Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dual-temperature photothermal tandem catalysis for CO2 conversion to olefins
Ziyang
Li
a,
Yuhang
Qi
 *a,
Lipu
Han
a,
Chu
Zhang
a,
Zhenwen
Yang
a,
Zhike
Wu
a and
Shanshan
Xu
*a,
Lipu
Han
a,
Chu
Zhang
a,
Zhenwen
Yang
a,
Zhike
Wu
a and
Shanshan
Xu
 *bc
*bc
aSchool of Electronics and Information Engineering, Hebei University of Technology, Tianjin 300130, China. E-mail: yuhangqi@hebut.edu.cn
bDepartment of Materials, School of Natural Science, The University of Manchester, Oxford Road, Manchester M13 9PL, UK. E-mail: shanshan.xu@manchester.ac.uk
cUniversity of Manchester at Harwell, Harwell Science and Innovation Campus, Didcot, Oxfordshire OX11 0DE, UK
First published on 18th March 2026
Abstract
We report a dual-temperature photothermal tandem system that overcomes temperature mismatch for CO2 hydrogenation to light olefins in a single reactor. A graphene-modified In2O3 top bed enables higher-temperature RWGS, and a K-doped Fe2O3 bottom bed facilitates lower-temperature FTS. This vertical configuration couples the two reactions, achieving a light-olefin production rate of 52.1 µmol g−1 h−1 with 69% selectivity at 0.05 MPa without external heating.
The increasing atmospheric CO2 level is aggravating climate change and energy insecurity, highlighting the urgent need for sustainable carbon-recycling technologies.1,2 Among the various strategies, photothermal CO2 reduction has attracted growing attention because it does not require external electrical input and enables full-spectrum solar utilization to activate inert CO2 molecules.3,4 Significant progress has been achieved in CO2 hydrogenation to C1 products such as CH4, CO, and CH3OH.5,6 However, overcoming the kinetic barriers associated with C–C bond formation to directly produce C2+ hydrocarbons remains highly challenging.7,8
For thermocatalytic CO2 hydrogenation towards C2+ products, two tandem strategies are most frequently explored. One approach involves coupling CO2-to-methanol with a subsequent methanol-to-olefins (MTO) process.9,10 Although this route can yield light olefins, methanol selectivity is highly pressure-dependent. It typically requires elevated pressures to suppress methanol decomposition, which is unfavourable for scalable operations under photothermal conditions. Another widely studied route converts CO2 to CO via the reverse water–gas shift (RWGS) reaction, followed by Fischer–Tropsch synthesis (FTS) to form hydrocarbons.11–13 Integrating RWGS and FTS in a single reactor is intrinsically difficult because the two steps require different optimal temperature windows: RWGS is endothermic and favours high temperatures >400 °C for efficient CO formation,14,15 whereas FTS requires moderate temperatures (200–350 °C) to promote C–C coupling and avoid excessive methane formation.16,17 This fundamental temperature mismatch is a key limitation to achieving direct CO2-to-olefin conversion in a single photothermal reactor.
Compared with the recently reported single-reactor tandem photothermal system,18 this work employs a dual-bed reactor in which the RWGS and FTS functions are spatially separated. This design enables cascade heat utilization, suppresses local overheating, and allows independent optimization of each catalytic bed for improved flexibility in tuning target products. The top layer, made of graphene-modified In2O3 (G–In2O3), exhibits excellent photothermal conversion and efficiently reduces CO2 to CO while simultaneously heating the gas phase and transferring heat to the downstream bed via combined radiative and convective heat transfer. This controlled heat flow determines the lower temperature of the potassium-doped Fe2O3 (K–Fe2O3) bed, where CO undergoes FTS to produce light olefins with high selectivity. The vertically stacked configuration establishes a well-defined temperature gradient, enabling the RWGS and FTS stages to operate within their respective optimal temperature ranges. Under low pressure (0.05 MPa), the tandem system achieves a light-olefin production rate of 52.1 µmol g−1 h−1 with 69% selectivity, demonstrating that engineering a temperature gradient is an effective strategy to overcome the inherent temperature mismatch in tandem photothermal catalysis.
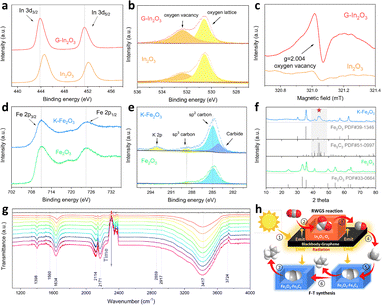
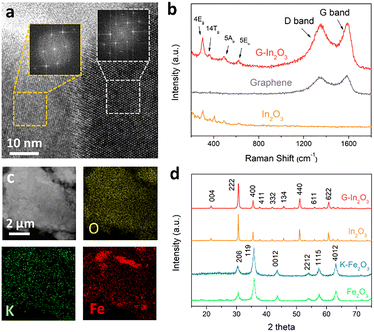
The G–In2O3 composite in the top bed was prepared by using the hydrothermal method. The average particle size of In2O3 particles is 50–100 nm, and they are uniformly dispersed on the graphene sheets (Fig. S1). The high-resolution transmission electron microscopy (HRTEM) image (Fig. 1a) shows a distinct interface between graphene and In2O3. The corresponding fast Fourier transform patterns provide atomic-resolution structural information: the cubic lattice spots can be assigned to In2O3, while the hexagonal pattern arises from the characteristic six-fold symmetry of graphene. Raman spectroscopy further confirms the formation of the G–In2O3 composite (Fig. 1b). The D band at ∼1300 cm−1 and the G band at ∼1400 cm−1 are characteristic of graphene, whereas the peaks at 307, 366, 494, and 628 cm−1 are aligned with the In–O vibrations of InO6 structural units in the body-centred cubic In2O3 structure.
Potassium modification of Fe2O3 in the lower catalyst bed was verified by SEM and elemental mapping, which shows that K is homogeneously distributed on Fe2O3 (Fig. 1c). TEM and HRTEM analyses indicate that the lattice spacings of K–Fe2O3 are 0.251, 0.208, and 0.295 nm, corresponding to the (311), (400), and (220) crystal planes of γ-Fe2O3 (Fig. S2). As shown in the X-ray diffraction (XRD) patterns (Fig. 1d), all diffraction peaks can be indexed to Fe2O3 (JCPDS No. 39-1346) and In2O3 (JCPDS No. 71-2194),19,20 indicating that the crystalline structures of both oxides are preserved after graphene and K modification.
In the catalyst selection for this dual-bed reactor, In2O3 was chosen because it is an efficient catalyst for the RWGS reaction and can produce CO with nearly 100% selectivity. The generated CO acts as the substrate for the FTS reaction, thereby initiating the tandem reaction. Fe2O3 was selected instead of Fe5C2 because it offers advantages such as facile preparation, low cost, and good structural stability. In addition, Fe2O3 can be transformed into the active Fe5C2 phase through in situ phase conversion under reaction conditions.
To determine the optimal composition for CO2 conversion, we evaluated G–In2O3 composites with various graphene loadings in the photothermal RWGS reaction (Fig. S3) and K-modified Fe2O3 with different K contents in FTS (Fig. S4). As shown in Fig. S5, all the G–In2O3 composites produce CO as the only product, confirming that graphene incorporation does not alter the reaction pathway. The CO production rate increases progressively, reaching a maximum for G–In2O3-10%, but drops when the loading is further increased to 15 wt%. At moderate loadings (<10 wt%), graphene improves the dispersion of In2O3 and its specific surface area (Fig. S6) and enhances CO2 adsorption and activation (Fig. S7).21 The strong broadband absorption of graphene raises the catalyst surface temperature under illumination, thereby accelerating the endothermic RWGS reaction (Fig. S8).22 Graphene also suppresses non-radiative recombination of photogenerated carriers in In2O3, resulting in a higher photothermal conversion efficiency (Fig. S9).23,24 Such effects contribute to the enhanced CO production at the optimal 10 wt% loading. However, excessive graphene loadings will block In2O3 active sites and reduce the accessible surface area for gas adsorption, leading to reduced CO production.
In the following FTS reaction, introducing potassium significantly changes both hydrocarbon distribution and light-olefin productivity. As shown in Fig. S10, the K–Fe2O3 catalyst containing 2.5 wt% K exhibits the highest light-olefin production rate (i.e., 0.55 mmol g−1 h−1 for C2H4 and 0.37 mmol g−1 h−1 for C3H6). In contrast, although the overall olefin production rate declines at higher K loadings, the K–Fe2O3 with 5 wt% K catalyst shows the highest selectivity toward C2–3![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) olefins (51%), accompanied by a suppressed formation of CH4. A moderate amount of potassium improves the catalytic performance by slightly reducing the photoluminescence intensity of Fe2O3 (Fig. S11) and enhancing CO adsorption and activation (Fig. S12). However, excessive K loading further neutralises surface acidity and suppresses H2 chemisorption, thus lowering the FTS reaction rate and shifting the product distribution away from CH4 and toward heavier hydrocarbons and a higher olefin fraction, which agrees with the trends in Fig. S10.25,26 Furthermore, the photothermal FTS performance of K–Fe2O3 was evaluated under identical conditions while varying the light intensity, as detailed in Fig. S13.
olefins (51%), accompanied by a suppressed formation of CH4. A moderate amount of potassium improves the catalytic performance by slightly reducing the photoluminescence intensity of Fe2O3 (Fig. S11) and enhancing CO adsorption and activation (Fig. S12). However, excessive K loading further neutralises surface acidity and suppresses H2 chemisorption, thus lowering the FTS reaction rate and shifting the product distribution away from CH4 and toward heavier hydrocarbons and a higher olefin fraction, which agrees with the trends in Fig. S10.25,26 Furthermore, the photothermal FTS performance of K–Fe2O3 was evaluated under identical conditions while varying the light intensity, as detailed in Fig. S13.
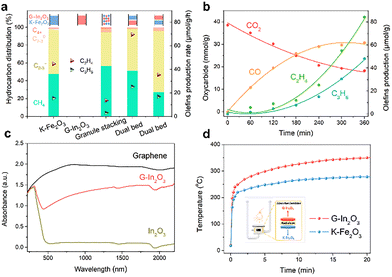
After optimising the compositions of G–In2O3 and K–Fe2O3, we evaluated tandem photothermal CO2 hydrogenation over the G–In2O3-10% and K–Fe2O3-5% catalyst. The hydrocarbon selectivity and product distribution depend strongly on the integration configuration of the two components. The single K–Fe2O3 component shows low activity and poor olefin selectivity (i.e., 50%) because it cannot accumulate sufficiently high CO concentrations, allowing the Sabatier reaction to proceed and producing CH4 (i.e., 48%) as the product (Fig. 2a and Fig. S14). In contrast, single In2O3 primarily generates CO as a product with negligible hydrocarbons, as the low-pressure and high-temperature conditions are unfavourable for methanol or olefin formation. Simply mixing two components physically (granule stacking) shows only a minor tandem effect, indicating that excessive proximity does not enable optimal photothermal CO2-to-olefin performance.
Therefore, we adopt a dual-bed configuration, with G–In2O3-10% in the upper layer and K–Fe2O3-5% in the lower layer, separated by an inert screen (Fig. 2a). When K–Fe2O3-5% is positioned beneath G–In2O3-10%, the light-olefin production rate achieved was 52.1 µmol g−1 h−1 with a selectivity of 69%. In contrast, reversing the order—placing K–Fe2O3-5% above G–In2O3-10%—resulted in a significantly lower olefin productivity, confirming that efficient tandem CO2 reduction and F–T coupling require the correct thermal and reactant-flow sequence. This improvement arises from the temperature distribution and reaction dynamics revealed in Fig. 2b. In the dual-bed configuration, the upper G–In2O3-10% layer, operating at a higher photothermal temperature, rapidly converts CO2 to CO and steadily increases the CO concentration throughout the reaction (Fig. 2b). As CO continues to accumulate, the concentration surpasses the threshold required to trigger chain-growth reactions in the downstream FTS bed. Olefin production begins to rise significantly after 120 min and continues to increase over time. Meanwhile, the CO formation rate gradually approaches a quasi-steady state, while CO2 is continuously consumed. These results demonstrate that the upper G–In2O3 bed must supply CO with high selectivity and rate to initiate and sustain FTS in the lower K–Fe2O3 bed, thereby enabling efficient tandem photothermal CO2-to-olefin conversion. Detailed analysis of the stability test is provided in Fig. S15 of the SI.
As a result, the photothermal conversion efficiency of the upper G–In2O3 layer is crucial. The light source primarily irradiates the top bed containing G–In2O3. G–In2O3 was selected because the tandem process is initiated by light-driven RWGS, which produces CO as the key intermediate for the subsequent FTS reaction in the lower bed. Positioning In2O3 in the upper bed promotes the formation of a CO-rich stream under continuous unidirectional gas flow. Moreover, UV-Vis-NIR spectra (Fig. 2c) show that pristine In2O3, as a wide-band-gap semiconductor (3.2 eV, Fig. S16), shows weak absorption across visible and near-infrared regions. Graphene endows G–In2O3 with strong, nearly broadband light absorption, enabling efficient photothermal conversion. The heat generated in the upper bed can also be transferred to the downstream K–Fe2O3 bed through thermal radiation and gas-phase convection, helping to maintain the temperature required for the tandem reaction. The resulting thermal distribution in the dual-bed configuration is confirmed under photothermal operation (Fig. 2d): the upper G–In2O3 layer rapidly reaches ∼360 °C within 10 min, while the lower K–Fe2O3 layer stabilises at ∼260 °C. The respective temperatures also precisely correspond to the optimal adsorption range (Fig. S17).27
To identify the active sites in G–In2O3 for CO2 reduction, XPS and EPR measurements were carried out. In comparison, the corresponding peaks of G–In2O3 shift to lower binding energies, which can be attributed to electron transfer at the graphene/In2O3 interface and an increased concentration of oxygen vacancies (Fig. 3a). The O 1s spectra (Fig. 3b) can be deconvoluted into two contributions: the peak at 529.8 eV is assigned to lattice oxygen in In–O–In, whereas the peak at 531.4 eV is associated with oxygen vacancies.28 Furthermore, the stronger EPR signal for G–In2O3 shows that it has a higher oxygen-vacancy level (Fig. 3c),29 aligning with the XPS results. This suggested that carbon at the graphene/In2O3 interface lowers the energy needed to create oxygen vacancies in the In2O3 lattice.
Because the oxygen vacancy concentration evolves under photothermal reaction conditions, we compared the XRD, PL, EPR and UV-vis-NIR spectra of G–In2O3 before and after the reaction (Fig. S18–S21). Detailed characterization and analysis are provided in the SI.
Regarding the active phase of the K–Fe2O3 catalyst in the FTS reaction, XPS measurements were carried out to determine the surface elemental composition and chemical states (Fig. 3d). In the Fe 2p spectrum, upon K promotion, the Fe 2p3/2 peak shifts to a lower binding energy, indicating a more reduced Fe environment and thus facilitating the transformation of Fe species into Fe5C2, which is widely recognised as the active phase for FTS in Fe-based catalysts.30–32 In the C 1s spectrum (Fig. 3e), three C 1s components together with an additional K 2p signal are observed. Compared with pristine Fe2O3, the K–Fe2O3 sample exhibits new carbide-related C 1s features, aligning with the leftward shift of the Fe 2p3/2 peak.33 XRD patterns of Fe2O3 and K–Fe2O3 (Fig. 3f) further reveal that additional reflections in K–Fe2O3 can be indexed to the main diffraction peaks of Fe5C2, with no Fe3C phase detected (Fig. S22).34,35 K doping plays multiple roles in the Fe-based catalyst by enhancing CO adsorption and activation, increasing chain-growth probability, and suppressing the secondary hydrogenation of olefins, thereby improving light-olefin selectivity. In addition, K facilitates the formation and stabilization of the Fe5C2 active phase, which contributes to the enhanced FTS performance.36
Although In2O3 is widely recognized as an effective catalyst for CO2 hydrogenation to methanol under relatively high pressures, the reaction in this work was performed at 0.5 atm, which is less favorable for methanol formation.37 To elucidate the CO2 activation pathway, in situ FTIR spectroscopy was conducted (Fig. 3g). The spectra show a band at 1398 cm−1 assigned to carbonate species and a band at 1560 cm−1 attributed to COOH* intermediates. Since COOH* species are generally associated with the RWGS pathway to CO rather than the methanol pathway via HCOO*, these results indicate that CO is the predominant product in the present system. The feature at 1634 cm−1 originates from adsorbed water, and the bands at 2114 and 2171 cm−1 are ascribed to linearly and bridge-bonded adsorbed CO*, respectively. The band at 2917 cm−1 is characteristic of CH3* species, and the broad absorptions at 3417 and 3724 cm−1 are associated with surface hydroxyl groups and adsorbed water molecules.38 Accordingly, these surface intermediates provide direct spectroscopic evidence for a tandem RWGS–FTS mechanism, wherein CO2 is first reduced to CO via RWGS, and then the resulting CO is subsequently hydrogenated through CHx* intermediates to form light hydrocarbons.39–41
In summary, we have developed a tandem dual-temperature catalytic configuration that enables controlled multi-stage temperature distribution and efficient energy utilisation for the photothermal hydrogenation of CO2 into high-value hydrocarbons. In the upper bed, G–In2O3 drives the RWGS reaction. The absorbed light energy is converted into heat and transmitted to the underlying K–Fe2O3 bed via both radiative and convective heat transfer. In the lower bed, K–Fe2O3 drives the FTS process (Fig. 3h). By synergistically engineering the dual-bed catalysts with optimal operational temperatures in a single photothermal reactor, we achieve direct CO2-to-light-olefin conversion with a production rate of 52.1 µmol g−1 h−1 and a selectivity of 69%, demonstrating a promising strategy for solar-driven CO2 valorisation.
Z. L.: Investigation, Methodology, Writing – original draft. L. H. and C. Z.: Formal analysis, Data curation. Z. Y. and Z. W.: Investigation, Validation, Visualization. Y. Q.: Conceptualization, Supervision, Project administration. S. X.: Conceptualization, Writing – review & editing.
Conflicts of interest
There are no conflicts to declare.Data availability
Data will be made available on request. Supplementary information is available. See DOI: https://doi.org/10.1039/d5cc07291c.Acknowledgements
The authors appreciate the support from the National Natural Science Foundation of China (52402246) and the Natural Science Foundation of Hebei (E2023202267). S. X. acknowledges the start-up funding from the University of Manchester and the University of Manchester Harwell Campus.References
- F. Xu, F. Zhao, X. Deng, J. Zhang, J. Zhang, C. Ai, J. Yu and H. García, Nat. Commun., 2025, 16, 6882 CrossRef CAS PubMed.
- X. Wan, Y. Li, Y. Chen, J. Ma, Y.-A. Liu, E.-D. Zhao, Y. Gu, Y. Zhao, Y. Cui, R. Li, D. Liu, R. Long, K. M. Liew and Y. Xiong, Nat. Commun., 2024, 15, 1273 CrossRef CAS PubMed.
- Z. Xiao, L. Zhang, X. Tan, K. Sun, J. Li, L. Pan, J. J. Zou, G. Li and D. Wang, Adv. Funct. Mater., 2025, 35, 2500339 CrossRef CAS.
- X. Ding, W. Liu, J. Zhao, L. Wang and Z. Zou, Adv. Mater., 2025, 37, 2312093 CrossRef CAS PubMed.
- W. Gao, Y. Li, D. Xiao and D. Ma, J. Energy Chem., 2023, 83, 62–78 CrossRef CAS.
- S. Khan, X. Dai, T. Ali, S. Mahmood, M. U. Haq, M. S. Riaz and Y. Hu, Int. J. Hydrogen Energy, 2023, 48, 24756–24787 CrossRef CAS.
- E. Ø. Pedersen, I.-H. Svenum and E. A. Blekkan, J. Catal., 2018, 361, 23–32 CrossRef CAS.
- Z. Li, L. Zhong, F. Yu, Y. An, Y. Dai, Y. Yang, T. Lin, S. Li, H. Wang, P. Gao, Y. Sun and M. He, ACS Catal., 2017, 7, 3622–3631 CrossRef CAS.
- S. Garg, Z. Xie and J. G. Chen, Nat. Chem. Eng., 2024, 1, 139–148 CrossRef CAS.
- L. Wang, Y. Sun, F. Zhang, J. Hu, W. Hu, S. Xie, Y. Wang, J. Feng, Y. Li, G. Wang, B. Zhang, H. Wang, Q. Zhang and Y. Wang, Adv. Mater., 2023, 35, 2205782 CrossRef CAS PubMed.
- S. Ning, H. Ou, Y. Li, C. Lv, S. Wang, D. Wang and J. Ye, Angew. Chem., Int. Ed., 2023, 62, e202302253 CrossRef CAS PubMed.
- H. Song, K. Sun, H. Huang, S. Ning, S. Wang, Z. Wang, Y. Weng, Y. Cui, Y. Li, X.-S. Wang, D. Wang, L. Liu, Z.-J. Wang and J. Ye, Nat. Commun., 2025, 16, 2831 CrossRef CAS PubMed.
- R. Li, Y. Li, Z. Li, S. Ouyang, H. Yuan and T. Zhang, Adv. Mater., 2023, 35, e2307217 CrossRef PubMed.
- S. Mo, S. Li, J. Zhou, X. Zhao, H. Zhao, X. Zhou, Y. Fan, Z. Zhu, B. Li, Q. Xie, W. Si, Y. Chen, D. Ye and J. Li, ACS Catal., 2025, 15, 2796–2808 CrossRef CAS.
- L. Wang, Y. Dong, T. Yan, Z. Hu, F. M. Ali, D. M. Meira, P. N. Duchesne, J. Y. Y. Loh, C. Qiu, E. E. Storey, Y. Xu, W. Sun, M. Ghoussoub, N. P. Kherani, A. S. Helmy and G. A. Ozin, Nat. Commun., 2020, 11, 2432 CrossRef CAS PubMed.
- Z. Xie, X.-Y. Yu, Z. Zhang, X. Wang and T. Xie, ACS Appl. Mater. Interfaces, 2025, 17, 19617–19628 CrossRef CAS PubMed.
- Q. Hao, Z. Li, Y. Shi, R. Li, Y. Li, S. Ouyang, H. Yuan and T. Zhang, Nano Energy, 2022, 102, 107723 CrossRef CAS.
- S. Ning, X. Wu, H. Song, X. Ma, S. Yue, S. Zhang, L. Tang, R. Liu, X. Yin, S. Ouyang and J. Ye, J. Am. Chem. Soc., 2025, 148, 1728–1740 CrossRef PubMed.
- P. Cao, X. Xu, F. Jia, Y. Zeng, W. Liu, C. Wang, S. Han, M. Fang, X. Liu, D. Zhu and S. T. Navale, Appl. Surf. Sci., 2025, 688, 162424 CrossRef CAS.
- C.-Q. Guo, H.-S. Zheng, J. Yang, X. Xiang and Z.-Y. Zhao, Appl. Surf. Sci., 2025, 685, 162063 CrossRef CAS.
- F. Hu, X. Chen, Z. Tu, Z.-H. Lu, G. Feng and R. Zhang, Ind. Eng. Chem. Res., 2021, 60, 12235–12243 CrossRef CAS.
- Z. Zhang and Z. Zhu, Appl. Surf. Sci., 2022, 601, 154243 CrossRef CAS.
- X. Jin, Q. Guan, T. Tian, H. Li, Y. Han, F. Hao, Y. Cui, W. Li, Y. Zhu and Y. Zhang, Appl. Surf. Sci., 2020, 504, 144241 CrossRef CAS.
- M. A. M. Khan, R. Siwach, S. Kumar, J. Ahmed and M. Ahamed, J. Alloys Compd., 2020, 846, 156479 CrossRef CAS.
- J. Huang, G. Zhang, M. Wang, J. Zhu, F. Ding, C. Song and X. Guo, Front. Energy Res., 2023, 10, 995800 CrossRef.
- H. Zhang, H. Ma, H. Zhang, W. Ying and D. Fang, Catal. Lett., 2011, 142, 131–137 CrossRef.
- P. Bredy, D. Farrusseng, Y. Schuurman and F. C. Meunier, J. Catal., 2022, 411, 93–96 CrossRef CAS.
- C. Shen, K. Sun, R. Zou, Q. Wu, D. Mei and C.-J. Liu, Appl. Catal., B, 2025, 361, 124683 CrossRef CAS.
- X. Yu, Y. Chen, Q. Zhang, Y. Yin, D. Sun, Y. Ru and G. Tian, Surf. Interfaces, 2023, 38, 102789 CrossRef CAS.
- L. Song, S. Ouyang, P. Li and J. Ye, J. Mater. Chem. A, 2022, 10, 16243–16248 RSC.
- Z. Zhang, B. Chen, L. Jia, W. Liu, X. Gao, J. Gao, B. Meng, Y. Tan, Y. He, W. Tu and Y.-F. Han, Appl. Catal., B, 2023, 327, 122449 CrossRef CAS.
- H. Wang, X. Nie, Y. Liu, M. J. Janik, X. Han, Y. Deng, W. Hu, C. Song and X. Guo, ACS Appl. Mater. Interfaces, 2022, 14, 37637–37651 CrossRef CAS PubMed.
- M. A. Hoque, M. I. Guzman, J. P. Selegue and M. K. Gnanamani, Materials, 2022, 15, 7378 CrossRef CAS PubMed.
- S. Najari, S. Saeidi, A. Sápi, Á. Szamosvölgyi, Á. Papp, A. Efremova, H. Bali and Z. Kónya, Chem. Eng. J., 2024, 485, 149787 CrossRef CAS.
- Y. Liu, Y. Ye, Y. Li, H. Du, Z. Yin, M. Li, Z. Huang, X. Zheng, H. Wang, Y. Wang and Y. Deng, Mol. Catal., 2024, 556, 113947 CAS.
- L. Niu, X. Liu, X. Wen, Y. Yang, J. Xu and Y. Li, Catal. Today, 2020, 343, 101–111 CrossRef CAS.
- J. Ye, C. Liu, D. Mei and Q. Ge, ACS Catal., 2013, 3, 1296–1306 CrossRef CAS.
- T. Yan, L. Wang, Y. Liang, M. Makaremi, T. E. Wood, Y. Dai, B. Huang, F. M. Ali, Y. Dong and G. A. Ozin, Nat. Commun., 2019, 10, 2521 CrossRef PubMed.
- M. Zhang, J. Ren and Y. Yu, ACS Catal., 2019, 10, 689–701 CrossRef.
- R. Li, Y. Li, Z. Li, W. Wei, Q. Hao, Y. Shi, S. Ouyang, H. Yuan and T. Zhang, ACS Catal., 2022, 12, 5316–5326 CrossRef CAS.
- J. Han, W. Liu, L. Zhang, H. Ren, C. Wu, J. Zhang, C. Gong, G. Yang, H. Yang, S. Zhang, H. Wang, T. Ji, J. Li and P. Gao, ACS Catal., 2025, 15, 3940–3954 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: