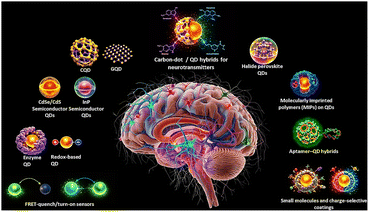
Quantum dot-enabled optical codes for neurotransmitter sensing at the bio-nano interface with translational relevance
Shrodha
Mondal
and
Prithidipa
Sahoo
 *
*
Department of Chemistry, Visva-Bharati University, Santiniketan-731235, India. E-mail: prithidipa.sahoo@visva-bharati.ac.in
First published on 11th February 2026
Abstract
Quantum dots (QDs), including semiconductor (Cd-based and III–V), carbon/graphene, and emerging halide perovskite QDs, offer size-tunable, bright, and photostable optical signals, making them uniquely suited as nanoreporters for neurotransmitter sensing. This review surveys recent advances in QD chemistry and surface engineering, recognition strategies (aptamers, molecularly imprinted polymers, enzymes, and small-molecule ligands), and signal transduction modalities (photoluminescence quenching/turn-on, FRET, electrochemiluminescence, and photoelectrochemical detection). We emphasise integration at the bio-nano interface for clinically relevant, minimally invasive platforms such as microfluidic sampling, wearable patches, and implantable probes, and analyse the principal barriers to translation (toxicity, stability in aqueous/biofluids, selectivity vs. interferents, and quantitation). Finally, we highlight promising directions: multiplexed spectral coding, ratiometric and lifetime-based readouts, renal-clearable/biodegradable QDs, and hybrid QD–polymer platforms for continuous monitoring of neurochemical signatures in diagnostic settings.
1. Introduction
Neurotransmitter dynamics – the rapid, local changes in the concentration of small signalling molecules such as dopamine, serotonin, acetylcholine, and γ-aminobutyric acid – underpin brain function and are central biomarkers for neurodegenerative diseases, psychiatric disorders, and acute neurological injury (Fig. 1 and Table 1).1 Recent studies emphasize that neurotransmitter signalling is not only central to classical functions such as mood, motor control, cognition, and reward but also in dynamically regulating complex behaviors and integrating neural networks at precise temporal and spatial scales. For instance, advanced human research has shown that dopamine and serotonin exhibit distinct yet overlapping influences on social decision-making and value-driven behaviors, with rapid in vivo fluctuations observed during interactive tasks. This indicates that neurotransmitter activity extends beyond steady baseline levels to encode real-time behaviors.2,3 Additionally, serotonin synthesis dynamics are associated with reward anticipation and feedback processing, implying that small variations in its availability can affect cognitive valuation and emotional responses. Monoaminergic systems also crucially affect motor pathway excitability, as serotonergic and noradrenergic signals both enhance corticospinal and motor neuron outputs, suggesting they work together across functional networks.4,5 Furthermore, alterations in inhibitory and excitatory neurotransmitters, such as GABA and glutamate, are increasingly linked to neurodegenerative and neuropsychiatric disorders, and GABA metabolism is now accessible through non-invasive methods for assessing region-specific inhibitory activity in the human brain.6,7 Conventional laboratory assays (HPLC, mass spectrometry, enzyme-linked methods) offer high chemical specificity but are generally limited by discrete sampling, lengthy preparation, and centralised infrastructure, thereby constraining their utility for time-resolved, bedside, or ambulatory phenotyping.| Neurotransmitter class | Neurotransmitter | Primary physiological roles | Representative clinical relevance |
|---|---|---|---|
| Monoamines | Dopamine | Reward and motivation, motor control, and endocrine modulation | Parkinson's disease, schizophrenia, and addiction |
| Serotonin (5-HT) | Mood regulation, sleep–wake cycle, appetite, and digestion | Depression, anxiety disorders, migraine | |
| Norepinephrine (noradrenaline) | Attention and alertness, stress response, and autonomic regulation | ADHD, PTSD, major depressive disorder | |
| Epinephrine (adrenaline) | Acute stress, cardiovascular, and metabolic control | Acute neurological injury, stress-related disorders | |
| Histamine | Wakefulness and arousal, appetite control, cognitive modulation | Sleep disorders, neuroinflammation | |
| Amino acids | Glutamate | Primary excitatory neurotransmission, learning and memory, synaptic plasticity | Alzheimer's disease, stroke, and epilepsy |
| GABA (γ-aminobutyric acid) | Primary inhibitory neurotransmission: anxiety and sleep regulation | Anxiety disorders, epilepsy, and insomnia | |
| Glycine | Inhibitory signalling in spinal cord and brainstem, NMDA receptor co-agonist | Motor disorders, schizophrenia | |
| Cholinergic | Acetylcholine | Memory and learning, attention, muscle contraction, and autonomic function | Alzheimer's disease, myasthenia gravis |
The growing clinical and research demand for minimally invasive, continuous neurochemical monitoring has therefore stimulated interest in nanomaterial transducers that combine high sensitivity, fast response, and compatibility with compact optical/electronic readouts.8–20
Quantum dots (QDs) are an up-and-coming class of transducers for this challenge because they offer exceptionally bright, size-tunable, and narrowband photoluminescence, high photostability, and a surface chemistry that can be engineered to couple molecular recognition with robust optical signals. These optical attributes make QDs ideal for multiplexed and ratiometric schemes, where distinct spectral “codes” report multiple analytes simultaneously, or for internal reference channels that improve quantitation in complex biofluids. Recent survey articles and focused reviews highlight how advances in QD synthesis and surface passivation have translated these photophysical advantages into practical sensing platforms.
Within the broader QD family, carbon and graphene quantum dots (CQDs/GQDs) have attracted disproportionate attention for biomedical sensing due to their facile, low-cost synthesis routes, versatile surface functionalization, and comparatively benign toxicity profiles. Carbon-based QDs have been integrated into fluorescence, electrochemical and electrochemiluminescence (ECL) sensors for monoamine neurotransmitters, delivering low-nanomolar to picomolar limits of detection in controlled media and enabling routes toward wearable optical or electrochemical patches. Their tunable surface states also enable ratiometric responses that mitigate matrix effects encountered in real samples.21–38
At the same time, emerging halide perovskite quantum dots (PQDs) exhibit ultrahigh brightness and narrow emission profiles, making them exceptionally attractive for low-light, multiplexed readouts and enabling sensitive optical and photoelectrochemical biosensing platforms.39,40 However, intrinsic water/air instability and concerns about lead content pose significant translational barriers to biological applications.39 Recent hybrid strategies, including embedding PQDs within protective matrices or constructing heterostructures with conventional semiconductor quantum dots, have demonstrated significant improvements in aqueous stability and resistance to signal interference, thereby enabling reliable neurotransmitter detection under physiologically relevant conditions.41–43 These hybrid approaches offer a pragmatic pathway to harness PQD's superior photophysical properties while mitigating limitations in chemical robustness and biosafety.39–41
Equally important to the optical reporter are the molecular recognition motifs that confer selectivity in complex biofluids. Aptamer–QD constructs, molecularly imprinted polymer shells, enzyme-linked transduction and tailored ligand coatings each offer complementary advantages: aptamers enable conformationally driven, highly specific binding that can be transduced optically at very low concentrations; MIPs provide rugged, low-cost selectivity; and enzyme or redox-mediated schemes are well suited to electrochemical or photoelectrochemical coupling.44–46 Notably, aptamer-based nanosensors have recently achieved femtomolar sensitivity in specialized nanopores and optical geometries, illustrating the power of coupling high-affinity recognition with nanoscale transducers.44–49
Finally, the push toward dynamic and time-resolved human diagnostics requires device integration, e.g., microfluidics for controlled sampling, soft polymer and textile matrices for epidermal/wearable monitoring, and compact photonic/electronic interrogators for ambulatory readout. Advances in wearable sweat sensors and flexible electrochemical platforms point to realistic pathways for embedding QD-based neurochemical reporters into noninvasive diagnostic formats. Taken together, recent material innovation, surface-chemistry strategies and device engineering converge to make QD-enabled neurotransmitter sensing an exciting frontier for advanced human diagnostics; this review surveys those developments and identifies the key opportunities and obstacles on the route to clinical translation.
2. Quantum dot types and optical properties relevant to sensing
2.1. Semiconductor QDs (CdSe/CdS, InP)
Traditional II–VI (e.g., CdSe, CdTe) and III–V (e.g., InP) colloidal semiconductor QDs remain the benchmark for optical sensing because they combine very high photoluminescence quantum yields, narrow, size-tunable emission bands and high photostability properties that make them exceptional FRET donors/acceptors and ideal for multiplexed optical assays. Core/shell engineering (CdSe/CdS, CdSe/ZnS) and graded-alloy shells reduce surface trap states, increase quantum yield and broaden the usable excitation/emission windows for ratiometric or lifetime-based sensing. Ligand-exchange and surface functionalization strategies further control aqueous solubility and bioconjugation while balancing trap-state passivation against charge-transfer sensitivity required for photoelectrochemical sensors. For applications needing lower toxicity, InP-based QDs have become viable Cd-free alternatives with improved brightness owing to advanced shelling and surface chemistry, although their synthetic control and long-term stability still lag behind the best Cd-based systems.Narrow linewidths (tens of nm or less) allow spectral multiplexing and precise FRET pairs; high QY boosts signal-to-noise in dilute biological fluids.
Shelling and ligand design are often the key trade-offs: thick shells/passivating ligands improve brightness/stability but can reduce electron/hole tunnelling needed for some electrochemical or PEC transduction modes.25,50–55
2.2. Carbon and graphene quantum dots (CQDs/GQDs)
Carbon quantum dots (CQDs) and graphene quantum dots (GQDs) are attractive sensor building blocks for biological and field-deployable assays because they are inexpensive to synthesize, readily doped with heteroatoms (N, S, P) and typically show lower cytotoxicity than heavy-metal QDs.20,56 Their emissive behavior is often dominated by surface states, molecular fluorophores formed during synthesis, and dopant-related traps, which give rise to excitation-dependent emission and tunable ratiometric responses that can be exploited to suppress matrix interference in complex biofluids.20,56 CQDs and GQDs are widely used in electrochemical, electrochemiluminescence (ECL) and dual optical-electrochemical sensors for monoamine neurotransmitters (dopamine, serotonin);57 in many reported devices, the carbon dots act both as conductive surface modifiers (improving electron transfer) and as optical reporters for fluorescence/ECL readout.20,56,58,59 Recent reviews and method papers summarize design rules (doping, passivation, composite formation) and present numerous dopamine-sensing platforms with detection limits in the nM–µM range.20,56,60Excitation-dependent emission can be exploited for ratiometric sensing (internal referencing) but requires careful calibration across excitation wavelengths and sample matrices.20
GQDs combine π–π interactions and high surface area for analyte adsorption and are useful for monoamine capture and electrochemical signal amplification when integrated with metal nanoclusters or redox polymers.58–60
2.3. Halide perovskite QDs
Lead halide perovskite quantum dots (PQDs; e.g., CsPbX3 and related phases) are frequently highlighted for sensing applications due to their ultrahigh brightness, near-unity photoluminescence quantum yields, extremely narrow emission linewidths (few nm FWHM), and facile bandgap tunability across the visible spectrum via halide composition.40,61 These optical attributes translate into exceptional signal-to-noise ratios in fluorescence and photoelectrochemical-based sensors, enabling ultralow detection limits for neurotransmitters and other bioanalytes.40,62Despite these analytical advantages, lead toxicity represents a fundamental and largely non-negotiable barrier for clinical and in vivo sensing. The presence of labile Pb2+ ions, combined with the known moisture and oxygen instability of lead halide perovskites, severely limits their suitability for biomedical deployment.40 Even trace lead leaching is unacceptable under current regulatory frameworks, and the long-term integrity of encapsulation cannot be guaranteed under physiological conditions that involve mechanical stress, ionic strength fluctuations, and enzymatic activity.
Recent studies have therefore shifted toward hybrid or heterojunction designs as pragmatic, short-term engineering solutions rather than true biocompatible materials. Examples include CsPb2Br5/CdSe heterojunctions, perovskite-semiconductor composites, or embedding PQDs within polymers, covalent organic frameworks (COFs), or inorganic matrices.40,62 Such architectures can substantially improve water stability and operational reproducibility, and CsPb2Br5/CdSe systems have demonstrated water-stable photoelectrochemical dopamine sensing with low-nanomolar detection limits and good anti-interference performance.41 These results demonstrate that perovskite-derived brightness can be indirectly harnessed for in vitro or ex vivo analytical sensing when Pb exposure is physically isolated from the sample environment.
However, encapsulation strategies should be viewed as risk-mitigation approaches rather than permanent solutions, as most coatings are susceptible to degradation, cracking, or ion diffusion over extended use. Consequently, regulatory acceptance of lead-based perovskite sensors for implantable, wearable, or clinical diagnostic applications is doubtful in the near term.40
For aqueous neurotransmitter sensing, hybridisation remains the most effective route to exploit perovskite optical performance while partially addressing stability concerns; however, such systems are best suited for laboratory-based or disposable analytical devices. From a translational perspective, future research should prioritize lead-free perovskite analogues (e.g., tin, bismuth, or double perovskite systems) or alternative QD platforms, such as carbon dots or silicon QDs, that inherently align better with biocompatibility and regulatory requirements.40
3. Molecular recognition strategies at the QD surface
To enable direct comparison across sensing platforms, the key molecular recognition strategies employed at quantum dot surfaces are summarized in Table 2. The table summarizes the recognition strategy, targeted neurotransmitters, surface chemistry, representative performance, selectivity and stability limitations, real-sample relevance, and matrix tests, enabling systematic comparison across different approaches.| Recognition strategy at QD/related surface | Target neurotransmitters | Mechanism/surface chemistry | Representative performance/notes | Selectivity and stability limitations | Real-sample relevance | Matrix tested (buffer/serum/CSF/in vivo) | Ref. |
|---|---|---|---|---|---|---|---|
| MIP-coated QDs (fluorescence sensors) | Dopamine | Molecular imprinting creates analyte-specific cavities on a polymer shell coated on ZnS/CQDs or graphene QDs | MIP@ZnS QDs for dopamine: linear range ∼0.01–1 µM, LOD ∼3.6 nM; high imprinting factor vs. non-imprinted polymer, tested in urine samples | Fouling/oxidative passivation of imprinted sites can reduce long-term stability in complex bio samples and affect the baseline | Limited, often restricted to spike-recovery tests with brief discussion | Mostly buffer, some diluted serum/urine, rarely CSF, no true in vivo | 63 |
| Carbon QDs/graphene QDs (label-free photoluminescence) | Dopamine | Non-specific surface interactions; fluorescence quenching via photoinduced electron transfer upon analyte binding | Boron/sulfur-doped GQDs show sensitive fluorescence quenching with dopamine over a wide range | Lacks tuning for receptor-like selectivity; sensitive to interferents (e.g., ascorbic acid) unless surface passivation added | Weak, real-sample effects are often acknowledged but not deeply analyzed | Primarily buffer, occasional serum dilution | 64 |
| MIP on carbon QDs in optical fiber sensors | Dopamine | Silica-based MIP on CQDs with optical loss mode resonance transduction | Dopamine detection over ∼0.3–100 µM with selectivity vs. typical interferents | Optical surfaces are still susceptible to fouling and baseline shifts without robust passivation | Limited, real-sample stability is rarely explored | Mostly buffer, a few serum examples | 65 |
| Aptamer-functionalized surfaces (FET/optical) | Dopamine, serotonin | DNA/RNA aptamer binding via conformational change or charge modulation | Aptamer sensors on graphene FETs can achieve fM–aM LODs and wide dynamic ranges (e.g., 1 aM–100 µM for dopamine) | Aptamer affinity can vary with the ionic environment; fouling and non-specific binding can still distort the baseline in complex matrices | Moderate, some studies discuss biological relevance and interference | Buffer, serum, occasionally CSF (ex vivo), rare in vivo | 44 |
| Electrical aptamer biosensors | Serotonin, dopamine | Aptamer conformational gating in nanopores or FET architectures | Aptamer nanopore sensors exhibit distinct electrical responses to dopamine vs. serotonin | Baseline drift with repeated use, ionic strength, and fouling impact signal regulation | Strong when imaging is demonstrated | In vivo (zebrafish/cells), ex vivo tissue | 45 |
| Nanoplasmonic aptasensors (non-QDs) | Dopamine | Surface plasmon resonance + aptamer monolayer for label-free detection | High specificity aptamer-functionalized plasmonic sensor tested in blood and CSF | Surface fouling and non-specific adsorption remain challenges in whole biofluids | Good conceptual relevance, but stability issues noted | Buffer, serum, rare in vivo | 66 |
| MIP + aptamer hybrid on electrodes | Dopamine | Dual recognition combining MIP cavities and aptamer sequences | MIP-aptamer on AuNPs/rGO electrodes: LOD ∼47 nmol/L with improved selectivity vs. individual MIP or aptamer systems | Complex synthesis, stability of the aptamer layer vs. fouling requires optimization | Moderate, but often proof-of-concept | Buffer and spiked serum | 44 |
3.1. Aptamer–QD hybrids
Aptamer–QD hybrids combine the molecular recognition and conformational dynamics of nucleic-acid aptamers with the bright, photostable optical signals of quantum dots (QDs).47,67–72 In the canonical designs, an aptamer that binds a small neurotransmitter (e.g., dopamine, serotonin) is chemically attached to a QD (or to a QD-nanostructure assembly) such that target binding triggers a distance/orientation change between the QD and a quencher/acceptor (organic dye, graphene oxide, Au nanoparticles) or between two QDs.47,69,71 This conformational change translates into an optical readout via one or more mechanisms: distance-dependent changes in FRET efficiency, photoinduced electron/energy transfer (quenching or turn-on), ratiometric emission shifts (two-colour QD constructs), or lifetime changes.67–70 The high QY and narrow emission of QDs increase signal-to-noise, extend the dynamic range in dilute samples, and improve multiplexing via colour encoding.68,70Optical aptamer–QD assays have routinely reported sub-nanomolar limits47,72 with careful signal amplification (e.g., enzymatic or nanoparticle amplification) and ultra-low background; some hybrid platforms (including nanopore–aptamer systems described below) achieve femtomolar detection for dopamine under buffered conditions.72,73 Always check matrix effects: limits measured in buffers rarely translate intact to serum/CSF without calibration and anti-fouling measures.73
Ratiometric and multi-emitter schemes: two QDs with distinct emission wavelengths can be attached to different aptamer sites or to a reporting strand: analyte-induced structural shifts change relative intensities, giving internal referencing that cancels many matrix effects – useful for complex biofluids. Ratiometric aptamer–QD sensors have been used to improve robustness in serum/saliva measurements.47,73–76
Carbon-dot/QD hybrids for neurotransmitters: carbon dot constructs functionalized with dopamine aptamers have produced highly selective fluorescence responses and have been incorporated into simple turn-on assays. These systems highlight how non-toxic carbon QDs can be combined with aptamers for sensitive neurotransmitter readout.46
Small-molecule conformational signalling: many small-molecule aptamers undergo pronounced conformational rearrangements upon binding, producing large changes in distance or energy-transfer that map well to FRET or quenching transduction.45,46
Optical brightness and multiplexing: QDs’ high brightness and narrow emission spectra enable simultaneous multiplexed detection of multiple neurotransmitters using different aptamers/QD colours.47,77
3.2. Molecularly imprinted polymers (MIPs) on QDs
MIP-coated quantum dots (QD@MIP) are hybrid nanomaterials in which a luminescent QD core (Cd-based, ZnS, carbon dots, etc.) is cloaked with a thin molecularly imprinted polymer layer that contains template-shaped cavities complementary in size, shape and functional groups to the target analyte. The polymer shell supplies synthetic, robust molecular recognition (mimicking antibodies/enzymes) while the QD provides an optical reporter (fluorescence intensity, lifetime, or ratiometric signal). This combination is attractive where biomolecular receptors (antibodies, enzymes, aptamers) are unstable, expensive, or unavailable, because MIPs are inexpensive, thermally and chemically robust and can be tuned for small molecules such as monoamine neurotransmitters.Core–shell silica templating. QDs are first embedded in a silica shell, and silica is subsequently modified and imprinted with organic monomers to yield a stable, silica-supported MIP. This approach improves colloidal stability and reduces QD aggregation.
Grafting/“grafting-from” polymerization and electropolymerization. Surface-initiated polymerization or electropolymerization of monomers (e.g., polypyrrole, polyaniline derivatives) creates thin MIP films around QDs or on QD-modified electrodes, enabling integration of optical QDs with electrochemical readout.
Robustness and shelf stability: MIPs tolerate harsh sample preps, temperature swings and long storage – important for field or point-of-care neurotransmitter assays where enzyme or antibody decay would cripple performance.
Low cost/scalable synthesis: monomers and polymerization methods are inexpensive and compatible with large-scale sensor fabrication (paper strips, arrays).
Sensitivity: many QD@MIP fluorescence sensors report detection limits in the low nM to sub-nM range for small organic analytes. With ratiometric designs or signal amplification, some systems achieve even lower LODs in controlled buffers.
Selectivity: properly designed MIPs discriminate between close structural analogues (e.g., dopamine vs. tyramine) based on shape and functional group complementarity; however, selectivity is rarely as absolute as that of a high-affinity aptamer or antibody, and should always be evaluated against likely interferents (ascorbate, uric acid, and metabolites).
Real sample testing: several studies have validated QD@MIP sensors in serum, urine, and food matrices, as well as on paper-based platforms, demonstrating their practicality for field or biomedical samples when combined with simple sample prep or dilution.
Non-specific adsorption/fouling. Biological fluids foul polymer surfaces; strategies include PEGylated outer layers, zwitterionic co-monomers, or employing ratiometric readout to cancel background changes.78,80–83
QD photophysics and polymer thickness. Thick MIP shells increase the distance between the analyte and the QD and can blunt quenching-type mechanisms; thin, conformal shells (surface imprinting) are preferred to maintain strong optical coupling while still forming selective cavities.63,80,81
Heavy-metal QD safety and leaching. For biomedical applications, use ZnS, doped ZnS, carbon QDs, or robust encapsulation (silica or polymer barrier) to prevent toxic metal-ion leaching; always measure metal release (ICP-MS) if intended for biological/clinical use.78,82,84
3.3. Enzyme and redox-based recognition
Enzyme-based neurotransmitter sensors use a biocatalyst (most commonly an oxidase or peroxidase) to convert a non-electroactive or weakly fluorescent neurotransmitter into a redox-active product (e.g., dopamine → dopamine–quinone + H2O2). The generated product(s) – H2O2, quinones, semiquinones, or polymerized products (polydopamine) – interact with the QD core or QD surface chemistry and thereby modulate the optical/electrochemical signal. This modulation can occur via (a) photoinduced electron transfer (PET) or charge transfer between QDs and oxidized analytes/oxidation products (causing quenching or altered lifetimes), (b) FRET or inner-filter effects when enzymatically produced chromophores absorb QD emission, or (c) chemical modification/deposition (e.g., formation of insulating polymer films at the QD surface) that changes emission intensity or electron/charge transfer to electrodes. Enzymes, therefore, convert a molecular recognition event into a chemical species that interacts strongly (and often irreversibly) with the QD reporter.76PEC (photoelectrochemical) detection: QDs are excellent light-harvesting materials for PEC devices (they produce photocurrent under illumination). Redox reactions of neurotransmitters at the QD-modified electrode surface (or mediated by enzyme products) change charge-transfer kinetics and photocurrent amplitude. The recent hybrid perovskite/CdSe QD PEC dopamine sensor is an illustrative example in which a water-stable perovskite-QD heterojunction provides sensitive photoelectrochemical readout of dopamine via redox interactions with photoexcited carriers.41
Advantages. High sensitivity via enzymatic amplification: oxidases produce multiple redox equivalents per analyte molecule (e.g., H2O2), enabling signal amplification before QD transduction.41,76
Versatile readouts: enzymatic products can be detected optically (fluorescence quench/turn-on, chemiluminescence) or electrochemically (amperometry, ECL, PEC), enabling sensor formats spanning the lab to point-of-care.37,76
Tradeoffs/limitations. Enzyme fragility/operational stability: natural enzymes are temperature, pH and protease sensitive; immobilization strategies (entrapment, crosslinking, covalent attachment, protective matrices) or replacement with nanozymes are common remedies.76,85
Secondary chemistry and fouling: oxidation products (quinones, polydopamine) can irreversibly modify QD surfaces or electrode interfaces, causing signal drift or loss of reversibility – this is useful in single-use assays but problematic for continuous monitoring. Careful choice of QD passivation and anti-fouling coatings (PEG, zwitterions) helps.37,86
Specificity and cross-reactivity: oxidases may act on multiple substrates; pairing a selective oxidase (or combining enzymatic recognition with molecular imprinting/aptamer layers) improves selectivity.85–87
3.4. Small molecules and charge-selective coatings
Charge-selective coatings, tailored ligands and metal-coordination motifs on quantum dot (QD) surfaces are a pragmatic, widely used strategy to bias which small molecules adsorb, bind, or exchange electrons at the QD interface. For neurotransmitter sensing (most commonly dopamine, DA), these surface chemistries enable two complementary selectivity mechanisms: (A) thermodynamic/affinity selectivity – preferential adsorption or reversible complexation of the analyte versus interferents; and (B) kinetic/electrochemical selectivity – differences in electron-transfer rates or oxidation chemistry at the functionalized surface that change signal kinetics or amplitude.Electrostatic (polyelectrolyte) coatings: exclude or concentrate charged interferents. Polyelectrolytes and ion-exchange coatings (classical example: Nafion) create an electrostatic sieve at the sensor surface. At physiological pH, dopamine is largely cationic (protonated amine), while ascorbic acid (AA) is anionic, and uric acid (UA) is negatively charged or neutral depending on pH.88–90 A negatively charged coating (e.g., Nafion) repels anions (AA, UA) while allowing cationic DA to approach the QD/electrode, improving selectivity in electrochemical and QD-modified electrode sensors. Nafion and other sulfonated polymers remain widely used as simple, robust exclusion layers in electrochemical platforms and in hybrid QD composites.89,90 Reviews and experimental studies summarising Nafion/charged-polymer strategies and their use for DA vs. AA/UA discrimination are available.90
Electrostatic selectivity is pH- and ionic-strength-dependent; high salt screens charges and reduces exclusion effectiveness, so report conditions carefully.88
Boronic-acid motifs and diol recognition: covalent reversible capture of catechols. Boronic acids selectively and reversibly form boronate esters with cis-diol motifs such as the catechol group on dopamine. Functionalizing QDs (or their surface ligands) with phenylboronic acid derivatives provides a chemically selective binding pocket that discriminates catechols from non-diol interferents like AA/UA.90 This motif is especially powerful for optical sensors (turn-on/turn-off fluorescence) and has been demonstrated using boronic-acid-modified carbon dots, MoS2/QD hybrids and polymer coatings to achieve low-nM to µM dopamine sensing with strong selectivity. Representative implementations (including recent carbon-dot and GQD work) demonstrate that boronate chemistry is a practical route for catechol recognition.37 Boronate ester formation is pH-sensitive (better at alkaline pH, where boronate is activated), so designs for physiological samples often balance buffer conditions or use local microenvironments to favour binding.90
Inclusion complexes and host motifs (β-cyclodextrin, crown ethers). Supramolecular hosts such as β-cyclodextrin (β-CD) can be grafted to QDs or used as stabilizers to form inclusion complexes that preferentially bind small hydrophobic/aromatic molecules. β-CD functionalization on carbon dots or QD composites has been used to capture dopamine and suppress interference from hydrophilic anions. Several recent works demonstrate β-CD-stabilized CDs or Se-QDs embedded in composite electrodes producing selective DA readout. Host motifs are modular and easy to combine with other surface chemistries.91,92
Metal-coordination/metal-doped motifs: catechol–metal complexation and catalytic redox tuning. Catechols coordinate strongly to transition metal centres (Fe3+, Cu2+, etc.), forming stable catechol–metal complexes. QD surfaces that present metal-binding sites (via surface-bound metal ions, metal-doping, or coordination ligands) can therefore selectively capture or catalyse oxidation of dopamine relative to AA/UA. Metal centers can also modulate electron-transfer rates: e.g., Cu- or Fe-doped CQDs or metal-oxide-QD hybrids can accelerate oxidation of catechols, change quenching behaviour, or produce catalytic (nanozyme) activity that yields a distinctive optical/electrochemical signature for DA. Reviews of metal–catecholate complexes and recent papers on metal-doped QD/nanocomposites illustrate how metal coordination can be used to tune selectivity and sensitivity.93
Metal centres may catalyse dopamine autoxidation (producing quinones/polymers), which can give high sensitivity but also lead to surface fouling; control experiments and surface passivation are essential.94
π–π/hydrophobic interactions and aromatic ligands. Graphitic/π-rich QDs (graphene QDs, carbon dots with aromatic surface groups) and aromatic ligands promote π–π stacking with aromatic neurotransmitters (dopamine, serotonin), increasing residence time and enhancing quenching or electron transfer relative to non-aromatic interferents.35,92 Several recent carbon–QD composites illustrate improved DA sensing via π–π and hydrophobic adsorption when combined with auxiliary selective motifs.35
Kinetic (rate)-based discrimination. Even when multiple species bind, differences in adsorption/desorption kinetics and electron-transfer rates can be exploited: coatings that slow access (thicker polymers, steric barriers) will penalize fast-diffusing anions differently than small cations, and catalytic metal sites often oxidize DA faster than AA – so time-resolved readout (chronoamperometry, transient fluorescence) can separate analytes by their kinetic fingerprint. Recent sensor papers emphasize combining steady-state and time-resolved metrics to improve selectivity.88,94
Above all, most QD-based platforms discussed in this review (Fig. 2) should be viewed as preclinical or translational research tools rather than established diagnostic devices. Their primary contribution lies in defining sensing mechanisms, material design rules, and integration strategies that could inform future clinically compliant systems. Explicitly recognizing this distinction is essential to avoid overstating readiness while still acknowledging the substantial progress made toward dynamic in situ neurochemical monitoring.
4. Signal transduction modalities
Instead of reiterating well-established photophysical theory, this section explains how different signal-transduction mechanisms influence sensitivity, selectivity, robustness, and multiplexing capacity in quantum-dot-based neurotransmitter sensors, and why certain mechanisms are preferred in specific biological contexts.4.1. Photoluminescence (PL) quenching and turn-on sensors
PL quenching and recovery remain the most widely used transduction modes in QD-based neurotransmitter sensing because they exploit direct coupling between analyte binding and exciton recombination pathways. In practice, the choice between quenching- and turn-on-type responses reflects trade-offs among background suppression, matrix tolerance, and reversibility.Turn-on formats are often implemented using displacement or competitive binding assays, which are generally preferred for dilute biofluids, where autofluorescence and scattering dominate background signals. In contrast, turn-off (quenching) sensors are simpler to construct but are more prone to false positives due to nonspecific quenchers present in serum, sweat, or tissue environments. Importantly, lifetime-based PL readouts decouple signal intensity from excitation fluctuations and optical path length, offering greater robustness in colored or turbid samples and enabling more reliable operation in complex biological matrices.95
Carbon-based QDs (CQDs, GQDs, polymer dots) are frequently employed in this regime due to their abundant surface states and accessible excited-state pathways. While this enhances sensitivity, it also increases susceptibility to nonspecific interactions, underscoring the need for careful surface passivation and lifetime-resolved validation.95,96
The principal trade-off is architectural complexity: precise donor–acceptor spacing (1–10 nm) must be engineered and maintained, which can be challenging under physiological conditions where conformational drift or nonspecific adsorption occurs. Consequently, FRET-based neurotransmitter sensors are most successful in aptamer-, DNA-, or scaffold-guided designs where geometry is tightly constrained.97,98
SET and Dexter transfer appear less frequently in neurotransmitter sensing but offer niche advantages. SET enables longer-range distance sensitivity near metal interfaces, useful in surface-immobilized architectures, while Dexter transfer requires direct orbital overlap and is therefore restricted to tightly packed or solid-state assemblies. In practice, their stringent geometric constraints limit widespread adoption, but they can provide high contrast in carefully engineered nanoscale systems.
4.2. Förster resonance energy transfer (FRET) and spectral coding
Narrow QD emission and tunable absorption spectra enable precise FRET pairs for distance-dependent, ratiometric sensing.103 Multiplexed monitoring of several neurotransmitters can be achieved by assigning unique QD spectral codes and orthogonal recognition elements. FRET-based QD platforms have demonstrated multianalyte discrimination in model fluids.104,105Beyond single-analyte detection, QDs uniquely enable spectral and temporal coding strategies that address one of the major bottlenecks in neurochemical sensing: simultaneous detection of multiple neurotransmitters with overlapping chemistry.105,106
The narrow emission linewidths and size-tunable emission of QDs allow multiple FRET pairs to coexist with minimal spectral cross-talk, supporting multiplexed ratiometric sensing.103,107 In practice, this capability is most impactful when combined with orthogonal recognition elements (such as aptamers, enzymes, or molecularly imprinted polymers), rather than relying solely on photophysics.76,104
Spectral coding expands multiplexing capacity but introduces trade-offs in device complexity, calibration burden, and data processing, particularly when moving from buffered solutions to biological samples.106,108–111 Time-domain coding, using fluorescence lifetime or time-gated detection, offers a complementary route by providing concentration-independent identifiers that are robust to photobleaching and excitation instability.104,109,112–115 However, lifetime-based systems require more sophisticated instrumentation, limiting near-term point-of-care translation.
Overall, FRET combined with spectral or temporal coding represents a powerful but design-intensive strategy, best suited for high-information-content assays rather than minimalistic diagnostic devices.
4.3. Electrochemiluminescence (ECL) and photoelectrochemical (PEC) transduction
ECL and PEC transduction represent two complementary signal-generation paradigms that convert molecular or biomolecular recognition events into quantifiable outputs via electrochemical or photophysical processes.116,117 In ECL, an applied potential at an electrode triggers redox reactions of a luminophore and coreactants (e.g., classic Ru2+/TPA systems or newer semiconductor nanocrystal emitters), leading to electronically excited states that emit light; this light output can be measured with high sensitivity, low background noise and a wide dynamic range, making ECL particularly suited for ultrasensitive detection of biomarkers, nucleic acids, proteins, and small molecules.118,119 In PEC transduction, on the other hand, a photoactive material (semiconductor, quantum dot or MOF-derived) absorbs light to generate electron–hole pairs; the resulting photocurrent or photovoltage is modulated by interactions between the recognition element and the analyte that serves as the detection signal.116,117,120 Recent research increasingly merges ECL and PEC modalities (e.g., via hybrid nanomaterials such as quantum dot/metal–organic-framework composites) to create dual-mode sensors that deliver both luminescence and photocurrent outputs, thereby improving robustness, reducing false signals, and enabling multiplexed detection.118,119ECL using QDs yields low-background signal generation ideal for low-abundance targets; carbon QDs and doped CQDs are frequently integrated into ECL sensors for monoamine detection. Photoelectrochemical sensors using perovskite/CdSe QD hybrids have shown sensitive dopamine detection by coupling light-driven current changes to analyte concentration.117–119
Hybrid designs such as QD/MOF or perovskite/QD composites enable dual-mode readout (optical + electrochemical), improving robustness against false positives. However, PEC systems introduce additional constraints related to electrode stability, light management, and (for perovskites) material toxicity, which currently limit biomedical translation.
5. Platform integration: from benchtop to wearable and implantable diagnostics
While QD-based neurochemical sensors demonstrate exceptional sensitivity and tunable photophysics under controlled benchtop conditions, their clinical relevance ultimately depends on how effectively these properties can be embedded into deployable sensing architectures. Platform integration, therefore, represents not a peripheral engineering step but a central translational strategy that determines whether QD sensors remain laboratory demonstrations or mature into real-world diagnostic tools. In this context, integration must simultaneously address sample handling, signal stability, user accessibility, and physiological compatibility. This section adopts a systems-level perspective, organizing recent advances along a clear translational continuum: microfluidic and lab-on-chip platforms that enable miniaturization and automation; wearable and epidermal devices that support continuous, noninvasive monitoring; and implantable or imaging-compatible probes that permit direct interrogation of neurochemical dynamics in living systems. Together, these integration levels illustrate how QD-based sensing evolves from static assays into clinically and biologically actionable technologies.5.1. Microfluidics and lab-on-chip
The first and most immediate step toward translation involves moving QD-based sensors off the benchtop and into microfluidic and lab-on-chip (LOC) architectures. At this level, the primary challenges are not biological compatibility, but rather fluid control, assay miniaturization, reaction kinetics, and reduction of sample and reagent volumes. Microfluidic integration provides a controlled environment in which the intrinsic optical sensitivity of QDs can be leveraged while overcoming the mass-transport and scalability limitations of bulk assays.Ankireddy et al. developed dopamine-functionalized InP/ZnS quantum dots (QDs-DA) as fluorescent probes for adenosine detection in a PDMS-based microfluidic chip. The photoluminescence (PL) of QDs-DA was quenched by Zn2+ due to strong coordination interactions, while the presence of adenosine preferentially sequestered Zn2+, resulting in PL recovery. Water-soluble InP/ZnS QDs were prepared using MPA and MUA ligands and immobilized on polystyrene microbeads via EDC/sulfo-NHS coupling, followed by DA conjugation. The QDs-DA-loaded microbeads were packed into microfluidic channels, enabling reversible Zn2+-mediated fluorescence quenching and adenosine-triggered recovery, with shorter linker lengths yielding faster response kinetics. This fluorescent bioprobe demonstrated sensitive adenosine detection in solution and serum, highlighting its potential for biological sensing applications.121
Dadkhah et al. developed a dual-mode fluorescence and colorimetric biosensor based on nitrogen–boron co-doped carbon quantum dots (N–B CQDs) for rapid and sensitive dopamine (DA) detection. Dopamine detection was achieved through a fluorescence “turn-off” mechanism, governed by the inner filter effect (IFE) and Förster resonance energy transfer (FRET), at basic pH, along with a smartphone-assisted colorimetric readout (Fig. 3). The smartphone-based method enabled reliable quantitative analysis of color changes beyond naked-eye observation. Linear detection ranges of 0.25–50 µM (fluorescence) and 5–500 µM (colorimetric) were achieved. The biosensor demonstrated good selectivity and sensitivity toward DA, even in the presence of interfering species, and was successfully applied to human urine and serum samples. Although not universally more sensitive than all existing sensors, the dual-readout strategy combines the high sensitivity of fluorescence with the portability and rapid on-site analysis of colorimetric detection, highlighting its promise for point-of-care diagnostics and future integration into µPADs.122
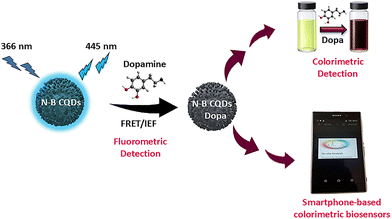 | ||
| Fig. 3 Dual-mode biosensor for dopamine detection.122 | ||
Chellasamy, with his lab members, reported that a simple and cost-effective dopamine (DA) sensing platform was developed using green-synthesised honey-derived carbon quantum dots (H-CQDs, 1–6 nm) (Fig. 4), which exhibit bluish-green fluorescence with an emission maximum at 445 nm and a quantum yield of ∼44%. The H-CQDs, rich in polar surface functional groups and highly water-soluble, enabled DA detection via electron-transfer-induced fluorescence and colour changes without the need for surface functionalization or additives. In fluorometric mode, the sensor showed high selectivity toward DA, with limits of detection of 6 nM (deionized water), 8.5 nM (male geriatric plasma), and 8 nM (female geriatric plasma) over a linear range of 100 nM–1000 µM. In colorimetric mode, a visible color change occurred within 5 min with an LOD of 163 µM. Integration with a smartphone-based colorimetric sensor array enabled precise DA quantification using a custom imaging and analysis application (Fig. 4). The H-CQDs were comprehensively characterized before and after DA detection, demonstrating the platform's robustness and applicability for biomedical sensing in real biological matrices.123
 | ||
| Fig. 4 Schematic overview of the fabrication process and sensing mechanism of a smartphone-integrated sensor array based on H-CQDs for dopamine detection. Adapted with permission from ref. 123. Copyright 2021, Elsevier. | ||
Across these studies, a recurring theme is the use of platform design, not solely nanomaterial chemistry, to enhance analytical performance. Whether through controlled residence times in microchannels, bead-based immobilization strategies, or smartphone-assisted optical readout, these works demonstrate that LOC integration can amplify the practical utility of QD sensors without fundamentally altering their photophysical mechanisms.
Jyoti Duhan and Sangeeta Obrai reported a simple bottom-up hydrothermal strategy for the synthesis of lanthanum- and nitrogen-codoped carbon quantum dots (La,N-CQDs). The La,N-CQDs functioned as fluorescent probes for highly sensitive and selective serotonin detection, achieving a low limit of detection of 7.4 nM. A portable smartphone-based sensing platform was developed to analyze fluorescence color changes, enabling reliable on-site and real-sample analysis. The probe exhibited excellent photostability, selectivity, sensitivity, and accuracy, with performance surpassing many reported methods. The approach is low-cost, fast, stable, and user-friendly, and was also demonstrated in colorimetric test papers and anti-counterfeiting ink applications. Real-sample analysis showed excellent accuracy, with serotonin recoveries ranging from 98% to 104% in spiked samples, confirming its practical applicability. Fig. 5 demonstrates the serotonin-induced fluorescence quenching of La,N-CQDs and its straightforward use in a paper-based analytical format.124
 | ||
| Fig. 5 Schematic illustration of the fluorescence-based serotonin sensing mechanism using La,N-CQDs and their paper-based implementation. (A) Photographic images of the handwritten pattern “NIT.J” on filter paper observed under (a) visible light, (b) under UV illumination, and (c) after spraying with water (left) and serotonin solution under UV light, showing fluorescence quenching. (B) Fluorescence response of the La,N-CQD-modified colorimetric paper under UV light following the sequential addition of 0, 10, 20, 30, 40, 50, 60, and 70 µL of serotonin solution. Reproduced and adapted with permission from ref. 124. Copyright 2023, Elsevier. | ||
Sarkar and co-workers developed bioreduced graphene quantum dots (rGQDs) using a Lawsonia inermis bioextract, yielding multifunctional surface groups for selective epinephrine sensing. The rGQDs enabled a multimodal sensing platform that combines ratiometric fluorescence (inner-filter effect) and enhanced electrical conductivity, implemented on a flexible, biopolymer-coated paper strip. A smartphone-assisted portable system allowed visual RGB-based quantification of epinephrine (Fig. 6), achieving low detection limits in serum, urine, and sweat. This eco-friendly, wearable-compatible platform highlights the potential of QD-based multimodal diagnostics for real-time healthcare monitoring.125
 | ||
| Fig. 6 Conceptual illustration of a disposable smart sensing platform based on rGQDs for multimodal epinephrine detection. Adapted with permission from ref. 125. Copyright 2026, American Chemical Society. | ||
A major limitation of benchtop QD-based neurotransmitter sensors is their dependence on bulky instrumentation, high sample volumes, and poor control over mass transport in complex biofluids. Microfluidic and lab-on-chip (LOC) platforms directly address these issues by enabling precise fluid handling, on-chip preconcentration, rapid reaction kinetics, and miniaturized optical readout, making them highly compatible with QD-based sensing architectures for neurochemical analysis. QD sensors integrated into microfluidic channels may enable controlled sampling, on-chip preconcentration and rapid optical information with low sample volumes, which is vital for tear/saliva diagnostics and for reducing matrix effects. Microfluidic and lab-on-chip (LOC) platforms integrate sample handling, reaction chambers, optical pathways, and detection electrodes onto miniaturized chips that require only microliters of biological fluid. When combined with QDs, these systems could enable rapid mixing, controlled reaction kinetics, laminar-flow-based separation, and multiplexed optical readout, all of which are valuable for detecting dopamine, serotonin, and related neurochemicals in real time. LOC devices also support point-of-care applications such as sweat, saliva, or microdialysate monitoring, and can integrate LEDs, photodiodes, or smartphone-based fluorescence imaging for portable diagnostics. Their small volumes enhance sensitivity, reduce reagent consumption, and minimize matrix effects, making them ideal hosts for QD-based aptamer, MIP, or enzymatic sensing layers.
Taken together, these examples demonstrate that microfluidic and LOC platforms systematically address several core limitations of benchtop QD-based neurotransmitter sensors, including uncontrolled sampling, high sample consumption, and limited portability. By enabling precise fluid handling, rapid reaction kinetics, and compact optical readout, LOC integration transforms QD probes from passive reporters into actively engineered diagnostic systems. Importantly, this level of integration establishes the technological foundation for point-of-care testing and continuous monitoring, setting the stage for more intimate contact with the human body in wearable formats.
5.2. Wearable patches and epidermal sensors
While microfluidic and LOC systems bring QD-based sensing closer to point-of-care deployment, they remain largely episodic and externally operated. Wearable and epidermal sensors represent a conceptual shift from discrete measurements toward continuous, user-integrated monitoring, where conformability, mechanical robustness, and long-term signal stability become as critical as analytical sensitivity.At this integration level, QDs are no longer evaluated solely as fluorescent probes, but as multifunctional signal enhancers embedded within flexible substrates, microneedles, or transistor architectures. The emphasis shifts toward multimodal transduction, resistance to biofouling, and compatibility with dynamic physiological environments such as sweat, interstitial fluid, or skin interfaces.
Głowacz reported a pattern-based neurotransmitter-sensing strategy using glutathione-capped CdSeS/ZnS quantum dots as non-specific nanoreceptors, combined with excitation–emission fluorescence spectroscopy (2D fluorescence). Multidimensional fluorescence response patterns arising from differentiated QD–analyte interactions enabled accurate identification of dopamine, norepinephrine, epinephrine, serotonin, GABA, and acetylcholine using unfolded partial least squares-discriminant analysis. Among zeroth-, first-, and second-order fluorescence data, excitation–emission matrices provided the best analytical performance, achieving an accuracy of 88.9–93.3%, a sensitivity of 0.89–0.93, and a specificity of 0.99–1.00. The approach further enabled quantitative determination of catecholamines in the micromolar range with R2 values of 0.916–0.987. This excitation–emission-assisted pattern-recognition strategy simplifies sensor design by relying on a single QD nanoreceptor while expanding the scope of neurotransmitters detectable using QD-based assays.126
Kotagiri and co-workers developed a wearable microneedle-based sensor for continuous monitoring of serotonin (5-hydroxytryptamine, 5-HT) in interstitial fluid (ISF) (Fig. 7). The microneedle array employed Ag/rGO-modified carbon-paste electrodes and enabled orthogonal electrochemical detection using square-wave voltammetry and amperometry, providing real-time and complementary analytical information. The working electrode surfaces were characterized using electroanalytical and morphological techniques. The sensor exhibited linear responses to 5-HT in phosphate-buffered saline over the ranges 3–21 µM and 6–60 µM, and demonstrated stable, continuous monitoring in artificial ISF containing bovine serum albumin, with linearity from 13.5 to 95 µM. Long-term operation, resistance to biofouling, and high selectivity against common interferents were confirmed. Performance validation in a skin-mimicking model highlights the platform's potential for real-time 5-HT monitoring in ISF.127
 | ||
| Fig. 7 Schematic representation of a microneedle-based electrochemical sensor for serotonin (5-HT) detection. (i) Illustration of the wearable sensor patch mounted on the skin, showing microneedle penetration into the tissue for interstitial-fluid sampling. (ii) Structural design of the working electrode (WE) and its surface functionalization, including conductive polymer (CP), chitosan, Ag/rGO nanocomposite, and a Nafion antifouling layer, together with the associated electron-transfer mechanism at the modified interface. (iii) Portable Bluetooth-enabled handheld potentiostat integrated with a smartphone for signal acquisition and data analysis. (iv) FE-SEM micrographs and optical photographs of the microneedle array prior to packaging. Adapted with permission from ref. 127. Copyright 2023, American Chemical Society. | ||
A compact organic electrochemical transistor (OECT) sensor incorporating carbon quantum dots (CQDs) was developed to enhance the transconductance of an electropolymerized poly(3,4-ethylenedioxythiophene) (PEDOT) channel for sensitive and selective dopamine (DA) detection (Fig. 8). Precise monitoring of DA is essential for the diagnosis and management of dopamine-related disorders. Integration of CQDs into the PEDOT matrix resulted in a threefold increase in the oxidation current at the DA peak in differential pulse voltammetry, while maintaining excellent selectivity even in the presence of high concentrations of common interferents, such as uric acid and ascorbic acid. The CQD-modified OECT exhibited an enhanced amperometric response, with a low detection limit of 55 nM and a wide linear range of 1–500 µM. These results highlight the effectiveness of low-dimensional carbonaceous nanomaterials in improving OECT sensitivity and demonstrate their promise for cost-effective, high-performance biosensing platforms targeting DA and other biologically relevant analytes, with clear implications for point-of-care diagnostics.35
Collectively, QD-integrated wearable platforms illustrate how nanomaterial-enabled signal amplification can be harmonized with skin-compatible device architectures to enable real-time, noninvasive neurochemical monitoring. By combining optical pattern recognition, electrochemical detection, and transistor-based amplification, these systems address key challenges of selectivity, signal drift, and user mobility. Importantly, wearable integration marks a transition from controlled microenvironments to continuously fluctuating physiological conditions, foreshadowing the even stricter demands placed on implantable and in vivo sensing platforms.
5.3. Implantable probes – in vivo and in vitro imaging
Despite the advantages of wearable sensors, direct interrogation of neurotransmitter dynamics within tissues and organs remains inaccessible without implantable or in vitro/in vivo-compatible platforms. At this final stage of translation, analytical performance alone is insufficient; long-term biocompatibility, immune response, signal persistence, and safe clearance become dominant constraints governing sensor viability.The studies discussed in this section reflect a convergence of QD-based optical sensing, electrochemical transduction, and bioimaging strategies to preserve sensitivity in complex living environments. Rather than prioritizing portability, these platforms emphasize functional stability and physiological relevance, enabling real-time mapping of neurotransmitter dynamics in cellular models, animal systems, and clinically relevant biofluids.
Zhao et al. synthesized nitrogen-doped carbon quantum dots (N-CQDs) via a simple, cost-effective hydrothermal method using polyacrylamide as both a carbon and a nitrogen source. The resulting N-CQDs exhibited a high quantum yield of 23.1%, excellent water solubility, and stable fluorescence. These N-CQDs served as effective fluorescent probes for the sensitive and selective detection of dopamine. Under alkaline conditions, dopamine is oxidized to dopamine–quinone, leading to efficient fluorescence quenching of the N-CQDs. A good linear response was observed over the dopamine concentration range of 0.1–200 µM, with a low detection limit of 0.05 µM. The probe demonstrated high selectivity toward dopamine in the presence of common interfering species and was successfully applied to the determination of dopamine in urine samples, with satisfactory recoveries.128
In this study, CQDs were synthesized via a microwave-assisted method and characterized by dynamic light scattering (DLS) and Fourier transform infrared spectroscopy (FTIR) to determine size distribution and chemical purity. Fluorescence studies revealed that the emission intensity of CQDs is strongly dependent on pH and temperature, enabling their application as pH and temperature sensors via fluorescence quenching. Additionally, the sensing performance of CQDs toward dopamine was investigated, achieving a detection limit of 0.2 mM. Dopamine detection was carried out at room temperature using both steady-state and time-resolved fluorescence quenching techniques, and bimolecular quenching rate constants were determined for different dopamine concentrations. These results demonstrate that fluorescence quenching of CQDs provides an effective approach for dopamine detection and multisensing applications.129
In parallel, Raut et al. reported N-doped carbon quantum dot-based fluorescent nanosensors for in vivo visualization of neurotransmitters such as glycine (LOD: 0.01 µM) (Fig. 9)130 and GABA (LOD: 0.16 µM) (Fig. 10)131 in zebrafish models, enabling real-time mapping of inhibitory neurotransmission with high sensitivity and biocompatibility.
 | ||
| Fig. 9 Schematic illustration of the synthesis and glycine-sensing mechanism of the N-CQDs/Cu fluorescent probe and its in vivo imaging application. The bottom panels show fluorescence imaging of zebrafish brainstem tissue treated with N-CQDs/Cu in the absence and presence of glycine, demonstrating selective detection of endogenous glycine in vivo. Adapted with permission from ref. 130. Copyright 2023, American Chemical Society.130 | ||
 | ||
| Fig. 10 Schematic representation of the GABA-sensing mechanism of N-CQDs and their application for in vivo neuroimaging. The interaction of GABA with N-CQDs induces fluorescence quenching, leading to the formation of the N-CQDs/GABA complex via a turn-off sensing mechanism. The lower panels show fluorescence images of zebrafish forebrain tissue treated with N-CQDs alone, after exposure to exogenous GABA, and after treatment with a glutamic acid decarboxylase (GAD) inhibitor, demonstrating the probe's ability to visualize changes in endogenous GABA levels in vivo. Adapted with permission from ref. 131. Copyright 2024, American Chemical Society. | ||
A porous platinum nanoflower-based ternary nanocomposite (Pt NFs/MWCNTs-GQDs) was fabricated and modified onto a glassy carbon electrode for electrochemical epinephrine sensing. The hybrid electrode exhibited excellent electrocatalytic activity toward epinephrine oxidation, enabling sensitive detection over a wide linear range (0.1–800 µM) with a low detection limit of 14 nM. The sensor showed good stability, reproducibility, and selectivity and was successfully applied to the determination of epinephrine in pharmaceutical formulations and biological fluids, demonstrating its practical relevance for medical diagnostics.132
Researchers reported a facile and green one-step hydrothermal synthesis of graphitic carbon nitride quantum dots (g-CNQDs) using polyethylenimine (PEI) as a structure-directing agent. The branched architecture and abundant amine groups of PEI promote self-cyclization, leading to the formation of triazine-based g-CNQDs with bright blue fluorescence (Fig. 11). Compared to conventional multistep methods that employ hazardous reagents, this streamlined approach yields g-CNQDs with moderate quantum yields, excellent aqueous dispersibility, and good biocompatibility. The positively charged g-CNQDs enable sensitive dopamine (DA) detection through electrostatic interactions, forming a non-fluorescent g-CNQDs–DA complex that produces a fluorescence “turn-off” response (Fig. 11). The probe exhibits a wide linear range of 0.1–300 µM and a low detection limit of 43 nM (S/N = 3). High selectivity was confirmed in human urine and serum samples, with recoveries of 97–106%, highlighting its potential for sustainable and selective bioanalytical sensing applications.133
 | ||
| Fig. 11 Diagrammatic representation of dopamine detection employing PEI-functionalized g-CNQDs synthesized through a facile hydrothermal route. | ||
Saisree et al. developed a highly stable copper nanocluster (CuNC) system that remained stable for over one year. The nanoclusters were synthesized using nitrogen-doped graphene quantum dots (N-GQDs) as both reducing and capping agents, with glutathione serving as an additional stabilizing ligand. The resulting CuNC@N-GQD composite enabled simultaneous electrochemical detection of dopamine (DA), serotonin (SER), and nicotine (NIC), producing well-defined oxidation peaks with good peak-to-peak separation. Notably, neither bare CuNCs nor N-GQDs alone exhibited such simultaneous sensing capability. The composite showed significantly enhanced sensitivity, with current responses approximately fourfold higher for DA and SER and twofold higher for NIC compared to the controls. The limits of detection were 0.001 nM (DA), 1.0 nM (SER), and 0.01 nM (NIC). The superior sensitivity and multiplexed sensing performance arise from the synergistic interaction between CuNCs and N-GQDs. The sensor was successfully validated in spiked blood and urine samples, demonstrating excellent applicability in real biological matrices.58
De et al. further extended this approach to intracellular imaging of adenine using an N-CQDs/Ag composite (LOD: 0.076 µM, response time: 10 s), demonstrating that metal-enhanced carbon nanodots can function reliably in complex biological environments (Fig. 12).134
 | ||
| Fig. 12 Schematic illustration of the adenine-sensing mechanism of the N-CQDs/Ag composite and its application in biological imaging. Coordination of Ag+ ions with N-CQDs results in fluorescence quenching (FL OFF). Upon selective binding of adenine, Ag+ preferentially forms an adenine–Ag+ complex, leading to fluorescence recovery of the N-CQDs through a turn-on sensing mechanism. The lower panels display fluorescence imaging results in vitro and in vivo, along with comparisons to control samples, demonstrating the capability of the N-CQDs/Ag probe for sensitive intracellular adenine detection in biological systems. Adapted with permission from ref. 134. Copyright 2025, American Chemical Society. | ||
Extensive research has shown that coordinated signalling between multiple neurotransmitters, particularly serotonin, glutamate, and dopamine (DA), is crucial for striatal function and its associated behaviors and pathologies. A CQD-modified electrochemical platform was developed by Mukherjee and his co-workers, integrating carbon quantum dots derived from corn seeds with an electrochemically deposited NiWO4 thin film on an FTO electrode for enzyme-free detection of serotonin (Fig. 13). The CQDs enhanced electron transfer and serotonin adsorption, enabling sensitive and selective detection with a nanomolar detection limit (134 nM), good resistance to common interferents, and stable performance over repeated sensing cycles, demonstrating the potential of CQD-semiconductor nanocomposites for electrochemical neurotransmitter sensing in physiologically relevant concentration ranges.135
 | ||
| Fig. 13 (i) Graphic representation of the synthesis of the NiWO4/CQD nanocomposite and subsequent fabrication of the modified FTO/NiWO4/CQD sensing electrode. (ii) Illustration of the electrochemical sensing mechanism for serotonin at the CQD-functionalized NiWO4 electrode, highlighting the role of CQDs in facilitating adsorption and charge transfer during the detection process. Adapted with permission from ref. 135. Copyright 2026, American Chemical Society. | ||
Glutamate and dopamine (DA) are critical neurotransmitters governing striatal function, motor coordination, and motivated behavior. While electroanalytical techniques allow rapid, spatially resolved detection of DA, glutamate is non-electroactive and is typically measured using enzyme-based amperometric biosensors, which suffer from limited selectivity and complex surface modifications. To overcome these limitations, a novel glutamate microbiosensor fabricated on a carbon-fiber microelectrode and integrated with fast-scan cyclic voltammetry (FSCV) was developed to enable the simultaneous, subsecond quantification of glutamate and DA at single brain sites. The sensor demonstrated high sensitivity, stability, and selectivity using an optimized voltammetric waveform. Its applicability was validated in the rat ventral striatum, where electrically evoked glutamate and DA release was monitored at micrometer-scale locations before and after pharmacological modulation. This technological advance provides a powerful platform for probing glutamate–dopamine interactions underlying neural circuitry, motor integration, and maladaptive behaviors.136
Together, implantable and in vivo platforms complete the translational arc of QD-based neurochemical sensing, demonstrating that carefully engineered nanomaterial–platform combinations can operate within living systems while retaining sensitivity and selectivity. When viewed alongside microfluidic and wearable integrations, these advances reveal that platform integration is the unifying principle governing the successful transition of QD sensors from benchtop demonstrations to clinically and biologically meaningful neurodiagnostic technologies.
6. Key analytical performance metrics and selectivity challenges
6.1. Limits of detection (LOD) – what the literature shows and what the numbers mean
QD-based neurotransmitter sensors report a wide range of LODs, depending on the transduction mode (optical fluorescence, ECL, PEC, electrochemical), QD type, and whether signal amplification (enzymes, nanozymes, nanopores) is used. In controlled buffer assays, many fluorescence/ECL/PEC platforms achieve LODs in the low-nanomolar to picomolar range; doped carbon-dot electrodes and chemiluminescent QD systems have reported LODs in the high-pM to low-nM range under optimized conditions.137The current best-reported single-molecule/electrical sensitivity for small neurotransmitters comes from aptamer-modified nanopore platforms rather than conventional QD fluorescence assays. Stuber et al. demonstrated femtomolar detection of dopamine with an aptamer-functionalized nanopore and carefully correlated binding kinetics with signal statistics. Such femtomolar claims are technically achievable in highly controlled, low-volume experiments but require stringent validation before being accepted as practical LODs in complex matrices (serum, CSF).45
Practical takeaway: expect a realistic performance gap between buffer LODs and matrix LODs – many QD sensors lose one to several orders of magnitude in sensitivity when moved into serum/CSF because of background fluorescence, quenching by matrix components, and nonspecific adsorption. Reviews and method papers emphasize reporting both buffer and real-sample LODs.137
6.2. Selectivity – the persistent problem (DA vs. AA/UA) and practical solutions
Discriminating dopamine (DA) from ascorbic acid (AA) and uric acid (UA) is a recurring analytical challenge because these interferents are present at much higher concentrations in biological fluids and often undergo similar redox/electrochemical or quenching interactions at nanomaterial surfaces. Simple strategies – e.g., a negatively charged exclusion layer (Nafion) or size-/charge-selective polymer coatings – work well in electrochemical setups but are less effective in fluorescence assays where nonspecific adsorption and inner-filter effects dominate.138Combining orthogonal recognition elements is one of the most effective ways to improve specificity: aptamer + MIP (dual recognition), aptamer + exclusion coating, or aptamer–QD hybrids combined with electrochemical pretreatment show markedly better discrimination in recent reports. For example, Shen et al. demonstrated a dual aptamer–MIP gold platform with enhanced selectivity over AA/UA by requiring both shape/functional complementarity (MIP) and sequence-level binding (aptamer).138
Other effective approaches:
•Boronic-acid motifs (selective for catechol cis-diols) or β-cyclodextrin host motifs for aromatic inclusion when appropriate.
•Kinetic discrimination (time-resolved readout) exploiting different adsorption/oxidation rates of DA vs. AA/UA.
•Pre-oxidation/electrochemical pretreatment to shift interferent potentials away from the DA response.138
6.3. Stability and reproducibility: material and practical bottlenecks
Three stability axes are most important for QD sensors:Photostability. Colloidal QDs are more photostable than organic dyes but still can photobleach or undergo photo-oxidation under prolonged illumination; photostability varies with QD composition and surface passivation. Accelerated photostability studies and reviews point out that ligand chemistry and shell quality (graded shells, ZnS capping) strongly influence operational photostability.139
Colloidal/chemical stability in saline and proteinaceous media. Ionic strength and serum proteins cause ligand displacement, aggregation, and quenching. This is a particularly acute problem for halide perovskite QDs, which are intrinsically moisture- and oxygen-sensitive and susceptible to lead leaching unless embedded in protective matrices or converted into more robust phases (e.g., CsPb2Br5/CdSe heterojunctions or polymer/SiO2 encapsulation). Recent papers demonstrate water-stable perovskite-QD heterojunctions and encapsulation schemes that substantially improve operational stability for PEC sensing of dopamine.41
Batch-to-batch reproducibility and synthetic variability. Small differences in precursor purity, ligand coverage and shell thickness can produce large differences in PL quantum yield, surface trap densities and therefore sensor response. Reviews of QD sensing platforms call out reproducible synthesis, rigorous characterization (PL quantum yield, FWHM, zeta potential) and inter-batch QC as necessary steps before claiming clinical applicability.139
Practical implication: for clinical or long-term biosensing studies, passivated QDs (thick graded shells or silica/polymer encapsulation), demonstrate metal-ion leaching tests (ICP), and show photostability under intended illumination.41,139
7. Toxicity, safety and regulatory considerations
7.1. Intrinsic material toxicity: heavy-metal content and leaching
•Many common QDs (e.g., CdSe, CdTe, and similar II–VI QDs) contain cadmium (a heavy metal), and their toxicity is well documented. In particular, cytotoxicity arises largely from the leakage of Cd2+ ions when the QD core degrades or the shell/ligand coating fails.140•In vivo studies show that QDs tend to accumulate in major organs (liver, kidney, etc.), with associated oxidative stress, biochemical disruption, and potential long-term damage. For example, a recent in vivo study in rats showed biochemical and oxidative damage after administration of CdSe QDs.141
•Reviews summarizing decades of nanotoxicology of QDs highlight that free metal-ion release and surface-mediated ROS generation are the main mechanisms of cytotoxicity.142
•Even if a QD is coated with a protective shell (e.g., ZnS, silica, polymer), such protection is not always sufficient: over time, or under physiological conditions (saline, proteins, varying pH), shells/ligands may degrade or be displaced – leading to core exposure and ion release.143
7.2. Long-term fate, biodistribution and clearance
•A critical concern is what happens to QDs after administration: whether they accumulate in organs, how long they persist, and whether they are excreted or degraded safely. Many studies show that QDs – especially heavy-metal QDs – tend to accumulate in the liver, kidneys, spleen, lungs, etc.144•For example, in a 2020 study on inhaled InP/ZnS (cadmium-free) QDs, the authors observed that after translocation from the lungs to circulation, QDs accumulated in major organs and altered some blood and serum markers – even though no gross histopathological changes were observed in most organs after 15 days.145
•This indicates that even presumed “safer” QDs (cadmium-free, e.g., InP-based) may still accumulate and impact physiology – albeit at levels less dramatic than Cd-based QDs.
•The long-term fate (months, years) is often unknown – few studies track chronic biodistribution, biodegradation, or excretion over long times. This is a serious unknown that complicates translation to clinical or repeated-dose applications. Reviews of regulatory outlook also highlight this as a major barrier.146
7.3. Safer alternatives: carbon QDs, InP QDs, heavy-metal-free QDs but not free of concerns
Because of these toxicity and safety limitations, researchers have increasingly turned to heavy-metal-free or low-toxicity QDs, such as carbon quantum dots (CQDs), graphene QDs, or III–V QDs like InP as alternatives.•Carbon QDs (CQDs): many in vitro and some in vivo studies indicate low cytotoxicity and good biocompatibility compared to heavy-metal QDs. For example, one study reported that CQDs had no significant adverse effects on cell viability, apoptosis, or major functional readouts in mammalian cells – supporting their suitability for bioimaging and potentially biosensing.147
•InP/other non-Cd/Qd QDs: compared side by side with Cd-based QDs, cadmium-free QDs such as InP/ZnS or other I–III–V materials show lower cytotoxicity, lower ROS generation, and generally better cell viability in many cell lines.148
•Indeed, a recent comparative toxicity study on liver-derived cell lines (HepG2 and THLE-2) found that nitrogen-doped carbon dots (NCDs) had the least toxicity, while InP/ZnS and other cadmium-free QDs still exhibited some cytotoxicity and ROS generation at higher concentrations.149
•Reviews and recent assessments of QD biocompatibility conclude that surface chemistry, shell composition, ligand choice, and dosage often matter more than the core material itself; with good passivation and carefully optimized coatings, even some heavy-metal QDs can have acceptable short-term biocompatibility – though long-term safety remains uncertain.
8. Outlook: emerging directions and recommendations
8.1. Multiplexed optical coding and lifetime imaging
Narrowband QD emissions and lifetime-resolved detection enable simultaneous monitoring of multiple neurotransmitters while correcting for intensity fluctuations and matrix effects. It empowers simultaneous detection of multiple neurotransmitters by assigning unique spectral, intensity, or fluorescence-lifetime “codes” to different QDs or recognition elements. Because fluorescence lifetime is largely independent of probe concentration and excitation fluctuations, lifetime imaging microscopy (FLIM) offers robust, interference-resistant readouts in complex biofluids or tissues. Together, these strategies enhance analytical specificity and allow parallel, high-fidelity neurotransmitter sensing.8.2. Ratiometric and lifetime-based sensors for robust quantitation
Ratiometric sensors using dual-emission QDs, or lifetime shifts that are immune to excitation intensity and scattering, will improve quantitative accuracy in complex biofluids. These improve quantitative accuracy by using internal optical references that compensate for fluctuations in excitation intensity, probe concentration, and matrix effects. In ratiometric systems, analyte binding modulates two emission bands with opposite or proportional changes, producing self-calibrated signals. Lifetime-based sensing leverages analyte-induced shifts in fluorescence decay, which are inherently resistant to scattering and photobleaching, enabling more reliable neurotransmitter quantitation in complex biological environments.8.3. Biodegradable/renal-clearable QDs
Designing QDs that degrade into non-toxic fragments or are small/modified enough for renal clearance will be pivotal for in vivo translation. These dots are engineered to overcome long-term retention and toxicity concerns by using ultrasmall cores, degradable surface ligands, or polymer/silica scaffolds that break down into excretable fragments. Sizes below the renal filtration threshold (∼5–6 nm) enable rapid urinary clearance, while biodegradable coatings prevent heavy-metal release. These designs improve biocompatibility and significantly broaden the clinical translation potential of QD-based neurotransmitter sensors.8.4. Hybrid QD–polymer wearable patches with wireless readout
Integrating QD sensors into soft, conformable films with on-board excitation/detection and wireless telemetry can enable ambulatory neurochemical phenotyping, a major leap for psychiatry and personalised neurology. These patches integrate fluorescent or electro-optical QDs within stretchable, breathable polymer matrices to create skin-conformal sensing platforms. These patches can monitor neurotransmitter-related biomarkers in sweat or interstitial fluid, while embedded micro-LEDs, photodetectors, or NFC modules enable wireless, real-time readout. The hybrid design provides mechanical durability, biocompatibility, and stable signal transduction, supporting continuous, noninvasive physiological monitoring.8.5. Standardization and validation in human samples
To move beyond proof-of-concept, researchers should prioritize head-to-head comparisons in clinically relevant matrices (blood, CSF, saliva), interlaboratory reproducibility studies, and blinded clinical validations against established assays (HPLC, electrochemical microdialysis). Standardisation and validation are essential for translating QD-based neurotransmitter sensors into clinical practice. This requires rigorous calibration in physiologically relevant matrices (plasma, serum, CSF), evaluation of interferents, and cross-validation against gold-standard methods such as HPLC-MS. Batch-to-batch reproducibility, storage stability, and patient-to-patient variability must be assessed. Clear reporting of accuracy, precision, recovery, and robustness ensures reliable performance in real-world diagnostics.9. A brief comparison with recent reviews
This section contrasts the scope and focus of the current review with recent articles on neurotransmitter sensing to highlight its unique contributions (Table 3). Existing reviews on neurotransmitter sensing largely treat electrochemical, fluorescence, and aptamer-based platforms in isolation, with a strong bias toward carbon quantum dot electrochemistry or single-analyte fluorescence assays. They rarely address how quantum dot photophysics enables spectral/temporal coding, ECL/PEC transduction, and genuine multiplexed detection. This review uniquely integrates these mechanisms with advanced recognition strategies to define practical routes toward real-time, multi-neurotransmitter diagnostics, a gap that is not systematically addressed in recent literature.| Review (topic/focus) | Main scope | How this review differs/adds value | Ref. |
|---|---|---|---|
| Fluorescent-based neurotransmitter sensors | Focused on fluorescence-based biosensors, including metal clusters, carbon dots, and QDs for NT detection; discusses mechanisms such as FRET, PET, and ICT, and challenges for fluorescence NT sensing | The present review goes beyond fluorescence to comprehensively cover optical (FRET/temporal codes), electrochemical, ECL, PEC, and hybrid approaches with QDs, including device integration and translational barriers | 76 |
| Carbon-based quantum dots for electrochemical detection | Examines carbon QD and graphene QD electrochemical sensors for monoamine NTs over the past decade, focusing on CQD/GQD properties, synthesis, and electrochemical performance | The present review expands beyond CQD/GQD electrochemical sensors to include PQDs, hybrid QD systems, optical codes, aptamer/QD constructs, nanomaterial integration, and real-world diagnostic contexts | 19 |
| Recent advances in aptamer-based sensors for neurotransmitters | Summarises the latest progress in aptamer-based biosensor technologies for NTs, focusing on biomaterials and selectivity mechanisms | The present review incorporates aptamer approaches and contextualises them within QD transduction platforms, ratiometric/spectral coding schemes, and system-level integration, aspects not covered in purely aptamer-focused reviews | 44 |
| Nanotechnology-driven electrochemical neurotransmitter sensing | Reviews developments in electrochemical sensors, including nanostructured electrodes, lab-on-chip/wearable formats, and analytical performance improvements | The present review emphasises optical transduction (FRET/ECL/PEC) and multiplexed, spectral-coded QD strategies, which are largely absent in purely electrochemical summaries | 150 |
| Latest trends in electrochemical NT sensors (2019) | Historical overview of electrochemical NT sensors, enzyme-based and enzyme-free, focusing on sensitivity and nanomaterial enhancements | The present review updates this chronologically and extends into new sensing paradigms (optical codes, QD hybrids, temporal coding) and translational diagnostics | 151 |
| Recent advancements in nanosensors for NT detection | Covers advances in optical and electrochemical nanosensors for neurotransmitters | The present work uniquely integrates spectral/temporal coding, hybrid QDs, aptamer/MIP combinations, and device translation issues, giving a more unified QD-centric perspective | 152 |
| A review of carbon quantum dots in depression-related NT sensing | Focused specifically on CD-based electrochemical NT sensors in the context of depression, and the synthesis/structure of CDs | The present review extends to all QD types (CQDs, PQDs, hybrid) and multiple analytical modalities (optical, electrochemical, ECL, PEC, ratiometric codes), as well as translational diagnostics | 153 |
| Electrochemical nanobiosensors for NT detection | General overview of electrochemical biosensors with nanomaterials for NTs, including cell-based and in vitro/in vivo monitoring | The present article complements this by incorporating optical quantum-dot codes and detailed surface-chemistry selectivity motifs that have not been comprehensively covered before | 154 |
| Aptamer renaissance for neurochemical biosensing | Highlights the role of aptamers integrated with electronic platforms in addressing limitations in neurochemical sensing | The present work integrates this aptamer focus into a broader QD and optical code framework, illustrating how aptamers can be coupled to multifaceted QD sensing architectures | 155 |
| Carbon dots review (CD sensors in general) | Focuses on carbon dots and their various biosensing applications, comparing categories of CD sensors and performance metrics | The present review situates CDs within a broader context of neurotransmitter sensing, connecting CD performance to QD spectral coding, multiplexing, and translation into real biological matrices | 156 |
| Carbon dots and their nanocomposite-based sensors for neurotransmitter detection | Focuses on carbon dots (CDs) and CD-based nanocomposites used for optical and electrochemical sensing of neurotransmitters (primarily epinephrine and norepinephrine). Emphasizes synthesis routes, photoluminescence mechanisms, and representative sensing platforms | The present review goes beyond CD-centric systems by systematically comparing multiple quantum dot classes (carbon, metal chalcogenide, perovskite, hybrid QDs) and multiple transduction modes (fluorescence, ECL, PEC, electrochemical, nanopore-based electrical sensing). It critically analyzes LOD realism (buffer vs. biofluids), selectivity challenges (DA vs. AA/UA), stability, reproducibility, and clinical translatability, which are only briefly discussed or not quantitatively benchmarked in this review | 157 |
Conclusions
Quantum dots (QDs) are poised to redefine molecular neurodiagnostics, offering an unprecedented combination of brightness, tunable emission, and modular surface chemistry that enables highly sensitive, multiplexed detection architectures. Their compatibility with diverse recognition elements, such as aptamers, MIPs, redox enzymes, and charge-selective coatings, allows the construction of hybrid sensing platforms that couple molecular specificity with powerful optical or electrochemical transduction. Carbon QDs and engineered InP, CdSe/ZnS, and perovskite/polymer composites are emerging as particularly promising candidates for wearable, implantable, and point-of-care devices, where flexibility, robustness, and biocompatibility are essential.Yet, the path to clinical translation is not solely a materials challenge; stability in complex human biofluids, selective neurotransmitter discrimination against abundant interferents, and predictable in vivo safety profiles remain formidable barriers. Addressing these requires integrative strategies of encapsulation and graded-shell passivation for photostability, dual-recognition architectures for selectivity, and biodegradable or renal-clearable designs for safety. Equally crucial are standardized analytical protocols, cross-validation with gold-standard clinical assays, and multi-center assessments to ensure reproducibility across batches and patient cohorts.
With coordinated progress in material design, biointerface engineering, and regulatory-grade validation, QD-enabled neurotransmitter sensors can evolve from laboratory prototypes into reliable clinical tools, supporting precision neurochemical monitoring and next-generation neurological diagnostics.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this feature article.Acknowledgements
SM and PS acknowledge the Department of Chemistry, Visva-Bharati, for providing the infrastructural facilities.References
- À. García-Cazorla and R. Artuch, in Rosenberg's Molecular and Genetic Basis of Neurological and Psychiatric Disease, ed. R. N. Rosenberg and J. M. Pascual, Academic Press, 5th edn, 2015, pp. 703–712 Search PubMed.
- S. R. Batten, D. Bang, B. H. Kopell, A. N. Davis, M. Heflin, Q. Fu, O. Perl, K. Ziafat, A. Hashemi, I. Saez, L. S. Barbosa, T. Twomey, T. Lohrenz, J. P. White, P. Dayan, A. W. Charney, M. Figee, H. S. Mayberg, K. T. Kishida, X. Gu and P. R. Montague, Nat. Hum. Behav., 2024, 8, 718–728 Search PubMed.
- L. Shuyi, G. Mingzhou, M. Zijun, Z. Haiyan, W. Ying and Z. Yujiao, Prog. Neuro-Psychopharmacol. Biol. Psychiatry, 2025, 141, 111475 CrossRef PubMed.
- A. Hahn, M. B. Reed, M. Murgaš, C. Vraka, S. Klug, C. Schmidt, G. M. Godbersen, B. Eggerstorfer, D. Gomola, L. R. Silberbauer, L. Nics, C. Philippe, M. Hacker and R. Lanzenberger, Mol. Psychiatry, 2025, 30, 600–607 CrossRef CAS PubMed.
- J. R. Thorstensen, T. T. Henderson and J. J. Kavanagh, Neuropharmacology, 2024, 242, 109761 CrossRef CAS PubMed.
- L. An, S. Hong, T. Turon, A. Pavletic, C. S. Johnson, J. A. Derbyshire and J. Shen, Magn. Reson. Med., 2025, 94, 4–14 CrossRef CAS PubMed.
- R. I. Teleanu, A.-G. Niculescu, E. Roza, O. Vladâcenco, A. M. Grumezescu and D. M. Teleanu, Int. J. Mol. Sci., 2022, 23, 5954 Search PubMed.
- M. Salehirozveh, P. Dehghani and I. Mijakovic, ACS Nano, 2025, 19, 24404–24424 CrossRef CAS PubMed.
- E. S. Bucher and R. M. Wightman, Annu. Rev. Anal. Chem., 2015, 8, 239–261 CrossRef CAS PubMed.
- H. Brennenstuhl, S. Jung-Klawitter, B. Assmann and T. Opladen, Neuropediatrics, 2019, 50, 2–14 Search PubMed.
- J. Ng, S. J. Heales and M. A. Kurian, Paediatr. Drugs, 2014, 16, 275–291 CrossRef CAS PubMed.
- T. Manimekala, R. Sivasubramanian and G. Dharmalingam, J. Electron. Mater., 2022, 51, 1950–1973 Search PubMed.
- Y. Da, S. Luo and Y. Tian, ACS Appl. Mater. Interfaces, 2023, 15, 138–157 Search PubMed.
- P. Dabnichki and T. Y. Pang, Biosensors, 2024, 14, 628 CrossRef PubMed.
- G. Rizzi and K. R. Tan, Front. Neural Circuits, 2017, 11, 110 CrossRef PubMed.
- Y.-F. Zhang and S. J. Cragg, Neuron, 2021, 109, 1070–1071 CrossRef CAS PubMed.
- E. H. Kim, G. Chin, G. Rong, K. E. Poskanzer and H. A. Clark, Acc. Chem. Res., 2018, 51, 1023–1032 Search PubMed.
- M. Perry, Q. Li and R. T. Kennedy, Anal. Chim. Acta, 2009, 653, 1–22 Search PubMed.
- S. E. Elugoke, A. S. Adekunle, O. E. Fayemi, B. B. Mamba, E. M. Sherif and E. E. Ebenso, Biosensors, 2020, 10, 162 CrossRef CAS PubMed.
- C. Liu, X. Lin, J. Liao, M. Yang, M. Jiang, Y. Huang, Z. Du, L. Chen, S. Fan and Q. Huang, Chin. Chem. Lett., 2024, 35, 109598 CrossRef CAS.
- C. Yang, M. E. Denno, P. Pyakurel and B. J. Venton, Anal. Chim. Acta, 2015, 887, 17–37 CrossRef CAS PubMed.
- S. Y. Lim, W. Shen and Z. Gao, Chem. Soc. Rev., 2015, 44, 362–381 RSC.
- W. Su, H. Wu, H. Xu, Y. Zhang, Y. Li, X. Li and L. Fan, Mater. Chem. Front., 2020, 4, 821–836 RSC.
- V. Magesh, A. K. Sundramoorthy and D. Ganapathy, Front. Mater., 2022, 9, 906838 CrossRef.
- A. Loskutova, A. Seitkali, D. Aliyev and R. Bukasov, Int. J. Mol. Sci., 2025, 26, 6674 CrossRef CAS PubMed.
- Z. A. Qureshi, H. Dabash, D. Ponnamma and M. K. G. Abbas, Heliyon, 2024, 10, e31634 CrossRef CAS PubMed.
- P. Abdul Rasheed, M. Ankitha, V. K. Pillai and S. Alwarappan, RSC Adv., 2024, 14, 16001 RSC.
- B. D. Mansuriya and Z. Altintas, Sensors, 2020, 20, 1072 CrossRef CAS PubMed.
- T. H. Le, D. H. Lee, J. H. Kim and S. J. Park, Korean J. Chem. Eng., 2020, 37, 1000–1007 CrossRef CAS.
- S. R. Lodha, J. G. Merchant, A. J. Pillai, A. H. Gore, P. O. Patil, S. N. Nangare, G. G. Kalyankar, S. A. Shah, D. R. Shah and S. P. Patole, Heliyon, 2024, 10, e41020 CrossRef CAS PubMed.
- Y. Wang, R. Hu, G. Lin, I. Roy and K.-T. Yong, ACS Appl. Mater. Interfaces, 2013, 5, 2786–2799 CrossRef CAS PubMed.
- R. Ding, Y. Chen, Q. Wang, Z. Wu, X. Zhang, B. Li and L. Lin, J. Pharm. Anal., 2022, 12, 355–364 CrossRef CAS PubMed.
- A. Tiwari, S. Walia, S. Sharma, S. Chauhan, M. Kumar, T. Gadly and J. K. Randhawa, J. Mater. Chem. B, 2023, 11, 1029–1043 RSC.
- J. Hu, J. Li, Q. Guo, G. Du, C. Li, R. Li, R. Zhou and H. He, Int. J. Mol. Sci., 2024, 25, 10346 CrossRef CAS PubMed.
- J. Gamboa, R. El Attar, D. Thuau, F. Estrany, M. Abbas and J. Torras, Mikrochim. Acta, 2024, 191, 639 CrossRef CAS PubMed.
- L. Han, Y. Guo, H. Zhang, Z. Wang, F. Zhang, Y. Wang, X. Li, Y. Wang and J. Ye, RSC Adv., 2024, 14, 1813–1821 Search PubMed.
- T. Y. Kao, C. H. Kuo, Y. W. Wu and S. C. Luo, ACS Meas. Sci. Au, 2024, 4, 712–720 CrossRef CAS PubMed.
- C. Ma, Y. Wen, Y. Qiao, K. Z. Shen and H. Yuan, Molecules, 2024, 29, 2925 CrossRef CAS PubMed.
- R. H. Al Omari, M. E. A. Zaki, G. P. Priya, Q. H. Aziz, Y. Sasikumar, A. Aldulaimi, R. Sharma, S. M. Gomha and N. Sarhan, Electroanalysis, 2025, 37, e70083 CrossRef.
- R. H. Al Omari, G. PadmaPriya, S. Al-Hasnaawei, S. Ray, K. Chennakesavulu, R. Sharma, A. S. Chauhan and N. Sarhan, Chin. J. Anal. Chem., 2025, 100677 Search PubMed.
- G. Zhao, X. Sun, S. Li, J. Zheng, J. Liu and M. Huang, Nanoscale, 2024, 16, 2621–2631 Search PubMed.
- S. Xiao, Y.-T. Yang, Y.-F. Chen, J.-L. Liu, Y.-Q. Chai and R. Yuan, Biosens. Bioelectron., 2024, 254, 116193 CrossRef CAS PubMed.
- F. Yang, X. Han, Y. Ai, B. Shao, W. Ding, K. Tang and W. Sun, Molecules, 2024, 29, 744 CrossRef CAS PubMed.
- J.-H. Park, Y.-S. Eom and T.-H. Kim, Biosensors, 2023, 13, 413 CrossRef CAS PubMed.
- A. Stuber, A. Douaki, J. Hengsteler, D. Buckingham, D. Momotenko, D. Garoli and N. Nakatsuka, ACS Nano, 2023, 17, 19168–19179 CrossRef CAS PubMed.
- Y. Ouyang, Y. Biniuri, M. Fadeev, P. Zhang, R. Carmieli, M. Vázquez-González and I. Willner, J. Am. Chem. Soc., 2021, 143, 11510–11519 CrossRef CAS PubMed.
- D. Kim and S. Yoo, Chemosensors, 2021, 9, 318 CrossRef CAS.
- Y. Mu, Z. Chen, J. Zhan and J. Zhang, Anal. Sens., 2024, 4, e202400027 Search PubMed.
- H. M. N. Ahmad, A. Andrade and E. Song, Biosensors, 2023, 13, 983 CrossRef CAS PubMed.
- M. A. Boles, D. Ling, T. Hyeon and D. V. Talapin, Nat. Mater., 2016, 15, 141–153 Search PubMed.
- O. Chen, J. Zhao, V. P. Chauhan, J. Cui, C. Wong, D. K. Harris, H. Wei, H. S. Han, D. Fukumura, R. K. Jain and M. G. Bawendi, Nat. Mater., 2013, 12, 445–451 Search PubMed.
- H. He, S. Deng and Y. Liu, Discov. Nano, 2025, 20, 132 CrossRef PubMed.
- P. Linkov, V. Krivenkov, I. R. Nabiev and P. S. Samokhvalov, Mater. Today Proc., 2016, 3, 104–108 CrossRef.
- X. Lu, X. Hou, H. Tang, X. Yi and J. Wang, Nanomaterials, 2022, 12, 4031 Search PubMed.
- I. L. Medintz, A. R. Clapp, H. Mattoussi, E. R. Goldman, B. Fisher and J. M. Mauro, Nat. Mater., 2003, 2, 630–638 CrossRef CAS PubMed.
- S. E. Elugoke, G. E. Uwaya, T. W. Quadri and E. E. Ebenso, Carbon Quantum Dots: Basics, Properties, and Fundamentals, ACS Symposium Series, 2024, vol. 1465, ch. 1 Search PubMed.
- Z. Nazari, M. H. Nematollahi, F. Zareh, B. Pouramiri and M. Mehrabani, ChemistrySelect, 2023, 8, e202203630 CrossRef CAS.
- S. Saisree, J. S. Arya Nair and K. Y. Sandhya, J. Mater. Chem. B, 2022, 10, 3974–3988 RSC.
- A. M. Mahmoud, M. H. Mahnashi, K. Alhazzani, A. Z. Alanazi, M. M. Algahtani, A. Alaseem, B. A. Alyami, A. O. AlQarni and M. M. El-Wekil, Spectrochim. Acta, Part A, 2021, 252, 119516 CrossRef CAS PubMed.
- M. S. Al Ja’farawy, D. Thirumalai, J. Lee, H. S. Jung, S.-C. Chang, J.-H. Yoon and D.-H. Kim, J. Anal. Sci. Technol., 2023, 14, 29 CrossRef.
- P. Feng, X. Yang, X. Feng, G. Zhao, X. Li, J. Cao, Y. Tang and C.-H. Yan, ACS Nano, 2021, 15, 6266–6275 CrossRef CAS PubMed.
- M. Kedhim, V. Jain, S. Ballal, A. Kumar, A. Singh, V. Kavitha, R. Panigrahi and H. Noorizadeh, RSC Adv., 2025, 15, 18875–18892 RSC.
- X. Zhang, M. Wang, Y. Zhang, P. Zhao, J. Cai, Y. Yao and J. Liang, Molecules, 2023, 28, 3646 CrossRef CAS PubMed.
- M. Chatterjee, P. Nath, S. Kadian, A. Kumar, V. Kumar, P. Roy, G. Manik and S. Satapathi, Sci. Rep., 2022, 12, 9061 CrossRef CAS PubMed.
- T. Azargoshasb, H. A. Navid, S. Yadollahzadeh, R. Aghbolaghi, R. Parvizi, G. Parsanasab and S. Kumar, IEEE Sens. J., 2024, 24, 4510–4522 Search PubMed.
- A. Biswas, S. Lee, P. Cencillo-Abad, M. Karmakar, J. Patel, M. Soudi and D. Chanda, Sci. Adv., 2024, 10, eadp7460 CrossRef CAS PubMed.
- A. M. Dennis, W. J. Rhee, D. Sotto, S. N. Dublin and G. Bao, ACS Nano, 2012, 6, 2917–2924 CrossRef CAS PubMed.
- G. Boas, Photonics Spectra, 2011, 45(9), 78–83 Search PubMed.
- M. C. Dos Santos, W. R. Algar, I. L. Medintz and N. Hildebrandt, TrAC, Trends Anal. Chem., 2020, 125, 115819 CrossRef.
- M. Chern, R. Toufanian and A. M. Dennis, Analyst, 2020, 145, 5754–5767 RSC.
- K. Boeneman, J. B. Delehanty, K. Susumu, M. H. Stewart, J. R. Deschamps and I. L. Medintz, Adv. Exp. Med. Biol., 2012, 733, 63–74 CrossRef CAS PubMed.
- A. Yadav, P. Dogra, P. Sagar, M. Srivastava, A. Srivastava, R. Kumar and S. K. Srivastava, Spectrochim. Acta, Part A, 2025, 335, 126002 CrossRef CAS PubMed.
- C. Moraldo, E. Vuille-dit-Bille, B. Shkodra, T. Kloter and N. Nakatsuka, J. Neurosci. Methods, 2022, 365, 109386 Search PubMed.
- X. Wu, X. Zhang, J. Ma, Y. Zhang and M. Li, Spectrochim. Acta, Part A, 2024, 304, 123374 CrossRef CAS PubMed.
- R. Zhu, L. Yu, L. Guo, X. Ma and Z. Meng, Microchem. J., 2024, 201, 110577 CrossRef CAS.
- R. Govindaraju, S. Govindaraju, K. Yun and J. Kim, Biosensors, 2023, 13, 1008 Search PubMed.
- T. Cheng, N. Afshan, J. Jiao and J. Jiao, Biosens. Bioelectron. X, 2024, 20, 100528 Search PubMed.
- R. Liu and C. C. Ko, Biosensors, 2023, 13, 295 Search PubMed.
- A. G. Ayankojo, J. Reut and V. Syritski, Biosensors, 2024, 14, 71 CrossRef CAS PubMed.
- X. Zhou, A. Wang, C. Yu, S. Wu and J. Shen, ACS Appl. Mater. Interfaces, 2015, 7, 11741–11747 CrossRef CAS PubMed.
- R. Zhu, M. Lai, M. Zhu, H. Liang, Q. Zhou, R. Li, W. Zhang and H. Ye, Spectrochim. Acta, Part A, 2021, 244, 118845 CrossRef CAS PubMed.
- D. M. El-Husseini, D. M. A. Elmasry, E. M. Abo Hatab and S. Kassem, RSC Adv., 2024, 14, 27438–27448 Search PubMed.
- J. Yan, S. Liu, D. Sun, S. Peng, Y. Ming, A. Ostovan, Z. Song, J. You, J. Li and H. Fan, Sensors, 2024, 24, 7068 CrossRef CAS PubMed.
- T. Azargoshasb, R. Parvizi, F. Bozorgzadeh, H. A. Navida and H. Heidari, Nanoscale Adv., 2023, 5, 459–470 RSC.
- W. Zou, Y. Liu, R. Li and R. Guo, ACS Sustainable Chem. Eng., 2022, 10, 10057–10067 CrossRef CAS.
- X. Gao, H. Wei, W. Ma, W. Wu, W. Ji, J. Mao, P. Yu and L. Mao, Nat. Commun., 2024, 15, 7915 CrossRef CAS PubMed.
- X. Li, F. Shi, L. Wang, S. Zhang, L. Yan, X. Zhang and W. Sun, Molecules, 2023, 28, 6151 CrossRef CAS PubMed.
- T. Rattanaumpa, S. Maensiri and K. Ngamchuea, RSC Adv., 2022, 12, 18709–18721 RSC.
- H.-S. Jang, D. Kim, C. Lee, B. Yan, X. Qin and Y. Piao, Inorg. Chem. Commun., 2019, 105, 174–181 CrossRef CAS.
- S. Lakard, I.-A. Pavel and B. Lakard, Biosensors, 2021, 11, 179 Search PubMed.
- R. Sangubotla and J. Kim, Ceram. Int., 2023, 49, 16272–16282 Search PubMed.
- A. Foerster and N. A. Besley, J. Phys. Chem. A, 2022, 126, 2899–2908 CrossRef CAS PubMed.
- Y. Zou, X. Wang, Y. Li and Y. Cheng, Mater. Today, 2024, 79, 112–133 CrossRef CAS.
- R. Darabi, H. Karimi-Maleh, M. Akin, K. Arikan, Z. Zhang, R. Bayat, M. Bekmezci and F. Sen, Electrochim. Acta, 2023, 457, 142402 CrossRef CAS.
- A. S. Sharma and N. Y. Lee, Analyst, 2024, 149, 4095–4115 Search PubMed.
- L. Sun, F. J. Warren and M. J. Gidley, Trends Food Sci. Technol., 2019, 91, 262–273 CrossRef CAS.
- A. K. Verma, A. Noumani, A. K. Yadav and P. R. Solanki, Diagnostics, 2023, 13, 1375 Search PubMed.
- C. Ma, F. Zeng, L. Huang and S. Wu, J. Phys. Chem. B, 2011, 115, 874–882 CrossRef CAS PubMed.
- A. P. de Silva, T. S. Moody and G. D. Wright, Analyst, 2009, 134, 2385–2393 RSC.
- H. Niu, J. Liu, H. M. O’Connor, T. Gunnlaugsson, T. D. James and H. Zhang, Chem. Soc. Rev., 2023, 52, 2322–2357 RSC.
- Y. Yang, T. Zou, Z. Wang, X. Xing, S. Peng, R. Zhao, X. Zhang and Y. Wang, Nanomaterials, 2019, 9, 738 CrossRef CAS PubMed.
- K. S. Keremane, M. G. Acharya, P. Naik, C. C. Malakar, K. Wang and B. Poudel, Chemosensors, 2025, 13, 174 Search PubMed.
- L. Wu, C. Huang, B. P. Emery, A. C. Sedgwick, S. D. Bull, X.-P. He, H. Tian, J. Yoon, J. L. Sessler and T. D. James, Chem. Soc. Rev., 2020, 49, 5110–5139 RSC.
- J. Wang, Q. Xiang, H. Dong and X. Zhang, Sens. Diagn., 2024, 3, 914–932 RSC.
- J. Korram, L. Dewangan, R. Nagwanshi, I. Karbhal, K. K. Ghosh and M. L. Satnami, New J. Chem., 2019, 43, 6874–6882 RSC.
- X. Sun, T. Xiang, L. Xie, Q. Ren, J. Chang, W. Jiang, Z. Jin, X. Yang, W. Ren and Y. Yu, Mater. Today Bio, 2025, 32, 101763 CrossRef CAS PubMed.
- H. Y. Tsai and W. R. Algar, ACS Nano, 2022, 16, 8150–8160 CrossRef CAS PubMed.
- H. Kim, J. Song, S. Kim, S. Lee, Y. Park, S. Lee, S. Lee and J. Kim, Biosensors, 2023, 13, 470 CrossRef CAS PubMed.
- J. Song, F. Wang, X. Yang, B. Ning, M. G. Harp, S. H. Culp, S. Hu, P. Huang, L. Nie, J. Chen and X. Chen, J. Am. Chem. Soc., 2016, 138, 7005–7015 CrossRef CAS PubMed.
- C. Tortolini, F. Tasca, M. A. Venneri, C. Marchese and R. Antiochia, Chemosensors, 2021, 9, 229 CrossRef CAS.
- R. Gao, M. S. Kodaimati and D. Yan, Chem. Soc. Rev., 2021, 50, 5564–5589 RSC.
- M. C. Dos Santos and N. Hildebrandt, TrAC, Trends Anal. Chem., 2016, 84, 1–15 Search PubMed.
- L. Zhou, Y. Fan, R. Wang, X. Li, L. Fan and F. Zhang, Angew. Chem., Int. Ed., 2018, 57, 12824–12829 CrossRef CAS PubMed.
- M. Adhikari, R. Houhou, J. Hniopek and T. Bocklitz, J. Exp. Theor. Anal., 2023, 1, 44–63 CrossRef.
- S. M. Levchenko, A. Pliss, X. Peng, P. N. Prasad and J. Qu, Light: Sci. Appl., 2021, 10, 224 CrossRef CAS PubMed.
- X. Zhang, Y. Guo, M. Liu and S. Zhang, RSC Adv., 2013, 3, 2846–2857 RSC.
- Y. Zang, J. Lei and H. Ju, Biosens. Bioelectron., 2017, 96, 8–16 CrossRef CAS PubMed.
- J. Qin, J. Li, H. Zeng, J. Tang and D. Tang, Analyst, 2023, 148, 2200–2213 RSC.
- Y. Xue, W. Dong, B. Wang and G. Jie, Analyst, 2023, 148, 4456–4462 RSC.
- G. Xiao, J. Li, B. Zhao and Z. Yue, Chemosensors, 2025, 13, 344 CrossRef CAS.
- S. R. Ankireddy and J. Kim, Int. J. Nanomed., 2015, 10, 121–128 CAS.
- S. Dadkhah, A. Mehdinia, A. Jabbari and A. Manbohi, Microchim. Acta, 2020, 187, 569 CrossRef CAS PubMed.
- G. Chellasamy, S. R. Ankireddy, K. N. Lee, S. Govindaraju and K. Yun, Mater. Today Bio, 2021, 12, 100168 CrossRef PubMed.
- J. Duhan and S. Obrai, Opt. Mater., 2023, 145, 114466 CrossRef CAS.
- S. Borah, K. J. Goswami, N. S. Sarma and D. Sarkar, ACS Appl. Bio Mater., 2026, 9, 1670–1681 CrossRef CAS PubMed.
- K. Głowacz, M. Drozd and P. Ciosek-Skibińska, Microchim. Acta, 2021, 188, 343 CrossRef PubMed.
- L. R. Panicker, F. Shamsheera, R. Narayan and Y. G. Kotagiri, ACS Appl. Nano Mater., 2023, 6, 20601–20611 CrossRef CAS.
- C. Zhao, Y. Jiao, J. Hua, J. Yang and Y. Yang, J. Fluoresc., 2018, 28, 269–276 CrossRef CAS PubMed.
- D. Bharathi, B. Siddlingeshwar, R. H. Krishna, V. Singh, N. Kottam, D. D. Divakar and A. A. Alkheraif, J. Fluoresc., 2018, 28, 573–579 CrossRef CAS PubMed.
- J. Raut, R. D. Sherpa, S. K. Jana, S. M. Mandal, S. Mandal, S. P. Hui and P. Sahoo, ACS Appl. Nano Mater., 2023, 6, 23611–23619 CrossRef CAS.
- J. Raut, R. D. Sherpa, S. K. Jana, S. M. Mandal, S. Mandal, S. P. Hui and P. Sahoo, ACS Appl. Nano Mater., 2024, 7, 23278–23287 CrossRef CAS.
- H. Liu, J. He, Y. Li, B. Li, J. Zhou, B. Wu, D. Li, L. Sun, D. Yu and Z. Cao, Microchim. Acta, 2025, 192, 794 CrossRef CAS PubMed.
- L. Wang, R. Wang, B. Yang, F. Zhang, J. Zhao, S. Wang, M. Dong, Y. Cheng and Y. Li, Microchim. Acta, 2025, 192, 719 CrossRef CAS PubMed.
- D. De, S. K. Jana, S. Mondal, S. Mandal and P. Sahoo, ACS Appl. Nano Mater., 2025, 8, 22038–22048 CrossRef CAS.
- R. Mondal, N. Bose, A. Pangas, M. Sarkar, S. F. Ahmed and N. Mukherjee, ACS Appl. Nano Mater., 2026, 9, 2486–2500 CrossRef CAS.
- L. C. Kimble, J. S. Twiddy, J. M. Berger, A. G. Forderhase, G. S. McCarty, J. Meitzen and L. A. Sombers, ACS Sens., 2023, 8, 4091–4100 CrossRef CAS PubMed.
- A. Padmapriya, P. Thiyagarajan, M. Devendiran, R. A. Kalaivani and A. M. Shanmugharaj, J. Electroanal. Chem., 2023, 943, 117609 CrossRef CAS.
- M. Shen and X. Kan, Electrochim. Acta, 2021, 367, 137433 CrossRef CAS.
- Z. U. Khan, L. U. Khan, H. F. Brito, M. Gidlund, O. L. Malta and P. Di Mascio, ACS Omega, 2023, 8, 34328–34353 CrossRef CAS PubMed.
- N. Chen, Y. He, Y. Su, X. Li, Q. Huang, H. Wang, X. Zhang, R. Tai and C. Fan, Biomaterials, 2012, 33, 1238–1244 CrossRef CAS PubMed.
- K. Das, R. Meena, U. S. Gaharwar, E. Priyadarshini, K. Rawat, R. Paulraj, Y. K. Mohanta, M. Saravanan and H. B. Bohidar, Oxid. Med. Cell. Longev., 2023, 7707452 CAS.
- L. Hu, H. Zhong and Z. He, Colloids Surf., B, 2021, 200, 111609 CrossRef CAS PubMed.
- B. Uprety and H. Abrahamse, Front. Chem., 2022, 10, 946574 CrossRef CAS PubMed.
- A. Neelam, J. Egypt Natl. Canc. Inst., 2025, 37, 73 CrossRef PubMed.
- G. Lin, T. Chen, Y. Pan, Z. Yang, L. Li, K. T. Yong, X. Wang, J. Wang, Y. Chen, W. Jiang, S. Weng, X. Huang, J. Kuang and G. Xu, Nanotheranostics, 2020, 4, 173–183 CrossRef PubMed.
- C. Campalani and J.-C. M. Monbaliu, Mater. Sci. Eng., R, 2025, 163, 100940 CrossRef.
- A. Kumar, D. Kumar, M. Saikia, N. Renuga Devi and A. Subramania, Mater. Adv., 2023, 4, 3951–3966 RSC.
- N. Le, M. Zhang and K. Kim, Int. J. Mol. Sci., 2022, 23, 10763 CrossRef CAS PubMed.
- S. Harris and K. Kim, Nanomaterials, 2024, 14, 1086 CrossRef CAS PubMed.
- M. U. Nisa, A. Yaqub, M. H. Khan, F. Yaseen, S. Jilani, H. Ajab, N. S. Shah and A. Al-Anazi, Sens. Actuators Rep., 2025, 9, 100292 CrossRef.
- Z. Tavakolian-Ardakani, O. Hosu, C. Cristea, M. Mazloum-Ardakani and G. Marrazza, Sensors, 2019, 19, 2037 CrossRef CAS PubMed.
- N. Chauhan, S. Soni, P. Agrawal, Y. P. S. Balhara and U. Jain, Process Biochem., 2020, 91, 241–259 CrossRef CAS.
- J. Xu, J. Tao, L. Su, J. Wang and T. Jiao, Materials, 2021, 14, 3987 CrossRef CAS PubMed.
- H. K. Choi, J. H. Choi and J. Yoon, Biosensors, 2023, 13, 892 CrossRef CAS PubMed.
- A. Stuber and N. Nakatsuka, ACS Nano, 2024, 18, 2552–2563 CrossRef CAS PubMed.
- X. Terzapulo, A. Kassenova, A. Loskutova and R. Bukasov, Sens. Bio-Sens. Res., 2025, 47, 100771 CrossRef.
- L. J. Mohammed and K. M. Omer, Chem. Pap., 2026, 1, 4616 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |