A plasmon-enhanced fluorescent nanoprobe for ultrasensitive and selective detection of nitroaniline derivatives†
Farhin
Akhtar‡
a,
Santanu
Dolai‡
b,
Sabyasachi
Pramanik
 *a and
Satyapriya
Bhandari
*a and
Satyapriya
Bhandari
 *c
*c
aDepartment of Engineering Sciences, Rajiv Gandhi Institute of Petroleum Technology Sivasagar Campus, Sivasagar, Assam-785697, India. E-mail: spramanik@rgipt.ac.in
bDepartment of Chemistry, Indian Institute of Technology Guwahati, Assam-781039, India
cDepartment of Chemistry, Kandi Raj College, Affiliated to University of Kalyani, West Bengal-742137, India. E-mail: satyapriya.bhandari@krc.edu.in
First published on 12th March 2026
Abstract
A water-soluble, plasmon-enhanced fluorescent nanoprobe is engineered through Zn2+-directed assembly of HQS-capped silver nanoparticles and zinc quinolate complexes. Plasmonic coupling amplifies emission, enabling selective ppb-level detection of 4-nitroaniline and 2,4-dinitroaniline via NH2 → Zn2+ coordination-induced static quenching, delivering excellent sensitivity in real water matrices with high selectivity, sensitivity, and environmental applicability.
Plasmon-enhanced fluorescence (PEF) represents a transformative approach in optical detection, enabling conventional fluorophores to function as ultra-bright probes for analyte sensing at extremely low concentrations.1–9 By exploiting localized surface plasmon resonance (LSPR) in noble-metal nanostructures, particularly silver and gold nanoparticles, PEF amplifies fluorescence through strong near-field electromagnetic enhancement and plasmon-mediated radiative processes.1–3 Fluorophores positioned within these nanoscale regions exhibit accelerated excitation rates, improved radiative decay, and reduced non-radiative losses, collectively yielding substantial gains in quantum yield, brightness, and photostability.1,3–9 Over the past decade, plasmonic coupling has advanced applications in biomedical imaging, single-molecule tracking, catalysis monitoring, and environmental sensing.2–7 Ongoing innovations in nanoparticle design, functionalization, and distance control are extending PEF toward robust, deployable sensing architectures.
Environmental monitoring is a critical domain where advanced optical probes are urgently required, particularly for detecting persistent organic pollutants such as nitroaromatic amines.5,7,10–14 Among these, 2,4-dinitroaniline (2,4-DNA) and 4-nitroaniline (4-NA) are especially concerning due to their extensive use in dye synthesis, agricultural formulations, polymer intermediates, and energetic material precursors.10–17 Their strong electron-withdrawing nitro groups confer remarkable stability, allowing them to persist in natural waters, soils, and industrial effluents without rapid degradation.10–12 This persistence, coupled with documented toxicity – including oxidative stress, endocrine disruption, cytotoxicity, and methemoglobin formation – poses serious risks to ecosystems and human health.11,12 Conventional analytical methods such as GC–MS, LC–MS, ion mobility spectrometry, and electrochemical detection provide excellent accuracy but are limited by high costs, complex sample preparation, and poor portability.14–16 Consequently, fluorescence-based sensing has emerged as a rapid, inexpensive, and field-friendly alternative. Recent advances include carbon dots, covalent and metal–organic frameworks, luminescent complexes, and semiconductor nanocomposites, achieving ppb-to-nM detection of nitroanilines.10–16 Despite notable sensitivity, many systems suffer from multistep fabrication, limited water dispersibility, or modest photostability.10–16 These limitations underscore the urgent need for a simple, water-soluble, and stable plasmon-enhanced fluorescence nanoprobes capable of reliable nitroaniline discrimination.
Herein, we report a water-soluble plasmon-enhanced fluorescence nanoprobe (PEFN) constructed via Zn2+-mediated assembly of HQS-stabilized silver nanoparticles (Ag NPs, 20 nm) and an in situ zinc quinolate complex (ZQC). HQS sulfonic groups ensure strong stabilization and controlled surface arrangement, while Zn2+ coordination generates a bright green-emissive ZQC within the plasmonic near-field. This hybrid system exhibits enhanced fluorescence, a quantum yield of 30%, and an extended lifetime of 6.4 ns compared to ZQC alone (2.9 ns), with excellent colloidal stability and photostability. The PEFN shows high selectivity toward 2,4-DNA and 4-NA through NH2 → Zn2+ coordination and ground-state complexation, achieving detection limits of 1.5 and 1.4 ppb respectively. Robust recoveries in tap and river water establish the PEFN as a stable, cost-effective sensing platform.
Scheme 1 depicts the Zn2+-mediated assembly of HQS-stabilized Ag NPs into a zinc quinolate complex (ZQC), where sulfonate groups stabilize Ag NPs and Zn2+ coordinates at pyridine and phenolic sites. Detection of 4-NA and 2,4-DNA occurs via NH2 → Zn2+ coordination, ground-state complexation, and static fluorescence quenching. Ag NPs were synthesized using reported methods, and detailed preparation of PEFNs with their fluorescence-based sensing applications is provided in the SI.
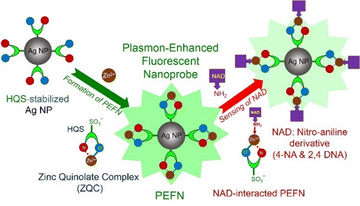 | ||
| Scheme 1 Schematic illustration of the formation of a PEFN and the proposed sensing mechanism toward 4-NA and 2,4-DNA. | ||
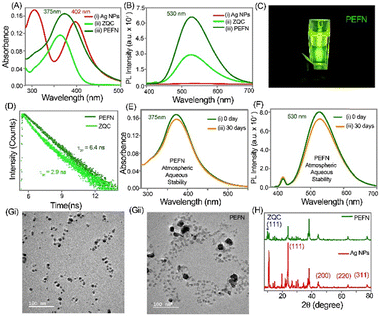
The UV-Vis spectrum of HQS-stabilized Ag NPs (Fig. 1A) exhibits a distinct localized surface plasmon resonance (LSPR) band at 402 nm, confirming successful nanosilver formation. Upon assembly into the plasmon-enhanced fluorescence nanoprobe (PEFN), an additional absorption band emerges at 375 nm, attributed to the π–π* transition of the quinolate unit, thereby validating effective incorporation of the ZQC.18–20 Under 365 nm excitation, the PEFN displays intense green fluorescence with a maximum at 530 nm (Fig. 1B), originating from the HOMO–LUMO transition of the ZQC.19,20 The emission is amplified 2.5-fold relative to the ZQC alone, consistent with the plasmon-mediated enhancement of excitation and radiative processes. Photographic images under UV illumination (Fig. 1C) visually confirm the increased brightness of the hybrid system. Time-resolved fluorescence (Fig. 1D and Table S1, SI) reveals an average lifetime of 6.4 ns, significantly longer than that of pristine ZQC (2.9 ns), supporting plasmon-assisted radiative decay enhancement. Fluorescence and absorption titrations (Fig. S1, SI) identified optimal Zn2+ concentrations for efficient coordination-driven assembly. Long-term optical stability in the aqueous phase was demonstrated by spectra that remained unchanged over 30 days (Fig. 1E and F), confirming colloidal robustness. The PEFN achieved an absolute photoluminescence quantum yield (PLQY) of 30%, markedly higher than that of pristine ZQC (13%), and retained over 95% of its emission intensity after 30 minutes of continuous UV irradiation (Fig. S2, SI), indicating excellent photostability. TEM analysis (Fig. 1G and Fig. S3, SI) showed uniformly dispersed spherical Ag NPs (20.0 ± 1.0 nm), while Zn2+ coordination induced ordered assembly characteristic of the PEFN. HRTEM (Fig. S4, SI) revealed narrow size distributions and lattice fringes of the Ag (111) plane, confirming crystallinity. XRD patterns (Fig. 1H) verified the FCC structure of Ag NPs, with additional ZQC (111) peaks confirming nanocomposite formation.21–23 Benesi–Hildebrand analysis yielded a binding constant of 1.97 × 105 M−1 (Fig. S5, SI), indicating strong Zn2+ coordination with HQS-functionalized Ag NPs. FTIR spectra (Fig. S6 and S7, SI) showed shifts in sulfonate stretching bands and the emergence of Ag–O and Zn–N/O features, confirming Zn–HQS coordination.19–24 Zeta-potential changes (Fig. S8 and Table S2, SI) further validated systematic surface-charge modulation. Collectively, HQS acts as both a stabilizer and a coordination linker, guiding the integration of plasmonic Ag NPs with luminescent ZQC domains into a structurally robust PEFN architecture.
The sensing performance of the PEFN was systematically evaluated using 2,4-DNA and 4-NA as representative analytes. Incremental addition of either compound produced a progressive decrease in the emission band at 530 nm, confirming efficient fluorescence quenching (Fig. 2A). The calculated detection limits were 1.5 ppb for 2,4-DNA and 1.4 ppb for 4-NA (Fig. 2B and Table S3, SI), demonstrating sub-ppm sensitivity.10–17 Notably, the lower LOD for 4-NA indicates stronger interaction with the PEFN compared to 2,4-DNA. Stern–Volmer (SV) analysis followed the classical relation I0/I = 1 + Ksv[Q]), yielding linear plots across the tested linear ranges of 0.2–1.0 µM (Fig. 2C).23–25 The extracted quenching constants were Ksv (2,4-DNA) = 1.6 × 106 M−1 and Ksv (4-NA) = 1.7 × 106 M−1, confirming strong affinity. A time-dependent fluorescence study showed that the PEFN reached ∼90% of the maximum signal change shortly after analyte addition, indicating rapid sensing activity with a response time of 60–70 s (Fig. S9, SI). Selectivity studies revealed that only 2,4-DNA and 4-NA induced significant quenching, while related nitroaromatics, common amines, and metal ions produced negligible responses (Fig. 2D). Digital photographs under UV illumination corroborated the selective quenching (Fig. 2E). Compared with earlier reported probes (Table S4, SI), the PEFN exhibited superior detection limits, selectivity, and aqueous stability.10–17 As shown in Table S5, the sensor achieved recovery values in the range of 95.83–108.97% with relative standard deviations below 5% (n = 3), demonstrating excellent analytical accuracy, reproducibility, and tolerance to complex sample matrices. These results confirm that the PEFN maintains strong sensing performance under environmentally relevant conditions, underscoring its suitability for real-world monitoring of nitroaniline contaminants.
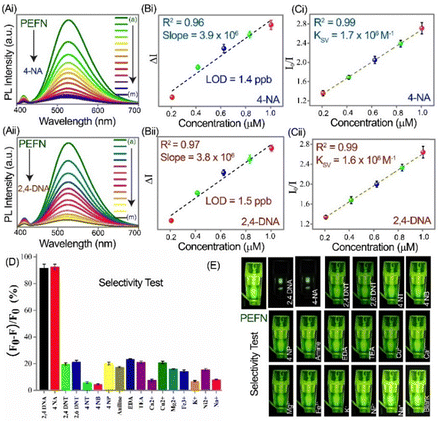 | ||
| Fig. 2 Fluorometric detection, selectivity and interference studies of nitroaromatic analytes using the PEFN. (A) Representative fluorescence emission spectra of the PEFN (λex = 365 nm) upon incremental addition of (a) 0.000; (b) 0.208; (c) 0.416; (d) 0.625; (e) 0.833; (f) 1.004; (g) 1.458; (h) 1.875; (i) 2.708; (j) 3.541; (k) 4.375; (l) 5.208; and (m) 6.041 µM concentrations of (i) 4-NA and (ii) 2,4-DNA. (B) Calibration plots showing the linear relationship between fluorescence intensity (ΔI) and analyte concentration, enabling the determination of the sub-ppm detection limits of (i) 4-NA and (ii) 2,4-DNA. (C) Stern–Volmer plots showing the linear relationship between fluorescence intensity (I0/I) and analyte concentration for (i) 4-NA and (ii) 2,4-DNA. Error bars represent 3 independent measurements. (D) Bar diagram showing the fluorescence response of the PEFN toward target analytes (2,4-DNA and 4-NA) versus structurally similar nitroaromatics (2,4-DNT, 2,6-DNT, 4-NT, 4-NB and 4-NP), common amines (aniline, EDA and TEA), and metal ions (Cu2+, Ca2+, Mg2+, Fe3+, K+, Ni2+ and Na+). Error bars represent 3 independent measurements. (E) Corresponding fluorescence photographs under 365 nm excitation, confirming high selectivity and minimal cross-reactivity. A concentration of 15 µM (2.5-fold higher than the analyte concentration) of each interfering substance was used in Fig. 2D and E. | ||
The interaction mechanism between the PEFN and nitroaromatic amines is governed by ground-state complex formation. Both 4-NA and 2,4-DNA contain electron-rich –NH2 groups that donate lone-pair electrons to Zn2+ centers embedded within the HQS-stabilized Ag NP framework, forming stable NH2 → Zn2+ complexes.26–30 This association perturbs the local electronic structure of the fluorophore, resulting in pronounced emission quenching (Scheme 1). Zeta-potential analysis (Fig. S8, S10 and Table S2, SI) supports complexation: the weakly positive surface charge of the PEFN (+0.29 mV) shifts to negative values upon analyte addition, consistent with the adsorption of nitro-rich molecules.27–30 FTIR spectra (Fig. S11, SI) reveal downshifts and broadening of NH2 stretching bands (3300–3500 cm−1), confirming electron donation to Zn2+. Binding constant analysis shows the stronger affinity of 4-NA (2.6 × 106 M−1) compared to 2,4-DNA (1.5 × 106 M−1) (Fig. S12, SI), explaining its lower LOD (Table S3, SI). The reduced electron-donating ability of 2,4-DNA, due to two –NO2 substituents, diminishes NH2 → Zn2+ coordination efficiency, consistent with higher LOD values. Fluorescence lifetime studies (Fig. S13 and Table S6, SI) show unchanged fluorescence lifetime decay of PEFN with 4-NA or 2,4-DNA, confirming static quenching via NH2 → Zn2+ coordination. This evidences ground-state complex formation between the PEFN and nitro-analytes.26,30 The observed absorption shift upon interaction with nitroaniline indicates perturbation of the Zn-quinolate electronic structure due to changes in the Zn2+ coordination environment, likely via –NH2 interaction (Fig. S14, SI). UV-Vis, FTIR, TRPL, and fluorescence results support stable ground state complex formation and static quenching rather than dynamic quenching. The fluorescence response of the PEFN (originally synthesized at pH 6.8) to nitroaniline remained unchanged across the pH range of 5.7–7.5, even at the highest concentration within the linear range (Fig. S15, SI), confirming pH-independent sensing behavior. Although HQS strongly chelates Zn2+, the PEFN retains partially accessible Zn2+ sites that allow –NH2 groups of nitroaniline to coordinate, facilitated by –NO2 polarization, with stronger binding observed for 4-NA than for 2,4-DNA. Overall, PEFN sensing occurs via NH2 → Zn2+ coordination, ground-state complex formation, and static quenching, with 4-NA binding enhancing sensitivity and selectivity.
In summary, we report a highly efficient PEFN constructed through Zn2+-mediated assembly of HQS-stabilized Ag NPs, producing a bright green-emissive ZQC tailored for nitroaniline detection. The strong LSPR of Ag NPs amplifies ZQC fluorescence, yielding enhanced intensity, quantum yield, and lifetime, with excellent aqueous stability and photostability. The PEFN achieves ppb-level detection with low LODs and strong linearity, while resisting interference from related nitroaromatics, amines, and metal ions. Mechanistic studies confirm NH2 → Zn2+ coordination and ground-state complexation as the basis of static quenching. High recoveries and low RSDs in tap and river water validate robustness, establishing this cost-effective, stable platform for environmental and forensic monitoring.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental procedures, characterization, and sensing studies; Fig. S1–S15 and Tables S1–S6. See DOI: https://doi.org/10.1039/d5cc07258a.Acknowledgements
S. P. gratefully acknowledges funding from the Rajiv Gandhi Institute of Petroleum Technology Seed Grant (P-2123). We thank the Department of Chemistry and the Center for Nanotechnology at IIT Guwahati, Dr Mihir Manna, and Kandi Raj College for technical support. Special thanks to Prof. Arun Chattopadhyay (IIT Guwahati) for guidance and laboratory access.References
- J. F. Li, C. Y. Li and R. F. Aroca, Chem. Soc. Rev., 2017, 46, 3962–3979 RSC
.
- M. Wang, M. Wang, G. Zheng, Z. Dai and Y. Ma, Nanoscale Adv., 2021, 3, 2448–2465 RSC
.
- S. H. Cho, S. Choi, J. M. Suh and H. W. Jang, J. Mater. Chem. C, 2025, 13, 6484–6507 RSC
.
- N. Hildebrandt, M. Lim, N. Kim, D. Y. Choi and J.-M. Nam, Chem. Commun., 2023, 59, 2352 RSC
.
- K. Chung, S. Yu and L. P. Lee, Chem. Soc. Rev., 2024, 53, 10491–10522 RSC
.
- S. H. Oh and H. Altug, Nat. Commun., 2018, 9, 5263 CrossRef
.
- M. Li, S. K. Cushing and N. Wu, Analyst, 2015, 140, 386–406 RSC
.
- K. Jo, E. Marino, J. Lynch, Z. Jiang, N. Gogotsi, T. P. Darlington, M. Soroush, P. J. Schuck, N. J. Borys, C. B. Murray and D. Jariwala, Nat. Commun., 2023, 14, 2649 CrossRef
.
- Y. Fu, J. Zhang and J. R. Lakowicz, Chem. Commun., 2012, 48, 9726–9728 RSC
.
- P. Das, G. Chakraborty and S. K. Mandal, ACS Appl. Mater. Interfaces, 2020, 12, 10224–10232 CrossRef
.
- R. Zheng, Y. Zhang, G. Bao, P. Wu, W. Wei, X. Yuan, T. Zou, T. Zhang and J. Wang, ACS Sustainable Chem. Eng., 2023, 11, 9077–9086 CrossRef
.
- S. Li, Q. Zhou, Z. Li, M. Liu, Y. Li and C. Chen, Spectrochim. Acta, Part A, 2023, 286, 122040 CrossRef
.
- A. Chanda and S. K. Mandal, ACS Appl. Nano Mater., 2025, 8, 12784–12798 CrossRef
.
- T. S. Priya, R. Sasikumar, T. W. Chen, S. M. Chen, B. Kim and T. Kokulnathan, Appl. Clay Sci., 2023, 244, 107103 CrossRef
.
- B. Hu, X.-T. Li, Y. Chen and W.-Y. Xie, Chemosphere, 2010, 81, 430–435 CrossRef PubMed
.
- J. S. Chiang and S.-D. Huang, Talanta, 2008, 75, 70 CrossRef
.
- H. Xiao and Z. Zhang, Anal. Methods, 2023, 15, 3940–3947 RSC
.
- S. D. Solomon, M. Bahadory, A. V. Jeyarajasingam, S. A. Rutkowsky, C. Boritz and L. Mulfinger, J. Chem. Educ., 2007, 84, 322 CrossRef
.
- S. Singha, M. Paul, P. Ghosh, M. Manna, S. Pramanik, A. Misra and S. Bhandari, J. Phys. Chem. Lett., 2025, 16, 6066 CrossRef PubMed
.
- S. Bhandari, S. Roy, S. Pramanik and A. Chattopadhyay, Langmuir, 2019, 35, 14399 CrossRef PubMed
.
- Y. Liu, C.-H. Liu, T. Debnath, Y. Wang, D. Pohl, L. V. Besteiro, D. M. Meira, S. Huang, F. Yang, B. Rellinghaus, M. Chaker, D. F. Perepichka and D. Ma, Nat. Commun., 2023, 14, 541 CrossRef PubMed
.
- P. Maiti, S. Sarkar, T. Singha, S. D. Roy, M. Mahato, P. Karmakar, S. Paul and P. K. Paul, Langmuir, 2023, 39, 6713 Search PubMed
.
- A. Manivel and S. Anandan, Colloids Surf., A, 2012, 395, 38 Search PubMed
.
- B. Sureshkumar, Y. S. Mary, C. Y. Panicker, K. S. Resmi, S. Armaković, S. J. Armaković, C. Van Alsenoy and S. Suma, J. Mol. Struct., 2017, 1150, 540–552 CrossRef
.
- R. Siegel and S. Glazier, J. Chem. Educ., 2021, 98, 2643–2648 CrossRef
.
- A. Sharma, D. Kim, J.-H. Park, S. Rakshit, J. Seong, G. H. Jeong, O.-H. Kwon and M. S. Lah, Commun. Chem., 2019, 2, 39 CrossRef
.
- F. A. Alharthi, H. K. Aldubeikl, H. S. Alanazi, W. S. Al-Nafaei and I. Hasan, Nanomaterials, 2023, 13, 362 CrossRef PubMed
.
- L. Li, J.-Y. Zou and S.-Y. You, Inorg. Chim. Acta, 2020, 509, 119703 CrossRef
.
- P. R. Reddy, M. Radhika and P. Manjula, J. Chem. Sci., 2005, 117, 239–246 CrossRef
.
- L. Cai, Z. Zhang, H. Xiao, S. Chen and J. Fu, RSC Adv., 2019, 9, 41383–41391 Search PubMed
.
Footnotes |
| † Dedicated to Prof. Chattopadhyay on his 61st birthday. |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |