Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Copper nanoclusters with atomic precision as catalyst for organic reactions
Rupa
Sarma
,
Sourav
Mandal
,
Dipanwita
Rout
,
Mandira
Ghosh
,
Tokuhisa
Kawawaki
 ,
Sourav
Biswas
,
Sourav
Biswas
 * and
Yuichi
Negishi
* and
Yuichi
Negishi
 *
*
Institute of Multidisciplinary Research for Advanced Materials, Tohoku University, Katahira 2-1-1, Aoba-ku, Sendai 980-8577, Japan. E-mail: Sourav.biswas210@gmail.com; yuichi.negishi.a8@tohoku.ac.jp
First published on 2nd April 2026
Abstract
Atomically precise copper nanoclusters (Cu NCs) have emerged as a distinctive class of catalysts that bridge molecular complexes and conventional nanoparticles. Their well-defined nuclearity, discrete electronic structures, and tunable ligand environments enable direct structure–activity correlations that are inaccessible with traditional Cu catalysts. This review summarizes recent advances in the use of atomically precise Cu NCs as organocatalysts for a broad range of organic transformations, including cycloadditions, C–C and C–N coupling reactions, hydroboration, hydrogenation, oxidation, protosilylation, sulfonylation, and multicomponent reactions. Emphasis is placed on how subtle structural features—such as nuclearity, hydride content, ligand dynamics, surface defects, mixed-valence states, and exposed metal sites—govern catalytic activity, selectivity, and reaction mechanisms. Representative examples illustrate that Cu NCs can operate via radical or polar pathways, act as intrinsic photo- and redox catalysts, and exhibit single-site behavior in both homogeneous and heterogeneous systems. Overall, this review highlights key structure–activity relationships and outlines emerging design principles and future opportunities for developing efficient, sustainable, and earth-abundant Cu-based catalysts.
1. Introduction
Research on atomically precise metal nanoclusters (NCs) originated from their extraordinary photophysical properties, which arise from size-dependent quantum confinement effects and discrete electronic energy levels.1–5 Unlike conventional metal nanoparticles, NCs possess quantized electronic orbitals and well-defined molecular-like electronic structures, enabling strong correlations between their atomic arrangement, electronic configuration, and optical response.6–13 In parallel, their fascinating structural architectures—ranging from core–shell motifs to ligand-protected metal frameworks—have stimulated extensive efforts toward understanding structure–property relationships and expanding the library of atomically precise NCs.14–17In the early stages of NC research, a major challenge was their limited stability in solution, which severely restricted detailed characterization and hindered exploration of their functional applications.18,19 This instability was primarily associated with weak metal–ligand interactions and susceptibility to aggregation or oxidation.20,21 However, rapid advances in synthetic methodologies and surface protection strategies have largely overcome these limitations.15,22–27 The development of robust ligand shells—such as thiolates, phosphines, N-heterocyclic carbenes, carborane and alkynyl ligands—has enabled strong metal–ligand interactions, significantly enhancing the chemical and thermal stability of NCs.28–33 These advances further facilitated the transition from noble-metal-based NCs to those composed of first-row transition metals, thereby broadening compositional diversity while maintaining structural precision and stability. The availability of stable and well-defined NCs marked a pivotal shift in research focus from purely photophysical investigations toward functional applications. In particular, the atomically precise structures, high surface atom exposure, and tunable electronic states of NCs make them highly attractive candidates for catalysis.4,34–37 Unlike traditional heterogeneous catalysts, which often suffer from ill-defined active sites and limited structure–activity correlations, NCs offer an unparalleled platform for correlating catalytic performance with precise atomic structures and electronic configurations. Consequently, metal NCs have emerged as a new class of catalysts capable of overcoming many intrinsic limitations associated with conventional transition-metal catalysts, such as poor selectivity, low atom efficiency, and limited mechanistic insight.38–42
As the scope of NC research expanded beyond noble metals, copper (Cu) NCs rapidly gained attention due to earth abundance of elemental Cu, low cost, multiple accessible oxidation states, and favorable redox potentials.43–52 These intrinsic properties, combined with atomic precision and ligand-induced electronic modulation, render Cu NCs particularly promising for catalytic applications. As a result, a rapidly growing body of literature now demonstrates the extensive use of Cu NCs as catalysts in a wide range of chemical transformations. These applications span diverse areas, including organic transformations, electrocatalytic and photocatalytic reduction reactions, and the conversion of both organic and inorganic substrates.53–59 Notably, the most pronounced diversity and performance enhancements have been observed in organic transformation reactions, where the catalytic activity, selectivity, and reaction pathways are highly sensitive to the structural architecture of the Cu NCs (Fig. 1).60 Importantly, organic catalytic transformations play a pivotal role in modern synthetic chemistry by enabling the efficient construction and functionalization of molecular architectures through controlled substrate activation, electron-transfer processes, and radical or polar reaction pathways. Accordingly, catalysts with atomically well-defined structures are particularly valuable because they allow direct correlation between electronic structure, active-site geometry, and catalytic reactivity in complex organic transformation. Parameters such as nuclearity, ligand environment, surface defects, and oxidation state distribution play critical roles in governing catalytic behavior. Despite several earlier review efforts summarizing aspects of Cu-based NC catalysis, the exceptionally rapid growth of this field necessitates an updated and comprehensive assessment that incorporates recent discoveries, particularly those emphasizing structure–activity relationships.60,61 However, although a range of analytical techniques—including single-crystal X-ray diffraction (SCXRD), electrospray ionization mass spectrometry (ESI-MS), Extended X-ray absorption fine structure (EXAFS), and high-resolution transmission electron microscopy (HR-TEM)—are available for determining the structures of NCs, SCXRD remains the most comprehensive and reliable method (Scheme 1). This is because it provides precise atomic-level insights into both their geometric and electronic structures. Therefore, in this review, we primarily focus on the catalytic reactivity of nanoclusters whose structural architectures have been unambiguously resolved by SCXRD, enabling a clear correlation between their well-defined structures and observed catalytic properties.
 | ||
| Fig. 1 Schematic representation of utilization of diverse Cu NCs in diverse organocatalytic reactions. | ||
In this review, we systematically summarize recent progress in the application of Cu NCs in organic transformation reactions, with a particular emphasis on elucidating the roles of atomic precision, structural architecture, and electronic structure in determining catalytic performance (Fig. 1). Comparisons with conventional catalytic systems are provided to highlight the unique advantages of Cu NCs. Finally, we discuss emerging trends and future directions in this rapidly evolving area, focusing on how strategic structural evolution and rational ligand design are shaping the next generation of Cu–NC-based catalysts.
2. Utilization of Cu NCs in organic transformation reactions
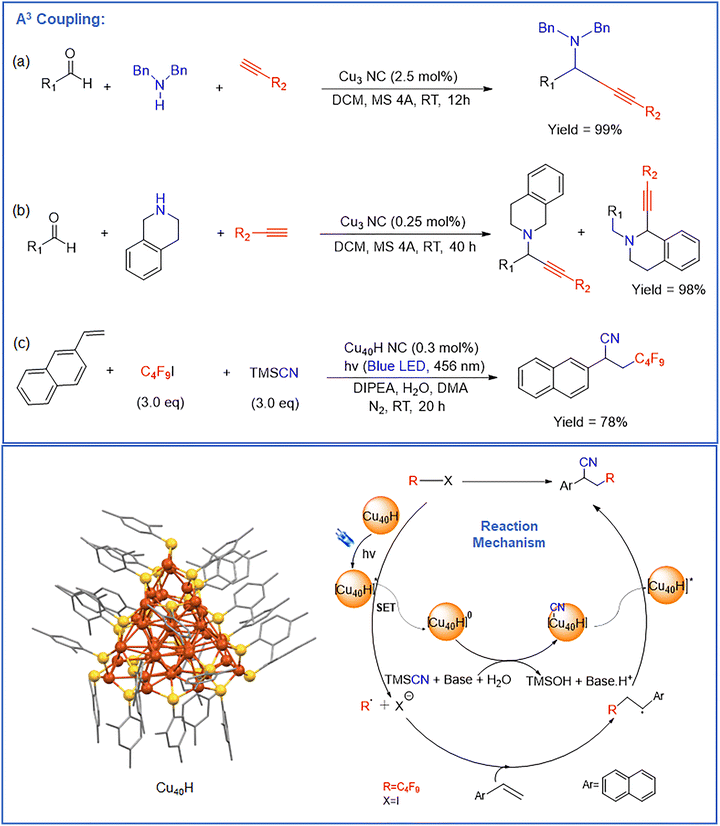
The development of Cu NCs can be traced back to the early synthesis of copper-hydride (CuH) NCs, which represented one of the first attempts to isolate atomically precise Cu-based species with cluster-like electronic structures.62–67 These CuH NCs attracted significant interest due to their unique electronic characteristics and their conceptual similarity to molecular metal hydride complexes.64 However, the intrinsic instability of CuH NCs—stemming from weak metal–H bonding, high susceptibility to oxidation, and structural lability in solution—severely limited their long-term stability and practical applicability. As a result, systematic studies on their catalytic behavior, mechanistic pathways, and recyclability remained challenging. The introduction of surface protection strategies marked a turning point in Cu NC research.68–70 The incorporation of strongly coordinating ligands significantly enhanced the stability without compromising atomic precision. These surface-protecting units not only prevented aggregation and oxidation of the Cu core but also played an active role in modulating the electronic structure and surface reactivity of the NCs. Consequently, ligand-protected Cu NCs emerged as a stable and versatile platform for both fundamental investigations and applied catalysis.53,71It is noteworthy that CuH complexes and related C–H–Cu species are well established as efficient catalysts for a variety of organic transformations, including hydrogenation, hydroamination, hydrosilylation, and C–H functionalization reactions.72–75 Nevertheless, when translated to the regime, the weak stability of hydride-containing Cu NCs restricts their isolation and hinders direct evaluation of their catalytic potential under practical reaction conditions. This limitation underscores the importance of ligand-stabilized Cu NCs, where the hydride motif may be transient or generated in situ, while the overall cluster framework remains structurally intact. The availability of a diverse library of stable Cu NCs has therefore significantly broadened the scope of Cu-based NC catalysis.68 These well-defined NCs enable systematic exploration of structure–activity relationships, revealing how parameters such as nuclearity, ligand environment, surface defects, and oxidation-state distribution influence catalytic performance. As a result, Cu NCs have been successfully employed in a wide range of organic transformation reactions, often exhibiting superior selectivity, enhanced atom efficiency, and unique reaction pathways compared to conventional copper catalysts and nanoparticles. In the following sections, we comprehensively summarize representative organic transformation reactions catalyzed by Cu NCs, with particular emphasis on the role of cluster stability and structural architecture in governing catalytic activity and selectivity.
2.1. Click reaction
Cook et al. reported a mixed-valent [Cu20(CCPh)12(OAc)6] (Cu20) (CCPh = phenyl acetylide; OAc = acetate) NC, where a tetrahedral [Cu4]2+ kernel is encapsulated within a [Cu16(CCPh)12(OAc)6]2− shell.76 This structure represents the first example of a Cu-only superatom featuring a tetrahedral core, hosting a two-electron superatomic configuration. Cu20 efficiently catalyzed the [3 + 2] cycloaddition between benzyl azide (PhCH2N3) and a broad range of terminal alkynes—including phenylacetylene (HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh), ethyl propiolate (HC
CPh), ethyl propiolate (HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CCO2Et), and tert-butyl acetylene (HC
CCO2Et), and tert-butyl acetylene (HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CtBu)—producing the corresponding 1,2,3-triazoles in excellent yields (Fig. 2). The well-defined Cu20 framework provides accessible Cu(I) sites that can promote azide–alkyne cycloaddition without requiring catalyst pretreatment, while the ligand-protected structure prevents aggregation during catalysis. Remarkably, high activity was maintained even at catalyst loadings as low as 0.05 mol%. An intriguing observation was the formation of 1-benzyl-4-phenyl-1H-1,2,3-triazole when alkynes other than HC
CtBu)—producing the corresponding 1,2,3-triazoles in excellent yields (Fig. 2). The well-defined Cu20 framework provides accessible Cu(I) sites that can promote azide–alkyne cycloaddition without requiring catalyst pretreatment, while the ligand-protected structure prevents aggregation during catalysis. Remarkably, high activity was maintained even at catalyst loadings as low as 0.05 mol%. An intriguing observation was the formation of 1-benzyl-4-phenyl-1H-1,2,3-triazole when alkynes other than HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh were employed. This indicated direct involvement of the phenylacetylide ligands in the catalytic cycle. The authors proposed a mechanism in which PhCH2N3 first reacts with a ligand-derived phenyl acetylide to form a Cu-bound triazolate intermediate. Subsequent protonolysis by an external alkyne releases the triazole product and regenerates the copper-acetylide site, sustaining the catalytic turnover. The study further distinguished the performance of Cu20 NC in homogeneous versus heterogeneous environments. For the heterogeneous variant, the Cu20 NC was grafted onto dry, partially dehydroxylated silica, with immobilization accompanied by the loss of one phenylacetylide ligand per cluster, generating a Cu–O–Si linkage. While the homogeneous catalyst exhibited signs of structural degradation during reaction, the silica-supported cluster retained both its structural fidelity and catalytic efficiency across multiple cycles. No detectable Cu leaching was observed, confirming that catalysis originated from the intact immobilized NC rather than from dissolved Cu+ species. Overall, this work establishes Cu NCs as a robust and versatile catalyst that performs efficiently in both homogeneous and supported heterogeneous forms. It further shows that immobilization on a solid support preserves the structural integrity of atomically precise Cu NCs, enhancing their stability, preventing leaching, and enabling reusability—crucial features for next-generation copper-based catalysts.
CPh were employed. This indicated direct involvement of the phenylacetylide ligands in the catalytic cycle. The authors proposed a mechanism in which PhCH2N3 first reacts with a ligand-derived phenyl acetylide to form a Cu-bound triazolate intermediate. Subsequent protonolysis by an external alkyne releases the triazole product and regenerates the copper-acetylide site, sustaining the catalytic turnover. The study further distinguished the performance of Cu20 NC in homogeneous versus heterogeneous environments. For the heterogeneous variant, the Cu20 NC was grafted onto dry, partially dehydroxylated silica, with immobilization accompanied by the loss of one phenylacetylide ligand per cluster, generating a Cu–O–Si linkage. While the homogeneous catalyst exhibited signs of structural degradation during reaction, the silica-supported cluster retained both its structural fidelity and catalytic efficiency across multiple cycles. No detectable Cu leaching was observed, confirming that catalysis originated from the intact immobilized NC rather than from dissolved Cu+ species. Overall, this work establishes Cu NCs as a robust and versatile catalyst that performs efficiently in both homogeneous and supported heterogeneous forms. It further shows that immobilization on a solid support preserves the structural integrity of atomically precise Cu NCs, enhancing their stability, preventing leaching, and enabling reusability—crucial features for next-generation copper-based catalysts.
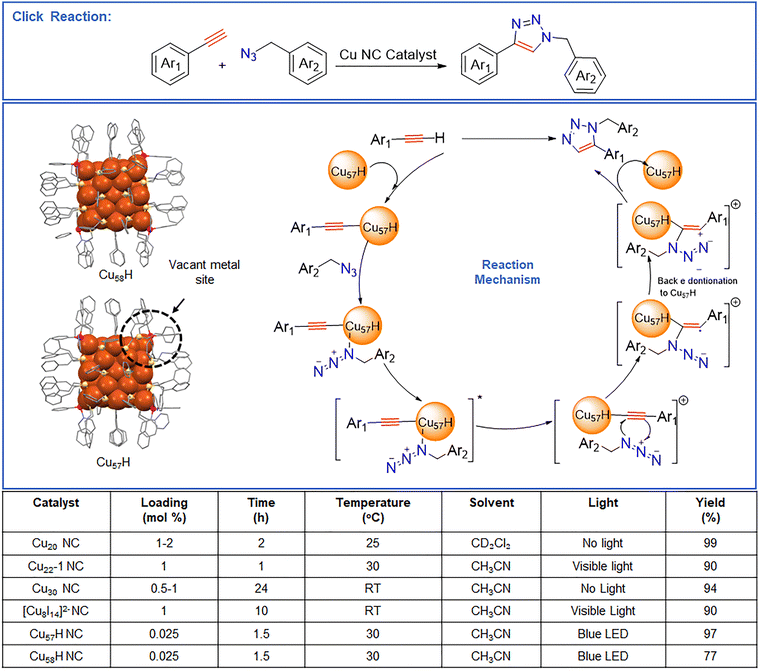 | ||
| Fig. 2 Representation of click reaction and utilization of diverse Cu NCs with diverse yields and the associated reaction mechanism by utilizing Cu57H NC. | ||
Thus, the previous study established an important model for employing Cu NCs as heterogeneous catalysts. This concept subsequently influenced several related works, where Cu NCs were used heterogeneously even when water-soluble variants were available. In this context, Yang et al. reported such water-soluble variant, [Cu30(MBA)16] (Cu30) (MBA = 4-mercaptobenzoic acid) NC, and evaluated its catalytic performance in azide–alkyne cycloaddition under heterogeneous conditions by immobilizing it onto amine-functionalized TiO2 nanoparticles (TiO2–NH2) (Fig. 2).77 The supported catalyst exhibited a substantially higher yield (94%) than the unsupported cluster at room temperature. This improvement was attributed to enhanced dispersion of Cu30 NC on the support surface, which increased the number of accessible active sites and provided superior structural stability during repeated catalytic cycles. In particular, anchoring the nanocluster on TiO2–NH2 significantly enhances its structural stability and prevents aggregation, allowing the catalyst to be reused over multiple reaction cycles. X-ray photoelectron spectroscopy (XPS) analysis revealed that ∼67% of the Cu centers were present as Cu(I), the oxidation state primarily responsible for high activity in click-type cycloadditions. Substrate-scope studies showed that the nature of alkyne substituents strongly affected reaction efficiency: electron-donating or sterically bulky groups led to reduced yields due to steric hindrance or lower alkyne reactivity, whereas smaller or electron-withdrawing substituents afforded higher conversions. Thus, the findings highlight the combined steric and electronic factors that govern the catalytic performance of the Cu30 NC in promoting azide–alkyne cycloaddition.
The individual Cu NCs demonstrate significant potential for cycloaddition reactions; however, understanding how their precise structural architectures influence catalytic behaviour requires direct comparison of well-defined structural isomers. Such controlled studies become feasible when isomeric NCs are available. Zhou et al. reported the catalytic activity of two such structural isomers, Cu22-1 and Cu22-2, both formulated as [Cu22(SePh)10(Se)6[P(Ph-4F)3]8] (SePh = phenylselenide, P(Ph-4F)3 = tris(4-fluorophenyl)phosphine) in cycloaddition reaction (Fig. 2).78 Single-crystal X-ray diffraction (SCXRD) data revealed that each NC contains a similar Cu16Se6 core which is capped by Cu(SeR)3(P(Ph-4F)3) surface motifs. The isomerism originates from different spatial arrangements of these capping motifs, which leads to distinct bonding environments and packing interactions through C–F⋯H–C and π⋯π interactions. These structural differences translate into measurable electronic and optical distinctions: Cu22-1 exhibits a shoulder at 420 nm and a band gap of 2.60 eV, whereas Cu22-2 shows a weaker absorption at 382 nm with a band gap of 2.73 eV. DFT (density functional theory) and TDDFT (time dependent density functional theory) analyses further corroborated subtle differences in orbital distributions and oscillator strengths. The two isomers displayed markedly different catalytic activities in the [3 + 2] azide–alkyne cycloaddition between PhCH2N3 and HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh. Under thermal conditions (50 °C, 0.06 mol% catalyst), Cu22-1 delivered a 92% yield in 24 h, while Cu22-2 achieved only 36%. The disparity was even more pronounced under 405 nm irradiation: Cu22-1 reached 90% yield within 1 h, whereas Cu22-2 afforded just 10%. The superior performance of Cu22-1 arises from its more favourable surface-motif configuration and higher oscillator strength (0.01004 vs. 0.00405), which enhance light absorption and electron–hole pair generation, thereby boosting photocatalytic efficiency. Ultraviolet-visible (UV-vis) absorbance and powder X-ray diffraction (PXRD) analyses confirmed that both NCs remained structurally intact after catalysis. Thus, this study demonstrates that even subtle variations in the structural architecture of Cu NCs can significantly alter their overall catalytic activity. Therefore, designing the appropriate geometry is crucial for maximizing the catalytic performance of NCs. However, several strategies reported in the literature show that structural activation of existing NCs can also enhance their catalytic activity, enabling otherwise inert clusters to function as efficient catalysts.
CPh. Under thermal conditions (50 °C, 0.06 mol% catalyst), Cu22-1 delivered a 92% yield in 24 h, while Cu22-2 achieved only 36%. The disparity was even more pronounced under 405 nm irradiation: Cu22-1 reached 90% yield within 1 h, whereas Cu22-2 afforded just 10%. The superior performance of Cu22-1 arises from its more favourable surface-motif configuration and higher oscillator strength (0.01004 vs. 0.00405), which enhance light absorption and electron–hole pair generation, thereby boosting photocatalytic efficiency. Ultraviolet-visible (UV-vis) absorbance and powder X-ray diffraction (PXRD) analyses confirmed that both NCs remained structurally intact after catalysis. Thus, this study demonstrates that even subtle variations in the structural architecture of Cu NCs can significantly alter their overall catalytic activity. Therefore, designing the appropriate geometry is crucial for maximizing the catalytic performance of NCs. However, several strategies reported in the literature show that structural activation of existing NCs can also enhance their catalytic activity, enabling otherwise inert clusters to function as efficient catalysts.
Xiao et al. reported a controlled synthetic approach to confine a dimeric [Cu8I14]2− NC in a supramolecule through host–guest interaction and identified its catalytic activity azide–alkyne cycloaddition (Fig. 2).79 The confined supramolecular [Cu8I14]2− NC provides multiple accessible Cu(I) sites and promotes efficient charge separation under visible light irradiation, thereby enhancing photocatalytic performance. In benzylamine oxidation, it achieved up to 95% conversion and 76% yield under 455 nm irradiation and O2, outperforming free CuI and the ligand alone. Although minor structural degradation was observed, the catalyst retained most of its activity over three cycles. In addition, this self-assembly also delivered up to 90% yield for the coupling of HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh and PhCH2N3 under blue light within 10 hours. Electron-donating substituents enhanced yields, whereas electron-withdrawing groups decreased them (56–77%). 2,2,6,6-Tetramethylpiperidine-1-oxyl (TEMPO) quenching significantly suppressed reactivity, confirming a photoinduced radical pathway. Mechanistic investigations showed that light excitation generates from the confined NC which produces reactive oxygen species (O2˙−, ˙OH) that drive electron-transfer processes. DFT calculations further supported a radical mechanism and highlighted the role of multiple Cu(I) centers in enabling efficient charge separation. Thus, simply confining a Cu NC can activate its surface and impart exceptional catalytic reactivity.
CPh and PhCH2N3 under blue light within 10 hours. Electron-donating substituents enhanced yields, whereas electron-withdrawing groups decreased them (56–77%). 2,2,6,6-Tetramethylpiperidine-1-oxyl (TEMPO) quenching significantly suppressed reactivity, confirming a photoinduced radical pathway. Mechanistic investigations showed that light excitation generates from the confined NC which produces reactive oxygen species (O2˙−, ˙OH) that drive electron-transfer processes. DFT calculations further supported a radical mechanism and highlighted the role of multiple Cu(I) centers in enabling efficient charge separation. Thus, simply confining a Cu NC can activate its surface and impart exceptional catalytic reactivity.
Beyond confinement, introducing defects on the NC surface can also strongly influence catalytic activity. This effect was clearly demonstrated by Dong et al., who showed that single-atom surface engineering can markedly enhance catalytic performance while preserving the overall structural integrity of the Cu NC.80 They synthesized the parent [Cu58H20(PET)36(PPh3)4]2+ (Cu58H) (PET = phenylethanethiolate; PPh3 = triphenylphosphine) NC, via an optimized reduction protocol. The Cu58H NC features a five-shell concentric architecture surrounding a Cu8 cubic core. Within this framework, the outermost metal–ligand shell contains a Cu8 cube bound to four PPh3 ligands. Based on the coordination pattern, the PPh3 ligands bind in a μ1 mode, leaving four surface Cu atoms coordinatively accessible and thus more susceptible to selective surface modification. Utilising this feature, the authors treated the NC with excess PPh3 (27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio), enabling the controlled removal of a single surface Cu atom and generating the defective analogue [Cu57H20(PET)36(PPh3)4]+ (Cu57H). Structural characterization confirmed that Cu57H preserves the parent geometric framework with the exception of a single-atom vacancy. Accommodation of this vacancy induces subtle rearrangements in adjacent thiolate coordination. Despite the minimal structural alteration, the defect has profound electronic consequences. Fig. 2 depicts the proposed catalytic cycle for Cu57H-mediated photoinduced azide–alkyne cycloaddition. The reaction is initiated by coordination and activation of the alkyne to generate a Cu–σ,π-alkynyl intermediate. Subsequent light-driven radical processes enable coupling with the azide, forming the triazole framework. Notably, the presence of a surface Cu vacancy in Cu57H promotes substrate activation and facilitates efficient regeneration of the catalytic site, thereby enhancing overall catalytic performance. DFT calculations indicated a localized region of reduced electron density at the vacancy site, while time-resolved spectroscopy revealed a significantly prolonged excited-state lifetime (248.7 ps for Cu57H vs. 108.8 ps for Cu58H), consistent with enhanced charge separation. These effects translated directly into catalytic performance: in photoinduced azide–alkyne cycloaddition reactions, Cu57 achieved a 97% yield compared to 77% for Cu58H under identical conditions (Fig. 2). The enhanced activity arises from three key factors: (i) the vacancy generates an exposed, coordinatively unsaturated Cu site that facilitates substrate binding; (ii) local surface rearrangement produces a geometry more reminiscent of highly reactive Cu (111) facets rather than the original Cu (100)-like arrangement; and (iii) the extended charge-separated state improves photocatalytic efficiency. Mechanistic studies further supported a radical pathway, as TEMPO quenching reduced the product yield to below 10%.
1 molar ratio), enabling the controlled removal of a single surface Cu atom and generating the defective analogue [Cu57H20(PET)36(PPh3)4]+ (Cu57H). Structural characterization confirmed that Cu57H preserves the parent geometric framework with the exception of a single-atom vacancy. Accommodation of this vacancy induces subtle rearrangements in adjacent thiolate coordination. Despite the minimal structural alteration, the defect has profound electronic consequences. Fig. 2 depicts the proposed catalytic cycle for Cu57H-mediated photoinduced azide–alkyne cycloaddition. The reaction is initiated by coordination and activation of the alkyne to generate a Cu–σ,π-alkynyl intermediate. Subsequent light-driven radical processes enable coupling with the azide, forming the triazole framework. Notably, the presence of a surface Cu vacancy in Cu57H promotes substrate activation and facilitates efficient regeneration of the catalytic site, thereby enhancing overall catalytic performance. DFT calculations indicated a localized region of reduced electron density at the vacancy site, while time-resolved spectroscopy revealed a significantly prolonged excited-state lifetime (248.7 ps for Cu57H vs. 108.8 ps for Cu58H), consistent with enhanced charge separation. These effects translated directly into catalytic performance: in photoinduced azide–alkyne cycloaddition reactions, Cu57 achieved a 97% yield compared to 77% for Cu58H under identical conditions (Fig. 2). The enhanced activity arises from three key factors: (i) the vacancy generates an exposed, coordinatively unsaturated Cu site that facilitates substrate binding; (ii) local surface rearrangement produces a geometry more reminiscent of highly reactive Cu (111) facets rather than the original Cu (100)-like arrangement; and (iii) the extended charge-separated state improves photocatalytic efficiency. Mechanistic studies further supported a radical pathway, as TEMPO quenching reduced the product yield to below 10%.
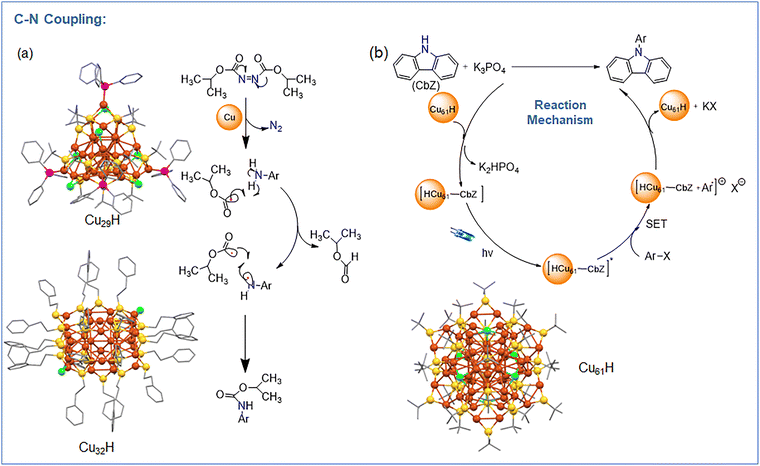
2.2. Coupling reactions
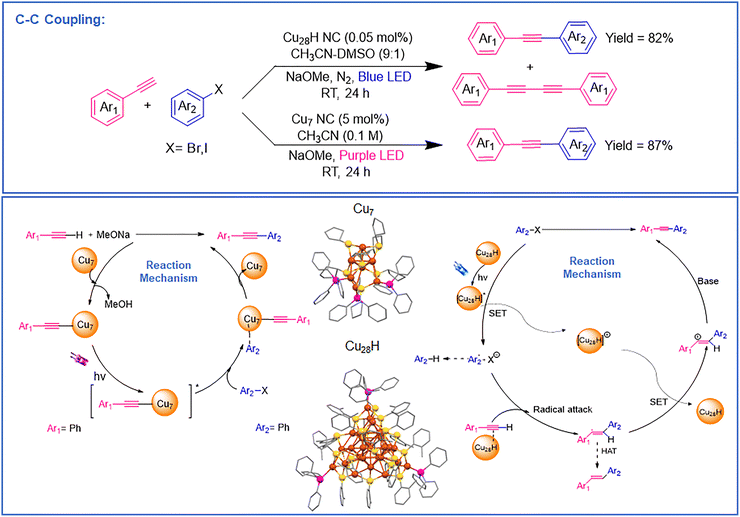
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh in the presence of 5 mol% Cu7 NC and NaOMe in MeCN produced 1-phenyl-2-phenylacetylene in 87% yield. Substituting purple LEDs with blue LEDs or CFL bulbs reduced the yield to 79% and 43%, respectively. Replacing the NC with CuCl gave a 37% yield accompanied by homocoupled 1,3-diyne byproducts, highlighting its selectivity. To rationalize this behavior, the proposed catalytic cycle is illustrated in Fig. 4 which depicts the hydride free Cu7 NC follows a polar catalytic pathway initiated by the formation of a Cu–σ-alkynyl intermediate upon alkyne activation. Subsequent photoexcitation enables oxidative addition of the aryl halide at the Cu center, identified as the rate-determining step, leading to a Cu–C intermediate. This is followed by reductive elimination to yield the C(sp2)–C(sp) coupled product, with regeneration of the active Cu7 NC catalyst completing the catalytic cycle. Absence of light, base, or catalyst resulted in no reaction. The catalyst was recyclable up to 10 runs with 61% yield retained, and post-catalysis UV-vis/transmission electron microscopy (TEM)/ESI-MS confirmed the overall structural integrity, though minor ligand loss occurred through PPh3 ligands. Substrate scope tests showed that both aryl iodides and aryl bromides bearing electron-donating (–CH3, –OCH3) or electron-withdrawing (–NO2, –CO2Et, –CF3, –F) substituents yielded corresponding alkynylated products in high selectivity. For example, p-nitroiodobenzene with HC
CPh in the presence of 5 mol% Cu7 NC and NaOMe in MeCN produced 1-phenyl-2-phenylacetylene in 87% yield. Substituting purple LEDs with blue LEDs or CFL bulbs reduced the yield to 79% and 43%, respectively. Replacing the NC with CuCl gave a 37% yield accompanied by homocoupled 1,3-diyne byproducts, highlighting its selectivity. To rationalize this behavior, the proposed catalytic cycle is illustrated in Fig. 4 which depicts the hydride free Cu7 NC follows a polar catalytic pathway initiated by the formation of a Cu–σ-alkynyl intermediate upon alkyne activation. Subsequent photoexcitation enables oxidative addition of the aryl halide at the Cu center, identified as the rate-determining step, leading to a Cu–C intermediate. This is followed by reductive elimination to yield the C(sp2)–C(sp) coupled product, with regeneration of the active Cu7 NC catalyst completing the catalytic cycle. Absence of light, base, or catalyst resulted in no reaction. The catalyst was recyclable up to 10 runs with 61% yield retained, and post-catalysis UV-vis/transmission electron microscopy (TEM)/ESI-MS confirmed the overall structural integrity, though minor ligand loss occurred through PPh3 ligands. Substrate scope tests showed that both aryl iodides and aryl bromides bearing electron-donating (–CH3, –OCH3) or electron-withdrawing (–NO2, –CO2Et, –CF3, –F) substituents yielded corresponding alkynylated products in high selectivity. For example, p-nitroiodobenzene with HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh produced 4-nitrophenylphenylacetylene, while 2-bromopyridine yielded 2-(phenylethynyl)pyridine. Similarly, various alkynes such as p-tolylacetylene, p-aminophenylacetylene, p-trifluoromethylphenylacetylene, and 2-thiophenylacetylene gave their respective C(sp2)–C(sp) coupled products in good yields, confirming broad substrate tolerance and full suppression of alkyne homocoupling. Mechanistic studies combining radical-trapping and DFT calculations established a non-radical, polar pathway distinct from the previous study. DFT calculation revealed that the active Cu site lies at the triangular base of the Cu4 head, where adsorption of HC
CPh produced 4-nitrophenylphenylacetylene, while 2-bromopyridine yielded 2-(phenylethynyl)pyridine. Similarly, various alkynes such as p-tolylacetylene, p-aminophenylacetylene, p-trifluoromethylphenylacetylene, and 2-thiophenylacetylene gave their respective C(sp2)–C(sp) coupled products in good yields, confirming broad substrate tolerance and full suppression of alkyne homocoupling. Mechanistic studies combining radical-trapping and DFT calculations established a non-radical, polar pathway distinct from the previous study. DFT calculation revealed that the active Cu site lies at the triangular base of the Cu4 head, where adsorption of HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh and NaOMe forms a Cu–acetylide intermediate (Cu–C ≈ 1.90 Å, ΔG = –1.06 eV). Subsequent oxidative addition of aryl iodide at this Cu center (rate-determining, ΔG‡ = 1.15 eV) leads to a Cu(III)-like intermediate, followed by reductive elimination generating the cross-coupled product and regenerating Cu7. The overall reaction free energy is –2.28 eV, confirming strong thermodynamic favorability. The mechanistic cycle thus proceeds via Cu-σ-alkynyl intermediate → photoexcited Cu-acetylide → oxidative addition → reductive elimination, forming the C(sp2)–C(sp) bond cleanly through a polar process. Thus, this work demonstrates that hydride-free Cu NCs enable efficient and highly selective C–C bond formation under mild conditions. The absence of hydrides shifts the mechanism from a radical to a polar pathway, affording clean, high-yield Sonogashira-type couplings and highlighting hydride-free Cu NCs as promising catalysts for sustainable photochemical synthesis.
CPh and NaOMe forms a Cu–acetylide intermediate (Cu–C ≈ 1.90 Å, ΔG = –1.06 eV). Subsequent oxidative addition of aryl iodide at this Cu center (rate-determining, ΔG‡ = 1.15 eV) leads to a Cu(III)-like intermediate, followed by reductive elimination generating the cross-coupled product and regenerating Cu7. The overall reaction free energy is –2.28 eV, confirming strong thermodynamic favorability. The mechanistic cycle thus proceeds via Cu-σ-alkynyl intermediate → photoexcited Cu-acetylide → oxidative addition → reductive elimination, forming the C(sp2)–C(sp) bond cleanly through a polar process. Thus, this work demonstrates that hydride-free Cu NCs enable efficient and highly selective C–C bond formation under mild conditions. The absence of hydrides shifts the mechanism from a radical to a polar pathway, affording clean, high-yield Sonogashira-type couplings and highlighting hydride-free Cu NCs as promising catalysts for sustainable photochemical synthesis.
2.3. Hydroboration
Alamer et al. reported a robust [Cu45H14(TBBT)29(PPh3)4(tBuNH2)2]2+ (Cu45H) (TBBT = 4-tert-butylbenzenethiolate; tBuNH2 = tert-butylamine) NC, featuring a planar hexagonal Cu7 core enclosed by a Cu38 shell composed of two Cu–thiolate layers linked through a phosphine-stabilized macrocyclic Cu14(TBBT)16P4 ring.89 The NC contains 14 hydrides and is structurally well-defined by SCXRD. Cu45H proved to be a highly efficient catalyst for hydroboration of C–C multiple bonds, delivering up to 90% isolated yield and >95% E-selectivity for terminal alkynes at only 0.025 mol% loading under ambient, open-air conditions (Fig. 6). Air and water were beneficial to the catalytic cycle, and control studies identified the macrocyclic Cu–S–P interface as the active site, outperforming Cu–TBBT, Cu–TPP, Cu61H, and even CuCl (5 mol%). The catalyst tolerated diverse functional groups and showed broad substrate scope, including aryl, heteroaryl, and aliphatic alkynes, internal alkynes, and styrenes, with recyclability up to five cycles and gram-scale applicability. As shown in Fig. 7, the Cu45H NC catalyzes hydroboration via a radical-mediated pathway. Activation of the boron reagent at the Cu–thiolate–phosphine interface generates reactive radical species, which subsequently add across the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C bond of the alkyne to form a vinyl intermediate. This intermediate undergoes further transformation to yield the hydroborated product with high regio- and stereoselectivity. The catalytic cycle is sustained by regeneration of the active Cu45H NC, with the cluster interface playing a crucial role in facilitating substrate activation and stabilizing reactive intermediates. Mechanistic experiments indicated a radical pathway, supported by inhibition with TEMPO/BHT/DMPO (BHT = butylated hydroxytoluene; DMPO = 5,5-dimethyl-1-pyrroline N-oxide) and detection of radical adducts by GC–MS. Thus, Cu45H NC demonstrates how precise Cu–thiolate–phosphine interfaces in atomically defined clusters enable highly selective C–B bond formation under mild conditions.
C bond of the alkyne to form a vinyl intermediate. This intermediate undergoes further transformation to yield the hydroborated product with high regio- and stereoselectivity. The catalytic cycle is sustained by regeneration of the active Cu45H NC, with the cluster interface playing a crucial role in facilitating substrate activation and stabilizing reactive intermediates. Mechanistic experiments indicated a radical pathway, supported by inhibition with TEMPO/BHT/DMPO (BHT = butylated hydroxytoluene; DMPO = 5,5-dimethyl-1-pyrroline N-oxide) and detection of radical adducts by GC–MS. Thus, Cu45H NC demonstrates how precise Cu–thiolate–phosphine interfaces in atomically defined clusters enable highly selective C–B bond formation under mild conditions.
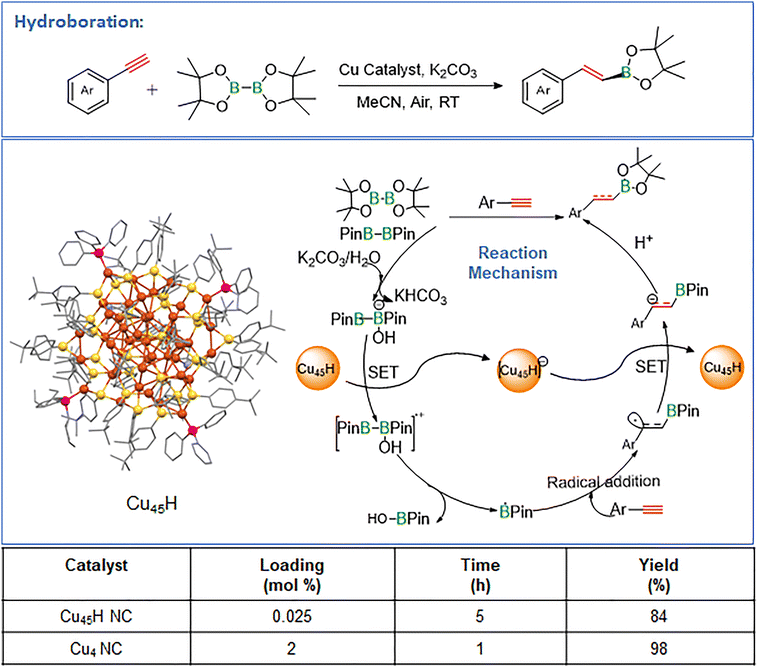 | ||
| Fig. 7 Representation of hydroboration reactions and the performance of various Cu NCs with corresponding yields. | ||
Jia et al. reported dual catalytic sites for highly chemoselective alkyne hydroboration in synthesized NCs.90 They reported ([Cu4(NHT)4]) (Cu4) NC and [Cu8(NHT)4(StBu)4] (Cu8) (NHT = N-heterocyclic thione; StBu = tert-butyl thiolate) in which N-heterocyclic thione ligands provide both strong Cu–S and reversible Cu–N coordination. SCXRD revealed a Cu4 core bridged by NHT ligands in Cu4 NC and a fused Cu4–Cu4 architecture in Cu8 NC. This combination of structural stability and dynamic Cu–N flexibility enabled efficient heterogeneous catalysis under ambient conditions. Cu4 NC showed exceptional hydroboration activity with bis(pinacolato)diboron (B2Pin2), delivering vinylboronates in up to 98% yield and achieving a TON of 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 786—far exceeding conventional Cu catalysts (Fig. 6). It exhibited excellent regio-, stereo-, and chemoselectivity across diverse aryl, heteroaryl, and sterically hindered alkynes, preferentially reducing C
786—far exceeding conventional Cu catalysts (Fig. 6). It exhibited excellent regio-, stereo-, and chemoselectivity across diverse aryl, heteroaryl, and sterically hindered alkynes, preferentially reducing C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C bonds even in the presence of alkenes or carbonyls. Cu8 NC, in contrast, showed only moderate activity (64% yield). Mechanistic studies indicated that reversible Cu–N bond dissociation generates open active sites, while robust Cu–S bonds maintain cluster integrity. The reaction proceeds via cluster borylation, alkyne insertion, and protonation. DFT results showed a lower activation barrier for Cu4 NC (22.9 kcal mol−1) than for Cu8 NC (24.7 kcal mol−1), rationalizing its superior reactivity.
C bonds even in the presence of alkenes or carbonyls. Cu8 NC, in contrast, showed only moderate activity (64% yield). Mechanistic studies indicated that reversible Cu–N bond dissociation generates open active sites, while robust Cu–S bonds maintain cluster integrity. The reaction proceeds via cluster borylation, alkyne insertion, and protonation. DFT results showed a lower activation barrier for Cu4 NC (22.9 kcal mol−1) than for Cu8 NC (24.7 kcal mol−1), rationalizing its superior reactivity.
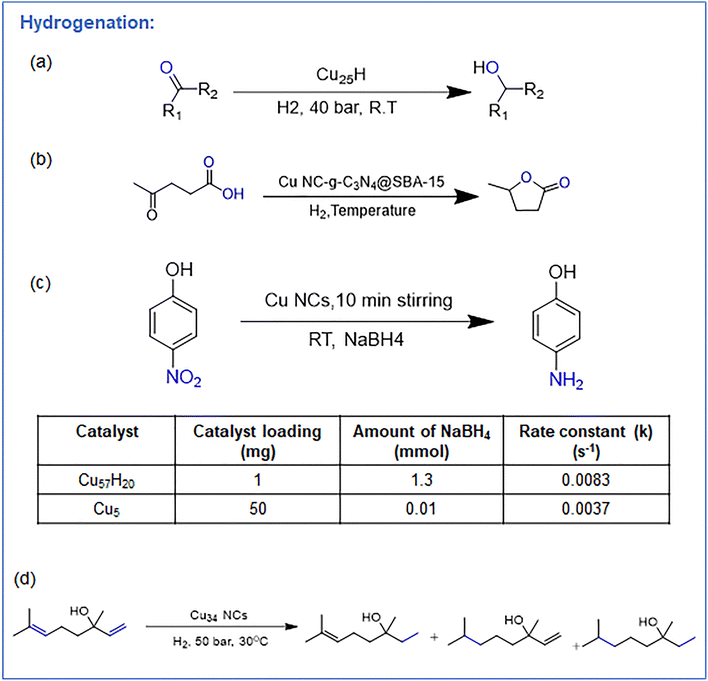
2.4. Hydrogenation
Sun et al. reported the hydride-rich [Cu25H10(SPhCl2)18]3− (Cu25H), featuring a Cu13 icosahedral core capped by twelve Cu atoms and stabilized by ten hydrides and eighteen thiolates.91 This atomically precise NC exhibits remarkable catalytic activity for the room-temperature hydrogenation of 2-hexanone and 3-hexanone to the corresponding alcohols (Fig. 8a). Notably, D2-labeling experiments unambiguously revealed that the hydride ligands themselves act as the hydrogen source, directly transferring hydrogen to the substrate during reduction. Consequently, the hydrides play a dual role: they not only stabilize the Cu framework but also constitute a readily accessible and reactive hydride reservoir. Importantly, SCXRD and ESI-MS analyses confirmed that the NC structure remained intact after catalysis. This unique structural and functional synergy endows Cu25H NC with distinctive single-site catalytic behavior and underpins its high efficiency in selective hydrogenation reactions.Later, Lan et al. reported an efficient catalytic system for converting levulinic acid to γ-valerolactone using Cu NCs stabilized by graphitic carbon nitride (g-C3N4) within Santa Barbara Amorphous-15 (SBA-15) mesoporous silica to check the effectivity in heterogenous medium.92 The catalyst, Cu/NC@SBA-15, prepared through impregnation, calcination, and H2 reduction, benefits from in situ-formed g-C3N4, whose N–Cu coordination stabilizes mixed-valent Cu0/Cu+/Cu2+ sites and suppresses agglomeration. Cu/NC@SBA-15 delivered 92% yield at 140 °C and 3 MPa H2—comparable to Ru catalysts—and remained highly effective under milder conditions (93% conversion at 120 °C; 53% yield at 0.1 MPa H2) (Fig. 8b). It exhibited a high TOF (5.42 h−1), low activation energy (35.6 kJ mol−1), and outstanding durability, maintaining activity for 100 h of continuous operation. DFT calculations revealed that the key hydrogenation step proceeds with a significantly lower barrier on Cu NCs (1.12 eV) than on extended Cu surfaces (1.93 eV), attributed to abundant low-coordination edge/corner sites. Combined effects of N–Cu stabilization, and mesoporous confinement enhance H2 activation and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O hydrogenation. Thus, hydrogenation can be achieved using Cu NCs under both homogeneous and heterogeneous conditions. However, variations in their structural architecture play a crucial role in determining their hydrogenation activity. Such effect is visible when Dong et al. reported an atomically precise chiral Cu NC, [Cu18H(PET)14(PPh3)6(NCS)3] (Cu18H) (NCS = isothiocyanate), featuring a Cu15 triple-helical core stabilized by PET, PPh3, and NCS ligands with one hydride.93 Structurally, Cu18H forms a three-layer chiral sandwich, which packs in the crystal as CCC- and AAA-enantiomers that assemble into a double-helical supramolecular structure supported by C–H⋯H–C, C–H/π, and C–H/S contacts. Catalytically, Cu18 shows exceptional electron-transfer activity in reducing [Fe(CN)6]3− → [Fe(CN)6]4−. Without the NC, the NaBH4-driven reduction requires ∼16 min, whereas Cu18H NC accelerates it to 14 s. The tightly packed double helix provides extended pathways for electron delocalization, analogous to DNA-mediated transfer, while the hydride ligand may assist in electron shuttling. UV-vis analysis confirmed that the cluster remains intact after reaction, highlighting its robustness and recyclability. So, Cu18H NC functions as a highly efficient electron-transfer catalyst, where 3D chirality and double-helical assembly directly enhance stability and reactivity.
O hydrogenation. Thus, hydrogenation can be achieved using Cu NCs under both homogeneous and heterogeneous conditions. However, variations in their structural architecture play a crucial role in determining their hydrogenation activity. Such effect is visible when Dong et al. reported an atomically precise chiral Cu NC, [Cu18H(PET)14(PPh3)6(NCS)3] (Cu18H) (NCS = isothiocyanate), featuring a Cu15 triple-helical core stabilized by PET, PPh3, and NCS ligands with one hydride.93 Structurally, Cu18H forms a three-layer chiral sandwich, which packs in the crystal as CCC- and AAA-enantiomers that assemble into a double-helical supramolecular structure supported by C–H⋯H–C, C–H/π, and C–H/S contacts. Catalytically, Cu18 shows exceptional electron-transfer activity in reducing [Fe(CN)6]3− → [Fe(CN)6]4−. Without the NC, the NaBH4-driven reduction requires ∼16 min, whereas Cu18H NC accelerates it to 14 s. The tightly packed double helix provides extended pathways for electron delocalization, analogous to DNA-mediated transfer, while the hydride ligand may assist in electron shuttling. UV-vis analysis confirmed that the cluster remains intact after reaction, highlighting its robustness and recyclability. So, Cu18H NC functions as a highly efficient electron-transfer catalyst, where 3D chirality and double-helical assembly directly enhance stability and reactivity.
Luo et al. reported another chiral [Cu57H20(PET)36(PPh3)4] (Cu57H20) NC and assessed its reactivity for hydrogenation reaction (Fig. 8c).94 SCXRD showed a Cu8 cubic core encapsulated by a Cu49(PET)36(PPh3)4 shell that contains a single Cu vacancy, creating a mono-lacunary defect analogous to polyoxometalates. This missing metal site exposes a μ3-hydride on a Cu3 triangle, a key structural feature absent in its fully occupied Ag analogue. Cu57H20 efficiently catalyzed the NaBH4 mediated hydrogenation of p-nitrophenol to p-aminophenol, with the 400 nm p-NP band disappearing and the 300 nm product band emerging, reaching complete conversion in 20 min (k = 0.18 min−1). No reaction occurred without the cluster. Post-catalysis UV-vis and TEM confirmed that the Cu57H20 NC remained intact. Although slightly slower than the smaller Cu11H3 catalyst, Cu57H20 significantly outperformed hydride-containing Ag clusters, highlighting the beneficial role of its vacancy defect and exposed hydride site in enabling efficient hydrogen transfer.
The effect of the structural architecture is more prominent when Liu et al. reported two nearly identical—[Cu20H9(Tf-dpf)10]+ (Cu20H9) and [Cu20H8(Tf-dpf)10]2+ (Cu20H8) (Tf-dpf = N,N′-di(5-trifluoromethyl-2-pyridyl)formamidinate) NCs—that differ by only a single hydride yet display strikingly different structures and reactivity.95 Their catalytic behaviour was evaluated in the selective hydrogenation of cinnamaldehyde to 3-phenylpropanal using Ph2SiH2. Cu20H8 showed excellent performance, giving 96.7% yield at 0.2 mol% loading within 4 h, whereas Cu20H9 produced only 3.7%, even with increased loading or extended time. ESI-MS revealed that Cu20H8 undergoes partial ligand dissociation, forming species such as [Cu20H8(Tf-dpf)9(EtO)(EtOH)]2+ during catalysis—evidence of accessible Cu sites. In contrast, Cu20H9 remains fully intact, showing no ligand loss. DFT calculations support this difference: Tf-dpf− dissociation is 0.3 eV more favorable in Cu20H8, explaining its ability to open coordination sites for substrate activation. Thus, despite their near-identical compositions, Cu20H8 is catalytically active due to ligand lability, whereas Cu20H9 is inert because of its overly rigid coordination environment. Basu et al. reported a BSA-stabilized, water-soluble Cu5 NC (<2 nm) in which Cu(I) centers are stabilized through coordination with thiol, amine, and carboxylate groups of the protein matrix.96 The catalytic activity of the BSA–Cu NCs was evaluated for the NaBH4-mediated reduction of 4-nitrophenol, where UV-vis monitoring showed the decay of the 400 nm 4-nitrophenolate band and the appearance of the 300 nm 4-aminophenol band (Fig. 8c). Li et al. developed an atomically precise Cu NCs featuring built-in frustrated Lewis pair (FLP) sites by anchoring methoxy-substituted thiolate ligands onto a Cu NC framework.97 The resulting cluster, [Cu34S7(RS)18(PPh3)4]2+ (Cu34) NC, contains a Cu21S5 core wrapped by a Cu13S2(RS)18(PPh3)4 shell. The methoxy groups create surface FLPs in which Cu(I) atoms function as Lewis acids and ligand O atoms as Lewis bases, enabling heterolytic H2 activation without forming acid–base adducts. For catalytic testing, linalool hydrogenation was chosen because it contains two sterically distinct C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds. The supported catalyst (Cu34/XC-72) delivered nearly quantitative conversion with >99% selectivity to 1,2-dihydrolinalool at room temperature (Fig. 8d). Other Cu NCs capped with nonfunctional ligands—and even a methoxy-thiolate-protected Cu9 cluster lacking frustrated Cu–O pairs—were completely inactive, confirming the necessity of FLP sites for H2 activation. Poisoning experiments using external Lewis acids (CO2, pyrrole) or bases (pyridine, NH3) strongly suppressed reactivity, verifying that Cu–O FLPs were the active centers. DFT calculations showed that hydrogenation to product A is favored by 2.95 eV and proceeds with a barrier of 2 eV lower than formation of the alternative regioisomer, explaining the exclusive selectivity. Overall, this study demonstrates that embedding FLP motifs directly onto Cu NC surfaces enables efficient and highly regioselective hydrogenation, establishing a well-defined model platform for heterogeneous FLP catalysis using earth-abundant metals.
C bonds. The supported catalyst (Cu34/XC-72) delivered nearly quantitative conversion with >99% selectivity to 1,2-dihydrolinalool at room temperature (Fig. 8d). Other Cu NCs capped with nonfunctional ligands—and even a methoxy-thiolate-protected Cu9 cluster lacking frustrated Cu–O pairs—were completely inactive, confirming the necessity of FLP sites for H2 activation. Poisoning experiments using external Lewis acids (CO2, pyrrole) or bases (pyridine, NH3) strongly suppressed reactivity, verifying that Cu–O FLPs were the active centers. DFT calculations showed that hydrogenation to product A is favored by 2.95 eV and proceeds with a barrier of 2 eV lower than formation of the alternative regioisomer, explaining the exclusive selectivity. Overall, this study demonstrates that embedding FLP motifs directly onto Cu NC surfaces enables efficient and highly regioselective hydrogenation, establishing a well-defined model platform for heterogeneous FLP catalysis using earth-abundant metals.
2.5. Indolizine
Wang et al. reported an atomically precise pyrazolate-protected Cu8 NC notable for its extraordinary chemical stability.98 The D2h-symmetric Cu8 ring, stabilized by eight strongly coordinating μ-pyrazolate ligands, resists conditions that typically decompose Cu NCs. Remarkably, Cu8 remained crystalline after exposure to boiling water, concentrated H2O2, 1 M HCl, and saturated KOH, and persisted in aerated solution for over 20 days without degradation. XPS confirmed Cu(I) retention even after months in air, and thermogravimetric analysis (TGA) indicated decomposition only near 300 °C. This exceptional robustness arises from the rigid, multidentate Cu–N coordination network and the hydrophobic ligand shell, which together block access of water, acids, bases, and oxidants to the metal core. DFT calculations further revealed a large HOMO–LUMO gap (4.63 eV), consistent with the cluster's remarkable kinetic inertness. They demonstrated its exceptional chemical robustness of Cu8 NC enables its use as an efficient homogeneous catalyst for the three-component synthesis of indolizines (Fig. 9). With only 0.5 mol% loading, Cu8 NC delivered high yields, whereas common Cu salts and other Cu NCs showed poor activity. The rigid pyrazolate framework imparts unusual resistance to acid, base, and oxidation, allowing Cu8 NC to remain structurally intact under reaction conditions. This stability underpins its superior performance, enabling gram-scale synthesis with catalyst loadings as low as 0.005 mol% (97% yield) and allowing more than ten recyclings without loss of activity. The reaction exhibited broad substrate scope: aryl alkynes with both electron-donating and electron-withdrawing groups gave excellent yields, while aliphatic alkynes performed moderately due to weaker π-coordination. A wide range of secondary amines—including cyclic and drug-derived substrates—also reacted efficiently. This study demonstrates that structural design and chemical stability—not merely metal content—are decisive factors in unlocking the catalytic potential of atomically precise Cu NCs.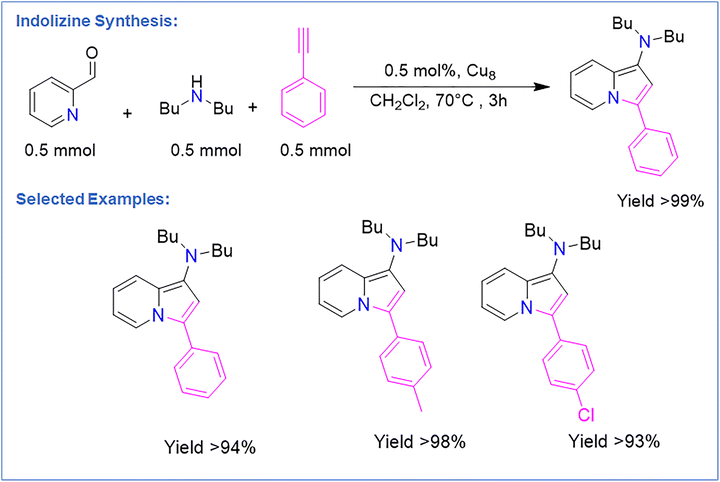 | ||
| Fig. 9 Representation of indolizine reaction and the performance of Cu8 NCs with corresponding yields by substrate variation. | ||
2.6. Oxidation
Bodiuzzaman et al. introduced an atomically precise Cu NC, [Cu13H10(Nap)3(PPh3)7] (Cu13Nap) (Nap = 1-naphthalene-thiolate), as an efficient photocatalyst for decarboxylative oxygenation under mild conditions (Fig. 10a).99 A structurally analogous cluster, [Cu13H10(DCBT)3(PPh3)7] (Cu13DCBT) (DCBT = 2,4-dichlorobenzenethiolate) served as a comparison but showed markedly lower activity (Fig. 12). Cu13Nap catalyzed the decarboxylative oxidation of Ibuprofen with 0.33 mol% loading under blue-light irradiation and O2, delivering the corresponding ketone in 90% yield. Among reported Cu NCs (Cu28, Cu29, Cu45, Cu57, Cu61), Cu13Nap showed the highest efficiency, whereas Cu13DCBT gave only 28% yield. The NC also displayed broad substrate scope, converting primary and secondary carboxylic acids—including heteroaryl, functionalized, aliphatic, and pharmaceutical derivatives—into aldehydes or ketones in up to 94% yield. Mechanistic studies ruled out SET pathways, as the oxidation potential of Cu13Nap was insufficient for direct substrate oxidation. Instead, radical traps, oxygen quenchers, NIR PL, and EPR experiments supported a singlet-oxygen (1O2) mediated mechanism. TD-DFT calculations further showed that the triplet excited state of Cu13Nap efficiently sensitizes O2 to 1O2, which drives decarboxylation followed by C–O bond formation. The superior performance of Cu13Nap relative to Cu13DCBT was attributed to stronger visible-light absorption and longer excited-state lifetimes imparted by the naphthalene thiolate ligands. Post-reaction ESI-MS analysis confirmed that the Cu13Nap NC retained its original molecular composition after 16 h of irradiation under catalytic conditions, demonstrating excellent structural stability during photocatalysis. Thus, this work establishes atomically precise Cu NCs as effective photosensitizers for 1O2-mediated decarboxylative oxygenation and expands the mechanistic landscape of Cu–NC photocatalysis beyond electron-transfer pathways.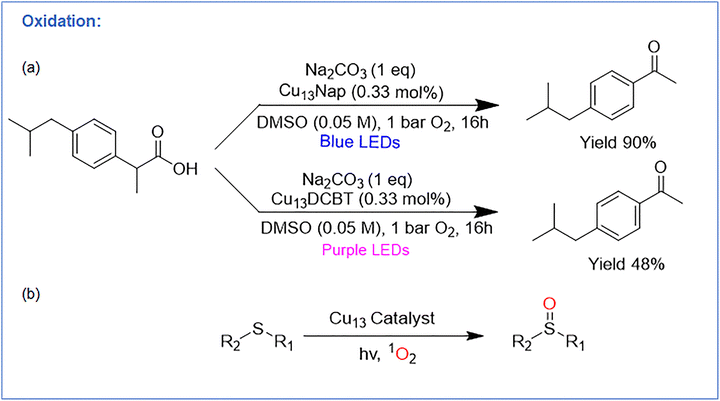 | ||
| Fig. 10 Representation of oxidation reaction and the performance of two Cu13 NCs with corresponding yields. | ||
Zhang et al. developed a solvent-controlled route to two quasi-isomeric Cu NCs, Cu13a and Cu13b, stabilized by 9-(prop-2-yn-1-yl)-9H-carbazole (CZ-PrAH) and p-tert-butylthiacalix[4]arene (H4TC4A) ligands.100 The isomerism arose from selective chloride release: CHCl3 furnished Cu13a with a coordinated Cl− ligand, whereas CH2Cl2 yielded Cl-free Cu13b. SCXRD showed both clusters possess similar Cu13 kernels with strong cuprophilic interactions, but the bound chloride in Cu13a induced greater structural distortion. Electronic analyses and TD-DFT revealed that chloride incorporation generated a distinct X(Cl) → M(Cu) charge-transfer transition, narrowing the HOMO–LUMO gap and enhancing spin–orbit coupling. As a result, Cu13a displayed more efficient intersystem crossing and stronger singlet-oxygen (1O2) sensitization, while Cu13b mainly produced superoxide (O2˙−). Both NCs photocatalyzed aerobic sulfide oxidation with full substrate conversion (Fig. 10b). However, Cu13a consistently delivered higher sulfoxide selectivity—for example, >99.9% for dimethyl sulfide compared to 93.4% for Cu13b. Scavenger experiments confirmed 1O2 as the dominant reactive species for Cu13a, whereas O2˙− contributed substantially in Cu13b. Oxygen exclusion suppressed reactivity, verifying O2 from air as the oxidant. Furthermore, PXRD analysis before and after catalysis confirmed that the NC framework remained unchanged, and both Cu13a and Cu13b maintained similar photocatalytic performance over multiple cycles, indicating good structural stability under reaction conditions. So, this study highlights how a single chloride ligand can finely modulate charge-transfer pathways, excited-state dynamics, and oxygen-species selectivity in atomically precise Cu13 photocatalysts.
Liu et al. reported the mixed-valence [Cu54S13O6(StBu)20(tBuSO3)12] (Cu54) (tBuSO3 = tert-butanesulfonate) NC, the largest atom-precise CuI/CuII cluster known to date.101 Its Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m cubic structure features a multilayer core–shell arrangement: a central sulfide anion inside a Cu12O6 dodecahedron, surrounded by an S12 icosahedron, and an outer shell of 42 Cu atoms coordinated by 20 StBu− and 12 tBuSO3− ligands. Spectroscopy indicates a CuI/CuII ratio of ∼38
m cubic structure features a multilayer core–shell arrangement: a central sulfide anion inside a Cu12O6 dodecahedron, surrounded by an S12 icosahedron, and an outer shell of 42 Cu atoms coordinated by 20 StBu− and 12 tBuSO3− ligands. Spectroscopy indicates a CuI/CuII ratio of ∼38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 (average oxidation state +1.3). Cu54 NC shows broad absorption up to 580 nm with a 2.05 eV band gap. When supported on titanium dioxide (TiO2), the composite Cu54/TiO2 exhibits excellent visible-light photocatalytic activity for phenol degradation. Upon irradiation (λ > 420 nm), phenol is rapidly converted to intermediates such as benzoquinone, hydroquinone, and catechol. The optimal 1.0 wt% Cu54/TiO2 achieves >95% phenol removal within 1.5 h, outperforming TiO2 alone. Mechanistically, both Cu54 and TiO2 are photoexcited, and because the Cu54 conduction band (−1.94 eV) is more negative than that of TiO2 (−1.16 eV), electrons flow from Cu54 to TiO2, generating O2˙− for phenol oxidation. Holes in the Cu54 valence band (+0.11 eV) further contribute to oxidation. This efficient charge separation boosts activity, while excessive Cu54 loading blocks TiO2 surface sites and lowers performance. The catalyst retains structure and activity over repeated cycles. Overall, this work establishes Cu54 NC as a robust mixed-valence photocatalyst and provides a promising design strategy for future Cu-based environmental catalysts.
16 (average oxidation state +1.3). Cu54 NC shows broad absorption up to 580 nm with a 2.05 eV band gap. When supported on titanium dioxide (TiO2), the composite Cu54/TiO2 exhibits excellent visible-light photocatalytic activity for phenol degradation. Upon irradiation (λ > 420 nm), phenol is rapidly converted to intermediates such as benzoquinone, hydroquinone, and catechol. The optimal 1.0 wt% Cu54/TiO2 achieves >95% phenol removal within 1.5 h, outperforming TiO2 alone. Mechanistically, both Cu54 and TiO2 are photoexcited, and because the Cu54 conduction band (−1.94 eV) is more negative than that of TiO2 (−1.16 eV), electrons flow from Cu54 to TiO2, generating O2˙− for phenol oxidation. Holes in the Cu54 valence band (+0.11 eV) further contribute to oxidation. This efficient charge separation boosts activity, while excessive Cu54 loading blocks TiO2 surface sites and lowers performance. The catalyst retains structure and activity over repeated cycles. Overall, this work establishes Cu54 NC as a robust mixed-valence photocatalyst and provides a promising design strategy for future Cu-based environmental catalysts.
2.7. Protosilylation
Wen et al. reported two atomically defined [Cu6(StBu)2(dppm)2(mnt)2]·CHCl3 (Cu6-1) and [Cu6(StBu)2(dppe)2(mnt)2] (Cu6-2) (mnt = maleonitriledithiolate, dppm = (bis(diphenylphosphino)methane), dppe = (1,2-bis(diphenylphosphino)ethane) NCs.102 SCXRD revealed that Cu6-1 features a fused Cu4 tetrahedral core capped by mnt and bisphosphine ligands, leaving two Cu sites exposed for catalysis. Compared with Cu6-2, Cu6-1 exhibits shorter Cu⋯Cu distances and stronger C–H⋯π interactions, giving markedly enhanced structural stability. Cu6-1 proved highly active for open-air protosilylation of C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C and C
C and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds (Fig. 11). Using HC
C bonds (Fig. 11). Using HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPh, (dimethylphenylsilyl)boronic acid pinacol ester (PhMe2SiBpin), 1,8-Diazabicycloundec-7-ene (DBU), and MeCN/MeOH, Cu6-1 afforded the (E)-silylalkene in 91% yield, outperforming Cu6-2 and larger NCs (Cu8, Cu15, Cu54) or classical Cu salts. These NCs tolerated a broad substrate scope—including aryl/alkyl acetylenes, TMS-alkynes, internal alkynes, alkenes, and enynes—producing trans-silylalkenes, silyl-allenes, and even disilylated allenes. The catalyst was robust under gram-scale conditions, delivering TON up to 14
CPh, (dimethylphenylsilyl)boronic acid pinacol ester (PhMe2SiBpin), 1,8-Diazabicycloundec-7-ene (DBU), and MeCN/MeOH, Cu6-1 afforded the (E)-silylalkene in 91% yield, outperforming Cu6-2 and larger NCs (Cu8, Cu15, Cu54) or classical Cu salts. These NCs tolerated a broad substrate scope—including aryl/alkyl acetylenes, TMS-alkynes, internal alkynes, alkenes, and enynes—producing trans-silylalkenes, silyl-allenes, and even disilylated allenes. The catalyst was robust under gram-scale conditions, delivering TON up to 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 with excellent functional-group compatibility, exhibiting excellent post-catalytic stability with no detectable structural change as confirmed by UV-vis, NMR and ESI-MS analyses. Silylalkene products were easily diversified through halogenation and Pd-catalyzed coupling. Importantly, Cu6-1 retained full activity over eight cycles with no structural change, while Cu6-2 progressively deactivated due to weaker intracluster interactions. DFT studies supported a dual-Cu mechanism: PhMe2SiBpin first forms a Cu–Si species, followed by alkyne coordination and StBu dissociation. A two-Cu migratory insertion step with a low barrier (23.1 kcal mol−1) accounts for the high activity and broad reactivity at room temperature.
200 with excellent functional-group compatibility, exhibiting excellent post-catalytic stability with no detectable structural change as confirmed by UV-vis, NMR and ESI-MS analyses. Silylalkene products were easily diversified through halogenation and Pd-catalyzed coupling. Importantly, Cu6-1 retained full activity over eight cycles with no structural change, while Cu6-2 progressively deactivated due to weaker intracluster interactions. DFT studies supported a dual-Cu mechanism: PhMe2SiBpin first forms a Cu–Si species, followed by alkyne coordination and StBu dissociation. A two-Cu migratory insertion step with a low barrier (23.1 kcal mol−1) accounts for the high activity and broad reactivity at room temperature.
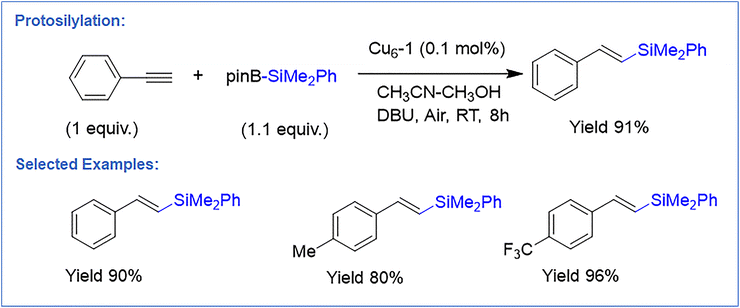 | ||
| Fig. 11 Representation of protosilylation reaction by Cu6-1 NCs with substrate scope and corresponding yields. | ||
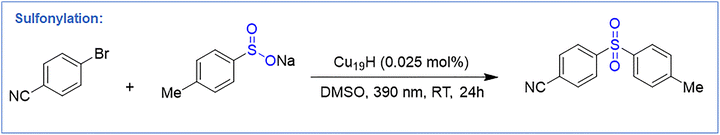
2.8. Sulfonylation
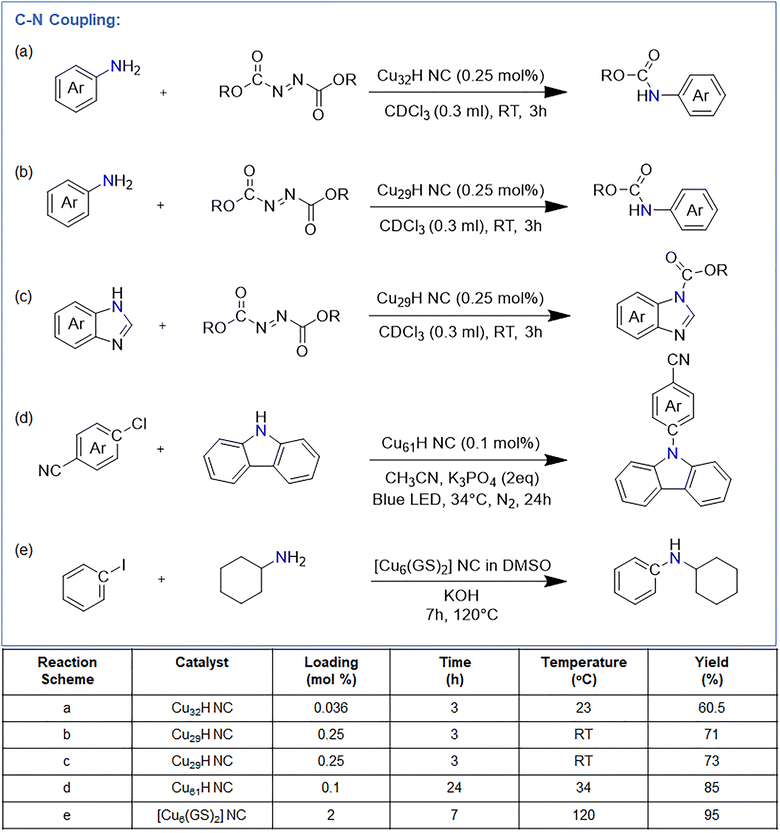
Alotaibi et al. reported [Cu19H6(StBuPhCH2)12(PPh3)6Br] (Cu19H) (StBuPhCH2 = (tert-butylthio)methylbenzene)) NC, featuring a Cu13 Ino-decahedral core capped by four ligand motifs, one bridging bromide, and six hydrides.103 A key structural feature is that three faces of the metal core remain exposed, providing accessible Cu sites for catalysis. They utilized this Cu19H NC as a highly efficient visible-light photocatalyst for the sulfonylation of aryl halides. Using only 0.025 mol% catalyst, 4-bromobenzonitrile and sodium 4-methylbenzenesulfinate were converted to 4-(methylsulfonyl)benzonitrile in 88% yield under 390 nm irradiation in DMSO (Fig. 12). No reaction occurred without light or catalyst, consistent with a photo-induced SET pathway. Cu19 outperformed CuX (X = Cl, Br or I) salts and other Cu clusters (Cu28, Cu29, Cu45, Cu58, Cu61), attributed to its unshielded Cu faces that directly interact with sulfinates. Immobilization on silica (Cu19–S) enabled heterogeneous catalysis with full recyclability over five cycles. The reaction showed broad scope: aryl bromides bearing electron-donating, electron-withdrawing, steric, or heteroaryl substituents (–CN, –OMe, –tBu, –F, –COPh, –CF3; pyridyl, quinolinyl, thiophenyl) afforded sulfones in 61–88% yield. Various sodium sulfinates (aryl and alkyl) also coupled efficiently (68–86% yield). Late-stage sulfonylation of drug-derived molecules proceeded in 71–74% yield, and the TOF was ∼400× higher than [Cu(CH3CN)4]BF4. Mechanistic experiments (radical clocks, olefin trapping, TEMPO quenching) confirmed a radical pathway. Upon irradiation, Cu19H NC forms a NC–SO2R species that undergoes SET to the aryl halide, generating an aryl radical and oxidized NC–SO2R+. Radical coupling or reductive elimination furnishes the sulfone and regenerates the cluster. DFT calculations supported favorable formation of NC–SO2R (ΔE = –0.98 eV) and its strong excited-state reducing power (–2.34 V vs. Ag/AgCl). This is the sole reported example of a Cu NC serving as a robust, reusable, visible-light-driven photocatalyst for broad C–S bond formation under mild conditions.3. Conclusion and future outlook
This review summarizes recent advances in Cu NC catalysis for organic transformations, highlighting how the evolution from early, unstable Cu–hydride species to robust, atomically precise, ligand-protected architectures has reshaped Cu catalysis. In contrast to conventional Cu salts and nanoparticles, Cu NCs possess discrete electronic structures, well-defined active sites, and tunable nuclearity, ligand environments, hydride content, and surface accessibility. These features enable direct structure–activity correlations and provide mechanistic insight that is largely inaccessible in traditional catalytic systems.Cu NCs display exceptional versatility across a broad range of reactions, including cycloadditions, C–C and C–N couplings, hydroboration, hydrogenation, oxidation, protosilylation, sulfonylation, and multicomponent transformations. Their catalytic performance is governed not merely by Cu identity but by subtle structural factors such as single-atom vacancies, ligand dynamics, mixed-valence states, hydride distribution, and exposed metal sites. Small structural variations can dramatically alter reaction pathways, switching between radical and polar mechanisms, modulating selectivity, and enhancing catalytic efficiency. Collectively, these studies establish Cu NCs as molecularly defined catalysts that bridge homogeneous, heterogeneous, and photocatalytic regimes.
Looking ahead, several directions are expected to drive further progress. Atomically precise bimetallic and multimetallic NCs offer opportunities to exploit synergistic effects and unlock new reactivity through controlled heterometal incorporation. Defect and vacancy engineering will continue to emerge as a powerful tool for tuning electronic structure and generating highly active catalytic sites. Advances in dynamic and adaptive ligand design are likely to enable stimulus-responsive catalysis and enzyme-like flexibility. Deeper understanding of hydride chemistry will expand Cu NC applications in selective hydrogenation and hydrogen-transfer processes, while integration of photocatalytic and electrocatalytic platforms may enable efficient solar- and electricity-driven transformations. Finally, addressing challenges related to stability, scalability, and catalyst recyclability will be essential for translating Cu NCs into practical and sustainable catalytic systems. Overall, Cu NCs represent a unique and rapidly advancing class of catalysts in which atomic precision enables rational design, mechanistic clarity, and exceptional performance. Given the central importance of organocatalytic transformations in modern synthetic chemistry, continued efforts toward developing well-defined catalytic systems are essential for achieving deeper mechanistic insight and improved control over catalytic activity and selectivity. Continued synergy between synthesis, advanced characterization, and theory will be critical for realizing their full potential in organic synthesis and beyond.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (grant no. 23H00289 and 22K19012), Scientific Research on Innovative Areas “Aquatic Functional Materials” (grant no. 22H04562), the Yazaki Memorial Foundation for Science and Technology, and the Ogasawara Foundation for the Promotion of Science and Engineering.References
- R. Jin, C. Zeng, M. Zhou and Y. Chen, Chem. Rev., 2016, 116, 10346–10413 CrossRef CAS PubMed.
- I. Chakraborty and T. Pradeep, Chem. Rev., 2017, 117, 8208–8271 CrossRef CAS PubMed.
- H. Hirai, S. Ito, S. Takano, K. Koyasu and T. Tsukuda, Chem. Sci., 2020, 11, 12233–12248 RSC.
- Y. Du, H. Sheng, D. Astruc and M. Zhu, Chem. Rev., 2019, 120, 526–622 CrossRef PubMed.
- Z. Hens and J. De Roo, J. Am. Chem. Soc., 2020, 142, 15627–15637 CrossRef CAS PubMed.
- R. Jin, Nanoscale, 2015, 7, 1549–1565 RSC.
- H. Qian, M. Zhu, Z. Wu and R. Jin, Acc. Chem. Res., 2012, 45, 1470–1479 CrossRef CAS PubMed.
- R. Jin, Y. Charles Cao, E. Hao, G. S. Métraux, G. C. Schatz and C. A. Mirkin, Nature, 2003, 425, 487–490 CrossRef CAS PubMed.
- X. Kang and M. Zhu, Chem. Soc. Rev., 2019, 48, 2422–2457 RSC.
- Y.-Z. Huang, R. K. Gupta, G.-G. Luo, Q.-C. Zhang and D. Sun, Coord. Chem. Rev., 2024, 499, 215508 CrossRef CAS.
- Y. Lu and W. Chen, Chem. Soc. Rev., 2012, 41, 3594–3623 RSC.
- H. Yu, B. Rao, W. Jiang, S. Yang and M. Zhu, Coord. Chem. Rev., 2019, 378, 595–617 CrossRef CAS.
- Z.-Y. Liu, Q.-B. Nie, B.-L. Han, R. K. Gupta, G.-L. Dong, G.-G. Luo, Z.-L. Yang and D. Sun, Chem. Soc. Rev., 2025, 54, 9092–9115 RSC.
- Y. Jin, C. Zhang, X.-Y. Dong, S.-Q. Zang and T. C. Mak, Chem. Soc. Rev., 2021, 50, 2297–2319 RSC.
- S. Biswas, S. Das and Y. Negishi, Coord. Chem. Rev., 2023, 492, 215255 CrossRef CAS.
- R. K. Gupta, Z. Wang, B. Mohan, C. H. Tung and D. Sun, Adv. Mater., 2024, 36, 2410054 CrossRef CAS PubMed.
- Y. Li, M. Zhou and R. Jin, Adv. Mater., 2021, 33, 2006591 CrossRef CAS PubMed.
- A. W. Cook and T. W. Hayton, Acc. Chem. Res., 2018, 51, 2456–2464 CrossRef CAS PubMed.
- Z. J. Guan, J. J. Li, F. Hu and Q. M. Wang, Angew. Chem., Int. Ed., 2022, 61, e202209725 CrossRef CAS PubMed.
- Q. Tang, G. Hu, V. Fung and D.-E. Jiang, Acc. Chem. Res., 2018, 51, 2793–2802 CrossRef CAS PubMed.
- Z. Lei, X.-K. Wan, S.-F. Yuan, Z.-J. Guan and Q.-M. Wang, Acc. Chem. Res., 2018, 51, 2465–2474 CrossRef CAS PubMed.
- S. Biswas, A. K. Das and S. Mandal, Acc. Chem. Res., 2023, 56, 1838–1849 CrossRef CAS PubMed.
- B. Zhang, J. Chen, Y. Cao, O. J. H. Chai and J. Xie, Small, 2021, 17, 2004381 CrossRef CAS PubMed.
- J. Yan, B. K. Teo and N. Zheng, Acc. Chem. Res., 2018, 51, 3084–3093 CrossRef CAS PubMed.
- T. Higaki, Q. Li, M. Zhou, S. Zhao, Y. Li, S. Li and R. Jin, Acc. Chem. Res., 2018, 51, 2764–2773 CrossRef CAS PubMed.
- Z. Lei, X.-K. Wan, S.-F. Yuan, J.-Q. Wang and Q.-M. Wang, Dalton Trans., 2017, 46, 3427–3434 RSC.
- Q. Yao, T. Chen, X. Yuan and J. Xie, Acc. Chem. Res., 2018, 51, 1338–1348 CrossRef CAS PubMed.
- Y.-P. Xie, J.-L. Jin, G.-X. Duan, X. Lu and T. C. Mak, Coord. Chem. Rev., 2017, 331, 54–72 CrossRef CAS.
- Q. Yao, M. Zhu, Z. Yang, X. Song, X. Yuan, Z. Zhang, W. Hu and J. Xie, Nat. Rev. Mater., 2025, 10, 89–108 CrossRef CAS.
- S. Maity, S. Kolay, S. Chakraborty, A. Devi and A. Patra, Chem. Soc. Rev., 2025, 54, 1785–1844 RSC.
- J.-H. Huang, Y. Cui, Z.-Y. Wang and S.-Q. Zang, Acc. Chem. Res., 2025, 58, 1249–1261 CrossRef CAS PubMed.
- J. Qian, Z. Yang, J. Lyu, Q. Yao and J. Xie, Precision Chem., 2024, 2, 495–517 CrossRef CAS PubMed.
- Z. Yang, Y. Wang, R. Zhang, T. Chen and J. Xie, Adv. Mater., 2025, e08578 CrossRef CAS PubMed.
- S. Biswas, Y. Shingyouchi, M. Ogami, M. Kamiyama, T. Kawawaki and Y. Negishi, EcoEnergy, 2024, 2, 400–418 CrossRef CAS.
- Z.-H. Zheng, F.-X. Xiao and Y.-J. Xu, Small, 2025, 21, 2505282 CrossRef CAS PubMed.
- X. Du and R. Jin, ACS Nano, 2019, 13, 7383–7387 CrossRef CAS PubMed.
- S. Rodríguez-Nuévalos, M. Espinosa and A. Leyva-Pérez, Commun. Chem., 2024, 7, 76 CrossRef PubMed.
- X. Pan, Y. Yao, M. Zhang, X. Yuan, Q. Yao and W. Hu, Nanoscale, 2024, 16, 8196–8215 RSC.
- J. Q. Fan, J. X. Guo and M. B. Li, Chem. – Eur. J., 2025, e02620 CrossRef CAS PubMed.
- D. Yang, J. Wang, Q. Wang, Z. Yuan, Y. Dai, C. Zhou, X. Wan, Q. Zhang and Y. Yang, ACS Nano, 2022, 16, 15681–15704 CrossRef CAS PubMed.
- P. Kumar and M. Nemiwal, Chem. Asia. J., 2024, 19, e202400062 CrossRef CAS PubMed.
- R. K. Gupta, Z. Wang, B. Mohan, C. H. Tung and D. Sun, Adv. Funct. Mater., 2025, 2507047 CrossRef CAS.
- X. Liu and D. Astruc, Coord. Chem. Rev., 2018, 359, 112–126 CrossRef CAS.
- M. Kamiyama, Y. Shingyouchi, R. Sarma, M. Ghosh, T. Kawawaki, S. Biswas and Y. Negishi, Chem. Commun., 2025, 61, 1048–1062 RSC.
- A. Baghdasaryan and T. Bürgi, Nanoscale, 2021, 13, 6283–6340 RSC.
- M. Chen, C. Guo, L. Qin, L. Wang, L. Qiao, K. Chi and Z. Tang, Nano-Micro Lett., 2025, 17, 83 CrossRef CAS PubMed.
- A. K. Das, S. Biswas, V. S. Wani, A. S. Nair, B. Pathak and S. Mandal, Chem. Sci., 2022, 13, 7616–7625 RSC.
- A. K. Das, S. Hegde, T. J. Woods, V. S. Wani and M. P. Backlund, Nano Lett., 2025, 25, 7491–7498 CrossRef CAS PubMed.
- A. Jana, S. Duary, A. Das, A. R. Kini, S. Acharya, J. Machacek, B. Pathak, T. Base and T. Pradeep, Chem. Sci., 2024, 15, 13741–13752 RSC.
- A. Patra, K. Shinde, S. Mandal, A. Kudlu, W.-D. Si, M. Francis, A. Das, D. Sun and S. Roy, Inorg. Chem., 2025, 64, 20666–20678 CrossRef CAS PubMed.
- B. L. Han, F. Alkan, Z. R. Yuan, P. Mahato, Z. Wang, C. H. Tung and D. Sun, Angew. Chem., Int. Ed., 2025, 64, e202507412 CrossRef CAS PubMed.
- H. Zhao, C. Zhang, B.-L. Han, P. Mahato, C.-Z. Yang, P.-X. Yu, Z. Wang, C.-H. Tung and D. Sun, Nano Lett., 2025, 25, 9132–9138 CrossRef CAS PubMed.
- S. Biswas, S. Das and Y. Negishi, Nanoscale Horiz., 2023, 8, 1509–1522 RSC.
- S. Biswas, Y. Shingyouchi, M. Kamiyama, M. K. Jena, M. Ogami, T. Kawawaki, B. Pathak and Y. Negishi, Small, 2025, 21, 2500302 CrossRef CAS PubMed.
- S. Biswas, T. Tanaka, H. Song, M. Ogami, Y. Shingyouchi, S. Hossian, M. Kamiyama, T. Kosaka, R. Nakatani, Y. Niihori, S. Das, T. Kawawaki, D.-E. Jiang and Y. Negishi, Small Sci., 2025, 5, 2400465 CrossRef CAS PubMed.
- S. Biswas, Y. Shingyouchi, M. Kamiyama, M. Ogami, H. Song, B. Li, S. Wang, T. Kawawaki, D.-E. Jiang and Y. Negishi, J. Am. Chem. Soc., 2025, 147, 23733–23742 CrossRef CAS PubMed.
- Y. Shingyouchi, M. Ogami, S. Biswas, T. Tanaka, M. Kamiyama, K. Ikeda, S. Hossain, Y. Yoshigoe, D. Osborn and G. F. Metha, Small, 2025, 21, 2409910 CrossRef CAS PubMed.
- J. Oliver-Messeguer, L. Liu, S. García-García, C. Canós-Giménez, I. Domínguez, R. Gavara, A. Doménech-Carbó, P. Concepción, A. Leyva-Pérez and A. Corma, J. Am. Chem. Soc., 2015, 137, 3894–3900 CrossRef PubMed.
- R. Greco, E. Tiburcio, B. Palomar-De Lucas, J. Ferrando-Soria, D. Armentano, E. Pardo and A. Leyva-Pérez, Mol. Catal., 2022, 522, 112228 CAS.
- A. Pal, S. Biswas, B. Sahoo and Y. Negishi, Synlett, 2025, 1123–1129 CAS.
- A. Sagadevan, K. Murugesan, O. M. Bakr and M. Rueping, Chem. Commun., 2024, 60, 13858–13866 RSC.
- R. S. Dhayal, W. E. van Zyl and C. W. Liu, Acc. Chem. Res., 2016, 49, 86–95 CrossRef CAS PubMed.
- P.-K. Liao, C.-S. Fang, A. J. Edwards, S. Kahlal, J.-Y. Saillard and C. W. Liu, Inorg. Chem., 2012, 51, 6577–6591 CrossRef CAS PubMed.
- R. S. Dhayal, J.-H. Liao, Y.-R. Lin, P.-K. Liao, S. Kahlal, J.-Y. Saillard and C. W. Liu, J. Am. Chem. Soc., 2013, 135, 4704–4707 CrossRef CAS PubMed.
- J.-H. Liao, R. S. Dhayal, X. Wang, S. Kahlal, J.-Y. Saillard and C. W. Liu, Inorg. Chem., 2014, 53, 11140–11145 CrossRef CAS PubMed.
- A. J. Edwards, R. S. Dhayal, P. K. Liao, J. H. Liao, M. H. Chiang, R. O. Piltz, S. Kahlal, J. Y. Saillard and C. W. Liu, Angew. Chem., Int. Ed., 2014, 53, 7214–7218 CrossRef CAS PubMed.
- R. S. Dhayal, J. H. Liao, X. Wang, Y. C. Liu, M. H. Chiang, S. Kahlal, J. Y. Saillard and C. W. Liu, Angew. Chem., Int. Ed., 2015, 54, 13604–13608 CrossRef CAS PubMed.
- S. Biswas and Y. Negishi, Dalton Trans., 2024, 53, 9657–9663 RSC.
- S. Biswas and Y. Negishi, J. Phys. Chem. Lett., 2024, 15, 947–958 CrossRef CAS PubMed.
- X. Ge, S. Zeng, H. Deng, B. K. Teo and C. Sun, Coord. Chem. Rev., 2024, 503, 215667 CrossRef CAS.
- M. Ghosh, R. Sarma, M. Kamiyama, T. Kawawaki, S. Biswas and Y. Negishi, Nanoscale Horiz., 2025, 10, 1250–1267 RSC.
- W. S. Mahoney, D. M. Brestensky and J. M. Stryker, J. Am. Chem. Soc., 1988, 110, 291–293 CrossRef CAS.
- B. H. Lipshutz and B. A. Frieman, Synfacts, 2006, 0047 CrossRef.
- C. Deutsch, N. Krause and B. H. Lipshutz, Chem. Rev., 2008, 108, 2916–2927 CrossRef CAS PubMed.
- T. H. Lemmen, K. Folting, J. C. Huffman and K. G. Caulton, J. Am. Chem. Soc., 1985, 107, 7774–7775 CrossRef CAS.
- A. W. Cook, Z. R. Jones, G. Wu, S. L. Scott and T. W. Hayton, J. Am. Chem. Soc., 2018, 140, 394–400 CrossRef CAS PubMed.
- G. Yang, Y. Xie, Y. Wang, Y. Tang, L. L. Chng, F. Jiang, F. Du, X. Zhou, J. Y. Ying and X. Yuan, Nano Res., 2023, 16, 1748–1754 CrossRef CAS.
- H. Zhou, T. Yang, H. Deng, Y. Yun, S. Jin, L. Xiong and M. Zhu, Nanoscale, 2024, 16, 10318–10324 RSC.
- Y.-Q. Xiao, P. Shang, X.-Y. Chen, X.-Q. Pu, K.-W. Jiang and X.-F. Jiang, Chem. Mater., 2024, 36, 2027–2038 CrossRef CAS.
- C. Dong, R. W. Huang, A. Sagadevan, P. Yuan, L. Gutiérrez-Arzaluz, A. Ghosh, S. Nematulloev, B. Alamer, O. F. Mohammed, I. Hussain, M. Rueping and O. M. Bakr, Angew. Chem., Int. Ed., 2023, 62, e202307140 CrossRef CAS PubMed.
- T. Jia, Y.-X. Li, X.-H. Ma, M.-M. Zhang, X.-Y. Dong, J. Ai and S.-Q. Zang, Nat. Commun., 2023, 14, 6877 CrossRef CAS PubMed.
- L.-J. Liu, M.-M. Zhang, Z. Deng, L.-L. Yan, Y. Lin, D. L. Phillips, V. W.-W. Yam and J. He, Nat. Commun., 2024, 15, 4688 CrossRef CAS PubMed.
- S. Nematulloev, A. Sagadevan, B. Alamer, A. Shkurenko, R. Huang, J. Yin, C. Dong, P. Yuan, K. E. Yorov, A. A. Karluk, W. J. Mir, B. E. Hasanov, M. N. Hedhili, N. M. Halappa, M. Eddaodi, O. F. Mohammed, M. Rueping and O. M. Bakr, Angew. Chem., Int. Ed., 2023, 62, e202303572 CrossRef CAS PubMed.
- S. Biswas, A. Pal, M. K. Jena, S. Hossain, J. Sakai, S. Das, B. Sahoo, B. Pathak and Y. Negishi, J. Am. Chem. Soc., 2024, 146, 20937–20944 CrossRef CAS PubMed.
- S. Lee, M. S. Bootharaju, G. Deng, S. Malola, W. Baek, H. Hakkinen, N. Zheng and T. Hyeon, J. Am. Chem. Soc., 2020, 142, 13974–13981 CrossRef CAS PubMed.
- A. K. Das, S. Biswas, A. Pal, S. S. Manna, A. Sardar, P. K. Mondal, B. Sahoo, B. Pathak and S. Mandal, Nanoscale, 2024, 16, 3583–3590 RSC.
- A. Sagadevan, A. Ghosh, P. Maity, O. F. Mohammed, O. M. Bakr and M. Rueping, J. Am. Chem. Soc., 2022, 144, 12052–12061 CrossRef CAS PubMed.
- B. Mondal, K. Basu, R. Jana, P. Mondal, B. Hansda, A. Datta and A. Banerjee, ACS Appl. Nano Mater., 2022, 5, 7932–7943 CrossRef CAS.
- B. Alamer, A. Sagadevan, M. Bodiuzzaman, K. Murugesan, S. Alsharif, R.-W. Huang, A. Ghosh, M. H. Naveen, C. Dong, S. Nematulloev, J. Yin, A. Shkurenko, M. Abulikemu, X. Dong, Y. Han, M. Eddaudi, M. Rueping and O. M. Bakr, J. Am. Chem. Soc., 2024, 146, 16295–16305 CrossRef CAS PubMed.
- T. Jia, J. Ai, X. Li, M.-M. Zhang, Y. Hua, Y.-X. Li, C.-F. Sun, F. Liu, R.-W. Huang, Z. Wang and S.-Q. Zang, Nat. Commun., 2024, 15, 9551 CrossRef CAS PubMed.
- C. Sun, N. Mammen, S. Kaappa, P. Yuan, G. Deng, C. Zhao, J. Yan, S. Malola, K. Honkala, H. Häkkinen, B. K. Teo and N. Zheng, ACS Nano, 2019, 13, 5975–5986 CrossRef CAS PubMed.
- F. Lan, J. Aarons, Y. Shu, X. Zhou, H. Jiao, H. Wang, Q. Guan and W. Li, Appl. Catal., B, 2021, 299, 120651 CrossRef CAS.
- G. Dong, Z. Pan, B. Han, Y. Tao, X. Chen, G.-G. Luo, P. Sun, C. Sun and D. Sun, Angew. Chem., Int. Ed., 2023, 62, e202302595 CrossRef CAS PubMed.
- G.-G. Luo, Z.-H. Pan, B.-L. Han, G.-L. Dong, C.-L. Deng, M. Azam, Y.-W. Tao, J. He, C.-F. Sun and D. Sun, Angew. Chem., Int. Ed., 2023, 62, e202306849 CrossRef CAS PubMed.
- C.-Y. Liu, T.-Y. Liu, Z.-J. Guan, S. Wang, Y.-Y. Dong, F. Hu, D.-E. Jiang and Q.-M. Wang, CCS Chem., 2024, 6, 1581–1590 CrossRef CAS.
- K. Basu, S. Paul, R. Jana, A. Datta and A. Banerjee, ACS Sustainable Chem. Eng., 2019, 7, 1998–2007 CrossRef CAS.
- S. Li, Q. Wu, X. You, X. Ren, P. Du, F. Li, N. Zheng and H. Shen, J. Am. Chem. Soc., 2024, 146, 27852–27860 CrossRef CAS PubMed.
- Y.-M. Wang, X.-C. Lin, K.-M. Mo, M. Xie, Y.-L. Huang, G.-H. Ning and D. Li, Angew. Chem., Int. Ed., 2023, 62, e202218369 CrossRef CAS PubMed.
- M. Bodiuzzaman, K. Murugesan, P. Yuan, B. Maity, A. Sagadevan, N. Malenahalli H, S. Wang, P. Maity, M. F. Alotaibi, D. E. Jiang, M. Abulikemu, O. F. Mohammed, L. Cavallo, M. Rueping and O. M. Bakr, J. Am. Chem. Soc., 2024, 146, 26994–27005 CrossRef CAS PubMed.
- C. Zhang, Z. Wang, W. D. Si, L. Wang, J. M. Dou, Z. Y. Gao, C. H. Tung and D. Sun, ACS Nano, 2022, 16, 9598–9607 CrossRef CAS PubMed.
- L.-J. Liu, J.-W. Zhang, M. Asad, Z.-Y. Wang, S.-Q. Zang and T. C. Mak, Chem. Commun., 2021, 57, 5586–5589 RSC.
- S. Wen, C. Zhang, L. J. Liu, Z. Wang, D. Sun and J. He, Angew. Chem., Int. Ed., 2025, 64, e202416851 CrossRef CAS PubMed.
- M. F. Alotaibi, A. Sagadevan, P. Yuan, M. Bodiuzzaman, K. Murugesan, R. Zhou, J. Yin, S. Thomas, R. W. Huang, N. M. Halappa, C. Dong, M. Abulikemu, H. N. Alshareef, O. F. Mohammed, M. Rueping and O. M. Bakr, J. Am. Chem. Soc., 2025, 147, 28932–28942 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |