 Open Access Article
Open Access ArticleNonspherical polymer nano/micro particles: a guide to shape engineering
Jeremiah
James
ab,
Emma
Leung
b and
Rong
Yang
 *a
*a
aRobert Frederick Smith School of Chemical and Biomolecular Engineering, Cornell University, Ithaca, NY 14853, USA. E-mail: ryang@cornell.edu
bNancy E. and Peter C. Meinig School of Biomedical Engineering, Cornell University, Ithaca, NY 14853, USA
First published on 20th March 2026
Abstract
Polymer micro- and nanoparticles underpin applications from drug delivery to building materials, yet virtually all commercial grades of polymer particles are spherical, forfeiting the performance gains unlocked by shape engineering. In drug delivery, for instance, shape-engineered polymer particles promise longer circulation, targeted tissue penetration, programmable assembly, and tailored photonic responses, positioning geometry as an under-exploited design axis. Although three decades of lab-scale research have produced dozens of protocols for producing shaped polymer particles, a mechanism-centric roadmap that links fabrication route to attainable morphology has been missing. This review closes that gap by surveying more than 70 documented geometries and the physicochemical principles that create them. In brief, starting from the spherical polymer particles, mechanical deformation, such as film stretching, compression, and shear, reshapes spheres into rods, ellipsoids, discs, and polyhedra using low-cost tensile/compressive bench-top rigs. Increased complexity in particle shapes can be obtained via lithographic and template-molding platforms, yielding cuboids, prisms, rings, and sub-100 nm flakes with dimensional fidelity. By expanding upon the polymerization mechanism beyond photopolymerization, seeded emulsion polymerization exploits polymerization-induced phase separation to generate lobed, Janus, porous, and indented architectures while retaining a reasonable production throughput. Polymerization under shear, such as masked photopolymerization under continuous microfluidic confinement, cast plugs, prisms, high-curvature “hamburger” or “olive” motifs, and other extruded forms. Emerging template-free and confinement-free techniques, such as polymer plasticization, condensed-droplet polymerization, and electrospraying, expand the landscape to asymmetric shapes, leveraging interfacial tensions, condensation kinetics, and electric fields to sculpt morphology. Looking ahead, green chemistries and scalable process design constitutes critical steps that will unlock the societal impact of this emerging class of materials. This review aims to organize those disparate methods based on their fundamental mechanisms, thereby accelerating the design, synthesis, and production of shape-engineered soft materials, pointing to novel solutions in healthcare, consumer goods, structural materials, and active matter and robotics.
1. Introduction
Virtually all commercially available polymer micro- and nanoparticles share a single geometry: sphere. This ubiquity is a direct consequence of the synthesis and production technology. The modern polymer particle industry traces its roots to World War II, when the U.S. government launched a synthetic rubber initiative to replace natural rubber imports cut off from Southeast Asia.1 The effort culminated in the industrial-scale adoption of emulsion polymerization, a process that rests on the fundamental principle of polymerization in monomer-swollen micelles.2,3 In a typical process, hydrophobic monomers are dispersed within an aqueous medium, using surfactants to form stable micellar droplets, which are then polymerized by radical initiators into solid particles.4 Because the micelles adopt a spherical shape to minimize their interfacial energy in water, this symmetry is preserved as polymerization locks the morphology in place.5 The dominance of emulsion polymerization in the particle manufacturing industry thus entrenched the spherical form as the industry standard, inadvertently constraining access to the performance gains that shape engineering could unlock.6The ability to deliberately engineer particle shape expands the design space of polymer-based micro- and nanostructures, unlocking functions inaccessible to conventional spherical particles. The opportunities afforded by shape-engineered polymer particles are most evident in drug delivery, where decades of research have optimized materials and formulations but seldom geometry until recently. Since their emergence in the mid-to-late twentieth century, liposomes and polymer-based delivery vehicles have served as foundations for modern nanomedicine by modulating the bioavailability of therapeutics, delaying their clearance, and reducing off-target toxicity.7–9 Research over the last decades has further expanded the material palette to metals (for electromagnetic or catalytic properties), dendrimers (to enable large payloads), carbon nanomaterials, and combinations of those constituents that give rise to hybrid materials.10 Among these, polymer-based delivery vehicles stand apart for their tunability in three critical design axes: chemistry, size, and shape. Through monomer and polymer selection, the chemistry is tuned to regulate release kinetics and responsiveness to biological or environmental stimuli.11–13 The sizable library of biocompatible polymers, ranging from natural biopolymers and polysaccharides to synthetic biodegradable amides or esters, and non-biodegradable siloxanes or acrylics, has created a rich chemical toolbox.14 Similarly, size engineering has been extensively optimized to balance circulation time, tissue penetration, and biological interactions for targeted delivery and/or pharmacokinetics.15–18 Taken together, these advances have propelled polymer particles toward clinical translation: almost 40 nanoparticle formulations have been clinically approved with more on the way, such as docetaxel-loaded polymeric particles (CPC634) that have entered clinical testing.15 Despite decades of progress in chemistry and size control, nearly all clinically approved nanomedicine to date remains spherical or near-spherical. As such, shape engineering becomes a powerful yet underutilized axis in the design of drug delivery vehicles.16 Emerging evidence underscores that shape-engineered polymer particles can outperform their spherical counterparts across multiple therapeutic benchmarks. Low-curvature geometries prolong circulation by evading macrophage-mediated clearance pathways, while elongated or anisotropic particles improve margination, tumor penetration, and endothelial adhesion.17,18 Such findings reveal geometry as a potentially synergistic design variable alongside chemistry and size, which critically governs the flow, jam, and stimuli-responsive behaviors of polymer particles in biological systems.
Despite the growing recognition of geometry as a design variable, current synthesis strategies for nonspherical polymer particles remain confined largely to the laboratory scale. Few existing techniques have achieved the throughput, reproducibility, or process economy required for industrial adoption. The barriers are rooted in complex multistep procedures, narrow parameter windows, and limited scalability. This review surveys the current landscape of shape-engineering methods, linking attainable morphologies to their governing physicochemical mechanisms. Beyond the emulsion polymerization routes that established the dominance of spherical particles, emerging fabrication strategies have diversified the particle-shape repertoire.19–21 Lithographic and template-molding platforms offer high precision in the xy-plane, producing nano- to microscale cuboids, prisms, and rings through patterned photomasks, molds, or interference fields, albeit with throughput constraints inherent to planar processing. Seeded emulsion polymerization exploits polymerization-induced phase separation to access lobed, Janus, hollow, and dimpled architectures within industry-familiar emulsification systems. Mechanical deformation approaches, such as film stretching, compression, or shear, reshape spherical feedstock into rods, ellipsoids, and polyhedra using low-cost tools, though shape uniformity can vary. Microfluidic photopolymerization achieves high-fidelity confinement-defined geometries, generating monodisperse plugs, discs, and prisms, yet remains limited by device throughput. Finally, template-free strategies such as solvent-vapor annealing, vapor-phase polymerization, condensed-droplet polymerization (CDP), and electrospraying leverage interfacial tension, condensation dynamics, and electric fields to create shapes unattainable through conventional routes. Despite the rapid development in the past three decades, the guiding principles that connect the fabrication routes to their resulting geometries remain elusive. This review aims to close that gap by cataloguing over seventy experimentally realized geometries and mapping each to its fundamental mechanism. We hope to empower researchers to quickly gain access to their targeted particle shapes through the detailed description of the mechanism, instrumentation, and processes involved in each methodology that can be found in this review. In doing so, we aim to open up the polymer shape engineering toolbox and accelerate translation of some of the methodologies into industrial practice, driven by future use-inspired developments. To help readers compare the major synthesis methods on equal footing, Table 1 consolidates the key practical considerations for each approach. Specifically, it summarizes (i) the operative mechanism that generates anisotropy, (ii) the polymerization/solidification mode, and (iii) the dominant polymer/material classes reported, alongside size ranges, throughput/scale, biomedical relevance, and method-specific advantages and limitations.
| Field | 2.1 Lithography | 2.2 Seeded emulsion polymerization | 2.3 Mechanical deformation | 2.4 Microfluidic | 2.5 Misc |
|---|---|---|---|---|---|
| Summary | Anisotropy is set by a mask/interference pattern or mold cavity UV/thermal curing (or solidification) locks geometry, followed by release/harvest of freestanding particles or structures.22–39 | Anisotropy emerges during seed swelling and polymerization-induced phase separation/protrusion growth, with morphology controlled by crosslink density, monomer-seed ratio, and kinetics.40–80 | Pre-formed particles are softened (heat above Tg or use solvent/plasticizer) and mechanically deformed (stretching, hot-press, squeeze flow) to lock anisotropy the deformation step itself does not require polymerization.81–96 | Droplet or laminar-flow templates formed in microchannels are solidified (typically by UV FRP) confinement, flow, and multiphase droplet physics set nonsphericity and compartmentalization.97–116 | Anisotropy arises without lithographic molds: interface-/film-based reshaping by thermal/solvent/vapor annealing vapor CDP (condense monomer droplets → FRP) electrospray droplet drying/collapse ionotropic gelation and biomimetic templating.117–132 |
| Polymerization | Often UV-initiated free-radical photopolymerization/photocrosslinking (hydrogels, photoresists, acrylates), with some thermal FRP (e.g., triacrylate arrays) in micromolding of pre-formed polymers, the shaping step itself does not require polymerization.22–39 | Predominantly FRP (persulfate/azo/thermal or radiation-initiated) in seeded emulsion/dispersion workflows some systems add post-polymerization chemistry (e.g., sacrificial phases).40–80 | Deformation step does not require polymerization precursor particles are commonly made by emulsion solvent evaporation (e.g., PLGA/PCL) or by polymerization when using PS/PMMA/PDVB model systems.81–96 | Predominantly UV-initiated FRP of acrylate/methacrylate monomers in droplets or laminar streams some platforms include sol–gel/surface chemistry steps for wettability patterning.97–116 | Mixed: reshaping/annealing steps have no polymerization CDP uses TBPO-initiated FRP electrospray uses pre-formed polymers alginate uses ionic crosslinking (Ca2+) RBC-mimic shells are crosslinked chemically (e.g., glutaraldehyde).117–132 |
| Polymers | PEG-based hydrogels23,26,30,38 | PS systems43–52,61–65,72–77 | PLGA81,82,85,86,94 | Polyacrylate.97,100,103–105,109,112,115,116 | PS117–123 |
| PMMA-based44,49,54,57,78 | |||||
| Epoxy photoresists24,25,32 | Methacrylate/epoxy systems48,58,59,75 | PS84,89,91,93,94,96 | PEGDA.110,111,113–116 | Methacrylates124–127 | |
| Thermoplastics and biodegradable polyesters31,34 | Bio-derived composites42 | PLA87,94 | PETPTA.107 | PLGA129,130 | |
| Acrylate-based39 | TPM-based70,71 | PDVB95 | Alginate.131 | ||
| TPM-based70,71 | PIBCA92 | Protein/polyelectrolyte.132 | |||
| Polymer-inorganics47,66,68,69 | Polymer blends90 | ||||
| Sizes | 0–1 µm24–26,28,30,32,38 | 0–1 µm41,42,46–52,55–57,59,64–66,68,69,71–76,79,80 | 0–1 µm81,83–86,89–94,96 | 0–1 µm (not reported) | 0–1 µm122,124,125 |
| 1–10 µm.22,23,31,34–37 | 1–10 µm41,43–46,48,50–55,58–63,65,69–73,76–80 | 1–10 µm81–85,87–91,93,94 | 1–10 µm97,103,116 | 1–10 µm117,119–123,129,130 | |
| >10 µm.22,23,29,30,34–37,39 | >10 µm43,46,48,55,56,59 | >10 µm82,89,94,95 | >10 µm.97,100–115 | >10 µm.119,120,126,127,131 | |
| Shapes | Mask-/mold-defined planar and extruded geometries (e.g., polygons, rods, boomerrang)22,23,29,30,32,37,38 | Polygons/polyhedra40,42,61,63,65 | Polygons/polyhedra89,96 | Disks/rods/ellipsoids97,100 | Lenses/acorns/discs/bowls/peanuts and other concave morphologies117–123 |
| Cone31 | Rods40,61 | Rods/cylinders82,86,89,93 | Janus/shells/crescents101,102,104,108,109,112 | Domes/plates124–127 | |
| Flakes39 | Disks/discoids47,63,73 | Disks81,85,89–94 | Mask-defined extruded 2D shapes106,110,112–116 | Rods129,130 | |
| Ellipsoids/spheroids45,48,69 | Ellipsoids81–84,86–93 | Tadpole/tear shapes131 | |||
| Locks/keys70,71 | Lobed/clusters84 | RBC-mimic132 | |||
| Lobed/clusters40,42–51,54,57,61–63,68,70 | Concave/dimpled/bowls84,89,90 | ||||
| Concave/dimpled/bowls/lens-like morphologies41,50,52,59,65,66,73–76 | Capsules/shells95 | ||||
| Capsules/shells41,66,68,78 | Lens-like89 | ||||
| Janus46–48,57–59,64,68,72,76 | Jellyfish-like81,89 | ||||
| Limitations | Release-layer challenges/particle loss22 | Incomplete products.41,50 | Heating above Tg or solvent/plasticizer softening and film stretching limitations81,90 | Clogging/sticking97,103,105 | Challenging isolation117 |
| Supercritical CO2 drying25 | Time-intensive and secondary particles40,46,59 | IIrregular shapes83 | Initiator concentration limitations.100,103,105 | Hours-long exposures.119,123 | |
| Flakes can stick/break on PDMS release39 | Require specific stabilizer/surfactant and initiator choices44,50 | Particle fusions94 | Requires oxygen-inhibition edge effects.110 | Incomplete polymerization124–126 | |
| PFPE/PDMS mold cost/swelling.27 | Size-limited.57 | Rupture.81,88 | High-temperature curing.102 | Viscosity constraints129,130 | |
| Bio-Relevance | Protein photolithography reports biocompatibility and proteolytic degradability over weeks.22 | Bio-derived starch nanoparticles were used as non-cross-linked seeds/stabilizers in a soap-free styrene system.42 | Biodegradable PLGA/PCL/PDLLA systems.81,87,90,94 | PEGDA-based particles are presented as suitable for bioassays (graphical encoding and covalent incorporation of biomolecular probes protein conjugation).106,108,110,114 | Alginate particles are explicitly positioned as biocompatible/biodegradable and were used for cell encapsulation.131 |
| PRINT nanogels report low cytotoxicity in HeLa cells and were evaluated in vivo after IV injection30 | pH-responsive or reactive functionalization is explicitly reported in some systems.59,68 | Artemether-loaded PLGA nanorods reported ≈90% THP-1 viability across 0.001–100 µg mL−1 and <10% haemolysis nanorods showed less haemolysis than nanospheres.86 | sRBC platforms explicitly aim to mimic RBC deformability and use biocompatible/biodegradable PLGA templates limitations on PS biocompatibility are stated.132 | ||
| S-FIL PEG particles include enzyme-degradable peptide-crosslinked variants (Cathepsin B trigger)38 | PLGA discs released encapsulated dye faster than microspheres in PBS (0.05% Triton X-100).94 | ||||
| “pelmeni” capsules combine polyelectrolyte multilayers with a biodegradable PLGA lid.31 | |||||
| Advantages | Compared to conventional polydisperse/spherical carriers, lithographic/templated approaches emphasize deterministic shape and monodispersity (e.g., PDI ∼1 sharp edges/sidewalls).22,30 | Seeded routes are repeatedly framed as maintaining monodispersity while enabling nonspherical/compartmentalized morphologies.40,55 | Deformation platforms are described as scalable/reproducible and enable high-aspect-ratio nonspherical particles from common spherical precursors.81,89,90 | Microfluidic platforms emphasize narrow size distributions and continuous production (e.g., 100–500 particles/s 5.6 × 10^6 particles per h).100,110 | Several approaches emphasize access to nonspherical geometries without the use of molds or masks.119,124,126 |
| PRINT nanogels were noted to maintain size/PDI at 37 °C versus a liposome comparator and to reduce PK confounding from polydispersity.30 | One-pot nonspherical hollow latexes were reported to improve hiding/opacity versus nonspherical solid particles.41 | Elongated PLGA discs showed higher adhesion than spheres.85 | CDP is highlighted as rapid (seconds–minutes), solvent/template-free, and widely tunable in size.124–127 | ||
| S-FIL release via PVA dissolution was highlighted as a gentler alternative to scraping/shear harvesting workflows.38 | Activated swelling protocols were described as producing nonspherical particles with excellent monodispersity and reproducibility.59 | Artemether-loaded PLGA nanorods reduced haemolysis relative to nanospheres.86 | |||
| Throughput | ∼10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 particles in minutes (100 × 100 array)22 000 particles in minutes (100 × 100 array)22 |
∼1010 particles per mL61 | ∼107–108 particles per hour82 | 12–36 particles per min98 | CDP polymerization step is very fast (seconds; <45 s noted) and overall runs are minutes-scale124,125 |
| ∼109 particles per exposure (6 in. wafer, 10 µm film; estimate)25 | ∼0.1 g particles per mL reaction mixture70 | Roll-to-roll ∼108 particles in 1 mL from ref. 85 | ∼100–500 particles per s and ∼1.6 × 107 particles per h with 10-device chip100 | ∼4 × 10^5 particles per s131 | |
| ∼1010 particles per cm2 per cycle32 | ∼108 micrometer-sized tetrahedral particles per batch80 | ∼108–1012 particles per run89 | ∼200–1000 particles per s109 | ||
| ∼1.97 × 106 particles per 17.35 cm2 mold; yield 90.8 ± 1.9%34 | Up to several grams of particles84 | ∼5.6 × 106 particles per h110 | |||
| Up to ∼1012 or ∼4 × 1011 particles per 8-inch wafer38 | 2.5 × 109 particles94 | ∼400![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 particles per h116 000 particles per h116 |
|||
| Recovered mass example: 9.45 mg (PRINT nanogels radiolabeling run)30 |
2. Strategies for shape-engineered polymer nano/micro particles
2.1. Lithography
Lithographic platforms create nonspherical polymer particles by spatially patterning crosslinking within photosensitive films, delivering unmatched in-plane fidelity and narrow polydispersity.22 In conventional mask-based photolithography, a photoresist—typically composed of an oligomer/polymer matrix, a multifunctional acrylate crosslinker, and a radical photoinitiator—is spin-coated onto a substrate and exposed to UV light through a patterned photomask (Fig. 1a). When a positive photoresist is used, the exposed regions polymerize while unexposed areas are removed during development, and vice versa for a negative photoresist. The resulting photomask-defined pattern in the x-y plane penetrates through the resist thickness, defining the particle height in the z-direction.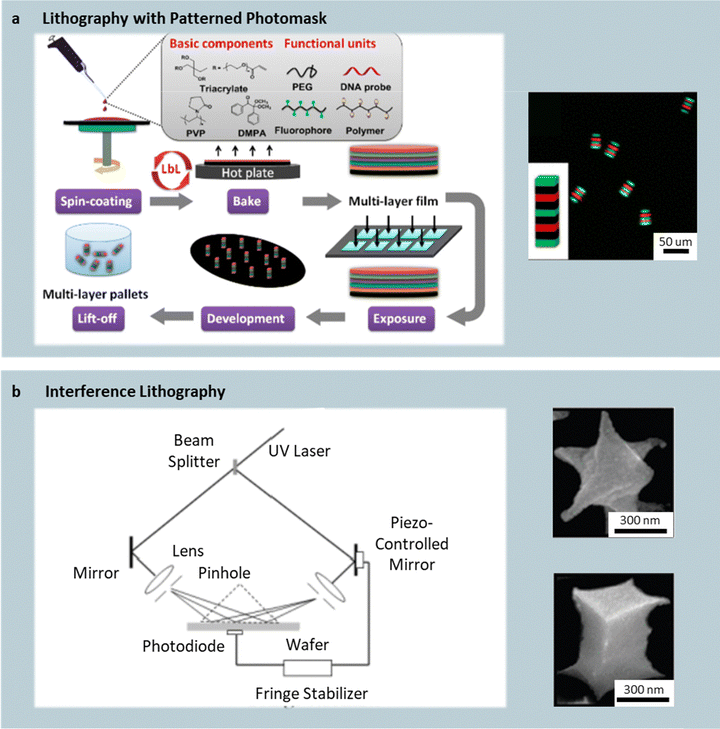 | ||
| Fig. 1 Mask-based and maskless photolithography techniques. (a) Schematic of mask-based photolithography showing the steps of spin-coating of a positive photoresist, UV exposure through a photomask, and development of cuboid particles. Fluorescent micrograph of cuboid particles produced using this mask-based lithography technique. Reprinted from ref. 24 with permission from American Chemical Society, copyright 2016. (b) Schematic of interference lithography using intersecting laser beams to polymerize a photosensitive resin without a mask, forming complex 3D microstructures. Reprinted from ref. 23 with permission from Wiley, copyright 2007. SEM image of spinning-top-shaped and cuboid particles produced using interference lithography. Reprinted from ref. 25 with permission from American Chemical Society, copyright 2007. | ||
Using a UV-curable oligomeric solution as the photoresist, cuboid particles with edge lengths between 5 and 200 µm have been fabricated (Fig. 1a).23 In addition to the common ingredients of a positive photoresist formulation, a volatile solvent such as dimethylformamide (DMF) can be included to improve film uniformity during spin-coating. As such, a representative positive photoresist formulation includes polyvinylpyrrolidone (PVP), trimethylolpropane ethoxylate triacrylate), DMF, and 2,2-dimethoxy-2-phenylacetophenone (DMPA, which can be activated during UV exposure). After spin-coating and solvent evaporation (typically via baking), curing is achieved via UV exposure through a square-patterned mask, generating radicals that crosslink the illuminated regions. Subsequent rinsing with isopropyl alcohol removes the unexposed material, revealing cuboid microparticles. Immersion in Tris–EDTA containing Tween-20 swells the lightly crosslinked network and reduces the wafer–particle interfacial tension, enabling clean detachment. Repeated spin-coat/bake cycles allow layer-by-layer (LbL) stacking of chemically distinct films—such as poly(ethylene glycol) (PEG200) or poly(allylamine hydrochloride) (PAH)—to build semi-interpenetrating polymer networks that enhance mechanical robustness or encode function. For instance, PEG or PAH layers can introduce porosity or DNA-binding capability, whereas alternating FITC- and TRITC-dextran-doped layers generate fluorescent “barcodes”.
Beyond mask-based approaches, lithographically fabricated polymer particles can also be produced without masks. Interference lithography (IL) is a maskless technique that uses the interference of coherent laser beams to create periodic, three-dimensional light intensity patterns within a photosensitive medium (Fig. 1b).24,25 Polymerization occurs only in regions of constructive interference, eliminating the need for costly photomasks and allowing rapid prototyping of complex geometries. After UV exposure, the solidified network forms a lattice of interconnected microstructures, which can be released as individual particles via mild dry etching or mechanical agitation. IL has been used to generate cubic, prismatic, and spinning-top particles (Fig. 1b). Although polymerization occurs volumetrically rather than on a planar substrate, the overall throughput is comparable to conventional photolithography—on the order of ∼109 particles per single exposure on a six-inch wafer, likely a result of the limited UV penetration. Nevertheless, IL has been considered to afford more flexible 3D design freedom compared to conventional photolithography.
Nonspherical polymer particles have been generated via hard-template replica synthesis, in which a pre-formed anisotropic particle serves as a sacrificial scaffold. In this approach, a non-spherical template, typically inorganic (e.g., silica rods, cubes, hematite ellipsoids) or polymeric, is first synthesized independently. A polymer shell is then deposited onto the template surface through surface-initiated polymerization, layer-by-layer (LbL) assembly, sol–gel coating, or crosslinking of adsorbed macromers. Subsequent selective removal of the core (e.g., by chemical etching, dissolution, or calcination) yields hollow or solid polymer replicas that preserve the original template geometry. This strategy has enabled polymeric replicas of rods, ellipsoids, cubes, dimpled particles, and lock-and-key geometries with high shape fidelity. For example, silica templates have been used to produce polymeric colloids with well-defined multicavities.35 Hollow silica and silica-polymer spheres (720–1000 nm) have also fabricated using LbL by sequentially depositing silica nanoparticles and polymer onto colloidal templates, followed by core removal via calcination or solvent decomposition. The wall thickness was tuned by adjusting the number of deposition cycles, while the final size and shape were dictated by the template morphology, yielding structures with potential applications in medicine, pharmaceutics, and materials science.36 While this method offers excellent geometric fidelity and versatility across material chemistries, scalability depends on the availability and cost of the initial anisotropic templates and the efficiency of core removal. Nevertheless, hard-template replication represents a widely adopted and complementary route to nonspherical polymer particles, particularly for hollow capsules and biomimetic structures.
When template-molding is used, the mold is often fabricated using photolithography and subsequent transfer or replication steps, which dictates the in-plane geometry of the particles while cavity depth defines the out-of-plane dimension (Fig. 2a). Virtually any planar shape – triangle, donut, heart, or ring – can be realized, and stacked structures can be obtained via precise control over mold depth and multistep procedures (Fig. 2a(i–vi)).
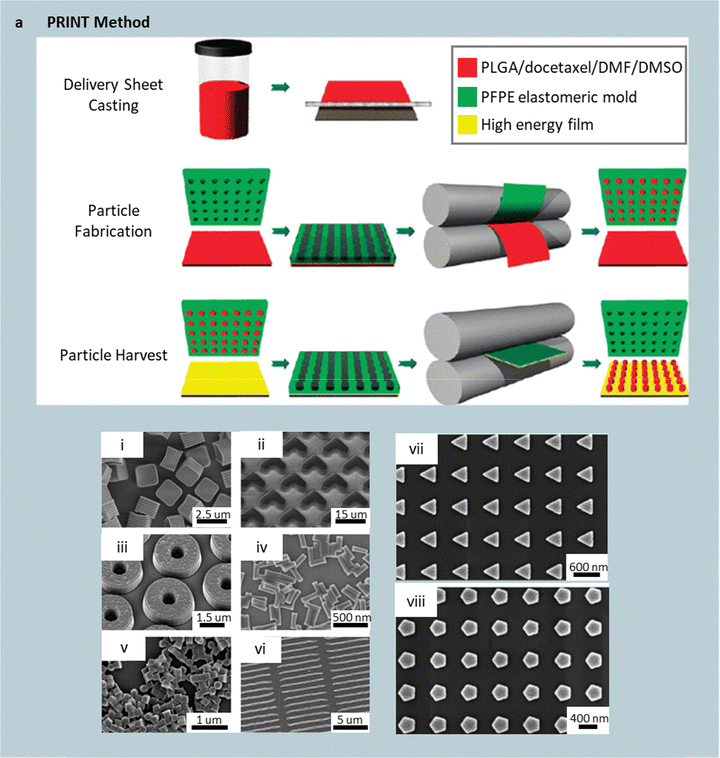 | ||
| Fig. 2 Template molding fabrication. Schematic of particle replication in non-wetting templates (PRINT) technique, comprising the steps of solution casting onto a delivery sheet, pressing the delivery sheet (red) against a patterned perfluoropolyether (PFPE) mold (green) using a heated roller to fill the mold with the particle-making material, pressing a harvesting substrate (yellow) against the filled mold using a heated roller to transfer the particles to a soluble substrate. PRINT has enabled particle shapes including (i) cuboids, (ii) boomerangs, (iii) rings, (iv) long cylinders, (v) short cylinders, and (vi) rods. Reprinted from ref. 26 with permission from American Chemical Society, copyright 2011. Step-and-flash imprint lithography (S-FIL) using a quartz mold pressed against a sacrificial PVA- and UV-curable precursor-coated wafer yields (vii) triangular and (viii) pentagonal particles after UV curing and PVA dissolution. Reprinted from ref. 38 with permission from Elsevier, copyright 2007. | ||
A representative example is particle replication in non-wetting templates (PRINT), which employs a lithographically patterned silicon master to define a perfluoropolyether (PFPE) mold. A photoresist pattern is first generated via lithography and used as an etch mask to transfer the design onto a silicon wafer.37 PFPE resin is then poured into the etched silicon cavities, cured, and peeled off to yield an elastomeric, non-wetting mold that reproduces the pattern. A polymer solution (e.g., degradable poly(D-lactic acid), biocompatible polyethylene glycol (PEG), or proteinaceous solutions) is cast onto a delivery sheet and partially dried to form a thin film. This coated sheet is pressed against the PFPE mold under mild heat and pressure, allowing the softened polymer to flow into the cavities. A high-surface-energy harvesting substrate is then laminated on top; due to the PFPE's low surface energy, the filled cavities release the solidified particles when the harvesting substrate is lifted and later dissolved, freeing the molded particles (Fig. 2a).
In addition to thermal curing, UV-assisted molding provides a complementary route for forming nanoscale features.38 For instance, step-and-flash imprint lithography (S-FIL) uses a quartz mold (transparent to UV light) patterned by high-resolution e-beam lithography, achieving feature sizes as small as 50 nm. The quartz mold is pressed onto a sacrificial PVA-coated silicon wafer coated with a UV-curable macromer (e.g., PEG diacrylate or dimethacrylate of varying molecular weights). Upon UV exposure, the monomer mixture polymerizes within the mold cavities. Removing the quartz reveals patterned protrusions joined by a thin residual film, which is removed by brief oxygen plasma etching. Dissolving the PVA layer releases the shaped nanoparticles (Fig. 2a(vii and viii)).
Template molding can also produce polymer cholesteric liquid crystal (PCLC) flakes with engineered optical properties for sensing or display applications. In one example, a PDMS mold replicated from a photoresist master is spin-coated with a thin PVA layer, then filled with a liquid-crystalline mixture containing diacrylates, monoacrylates, and chiral dopants. UV curing, followed by PVA dissolution, releases the PCLC flakes.39 Adjusting spin-coating speed precisely controls the PVA thickness and thus the final flake height, which is the difference between the molded height (10 µm in this case) and PVA thickness. A PVA coating obtained at 500 rpm yields a PVA thickness of ∼2 µm and thus flake heights of ∼8 µm, while 2000 rpm produces a PVA thickness of ∼0.5 µm, and thus ∼9.5 µm flakes, enabling tunable optical responses via controlled film thickness.
Lithography and template molding both deliver excellent shape fidelity and narrow size distribution, making them especially suitable for applications requiring rigorous dimensional control. However, scalability remains limited by planar processing throughput and mold-fabrication costs. Even small changes to the particle shape or size require remaking of the mold, making each design-to-test iteration laborious and costly. The achievable particle size is ultimately constrained by lithographic resolution, and true three-dimensional freedom is limited unless interference or multilayer approaches are employed. Despite these constraints, template molding provides a robust, reproducible path to nano- and microscale particles with diverse geometries and well-defined chemistries—an indispensable complement to photolithographic platforms.
2.2. Seeded emulsion polymerization
Seeded emulsion polymerization extends the classical emulsion polymerization framework to produce nonspherical polymer particles through polymerization-induced phase separation (PIPS).40–54 The process begins with narrowly dispersed seed particles, typically polystyrene (PS) or poly(methyl methacrylate) (PMMA), prepared via standard emulsion polymerization. Those seed particles are redispersed into a second emulsion containing fresh monomer, often accompanied by plasticizers, crosslinkers, or swelling agents. The monomer diffuses into and swells the seed particles. Upon initiation of its polymerization, a secondary polymer network forms inside the seed particles, often with a positive free energy of mixing (i.e., ΔGmix > 0), hence driving phase separation between the growing and pre-existing polymers. At the same time, chain mobility, determined by the relation between the reaction temperature and the glass-transition temperature (Tg), governs how far the phases can migrate before the matrix vitrifies. High chain mobility (T ≫ Tg) favors complete rearrangements, such as core–shell structures that often retain their overall spherical shape, whereas limited mobility locks transient domains into kinetically trapped nonspherical shapes. By tuning seed rigidity, monomer polarity, plasticizer content, inhibitor concentration, and swelling rate, each recipe can be positioned at a unique point on the thermodynamic-kinetic landscape, unlocking lobed, indented, Janus, and porous architectures. While emulsion polymerization is industrially relevant, there have been limited examples of scaled production of seeded emulsion polymerization, likely due to inconsistent particle morphology and properties55A representative example employs γ-ray polymerization of poly(styrene–divinyl benzene–acrylic acid) (p(S-DVB-AA)) seeds with 3-(methacryloxy)propyl-trimethoxysilane (MPS) and styrene (co-solvent).56 Varying styrene concentration modulates MPS solubility in the crosslinked seed and shifts the morphology sequence from raspberry-like (low styrene and small MPS domain size) to Mickey-Mouse (intermediate) to snowman-like (high styrene and large MPS domain size) particles (Fig. 3a). Similar lobed structures can be achieved using PMMA seeds at low reaction temperatures, where limited chain mobility confines polymerization to the seed surface, giving rise to raspberry-like particles.57 Subsequent annealing merges adjacent lobes as interfacial tension drives domain coalescence, reducing total polymer–water interfacial energy.
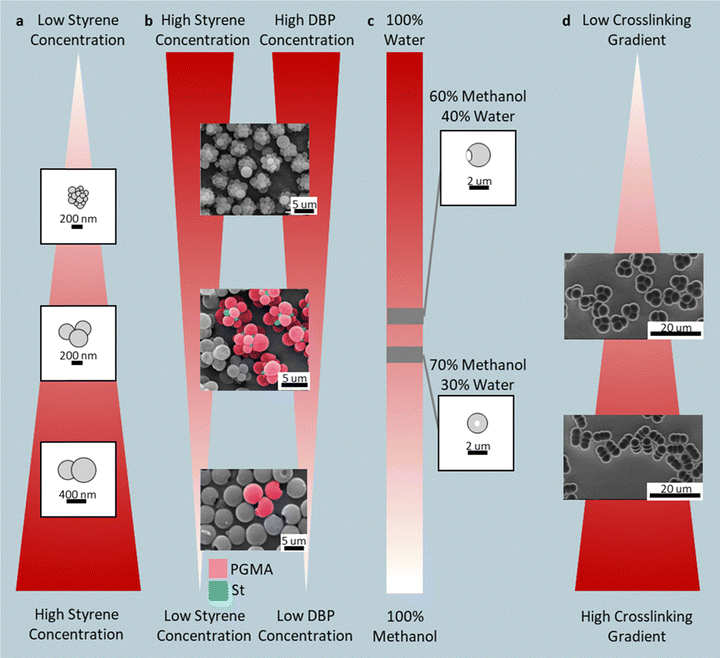 | ||
| Fig. 3 Monomer-driven swelling shapes: (a) raspberry, Mickey-Mouse, and snowman via styrene-MPS swelling of P(S-DVB-AA) seeds; (b) pineapple-, cluster-, and single-dimpled morphologies from PS/DBP swelling. Reprinted from ref. 58 with permission from American Chemical Society, copyright 2016; (c) single-dimpled and donut particles via APS-mediated water inclusion; (d) trimers, rods, and snowmen from multi-stage DVB gradients. Reprinted from ref. 60 with permission from Wiley, copyright 2007. | ||
Uncrosslinked seeds can also yield complex shapes. In one example, uncrosslinked poly(glycidyl methacrylate) (PGMA) seeds were partially swollen with dibutyl phthalate (DBP) and styrene.58 The swollen outer layer underwent phase separation, forming DBP-rich droplets that protrude from the seed surface. Upon seed removal (likely by centrifugation) from the swelling environment, the seeds are redispersed back into water, and the DBP diffuses back into the continuous phase; the protrusion pinches off, imprinting dimples on the PGMA seed. Next, these dimpled PGMA seeds are again swollen with controlled ratios of DVB, S, and azobis(isobutyronitrile) (AIBN). Polymerization leads to various-sized and numbers of PS protrusions. Adjusting DBP and styrene ratios produces pineapple-like (high DBP and St), cluster-like (moderate DBP and St), or single-dimpled spheres (low DBP and St) (Fig. 3b)
Reaction additives further enrich the morphology diversity. While the polymerization of a methacrylate mixture containing methyl methacrylate (MMA), glycidyl methacrylate (GMA), and ethylene glycol dimethacrylate (EGDMA) in PS seeds give rise to spherical particles, with a polymethacrylate shell and a PS core, the addition of inhibitors, such as 4-methoxyphenol (MEHQ) or dissolved O2 can slow the methacrylate polymerization to allow time for the shell to densify as the crosslinking density increases.59 That densification leads to a compressive mismatch between a rigid P(MMA–GMA–EGDMA) shell and a soft PS core, resulting in wrinkled raisin-like morphologies at methacrylate-to-PS mass ratios of 15–20. At mass ratios of 60, the seed absorbs far more monomer. Here, during polymerization, phase separation of the highly crosslinked region grows into a hemispherical shape while pushing into and greatly compressing the PS phase into a flat region, generating an acorn-like Janus particle. Introducing hydrophilic swelling agents such as ammonium persulfate (APS) draws water into the seed, forming trapped droplets that collapse into singular dimples or central voids upon drying.60 To control the particle morphology precisely, the polarity of the continuous phase can be adjusted by mixing methanol and water. Tuning the methanol-to-water ratio from 60/40 to 70/30 changes the droplet size, transitioning the particle morphology from single-dimpled spheres to donut-like particles (Fig. 3c).
Sequential emulsion polymerization steps can further encode asymmetry. For instance, PS seeds (∼1.5 µm) are first swollen and crosslinked in styrene with DVB, producing symmetric dimers.61 A second swelling step deliberately imposes a crosslinking-density gradient between the two bulbs by varying the DVB dose, establishing a tunable elasticity contrast that biases subsequent growth. In a third swelling, additional monomer and toluene are introduced; upon polymerization, morphology follows the preset gradient (Fig. 3d). Low elasticity contrast permits slow, near-isotropic monomer transport, yielding orthogonal triangular trimers. Intermediate contrast focuses growth at the softer bulb, producing linear three-bulb rods. At high contrasts, the more rigid bulb resists swelling while the softer bulb swells easily, suppressing new lobe formation and freezing the particle as a snowman-like dimer.
For example, seeded emulsion polymerization using uncrosslinked PS seeds swollen with 2-ethylhexyl methacrylate (EHMA) and hydrocarbon leads to particles that deform into predictable shapes upon solvent removal.63 Longer-chain hydrocarbons (e.g., dodecane, 12-carbon backbone) induce limited swelling, producing polyhedral particles, whereas shorter-chain hydrocarbons (e.g., hexane, 6-carbon backbone) enable greater deformation, yielding progressively flatter, saucer-like morphologies. Introducing PMMA particles as secondary seeds (i.e., dual-seeded emulsion polymerization) alters the polarity of the local environment, causing EHMA/hydrocarbon droplets to nucleate on regions of the PS seed farthest away from neighboring PMMA seeds, resulting in asymmetric swelling (Fig. 4a). This asymmetric swelling was observed with lower-affinity hydrocarbons to PS, such as dodecane. The dodecane nucleates on the PS seeds farthest away from the PMMA seeds, grow, and the subsequent PEHMA/hydrocarbon extraction (via 1-butanol), produced a bowl-shaped structure. Conversely, when hexane is used, its enhanced swelling capacity offsets the polarity imbalance caused by the PMMA seeds, thereby mitigating swelling asymmetry and nucleating evenly across the PS seed. After growth and extraction of the PEHMA/hydrocarbon domains, pits are left behind, forming a “Wiffle-ball-like” particle.
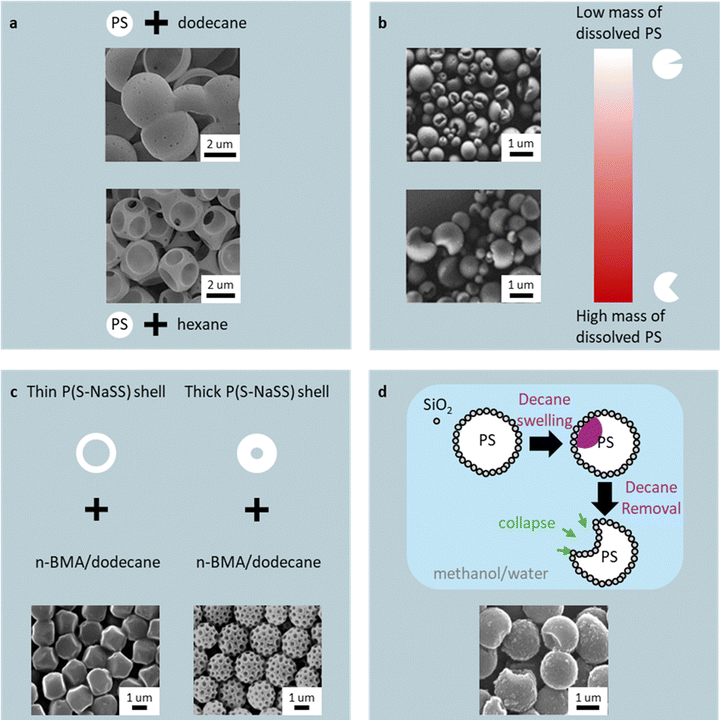 | ||
| Fig. 4 Hydrocarbon-swelling routes: (a) bowl and wiffle-ball morphologies from dodecane/hexane swelling. Reprinted from ref. 63 with permission from Springer, copyright 2012; (b) Pac-Man-like cavities from dissolved PS seeds. Reprinted from ref. 64 with permission from American Chemical Society, copyright 2018; (c) dodecahedral and golf-ball-like shapes from core–shell PS/P(S-NaSS) seeds. Reprinted from ref. 65 with permission from The Society of Polymer Science, copyright 2010; (d) bitten-apple PS–SiO2 particles. Reprinted from ref. 66 with permission from American Chemical Society, copyright 2018. | ||
Complete dissolution of the seed may also unlock new shapes. When PS seeds are pre-swollen with 1-chlorodecane (in water) and then an oil phase (acrylic acid (AA)) emulsion consisting of styrene (S), divinylbenzene (DVB), sodium dodecyl sulfate (SDS), and AIBN is added, an emulsified droplet containing untangled PS chains is achieved.64 Once polymerization begins, the dissolved PS chains adsorb onto the nascent poly(styrene-co-divinylbenzene) (PSDVB) nucleus and sterically block the nucleus's internal propagation sites. Due to acrylic acid (AA) being hydrophilic, contact with the nuclei confines propagation to the oil–water interface. The hydrophobic PS chains, in turn, retreat from this interface, shielding the interior nucleus while leaving reactive sites exposed at the boundary, resulting in preferential growth along the interface. Increasing the mass of dissolved PS (i.e., dissolving more seeds) increases site shielding, driving more polymerization at the interface. For example, an open-mouth Pac-Man-like particle (Fig. 4b) was achieved at a high concentration of dissolved PS due to extensive interior propagation site shielding, inhibiting polymer growth at the core of the droplet and preferential growth around the perimeter. Whereas when the concentration of PS chains is decreased, fewer internal propagation sites are shielded, resulting in increasingly more polymer being formed in the core, causing the Pac-Man mouth to progressively close.
Core–shell seed designs provide additional shape control. PS/poly(styrene-co-sodium styrene sulfonate (P(S-NaSS)) core–shell particles swollen with n-butyl methacrylate (n-BMA) and Dodecane yield dodecahedral or golf-ball-like morphologies depending on the shell thickness and PS molecular weight (Fig. 4c).65 Thinner shells and lower PS molecular weight enable the swollen n-BMA/dodecane domains to coalesce more readily. After polymerization, the composite particles were isolated by repeated centrifugation and methanol washing, then air-dried at room temperature. During drying, evaporation of Dodecane causes volumetric shrinkage of the few large P(n-BMA)/dodecane domains, driving interfacial-tension-induced collapse of the shell into a faceted, polyhedral structure. Conversely, seeds with thicker or higher-molecular-weight shells restrict domain coalescence, forming numerous smaller P(n-BMA)/dodecane domains; upon evaporation, shrinkage of these domains produces the characteristic golf-ball-like dimpling is synthesized. Similarly, silica-nanoparticle-coated PS seeds swollen with decane produce “bitten-apple”-shaped particles approximately 500 nm in diameter with up to 150 nm 'bite sizes' (Fig. 4d).66
During dispersion polymerization to create the PS–SiO2 seeds, 22 nm methanolic silica colloids electrostatically interact with the cationic initiator, 2,2′-azobis(isobutyramidine) dihydrochloride (AIBA).67 In conjunction with the low repulsive forces between silica nanoparticles, this interaction promotes the formation of a densely packed, contiguous silica-rich surface on the PS particles. During the shaping step, decane is added to a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol/water medium that partitions preferentially into the PS core because (i) decane is only sparingly soluble when water is present, and (ii) the permeable yet mechanically robust silica shell aids the particle in resisting deformation, leading to the formation of a single decane-rich bulge. As the suspension cools, decane evaporates more rapidly than the plasticized PS can relax, causing the silica shell to collapse and leave one deep crater—the characteristic “bitten-apple” shape.
1 methanol/water medium that partitions preferentially into the PS core because (i) decane is only sparingly soluble when water is present, and (ii) the permeable yet mechanically robust silica shell aids the particle in resisting deformation, leading to the formation of a single decane-rich bulge. As the suspension cools, decane evaporates more rapidly than the plasticized PS can relax, causing the silica shell to collapse and leave one deep crater—the characteristic “bitten-apple” shape.
Post-PIPS removal can imprint surface texture by selectively dissolving the sacrificial polymer while leaving the matrix intact. Uncrosslinked PGMA microspheres (4–8 µm in diameter) have been swollen with DBP in the presence of styrene (for 24 hours at 40 °C).75 Upon AIBN-initiated polymerization at 80 °C, the styrene polymerizes, giving rise to buds that nucleate and coarsen on the PGMA surface to produce raspberry-like clusters (Fig. 5a). A rapid (20 seconds) rinse in 85–95 vol% THF/H2O dissolves PS but swells PGMA, lifting the buds and a thin subsurface layer to leave “golf-ball” dimpling (Fig. 5a). The selective removal of PS can be explained using the Hildebrand solubility parameter (δ), as THF and PS have comparable δ's, which differ from PGMA. Similarly, programming the interfacial tensions of the two polymer phases against water prescribes whether hemispheres develop convex, flat, or concave bases (Fig. 5b). Uncrosslinked PS seeds (∼1 µm in diameter, plasticized with toluene) have been swollen with a sacrificial (meth)acrylate in the presence of a surfactant, giving rise to Janus particles after PIPS.76 With tert-butyl acrylate (tBA) and sodium dodecyl sulfate (SDS), the tBA–water tension is slightly lower than the PS–water tension, so the ptBA phase partially engulfs the PS phase, reducing the PS surface area in contact with the water. Dissolving the ptBA reveals PS hemispheres with a convex base. Keeping tBA but replacing SDS with cetyltrimethylammonium bromide (CTAB) lowers the PS–water tension below the tBA–water tension (due to better affinity to the PS); the higher-energy ptBA interface now bulges into the PS phase, and dissolving it leaves a concave base. Interfacial tensions were calculated by measuring the contact angle of water containing CTAB or SDS atop thin films of either polymer. Switching the monomer to tert-butyl methacrylate (tBMA) in SDS lifts that sacrificial-polymer tension almost to parity with the PS–water value, leaving neither phase favored. Hence, no bulge, thus, the internal boundary between the two polymers is flat, and removing the sacrificial half yields a flat-based hemispherical PS particle.
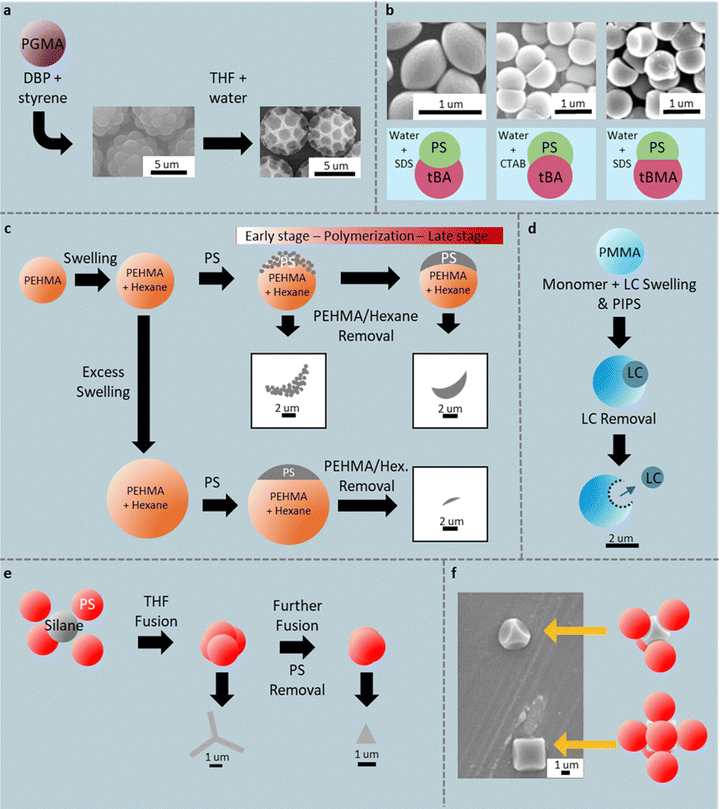 | ||
| Fig. 5 Sacrificial-phase and confinement routes to voided spheres, hemispheres, bowls, helmets, Y-shapes, pyramids, and concave-faced cubes. (a) PGMA-PS raspberries made by PIPS. Reprinted from ref. 75 with permission from American Chemical Society, copyright 2024; (b) golf balls made by selectively dissolving PS with a rapid rinse using THF/H2O. (c) Hemispheres with convex, concave, and flat bases via control over the interfacial tension. Reprinted from ref. 76 with permission from American Chemical Society, copyright 2017. (c) PHEMA/hexane routes to porous bowls and thick shells; saucers at high hexane content. (d) LC-templated PMMA helmets; (e) Y-shapes and pyramids from fused PS clusters. (f) TPM droplets polymerized in tetrahedral or octahedral colloidal cages formed by PS particles. Reprinted from ref. 80 with permission from American Chemical Society, copyright 2017. | ||
Combining hydrocarbons with a sacrificial phase or using an inert liquid-crystal droplet also expands the accessible bowl and shell morphologies. Moderately hexane-swollen PEHMA seeds have been shown to maintain sufficient rigidity to support the asymmetric formation of PS domains that coalesce and, upon PEHMA sacrifice, a porous bowl/shell is formed (Fig. 5c).77 The asymmetric precipitation is caused by dominant St uptake by the PS oligomers that nucleate first on the PEHMA particle. Further polymerization back-fills pores to thicken the shell (Fig. 5c). Excess hexane greatly enlarges the PEHMA seed, and the PS domain then forms a small cap sitting on a larger sphere leading to a flat, saucer- or disc-like morphology (Fig. 5c). Alternatively, a single E7 liquid-crystal droplet can be pushed outward from crosslinked PMMA seeds, swollen by styrene, MMA, and E7, during PIPS; methanol extraction of E7 leaves hollow helmet-like microcapsules stabilized by the crosslinked matrix (Fig. 5d) Here the shaped particle is meant to hold the LC, giving rise to controlled electro-optical particle properties; however, LC removal yields shaped polymer particles.78
Finally, geometric confinement imposed by pre-arranged seed packings acts as a colloidal mold that fixes otherwise in accessible polyhedral shapes. Partial THF-induced fusion of tetrahedral PS clusters lets a polymerizable silane oil ((3-glycidyloxypropyl) trimethoxy silane and 3-(trimethoxysilyl)propyl methacrylate) extrude through interstices and be trapped mid-transit on curing, giving Y-shaped hybrids (Fig. 5e); complete fusion closes interstices and yields pyramidal particles.79 Similarly, compressing monodispersed PS colloids creates tetrahedral clusters and octahedral cages that are capillary-wetted with 3-methacryloxypropyltrimethoxysilane (TPM) droplets.80In situ polymerization produces smooth-edged pyramids (in tetrahedral sites) and concave-faced cubes (in octahedral sites) in the same batch (Fig. 5f).
2.3. Mechanical deformation
Mechanical routes can reshape pre-made spherical particles, typically PS, PMMA, PLGA, or poly(ε-caprolactone) (PCL), into anisotropic shapes by softening the polymer (heat or solvent) and imposing shear, tensile, or compressive strain. Most methods highlighted here utilized feedstock particles ranging from a few hundred nanometers to tens of microns. Overall, these methods incorporate relatively inexpensive tools and straightforward processes to deliver large numbers of shape-controlled particles, although strain heterogeneity can lead to shape dispersity, and the frequent need for high temperature or plasticizing solvents further reduces the batch-to-batch reproducibility.81,82 Fundamentally, shapes emerge from three coupled levers: viscosity of the polymer melt that constitutes the particle (set by T–Tg and solvent uptake), interfacial wetting with the embedding matrix, and the spatial uniformity of the applied strain field. Basic operations, such as stirring a suspension of PS particles in the presence of plasticizers (e.g., 2 wt% PVP), yield rods and ellipsoids.83,84 Judicious control over viscosity and wetting, as detailed below, further expands the library to discs, barrels, bullets, UFOs, and facetted polyhedral (Fig. 6 and 7).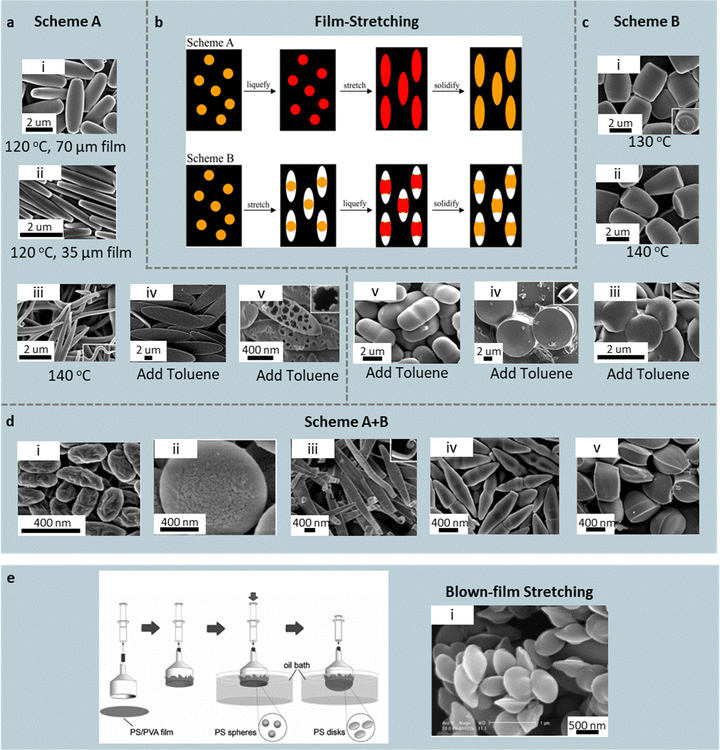 | ||
| Fig. 6 Film-stretching routes to anisotropic polymer particles. (b) Schematic of two film stretching methods: (a) Scheme A (liquefy, stretch, solidify) and (c) Scheme B (stretch, liquefy, solidify), for spheres embedded in a polymer film. Scheme A: (a-i) rods from a 70-µm film at 120 °C, (a-ii) flat rectangular disks from a 35-µm film at 120 °C, and (a-iii) worm-like particles at 140 °C. Using toluene instead of heat yields (a-iv) elliptical disks, which become (a-v) porous when quenched into IPA. (c) Scheme B: (c-i) barrels at 130 °C and (c-ii) bullets at 140 °C. With toluene: (c-iii) pill-shaped particles under 1D draw; (c-iv) pulleys (c-v) and biconvex lenses under moderate and strong biaxial draw, respectively. (d) Combining schemes yields (d-i, ii) wrinkled ellipsoids, (d-iii) ribbons, (d-iv) bicones, and (d-v) tacos. Reprinted from ref. 89 with permission from National Academy of Sciences, USA, copyright 2007. (e) Schematic of the blown-film stretching method, which produces (e-i) UFO-like particles from PS/SiO2 particles. Reprinted from ref. 91 with permission from Wiley, copyright 2008. | ||
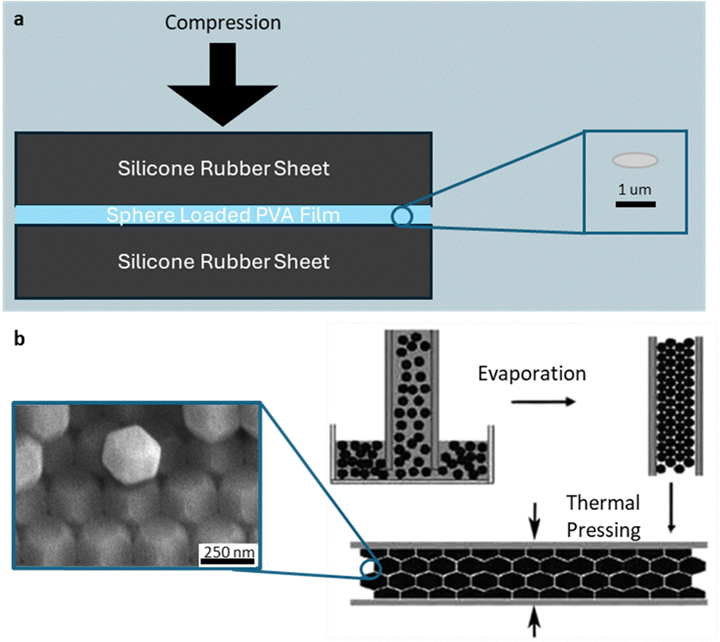 | ||
| Fig. 7 Compression-based shaping of polymer particles. (a) Film squeezing method: schematic showing PVA film embedded with PS spheres compressed between two elastic sheets at elevated temperature to induce biaxial deformation. Resulting monodispersed disc particles after cooling and film dissolution. (b) Thermal pressing of packed lattices: schematic illustrating close-packed polymer spheres confined between glass slides, heated slightly below Tg, and compressed to form faceted particles. Resulting polyhedral particles by thermal pressing. Reprinted from ref. 96 with permission from American Chemical Society, copyright 2005. | ||
In Scheme A, particle shape is governed by the particle size, particle viscosity (after liquefying), and film thickness (Fig. 6a). At 120 °C, a ∼2× uniaxial draw of spheres (∼2 µm in diameter) in a 70 µm film produces rods (Fig. 6a(i)), while the same temperature in a thinner, 35 µm film flattens particles (∼1 µm in diameter) into rectangular disks (Fig. 6a(ii)). Here, thinner films leads to the production of flatter particles due to strong particle association with the film arising from hydrogen bonding. Heating the particles to 140 °C lowers viscosity and enables skinny worm-like particles (Fig. 6a(iii)). Replacing heat with toluene further depresses viscosity; a 5-fold draw of particles (∼2 µm in diameter) embedded in a 35-µm film yields ultrathin elliptical disks (tens of nm thick) (Fig. 6a(iv)), which become porous when quenched directly into IPA (as solvent extraction outpaces chain relaxation; Fig. 6a(v)). Larger feedstock particles (∼6 µm) bias toward UFO-like caps, likely due to the limited plasticization at the center of the particle. Symmetric 2D strain gives oblate ellipsoids. Smaller feed particles (∼200 nm in diameter), upon toluene liquefaction, produce flat circular disks due to the more complete deformation compared to larger particles.
In Scheme B, the film is stretched while particles remain glassy, creating ellipsoidal voids that are then filled upon softening; shape is governed by both viscosity and particle–film wetting (Fig. 6c). At 130 °C, PS particles (∼3 µm in diameter) in a 35 µm film remain viscous (cP not reported) and thus partial void filling, yielding barrels (Fig. 6c(i)); slightly lowering viscosity drives asymmetric wetting at 140 °C favoring bullet shape (Fig. 6c(ii)). With toluene, viscosity is further lowered, causing uniform filling yielding pills (1D draw; Fig. 6c(iii)), while 2-fold (biaxial draws form pulleys and biconvex lenses, from ∼10 µm and ∼1 µm particles, respectively (Fig. 6c(vi and v)).
In addition, alternating Schemes A/B and changing draw axes generate wrinkled ellipsoids (Fig. 6d(i and ii)), ribbons (Fig. 6d(iii)), bicones (Fig. 6d(iv)), tacos (Fig. 6d(v)), and other hybrids. Typical lab batches yield 108–1012 particles (1–30 µm). Further strain-field engineering and automation are possible. An eight-clamp stretcher (equipped with an oil bath) creates deliberate strain heterogeneity in one pass—central true-biaxial zones form discs; inter-clamp uniaxial corridors produce rods; near-clamp regions give ellipsoids—useful for rapid generation of libraries of intrinsically shape-polydisperse particles. In addition to PS, poly(lactic-co-glycolic acid) (PLGA) and poly(ε-caprolactone) (PCL) microparticles have been used as feedstock.90 Blown-film stretching distributes tension more uniformly: a heated, particle-laden membrane is inflated over a 50-mm funnel, narrowing the size/shape distribution of ellipsoidal particles made of PVA or PS (Fig. 6e).91 Using this method, PS/SiO2 core–shell particles adopt UFO morphologies under higher inflation (Fig. 6e–i). For heat-sensitive cargo, low-temperature (≈50 °C) biaxial stretching of chitosan-grafted poly(isobutyl cyanoacrylate) (PIBCA) nanoparticles generates lemon-like drug-carrier particles with mucoadhesive shells.92
It is important to note that while throughput scales with particle loading in the film, increasing loading could induce contact and shape distortion. Automation (servo grips in a temperature-controlled chamber) reduces film tearing and improves run-to-run reproducibility; however, as with all stretching, it cannot eliminate strain gradients entirely.
A complementary approach, thermal pressing of packed lattices (Fig. 7b), uses capillary assembly to organize PS spheres (∼250 nm in diameter) into a close-packed lattice between glass slides.96 Applying a brief heating at sub-Tg temperature (∼110 °C, i.e., 6 °C lower than the Tg) and ∼5 MPa pressure eliminates voids while avoiding particle fusion. Cooling under the compressed state gives rise to a polyhedral lattice. The morphology of the polyhedral particles, e.g., square, hexagonal, triangular, reflects their packing symmetry. The resulting polyhedral arrays display tunable optical diffraction that scales with particle size and compression. While this is a facile method to create polymeric polyhedra, it is limited to faceted morphologies. In addition, the particles near the boundaries deform differently from those in the bulk, leading to inherent heterogeneities. The separation and recovery of individual particles from the compressed lattice is likely non-trivial.
2.4. Microfluidic
Microfluidic devices enable the continuous production of shaped polymer microparticles by coupling droplet formation with rapid photopolymerization. Typically, a syringe pump drives the flow in a PDMS-molded channel (designed using CAD and fabricated through lithography). The precursor solution (i.e., monomer/polymer) can be emulsified in-line by mixing with an immiscible solvent (e.g., water with small amounts of sodium dodecyl sulfate). Particle geometry is prescribed by (i) channel confinement (Fig. 8), (ii) interfacial tension in free droplets (Fig. 9), or (iii) patterned UV exposure of the flowing stream (Fig. 10). The Rayleigh–Plateau instability confines products to the 10–200 µm range, but the microfluidic devices deliver singly dispersed particles with deterministic control of their geometric dimensions. Simple channel designs yield plugs, rods, and disks; unconstrained coaxial droplets enable high-curvature “hamburger”, “olive”, and spherical-cap motifs; and mask projection produces virtually any planar extrusion.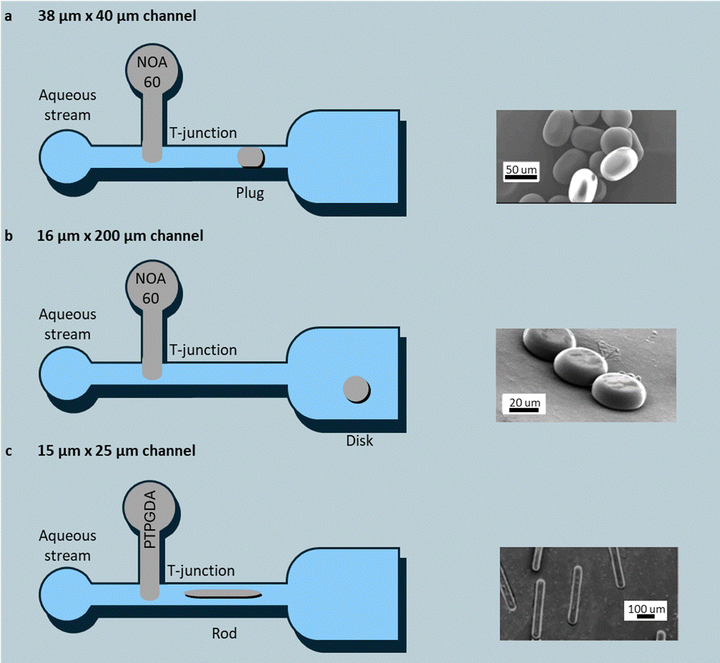 | ||
| Fig. 8 Microfluidic channel-confinement schemes for the production of shaped particles, including (a) plug particles formed in a tall rectangular channel; (b) disk particles formed in a shallow channel. Reprinted from ref. 97 with permission from American Chemical Society, copyright 2005; (c) rod particles formed by forcing droplets through a constriction before UV curing. Reprinted from ref. 100 with permission from American Chemical Society, copyright 2005. | ||
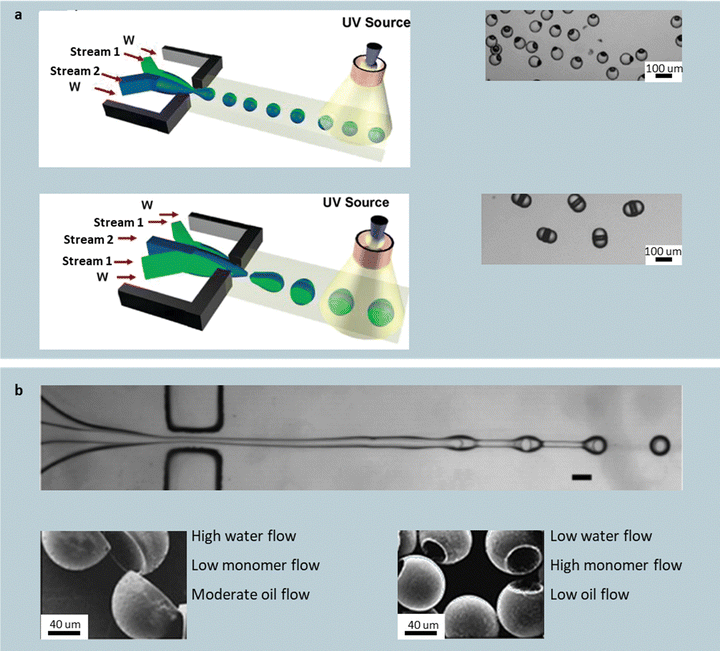 | ||
| Fig. 9 Unconfined, interfacial-tension-guided particle shape engineering. (a) Olive particles from asymmetric coflow of two streams; hamburger particles from core–shell coflow. Reprinted from ref. 108 with permission from American Chemical Society, copyright 2006. Sacrificial-core comprising silicone oil for particles with concave compartments (b); concave spherical caps obtained at low water and high monomer flow rates; pitted olive particles at high water and low monomer and oil flow. Reprinted from ref. 109 with permission from American Chemical Society, copyright 2005. | ||
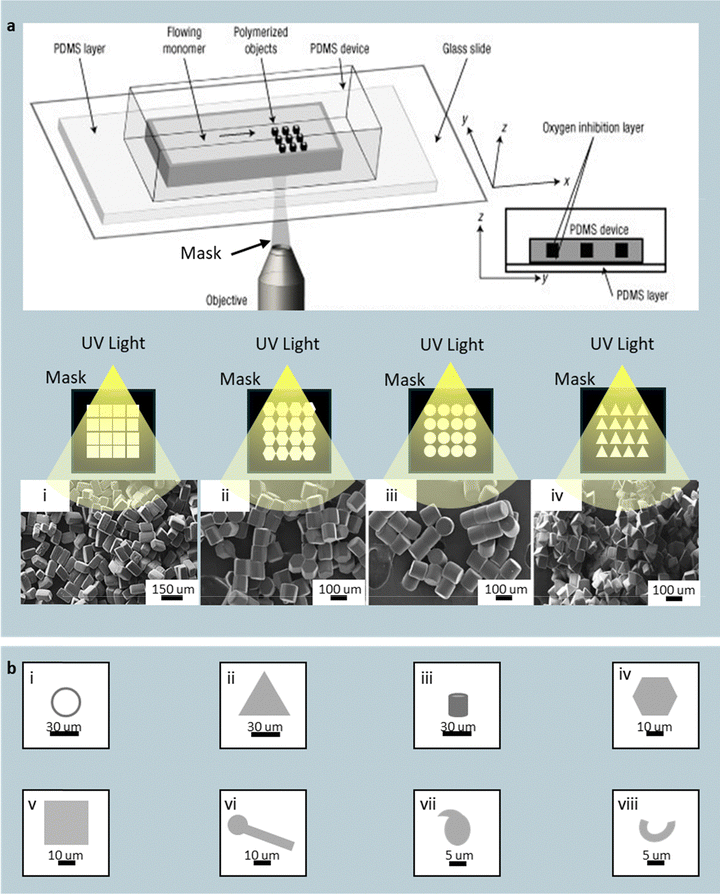 | ||
| Fig. 10 UV-mask photopolymerization of flowing prepolymer streams in microfluidic channels. (a) Schematic showing a continuous stream of UV-curable prepolymer exposed to patterned UV light through a photomask, where the illuminated cross-sections polymerize into solid particles. Mask-defined shapes fabricated via stop-flow projection lithography include (a-i) square, (a-ii) hexagonal, (a-iii) circular, and (a-iv) triangular particles. Reprinted from ref. 115 with permission (open access) from Springer, copyright 2011. (b) Continuous-flow patterned UV exposure extends the accessible morphologies to (b-i) rings, (b-ii) triangles, (b-iii) cylinders, (b-iv) hexagons, (b-v) cuboids, (b-vi) flat lollipops, (b-vii) commas, and (b-viii) horseshoe-like particles. Oxygen diffusion through PDMS channel walls forms a thin inhibition layer that prevents wall adhesion and enables continuous downstream transport of the patterned particles. | ||
In 40 µm-wide, 38-µm high channels, droplets were shaped into plugs (Fig. 8a). A sub-microsecond UV flash then “freezes” the droplets into shaped particles. Reducing the channel height to 16 µm flattens the droplets into disks (Fig. 8b), whereas constrictions narrower than the droplet diameter stretch them into cylinders (Fig. 8c). Length and thickness of the particles follow directly from flow-rate ratios of 0.01 to 0.1 µL min−1, as well as channel geometry, enabling deterministic tuning at throughputs up to 500 particles s−1 per chip.
While this method is straightforward to implement, the curing kinetics can introduce uncertainty. Insufficient initiator or UV exposure leads to a solid shell and liquid core, hence buckling upon extraction, whereas rapid cures can 'burst' droplets into irregular blobs from the exothermic reaction. A narrow window of reaction conditions and pulse energy that has been optimized empirically is often required for the reliable production of defect-free particles.
Replacing stream 1 with sacrificial silicone oil introduces a concave compartment into the spherical particles (Fig. 9b). An example system comprises a coaxial jet, where a central stream of silicone oil (containing 0.2–2.0 wt% sorbitan monooleate or SPAN 80 to stabilize the oil–monomer interface during droplet formation) is wrapped by an acrylate stream (e.g., tripropyleneglycol diacrylate or EGDMA) with 4 wt% of a photoinitiator (e.g., 1-hydroxycyclohexyl phenyl ketone (HCPK)), which is sheathed in water with 2 wt% SDS.109 Similarly to the previous method, the coaxial jet breaks into droplets due to Rayleigh–Plateau instability. Particles with a concave compartment can be achieved upon polymerization and oil extraction, with the morphology tunable via the flow rates of oil, monomer, or aqueous phase. Low water and high monomer flow rates produce concave caps, whose aspect ratio decreases with the oil flow rate. High water and lower monomer and oil flows impose greater shear and smaller encapsulated volumes, shifting products to off-center, pitted-olive particles. These routes are continuous and produce singly dispersed particles, but are limited to concave geometries.
To overcome these constraints, a continuous-flow variant integrates the same photomask principle but exposes the moving prepolymer stream to rapid (<1 s) patterned UV flashes (Fig. 10b).116 A PEGDA formulation, containing a commercially available photoinitiator, Darocur 1173 (an α-hydroxy-alkylphenone), continuously polymerizes into singly disperse rings (Fig. 10b(i)), triangles (Fig. 10b(ii)), cylinders (Fig. 10b(iii)), hexagons (Fig. 10b(iv)), cuboids (Fig. 10b(v)), flat lollipops (Fig. 10b(vi)), commas (Fig. 10b(vii)), and horseshoe-like particles (Fig. 10b(viii)). An AUTOCAD-designed photomask with ∼10 µm features is inserted at the microscope field stop, and the objective lens projects the pattern onto the flowing oligomer. A sub-second UV flash cures the illuminated cross-section, while oxygen diffusing through the PDMS channel prevents clogging by forming chain-terminating peroxide radicals near the channel surface (via a reaction with the initiator), leaving a thin unpolymerized lubrication layer. This continuous photolithographic microfluidic process achieves throughputs approaching 100 monodisperse particles s−1 per chip, while maintaining precise xy-plane fidelity. Although confined to planar extrusions and limited by optical depth of field, the UV-mask approach bridges conventional lithography and flow photopolymerization – offering mask-defined particle shapes without the need for molds or droplets.
2.5. Miscellaneous
A small class of unconventional fabrication methods broadens the shape palette of polymer particles by coupling polymer softening, vapor-phase condensation, or field-driven droplet shaping with straightforward bench-top setups. These approaches – often limited in throughput – demonstrate unique shape-forming mechanisms inaccessible to conventional lithography, emulsion, or microfluidic systems.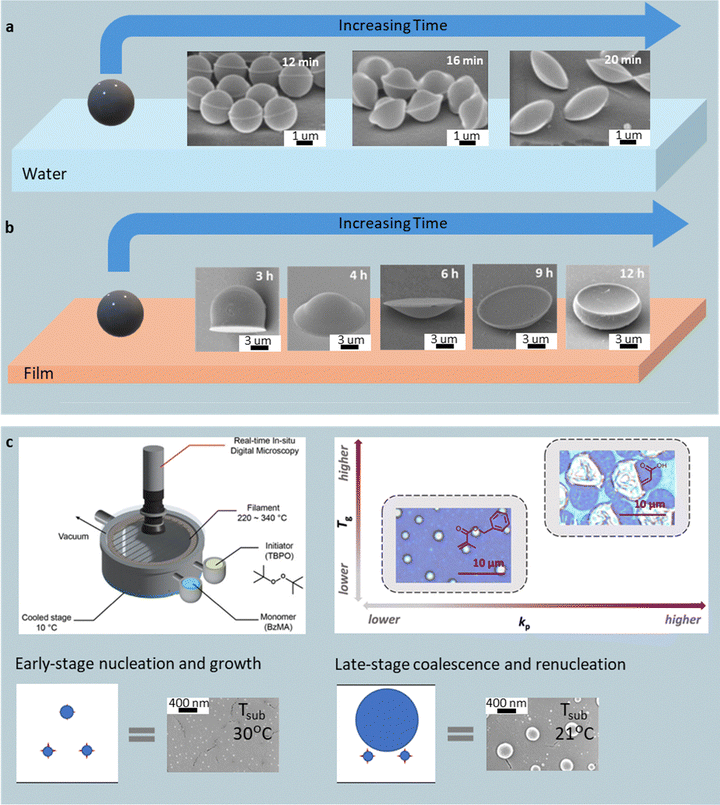 | ||
| Fig. 11 Solvent-vapor and condensed-droplet shaping of polymer particles. (a) Particle-on-water method in the presence of the vapor of an organic solvent relaxes PS spheres to form Saturn-, UFO-, and disk-shaped particles. Reprinted from ref. 122 with permission from Wiley, copyright 2016. (b) Particle-on-film in the presence of the vapor of an organic solvent produces bell, UFO, cymbal, contact lens, and bowl geometries. Reprinted from ref. 123 with permission from American Chemical Society, copyright 2017. (c) Schematic of CDP (Reprinted from ref. 127 with permission from Wiley, copyright 2025), combining vapor condensation and radical polymerization (Reprinted from ref. 126 with permission from American Chemical Society, copyright 2023) to yield a unimodal distribution of nanodomes or a bimodal distribution of larger synthesized microdomes. Reprinted from ref. 125 with permission from Thieme, copyright 2023. | ||
Replacing the water interface with a polymeric thin film expands the accessible morphologies. When PS spheres (10 µm) are spin-coated onto PMMA films and annealed in toluene vapor at 30 °C, swelling of both polymers enables wetting and partial sinking into the softened PMMA surface.123 The sequence of shapes evolves predictably with annealing time, i.e., bell (3 hours), UFO (4 hours), cymbal (6 hours), contact-lens (11 hours), and bowl (12 hours) geometries, reflecting progressive minimization of total surface and interfacial energies (Fig. 11b). To release the particles, PMMA can be dissolved using acetic acid. Contacting PS particles can fuse into various peanut-like shapes. Substituting toluene with cyclohexane, which swells PS but leaves PMMA rigid, halts the sinking behavior and produces uniformly flattened discs. The shape outcome can be tuned by solvent choice, vapor activity, and exposure duration, though the multi-hour processing limits throughput.
Polymer particles can also be synthesized directly from the vapor phase without any pre-existing seed. Condensed droplet polymerization (CDP) combines monomer condensation and radical-initiated polymerization under vacuum (1 mTorr–1 Torr).124–128 Monomers with a double bond are vaporized and condensed as droplets onto a thermoelectrically cooled, non-wetting substrate (usually kept at ≤10 °C). A tert-butyl peroxide vapor feed is thermally decomposed by a heated filament array to generate radicals that initiate polymerization within the condensed droplets to form solid spherical domes. The droplet contact angle controls curvature; early-stage nucleation and condensation yield sub-100 nm domes, while later coalescence generates a bimodal, micrometer-scale population. The interplay between propagation rate constant (kp) and Tg dictates the dome morphology: moderate kp and Tg values yield smooth hemispherical domes, while high values produce wrinkled or fragmented shells after unreacted monomer removal (Fig. 11c). The dome particle size and distribution can be controlled in real time using optical and interferometric monitoring. CDP thus offers rapid (<1 min), solvent- and template-free fabrication of morphologically tunable particles, though its current product range remains limited to hemispherical and disc-like geometries. Nevertheless, a broad range of functional monomers—including hydrophilic, aromatic, amine-containing, and crosslinkable types—have been made into dome particles using CDP.
In one representative setup, a 2 wt% alginate solution is electrosprayed through a 50 µm needle toward a calcium–chloride (CaCl2) crosslinking bath (Fig. 12a).131 The distance between the syringe and bath sets the droplet flight time, while CaCl2 concentration dictates the gelation rate. Short distances (3 mm) and low CaCl2 concentrations (1–2.5 wt%) yield tailed spherical particles, while longer distances (20 mm) promote gumdrop geometries, and high CaCl2 concentrations (7.5–10 wt%) rapidly fix hemispherical shapes upon impact, preserving the flattened base. Introducing shear to the bath (e.g., stirring at 50–500 rpm) elongates droplets into teardrop or comma-shaped particles as they gel. This method operates at room temperature, produces hundreds of microparticles per second, though particle sizes typically remain in the 100–300 µm range and are confined to hydrogel chemistries.
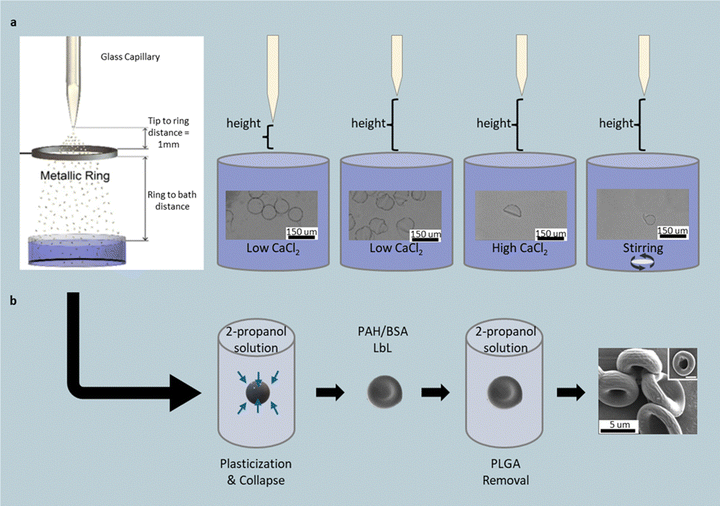 | ||
| Fig. 12 Electrospraying and electrohydrodynamic jetting to shape polymer particles. (a) Schematic of the electrospraying setup in which 2 wt% alginate droplets are crosslinked by CaCl2 to form acorn-, gumdrop-, and hemisphere-like particles. Stirring the crosslinking bath yields comma-shaped particles. Reprinted from ref. 131 with permission from Wiley, copyright 2018. (b) Hollow, red-blood-cell-like protein capsules generated from solvent-plasticized PLGA templates coated via layer-by-layer deposition and core removal. Reprinted from ref. 132 with permission from National Academy of Sciences, USA, copyright 2009. | ||
Electrospray principles can also be extended to solvent-driven deformation of solid polymer spheres. PLGA particles produced by electrohydrodynamic jetting are briefly immersed in 2-propanol to plasticize their outer shells, which buckle under internal stress to form biconcave discs.132 Subsequent layer-by-layer deposition of poly(allylamine hydrochloride) and bovine serum albumin, followed by PLGA dissolution, leaves hollow red-blood-cell-like protein capsules. Analogous structures can be fabricated from commercial hollow polystyrene beads by applying the same coating process and dissolving the core in tetrahydrofuran (Fig. 12b). These solvent-triggered plasticization routes enable reproducible formation of concave, biomimetic particles, though they rely on organic solvents and post-processing steps that limit scalability.
3. Discussion and conclusion
The diversity of accessible polymer particle morphologies, from rods and discs to bowls, lobes, and polyhedral, reflects a unified set of governing principles across fabrication families. Shape evolution is dictated by four dominant parameters: (i) geometric confinement (molds, microchannels, or colloidal packings), (ii) interfacial tension and wetting contrasts that prescribe curvature, (iii) polymer chain mobility relative to the reaction or deformation rate (T−Tg), and (iv) external field gradients (mechanical strain, shear, or electric fields). Together, these parameters define the thermodynamic and kinetic envelope that fixes particle geometry during chemical or physical solidification.Each method family occupies a distinct mechanistic and operational niche. Lithography and template molding deliver precise xy-plane fidelity for prismatic or extruded shapes, yet scale poorly. Seeded emulsion polymerization achieves diverse lobed or Janus morphologies via phase separation but depends strongly on polymer compatibility. Mechanical deformation reshapes spherical feedstock into rods, discs, or polyhedra using simple protocols but suffers from strain heterogeneity. Microfluidic photopolymerization produces monodisperse extruded or high-curvature particles but requires chip parallelization for scale-up. Vapor-based processes and CDP yield flattened or hemispherical shapes with minimal solvent use but require specialty reactors, while electrospraying converts the field force into shape anisotropy across hydrogel or thermoplastic systems.
Across methods, the attainable particle size is ultimately set by the specific length-scale bottleneck that creates and then preserves anisotropy. Templated lithography/imprint is constrained by mask or mold feature size and by release/harvest fidelity; mask-defined photolithography readily targets micron-scale features. At the same time, nanomolding can push to sub-100-nm features but is limited by rounding and feature fidelity at the smallest dimensions.22,36 Microfluidic methods are constrained by channel geometry and the droplet/confinement regimes that remain monodisperse, as particle dimensions track the channel width/height and the breakup windows, rather than being freely selectable.95,98,107 Seeded growth/seeded emulsion routes are constrained by the seed size and by swelling/monomer transport through (and compatibility with) the seed network; sufficiently small or highly constrained seeds can suppress the phase separation needed for anisotropy, and practical diffusion/annealing windows bound what is realizable.38,43,55,57 By contrast, mechanical deformation/reshaping is constrained by Tg-dependent chain mobility and strain homogeneity, as the shape-setting step requires softening above Tg or solvent plasticization and is additionally bounded by the available starting sphere sizes and handling/recovery limits.79,87,92 Accordingly, the demonstrated size windows and their limiting mechanisms are best interpreted together (and are tabulated as such) rather than treated as independent descriptors.22,36,38,79,95
At larger production scales, the dominant constraint is usually how effectively a method can be parallelized and how robustly particles can be harvested/collected, not simply the curing/polymerization chemistry. In templated approaches (lithography, imprinting, and related mold-replication), output scales most directly with patterned area (mask/mold/wafer size) and cycle repetition (fill-cure-release).25,32,36 Roll-to-roll mold templating (e.g., PRINT-style micromolding) may be well-positioned for scale-up because it replaces “one exposure at a time” with continuous cavity filling and repeated harvesting.29,35 Flow-based platforms are continuous, but the output is typically limited by device multiplexing (number of channels/emitters or illumination lanes) and downstream handling.98,101,103,107 Similar issues exist for continuous-flow and stop-flow lithographic platforms. Nevertheless, a productivity of multi-million particles per hour in multi-channel implementations has been reported.108,114 In summary, methods that are easiest to parallelize in hardware and easiest to harvest cleanly may be the most promising candidates for large-scale production.25,29,32,36,101,107,108
Future advancements hinge on four imperatives: standardization, sustainability, scalability, and use-inspired demonstration of emergent properties for the shaped particles. Reporting shape yield, throughput normalized by device area or polymer mass, and energy or solvent consumption will enable direct comparison between methods. Sustainable chemistries, from solvent-free polymerization to recyclable elastomers, and to PFAS-free molds, will improve process viability and scalability. Semi-continuous processes such as roll-to-roll PRINT or chemical vapor deposition hint that shape-engineered polymer particles are approaching market readiness. In situ, AI-supervised synthesis could soon automate parameter selection and deliver on-demand geometries. Finally, translating these shape libraries into real-world testing will reveal where geometry delivers tangible performance gains. For example, ellipsoid and discoidal are often favored in circulation and cellular uptake studies, due to their margination under flow (to prolong circulation time) and differential clearance times by specific immune cells.133,134 Hemispherical, concave, or convex shapes resemble optical lenses and can enable future optical metamaterials.127 The shapes of Janus and lobed/clustered particles can be leveraged to direct self-assembly, giving rise to emergent properties like superhydrophobicity or modulated reflectivity.135,136 Polyhedral and prismatic particles may assemble into materials with programmable photonic band gaps.137 While concave forms like bowls can enclose and/or transport cargo, extruded shapes like rods, plugs, or filaments have served as building blocks for higher-order assembly or interlocking interfaces for targeted combinations of mechanical, interfacial, and bulk properties.138,139 The future of shape-engineered polymer particles lies in their ability to be implemented in real-world applications. For example, tissue repair constructs that utilize particle-laden hydrogels may benefit from the anisotropic packing of rod particles for greater integration and faster tissue regeneration.140
In the near term, commercial adoption will likely favor single-shape, high-yield systems. Hybrid workflows that merge complementary mechanisms, e.g., microfluidic pre-shaping followed by vapor relaxation, could offer both precision and scalability. Ultimately, geometry will join chemistry and size as a core design variable in polymer particle engineering, reshaping the material logic of applications spanning healthcare, consumer goods, and structural composites.
Author contributions
JDJ conducted the literature search. EL and JDJ created the figures. JDJ and RY composed and revised the manuscript. RY directed the study.Conflicts of interest
R. Y. holds equity in Oto Biotech. EL and JDJ have no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
This work was supported in part by the National Science Foundation Faculty Early Career Development Program [grant number CMMI-2144171]. J. J. is supported by the NSF Graduate Research Fellowship Program (GRFP) under Grant No. DGE-2139899 and the Cornell Engineering Lance R. Collins Fellowship. E. L. is supported by the Cornell Engineering Learning Initiatives Undergraduate Research Fund.References
- American Chemical Society National Historic Chemical Landmarks. U.S. Synthetic Rubber Program.
- D. Foord, Enterp. Soc., 2023, 24, 395–424 CrossRef.
- M. Antonietti and K. Tauer, Macromol. Chem. Phys., 2003, 204, 207–219 CrossRef CAS.
- P. A. Lovell and F. J. Schork, Biomacromolecules, 2020, 21, 4396–4441 CrossRef CAS PubMed.
- D. N. Petsev, N. D. Denkov and P. A. Kralchevsky, J. Colloid Interface Sci., 1995, 176, 201–213 CrossRef CAS.
- P. Ahlin Grabnar and J. Kristl, J. Microencapsul., 2011, 28, 323–335 CrossRef PubMed.
- A. D. Bangham, M. M. Standish and J. C. Watkins, J. Mol. Biol., 1965, 13, 238-IN27 Search PubMed.
- K. K. Jain, in Drug Delivery Systems, ed K. K. Jain, Springer New York, New York, NY, 2020, vol. 2059, pp. 1–54 Search PubMed.
- A. Sultana, M. Zare, V. Thomas, T. S. S. Kumar and S. Ramakrishna, Med. Drug Discovery, 2022, 15, 100134 CrossRef CAS.
- S. Senapati, A. K. Mahanta, S. Kumar and P. Maiti, Signal Transduction Targeted Ther., 2018, 3, 7 CrossRef PubMed.
- N. Deirram, C. Zhang, S. S. Kermaniyan, A. P. R. Johnston and G. K. Such, Macromol. Rapid Commun., 2019, 40, 1800917 CrossRef PubMed.
- S. R. Sershen, S. L. Westcott, N. J. Halas and J. L. West, J. Biomed. Mater. Res., 2000, 51, 293–298 CrossRef CAS PubMed.
- A. Chilkoti, M. R. Dreher, D. E. Meyer and D. Raucher, Adv. Drug Delivery Rev., 2002, 54, 613–630 CrossRef CAS PubMed.
- O. Pillai and R. Panchagnula, Curr. Opin. Chem. Biol., 2001, 5, 447–451 CrossRef CAS PubMed.
- P. J. Gawne, M. Ferreira, M. Papaluca, J. Grimm and P. Decuzzi, Nat. Rev. Mater., 2023, 8, 783–798 CrossRef PubMed.
- V. Kozlovskaya, M. Dolmat and E. Kharlampieva, ACS Appl. Polym. Mater., 2021, 3, 2274–2289 CrossRef CAS.
- H. Hadji and K. Bouchemal, J. Controlled Release, 2022, 342, 93–110 CrossRef CAS PubMed.
- N. Kapate, J. R. Clegg and S. Mitragotri, Adv. Drug Delivery Rev., 2021, 177, 113807 CrossRef CAS PubMed.
- P. Li, J. Zhang, X. Liu, X. Zhang, J. Ma, G. Sun, L. Hou and S. Raza, Polym. Chem., 2025, 16, 774–799 RSC.
- D. Jiang, S. Liu and W. Tang, Micromachines, 2022, 13, 1659 CrossRef PubMed.
- P. Lagarrigue, F. Moncalvo and F. Cellesi, Pharmaceutics, 2022, 15, 32 CrossRef PubMed.
- R. K. Pal, N. E. Kurland, C. Jiang, S. C. Kundu, N. Zhang and V. K. Yadavalli, Eur. Polym. J., 2016, 85, 421–430 CrossRef CAS.
- B. Li, M. He, L. Ramirez, J. George and J. Wang, ACS Appl. Mater. Interfaces, 2016, 8, 4158–4164 CrossRef CAS PubMed.
- C. Lu and R. H. Lipson, Laser Photonics Rev., 2010, 4, 568–580 CrossRef CAS.
- J.-H. Jang, C. K. Ullal, S. E. Kooi, C. Koh and E. L. Thomas, Nano Lett., 2007, 7, 647–651 CrossRef CAS PubMed.
- L. E. Roode, H. Brighton, T. Bo, J. L. Perry, M. C. Parrott, F. Kersey, J. C. Luft, J. E. Bear, J. M. DeSimone and I. J. Davis, Nanomed. Nanotechnol. Biol. Med., 2016, 12, 1053–1062 CrossRef CAS PubMed.
- D. A. Canelas, K. P. Herlihy and J. M. DeSimone, Wiley Interdiscip. Rev.: Nanomed. Nanobiotechnol., 2009, 1, 391–404 CAS.
- K. B. Wiles, N. S. Wiles, K. P. Herlihy, B. W. Maynor, J. P. Rolland and J. M. DeSimone, in Soft lithography using perfluorinated polyether molds and PRINT technology for fabrication of 3-D arrays on glass substrates, ed M. J. Lercel, San Jose, CA, 2006, p. 61513F Search PubMed.
- A. Garcia, P. Mack, S. Williams, C. Fromen, T. Shen, J. Tully, J. Pillai, P. Kuehl, M. Napier, J. M. DeSimone and B. W. Maynor, J. Drug Delivery, 2012, 2012, 1–10 CrossRef PubMed.
- S. E. A. Gratton, P. D. Pohlhaus, J. Lee, J. Guo, M. J. Cho and J. M. DeSimone, J. Controlled Release, 2007, 121, 10–18 CrossRef CAS PubMed.
- V. Kudryavtseva, A. Bukatin, E. Vyacheslavova, D. Gould and G. B. Sukhorukov, Biomater. Adv., 2022, 136, 212762 CrossRef CAS PubMed.
- F. Buyukserin, M. Aryal, J. Gao and W. Hu, Small, 2009, 5, 1632–1636 CrossRef CAS PubMed.
- B. Xue, V. Kozlovskaya and E. Kharlampieva, J. Mater. Chem. B, 2017, 5, 9–35 RSC.
- N. Higuita-Castro, D. Gallego-Perez, K. Love, M. R. Sands, G. Kaletunç and D. J. Hansford, Ind. Biotechnol., 2012, 8, 365–371 CrossRef CAS.
- Y. Wang, Y. Wang, X. Zheng, G.-R. Yi, S. Sacanna, D. J. Pine and M. Weck, J. Am. Chem. Soc., 2014, 136, 6866–6869 CrossRef CAS PubMed.
- F. Caruso, R. A. Caruso and H. Möhwald, Science, 1998, 282, 1111–1114 CrossRef CAS PubMed.
- J. L. Perry, K. P. Herlihy, M. E. Napier and J. M. DeSimone, Acc. Chem. Res., 2011, 44, 990–998 CrossRef CAS PubMed.
- L. C. Glangchai, M. Caldorera-Moore, L. Shi and K. Roy, J. Controlled Release, 2008, 125, 263–272 CrossRef CAS PubMed.
- G. Zhou, S. Liu, W. Liu, D. Yuan and G. Zhou, Micromachines, 2019, 10, 441 CrossRef PubMed.
- J.-W. Kim and K.-D. Suh, J. Ind. Eng. Chem., 2008, 14, 1–9 CrossRef CAS.
- N. Sudjaipraparat, C. Kaewsaneha, S. Nuasaen and P. Tangboriboonrat, Polymer, 2017, 121, 165–172 CrossRef CAS.
- X. Pei, K. Zhai, X. Liang, Y. Deng, K. Xu, Y. Tan, X. Yao and P. Wang, J. Colloid Interface Sci., 2018, 512, 600–608 CrossRef CAS PubMed.
- M. Yang, Y. Guo, Q. Wu, Y. Luan and G. Wang, Polymer, 2014, 55, 1948–1954 CrossRef CAS.
- Y.-S. Cho, S.-H. Kim and J. H. Moon, Korean J. Chem. Eng., 2012, 29, 1102–1107 CrossRef CAS.
- H. R. Sheu, M. S. El-Aasser and J. W. Vanderhoff, J. Polym. Sci., Part A: Polym. Chem., 1990, 28, 653–667 CrossRef CAS.
- C. Wei, A. Plucinski, S. Nuasaen, A. Tripathi, P. Tangboriboonrat and K. Tauer, Macromolecules, 2017, 50, 349–363 CrossRef CAS.
- S. Wang, Y. Long, H. Liu and F. Liang, Macromolecules, 2025, 58, 5927–5936 CrossRef CAS.
- S. Jia, H. Wang, R. Tang, S. Ma, B. Gong and J. Ou, Polymer, 2023, 265, 125604 CrossRef CAS.
- B. Peng, H. R. Vutukuri, A. Van Blaaderen and A. Imhof, J. Mater. Chem., 2012, 22, 21893 RSC.
- X. Meng and D. Qiu, Polymer, 2020, 203, 122799 CrossRef CAS.
- S. Wang and N. Wu, Langmuir, 2015, 31, 7962–7969 CrossRef CAS PubMed.
- E. Y. K. Fung, K. Muangnapoh and C. M. Liddell Watson, J. Mater. Chem., 2012, 22, 10507 RSC.
- B. Peng and A. Imhof, Soft Matter, 2015, 11, 3589–3598 RSC.
- B. Peng, A. Van Blaaderen and A. Imhof, ACS Appl. Mater. Interfaces, 2013, 5, 4277–4284 CrossRef CAS PubMed.
- Y. Li, F. Liu, S. Demirci, U. K. Dey, T. Rawah, A. Chaudary, R. Ortega, Z. Yang, E. Pirhadi, B. Huang, X. Yong and S. Jiang, Nanoscale, 2025, 17, 88–112 RSC.
- F. Wang, H. Liu, L. Zhao and X. Zhang, Colloid Polym. Sci., 2014, 292, 1171–1179 CrossRef CAS.
- Y.-C. Lin, A. K. Tripathi and J. G. Tsavalas, ACS Appl. Polym. Mater., 2022, 4, 313–326 CrossRef CAS.
- L. Tian, X. Li, P. Zhao, Z. Ali and Q. Zhang, Macromolecules, 2016, 49, 9626–9636 CrossRef CAS.
- G. Russo and M. Lattuada, J. Colloid Interface Sci., 2022, 611, 377–389 CrossRef CAS PubMed.
- Y. Saadat, S. Hosseinzadeh, S. Abdolbaghi and F. Afshar-Taromi, Colloid Polym. Sci., 2013, 291, 1113–1120 CrossRef CAS.
- J.-W. Kim, R. J. Larsen and D. A. Weitz, Adv. Mater., 2007, 19, 2005–2009 CrossRef CAS.
- S. Hosseinzadeh and Y. Saadat, RSC Adv., 2015, 5, 35325–35337 RSC.
- Y. Saadat, S. Hosseinzadeh, F. Afshar-Taromi and H. Eslami, Colloid Polym. Sci., 2012, 290, 1099–1106 CrossRef CAS.
- J.-B. Fan, H. Liu, Y. Song, Z. Luo, Z. Lu and S. Wang, Macromolecules, 2018, 51, 1591–1597 CrossRef CAS.
- N. Konishi, T. Fujibayashi, T. Tanaka, H. Minami and M. Okubo, Polym. J., 2010, 42, 66–71 CrossRef CAS.
- H. Zou, D. Miao, H. Sun and X. Wang, Langmuir, 2018, 34, 14302–14308 CrossRef CAS PubMed.
- H. Zou, D. Miao, H. Sun and X. Wang, Langmuir, 2018, 34, 14302–14308 CrossRef CAS PubMed.
- Y. Sun, F. Liang, X. Qu, Q. Wang and Z. Yang, Macromolecules, 2015, 48, 2715–2722 CrossRef CAS.
- C. Li, Y. Yu, L. Wang, S. Zhang, J. Liu, J. Zhang, A.-B. Xu, Z. Wu, J. Tong, S. Wang, M. Xiao, Y. Fang, J. Yao, A. A. Solovev, B. Dong and L. He, Chem. Mater., 2019, 31, 9513–9521 CrossRef CAS.
- S. Sacanna, W. T. M. Irvine, P. M. Chaikin and D. J. Pine, Nature, 2010, 464, 575–578 CrossRef CAS PubMed.
- Y. Wang, Y. Wang, X. Zheng, G.-R. Yi, S. Sacanna, D. J. Pine and M. Weck, J. Am. Chem. Soc., 2014, 136, 6866–6869 CrossRef CAS PubMed.
- H. Adelnia, J. N. Gavgani and M. Soheilmoghaddam, Colloid Polym. Sci., 2015, 293, 2445–2450 CrossRef CAS.
- X. Xie, Z. Ling, D. Li and H. Zou, Langmuir, 2025, 41, 19914–19923 CrossRef CAS PubMed.
- J. Liao, C. Zhu, Z. He, J. Zhang, Y. Zeng and Z. Gu, Langmuir, 2022, 38, 12132–12139 CrossRef CAS PubMed.
- Y. Jiang, L. Zhao, R. Li and H. Zou, Langmuir, 2024, 40, 25811–25821 CrossRef CAS PubMed.
- W.-H. Chen, F. Tu, L. C. Bradley and D. Lee, Chem. Mater., 2017, 29, 2685–2688 CrossRef CAS.
- A. Namjoo, S. Abdolbaghi, Y. Saadat and S. Hosseinzadeh, Colloid Polym. Sci., 2015, 293, 1781–1789 CrossRef CAS.
- N.-H. Park, S.-I. Park and K.-D. Suh, Colloid Polym. Sci., 2001, 279, 1082–1089 CrossRef CAS.
- Z. Gong, T. Hueckel, G.-R. Yi and S. Sacanna, Nature, 2017, 550, 234–238 CrossRef PubMed.
- Y. Wang, J. T. McGinley and J. C. Crocker, Langmuir, 2017, 33, 3080–3087 CrossRef CAS PubMed.
- E. Ben-Akiva, K. R. Rhodes, R. A. Meyer and J. J. Green, J. Vis. Exp., 2018, 58332 Search PubMed.
- M. J. Heslinga, E. M. Mastria and O. Eniola-Adefeso, J. Controlled Release, 2009, 138, 235–242 CrossRef CAS PubMed.
- W. Li, T. Suzuki and H. Minami, Angew. Chem., Int. Ed., 2018, 57, 9936–9940 CrossRef CAS PubMed.
- M. K. Klein, N. R. Saenger, S. Schuetter, P. Pfleiderer and A. Zumbusch, Langmuir, 2014, 30, 12457–12464 CrossRef CAS PubMed.
- W. Tuntanatewin, P. Mekwatanakarn, H. Zhang and Y. Okamura, J. Appl. Polym. Sci., 2021, 138, 49798 CrossRef CAS.
- A. R. Bhide and A. B. Jindal, Int. J. Pharm., 2021, 605, 120820 CrossRef CAS PubMed.
- Q. Guo, C. J. Bishop, R. A. Meyer, D. R. Wilson, L. Olasov, D. E. Schlesinger, P. T. Mather, J. B. Spicer, J. H. Elisseeff and J. J. Green, ACS Appl. Mater. Interfaces, 2018, 10, 13333–13341 CrossRef CAS PubMed.
- F. Zhang, T. Zhao, D. Ruiz-Molina, Y. Liu, C. Roscini, J. Leng and S. K. Smoukov, ACS Appl. Mater. Interfaces, 2020, 12, 47059–47064 CrossRef CAS PubMed.
- J. A. Champion, Y. K. Katare and S. Mitragotri, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 11901–11904 CrossRef CAS PubMed.
- R. A. Meyer, R. S. Meyer and J. J. Green, J. Biomed. Mater. Res. A, 2015, 103, 2747–2757 CrossRef CAS PubMed.
- Y. Hu, J. Ge, T. Zhang and Y. Yin, Adv. Mater., 2008, 20, 4599–4602 CrossRef CAS.
- C. Palazzo, G. Ponchel, J. J. Vachon, S. Villebrun, F. Agnely and C. Vauthier, Int. J. Polym. Mater. Polym. Biomater., 2017, 66, 416–424 CrossRef CAS.
- S. J. Ahn, K. H. Ahn and S. J. Lee, Colloid Polym. Sci., 2016, 294, 859–867 CrossRef CAS.
- H. Zhang, W. Tuntanatewin, K. Ishikura, D. Sogabe, K. Sugawara, A. Tokui, A. Nakagawa and Y. Okamura, ACS Appl. Polym. Mater., 2020, 2, 3355–3364 CrossRef CAS.
- H. Minami, A. Kojima and T. Suzuki, Langmuir, 2017, 33, 1541–1546 CrossRef CAS PubMed.
- Z. Q. Sun, X. Chen, J. H. Zhang, Z. M. Chen, K. Zhang, X. Yan, Y. F. Wang, W. Z. Yu and B. Yang, Langmuir, 2005, 21, 8987–8991 CrossRef CAS PubMed.
- D. Dendukuri, K. Tsoi, T. A. Hatton and P. S. Doyle, Langmuir, 2005, 21, 2113–2116 CrossRef CAS PubMed.
- J. Kim and S. A. Vanapalli, Langmuir, 2013, 29, 12307–12316 CrossRef CAS PubMed.
- I. Kobayashi, M. A. Neves, T. Yokota, K. Uemura and M. Nakajima, Ind. Eng. Chem. Res., 2009, 48, 8848–8855 CrossRef CAS.
- M. Seo, Z. Nie, S. Xu, M. Mok, P. C. Lewis, R. Graham and E. Kumacheva, Langmuir, 2005, 21, 11614–11622 CrossRef CAS PubMed.
- S.-H. Kim, A. Abbaspourrad and D. A. Weitz, J. Am. Chem. Soc., 2011, 133, 5516–5524 CrossRef CAS PubMed.
- C.-H. Chen, R. K. Shah, A. R. Abate and D. A. Weitz, Langmuir, 2009, 25, 4320–4323 CrossRef CAS PubMed.
- T. Nisisako and T. Torii, Adv. Mater., 2007, 19, 1489–1493 CrossRef CAS.
- H.-Y. Yang, M.-J. Zhang, X.-H. Guo, Y. Deng, P. Zhang, L.-D. Qiu, W. Wang and L.-Y. Chu, Langmuir, 2025, 41, 30621–30632 CrossRef CAS PubMed.
- T. Nisisako and T. Hatsuzawa, Microfluid. Nanofluidics, 2010, 9, 427–437 CrossRef.
- D. C. Pregibon, M. Toner and P. S. Doyle, Science, 2007, 315, 1393–1396 CrossRef CAS PubMed.
- K. Feng, N. Gao, W. Zhang, K. Zhou, H. Dong, P. Wang, L. Tian, G. He and G. Li, Small, 2020, 16, 1903884 CrossRef CAS PubMed.
- Z. Nie, W. Li, M. Seo, S. Xu and E. Kumacheva, J. Am. Chem. Soc., 2006, 128, 9408–9412 CrossRef CAS PubMed.
- Z. Nie, S. Xu, M. Seo, P. C. Lewis and E. Kumacheva, J. Am. Chem. Soc., 2005, 127, 8058–8063 CrossRef CAS PubMed.
- G. C. Le Goff, J. Lee, A. Gupta, W. A. Hill and P. S. Doyle, Adv. Sci., 2015, 2, 1500149 CrossRef PubMed.
- M. Li, D. Joung, J. A. Kozinski and D. K. Hwang, Langmuir, 2017, 33, 184–190 CrossRef CAS PubMed.
- D. Dendukuri, T. A. Hatton and P. S. Doyle, Langmuir, 2007, 23, 4669–4674 CrossRef CAS PubMed.
- S. K. Suh, K. Yuet, D. K. Hwang, K. W. Bong, P. S. Doyle and T. A. Hatton, J. Am. Chem. Soc., 2012, 134, 7337–7343 CrossRef CAS PubMed.
- D. K. Hwang, J. Oakey, M. Toner, J. A. Arthur, K. S. Anseth, S. Lee, A. Zeiger, K. J. Van Vliet and P. S. Doyle, J. Am. Chem. Soc., 2009, 131, 4499–4504 CrossRef CAS PubMed.
- D. Baah, J. Tigner, K. Bean, N. Walker, B. Britton and T. Floyd-Smith, Microfluid. Nanofluid., 2012, 12, 657–662 CrossRef.
- D. Dendukuri, D. C. Pregibon, J. Collins, T. A. Hatton and P. S. Doyle, Nat. Mater., 2006, 5, 365–369 CrossRef CAS PubMed.
- B. J. Park and E. M. Furst, Langmuir, 2010, 26, 10406–10410 CrossRef CAS PubMed.
- M. Sabapathy, Y. Shelke, M. G. Basavaraj and E. Mani, Soft Matter, 2016, 12, 5950–5958 RSC.
- H.-F. Tseng, Y.-J. Chiu, B.-H. Wu, J.-W. Li and J.-T. Chen, ACS Appl. Nano Mater., 2018, 1, 4557–4565 CrossRef CAS.
- J.-T. Chen, P.-H. Lee, H.-F. Tseng, Y.-J. Chiu, Y.-H. Kao, K.-S. Jeng, C.-T. Liu and C.-C. Tsai, ACS Macro Lett., 2015, 4, 721–724 CrossRef CAS PubMed.
- H. Tseng, M. Cheng, K. Jeng, J. Li and J. Chen, Macromol. Rapid Commun., 2016, 37, 1825–1831 CrossRef CAS PubMed.
- L. Zheng, P. Huang, L. Zhang, D. Guo and Q. Yan, Part. Part. Syst. Charact., 2016, 33, 842–850 CrossRef CAS.
- H.-F. Tseng, M.-H. Cheng, J.-W. Li and J.-T. Chen, Macromolecules, 2017, 50, 5114–5121 CrossRef CAS.
- T. Franklin, D. L. Streever and R. Yang, Chem. Mater., 2022, 34, 5960–5970 CrossRef CAS.
- J. James and R. Yang, Org. Mater., 2023, 5(2), 148–157 CrossRef.
- T. Franklin and R. Yang, Chem. Mater., 2023, 35, 4955–4964 CrossRef CAS.
- K. Park, S. S. Liu, W. Tang and R. Yang, Adv. Mater., 2025, 37, 2419893 CrossRef CAS PubMed.
- P. Meakin, Phys. Scr., 1992, 1992, 31 CrossRef.
- H. Lee, S. An, S. Kim, B. Jeon, M. Kim and I. S. Kim, Sci. Rep., 2018, 8, 15725 CrossRef PubMed.
- J. Xu, K. Li, M. Liu, X. Gu, P. Li and Y. Fan, Eur. Polym. J., 2021, 148, 110372 CrossRef CAS.
- M. Jeyhani, S. Y. Mak, S. Sammut, H. C. Shum, D. K. Hwang and S. S. H. Tsai, Chem. Phys. Chem., 2018, 19, 2113–2118 CrossRef CAS PubMed.
- N. Doshi, A. S. Zahr, S. Bhaskar, J. Lahann and S. Mitragotri, Proc. Natl. Acad. Sci. U. S. A., 2009, 106, 21495–21499 CrossRef CAS PubMed.
- A. Dasgupta, T. Sun, R. Palomba, E. Rama, Y. Zhang, C. Power, D. Moeckel, M. Liu, A. Sarode, M. Weiler, A. Motta, C. Porte, Z. Magnuska, A. S. Elshafei, R. Barmin, A. Graham, A. McClelland, D. Rommel, E. Stickeler, F. Kiessling, R. M. Pallares, L. D. Laporte, P. Decuzzi, N. McDannold, S. Mitragotri and T. Lammers, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2218847120 CrossRef CAS PubMed.
- J. A. Champion and S. Mitragotri, Pharm. Res., 2009, 26, 244–249 CrossRef CAS PubMed.
- H. Yang, F. Liang, Y. Chen, Q. Wang, X. Qu and Z. Yang, NPG Asia Mater., 2015, 7, e176–e176 CrossRef CAS.
- L. Tian, X. Li, P. Zhao, Z. Ali and Q. Zhang, Macromolecules, 2016, 49, 9626–9636 CrossRef CAS.
- V. L. Colvin, MRS Bull., 2001, 26, 637–641 CrossRef CAS.
- H. Sun, Y. Gao, Y. Fan, J. Du, J. Jiang and C. Gao, Macromol. Rapid Commun., 2023, 44, 2300196 CrossRef CAS PubMed.
- J. E. Melzer and E. McLeod, Microsyst. Nanoeng., 2021, 7, 45 CrossRef PubMed.
- T. H. Qazi, J. Wu, V. G. Muir, S. Weintraub, S. E. Gullbrand, D. Lee, D. Issadore and J. A. Burdick, Adv. Mater., 2022, 34, 2109194 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |



