Open Access Article
Open Access ArticleChemoselective synthesis of α-fluoromethyl amides via the controlled addition of LiCH2F to N-aryl and N-alkyl isocyanates†
Davide
Castiglione
 a,
Alberto
Nardi
a,
Alberto
Nardi
 a,
Margherita
Miele
a,
Margherita
Miele
 a,
Laura
Castoldi
a,
Laura
Castoldi
 *b and
Vittorio
Pace
*b and
Vittorio
Pace
 *cd
*cd
aUniversity of Turin – Department of Chemistry, Via Giuria 7, 10125, Turin, Italy
bUniversity of Milan – Department of Pharmaceutical Sciences, General and Organic Chemisty Section “A. Marchesini” – Via Venezian 21, 20133, Milan, Italy. E-mail: laura.castoldi@unimi.it
cUniversity of Rome “La Sapienza” – Department of Chemistry, P.le A. Moro 5, 00185, Rome, Italy. E-mail: vittorio.pace@univie.ac.at; vittorio.pace@uniroma1.it
dUniversity of Vienna, – Department of Pharmaceutical Sciences, Division of Pharmaceutical Chemistry, Josef-Holaubek-Platz 2, 1090, Vienna, Austria
First published on 5th February 2026
Abstract
α-Fluoromethyl amides were prepared through the chemoselective nucleophilic addition of fluoromethyl-lithium to isocyanates.
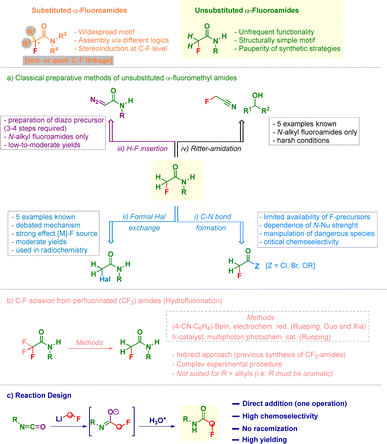
Forging the amide bond represents a fundamental operation in synthesis due to its ubiquitous presence in chemistry.1 The additional installation of a fluorine atom at the carbonyl vicinal position is a strategically convenient modulation of the chemical environment, while inducing minimal (steric) perturbation to the resulting cluster.2 This effect arises from the unique constitutional features of the fluorine atom, including comparable radius (1.47 A vs. 1.21 A for hydrogen)3 and length of C–H and C–F bonds.4 The continuous demand from interlinked fields5 for straightforward approaches enabling the assembly of α-monofluoroamides emerged as a remarkable endeavor due to the limited flexibility of α-functionalization techniques available for carbonyl motifs.6 Notwithstanding, the α-fluorination of amides can be now realized through different approaches leveraging on the construction of the C–F bond through the release—to an adequate amide precursor—of the halogen in both electrophilic7 and nucleophilic8 regimes. Upon the judicious control of the reaction conditions a high degree of stereocontrol can be imparted to α-fluorinative carbonyl processes, as showcased by Lectka,9 Rovis10 and Sun.11 The construction of α-fluoro-α-substituted amides can also be levered on rearrangement events starting from simple feedstocks. In this regard, Hu introduced a difluorocarbene precursor for merging sequential C–F and amide bond formations via a fluorinative-aminocarbonylation of aldehydes.12 Also, the groups of Lan and Song13 and Degennaro adapted the Meinwald transposition on fluorinated oxiranes for making fluoroamides.14 Despite these advancements—conducting to secondary and tertiary C–F linkages—there is a dearth of protocols allowing the gathering of –CH2F containing amides which, despite the relative structural simplicity, have not been thoroughly studied (Scheme 1 – top).15 The retrosynthetic analysis enables to individuate following tactics: (i) C–N bond construction starting from an amine and a monofluoromethylated carboxylic acid precursor (e.g. Schotten-Baumann procedure);16 (ii) C–F bond formation via nucleophilic substitution;17 (iii) H–F bond insertion into α-diazocarbonyls;18 (iv) tropylium-promoted Ritter-type amidation of alcohols with fluoroacetonitrile (Scheme 1a).19 More recently, the introduction of the hydrodefluorination tactic of perfluorinated carbon atoms (e.g. CF3)—formally relying on a C–F dissociation event-,20 are the basis for preparing α-fluoromethyl amides, as documented in the elegant electrochemical-mediated procedure developed by Rueping, Guo and Xia.21 Indeed, hydrodefluorinations could be implemented under photocatalytic conditions, as documented by Qu and Kang22 and the same group of Rueping (Scheme 1b).23 Unfortunately, the aforementioned protocols are not general and are dictated by the electronic nature [alkyl or (hetero)aryl] displayed by the substituent on nitrogen. As a matter of fact, hydrodefluorination methods are restricted to N-aryl systems,21–23 whereas the Ritter approach addresses uniquely the preparation of aliphatic analogues.19 A series of additional considerations supports the demand for modular methods en route to the targeted α-fluoromethyl amides: (1) using C–N bond formation protocols is limited by the availability of activated fluoromethylated carboxylic derivatives and by the nucleophilicity of the employed amine; (2) the adoption of Finkelstein or hydrodefluorinative strategies are hampered by the requirement of preparing the appropriate (α-bromomethyl or α-trifluoromethyl-) precursor non commercially available; (3) this latter drawback affects also Rueping's-type hydrodefluorinations requiring the previous synthesis of trifluoromethyl-amides precursors.21,23 Herein, is reported the rapid and chemoselective addition of CH2F-nucleophilic element to highly electrophilic isocyanates24 for preparing α-fluoromethyl amides (Scheme 1c). Notably, neither the nature of N-substituents nor the chirality in the starting materials were found detrimental for the generality of this single synthetic operation.
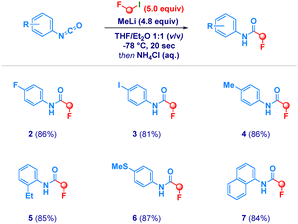
4-Fluorophenylisocyanate (1) was selected as the model substrate in the addition of LiCH2F, generated in situ from fluoroiodomethane25 and MeLi-LiBr (Table 1).26
| Entry | MeLi (equiv.) | Concentrat of 1 (M) | Time (s) | Ratioa2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2a 2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2b 2b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2c 2c |
2 (%)b |
|---|---|---|---|---|---|
| LiCH2F was formed using a slight excess (0.2 equiv. compared to MeLi) of ICH2F to ensure a quantitative I/Li exchange.a Determined by GC-MS analysis of the reaction crude.b Isolated yield.c ICH2F (5.2 equiv.). | |||||
| 1 | MeLi-LiBr (2.8) | 0.2 | 300 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66 66![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23- 23- |
Trace |
| 2 | MeLi-LiBr (2.8) | 0.1 | 300 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 56 56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :- :- |
10 |
| 3 | MeLi-LiBr (2.8) | 0.05 | 300 | 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
24 |
| 4 | MeLi-LiBr (2.8) | 0.05 | 60 | 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7 |
32 |
| 5 | MeLi-LiBr (2.8) | 0.05 | 20 | 53![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
48 |
| 6 | MeLi-LiBr (3.5) | 0.05 | 20 | 61![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
55 |
| 7 | MeLi-LiBr (4.0) | 0.05 | 20 | 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14 |
61 |
| 8 | MeLi-LiBr (4.5) | 0.05 | 20 | 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17 17 |
66 |
| 9 | MeLi-LiBr (5.0) | 0.05 | 20 | 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4:3 4:3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
65 |
| 10c | MeLi (5.0) | 0.05 | 20 | 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :-:-:- :-:-:- |
86 |
| 11 | MeLi (3.5) | 0.05 | 20 | 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :- :- |
64 |
Surprisingly, symmetrical 1,2-diarylurea 2a and 1,3,5-triarylbiuret 2b were isolated as the major products, together with traces of the desired N-(4-fluorophenyl)-fluoroacetamide (2, entry 1). Presumably, the amide anion generated after the carbenoid attack is nucleophilic enough to attack a second equivalent of isocyanate, thus forming (sequentially) 2a and 2b (vide infra for a plausible mechanistic rationale). With the aim of suppressing these di- and tri-merization adducts,27 we reasoned that lowering the initial concentration of the isocyanate 1 from 0.2–0.1 M (entries 1 and 2) to 0.05 M and, encouragingly 2 could be isolated in a promising 24% yield (entry 3). However, parallel to the switch of products distribution towards 2, the fluorine/bromine exchange product 2c was also formed. Aware of the extremely short life of the carbenoid (even at −78 °C)26,28 and, considering that the undesired events could be promoted following the nucleophilic attack, the final acidic quenching with NH4Cl was anticipated (entries 4–11). Thus, if realized just after 20 s from the end of the addition of MeLi-LiBr, the formation of the undesired products could be further mitigated giving 2 in good 48% yield (entry 5). Though the suppression of compounds 2a and 2b could be progressively achieved by employing the carbenoid in excess (3.5–5.0 equiv., entries 6–9) the presence of supra-stoichiometric LiBr (from MeLi-LiBr) boosted C–F to C–Br substitution. Under these circumstances the appearance of bromoamide 2c could not be suppressed, as suggested by the good correlation with the loading of LiBr. Fortunately, the replacement of MeLi-LiBr with its LiBr-free analogue secured the access to fluoroamide 2 in excellent 86% yield (entry 10). The attempt of decreasing the carbenoid loading to 3.5 equiv. increases the formation of 2a and 2b (entry 11), thus recommending a greater excess of nucleophile. It did—however—confirm that increasing the quantity of MeLi mitigates the risk of halogens substitution (entry 11 vs. entry 6).
With the optimal conditions in hand, we then explored the scope of the method (Scheme 2). The protocol demonstrated flexibility and allowed the installation of the α-fluoromethyl fragment onto diverse N-aryl isocyanates. The genuine chemocontrol underpinning the transformation was deduced by placing an iodine atom (3) on the aromatic ring of the isocyanate without observing concomitant C(sp2)–I/Li exchange. Heterocumulenes containing simple alkyl groups were amenable starting materials (4-5), as well as thioether- (6) and naphthyl- (7) containing analogues.
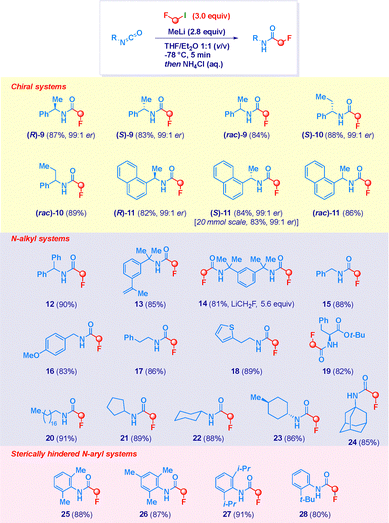
Encouraged by these preliminary results and considering that only scattered examples of chiral α-fluoromethylamides are known,16a,16b,29 we could successfully validate the protocol in the case of the enantiopure isocyanate (R)-8, thus giving the optically active α-fluoromethyl amide (R)-9 in 83% yield and 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 e.r. (Table 2, entry 1). A series of notes are pertinent: (a) the N-alkyl-type substituent—slightly decreasing the electrophilicity of the isocyanate and accounting for a more pronounced steric hindrance in proximity of the (nucleophilic) nitrogen—did not induce the formation of urea and biuret byproducts even with a more favourable stoichiometry (2.8 equiv. of LiCH2F), thus making not mandatory working at very low concentrations (entries 2 and 3); (b) the addition of MeLi completed within 5 min maximized the yield of (R)-9 (entry 4), whereas leaving the reaction for longer time was detrimental; (c) despite the basicity of the environment, the resident chiral center remained intact without detectable racemization.
1 e.r. (Table 2, entry 1). A series of notes are pertinent: (a) the N-alkyl-type substituent—slightly decreasing the electrophilicity of the isocyanate and accounting for a more pronounced steric hindrance in proximity of the (nucleophilic) nitrogen—did not induce the formation of urea and biuret byproducts even with a more favourable stoichiometry (2.8 equiv. of LiCH2F), thus making not mandatory working at very low concentrations (entries 2 and 3); (b) the addition of MeLi completed within 5 min maximized the yield of (R)-9 (entry 4), whereas leaving the reaction for longer time was detrimental; (c) despite the basicity of the environment, the resident chiral center remained intact without detectable racemization.
Having uncovered two sets of conditions for the fluoromethylative homologation of N-aryl ad N-alkyl-isocyanates, we next expanded the scope of the protocol (Scheme 3). The preservation of the optical purity was fully maintained during the nucleophilic addition for preparing the opposite (S)-9 enantiomer, as well as, the enantiopure homologue (S)-10 and both the naphthyl- analogues (R)-11 and (S)-11. Scaling up to 20 mmol was possible without affecting chemical yield or optical purity. It should be noted that the present approach to chiral fluoroamides consisting of a single synthetic operation favourably compares with the only other available based on an acylative/reductive process.16a,16b As expected, chemical yields for racemic counterparts (rac)-9-11 were similar. Benzhydryl isocyanate smoothly reacted to provide the corresponding derivative 12. The increase of the steric hindrance conferred by the benzyl-type substituent on the nitrogen was not detrimental for the addition, as noted in the tert-systems 13 and 14. Moreover, no cyclopropanation of the styryl substituent (13) was promoted by the carbenoid30 which here manifests a true carbanion-like reactivity. The concurrent presence of two isocyanates on the substrate framework was conveniently exploited for forming the bis-(α-fluoromethyamide) 14 by simply adjusting the stoichiometry of LiCH2F (5.6 mmol). Interestingly, in the series of N-alkyl isocyanates the gradual decreasing of the steric hindrance on the C(sp3)–NCO group did not trigger undesirable di- and trimerization processes observable with (more electrophilic) N-aryl analogues when employing the nucleophile in small excess (i.e. 2.8 equiv.). This aspect was evident from the reactivities of isocyanates leading—among others—to benzyl-(15–16) and homobenzyl- (17) type derivatives. Again, acidic C–H protons on a heteroaromatic nucleus (thiophene) were untouched during the reaction, thus forming compound 18. Of note is the fluoromethylation of the essential amino acid derivative—L-phenylalanine tert-butyl ester isocyanate—which occurs chemoselectively at the sp-hybridized carbon, thus making compatible the protocol for functionalizing complex systems (19). Furthermore, both acyclic (20) and cyclic (21–23) alkyl isocyanates promptly reacted to furnish the corresponding α-fluoromethyl amides in comparable chemical yields, again with stereochemical integrity (23). This behaviour was retained also in case of installing the high sterically demanding N-adamant-1-yl group (24). Collectively, the experimental evidence indicates that both the electronic properties and steric factors of the N-substituent play a critical role in determining the selectivity of the process. In this regard, it is instructive the clean and selective formation—with 2.8 equiv. of carbenoid (instead of 5.0 equiv., see Table 1) of—previously unreported—α-fluoromethyl amides 25–27 starting from aryl isocyanates presenting the sterically bulky 2,6-dimethyl-, 2,4,6-trimethyl- and 2,6-di-i-propyl-groups. Though the latter are representatives of the class of reactive aryl-isocyanates, the embodying of sterically demanding elements in critical positions eliminates the risk of generating the aforementioned urea and biuret byproducts. As a matter of fact, the switch from 2-ethylphenyl-(5, Scheme 2) to 2-tert-butylphenyl-(28) substituent on the isocyanate unambiguously illustrates the effect of sterics in the control of the reaction.
The formation of urea and biuret-type byproducts can be rationalized as follows (Scheme S1 – see SI). Upon the attack of LiCH2F to the non-congested aryl isocyanate A, lithium imidate B is generated. In the productive event of α-fluoromethyl amide (2) formation it is quenched with acid (path i) However, this nucleophilic species B might immediately attack a second equivalent of (unreacted) isocyanate, furnishing C which, reiterating the sequence with a third molecule (of isocyanate), gives the biuret precursor E (Scheme S1 – path ii). Both C and E could finally undergo a CO–N cleavage triggered by the intramolecular removal of the acidic fluorine-substituted methylenic proton (see C′ and E′) which eliminating fluoroketene D31 yield 2a and 2b. We believe that upon the removal of the acidic proton, enolate F′ could be obtained; afterwards, it could evolve via ejecting the nitrogen-centered anion (from which the urea or the biuret would be formed) and fluoroketene (Scheme S1 – path iii). Most probably, the weaker C(sp2)–N bond is broken instead of the C(sp2)–F one, as suggested by the BDE.32 A series of evidences are diagnostic for the hypothesized scenario:33 (a) the short-life of the carbenoid—disintegrated immediately after its generation—accounts for leaving in the reaction environment nucleophilic species like B and E which are intrinsically prone to intercept the remaining (not yet reacted) isocyanate; (b) by significantly diluting the mixture, these intermolecular processes are minimized or even precluded; (c) analogously, performing the acidic quenching just after 20 s—compatible with the generation and attack of the carbenoid—would guarantee to stop the sequence at the level of B, thus being productive; (d) this effect is strengthened when using an excess of carbenoid (5.0 equiv.) which maximizes the formation of B, thus decreasing the quantity of unreacted isocyanate; (e) the presence of sterically demanding factors on the isocyanate's nitrogen atom would impede undesired di- and tri-merizations; (f) on the other hand, the tamed electrophilicity of N-alkyl counterparts eliminates per se the risk of these undesired events. The fluoroketene elimination was confirmed by carefully quenching the reaction mixture with NaOEt (after 1 min).
In summa, we introduced a one-step protocol for the preparation of α-fluoromethyl amides via the addition of LiCH2F to isocyanates. The transformation exhibits good chemocontrol and chemical yields—though electron-withdrawing arylisocyanates are not competent substrates - and no racemization occurs on substrates embodying stereochemical information, thus making it primed for introducing CH2F moieties onto sensitive molecular architectures.
Conflicts of interest
The authors declare no competing interests.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental, characterization of products, NMR and HPLC charts. See DOI: https://doi.org/10.1039/d5cc06730h.
Acknowledgements
We thank All4Labels Group (Hamburg, Germany) for generous funding and Quzhou Fluorio Pharmtech Co. (Zhejiang, China) for a gift of fluoroiodomethane. Financial support from PRIN projects n. 20228W9TBL (L. C.) and n. 2022JLSZMY (V. P.), and FWF-Austria Project n. P 37068-B (V. P.) is gratefully acknowledged.References
- (a) A. Greenberg, C. M. Breneman and J. F. Liebman, The Amide Linkage. Selected Structural Aspects in Chemistry, Biochemistry and Materials Science, John Wiley & Sons Inc., New York, 2000 Search PubMed; (b) G. Li, S. Ma and M. Szostak, Trends Chem., 2020, 2, 914 CrossRef CAS.
- (a) D. O'Hagan, Chem. Soc. Rev., 2008, 37, 308 RSC; (b) C. Thiehoff, Y. P. Rey and R. Gilmour, Isr. J. Chem., 2017, 57, 92 CrossRef CAS.
- A. Bondi, J. Phys. Chem., 1964, 68, 441 CrossRef CAS.
- (a) K. Müller, C. Faeh and F. Diederich, Science, 2007, 317, 1881 CrossRef PubMed; (b) N. A. Meanwell, J. Med. Chem., 2018, 61, 5822 CrossRef CAS PubMed; (c) S. Purser, P. R. Moore, S. Swallow and V. Gouverneur, Chem. Soc. Rev., 2008, 37, 320 RSC; (d) J. Wang, M. Sánchez-Roselló, J. L. Aceña, C. del Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432 CrossRef CAS PubMed.
- E. Cosimi, N. Trapp, M.-O. Ebert and H. Wennemers, Chem. Commun., 2019, 55, 2253 Search PubMed.
- (a) S. J. Kalita, J. Qi, L. Xiao, D. Saha, Y.-Y. Huang and N. Shibata, Chem. Rev., 2025, 125, 8477 CrossRef CAS PubMed; (b) Z. Jiao, J. J. Beiger, Y. Jin, S. Ge, J. S. Zhou and J. F. Hartwig, J. Am. Chem. Soc., 2016, 138, 15980 CrossRef CAS PubMed.
- (a) F. Buckingham, A. K. Kirjavainen, S. Forsback, A. Krzyczmonik, T. Keller, I. M. Newington, M. Glaser, S. K. Luthra, O. Solin and V. Gouverneur, Angew. Chem., Int. Ed., 2015, 54, 13366 CrossRef CAS PubMed; (b) S. Y. Lee, S. Neufeind and G. C. Fu, J. Am. Chem. Soc., 2014, 136, 8899 CrossRef CAS PubMed; (c) K. Ishihara, K. Nishimura and K. Yamakawa, Angew. Chem., Int. Ed., 2020, 59, 17641 CrossRef CAS PubMed.
- (a) P. Adler, C. J. Teskey, D. Kaiser, M. Holy, H. H. Sitte and N. Maulide, Nat. Chem., 2019, 11, 329 CrossRef CAS PubMed; (b) H.-G. Cheng and G. Yin, Chemistry, 2019, 5, 1022 CrossRef CAS.
- D. H. Paull, M. T. Scerba, E. Alden-Danforth, L. R. Widger and T. Lectka, J. Am. Chem. Soc., 2008, 130, 17260 CrossRef CAS PubMed.
- P. Wheeler, H. U. Vora and T. Rovis, Chem. Sci., 2013, 4, 1674 RSC.
- (a) X. Dong, W. Yang, W. Hu and J. Sun, Angew. Chem., Int. Ed., 2015, 54, 660 CrossRef CAS PubMed; (b) B. Wang, S. Fan, C. Zhang and J. Sun, J. Am. Chem. Soc., 2025, 147, 10059 CrossRef CAS PubMed.
- (a) A. Liu, C. Ni, Q. Xie and J. Hu, Angew. Chem., Int. Ed., 2022, 61, e202115467 Search PubMed For important recent works on difluorocarbenes, see: ; (b) X. Ma and Q. Song, Chem. Soc. Rev., 2020, 49, 9197 Search PubMed; (c) F.-L. Qing, X.-Y. Liu, J.-A. Ma, Q. Shen, Q. Song and P. Tang, CCS Chem., 2022, 4, 2518 CrossRef CAS; (d) H.-J. Ai, X. Ma, Q. Song and X.-F. Wu, Sci. China: Chem., 2021, 64, 1630 CrossRef CAS.
- G. Zhang, Q. Shi, M. Hou, K. Yang, S. Wang, S. Wang, W. Li, C. Li, J. Qiu, H. Xu, L. Zhou, C. Wang, S.-J. Li, Y. Lan and Q. Song, CCS Chem., 2022, 4, 1671 CrossRef CAS.
- (a) G. De Santis, A. Maranzana, F. Lauria, M. Spennacchio, M. Andresini, R. Purgatorio, M. Colella, R. Luisi and L. Degennaro, Adv. Synth. Catal., 2025, 367, e70083 CrossRef CAS; (b) A. O. Amanetoullah, M. M. Chaabouni and A. Baklouti, Synth. Commun., 1996, 26, 1155 CrossRef CAS.
- (a) A. Fantinati, V. Zanirato, P. Marchetti and C. Trapella, ChemistryOpen, 2020, 9, 100 CrossRef CAS PubMed; (b) R. Britton, V. Gouverneur, J.-H. Lin, M. Meanwell, C. Ni, G. Pupo, J.-C. Xiao and J. Hu, Nat. Rev. Methods Primers, 2021, 1, 47 CrossRef CAS; (c) T. W. Butcher, W. M. Amberg and J. F. Hartwig, Angew. Chem., Int. Ed., 2022, 61, e202112251 Search PubMed; (d) M. Reichel and K. Karaghiosoff, Angew. Chem., Int. Ed., 2020, 59, 12268 CrossRef CAS PubMed; (e) A. Singh, H. Sindhe, A. Kamble, K. Rajkumar, V. Agrawal and S. Sharma, Org. Chem. Front., 2024, 11, 5605 RSC.
- (a) K. Aoki and K. Koga, Chem. Pharm. Bull., 2000, 48, 571 CrossRef CAS PubMed; (b) A. Wzorek, A. Kamizela, A. Sato and V. A. Soloshonok, J. Fluorine Chem., 2017, 196, 37 CrossRef CAS; (c) C. Dubiella, R. Baur, H. Cui, E. M. Huber and M. Groll, Angew. Chem., Int. Ed., 2015, 54, 15888 CrossRef CAS PubMed.
- (a) S. Mizuta, K. Kitamura, A. Kitagawa, T. Yamaguchi and T. Ishikawa, Chem. – Eur. J., 2021, 27, 5930 CrossRef CAS PubMed; (b) K. Gong, Z. Yin, P. Song, B. Xu and J. Han, Synlett, 2024, 1569 CAS; (c) T. Nishikata, S. Ishida and R. Fujimoto, Angew. Chem., Int. Ed., 2016, 55, 10008 CrossRef CAS PubMed; (d) J. S. Wright, R. Ma, E. W. Webb, W. P. Winton, J. Stauff, K. Cheng, A. F. Brooks, M. S. Sanford and P. J. H. Scott, Angew. Chem., Int. Ed., 2024, 63, e202316365 CrossRef CAS PubMed.
- (a) L. S. Munaretto, R. D. C. Gallo, L. P. M. O. Leão and I. D. Jurberg, Org. Biomol. Chem., 2022, 20, 6178 RSC; (b) E. E. Gray, M. K. Nielsen, K. A. Choquette, J. A. Kalow, T. J. A. Graham and A. G. Doyle, J. Am. Chem. Soc., 2016, 138, 10802 CrossRef CAS PubMed.
- S. H. Doan, M. A. Hussein and T. V. Nguyen, Chem. Commun., 2021, 57, 8901 RSC.
- Y.-J. Yu, F.-L. Zhang, T.-Y. Peng, C.-L. Wang, J. Cheng, C. Chen, K. N. Houk and Y.-F. Wang, Science, 2021, 371, 1232 CrossRef CAS PubMed.
- Z.-J. Shen, C. Zhu, X. Zhang, C. Yang, M. Rueping, L. Guo and W. Xia, Angew. Chem., Int. Ed., 2023, 62, e202217244 Search PubMed.
- H. Zhang, J.-X. Chen, J.-P. Qu and Y.-B. Kang, Nature, 2024, 635, 610 CrossRef CAS PubMed.
- J. Jia, K. Murugesan, C. Zhu, H. Yue, S.-C. Lee and M. Rueping, Chin. Chem. Lett., 2025, 36, 109866 CrossRef CAS.
- (a) V. Pace, S. Monticelli, K. de la Vega-Hernandez and L. Castoldi, Org. Biomol. Chem., 2016, 14, 7848 Search PubMed; (b) H. Ulrich, Chemistry and Technology of Isocyanates, Wiley, New York, 1996; Also non fluorinated carbenoids adds to isocyanates. However, no urea- and bi-urets are formed, presumably due to the higher stability of these carbenoids. See Search PubMed; (c) V. Pace, L. Castoldi and W. Holzer, Chem. Commun., 2013, 49, 8383 Search PubMed; (d) V. Pace, L. Castoldi, A. D. Mamuye and W. Holzer, Synthesis, 2014, 2897 CrossRefNotably, in case of adding LiCH2F to isothiocyanates, two consecutive homologations take place, giving iminothietanes, see: R. Senatore, M. Malik, T. Langer, W. Holzer and V. Pace, Angew. Chem., Int. Ed., 2021, 60, 24854 Search PubMed.
- (a) M. Colella, P. Musci, M. Andresini, M. Spennacchio, L. Degennaro and R. Luisi, Org. Biomol. Chem., 2022, 20, 4669 Search PubMed; (b) R. Senatore, M. Malik and V. Pace, Adv. Synth. Catal., 2022, 364, 2890 CrossRef CAS.
- G. Parisi, M. Colella, S. Monticelli, G. Romanazzi, W. Holzer, T. Langer, L. Degennaro, V. Pace and R. Luisi, J. Am. Chem. Soc., 2017, 139, 13648 CrossRef CAS PubMed.
- E. Delebecq, J.-P. Pascault, B. Boutevin and F. Ganachaud, Chem. Rev., 2013, 113, 80 CrossRef CAS PubMed.
- P. Musci, M. Colella, A. Sivo, G. Romanazzi, R. Luisi and L. Degennaro, Org. Lett., 2020, 22, 3623 Search PubMed.
- P. D. Bailey, A. N. Boa and J. Clayson, Tetrahedron, 2009, 65, 1724 Search PubMed.
- M. Colella, A. Tota, A. Großjohann, C. Carlucci, A. Aramini, N. S. Sheikh, L. Degennaro and R. Luisi, Chem. Commun., 2019, 55, 8430 RSC.
- B. M. Jacobson and P. D. Bartlett, J. Org. Chem., 1973, 38, 1030 CrossRef CAS . An alternative pathway leading to the formation of biuret products might consist in the reaction of the urea conjugate base directely with unreacted arylisocyanate. Likely, it would be more kinetically viable at low temperatures (−78 °C).
- Y.-R. Luo, Comprehensive Handbook of Chemical Bond Energies, CRC Press, Boca Raton, 2007 Search PubMed.
- (a) S. Perveen, S. M. Abdul Hai, R. A. Khan, K. M. Khan, N. Afza and T. B. Sarfaraz, Synth. Commun., 2005, 35, 1663 Search PubMed; (b) A. J. South, A. M. Geer, L. J. Taylor, H. R. Sharpe, W. Lewis, A. J. Blake and D. L. Kays, Organometallics, 2019, 38, 4115 CrossRef CAS; (c) A. Dasgupta, Y. van Ingen, M. G. Guerzoni, K. Farshadfar, J. M. Rawson, E. Richards, A. Ariafard and R. L. Melen, Chem. – Eur. J., 2022, 28, e202201422 CrossRef CAS PubMed; (d) H. Yang, D. Huang, K.-H. Wang, C. Xu, T. Niu and Y. Hu, Tetrahedron, 2013, 69, 2588 CrossRef CAS.
Footnote |
| † This work is dedicated to Professor Valeria Conte (University of Rome “Tor Vergata”) in the occasion of her retirement. |
| This journal is © The Royal Society of Chemistry 2026 |