrolyte–electrode interfaces, expose a higher density of electrochemically active sites, and consequently enhance the energy storage capacity of the material.
MoSSe components on HCSs as catalysts
To substantially enhance the performance of MoS2/HCS composites, researchers often employ MoS2-based alloys and heterostructure construction methodologies, which can increase the density of active sites through interface engineering.49 Due to the structural similarity to MoS2, MoSe2 has been another popular option for electrocatalyst fabrication, featuring a low Gibbs free energy for hydrogen adsorption. Its graphite-like layered structure, characterized by relatively wide interlayer spacing, is believed to facilitate charge transport through enhanced interlayer reflection and scattering. Nevertheless, the intrinsically low conductivity of MoSe2, along with its tendency to aggregate, often diminishes its catalytic efficiency, while its substantial volume expansion often further impedes mass transfer.101 Integrating layered MoSe2 with MoS2 to form heterostructures or alloyed structures is therefore a promising strategy. In this case, the incorporation of Se can enrich active sites and improve overall water splitting performance.102 When MoSe2 is anchored onto HCSs, its volume expansion can be effectively constrained, boosting the pseudocapacitive behaviour of the HCSs.103 Recent research has increasingly focused on constructing Se-doped MoS2 (MoSSe) composites on HCSs, where compositional tuning of MoSSe provides a versatile platform for developing high-performance catalysts.In general, MoSe2 features a graphite-like layered structure with relatively large interlayer spacing, where each Mo atomic layer sandwiched between two Se layers through strong intralayer covalent bonding and weak interlayer van der Waals interactions. This structural arrangement governs its intrinsic electronic properties, and its electrocatalytic activity is primarily associated with exposed Mo centres and Se-terminated edge sites.49 A range of reported MoSe2-based composites have demonstrated promising functional performance, for example, MoSe2/N-doped carbon anodes showed stable and efficient behaviour in sodium-ion batteries, while 2H/1T-MoSe2@graphene hybrids exhibited enhanced microwave absorption capabilities.101,103,104 However, both MoS2 and MoSe2 often exhibit rapid growth tendencies during synthetic processes, making it difficult to achieve homogeneous dispersion while avoiding aggregation. In such cases, employing HCSs as the base forming composite materials can be considered a practical solution. The tunable composition and surface chemistry of HCSs can regulate the nucleation and growth kinetics of MoS2 and MoSe2, while simultaneously offering efficient pathways for charge transport and mass diffusion. As a result, the incorporation of MoS2/MoSe2 composite nanocomponents onto the HCS framework leads to more exposed active sites and significantly enhanced energy storage performance.
The controlled growth of MoS2/MoSe2 composite nanosheets on HCSs has been widely explored as an effective strategy to enhance structural uniformity and catalytic performance. Yang and co-workers, for instance, employed SiO2 templates to fabricate HCSs and deposited MoS2−2xSe2x nanosheets with uniform spatial distribution onto this HCS base via a hydrothermal process.50 In this system, both the precursor concentration and the Se/S precursor ratio were found to be critical parameters governing the nucleation, deposition behaviour, and overall growth kinetics of MoS2 and MoSe2 on the carbon-based framework. Representative TEM images of the constructed MoS2−2xSe2x/HCSs composites (denoted C@MoS1.8Se0.2) are shown in Fig. 9a–d, confirming that the HCSs exhibit a consistent and well-defined size distribution conducive to homogeneous nanosheets assembly.
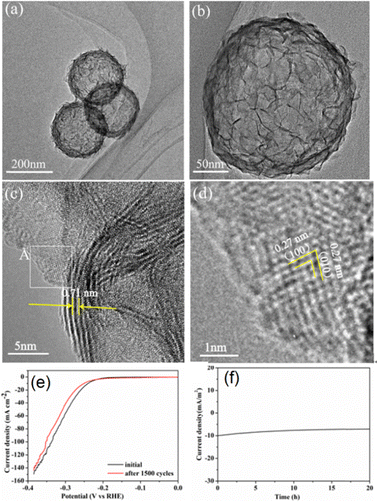 | ||
| Fig. 9 (a)–(c) TEM images of the reported C@MoS1.8Se0.2 composites; (d) corresponding enlarged white square area in (c); (e) HER polarization data for C@MoS1.8Se0.2 composites before/after 1500 cycles; (f) time-dependent current density curve for the C@MoS1.8Se0.2 composites at a static overpotential of 190 mV vs. reversible hydrogen electrode for 20 h. Reproduced with permission from ref. 50. Copyright 2021 American Chemical Society. | ||
The C@MoS1.8Se0.2 composites in Fig. 9 exhibit an average particle diameter of approximately 250 nm. As shown in the HRTEM image in Fig. 9, the MoS2−2xSe2x nanosheets are uniformly distributed and conformally anchored onto the surfaces of the HCSs.50 In contrast to the MoS2 modified HCSs presented in Fig. 5 and 7, the MoS2−2xSe2x nanosheet composites displayed much improved interfacial contact with the HCS substrates, which was attributed to the distinct growth behaviour induced by the incorporation of Se components. The presence of Se increased the density of nucleation sites, facilitating more effective deposition and co-growth of MoS2 and MoSe2 monomeric species.
The composite nanosheets grow both vertically and horizontally on the HCS surfaces, forming a well-integrated heterostructure, with the Se-doped MoS2 layers exhibiting an expanded interlayer spacing of 0.71 nm (Fig. 9c). Fig. 9d further presents a TEM image showing nanosheets lying horizontally on the HCS surface, where the characteristic honeycomb lattice of MoS2 is clearly resolved, with lattice spacings corresponding to the (100) and (010) planes. Notably, the interlayer spacing of the Se-doped structure was significantly larger than that of pristine 2H-MoS2.49,50 This expansion increased the exposure of catalytically active sites and was advantageous for promoting hydrogen adsorption and desorption during the hydrogen generation reactions. In this case, the enlarged interlayer spacing generated by Se incorporation increased the number of accessible catalytic sites and facilitated charge transport, thus enhanced the electrocatalytic activity of the MoS2−2xSe2x modified HCS system. The composition of the MoS2−2xSe2x nanosheet components could be precisely tuned by adjusting the relative amounts of S and Se precursors.50 According to Yang et al., when the Se/S molar ratio was set to 0.1, the resulting C@MoS2−2xSe2x composite system exhibited the lowest overpotential and the smallest Tafel slope among the tested samples.50 As shown in the durability and operational stability related HER data in Fig. 9e, the polarization curve of the composites after 1500 cycles showed only a slight increase in onset overpotential, indicating that the rate of H2 production remains essentially unchanged with the catalytic behaviour of the material largely preserved.50 The time-dependent current density profile over 20 h (Fig. 9f) with only minimal decay demonstrated that the composite catalyst maintained stable activity in acidic media and remained robust at a constant applied potential.50
MoSSe modified HCS systems are also popular choices for lithium–sulfur battery applications. Graphitic carbon materials with hollow structures offer several advantages for lithium–sulfur batteries, including the ability to accommodate high sulfur loadings, buffer the substantial volume expansion of sulfur during cycling, and suppress the shuttling of lithium polysulfides (LiPSs).105,106 Elemental doping in this case can further enhance the performance of these carbon hosts by modulating their electronic band structures and density of states, improving the catalytic activity of the composites towards LiPSs conversion.51 Such doping also promotes the formation of the conductive 1T-MoS2 phase and introduces S vacancies, collectively enhancing charge transport and overall conversion kinetics. Zhang et al. demonstrated these principles by growing MoSSe nanosheets on N-doped HCSs (MoSSe/NC in Fig. 10) for use as both the S host and separator modification, enabling efficient catalytic adsorption and conversion of LiPSs.51 When employed as an S host, the MoSSe/NC composite system significantly improved electron and ion transport while mitigating S volume expansion, leading to accelerated redox kinetics. Based on the SEM (Fig. 10a and b) and TEM (Fig. 10c–e) observation, it was confirmed that the MoSSe/NC composites maintained a uniform, monodisperse hollow morphology, with MoSSe nanosheets (having a moderate loading amount to avoid excessive aggregation) evenly distributed across the HCS surfaces. The HRTEM analysis in Fig. 10f–h reveals a carbon shell of approximately 13.3 nm thick, which functions as both a mechanical buffer and a conductive framework to facilitate reaction kinetics and accommodate volume changes.51 The few layered MoSSe/NC composites having an interlayer spacing of 0.69 nm along the (002) plane (which was larger than that of pristine MoS2) showed expanded spacing that was beneficial for alleviating structural strain associated with Li+ insertion and contributed to the improved electrochemical stability of the composite system.51
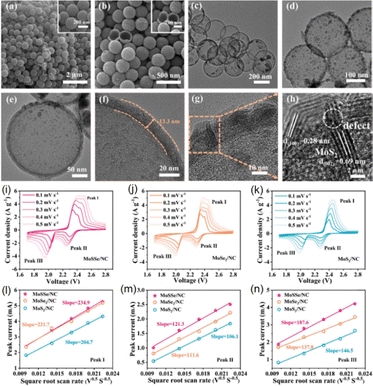 | ||
| Fig. 10 (a) and (b) SEM images of the MoSSe/NC system. (c)–(h) TEM images of MoSSe/NC collected at different magnifications. (i)–(k) CV curves of the MoSSe/NC, MoSe2/NC, and MoS2/NC composite systems at varying scan rates ranging from 0.1–0.5 mV s−1. (l)–(n) Logarithmic peak current versus logarithmic scan rate of the corresponding peaks. Reproduced with permission from ref. 51. Copyright 2023 Elsevier. | ||
The diffusion behaviour and oxidation kinetics of Li+ ions in the MoSSe/NC composite were also evaluated by Zhang and co-workers (Fig. 10i–k). The oxidation and reduction peak currents exhibited a linear dependence on the square root of the scan rate (Fig. 10l–n), demonstrating a diffusion-controlled electrochemical process. In this case, the Li+ diffusion rate was affected by variations in electrolyte viscosity arising from the dissolution of LiPSs and the subsequent deposition of Li2S2/Li2S on electrode surfaces.51 Due to the introduction of Se (together with expanded interlayer spacing and generated structural defects), the constructed MoSSe/NC system showed quite high Li+ diffusion rates across multiple reaction pathways. According to Zhang et al., the MoSSe/NC modified separator contributed to interfacial charge modulation and increased the number of exposed active sites, resulting in facilitated rapid anchoring and catalytic conversion of LiPSs with much improved uniform Li2S deposition.51
Co and Ni sulfide modified MoS2 composites on HCS as catalysts
Transition metal sulfides such as Co and Ni sulfides, along with their complexes, have attracted considerable interest as electrode materials for supercapacitors due to their high electrical conductivity and pronounced electrochemical activity. Because the crystal structures of MoS2 differ substantially from those of Ni- and Co-based sulfides, incorporating Ni or Co sulfides into MoS2 frameworks requires careful control to achieve a homogeneous distribution. Consequently, developing reliable synthetic strategies for Ni and Co sulfides involved systems remains a significant challenge. When combined with MoS2, the formation of heterostructures becomes essential due to the structural differences between the two components. Co- and Ni-based sulfides integrated with MoS2 have been widely investigated for their robust redox behaviour and enhanced electroactivity.107 For instance, among the various Co sulfides, Co9S8 has received particular attention because of its exceptionally high theoretical capacitance (5449 F g−1).108 Co9S8–MoS2 heterostructures, which leverage synergistic interactions between the two phases, have been reported to deliver promising performance in supercapacitor applications.109Liu et al. synthesized yolk–shell structured Co9S8–MoS2 nanosheet alloy modified HCSs composites (using SiO2 templates, Co9S8–MoS2 NSA@HCS, as shown in Fig. 11) with superior capacitive behaviour and cyclic stability in hybrid supercapacitors.7 The Co9S8–MoS2 precursor nanosheets were uniformly distributed across the HCS surface, exhibiting an average diameter of approximately 600 nm. According to Liu et al., the resulting Co9S8–MoS2 NSA@HCSs after sulfurization developed a nanoflower-like morphology, with nanosheets noticeably thinner than those of the original cobalt–molybdate silicate precursor.7 This Co9S8–MoS2 nanosheet array deposited HCS material with a constructed yolk–shell structure showed much enhanced electrical conductivity, cyclic durability and capacitive performance.7
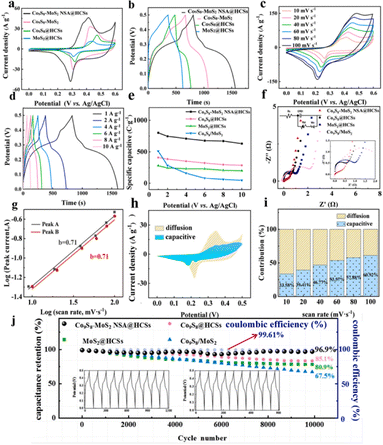 | ||
| Fig. 11 (a) CV curves of Co9S8 and MoS2 incorporated composite systems at 10 mV s−1; (b) GCD curves of Co9S8 and MoS2 incorporated composite systems at 1 A g−1; (c) CV curves at 10–100 mV s−1; (d) GCD curves at 1–10 A g−1; (e) capacity values obtained using GCD curves at different current densities. Data on (f) EIS curves and electrode equivalent circuits of the composite systems, (g) b-values, and (h) capacitive and diffusion-controlled contribution curves (at 10 mV s−1), and (i) relative contributions of capacitive and diffusion-controlled processes at various scan rates. (j) Comparison of cyclic stability and the corresponding coulombic efficiency at a current density of 10 A g−1. Reproduced with permission from ref. 7. Copyright 2024 Elsevier. | ||
Some of the detailed electrochemical evaluation data collected by Liu et al. on the Co9S8–MoS2 NSA@HCSs electrode is presented in Fig. 11. As shown in Fig. 11a, the Co9S8–MoS2@HCSs composite exhibited two pairs of redox peaks at 0.37/0.29 V and 0.40/0.29 V, which were shifted to lower potentials compared to that of Co9S8–MoS2.7 This decrease in redox peak potentials was associated with the incorporation of the highly conductive HCSs substrate, which significantly facilitated charge transport of the composite system.7 The redox features arising from the reversible oxidation and reduction of Co2+/Co3+ and Mo3+/Mo4+ in the presence of OH− can be described by eqn (4)–(7) below.7
| MoS2 + OH− ↔ MoS2OH + e− | (4) |
| MoS2OH + OH− ↔ MoS2O + H2O + e− | (5) |
| Co9S8 + OH− ↔ Co9S8OH + e− | (6) |
| Co9S8OH + OH− ↔ Co9S8O + H2O + e− | (7) |
In this case, the composite electrode showed the largest CV curve area, indicating its highest specific capacity and superior energy storage capability among all relevant composite samples.7 Consistently, the GCD profiles in Fig. 11b show that the Co9S8–MoS2NSA@HCS electrode delivered the longest discharge time, confirming its maximum specific capacity. The enhanced electrochemical activity of the composite system was further supported by the data presented in Fig. 11c–i, with the Nyquist plots (Fig. 11f) having the charge transport resistance (Rct) corresponding to the semicircle radius in the high-frequency region and the intercept at the x-axis representing the solution resistance (Rs) between the electrolyte and electrode.7 Based on the cyclic stability of the material evaluated at the current density of 10 A g−1 (Fig. 11j), the Co9S8–MoS2 NSA@HCSs electrode retained 96.9% of its initial capacity after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles with negligible distortion in curve shape.7 It was clear that integrating Co9S8 and MoS2 onto HCSs could significantly enhance the electrochemical activity of the composite system for energy conversion/storage applications.
000 charge–discharge cycles with negligible distortion in curve shape.7 It was clear that integrating Co9S8 and MoS2 onto HCSs could significantly enhance the electrochemical activity of the composite system for energy conversion/storage applications.
Among the variety of transition metal candidates, Ni incorporated materials are often considered hot choices for electrocatalyst fabrication due to their reliable catalytic properties. Layered NiS2, in particular, shares structural similarities with MoS2 and has therefore been frequently investigated for constructing NiS2/MoS2 hybrid systems for use in energy storage/conversion and related fields. Luan et al. reported the fabrication of a NiS2/MoS2 hetero-nanosheet modified double-layer HCS material with excellent HER activity.65 To preserve the nanosheet morphology, Luan et al. assembled the Ni(OH)2 nanosheets first onto DLHCs via hydrolysis of the nickel salts. The subsequent sulfurization of these Ni(OH)2 nanosheets occurred concurrently with MoS2 formation, leading to the in situ generation of NiS2/MoS2 composite nanosheets, with the resulting DLHCs@NiS2/MoS2 heterostructures (Fig. 12) exhibiting outstanding HER performance across a wide pH range.65 Based on the TEM data (Fig. 12a–c) collected for the DLHCs@NiS2/MoS2 heterostructures synthesized using optimized parameters (DLHCs@NiS2/MoS2-6), the average spacing between the dual spherical carbon shells is approximately 15 nm. As shown in Fig. 12c, the formation of NiS2/MoS2 heterojunctions was evident with quite large ultrathin NiS2/MoS2 nanosheets uniformly coated onto the surfaces of the DLHCs. The measured interlayer spacing of 0.65 nm corresponded to the (002) plane of 2H-MoS2, and the lattice fringes with a spacing of 0.25 nm were assigned to the (210) plane of NiS2.65,110 The outer carbon shell exhibiting uniformly distributed mesopores was crucial for providing abundant nucleation sites for heterojunction growth, with a well controlled efficient growth process leading to homogeneous distribution of NiS2 and MoS2 on the DLHC base.
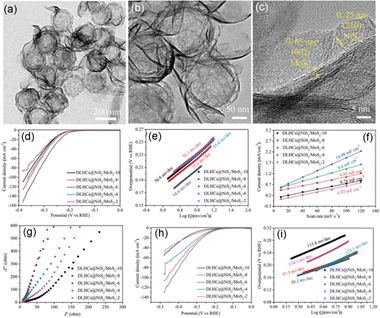 | ||
| Fig. 12 (a)–(c) TEM images of the DLHCs@NiS2/MoS2-6 composites. (d) HER polarization curves of the samples measured in 0.5 M H2SO4. (e) Corresponding Tafel plots derived from panel (d). (f) Double-layer capacitance (Cdl) values of the samples. (g) Electrochemical impedance spectra. (h) HER polarization curves obtained in 1 M KOH. (i) Corresponding Tafel plots derived from panel (h). Reproduced with permission from ref. 65. Copyright 2022 Wiley-VCH GmbH. | ||
Similar to other MoS2 involved catalyst systems, the HER pathway of this DLHCs@NiS2/MoS2 composite reported by Luan et al. primarily followed the Volmer–Tafel and Volmer–Heyrovsky mechanisms.13,64,65 As shown in the LSV curves in Fig. 12d, the DLHCs@NiS2/MoS2 electrode exhibited the lowest overpotential among all the tested composite systems. The corresponding Tafel slopes (Fig. 12e) increased with higher MoS2 loading, accompanied by a decline in HER activity, demonstrating that the incorporation of NiS2 played the key role in enhancing the catalytic performance of the composite system. This improvement was attributed to the increased number of exposed active sites introduced by the NiS2 components. Moreover, the S-edge sites of MoS2 nanosheets served as the primary catalytic centres for HER, and excessive MoS2 loading led to nanosheet agglomeration, thereby reducing their accessibility. The formation of a well-defined heterojunction between MoS2 and NiS2 also modulated the electronic structure, facilitated charge transport and promoted more efficient water dissociation of the material, collectively contributing to the observed superior HER activity of the composite system.65
The reaction kinetics at the electrode–electrolyte interface during the HER were further examined by Luan et al.65 The EIS spectra shown in Fig. 12g indicate that the DLHCs@NiS2/MoS2-6 composites exhibited the steepest slope in the low-frequency region, reflecting its superior electrochemical kinetics for the HER. The catalytic performance in alkaline media is presented in Fig. 12h and i. The DLHCs@NiS2/MoS2-6 system with optimized parameters showed the lowest onset potential (139 mV at 1 mA cm−2) and the smallest overpotential (230 mV at 10 mA cm−2). As the MoS2 loading increased, the Tafel slope decreased from 134.1 mV dec−1 (DLHCs@NiS2/MoS2-2) to 88.2 mV dec−1 (DLHCs@NiS2/MoS2-6), suggesting a shift in the rate-determining step from the Volmer step to the Heyrovsky step.65 However, excessive MoS2 loading led to performance deterioration, likely due to nanosheet agglomeration and reduced accessibility to active sites.
Ni and Co sulphides on HCSs as catalysts
Except for the MoS2 involved composite systems, a variety of Ni and Co sulfide components have also been directly employed to modify HCSs due to their excellent electrochemical properties, for instance, Ni sulphides exhibiting outstanding H2 generation activity and structural motifs that resemble the active centres of hydrogenase enzymes.111 Various Ni sulfide phases, including NiS, NiS2, Ni3S2, Ni3S4, and Ni7S6, have therefore emerged as promising electrocatalyst options for energy storage/conversion applications, especially when integrated with HCS substrates. Reddy et al. synthesized a core–shell HCS@nickel–sulfide nanocomposite that achieved a high specific capacitance of 1022 F g−1 at 1 A g−1.112 Recently, Liu and co-workers reported Ni sulfide composites supported on DLHCs for the HER, achieving a quite low onset overpotential of 162 mV, a Tafel slope of 88.23 mV dec−1, and a high specific capacitance of 1160 F g−1 at 1 A g−1 (Fig. 13, constructed using sol–gel-derived SiO2 templates, with the synthetic route illustrated in Fig. 13a).36 Furthermore, an asymmetric supercapacitor device was assembled by Liu et al. using DLHCs@NiS/Ni3S4 as the positive electrode and activated carbon as the negative electrode. This device delivered a specific capacitance of 306 F g−1 at 1 A g−1 and retained 78.4% of its capacitance when the current density increased to 10 A g−1, demonstrating excellent rate capabilities.36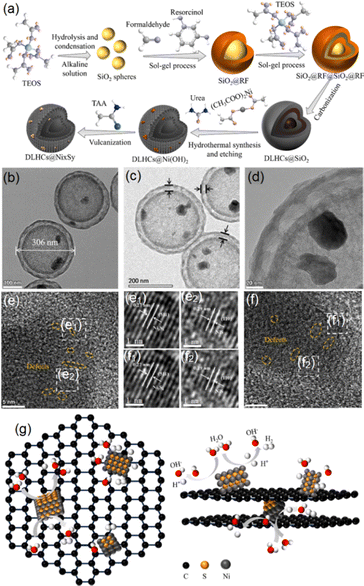 | ||
Fig. 13 Synthetic procedures (a) and TEM data at different magnifications (b)–(f) of the DLHCs@NixSy-1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 composite system: (e1) and (e2) enlarged areas circled in (e); (f1) and (f2) enlarged areas circled in (f); with orange dotted lines showing the structural defects. (g) HER mechanism proposed for the DLHCs@NixSy system in an alkaline environment. Reproduced with permission from ref. 36. Copyright 2024 Elsevier. 2 composite system: (e1) and (e2) enlarged areas circled in (e); (f1) and (f2) enlarged areas circled in (f); with orange dotted lines showing the structural defects. (g) HER mechanism proposed for the DLHCs@NixSy system in an alkaline environment. Reproduced with permission from ref. 36. Copyright 2024 Elsevier. | ||
According to Liu et al., the Ni sulfide components were deposited onto a double-layer HCS through hydrothermal reaction, producing a uniform and tightly adhered NiS/Ni3S4 coating, as shown in the TEM images in Fig. 13b–f.36 The further introduction of SiO2 precursors was essential for generating the characteristic double-layer carbon shells. During the following alkaline treatment, the SiO2 was etched away with Ni(OH)2 simultaneously deposited onto the carbon framework. This process preserved the mechanically robust double-layer hollow carbon structures, which functioned both as a conductive backbone and as a stable platform for anchoring the active Ni sulfide phases. High-resolution TEM images (Fig. 13e and f) show clear lattice spacings of 0.39 nm and 0.27 nm, corresponding to the (202) plane of NiS and the (511) plane of Ni3S4, with the clear boundary between these two sets of lattice fringes confirming the formation of the NiS/Ni3S4 heterointerfaces. As schematically illustrated in Fig. 13g, in this system, the highly conductive carbon shells acted as an efficient platform for anchoring NixSy species and enabled rapid electron transport from the graphitic carbon framework to the catalytic Ni sulfide domains where H2 generation took place. Previous studies have also demonstrated that Ni-based active sites can accelerate the initial water dissociation step while assisting in the subsequent removal of OH− species, improving the overall reaction kinetics.36,113,114 This DLHCs@NiS/Ni3S4 material were also used by Liu and co-workers for constructing an asymmetric supercapacitor that could deliver an energy density of 40 Wh kg−1 at a power density of 375 W kg−1, exhibiting strong durability in both the HER and supercapacitor testing.36
In addition, Ni–Co mixed sulfides have also emerged as a promising class of high-performance electrocatalysts, benefiting from the intrinsic catalytic activity of both Ni and Co. Since the HER and OER often require different active site environments and reaction pathways, the target composite catalysts must be tailored to exhibit distinct selectivity for each process.115,116 Besides, achieving exceptionally high performance with non-noble metal components remains challenging, largely due to interfacial limitations within nanosheets, nanotubes, and nanoneedles, each of which composite structures. Careful control over morphology and the deliberate design of nanostructured catalysts are thus essential strategies for improving catalytic behavior.117,118 In general, Ni–Co sulfides can be synthesized in various morphologies with significantly different catalytic responses. A recent example involved the growth of Ni–Co sulfides on a DLHC substrate, constructing NiCo2S4@DC composites using a hydrothermal method.96 The resulting NiCo2S4 hollow spherical tube structures contained internal cavities that helped buffer mechanical stress from volume changes during operation, while their high surface area increased the interfacial contact between the electrolyte and the active materials, facilitating efficient charge transport and enhancing catalytic performance.
Fig. 14a outlines the detailed synthetic strategy used to construct a Ni–Co mixed sulphide incorporated hybrid catalyst (NiCo2S4@DCs, with NiCo2S4 directly grown onto hollow double-layered carbon nanospheres (DCs)) developed by Yang's group.96 In this approach, the sol–gel-derived SiO2 spheres acted as the sacrificial templates.96 A resorcinol-formaldehyde polymer layer was deposited onto the SiO2 cores (SiO2@RF), followed by coating, carbonization, and template removal steps to fabricate the DLHC base. Moreover, NiCo(CO3)(OH)2 was deposited onto the carbon spheres and converted into the NiCo2S4 phase through reaction with sulfide ions achieving uniform dispersion of NiCo2S4 components and the formation of well-defined interfaces with the carbon framework. In this case, the hydrothermal treatment was crucial for the nucleation and direct growth of NiCo2S4 nanotubes onto DLHC surfaces. The TEM images of the constructed NiCo2S4@DC composites (Fig. 14b–d) demonstrated that the NiCo2S4 formed porous hollow tubular structures that distributed irregularly around the carbon base. The confined geometry of the HCSs restricted the longitudinal growth of NiCo2S4 tubes, resulting in their characteristic short tube morphology. The TEM data further confirmed the crystalline nature of this active phase, showing lattice spacings of 0.23 nm and 0.28 nm corresponding to the (400) and (311) planes of NiCo2S4.96
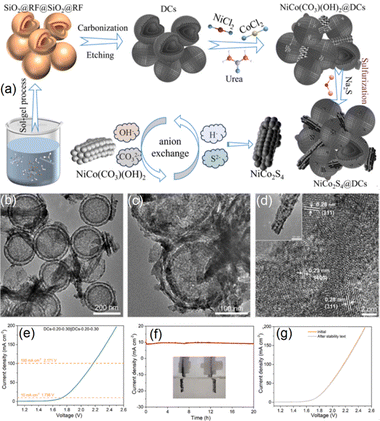 | ||
| Fig. 14 (a) Schematic illustration of the synthesis of NiCo2S4@DCs. (b)–(d) TEM data of the composite system. (e) LSV curve of the composites recorded in 1 M KOH. (f) Chronoamperometry measurement at 1.736 V for 20 h in 1 M KOH; the inset depicts the two-electrode operating configuration. (g) LSV data collected before/after the chronoamperometry test. Reproduced with permission from ref. 96. Copyright 2024 Elsevier. | ||
According to Yang et al., the Ni/Co precursor ratios were varied to synthesize a series of composites (DCs-x-y with x and y denoting the molar ratios of the Ni and Co precursors). The high performance of the optimized composition (DCs-0.20-0.30) arose from the high surface area and monodisperse HCSs, which promoted uniform NiCo2S4 distribution, improved electrical conductivity, and increased the number of accessible active sites. The NiCo2S4@DCs system exhibited reasonably high HER and OER activity in alkaline media and delivered excellent overall water splitting performance with excellent operational stability (for 20 h at 10 mA cm−2), requiring only 1.736 V to reach 10 mA cm−2 (Fig. 14e–g). The observed HER activity of this composite system in acidic media was superior to that observed in alkaline media, with the catalytic activity of the optimized NiCo2S4@DC system surpassing that of bare DCs, NiCo2S4, NixSy@DCs, and CoxSy@DCs across all tested conditions. These highlight the potential of such Ni–Co sulphide hybrid structures to guide the rational design of electrocatalysts optimized for different electrolyte environments in both H2 generation and overall water splitting. To facilitate a clear comparison of HER performance among the TMD decorated HCSs composites, Table 2 summarizes the overpotentials and Tafel slopes of some selected examples reported in the literature. The electrochemical activity of TMDs is highly sensitive to their synthesis conditions and spatial distribution, both of which critically influence their catalytic behavior.119
| Sample | Overpotential (mV/at 10 mA cm−2) | Tafel slope (mV dec−1) | Ref. |
|---|---|---|---|
| MoS2@HCSs | 207.0 | 62.0 | 64 |
| MoS2@ HCSs | 162.0 | 44.0 | 119 |
| P-MoS2@N-HCSs | 117.0 | 68.0 | 37 |
| MoS2@DLHCs | 174.4 | 74.7 | 35 |
| MoS2@HCSs | 190.0 | 79.9 | 50 |
| MoSSe@HCSs | 186.0 | 53.2 | 50 |
| NiS2/MoS2@DLHCs | 139.0 | 54.6 | 65 |
| NiS/Ni3S4@DLHCs | 162.0 | 88.2 | 36 |
| NiCo2S4@ DLHCs | 206.0 | 71.0 | 96 |
| Ni-MoS2@CNTs | 158 | 62.3 | 18 |
Challenges and perspectives
This review has examined strategies for controlling the growth of TMDs on HCSs to develop high-performance catalysts for sustainable energy storage and conversion, with particular emphasis on applications in water splitting and supercapacitors. We first summarized advances in HCS engineering, including hard-template synthesis, shell-thickness modulation, and non-metal heteroatom doping that enable precise structural control. The subsequent sections discussed the integration of MoS2 nanosheets with HCSs, followed by an analysis of how Se, Ni, and Co incorporation modifies the catalytic behaviour of MoS2/HCSs composites. Beyond MoS2 and MoSe2 systems, recent progress on Ni- and Co-sulfide/HCS hybrids was also highlighted, with attention to their catalytic performance and structural advantages. Throughout the review, we emphasized synthesis kinetics, heterostructure formation, and interfacial characteristics, all of which play critical roles in determining catalytic activity. Despite the substantial number of studies published on TMD/HCS composites, significant challenges remain. The pronounced differences in structure, bonding, and physicochemical properties between TMDs and carbon-based hosts mean that both fundamental understanding and practical implementation are still developing. Key research directions and remaining challenges for advancing these composite catalysts are outlined below.(i) Precise control over the shell thickness and structure of HCSs remains a long-standing challenge. Although hard template methods have been employed for decades, the template removal step is still hard to regulate with precision, often leading to variability in shell thickness. Residual template or precursor species can negatively influence catalytic performance and hinder scalability. Non-metal doping has been explored to introduce additional active sites, yet achieving uniform incorporation of dopant atoms throughout the carbon framework remains difficult. These limitations highlight the need for new synthetic strategies capable of delivering precise structural control, homogeneous doping, and improved catalytic activity.
(ii) TMD modified catalysts exhibit diverse crystal structures and bonding environments, making composite formation inherently complex. The formation of TMD composites often requires multistep or delicate synthesis routes, and further progress will depend on design strategies grounded in solid theoretical understanding. Because TMDs can form alloys, composite phases, homojunctions, and heterostructures, the spatial arrangement and interaction of each component strongly influence the final catalytic behavior. Progress in new synthetic methodologies, computational modelling, mechanism study, and advanced in-situ characterization will be essential for establishing clear structure–property relationships and guiding the rational design of next-generation TMDs incorporated catalysts.
(iii) Interface engineering remains a central but underdeveloped frontier. Interface engineering represents a crucial avenue for constructing TMD-based heterostructures and achieving high catalytic efficiency. For example, MoS2/MoSe2 heterostructures supported on HCSs involve multiple interfacial interactions, between MoS2 and MoSe2, between each TMD and the HCS hosts, and across the combined junctions. The quality and continuity of these interfaces largely determine the resulting catalytic performance of the material. Redox behavior, band gap alignment, and charge carrier transport within the composite system are all strongly governed by interfacial characteristics. Consequently, deeper investigation into interface formation, optimization, and stabilization is essential for advancing the TMD-HCS hybrid systems.
(iv) Long-term operational stability of e.g., HER and OER processes remains a central concern, as maintaining catalytic integrity under harsh electrochemical conditions is essential for real world water splitting and energy storage applications. Although the porous nanostructures of TMD-HCSs effectively immobilizes TMDs and mitigates aggregation or corrosion, further optimization is required to ensure consistent performance over extended cycling. Another challenge lies in maximizing interfacial electronic coupling. While current designs enhance active site exposure and charge transport pathways, the overall spatial utilization within the composite structure is still not fully exploited. Achieving more efficient electron/ion transport networks will be crucial for advancing both water splitting and supercapacitor technologies.120,121 Looking forward, the development of next-generation TMD–HCS nanostructures would depend on the strategic incorporation of new functional components. Improving energy conversion efficiency, particularly under practical operating conditions, remains a key objective. Emerging concepts, such as photothermal-responsive electrocatalysts engineered through porogen-mediated structural design, offer promising directions, with these dual-function systems integrating photothermal conversion with electrocatalysis, enabling broad spectrum light harvesting and significantly enhanced electrochemical activity. Future research could focus on e.g., refining structural stability, deepening interfacial coupling, and exploring multifunctional structures capable of leveraging external stimuli. Such advances will be essential for translating TMD-HCSs composites from laboratory demonstrations into scalable, high-performance energy conversion technologies.
Author contributions
Xiao Zhang: conceptualization, data curation, formal analysis, investigation, methodology, writing – original draft, funding acquisition, project administration, and writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was partly supported by JSPS KAKENHI Grant Number JP25K23518.Notes and references
- X. Zhang and P. Yang, Phys. Chem. Chem. Phys., 2025, 27, 25254–25265 RSC.
- W. Yan, Y. Mou, M. Li, K. Ma, Z. Xu, T. Lu, H. Du, C. Wang, H. Sun, L. Chen, Y. Tang, Y. Wang and G. Fu, Adv. Mater., 2025, 37, 2506936 Search PubMed.
- X. R. Zhang, Y. Lyu, J. Ding, X. Wang, B. Johannessen, S. P. Jiang and J. Zheng, Sci. Adv., 2025, 11, eadr8651 CrossRef CAS.
- X. Zhang, X. Xia and P. Yang, Environ. Sci.: Nano, 2024, 11, 4186–4195 RSC.
- X. Zhang, X. Ji and P. Yang, Appl. Surf. Sci., 2026, 723, 165666 CrossRef CAS.
- X. Wang, J. Hu, T. Lu, H. Wang, D. Sun, Y. Tang, H. Li and G. Fu, Angew. Chem., Int. Ed., 2025, 64, e202415306 CrossRef CAS.
- Q. Liu, Y. Sun, Y. Xie, F. Yao, X. Gao, H. Bai, K. Zhang, R. Liu and H. Yue, J. Power Sources, 2024, 605, 234537 CrossRef CAS.
- X. R. Zhang, Y. Lyu, C. Chen, J. Zheng, S. P. Jiang and S. Wang, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2311326121 CrossRef CAS PubMed.
- X. Zhang and P. Yang, Langmuir, 2025, 41, 7191–7211 CrossRef CAS.
- C. Wang, X. Zhang, Y. Zhu, K. Zhu, X. Luan and P. Yang, Electrochim. Acta, 2023, 454, 142386 CrossRef CAS.
- S. Huang, D. Ma, S. Zhai, K. Chen, W. Lai, W. Xiao, H. Liu, Z. Su, J. Zeng and Y. Tong, J. Power Sources, 2024, 598, 234195 CrossRef CAS.
- X. Zhang, X. Zhang, P. Yang and S. P. Jiang, J. Electroanal. Chem., 2021, 895, 115510 Search PubMed.
- X. Zhang, P. Yang and S. P. Jiang, Carbon, 2021, 175, 176–186 CrossRef CAS.
- X. Wang, M. Li, Y. Tang, H. Li and G. Fu, Prog. Mater. Sci., 2026, 155, 101539 CrossRef CAS.
- H. Lv, S. Wang, Z. Xiao, C. Qin, S. Zhai, G. Wang, Z. Zhao and Q. An, J. Power Sources, 2021, 493, 229679 Search PubMed.
- J. Chen, Y. Guo, Y. Wen, L. Huang, Y. Xue, D. Geng, B. Wu, B. Luo, G. Yu and Y. Liu, Adv. Mater., 2013, 25, 992–997 CrossRef CAS.
- Y. Liu, X.-Y. Yu, Y. Fang, X. Zhu, J. Bao, X. Zhou and X. W. Lou, Joule, 2018, 2, 725–735 CrossRef CAS.
- T. Dong, X. Zhang, P. Wang, H. S. Chen and P. Yang, Electrochim. Acta, 2020, 338, 135885 CrossRef CAS.
- X. Zhang, P. Yang and S. P. Jiang, Int. J. Hydrogen Energy, 2021, 46, 2065–2074 CrossRef CAS.
- Q. Shen, Z. Jiang, X. Tian, H. Zhu and Z. J. Jiang, J. Alloys Compd., 2021, 884, 161073 Search PubMed.
- F. Yang, W. Qiao, L. Yu, S. Wang and L. Feng, Chem. Eng. J., 2024, 483, 149055 CrossRef CAS.
- Z. J. Jiang, G. Xie, B. Deng and Z. Jiang, Electrochim. Acta, 2018, 285, 103–110 Search PubMed.
- Y. Kuang, W. Qiao, F. Yang and L. Feng, J. Energy Chem., 2023, 85, 447–454 CrossRef CAS.
- W. Chen, R. Qiao, C. Song, L. Zhao, Z. J. Jiang, T. Maiyalagan and Z. Jiang, J. Catal., 2020, 381, 363–373 Search PubMed.
- J. Q. Chi, W. K. Gao, J. H. Lin, B. Dong, K. L. Yan, J. F. Qin, B. Liu, Y. M. Chai and C. G. Liu, Catal. Today, 2019, 330, 259–267 Search PubMed.
- X. Zhang and P. Yang, Int. J. Miner., Metall. Mater., 2024, 31, 2368–2389 Search PubMed.
- G. Ghanashyam and H. Kim, J. Power Sources, 2024, 596, 234088 CrossRef CAS.
- T. Wang, X. Zhang, P. Yang and S. P. Jiang, Inorg. Chem. Front., 2020, 7, 3578–3587 Search PubMed.
- T. Dong, X. Zhang, P. Wang, H. S. Chen and P. Yang, Carbon, 2019, 149, 222–233 CrossRef CAS.
- T. Dong, X. Zhang, Y. Cao, H. S. Chen and P. Yang, Inorg. Chem. Front., 2019, 6, 1073–1080 RSC.
- Y. Zhang, X. Zong, L. Zhan, X. Yu, J. Gao, C. Xun, P. Li and Y. Wang, Electrochim. Acta, 2018, 284, 89–97 Search PubMed.
- L. Yu, X. He, L. Tang, X. Wang, W. Cai, Y. He, Y. Sun, G. Zhu, J. Rosen and H. Jin, J. Power Sources, 2025, 641, 236852 CrossRef CAS.
- X. Wei, X. Li, C. Lv, X. Mo and K. Li, Electrochim. Acta, 2020, 354, 136590 CrossRef CAS.
- H. Liao, H. Zhang, H. Hong, Z. Li and Y. Lin, Electrochim. Acta, 2017, 257, 210–216 CrossRef CAS.
- X. Luan, K. Zhu, X. Zhang and P. Yang, Electrochim. Acta, 2022, 407, 139929 CrossRef CAS.
- C. Liu, X. Zhang, Y. Cheng, X. Hao and P. Yang, Electrochim. Acta, 2024, 477, 143751 CrossRef CAS.
- Y. Wei, X. Zhang, Z. Zhao, H. S. Chen, K. Matras-Postolek, B. Wang and P. Yang, Electrochim. Acta, 2019, 297, 553–563 CrossRef CAS.
- Z. Chen, R. Cao, Y. Ge, Y. Tu, Y. Xia and X. Yang, J. Power Sources, 2017, 363, 356–364 Search PubMed.
- Y. Sun, Y. Shi, X. Zhang, F. Cao, L. Huang and Y. Chen, Carbon, 2025, 240, 120352 Search PubMed.
- W. Shi, X. Zhang, Y. Tian and P. Yang, J. Environ. Chem. Eng., 2024, 12, 114018 Search PubMed.
- S. Wang, X. Zhang, C. Li, Y. Tian and P. Yang, Int. J. Hydrogen Energy, 2024, 90, 853–864 CrossRef CAS.
- C. Wang, X. Wu, H. Sun, Z. Xu, C. Xu, X. Wang, M. Li, Y. Wang, Y. Tang, J. Jiang, K. Sun and G. Fu, Energy Environ. Sci., 2025, 18, 4276–4287 RSC.
- Y. Ma, S. Luo, M. Tian, J. E. Lu, Y. Peng, C. Desmond, Q. Liu, Q. Li, Y. Min, Q. Xu and S. Chen, J. Power Sources, 2020, 450, 227659 Search PubMed.
- S. Tong, X. Zhang and P. Yang, Environ. Res., 2023, 236, 116805 CrossRef CAS PubMed.
- C. Li, X. Zhang, T. Song, Y. Tian, S. Wang and P. Yang, J. Environ. Chem. Eng., 2024, 12, 113396 CrossRef CAS.
- Y. Chen, G. Meng, T. Yang, C. Chen, Z. Chang, F. Kong, H. Tian, X. Cui, X. Hou and J. Shi, J. Chem. Eng., 2022, 450, 138157 Search PubMed.
- H. Shi, Y. Zhang, N. Pang, S. Xu, D. Xiong, L. Wang, P. Yang and P. K. Chu, Electrochem. Acta, 2023, 439, 141596 Search PubMed.
- C. Li, Y. Tian, S. Wang, P. Yang and X. Zhang, J. Environ. Chem. Eng., 2024, 12, 114170 CrossRef CAS.
- X. Zhang, K. Zhu, C. Xie and P. Yang, Carbon, 2024, 220, 118884 CrossRef CAS.
- X. Luan, K. Zhu, X. Zhang and P. Yang, Langmuir, 2021, 37(27), 8314–8322 CrossRef CAS.
- J. Zhang, W. Xi, P. Du, Z. Xie, Y. Zhang, R. Wang, B. He, Y. Gong, H. Wang and J. Jin, Electrochim. Acta, 2023, 471, 143385 CrossRef CAS.
- X. Li, S. Bai, Z. Zhu, J. Sun, X. Jin, X. Wu and J. Liu, Langmuir, 2017, 33, 1248–1255 CrossRef CAS PubMed.
- L. Wang, L. Rao, B. Xia, L. Wang, L. Yue, Y. Liang and X. Hu, Carbon, 2018, 130, 31–40 CrossRef.
- D. Bin, Z. Chi, Y. Li, K. Zhang, X. Yang, Y. G. Sun and L. Wan, J. Am. Chem. Soc., 2017, 139, 13492–13498 Search PubMed.
- D. Zhu, Y. Wang, X. Yang, H. Wang, Y. Xu, Z. Wang, Y. Tian and H. Lu, Electrochim. Acta, 2025, 542, 147427 CrossRef.
- J. Du, Y. Zhang, H. Wu, S. Hou and A. Chen, Carbon, 2020, 156, 523–528 CrossRef.
- D. Khalafallah, X. Quan, C. Ouyang, M. Zhi and Z. Hong, Renew. Energy, 2021, 170, 60–71 CrossRef.
- J. Du, S. Zong, Y. Zhang, S. Hou and A. Chen, J. Colloid. Interface Sci., 2020, 565, 245–253 CrossRef PubMed.
- L. Zhou, Z. Zhuang, H. Zhao, M. Lin, D. Zhao and L. Mai, Adv. Mater., 2017, 29, 1602914 CrossRef PubMed.
- A. Fu, C. Wang, F. Pei, J. Cui, X. Fang and N. Zheng, Small, 2019, 15, 1804786 Search PubMed.
- H. Tian, J. Liang and J. Liu, Adv. Mater., 2019, 31, 1903886 Search PubMed.
- D. P. Li, Y. C. Sun, X. Wang, S. Wu, S. C. Han and Y. Yang, RSC Adv., 2017, 7, 37983–37989 RSC.
- L. L. Han, W. W. Wu, C. Yuan, Z. Wang, X. B. Zhou, X. M. Chen and P. Liu, J. Mater. Chem. C, 2023, 11, 5534–5545 RSC.
- C. Liu, P. Yang and X. Zhang, Int. J. Hydrogen Energy, 2025, 97, 1348–1356 CrossRef.
- X. Luan, K. Zhu, C. Wang, W. Shi, A. Szymska, K. Matras-Postolek and P. Yang, Adv. Mater. Interfaces, 2022, 2201661 CrossRef.
- H. Zhang, B. Wang, A. Feng, N. Zhang, Z. Jia, Z. Huang, X. Liu and G. Wu, Composites, Part B, 2019, 167, 690–699 Search PubMed.
- X. Wang, J. I. Feng, Y. Bai, Q. Zhang and Y. Yin, Chem. Rev., 2016, 116, 10983 Search PubMed.
- Z. Li, D. Wu, Y. Liang, R. Fu and K. Matyjaszewski, J. Am. Chem. Soc., 2014, 136, 4805–4808 CrossRef PubMed.
- S. Tanaka, H. Nakao, T. Mukai, Y. Katayama and Y. Miyake, J. Phys. Chem. C, 2012, 116, 26791–26799 CrossRef.
- X. L. Fang, J. Zang, X. L. Wang, M. S. Zheng and N. F. Zheng, J. Mater. Chem. A, 2014, 2, 6191–6197 RSC.
- M. Sevilla, P. Valle-Vigon and A. B. Fuertes, Adv. Funct. Mater., 2011, 21, 2781–2787 CrossRef.
- K. Wang, L. Huang, S. Razzaque, S. Jin and B. Tan, Small, 2016, 23, 3134–3142 CrossRef PubMed.
- C. Q. Yuan, X. H. Liu, M. Y. Jia, Z. X. Luo and J. N. Yao, J. Mater. Chem. A, 2015, 3, 3409–3415 RSC.
- X. L. Fang, J. Zang, X. L. Wang, M. S. Zheng and N. F. Zheng, J. Mater. Chem. A, 2014, 2, 6191–6197 RSC.
- Y. H. Dai, H. Jiang, Y. J. Hu, Y. Fu and C. Z. Li, Ind. Eng. Chem. Res., 2014, 53, 3125–3130 CrossRef.
- J. Wang, L. F. Shen, B. Ding, P. Nie, H. F. Deng, H. Dou and X. G. Zhang, RSC Adv., 2014, 4, 7538–7544 RSC.
- Q. G. Shao, J. Tang, Y. X. Lin, F. F. Zhang, J. S. Yuan, H. Zhang, N. Shinya and L. C. Qin, J. Mater. Chem. A, 2013, 1, 15423–15428 RSC.
- J. P. Han, G. Y. Xu, B. Ding, J. Pan, H. Dou and D. R. MacFarlane, J. Mater. Chem. A, 2014, 2, 5352–5357 RSC.
- M. C. Hsiao, C. Y. Chang, L. J. Niu, F. Bai, L. J. Li, H. H. Shen, J. Y. Lin and T. W. Lin, J. Power Sources, 2017, 345, 156–164 CrossRef.
- J. Liu, Y. Pan, L. Yu, Z. Gao, S. Zhang, D. Lan, Z. Jia and G. Wu, Carbon, 2025, 238, 120233 CrossRef.
- X. Xu, W. Zhong, M. Chen, L. Zhang, G. Liu and Y. Du, Int. J. Hydrogen Energy, 2021, 46, 8567–8577 CrossRef.
- D. C. Nguyen, T. L. L. Doan, S. Prabhakaran, D. T. Tran, D. H. Kim, J. H. Lee and N. H. Kim, Nano Energy, 2021, 82, 105750 CrossRef.
- S. Gao, S. Chen, F. Shi, W. Jiang and J. Chen, J. Power Sources, 2024, 591, 233866 CrossRef.
- J. Cheon, J. E. Gozum and G. S. Girolami, Chem. Mater., 1997, 9, 1847–1853 CrossRef CAS.
- Q. Li, J. T. Newberg, E. C. Walter, J. C. Hemminger and R. M. Penner, Nano Lett., 2004, 4, 277–281 CrossRef CAS.
- Y. Tian, X. Zhao, L. Shen, F. Meng, L. Tang, Y. Deng and Z. Wang, Mater. Lett., 2006, 60, 527–529 CrossRef CAS.
- S. J. Lee, Y. S. Son, J. H. Choi, S. S. Kim and S. Y. Park, Catalysts, 2021, 11, 1229 CrossRef CAS.
- F. Yang, H. Huang, J. Zhu, W. Dong, Q. Wu and H. Li, J. Mater. Sci.: Mater. Electron., 2022, 33, 21677–21687 CrossRef CAS.
- Y. Zhang, T. He, G. Liu, L. Zu and J. Yang, Nanoscale, 2017, 9, 10059–10066 RSC.
- S. G. Mohamed, I. Hussain and J. J. Shim, Nanoscale, 2018, 10, 6620–6628 RSC.
- S. J. Lee, Y. S. Son, J. H. Choi, S. S. Kim and S. Y. Park, Catalysts, 2021, 11, 1229 CrossRef CAS.
- W. Li, X. Xu, Y. Yang, Y. Huang, J. Jiang, W. Liu, M. Jing, Y. Bai, Y. Yang, T. Wu and X. Wang, J. Alloys Compd., 2023, 934, 167838 CrossRef CAS.
- X. Zhang, P. Yang and S. P. Jiang, J. Power Sources, 2021, 510, 230420 CrossRef CAS.
- J. Li, A. Listwan, J. Liang, F. Shi, K. Li and J. Jia, J. Chem. Eng., 2022, 422, 130100 CrossRef.
- K. Nie, N. Li, B. Li, Y. Yuan, Y. Zhang, P. Liu, S. Chong, J. Hu, Z. Liu and W. Huang, J. Chem. Eng., 2023, 475, 146066 CrossRef CAS.
- C. Liu, X. Zhang, X. Hao and P. Yang, J. Alloys Compd., 2024, 1002, 175110 Search PubMed.
- L. Ye, S. Chen, W. Li, M. Pi, T. Wu and D. Zhang, J. Phys. Chem. C, 2015, 119, 9560–9567 CrossRef CAS.
- H. Li, Q. Zhang, C. C. R. Yap, B. K. Tay, T. H. T. Edwin, A. Olivier and D. Baillargeat, Adv. Funct. Mater., 2012, 22, 1385–1390 CrossRef CAS.
- Y. Yang, X. Liu, Z. Zhu, Y. Zhong, Y. Bando, D. Golberg, J. N. Yao and X. Wang, Joule, 2018, 2, 1075–1094 Search PubMed.
- Y. Zhang, L. Zuo, Y. Huang, L. Zhang, F. Lai and W. Fan, ACS Sustain. Chem. Eng., 2015, 3, 3140–3148 CrossRef CAS.
- Y. Zhang, L. Zhang, B. Zhou, H. Cheng, Q. Zhang and B. Zhang, Carbon, 2023, 205, 562–572 CrossRef CAS.
- C. Liu, K. Wang, X. Zheng, X. Liu, Q. Liang and Z. Chen, Carbon, 2018, 139, 1–9 CrossRef CAS.
- X. Fu, Q. Zheng, L. Li and M. Cao, Carbon, 2022, 197, 324–333 CrossRef CAS.
- F. Zheng, W. Zhong, Q. Deng, Q. Pan, X. Ou, Y. Liu, X. Xiong, C. Yang, Y. Chen and M. Liu, Chem. Eng. J., 2019, 357, 226–236 Search PubMed.
- G. Liu, Q. Zeng, X. Sui, S. Tian, X. Sun, Q. Wu, X. Li, Y. Zhang, K. Tao, E. Xie and Z. Zhang, Small, 2023, 19, 2301085 CrossRef PubMed.
- D. Tian, X. Song, Y. Qiu, X. Sun, B. Jiang, C. Zhao, Y. Zhang, X. Xu, L. Fan and N. Zhang, ACS Nano, 2021, 15, 16515–16524 CrossRef CAS PubMed.
- G. Zhao and H. Zhu, Adv. Mater., 2020, 32, 1905756–1905777 CrossRef CAS.
- Z. Akbar Mohammadi, B. Ameri and S. Saeed Hossein Davarani, Chem. Eng. J., 2023, 470, 144132 CrossRef.
- T. Wang, J. Lei, Y. Wang, L. Pang, F. Pan, K. J. Chen and H. Wang, Small, 2022, 18, 2203307 Search PubMed.
- J. Hu, C. Zhang, P. Yang, J. Xiao, T. Deng, Z. Liu, B. Huang, M. K. H. Leung and S. Yang, Adv. Funct. Mater., 2020, 30, 1908520 Search PubMed.
- H. Wang, W. Zhang, X. Zhang, S. Hu, Z. Zhang, W. Zhou and H. Liu, Nano Res., 2021, 14, 4857–4864 Search PubMed.
- A. S. Justin, P. Vickramman and B. J. Reddy, Curr. Appl. Phys., 2018, 19, 295–302 Search PubMed.
- T. Chen, Z. Liu, Z. Liu, X. Tao, H. Fan and L. Guo, Mater. Lett., 2019, 248, 1–4 CrossRef CAS.
- J. Deng, Q. Gong, H. Ye, K. Feng, J. Zhou, C. Zha, J. Wu, J. Chen, J. Zhong and Y. Li, ACS Nano, 2018, 12, 1829–1836 CrossRef CAS.
- H. Chen, L. Yang, R. Wang, W. Zhang, R. Liu, Y. Yun, N. Wang, S. Ramakrishna, L. Jiao and Y. Long, Small, 2023, 19, 2304086 CrossRef CAS PubMed.
- S. Chen, H. Chen, M. Fan, C. Li and K. Shu, J. Sol–Gel Sci. Technol., 2016, 80, 119–125 CrossRef CAS.
- Z. Ji, L. Chen, K. Liu, D. Ma, S. Zhang, G. Zhu, X. Shen, P. Song and S. Premlatha, Appl. Surf. Sci., 2022, 574, 15172 CrossRef.
- J. Cheng, L. Lin, W. Hong, L. Lin, H. Chen and H. Lai, Electrochim. Acta, 2018, 283, 1245–1252 CrossRef CAS.
- W. W. Liu, M. H. Zhu and J. H. Liu, Inorg. Chem. Front., 2018, 5(9), 2198–2204 RSC.
- T. Yang, Q. Pang, G. Yang, R. Wang, J. Dang, H. Yang, Z. Wang, R. Feng, A. Arshad, X. Xu and S. Yun, Carbon, 2026, 246, 120858 CrossRef CAS.
- F. Liu, G. Li, H. Li, C. Hu, P. F. Msomi, H. Guo and Y. Liu, Carbon, 2025, 244, 120659 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |